Despite ongoing improvements in approaches to cancer
detection and treatment, cancer continues to pose a major public
health burden worldwide. For advanced malignancies, comprehensive
treatment strategies commonly include surgical resection,
chemotherapy, radiotherapy (RT) and combinations of these
modalities. As a cornerstone of oncologic treatment (1), RT has historically been effective
for managing advanced tumors (2-4).
Approximately 50 to 60% of patients with cancer receive RT for
primary or metastatic lesions, either as a standalone modality or
in combination with surgery and chemotherapy (5,6).
RT encompasses a broad range of regimens that can be
classified by fractionation, including conventional,
hypofractionation and hyperfractionation, and by delivery
technique, including three-dimensional (3D) conformal radiotherapy,
intensity-modulated radiotherapy (IMRT) and stereotactic body
radiotherapy (SBRT). The use of stereotactic RT has expanded
rapidly because it can deliver ablative doses to the target with
high precision, while limiting exposure of adjacent normal tissues
(7,8). Ionizing radiation directly damages
biomolecules such as DNA and indirectly generates reactive oxygen
species (ROS) via water radiolysis, thereby triggering oxidative
stress (9-11). This process results in DNA
lesions, such as double-strand breaks, and induces multiple forms
of cell death, including apoptosis, autophagy and necrosis,
ultimately eliminating cancer cells (12). The selection of definitive or
adjuvant RT depends on factors including tumor radiosensitivity.
Indications have broadened with high-precision fractionated
techniques such as stereotactic RT. For example, current guidelines
recommend stereotactic approaches in selected hepatocellular
carcinoma and in oligometastatic disease (8). However, RT efficacy is constrained
by multiple factors, among which the tumor microenvironment (TME)
is a key determinant.
The TME refers to the complex ecosystem surrounding
malignant cells, including immune cells, blood vessels,
extracellular matrix components and soluble mediators such as
cytokines. These components influence tumor evolution and treatment
responsiveness, creating a permissive niche that supports cancer
cell survival and proliferation (13,14). Cytokines are key mediators
through which immune cells coordinate immune responses, and major
classes include interleukins, interferons, members of the TNF
superfamily, chemokines and growth factors (15-19). More than 100 cytokines have been
identified, and their signaling cascades and biological functions
have been characterized in detail (16,20-26). Cytokines act through autocrine,
paracrine and endocrine modes and influence diverse cell
populations in the TME, thereby shaping clinical outcomes in cancer
(27). Accumulating evidence
supports the therapeutic value of cytokine administration and of
agents that block cytokine signaling. For instance,
granulocyte-macrophage colony-stimulating factor (GM-CSF) promotes
antitumor immunity via dendritic cell activation and has been used
in approaches including GM-CSF gene-transduced tumor cell vaccines
(28-31), sipuleucel-T (32) and the oncolytic virotherapy
Talimogene laherparepvec (33).
In addition, neutralizing antibodies and small molecule inhibitors
directed against cytokines or their receptors have demonstrated
activity in cancer and in immune-mediated diseases (34). Recent translational studies have
increasingly focused on integrating RT with cytokine-directed
interventions. For example, the colony stimulating factor 1
receptor (CSF1R) inhibitor PLX3397 combined with RT antagonizes
CSF1R signaling and can deplete irradiation-recruited M2-type
tumor-associated macrophages (TAMs), thereby mitigating
RT-associated immunosuppression (35-37). Conversely, RT combined with G-CSF
or GM-CSF can enhance neutrophil-mediated antitumor functions by
increasing ROS generation and promoting cytotoxic T-cell
activation, which can improve local tumor control and induce
abscopal responses (38). In
addition, a mitochondria-targeted radionuclide,
223Ra-TPP, has been engineered to trigger mitochondrial
DNA damage and engage the cyclic GMP-AMP synthase (cGAS)/stimulator
of interferon genes (STING)/IL-1β axis, thereby amplifying systemic
immunity (39). These findings
indicate that defining key cytokine networks within the TME is
important for the rational design of immunotherapy strategies.
Tumor cells evade immune surveillance by
establishing an immunosuppressive microenvironment, a hallmark of
cancer. This state is maintained by immunosuppressive cells such as
TAMs and regulatory T (Treg) cells together with specific cytokines
(40,41). RT exerts complex and frequently
bidirectional effects on the TME. On the one hand, RT can stimulate
antitumor immunity and can function as an in situ
vaccination approach. Irradiation triggers immune-activating
events, including damage-associated molecular pattern release,
pro-inflammatory cytokine production and engagement of the
cGAS/STING axis (42).
Radiation-induced immunogenic cell death recruits immune cells and
promotes dendritic cell antigen presentation through
damage-associated molecular patterns and activation of the
cGAS-STING pathway and pattern recognition receptors, ultimately
activating CD8+ T cells. Higher intratumoral
CD8+ T-cell density is associated with favorable
outcomes after RT (40,43-48). RT can also reprogram neutrophils
toward an antitumor phenotype. These RT-activated neutrophils
produce increased ROS, directly kill tumor cells and secrete
pro-inflammatory factors such as TNF-α and interferon (IFN)-γ to
promote systemic immunity (38).
Administration of G-CSF further increases the number of RT-induced
antitumor neutrophils and enhances ROS production, accompanied by
upregulation of the antitumor N1 marker ICAM-1 (38,49). On the other hand, RT can induce
immunosuppression. For example, irradiation-induced increases in
ROS and hypoxia-inducible factor (HIF)-1α can promote the release
of TGF-β and C-X-C motif chemokine ligand (CXCL)12, driving the
expansion of Treg cells, myeloid-derived suppressor cells (MDSCs)
and cancer-associated fibroblasts (CAFs), enhancing M2 polarization
and increasing immunosuppressive cytokine production. In parallel,
immune checkpoint pathways, including programmed cell death 1
(PD-1) and cytotoxic T-lymphocyte associated protein (CTLA)-4 can
be upregulated, further constraining antitumor immune responses
(50-53). RT can also promote tumor cell
secretion of M-CSF via the NF-κB-p65 pathway, inducing TAM
polarization toward an M2 phenotype and increasing secretion of
IL-10 and Arginase 1 (54).
Furthermore, irradiated tumor cells may release exosomes enriched
in microRNA (miR)-21 that are transferred to TAMs via the CCL2 and
CCL3 axis, contributing to an immunosuppressive milieu (35). This remodeling may partially
explain why, in locally advanced non-small cell lung cancer, adding
programmed cell death ligand (PD-L1) blockade to standard chemoRT
improves outcomes, while >50% of patients later develop
progressive disease (55).
Traditional RT research has largely focused on
direct cytotoxic effects, with less emphasis on how RT regulates
the tumor immune microenvironment and systemic immunity. Beyond
inducing lethal DNA damage, RT promotes intercellular communication
by generating mediators such as ROS. ROS can function as
ligand-like cues that activate cell-surface receptors, thereby
initiating apoptosis or senescence in a stress-dependent manner
(56), promoting proliferation
(12,57), and orchestrating immune signaling
through the induction of pro-inflammatory cytokines, including IL-1
and TNF (40). Numerous
preclinical studies have established that host immune competence is
a key determinant of RT efficacy (58-60). These observations support a
conceptual shift in which RT is viewed not only as a cytotoxic
modality but also as a driver of cytokine network remodeling within
the TME. Ionizing radiation can influence therapeutic success by
initiating immune signaling cascades, positioning RT as a core
component of radioimmunotherapy (61).
Different RT modalities can regulate cytokine
programs in distinct ways. For example, ultra-high dose-rate Fast
Linear Accelerator-based Scanning Hybrid (FLASH) RT reduces the
release of profibrotic factors such as TGF-β and IL-1β, thereby
alleviating radiation-induced lung injury, while promoting
CD4+ T-helper-cell secretion of tissue-reparative
cytokines, including IL-10 and IL-22 (62,63). By contrast, proton therapy
leverages the Bragg peak to better spare circulating lymphocytes
and, when combined with immunotherapy, increases the frequency of
intratumoral IFN-γ-positive T cells, while suppressing TGF-β
signaling (64). These advances
provide a rationale for RT selection and customization in
immunotherapy-compatible treatment designs.
Building on this background, the present study
proposes a multi-layered framework integrating five dimensions: RT
parameters, key signaling axes, effector cells, organ-specific
contexts and clinical translation. First, the dynamic roles of
major cytokine families in the post-irradiation tumor immune
microenvironment were summarized, including IFN-I/III, chemokines
and key mediators, such as IL-1, IL-6, IL-10 and TGF-β. The focus
is on antagonistic and cooperative networks that balance immune
activation and immunosuppression. Second, focusing on macrophages
as a key effector cell population, their hub function in RT
responses through CSF1R and CCR2 signaling and downstream metabolic
reprogramming were examined, and mechanisms of polarization control
and paracrine circuits were summarized. Third, the analysis was
extended to organ-specific radiation injury by summarizing
tissue-characteristic cytokine signatures and candidate
interventional targets. Glial-cell activation mediated by IL-1β,
IL-6 and TNF-α in radiation-induced brain injury (RBI) was
analyzed. The review then delineated the development of pulmonary
radiation fibrosis dominated by TGF-β and IL-13. Mucosal barrier
dysfunction linked to IL-33 and TSLP in gastrointestinal injury was
then discussed. Subsequently, the roles of IL-1 and TNF in the
inflammatory cascade of oral mucositis were summarized. The
coupling between IL-6/STAT3 axis-driven regenerative repair and
TGF-β-mediated fibrosis in liver injury was also described.
Finally, from a translational perspective, key parameters in
combined RT and immunotherapy strategies were evaluated, including
fractionation selection, immunotherapy sequencing, candidate
synergistic dose windows and predictive biomarkers. This synthesis
is intended to support individualized evidence-based integration of
RT with immunotherapy (Fig.
1).
IFN-I represents the largest IFN family and includes
IFN-α, IFN-β, IFN-ε and IFN-ω, which signal through the type I IFN
receptor (IFNAR) (65). Burnette
et al (66) showed that
RT-mediated tumor control is lost in IFNAR-deficient mice,
indicating that host IFN-I signaling is required for RT-induced
antitumor immunity. The magnitude and duration of IFN-I signaling
can influence RT efficacy (66,67). IFN-I pathways can therefore exert
both promotive and inhibitory effects on RT responses through
immune mechanisms.
Augmenting local IFN-I signaling in the TME can
counteract immunosuppression and promote antitumor immunity.
Irradiation can induce tumor-infiltrating myeloid cells to produce
IFN-α and IFN-β through autocrine signaling. These IFN-I signals
propagate within the hematopoietic compartment and enable
tumor-infiltrating dendritic cells to cross-prime CD8+ T
cells, supporting adaptive immune attack on the tumor (66). This response is coupled to a
cytosolic DNA sensing cascade involving cGAS, STING and IFN
regulatory factor 3 (IRF3) (67). In this cascade, cGAS functions as
a cytosolic DNA sensor that allows dendritic cells to detect DNA
derived from irradiated tumors and represents a key node through
which RT initiates antitumor immunity (68). Ionizing radiation also induces
DNA strand breaks and micronuclei formation, and micronuclear DNA
can activate STING and TANK-binding kinase 1 (TBK1), thereby
stimulating IFN production (69,70). The resulting IFN release promotes
dendritic cell maturation and major histocompatibility complex
class I (MHC-I) upregulation, enhances tumor antigen
cross-presentation and elicits antigen-specific cytotoxic
T-lymphocyte responses that mediate antitumor efficacy (66,71,72).
Poly (ADP-ribose) polymerase (PARP) inhibition can
lead to cytosolic double stranded DNA accumulation, engage the
cGAS/STING/TBK1/IRF3 axis and induce IFN-I production and
associated immune programs (67,73). In triple-negative breast cancer
models, olaparib activates cGAS-STING signaling and drives mediator
release that activates dendritic cells, resulting in increased
CD8+ T-cell infiltration and activation (74). These findings suggest that PARP
inhibitors can cooperate with RT to engage innate and adaptive
immunity through a double-stranded DNA-driven cGAS/STING/IRF3/IFN-I
signaling pathway. Activation of the cGAS-STING pathway is often
accompanied by PD-L1 upregulation, which has been linked to IFN-I
activity (75,76). Accordingly, STING agonists have
been proposed as candidate sensitizers to PD-1 or PD-L1 checkpoint
blockade (77). Beyond IRF3,
STING can also engage IκB kinase and NF-κB-inducing kinase to
activate NF-κB, supporting antitumor effects across tumor
initiation, progression and metastasis (77-80).
IFN-I signaling is also constrained by negative
feedback circuits. Irradiation-induced STING and IFN-I signaling in
dendritic cells can activate STAT2 and increase expression of the
m6A reader YTH domain-containing family protein 1 (YTHDF1). YTHDF1
promotes translation of cathepsin A and cathepsin B mRNAs,
increases lysosomal protease abundance and accelerates lysosomal
STING degradation, thereby reducing IFN-I production and limiting
dendritic cell antitumor activity (81). In addition, neoadjuvant RT
studies in rectal cancer have identified an inflammatory CAF subset
characterized by high IRF1 expression. This subset is polarized by
IFN-γ signaling and recruits T cells and dendritic cells through
secretion of CCL4 and CCL5, thereby augmenting antitumor immunity
(82).
By contrast, intratumoral IFN-I activation has been
associated with unfavorable outcomes and resistance to several
treatment modalities including RT (83-86). In settings combining RT with
CTLA-4 or PD-L1 checkpoint blockade, IFN signaling can contribute
to treatment resistance (87).
Chen et al (88) reported
that genetic ablation of Ifnar1 enhances CD8+ T
cell-dependent antitumor responses after RT, and they identified
serine protease inhibitor b9 as a critical factor that is reduced
in Ifnar1-deficient tumor cells.
CC chemokines comprise a chemokine subfamily defined
by an N-terminal C-C motif (89). CCL2, also known as monocyte
chemoattractant protein 1 (MCP-1), can bind multiple receptors, and
RT can induce CCL2 expression (90,91). Within radiation-induced immune
responses, CCL2 exerts pleiotropic effects. It can act downstream
of IL-6 to mediate macrophage infiltration into tumors after
irradiation (92). RT can
activate STING signaling and increase IFN-I production, which can
induce CCL2, CCL7 and CCL12 expression and drive monocyte
trafficking into tumors (93).
In murine head and neck squamous cell carcinoma, a 7.5-Gy dose
increased CCL2 and promoted infiltration of TNFα-producing
monocytes and CCR2+ Treg cells. Monocyte-derived TNFα
activated Treg cells and could undermine RT responses (94). Tumor-derived CCL2 has also been
implicated in adaptive radioresistance in pancreatic ductal
adenocarcinoma models, in which CCL2 blockade improved the efficacy
of RT (95). Upregulation of
CCL2 has been linked to radiation toxicities, including
radiation-induced lung injury, radiation-induced liver injury and
cognitive impairment after RBI (96-98). CCL3, also known as macrophage
inflammatory protein 1α, contributes to RT responses. Through CCR1
engagement, CCL3 promotes type 2 T-helper (Th2)-cell infiltration
and exacerbates radiation-associated lung injury and fibrotic
remodeling. In hepatocellular carcinoma, combining CCL3 with RT
enhanced CD8+ T cell-driven antitumor immunity, and CCL3
has also been evaluated as a candidate predictor of breast cancer
RT response (99). CCL5, also
known as regulated on activation, normal T cell expressed and
secreted, can exert context-dependent effects. RT induces tumor
cells and mesenchymal stromal cells to release CCL5, which recruits
macrophages and can promote M2 polarization and metastatic
progression (100,101). By contrast, CCL5 can also
recruit CD8+ T cells and enhance antitumor immunity,
indicating that its net effect depends on tumor context and
radiation dose (102). CCL8,
also known as MCP2, binds multiple receptors, including CCR1, CCR2,
CCR3 and CCR5, and mainly recruits monocytes and Treg cells.
Thoracic irradiation elevates CCL8, enhances macrophage
infiltration and has been associated with increased pulmonary
metastasis in a preclinical study (103). CCL11, also known as eotaxin 1,
recruits eosinophils through CCR3. Radiation-associated increases
in CCL11 have been linked to injury across multiple tissues. In
intestinal radiation fibrosis, irradiation drives mucosal
myofibroblasts to secrete CCL11, which recruits eosinophils and
accelerates fibrotic progression (104). After radiation injury to skin
and brain tissue, increased CCL11 can also promote migration of
eosinophils and other immune cells to damaged sites, aggravating
inflammation and tissue injury (105,106). CCL7 has been implicated in
radiation-induced lung fibrosis. In a radiation-sensitive murine
model, CCL7 was found to be markedly induced and was able to
promote lung fibrosis by recruiting profibrotic immune populations
(107). CCL22 is constitutively
secreted by specific dendritic cell subsets in secondary lymphoid
organs, particularly the CD103+ CD11b−
CD8+ subset. By binding CCR4 on Treg cells, CCL22
promotes direct dendritic cell- and Treg-cell contact that
suppresses effector T-cell activation and proliferation. Loss of
CCL22 enhances vaccine-induced antigen-specific CD8+
T-cell responses and antitumor immunity (108).
Overall, CC chemokines function as regulators of the
RT-conditioned TME by coordinating monocyte influx, macrophage
polarization, trafficking of T cells and Treg cells, and
eosinophil-associated remodeling. Table I summarizes their reported net
effects on antitumor immunity, radioresistance and RT-associated
toxicity. Conversely, pro-immunogenic chemokines such as XCL1 and
CXCL16 can recruit cross-presenting dendritic cells and effector
CD8+ T cells to strengthen antitumor immunity (109,110), whereas axes such as
CXCL12/CXCR4 and CXCL8/CXCR1/2 promote vasculogenesis, DNA damage
repair and survival signaling that support recurrence and
radioresistance (111-113). More broadly,
radiation-responsive chemokine programs are emerging as important
determinants of immune-cell trafficking and treatment outcome; a
recent review summarized the central role of CC chemokines in
radiation responses, and experimental evidence in lung cancer
further showed that RT can enhance activation of CD8+ T
cells with high CXCR3 expression by inducing IFN-γ-mediated CXCL10
and ICAM-1 expression (114,115).
RT kills tumor cells by inducing DNA double-strand
breaks and simultaneously triggers dynamic changes in
pro-inflammatory and anti-inflammatory cytokine networks (Fig. 3). These changes can have opposing
consequences. Pro-inflammatory cytokines, including TNF-α, IL-6 and
IL-1β, can activate NF-κB, STAT3 and angiogenesis-related pathways,
thereby promoting tumor-cell proliferation and invasion and
contributing to radioresistance. For example, TNF-α overexpression
in non-small cell lung cancer is associated with reduced
radiosensitivity, and increased serum IL-1β, IL-6 and TNF-α in head
and neck squamous cell carcinoma is associated with worse outcomes
(116,117). RT-induced immune reprogramming
can also involve IL-6-dependent regulation. In nasopharyngeal
carcinoma, irradiation increases mTOR complex 1 activity and alters
p62 phosphorylation, which suppresses p62-dependent selective
autophagy and ultimately increases endothelial protein C receptor
(PROCR) expression. By promoting IL-6 secretion, PROCR suppresses
Th1 differentiation and compromises CD8+ T-cell effector
function, thereby attenuating antitumor immunity (118). IL-34 suppresses IL-12 in
post-irradiation TAMs, limiting recruitment and activation of
IFN-γ-producing CD8+ T cells and blunting RT-induced
antitumor immunity. In IL-34-deficient tumors, RT promotes
pro-inflammatory macrophage differentiation and IL-12 induction,
strengthening immune-mediated tumor eradication (119). Cytokines are also key mediators
of immunogenic cell death. Under immunogenic conditions, they can
drive dendritic cell maturation and antigen cross-presentation and
enhance CD8+ T-cell responses through NF-κB and IFN-I
pathways, contributing to abscopal effects. In breast cancer
models, combining irradiation with valproic acid reprograms the
irradiated field TME, increases CD8+ T-cell infiltration
and M1-like polarization and suppresses growth of distant lesions
(120,121). Anti-inflammatory mediators such
as IL-10 can mitigate normal tissue toxicity. For example,
low-level laser therapy can ameliorate oral mucositis in part by
reducing IL-6 (122). However,
excessive anti-inflammatory signaling can suppress immune
surveillance and is associated with reduced survival. For example,
in glioblastoma, dexamethasone-induced anti-inflammatory cascades
correlate with shorter survival (123). In specific contexts, such as
combination with γ irradiation, IL-10 upregulation has also been
reported to promote tumor-cell apoptosis, as observed with ebselen
combination therapy in breast cancer models (124). Radiation dose is a determinant
of the immune microenvironment. Low- and intermediate-dose RT tends
to favor M2-macrophage polarization and an immunosuppressive
microenvironment (125),
whereas high-dose RT combined with PD-L1 blockade can activate the
cGAS-STING pathway, amplify pro-inflammatory cytokine release and
enhance CD8+ T-cell responses, thereby increasing
abscopal effects (126). When
pro- and anti-inflammatory signals are imbalanced, TGF-β-driven
pathways may promote radiation fibrosis or radioresistance
(127), and excessive
anti-inflammatory signaling may weaken immune clearance and
increase the risk of recurrence (121). Clinically, modulation of
cytokine networks has been explored to remodel the TME. For
example, URB937-mediated suppression of IL-1β, IL-6 and TNF-α
mitigates radiation-induced lung injury (128), siltuximab improves RT responses
in multiple myeloma (129) and
CTLA-4 blockade can potentiate RT-induced abscopal effects
(130). To maximize antitumor
efficacy while minimizing adverse effects, real-time monitoring and
rational modulation of this network remain important (131).
Macrophages are key immune components of the TME and
contribute to tumor initiation, immune evasion and antitumor
immunity (Fig. 4) (132). RT can reshape the TME by
altering the polarization of TAMs (133). Different radiation doses can
promote transitions toward a pro-inflammatory M1 phenotype or an
anti-inflammatory M2 phenotype through NF-κB and downstream
cytokine signaling, resulting in dose-dependent immunomodulation
(125,134-137). Low-dose RT (<2 Gy) can
inhibit NF-κB p65 activity, reduce expression of pro-inflammatory
factors such as TNF-α and IL-1β, and increase TGF-β release,
thereby promoting an immunosuppressive M2 phenotype (125,134). Moderate-dose RT from 2 to 10 Gy
can activate NF-κB p65 p50 heterodimers, increase the secretion of
TNF-α, IL-6 and IL-12, upregulate M1 markers including CD80, CD86
and MHC-II, and suppress M2 markers such as CD163 and IL-10,
thereby enhancing antitumor immune responses (135,136,138). By contrast, high-dose RT >10
Gy can increase the expression of chemokines such as CCL2, CCL5 and
CXCL12 through hypoxia-induced HIF-1α signaling, driving monocyte
differentiation toward M2-like TAMs. High-dose RT can also favor
NF-κB p50 homodimer activity, upregulate immunosuppressive factors
including IL-10 and TGF-β, and contribute to tumor immune escape
(111,139,140).
TAMs modulate tumor radiosensitivity by secreting
cytokines and chemokines. CCL2 and CXCL6 secreted by M2-like TAMs
can recruit MDSCs or activate EMT programs, thereby promoting
radioresistance (93,141-143). After irradiation, tumor-derived
exosomes can transfer miR-193b-3p and hsa_circ_0001610 to TAMs and
reduce radiosensitivity through MAPK kinase kinase 3 repression or
PD-L1 upregulation (144,145). High-dose RT can also induce
CAFs to secrete CCL2, which cooperates with TAM-derived VEGF and
heparin-binding EGF-like growth factor to promote angiogenesis and
DNA damage repair, thereby enhancing radioresistance (146-148). Conversely, M1-like TAMs secrete
TNF-α and IL-12, promote cytotoxic T-lymphocyte recruitment and
enhance antigen presentation, which can counteract
immunosuppression and improve radiosensitivity (149,150). In addition, RT-derived
microparticles can activate the JAK/STAT pathway, promote
polarization toward M1 macrophages and induce the release of danger
signals such as ATP and high mobility group box 1 (HMGB1), thereby
enhancing immunogenic cell death (151,152).
Given the role of TAMs in radiosensitivity,
targeting macrophage-associated cytokines has become an important
strategy to improve RT efficacy. Inhibition of CCL2/CCR2 or
CCL5/CCR5 signaling can limit macrophage infiltration and M2
polarization (153). Anti-CSF1R
antibodies can block monocyte to macrophage differentiation, and
combining these agents with RT can delay tumor recurrence (37,154). VEGF-neutralizing antibodies can
counteract macrophage-driven angiogenesis (147,155). Nanotechnology-based delivery
approaches, such as nanogels carrying Toll-like receptor (TLR)7 or
TLR8 agonists, can reprogram macrophages toward an M1 phenotype
(156-159). Iron oxide nanoparticles can act
as radiosensitizers by increasing ROS and promoting the secretion
of pro-inflammatory cytokines such as IL-1β (151,160). In addition, RT-derived
microparticles co-expressing tethered (t)IL-15 and tCCL19 combined
with PD-1 inhibitors can activate CD8+ T-cells and
cooperate with macrophages to enhance antitumor activity (161).
In summary, RT regulates TAM polarization through
cytokine networks, particularly NF-κB, CCL/CCR and CSF1R signaling,
which represent central determinants of tumor radiosensitivity.
Although cytokine-targeted approaches and nanotechnology-based
delivery strategies have shown efficacy in preclinical studies,
heterogeneity in dose effects, potential normal tissue toxicity and
immune feedback resistance mechanisms such as compensatory PD-L1
upregulation remain key challenges for clinical translation
(162). Future studies using
single-cell analyses and related technologies are needed to define
functional heterogeneity among macrophage subsets and to support
individualized strategies for combining RT with immunotherapy.
RBI is a common adverse effect of tumor RT and
effective treatments remain limited (163,164). Cellular senescence is
considered a major risk factor for RBI initiation and progression
(165-167). RT can induce pericyte
senescence and promote the secretion of senescence-associated
secretory phenotype factors, including IL-6, TNF-α, IL-1β and CCL2,
which can disrupt blood-brain barrier (BBB) integrity and impair
endothelial tight junctions in vitro (168). These factors can further impair
endothelial, vascular, glial and hippocampal neuronal function
through paracrine signaling (169-172). Radiation injury can also cause
autophagy defects and lysosomal dysfunction in perivascular cells,
leading to the accumulation of toxic proteins, accelerated
senescence and demyelination of microglia, neurons and
oligodendrocyte progenitor cells, thereby worsening cognitive
dysfunction (173). Based on
these mechanisms, activation of autophagy or elimination of
senescent cells has been explored as a strategy to mitigate RBI.
For example, rapamycin enhances lysosome-mediated protein
clearance, suppresses perivascular cell senescence and partially
restores proliferative capacity (174,175). Dasatinib plus quercetin and
all-trans retinoic acid can selectively eliminate senescent
pericytes and reduce senescence-associated secretory phenotype
secretion, thereby improving BBB integrity and cognitive function
(176-178). In models driven by microglial
inflammation and neuronal loss, pregabalin has shown
neuroprotective effects by blocking HMGB1-mediated TLR2, TLR4 and
RAGE/NF-κB signaling and by reducing the production of IL-6, IL-1β
and TNF-α (179).
In RT for high-grade brain tumors and metastases,
neuron-derived ectodysplasin A2 receptor (EDA2R) has been proposed
as a predictor of early responses, such as neurocognitive
impairment after cranial irradiation, although the mechanism
remains elusive (180). In
cachexia-associated muscle atrophy, oncostatin M upregulates EDA2R
expression through activation of the noncanonical NF-κB pathway
(181). Another report
suggested that after hypoxic injury, EDA2R can modulate
inflammatory mediators, including TNF-α and IL-1β (182). Based on these observations, a
plausible mechanism is that RT acutely activates multiple
immune-cell types, including microglia (183,184), and increases the secretion of
inflammatory mediators after the peak dose. Under these conditions,
oncostatin M may promote EDA2R upregulation through NF-κB signaling
and thereby modulate the local inflammatory microenvironment
(Fig. 5) (169,170,185,186). In addition, upregulation of
CCL8 in the hippocampus can mediate macrophage accumulation and
exacerbate neuroinflammation, contributing to cognitive impairment
after cranial irradiation (112).
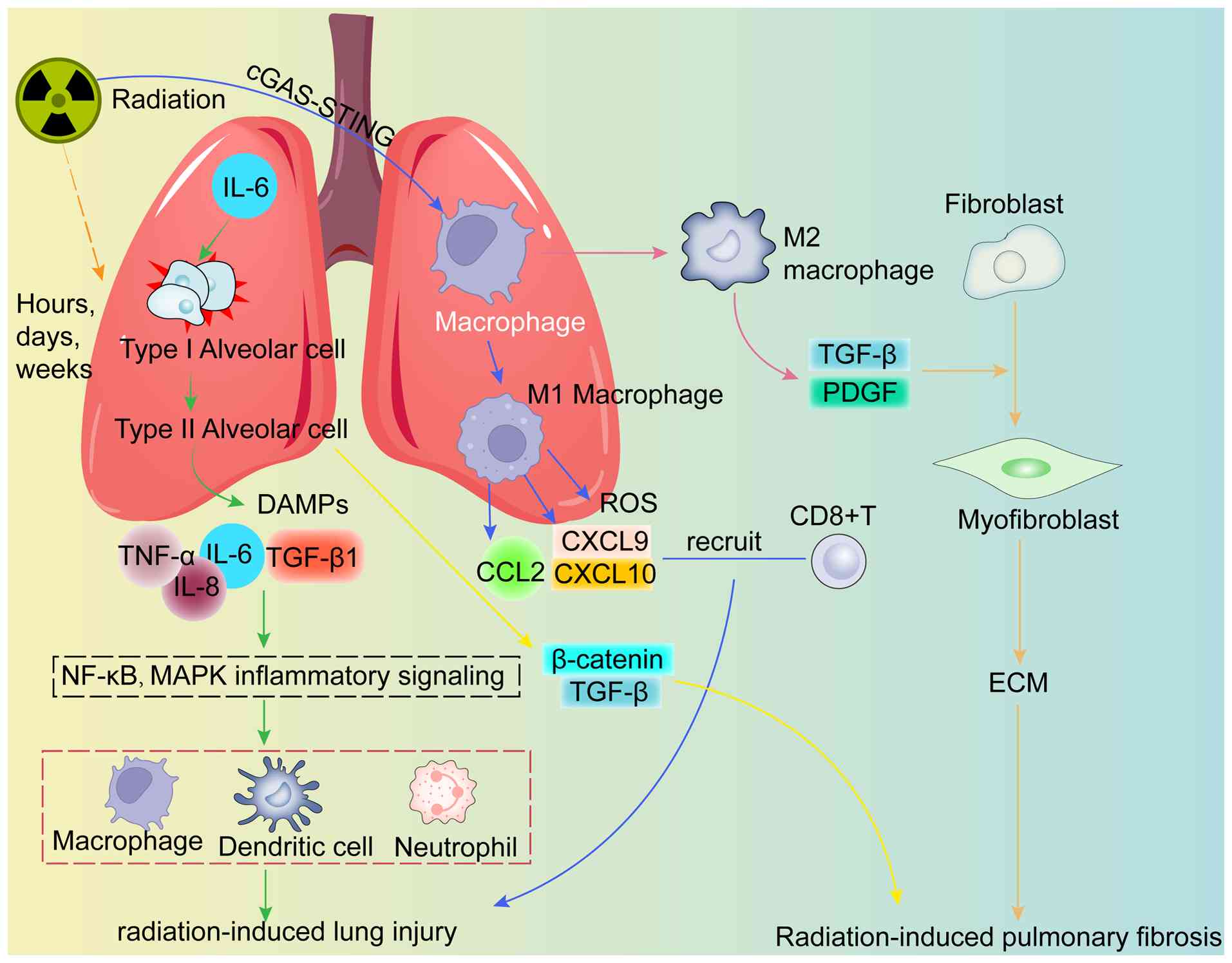
Radiation-induced lung injury arises from direct
irradiation-mediated cytotoxicity in normal lung tissue and the
subsequent inflammatory and fibrotic responses (Fig. 6) (187). Sustained post-irradiation
increases in cytokines such as IL-6 can damage type I alveolar
epithelial cells (AECI) and stimulate proliferation of AECII
(188). Subsequently, at ~6 to
8 weeks after irradiation, a second wave of cytokines including
TNF-α can contribute to acute pneumonitis (189). Ionizing radiation damages
alveolar epithelial cells and vascular endothelial cells, releasing
damage-associated molecular patterns (DAMPs) and cytokines
including TNF-α, IL-1β, IL-6, IL-8 and TGF-β1. These mediators
activate the NF-κB and MAPK pathways, promote the recruitment of
macrophages, neutrophils and dendritic cells, and amplify local
inflammation (190-192). In parallel, injured AECII show
reduced surfactant production, impaired tissue homeostasis and
activation of EMT via TGF-β1 and β-catenin signaling, promoting
fibrotic progression (193).
Radiation can also polarize lung resident macrophages toward an M1
phenotype, leading to the production of ROS and chemokines such as
CCL2, CXCL9 and CXCL10. This process promotes continued
CD8+ T-cell infiltration and exacerbates tissue injury
(133,194). In the late phase,
radiation-activated cGAS/STING signaling can promote macrophage
polarization toward an M2 phenotype through the regulation of CCL2
and can increase the secretion of profibrotic factors such as TGF-β
and platelet-derived growth factor (PDGF). These changes induce
fibroblast to myofibroblast differentiation, increase extracellular
matrix deposition and contribute to radiation-induced pulmonary
fibrosis (195-197). Macrophage-derived CCL22 is
selectively upregulated in rat models of radiation pneumonitis
(198). Targeting mediators
such as TGF-β, alone or in combination, and strategies that target
CCL22 through the TNF-related apoptosis-inducing ligand pathway
have been proposed for severe radiation lung toxicity (199). A previous study also indicated
that irradiation-activated bronchial club cells can modulate the
local immune microenvironment by secreting club cell secretory
protein, suppressing MDSCs and enhancing T-cell function,
suggesting that distinct pulmonary cell types contribute to
immunoregulation in lung injury and fibrosis (200). In addition, chemokines
including CCL5, CCL8 and CCL3 have been linked to the progression
of radiation-induced lung injury.
To reduce the risk of radiation-induced lung injury,
clinical management should integrate advanced planning and delivery
techniques such as IMRT and SBRT with evidence-based supportive
measures, including anti-inflammatory agents, immunomodulatory
strategies, and, in selected contexts, mesenchymal stromal
cell-based interventions, to control radiation-induced inflammation
and limit fibrotic progression. In parallel, the development of
therapeutics that target key cytokine pathways implicated in
pneumonitis and fibrosis remains an important research direction
for the prevention and treatment of radiation-induced lung
injury.
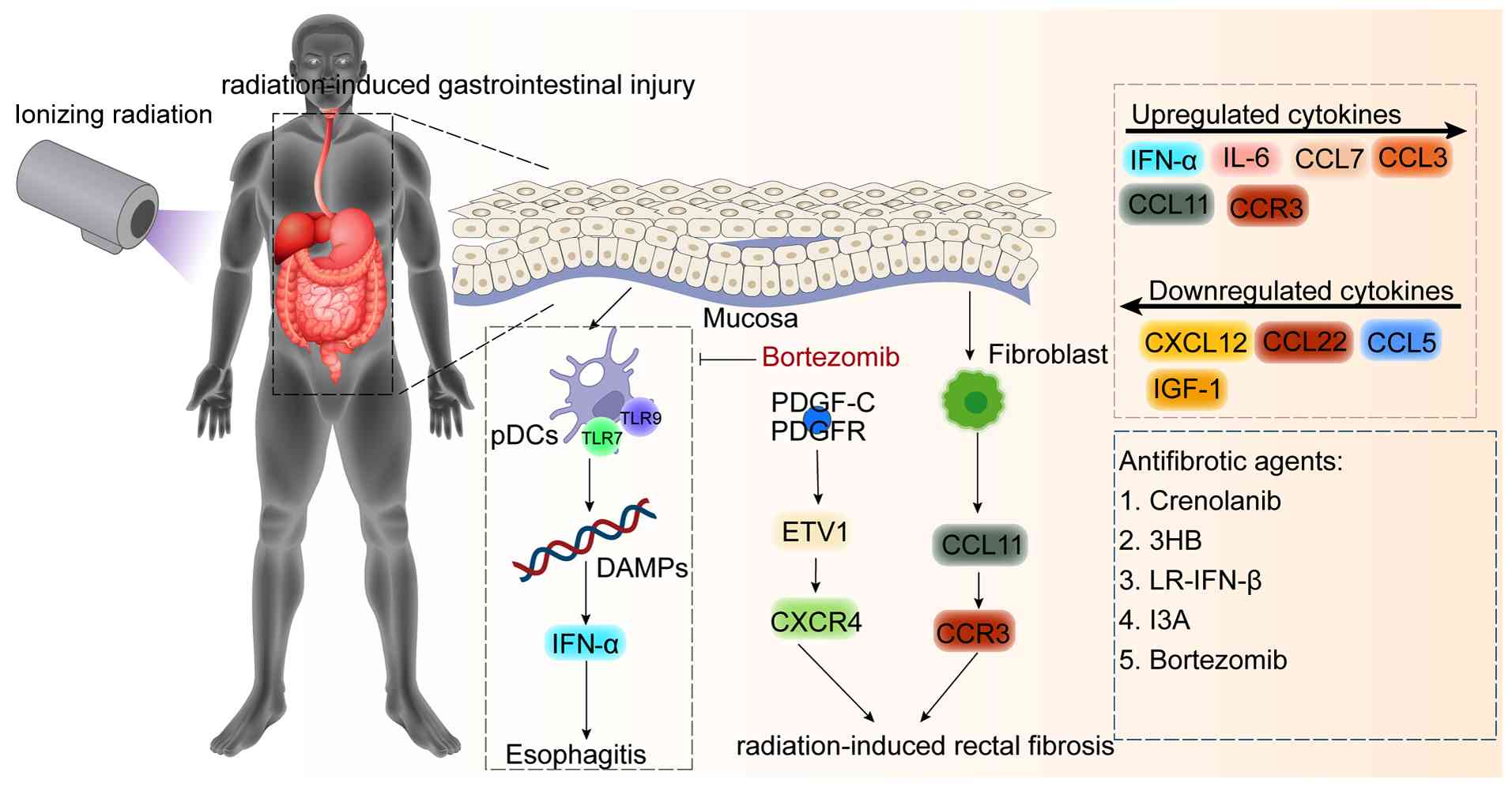
Radiation-induced gastrointestinal injury is a
common complication of cancer RT. Radiation esophagitis is a major
dose-limiting toxicity in RT for lung cancer and head and neck
squamous cell carcinoma (Fig.
7). Among patients with non-small cell lung cancer receiving
concurrent chemoRT, the incidence can reach 95 and 33% experience
grade 3 or higher events. Similar rates have been reported for 3D
conformal RT, IMRT and proton beam therapy (201-203). Evidence suggests that IFN-α
acts as a pro-inflammatory mediator in radiation esophagitis
(204). Plasmacytoid dendritic
cells (pDCs) in the esophageal mucosa can recognize endogenous RNA
and DNA released as damage-associated molecular patterns released
after tissue injury via TLR7 and TLR9, producing large amounts of
IFN-α and serving as a major source of IFN-I in radiation
esophagitis (205-207). Depletion of pDCs using
anti-CD317 antibodies or inhibition of pDCs function with
bortezomib can suppress IFN-α upregulation and alleviate mucosal
inflammation and tissue injury. These findings support a
pro-inflammatory plasmacytoid dendritic cell IFN-α pathway and
provide a rationale for targeted intervention. Beyond IFN-α,
upregulation of IL-16, CCL3 and CCL7 is associated with increased
esophagitis severity. By contrast, upregulation of CCL5, CXCL12,
CCL22 and IGF-1 is consistent with trends toward injury repair
(108).
Radiation-induced rectal injury refers to rectal
damage caused by RT for pelvic malignancies and is commonly
categorized as acute or chronic, with 3 months used as a practical
cutoff (208). Preclinical
studies indicate that PDGF-C, through engagement of PDGFR and
activation of a downstream ETS translocation variant 1-mediated
CXCR4 signaling axis, promotes colorectal inflammation and
fibrosis. Accordingly, the PDGFR antagonist crenolanib has been
proposed as a candidate agent for the prevention and treatment of
radiation-induced rectal lesions (209). During pelvic RT, depletion of
Akkermansia reduces concentrations of its metabolite
3-hydroxybutyrate in the gut and circulation, limiting activation
of the intestinal cell surface receptor G-protein-coupled receptor
43. This change relieves the suppression of IL-6, thereby driving
intestinal inflammation and tissue injury (210). In addition, irradiation induces
intestinal mucosal myofibroblasts to release CCL11, which recruits
eosinophils via CCR3 and exacerbates fibrosis (104). Hypoxia can also activate VEGF
and TGF-β pathways, promoting inflammation and fibrosis and
accelerating late-stage rectal injury (211,212). Several cytokines also represent
potential therapeutic targets. For example, CCL2 has been proposed
as a biomarker of late rectal toxicity after RT in prostate cancer,
and early assessment and intervention may reduce risk (213). LR-IFN-β released by the
probiotic Lactobacillus reuteri can alleviate
gastrointestinal acute radiation syndrome after total abdominal
irradiation (214). In
addition, the microbial metabolite indole-3-carboxaldehyde can
protect the intestine from radiation injury by activating Aryl
hydrocarbon receptor/IL-10/wingless-related integration site 3
signaling and by increasing the abundance of probiotic bacteria
(215).
Radiation-induced oral mucositis is a common
toxicity of RT for head and neck tumors, with an incidence ranging
from 26.4 to 100% (216,217).
Clinically, oral mucositis presents with erythema and ulceration in
the acute stage and can evolve into persistent chronic injury,
substantially compromising quality of life (218,219). Oral mucositis development is
closely linked to cytokine network dynamics (Fig. 8). RT damages mucosal cell DNA and
generates ROS, triggering the release of DAMPs (220). These signals activate TLR,
NF-κB and MAPK pathways, drive the secretion of pro-inflammatory
cytokines including TNF-α, IL-1β and IL-6, and increase apoptosis
and inflammation (218,220,221). Oral dysbiosis can further
enhance DAMPs signaling, sustaining epithelial NF-κB activation and
cytokine production (222,223). During progression, M1
macrophages accumulate in the submucosa and secrete TNF-α and
IL-1β, amplifying inflammation (224). CD4+ T-cells
influence the disease course through modulation of the balance
among Th1, Th17 and Treg-cell subsets (225,226). In the recovery phase, M2
macrophages predominate and support tissue repair (227). Based on these mechanisms,
multiple interventions have been evaluated. Melatonin reduces
pro-inflammatory cytokine expression by scavenging ROS and
inhibiting NF-κB signaling. Mouthwash and gel formulations have
reduced the incidence of severe mucositis by a ~34% in clinical
studies (228). Chlorhexidine
mucosal patches reduce pro-inflammatory cytokine levels through
activation of macrophage α2 receptors, and a phase II trial
reported a reduced incidence of severe mucositis (229,230). Pentoxifylline inhibits TNF-α
production and, when combined with vitamin E, can shorten mucositis
duration (231,232). Recombinant human IL-11
(rhIL-11) has been evaluated to reduce ulcer severity and
accelerate healing by promoting mucosal repair (233,234). Curcumin has also been reported
to reduce pain and clinical severity through inhibition of TNF-α
(233-237). Photobiomodulation can
accelerate mucosal repair by modulating cytokine activity and
promoting angiogenesis (238,239) and has been recommended for
prevention by the Multinational Association of Supportive Care in
Cancer and International Society of Oral Oncology guidelines with
level I to II evidence (240,241). Future work should evaluate
cost-effectiveness in larger clinical trials and integrate virtual
screening to develop specific cytokine inhibitors for prevention
and treatment (242,243).
Radiation-induced liver disease (RILD) is a serious
complication of RT for upper abdominal and thoracic tumors and can
limit the broader use of RT (244,245). RT can increase inflammatory
cytokine levels and promote the infiltration of immune cells,
leading to tissue fibrosis and hepatic dysfunction (Fig. 9) (246). However, immunoregulatory
mechanisms in RILD remain incompletely defined. Pyroptosis is an
inflammatory form of programmed cell death mediated by gasdermin D
(GSDMD) that can reshape the immune microenvironment through the
release of intracellular contents and cytokines (247-250). In a murine RILD model, RT
increases hepatocellular expression of full-length GSDMD (GSDMD-FL)
and its N-terminal fragment (GSDMD-N). This process promotes CXCL1
transcription through activation of STAT5A, while GSDMD-N pore
formation facilitates CXCL1 release. The resulting CXCL1 signaling
recruits neutrophils and exacerbates liver injury. Genetic deletion
or pharmacologic inhibition of GSDMD and neutralization of CXCL1
can alleviate radiation-induced liver injury (251). Accordingly, pharmacologic
approaches such as disulfiram that suppress both GSDMD-FL and
GSDMD-N have been proposed as targeted strategies to prevent RILD
(252-255).
RILD pathogenesis also involves complex cytokine
network regulation. Ionizing radiation activates the DNA damage
response through direct DNA damage and ROS bursts, initiating ATM
and ATR signaling and MAPK cascades, including ERK/JNK/p38. These
events promote autocrine growth factor release, including TGF-α and
TNF-α, radiation-induced bystander effects and inflammatory
responses (256-259). Hepatic non-parenchymal cells
act as key effector populations. After irradiation, Kupffer cells
secrete TNF-α, which drives inflammatory monocyte infiltration
through TLR4/NF-κB signaling, inducing hepatocyte apoptosis and
secondary injury (260-264). In parallel, sinusoidal
endothelial cells undergo apoptosis and lose fenestrae, causing
microcirculatory disturbance and promoting the secretion of
fibronectin EIIIA, which activates hepatic stellate cells (265,266). Activated hepatic stellate cells
respond to TGF-β1 signaling by increasing collagen synthesis
through the TGF-β1/Smad/connective tissue growth factor axis and by
suppressing extracellular matrix degradation, resulting in
radiation-induced liver fibrosis (267-271). Radiation-induced senescent
cells can also secrete pro-inflammatory factors such as IL-6
through the SASP, further worsening the microenvironment (272,273). Translational studies have
focused on targeting cytokine pathways implicated in these
processes. In animal studies, inhibiting Kupffer-cell function
reduces TNF-α release and alleviates sinusoidal endothelial cell
apoptosis and acute liver injury (260,274). Approaches targeting TGF-β1,
including antisense oligonucleotides and small-molecule inhibitors
such as pirfenidone, can suppress fibrotic signaling and improve
radiation-induced liver fibrosis (271,275). Natural antioxidants, including
curcumin and resveratrol, can modulate pathways such as nuclear
factor erythroid 2-related factor 2/Kelch-like ECH-associated
protein 1 and miR-34a-sirtuin 1 to suppress ROS/TNF-α/NF-κB-driven
inflammatory axes, thereby reducing oxidative stress and fibrotic
progression (276-279). Mesenchymal stem cell therapy
can inhibit TGF-β/Smad signaling and promote tissue repair through
the secretion of anti-inflammatory mediators such as hepatocyte
growth factor (266,280). However, targeted therapeutics
remain limited. Future work should integrate precision RT
approaches such as stereotactic RT and explore combinations with
immune checkpoint inhibitors to balance efficacy and hepatotoxicity
(281).
Given the role of IFN-I in RT-induced innate and
adaptive immunity, IFN-I has been explored as a therapeutic lever
in cancer (282). Early studies
showed that IFN-γ+CD8+ T-cells can increase
radiosensitivity in hypoxic tumors (283). RT-induced IFN-β can remodel the
antitumor immune microenvironment and has been associated with
improved disease-free survival in lung cancer (284). Preclinical murine tumor studies
indicated that loss of IFN signaling in the host or tumor after RT,
with or without immune checkpoint blockade, weakens local and
systemic immune responses (66,67,285). By contrast, a study in a murine
tumor model suggested that persistent IFN signaling can contribute
to resistance to immune checkpoint therapy (87). Tumor cells with high Argonaute 2
(AGO2) expression may suppress responsiveness to IFN-γ through a
negative feedback loop involving AGO2/protein tyrosine phosphatase
non-receptor type 6/STAT1, thereby promoting immune evasion.
Targeting this pathway has been proposed for tumors with high AGO2
expression (286). These
findings support a practical clinical strategy based on patient
stratification. Baseline and early post-RT IFN-related gene
signatures and circulating cytokine profiles can be used to
classify tumors by their IFN-I signaling state (83,91,213,287). In tumors with low baseline
IFN-I signaling, STING agonists or IFN-I agonists may be considered
to enhance priming (66,67,77,131,282). In tumors with chronically high
IFN-I signaling, the risk of tolerance and resistance should be
evaluated, and shortened exposure or intermittent dosing may be
more appropriate (83,85-88). This approach treats IFN-I as a
modifiable system variable rather than a single-direction target
(131).
Mechanistically, the cGAS-STING pathway lies
upstream of RT-induced IFN-β production and links dendritic cell
cross-presentation with CD8+ T-cell priming, forming a
central axis from innate sensing to adaptive immunity. STING
signaling is required for effective adaptive responses in multiple
models. Loss or inhibition of YTHDF1 reduces cathepsin A- and
cathepsin B-mediated STING degradation, restores STING stability,
increases irradiation-induced IFN-β secretion and dendritic cell
antigen cross-presentation, and promotes CD8+ T-cell
cytotoxicity, thereby strengthening RT efficacy. Based on this
mechanism, a dendritic cell vaccine generated by YTHDF1 knockout or
treatment with the small-molecule inhibitor salvianolic acid C
enhanced the efficacy of RT alone and RT combined with PD-L1
blockade in a preclinical model (81). In addition, combining RT with the
STING agonist cGAMP can reduce radioresistance and enhance host
antitumor immunity (67).
As described above, the CCL22/CCR4 axis contributes
to immune regulation. Targeting this axis may disrupt interactions
between Treg cells and DCs, enhance tumor antigen-specific T-cell
responses and potentially synergize with PD-1 or CTLA-4 inhibitors
(108). In a murine pancreatic
cancer model, stereotactic body RT combined with locally delivered
IL-12 fusion protein remodeled the TME, increased infiltration and
activity of antitumor macrophages and CD8+ T cells and
resulted in durable tumor regression (288).
The DNA damage response network is another hub
linking immunity and RT. When DNA repair is efficient, immunogenic
cell death and cGAS-STING activation may be reduced, whereas
impaired repair can increase cytosolic DNA signaling. Esophageal
squamous cell carcinoma often exhibits radioresistance and poor
prognosis (289). Overcoming
radioresistance remains a priority, and the DNA damage response
machinery that detects and repairs radiation-induced lesions
represents a mechanistic contributor (290,291). PARP1 functions as a central
sensor protein in this network (292). PARP inhibitors can activate
cGAS/STING signaling and promote innate immune activation (74), consistent with RT-driven
cytokine-mediated adaptive immunity (42,293). Although direct links between
PARPis and specific cytokines require further definition,
mechanistic intersections are plausible and clinically relevant. In
esophageal cancer, astrocyte elevated gene-1 recruits the
deubiquitinase biquitin-specific peptidase 10 to remove K48-linked
polyubiquitination at Lys425 of PARP1, reducing PARP1 degradation
and increasing recruitment to double-strand break sites. This
mechanism enhances homologous recombination-mediated repair and
reduces radiation lethality in esophageal squamous carcinoma cells
(294). Vav guanine nucleotide
exchange factor 2 (VAV2) is another candidate target. Increased
VAV2 promotes the formation and activity of the Ku70-Ku80 complex
involved in nonhomologous end joining repair and reduces
radiation-induced DSBs. Under irradiation, VAV2 can also activate
STAT1 signaling and promote radioresistance, and the STAT1
inhibitor fludarabine can reverse this phenotype in a preclinical
model (295).
Cytokine mechanisms of radiation injury are
discussed above. Translational studies also suggest that
irradiation can activate the constitutively expressed NLR family
pyrin domain containing 3 (NLRP3) inflammasome in bone
marrow-derived macrophages, inducing pyroptosis and IL-1β
production, and contributing to tissue injury and immune-cell loss
(296). A plausible mechanism
is that irradiation activates TLRs, leading to priming and
increased NLRP3 expression, which promotes caspase-1 activation,
IL-1β processing and pyroptosis (297-299). Caspase-1-dependent pyroptosis
is not restricted to NLRP3. Other inflammasomes, including NLRP1,
NLRC4 and the DNA-sensing absent in melanoma 2 (AIM2) inflammasome,
may also contribute to radiation-associated caspase-1 activation
(298-300). Irradiation can also induce the
release of M1-type pro-inflammatory cytokines and MCP-1 (301,302), and may amplify inflammatory
cascades through DAMPs and additional inflammasome pathways,
including AIM2 (300,303). These data support targeting
NLRP3-driven pyroptosis as a strategy to reduce
radiation-associated immune-cell loss, pro-inflammatory cytokine
cascades and tissue injury.
This section also summarizes cytokine-related
clinical studies, including therapeutic targets, drug classes and
combination paradigms involving RT with or without immunotherapy,
to support the development and clinical application of
cytokine-directed agents in the RT setting (Table II) (8,304). Table III provides a concise
cross-cytokine overview of representative clinical trials grouped
by cytokine axis, cancer type, RT-containing regimen and study
focus, thereby highlighting recurring translational patterns across
IFN-, IL-2-, GM-CSF- and TGF-β-directed strategies. Several themes
emerge from these trials. TGF-β inhibition has been explored across
multiple tumor types: Fresolimumab is being evaluated in
combination with RT in metastatic breast cancer (NCT01401062) and
with stereotactic ablative body radiotherapy in early-stage
non-small-cell lung cancer (NCT02581787), while bintrafusp α, a
bifunctional TGF-β/PD-L1 inhibitor, is under investigation with RT
in esophageal squamous cell carcinoma (NCT04595149) and in
combination with SBRT and IL-12 agonist M9241 in advanced
pancreatic cancer (NCT04327986). IL-2-based strategies represent
another active area, with trials combining SBRT and high-dose IL-2
in melanoma (NCT01416831), intralesional IL-2 with RT in refractory
metastatic NSCLC (NCT03224871) and bempegaldesleukin (NKTR-214)
plus nivolumab with RT in sarcoma (NCT03282344). GM-CSF has been
combined with SBRT in stage IV NSCLC after second-line chemotherapy
failure (NCT02623595) and with RT and PD-1 inhibition in advanced
recurrent or metastatic head and neck tumors (NCT05760196).
IFN-based approaches include adjuvant IFN-α2b with postoperative RT
for metastatic melanoma (NCT00003444) and IFN-β combined with
avelumab with or without RT for Merkel cell carcinoma
(NCT02584829). Collectively, these trials reflect a broad effort to
target cytokine axes across diverse tumor histologies and RT
delivery platforms.
The efficacy of combining RT with immune checkpoint
inhibitors depends on remodeling the immune state of the TME.
Identification and modulation of key immunoregulatory factors are
therefore important for optimizing combination strategies. IFN-I
signaling induced by irradiation is required to initiate systemic
antitumor immunity. For instance, in IFN-I receptor knockout
models, RT shows minimal efficacy, suggesting that IFN-I signaling
may have predictive value for response to RT combined with
immunotherapy (66,67). Clinically, IFN-related gene
signatures may help estimate radiosensitivity and the likelihood of
benefit from immunotherapy (83). This pathway also offers
actionable targets. STING agonists and PARPis can amplify
RT-induced innate immune signaling and may enhance responses to
PD-1 or PD-L1 blockade (77). In
parallel, TAMs and macrophage-mediated immunosuppression contribute
to radioresistance. After RT, tumors can exhibit increased
macrophage infiltration and M2 polarization, and chemokines such as
CCL2 produced in this setting can recruit MDSCs and weaken
antitumor immunity (92,94). High macrophage density and
hyperactivation of the CCL2/CCR2 axis have been associated with
poor outcomes in RT-immunotherapy combinations and may serve as
candidate biomarkers of response. Interventions targeting this
mechanism have shown benefit in preclinical models. CSF1R
inhibitors can deplete intratumoral macrophages, improve local
control and delay recurrence when combined with RT (35-37,154). Blockade of CCL2/CCR2 signaling
can reduce macrophage chemotaxis, remodel the TME and increase
radiosensitivity (95,153). These findings support an
approach in which IFN signaling and macrophage-related cytokine
profiles are assessed to guide the selection of adjunctive
immunomodulatory interventions.
Mechanism-based prevention and management of
RT-related toxicity is a key translational priority while pursuing
antitumor efficacy. Radiation-induced lung injury is linked to
dysregulated immune responses. RT can trigger an acute inflammatory
cascade in the lung with early increases in mediators such as IL-1,
TNF-α and IL-6, whereas persistent inflammatory signaling
contributes to chronic fibrotic remodeling (188,190-192). Excessive STING activation can
have context-dependent effects. In normal lung tissue, STING
signaling can exacerbate fibrosis by promoting CCR2-dependent
monocyte recruitment and M2 macrophage polarization (195). A preclinical study indicated
that CCR2 deficiency or pharmacologic blockade reduces inflammatory
cell infiltration and capillary damage and can reduce lung injury
after irradiation (96). These
findings support monitoring of inflammatory biomarkers such as IL-6
and TGF-β during RT to identify patients at increased risk and to
consider the timely use of anti-inflammatory or anti-fibrotic
interventions to limit progression from acute inflammation to
chronic injury (189,191). Radiation-induced liver injury
can also involve an innate immunity-driven inflammatory loop.
Ionizing radiation can activate the NLRP3 inflammasome in Kupffer
cells and induce pyroptosis, leading to the release of IL-1β and
other inflammatory mediators and exacerbating parenchymal injury
(250,251). Targeting
inflammasome-associated pyroptosis has therefore been proposed as
an interventional strategy. In preclinical models, disulfiram
inhibits GSDMD-mediated pyroptosis and reduces irradiation-induced
hepatocyte injury and neutrophil infiltration (252,254). In clinical practice, liver
function and relevant inflammatory biomarkers should be monitored
during RT, and hepatoprotective and anti-inflammatory measures
should be implemented when abnormalities are detected to prevent
irreversible decompensation (244,245).
Oral mucositis, a frequent consequence of head and
neck RT, reflects a cascade of immune responses initiated by
epithelial injury. After disruption of the oral mucosal barrier,
exposed basal cells and microbial products can activate pathways
such as NF-κB, induce the release of pro-inflammatory mediators
including TNF and IL-6, and promote neutrophil and macrophage
infiltration, leading to ulceration and pain (220,221). Consistent with this mechanism,
peripheral blood inflammatory markers correlate with mucositis
severity, and routine monitoring of TNF and IL-6 may provide an
adjunctive tool for assessing mucosal damage during treatment
(221). Multiple randomized
controlled trials have evaluated targeted interventions, including
topical recombinant human IL-11 mouthwash to promote mucosal
regeneration, anti-inflammatory mouth rinses and photobiomodulation
to accelerate ulcer healing (232,236,237). Photobiomodulation, supported by
its anti-inflammatory and pro-healing effects, has been recommended
by clinical guidelines as a first-line measure for preventing and
managing radiation-induced oral mucositis (239,240). Thus, mechanism-guided
monitoring and intervention may reduce the incidence and severity
of RT-related toxicities.
The selection of the RT modality and parameters can
alter immune trajectories and toxicity profiles through effects on
cytokine networks, providing an additional dimension for clinical
optimization. Hypofractionated regimens such as SBRT generate
systemic immune and inflammatory environments that can differ from
conventional fractionation because of high conformality and steep
dose gradients (305).
High-dose irradiation-induced cellular damage leads to the release
of ROS, inflammatory mediators and adhesion molecules that can
function as danger signals and enhance immune responses (306). In a clinical study evaluating
SBRT combined with IL-2 in metastatic melanoma or renal cell
carcinoma, the combination increased the frequency of proliferating
CD4+ T cells and early activated effector memory
phenotype cells, with an objective response rate exceeding
historical benchmarks (307).
Preclinical work has also evaluated SBRT combined with cytokine
modulation in pancreatic cancer models. In that setting,
IL-12-related effects depend on IFN-γ signaling and have been
linked to the reversal of T-cell exhaustion (308). A comparative study of RT
techniques from a cytokine perspective reported that
hypofractionated stereotactic RT reduced IL-10 and IL-17 with
limited effects on IL-1α, IL-6, macrophage inflammatory protein 1α
and TNF-α. These patterns were interpreted as consistent with lower
pulmonary toxicity and limited systemic immune perturbation. By
contrast, IMRT altered multiple cytokine pathways associated with
both antitumor and protumor effects, and outcomes may depend on
individual and protocol-specific factors (287).
FLASH RT delivered at ultra-high dose rates has
been reported to reduce normal tissue injury while maintaining
antitumor activity in preclinical models. In animal studies, FLASH
RT reduces free radical accumulation and endothelial injury in
normal tissues and suppresses the production of multiple
pro-inflammatory and profibrotic cytokines. Compared with
conventional RT, FLASH reduces mediators such as TGF-β and IL-1β in
lung tissue, alleviates lung injury and promotes CD4+
T-cell secretion of reparative cytokines, including IL-10 and
IL-22, which may support tissue regeneration (62,63). Proton therapy improves dose
distribution and can reduce normal tissue exposure. When combined
with immunotherapy, proton therapy has been associated with
increased intratumoral IFN-γ+ T cells and reduced TGF-β
signaling, potentially enhancing systemic antitumor immunity
(64). These observations
suggest that dose, fractionation, dose rate and irradiation field
should be considered controllable variables that influence the
balance between immune activation and immunosuppression, and
between antitumor efficacy and normal tissue toxicity.
In summary, integrating mechanistic understanding
of RT-induced immune signaling with clinical decision-making can
support measurable and adjustable therapeutic strategies. Candidate
approaches include profiling cytokines and immune-cell composition,
selecting RT regimens based on patient-specific immune features and
incorporating targeted agents to modulate detrimental inflammatory
or immunosuppressive pathways. These steps may enhance RT-driven
antitumor immunity while limiting normal tissue injury (192). With improved mechanistic
resolution and clinically implementable monitoring and intervention
strategies, RT can be developed as a component of systemic
immunomodulatory treatment, supporting individualized
radioimmunotherapy.
Not applicable.
SZ, YJ, JF, MG, YW and CD collected the relevant
literature and drafted the manuscript. CD designed the review
framework and revised the manuscript. Data authentication is not
applicable. All authors read and approved the final manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
This study received funding from the following sources: The Key
Research Project of Jiangsu Provincial Health Commission (grant no.
ZDB2020022) and the Guidance Science and Technology Plan Project
for Social Development of Zhenjiang City (grant no. FZ2023049).
|
1
|
Rodriguez-Ruiz ME, Vanpouille-Box C,
Melero I, Formenti SC and Demaria S: Immunological mechanisms
responsible for radiation-induced abscopal effect. Trends Immunol.
39:644–655. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Han B, Li C, Meng H, Gomes Romeiro F,
Mancuso A, Zhou Z, Levi Sandri GB, Xu Y, Han T, Han L, et al:
Efficacy and safety of external-beam radiation therapy for
hepatocellular carcinoma: An overview of current evidence according
to the different target population. Biosci Trends. 13:10–22. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kagawa Y, Smith JJ, Fokas E, Watanabe J,
Cercek A, Greten FR, Bando H, Shi Q, Garcia-Aguilar J, Romesser PB,
et al: Future direction of total neoadjuvant therapy for locally
advanced rectal cancer. Nat Rev Gastroenterol Hepatol. 21:444–455.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
McPhail S, Barclay ME, Swann R, Johnson
SA, Alvi R, Barisic A, Bucher O, Creighton N, Denny CA, Dewar RA,
et al: Use of radiotherapy in patients with oesophageal, stomach,
colon, rectal, liver, pancreatic, lung, and ovarian cancer: an
International Cancer Benchmarking Partnership (ICBP)
population-based study. Lancet Oncol. 25:352–365. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liauw SL, Connell PP and Weichselbaum RR:
New paradigms and future challenges in radiation oncology: an
update of biological targets and technology. Sci Transl Med.
5:173sr22013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Begg AC, Stewart FA and Vens C: Strategies
to improve radiotherapy with targeted drugs. Nat Rev Cancer.
11:239–253. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Luke JJ, Lemons JM, Karrison TG, Pitroda
SP, Melotek JM, Zha Y, Al-Hallaq HA, Arina A, Khodarev NN, Janisch
L, et al: Safety and clinical activity of pembrolizumab and
multisite stereotactic body radiotherapy in patients with advanced
solid tumors. J Clin Oncol. 36:1611–1618. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pollom EL, Chin AL, Diehn M, Loo BW and
Chang DT: Normal tissue constraints for abdominal and thoracic
stereotactic body radiotherapy. Semin Radiat Oncol. 27:197–208.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Vignard J, Mirey G and Salles B:
Ionizing-radiation induced DNA double-strand breaks: A direct and
indirect lighting up. Radiother Oncol. 108:362–369. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zou Z, Chang H, Li H and Wang S: Induction
of reactive oxygen species: An emerging approach for cancer
therapy. Apoptosis. 22:1321–1335. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang H, Mu X, He H and Zhang XD: Cancer
radiosensitizers. Trends Pharmacol Sci. 39:24–48. 2018. View Article : Google Scholar
|
|
12
|
Maier P, Hartmann L, Wenz F and Herskind
C: Cellular pathways in response to ionizing radiation and their
targetability for tumor radiosensitization. Int J Mol Sci.
17:1022016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Vitale I, Manic G, Coussens LM, Kroemer G
and Galluzzi L: Macrophages and metabolism in the tumor
microenvironment. Cell Metab. 30:36–50. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang H, Wang T, Yan S, Tang J, Zhang Y,
Wang L, Xu H and Tu C: Crosstalk of pyroptosis and cytokine in the
tumor microenvironment: from mechanisms to clinical implication.
Mol Cancer. 23:2682024. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Aggarwal BB, Gupta SC and Kim JH:
Historical perspectives on tumor necrosis factor and its
superfamily: 25 years later, a golden journey. Blood. 119:651–665.
2012. View Article : Google Scholar
|
|
16
|
Lazear HM, Schoggins JW and Diamond MS:
Shared and distinct functions of type I and III Interferons.
Immunity. 50:907–923. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rose-John S: Interleukin-6 family
cytokines. Cold Spring Harb Perspect Biol. 10:a0284152018.
View Article : Google Scholar
|
|
18
|
Zhang Y, Alexander PB and Wang XF:
TGF-beta family signaling in the control of cell proliferation and
survival. Cold Spring Harb Perspect Biol. 9:a0221452017. View Article : Google Scholar
|
|
19
|
Briukhovetska D, Dorr J, Endres S, Libby
P, Dinarello CA and Kobold S: Interleukins in cancer: from biology
to therapy. Nat Rev Cancer. 21:481–499. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Dougan M, Dranoff G and Dougan SK: GM-CSF,
IL-3, and IL-5 family of cytokines: Regulators of inflammation.
Immunity. 50:796–811. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Batlle E and Massague J: Transforming
growth factor-β signaling in immunity and cancer. Immunity.
50:924–940. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kang S, Tanaka T, Narazaki M and Kishimoto
T: Targeting interleukin-6 signaling in clinic. Immunity.
50:1007–1023. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Leonard WJ, Lin JX and O'Shea JJ: The ү(c)
family of cytokines: Basic biology to therapeutic ramifications.
Immunity. 50:832–850. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mantovani A, Dinarello CA, Molgora M and
Garlanda C: Interleukin-1 and related cytokines in the regulation
of inflammation and immunity. Immunity. 50:778–795. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
McGeachy MJ, Cua DJ and Gaffen SL: The
IL-17 family of cytokines in health and disease. Immunity.
50:892–906. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ouyang W and O'Garra A: IL-10 family
cytokines IL-10 and IL-22: From basic science to clinical
translation. Immunity. 50:871–891. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dranoff G: Cytokines in cancer
pathogenesis and cancer therapy. Nat Rev Cancer. 4:11–22. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dranoff G, Jaffee E, Lazenby A, Golumbek
P, Levitsky H, Brose K, Jackson V, Hamada H, Pardoll D and Mulligan
RC: Vaccination with irradiated tumor cells engineered to secrete
murine granulocyte-macrophage colony-stimulating factor stimulates
potent, specific, and long-lasting anti-tumor immunity. Proc Natl
Acad Sci USA. 90:3539–3543. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Berns AJ, Clift S, Cohen LK, Donehower RC,
Dranoff G, Hauda KM, Jaffee EM, Lazenby AJ, Levitsky HI, Marshall
FF, et al: Phase I study of non-replicating autologous tumor cell
injections using cells prepared with or without GM-CSF gene
transduction in patients with metastatic renal cell carcinoma. Hum
Gene Ther. 6:347–368. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hodi FS, Butler M, Oble DA, Seiden MV,
Haluska FG, Kruse A, Macrae S, Nelson M, Canning C, Lowy I, et al:
Immunologic and clinical effects of antibody blockade of cytotoxic
T lymphocyte-associated antigen 4 in previously vaccinated cancer
patients. Proc Natl Acad Sci USA. 105:3005–3010. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hodi FS, Mihm MC, Soiffer RJ, Haluska FG,
Butler M, Seiden MV, Davis T, Henry-Spires R, MacRae S, Willman A,
et al: Biologic activity of cytotoxic T lymphocyte-associated
antigen 4 antibody blockade in previously vaccinated metastatic
melanoma and ovarian carcinoma patients. Proc Natl Acad Sci USA.
100:4712–4717. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kantoff PW, Higano CS, Shore ND, Berger
ER, Small EJ, Penson DF, Redfern CH, Ferrari AC, Dreicer R, Sims
RB, et al: Sipuleucel-T immunotherapy for castration-resistant
prostate cancer. N Engl J Med. 363:411–422. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Andtbacka RH, Kaufman HL, Collichio F,
Amatruda T, Senzer N, Chesney J, Delman KA, Spitler LE, Puzanov I,
Agarwala SS, et al: Talimogene laherparepvec improves durable
response rate in patients with advanced melanoma. J Clin Oncol.
33:2780–2788. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Anderton H, Wicks IP and Silke J: Cell
death in chronic inflammation: Breaking the cycle to treat
rheumatic disease. Nat Rev Rheumatol. 16:496–513. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Xu J, Mo J, Jiang Y, Yang T, Lu Z, Han L,
Ding J, Shi F and Liu R: Tumor-associated macrophages
inradiotherapy: Mechanisms of polarization, immune regulation and
therapeutic strategies. Int Immunopharmacol. 161:1150092025.
View Article : Google Scholar
|
|
36
|
Yan D, Kowal J, Akkari L, Schuhmacher AJ,
Huse JT, West BL and Joyce JA: Inhibition of colony stimulating
factor-1 receptor abrogates microenvironment-mediated therapeutic
resistance in gliomas. Oncogene. 36:6049–6058. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Stafford JH, Hirai T, Deng L, Chernikova
SB, Urata K, West BL and Brown JM: Colony stimulating factor 1
receptor inhibition delays recurrence of glioblastoma after
radiation by altering myeloid cell recruitment and polarization.
Neuro Oncol. 18:797–806. 2016. View Article : Google Scholar
|
|
38
|
Takeshima T, Pop LM, Laine A, Iyengar P,
Vitetta ES and Hannan R: Key role for neutrophils in
radiation-induced antitumor immune responses: Potentiation with
G-CSF. Proc Natl Acad Sci USA. 113:11300–11305. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Li X, Wang C, Wu Y, Zhang J, Zhang H, Qin
S, Tang L and Yu F: Self-Powered α radionuclide nanomedicine:
Mitochondria-targeted multimodal energy recycling for amplified
radioimmunotherapy. Adv Mater. 37:e25046122025. View Article : Google Scholar
|
|
40
|
Barker HE, Paget JT, Khan AA and
Harrington KJ: The tumour microenvironment after radiotherapy:
Mechanisms of resistance and recurrence. Nat Rev Cancer.
15:409–425. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hanahan D: Hallmarks of cancer: New
dimensions. Cancer Discov. 12:31–46. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhang C, Liang Z, Ma S and Liu X:
Radiotherapy and cytokine storm: Risk and mechanism. Front Oncol.
11:6704642021. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Honeychurch J and Illidge TM: The
influence of radiation in the context of developing combination
immunotherapies in cancer. Ther Adv Vaccines Immunother. 5:115–122.
2017. View Article : Google Scholar
|
|
44
|
Weichselbaum RR, Liang H, Deng L and Fu
YX: Radiotherapy and immunotherapy: A beneficial liaison? Nat Rev
Clin Oncol. 14:365–379. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Colton M, Cheadle EJ, Honeychurch J and
Illidge TM: Reprogramming the tumour microenvironment by
radiotherapy: Implications for radiotherapy and immunotherapy
combinations. Radiat Oncol. 15:2542020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
De Martino M, Daviaud C and Vanpouille-Box
C: Radiotherapy: An immune response modifier for immuno-oncology.
Semin Immunol. 52:1014742021. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Krombach J, Hennel R, Brix N, Orth M,
Schoetz U, Ernst A, Schuster J, Zuchtriegel G, Reichel CA,
Bierschenk S, et al: Priming anti-tumor immunity by radiotherapy:
Dying tumor cell-derived DAMPs trigger endothelial cell activation
and recruitment of myeloid cells. Oncoimmunology. 8:e15230972018.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Fridman WH, Pages F, Sautes-Fridman C and
Galon J: The immune contexture in human tumours: impact on clinical
outcome. Nat Rev Cancer. 12:298–306. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Liu Q, Hao Y, Du R, Hu D, Xie J, Zhang J,
Deng G, Liang N, Tian T, Kasmann L, et al: Radiotherapy programs
neutrophils to an antitumor phenotype by inducing
mesenchymal-epithelial transition. Transl Lung Cancer Res.
10:1424–1443. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kachikwu EL, Iwamoto KS, Liao YP, DeMarco
JJ, Agazaryan N, Economou JS, McBride WH and Schaue D: Radiation
enhances regulatory T cell representation. Int J Radiat Oncol Biol
Phys. 81:1128–1135. 2011. View Article : Google Scholar
|
|
51
|
Arina A, Beckett M, Fernandez C, Zheng W,
Pitroda S, Chmura SJ, Luke JJ, Forde M, Hou Y, Burnette B, et al:
Tumor-reprogrammed resident T cells resist radiation to control
tumors. Nat Commun. 10:39592019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Vanpouille-Box C, Diamond JM, Pilones KA,
Zavadil J, Babb JS, Formenti SC, Barcellos-Hoff MH and Demaria S:
TGFβ is a master regulator of radiation therapy-induced antitumor
immunity. Cancer Res. 75:2232–2242. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Dovedi SJ, Adlard AL, Lipowska-Bhalla G,
McKenna C, Jones S, Cheadle EJ, Stratford IJ, Poon E, Morrow M,
Stewart R, et al: Acquired resistance to fractionated radiotherapy
can be overcome by concurrent PD-L1 blockade. Cancer Res.
74:5458–5468. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Liao Z, Wang Y, Yang Y, Liu X, Yang X,
Tian Y, Deng S, Hu Y, Meng J, Li J, et al: Targeting the cascade
amplification of macrophage colony-stimulating factor to alleviate
the immunosuppressive effects following radiotherapy. Research
(Wash D C). 7:04502024.PubMed/NCBI
|
|
55
|
Antonia SJ, Villegas A, Daniel D, Vicente
D, Murakami S, Hui R, Kurata T, Chiappori A, Lee KH, de Wit M, et
al: Overall survival with durvalumab after chemoradiotherapy in
stage III NSCLC. N Engl J Med. 379:2342–2350. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Redza-Dutordoir M and Averill-Bates DA:
Activation of apoptosis signalling pathways by reactive oxygen
species. Biochim Biophys Acta. 1863:2977–2992. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Ouellette MM, Zhou S and Yan Y: Cell
signaling pathways that promote radioresistance of cancer cells.
Diagnostics (Basel). 12:6562022. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Apetoh L, Ghiringhelli F, Tesniere A,
Obeid M, Ortiz C, Criollo A, Mignot G, Maiuri MC, Ullrich E,
Saulnier P, et al: Toll-like receptor 4-dependent contribution of
the immune system to anticancer chemotherapy and radiotherapy. Nat
Med. 13:1050–1059. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
59
|
Lee Y, Auh SL, Wang Y, Burnette B, Wang Y,
Meng Y, Beckett M, Sharma R, Chin R, Tu T, et al: Therapeutic
effects of ablative radiation on local tumor require CD8+ T cells:
Changing strategies for cancer treatment. Blood. 114:589–595. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Ngwa W, Irabor OC, Schoenfeld JD, Hesser
J, Demaria S and Formenti SC: Using immunotherapy to boost the
abscopal effect. Nat Rev Cancer. 18:313–322. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Price JM, Prabhakaran A and West CML:
Predicting tumour radiosensitivity to deliver precision
radiotherapy. Nat Rev Clin Oncol. 20:83–98. 2023. View Article : Google Scholar
|
|
62
|
Yan O, Wang S, Wang Q and Wang X: FLASH
radiotherapy: Mechanisms of biological effects and the therapeutic
potential in cancer. Biomolecules. 14:7542024. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Lu H, Li M, Quan C, Li C, Li D, Li Z, Xu
J, Zhang L, Liu Q, Dong G and Wang C: FLASH irradiation modulates
immune responses and accelerates lung recovery: A single-cell
perspective. Adv Sci (Weinh). 12:e017972025. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Hu Y, Paris S, Sahoo N, Wang Q, Wang Q,
Barsoumian HB, Huang A, Da Silva J, Bienassis C, Leyton CSK, et al:
Superior antitumor immune response achieved with proton over photon
immunoradiotherapy is amplified by the nanoradioenhancer NBTXR3. J
Nanobiotechnology. 22:5972024. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Schneider WM, Chevillotte MD and Rice CM:
Interferon-stimulated genes: A complex web of host defenses. Annu
Rev Immunol. 32:513–545. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Burnette BC, Liang H, Lee Y, Chlewicki L,
Khodarev NN, Weichselbaum RR, Fu YX and Auh SL: The efficacy of
radiotherapy relies upon induction of type I interferon-dependent
innate and adaptive immunity. Cancer Res. 71:2488–2496. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Deng L, Liang H, Xu M, Yang X, Burnette B,
Arina A, Li XD, Mauceri H, Beckett M, Darga T, et al:
STING-Dependent cytosolic DNA sensing promotes radiation-induced
type I interferon-dependent antitumor immunity in immunogenic
tumors. Immunity. 41:843–852. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Ishikawa H, Ma Z and Barber GN: STING
regulates intracellular DNA-mediated, type I interferon-dependent
innate immunity. Nature. 461:788–792. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Harding SM, Benci JL, Irianto J, Discher
DE, Minn AJ and Greenberg RA: Mitotic progression following DNA
damage enables pattern recognition within micronuclei. Nature.
548:466–470. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Mackenzie KJ, Carroll P, Martin CA, Murina
O, Fluteau A, Simpson DJ, Olova N, Sutcliffe H, Rainger JK, Leitch
A, et al: cGAS surveillance of micronuclei links genome instability
to innate immunity. Nature. 548:461–465. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Lim JY, Gerber SA, Murphy SP and Lord EM:
Type I interferons induced by radiation therapy mediate recruitment
and effector function of CD8(+) T cells. Cancer Immunol Immunother.
63:259–271. 2014. View Article : Google Scholar
|
|
72
|
Yamazaki T, Kirchmair A, Sato A, Buque A,
Rybstein M, Petroni G, Bloy N, Finotello F, Stafford L, Navarro
Manzano E, et al: Mitochondrial DNA drives abscopal responses to
radiation that are inhibited by autophagy. Nat Immunol.
21:1160–1171. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Shen J, Zhao W, Ju Z, Wang L, Peng Y,
Labrie M, Yap TA, Mills GB and Peng G: PARPi triggers the
STING-Dependent immune response and enhances the therapeutic
efficacy of immune checkpoint blockade independent of BRCAness.
Cancer Res. 79:311–319. 2019. View Article : Google Scholar
|
|
74
|
Pantelidou C, Sonzogni O, De Oliveria
Taveira M, Mehta AK, Kothari A, Wang D, Visal T, Li MK, Pinto J,
Castrillon JA, et al: PARP inhibitor efficacy depends on CD8(+)
T-cell recruitment via intratumoral STING pathway activation in
BRCA-deficient models of triple-negative breast cancer. Cancer
Discov. 9:722–737. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Ahn J, Konno H and Barber GN: Diverse
roles of STING-dependent signaling on the development of cancer.
Oncogene. 34:5302–5308. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Garcia-Diaz A, Shin DS, Moreno BH, Saco J,
Escuin-Ordinas H, Rodriguez GA, Zaretsky JM, Sun L, Hugo W, Wang X,
et al: Interferon receptor signaling pathways regulating PD-L1 and
PD-L2 expression. Cell Rep. 19:1189–1201. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Li A, Yi M, Qin S, Song Y, Chu Q and Wu K:
Activating cGAS-STING pathway for the optimal effect of cancer
immunotherapy. J Hematol Oncol. 12:352019. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Stilmann M, Hinz M, Arslan SC, Zimmer A,
Schreiber V and Scheidereit C: A nuclear poly(ADP-ribose)-dependent
signalosome confers DNA damage-induced IkappaB kinase activation.
Mol Cell. 36:365–378. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Demaria O, De Gassart A, Coso S,
Gestermann N, Di Domizio J, Flatz L, Gaide O, Michielin O, Hwu P,
Petrova TV, et al: STING activation of tumor endothelial cells
initiates spontaneous and therapeutic antitumor immunity. Proc Natl
Acad Sci USA. 112:15408–15413. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Chen Q, Boire A, Jin X, Valiente M, Er EE,
Lopez-Soto A, Jacob L, Patwa R, Shah H, Xu K, et al:
Carcinoma-astrocyte gap junctions promote brain metastasis by cGAMP
transfer. Nature. 533:493–498. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Wen C, Wang L, Piffko A, Chen D, Yu X,
Zawieracz K, Bugno J, Yang K, Naccasha EZ, Ji F, et al: YTHDF1 loss
in dendritic cells potentiates radiation-induced antitumor immunity
via STING-dependent type I IFN production. J Clin Invest.
134:e1816122024. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Huang L, Lu W, Wu R, Li Y, Ou Z, Chen J,
Liu Y, Yang W, Xue W, Mu P, et al: Interferon-driven CAF
reprogramming augments immunogenic response to neoadjuvant
radiotherapy in colorectal cancer. Cell Rep Med. 6:1022512025.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Weichselbaum RR, Ishwaran H, Yoon T,
Nuyten DS, Baker SW, Khodarev N, Su AW, Shaikh AY, Roach P, Kreike
B, et al: An interferon-related gene signature for DNA damage
resistance is a predictive marker for chemotherapy and radiation
for breast cancer. Proc Natl Acad Sci USA. 105:18490–18495. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Boelens MC, Wu TJ, Nabet BY, Xu B, Qiu Y,
Yoon T, Azzam DJ, Twyman-Saint Victor C, Wiemann BZ, Ishwaran H, et
al: Exosome transfer from stromal to breast cancer cells regulates
therapy resistance pathways. Cell. 159:499–513. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Erdal E, Haider S, Rehwinkel J, Harris AL
and McHugh PJ: A prosurvival DNA damage-induced cytoplasmic
interferon response is mediated by end resection factors and is
limited by Trex1. Genes Dev. 31:353–369. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Post AEM, Smid M, Nagelkerke A, Martens
JWM, Bussink J, Sweep FCGJ and Span PN: Interferon-stimulated genes
are involved in cross-resistance to radiotherapy in
tamoxifen-resistant breast cancer. Clin Cancer Res. 24:3397–3408.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Benci JL, Xu B, Qiu Y, Wu TJ, Dada H,
Twyman-Saint Victor C, Cucolo L, Lee DSM, Pauken KE, Huang AC, et
al: Tumor interferon signaling regulates a multigenic resistance
program to immune checkpoint blockade. Cell. 167:1540–1554 e12.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Chen J, Cao Y, Markelc B, Kaeppler J,
Vermeer JA and Muschel RJ: Type I IFN protects cancer cells from
CD8+ T cell-mediated cytotoxicity after radiation. J Clin Invest.
129:4224–4238. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Hughes CE and Nibbs RJB: A guide to
chemokines and their receptors. FEBS J. 285:2944–2971. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Bravata V, Minafra L, Forte GI, Cammarata
FP, Russo G, Di Maggio FM, Augello G, Lio D and Gilardi MC:
Cytokine profile of breast cell lines after different radiation
doses. Int J Radiat Biol. 93:1217–1226. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Wang P, Guo F, Han L, Wang X, Li J, Guo Y
and Lu Y: X-ray-induced changes in the expression of
inflammation-related genes in human peripheral blood. Int J Mol
Sci. 15:19516–19534. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Wang X, Yang X, Tsai Y, Yang L, Chuang KH,
Keng PC, Lee SO and Chen Y: IL-6 mediates macrophage infiltration
after irradiation via Up-regulation of CCL2/CCL5 in Non-small cell
lung cancer. Radiat Res. 187:50–59. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Liang H, Deng L, Hou Y, Meng X, Huang X,
Rao E, Zheng W, Mauceri H, Mack M, Xu M, et al: Host
STING-dependent MDSC mobilization drives extrinsic radiation
resistance. Nat Commun. 8:17362017. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Mondini M, Loyher PL, Hamon P, Gerbe de
Thore M, Laviron M, Berthelot K, Clemenson C, Salomon BL,
Combadiere C, Deutsch E and Boissonnas A: CCR2-dependent
recruitment of tregs and monocytes following radiotherapy is
associated with TNFα-mediated resistance. Cancer Immunol Res.
7:376–387. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Kalbasi A, Komar C, Tooker GM, Liu M, Lee
JW, Gladney WL, Ben-Josef E and Beatty GL: Tumor-derived CCL2
mediates resistance to radiotherapy in pancreatic ductal
adenocarcinoma. Clin Cancer Res. 23:137–148. 2017. View Article : Google Scholar
|
|
96
|
Wiesemann A, Ketteler J, Slama A,
Wirsdorfer F, Hager T, Rock K, Engel DR, Fischer JW, Aigner C,
Jendrossek V and Klein D: Inhibition of radiation-induced Ccl2
signaling protects lungs from vascular dysfunction and endothelial
cell loss. Antioxid Redox Signal. 30:213–231. 2019. View Article : Google Scholar
|
|
97
|
Lee SW, Haditsch U, Cord BJ, Guzman R, Kim
SJ, Boettcher C, Priller J, Ormerod BK and Palmer TD: Absence of
CCL2 is sufficient to restore hippocampal neurogenesis following
cranial irradiation. Brain Behav Immun. 30:33–44. 2013. View Article : Google Scholar
|
|
98
|
Malik IA, Moriconi F, Sheikh N, Naz N,
Khan S, Dudas J, Mansuroglu T, Hess CF, Rave-Frank M, Christiansen
H and Ramadori G: Single-dose gamma-irradiation induces
up-regulation of chemokine gene expression and recruitment of
granulocytes into the portal area but not into other regions of rat
hepatic tissue. Am J Pathol. 176:1801–1815. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Nimalasena S, Gothard L, Anbalagan S,
Allen S, Sinnett V, Mohammed K, Kothari G, Musallam A, Lucy C, Yu
S, et al: Intratumoral hydrogen peroxide with radiation therapy in
locally advanced breast cancer: Results from a phase 1 clinical
trial. Int J Radiat Oncol Biol Phys. 108:1019–1029. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Mi S, Qu Y, Chen X, Wen Z, Chen P and
Cheng Y: Radiotherapy increases 12-LOX and CCL5 levels in
esophageal cancer cells and promotes cancer metastasis via
THP-1-Derived macrophages. Onco Targets Ther. 13:7719–7733. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Zheng Z, Jia S, Shao C and Shi Y:
Irradiation induces cancer lung metastasis through activation of
the cGAS-STING-CCL5 pathway in mesenchymal stromal cells. Cell
Death Dis. 11:3262020. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Chen HY, Xu L, Li LF, Liu XX, Gao JX and
Bai YR: Inhibiting the CD8(+) T cell infiltration in the tumor
microenvironment after radiotherapy is an important mechanism of
radioresistance. Sci Rep. 8:119342018. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Yan C, Luo L, Urata Y, Goto S and Li TS:
Nicaraven reduces cancer metastasis to irradiated lungs by
decreasing CCL8 and macrophage recruitment. Cancer Lett.
418:204–210. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Takemura N, Kurashima Y, Mori Y, Okada K,
Ogino T, Osawa H, Matsuno H, Aayam L, Kaneto S, Park EJ, et al:
Eosinophil depletion suppresses radiation-induced small intestinal
fibrosis. Sci Transl Med. 10:eaan03332018. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Lee EJ, Kim JW, Yoo H, Kwak W, Choi WH,
Cho S, Choi YJ, Lee YJ and Cho J: Single high-dose irradiation
aggravates eosinophil-mediated fibrosis through IL-33 secreted from
impaired vessels in the skin compared to fractionated irradiation.
Biochem Biophys Res Commun. 464:20–26. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Bostrom M, Kalm M, Eriksson Y, Bull C,
Stahlberg A, Bjork-Eriksson T, Hellstrom Erkenstam N and Blomgren
K: A role for endothelial cells in radiation-induced inflammation.
Int J Radiat Biol. 94:259–271. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Johnston CJ, Williams JP, Okunieff P and
Finkelstein JN: Radiation-induced pulmonary fibrosis: Examination
of chemokine and chemokine receptor families. Radiat Res.
157:256–265. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Rapp M, Wintergerst MWM, Kunz WG, Vetter
VK, Knott MML, Lisowski D, Haubner S, Moder S, Thaler R, Eiber S,
et al: CCL22 controls immunity by promoting regulatory T cell
communication with dendritic cells in lymph nodes. J Exp Med.
216:1170–1181. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Bottcher JP, Bonavita E, Chakravarty P,
Blees H, Cabeza-Cabrerizo M, Sammicheli S, Rogers NC, Sahai E,
Zelenay S and Reise Sousa C: NK Cells stimulate recruitment of cDC1
into the tumor microenvironment promoting cancer immune control.
Cell. 172:1022–1037 e14. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Matsumura S, Wang B, Kawashima N,
Braunstein S, Badura M, Cameron TO, Babb JS, Schneider RJ, Formenti
SC, Dustin ML and Demaria S: Radiation-induced CXCL16 release by
breast cancer cells attracts effector T cells. J Immunol.
181:3099–3107. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Kioi M, Vogel H, Schultz G, Hoffman RM,
Harsh GR and Brown JM: Inhibition of vasculogenesis, but not
angiogenesis, prevents the recurrence of glioblastoma after
irradiation in mice. J Clin Invest. 120:694–705. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Belarbi K, Jopson T, Arellano C, Fike JR
and Rosi S: CCR2 deficiency prevents neuronal dysfunction and
cognitive impairments induced by cranial irradiation. Cancer Res.
73:1201–1210. 2013. View Article : Google Scholar
|
|
113
|
Lumniczky K, Szatmari T and Safrany G:
Ionizing radiation-induced immune and inflammatory reactions in the
brain. Front Immunol. 8:5172017. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Wang L, Jiang J, Chen Y, Jia Q and Chu Q:
The roles of CC chemokines in response to radiation. Radiat Oncol.
17:632022. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Wang CL, Ho AS, Chang CC, Sie ZL, Peng CL,
Chang J and Cheng CC: Radiotherapy enhances CXCR3(high)CD8(+) T
cell activation through inducing IFNgamma-mediated CXCL10 and
ICAM-1 expression in lung cancer cells. Cancer Immunol Immunother.
72:1865–1880. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Zhu R, Xue X, Shen M, Tsai Y, Keng PC and
Chen Y, Lee SO and Chen Y: NFĸB and TNFα as individual key
molecules associated with the cisplatin-resistance and
radioresistance of lung cancer. Exp Cell Res. 374:181–188. 2019.
View Article : Google Scholar
|
|
117
|
Alrehaili AA, Gharib AF, Bakhuraysah MM,
Alharthi A, Alsalmi O, Alsaeedi FA, Alhakami RA, Alasmari KA,
Mohammed N and Elsawy WH: Serum pro-inflammatory cytokines and
leptin as potential biomarkers for treatment response and toxicity
in locally advanced squamous cell carcinoma of the head and neck.
Diseases. 12:552024. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Chen W, Zhang C, Li Z, Xu Z, Ding C, Wu J,
Wei H, Deng Z, He T, Long L, et al: PROCR diminishes the efficacy
of radiation by impairing T-cell-mediated antitumour immunity. Nat
Commun. 16:71452025. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Han N, Wada H, Kobayashi T, Otsuka R and
Seino KI: A mechanism of IL-34-induced resistance against cytotoxic
anti-cancer therapies such as radiation by X-ray and chemotherapy
by Oxaliplatin. Oncoimmunology. 12:22384992023. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Spiotto M, Fu YX and Weichselbaum RR: The
intersection of radiotherapy and immunotherapy: mechanisms and
clinical implications. Sci Immunol. 1:EAAG12662016. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Kaur R, Lang DK, Singh H, Arora A, Garg N
and Saini B: Repurposing of various current medicines as
radioprotective agents. Anticancer Agents Med Chem. 23:1104–1121.
2023. View Article : Google Scholar
|
|
122
|
Oton-Leite AF, Silva GB, Morais MO, Silva
TA, Leles CR, Valadares MC, Pinezi JC, Batista AC and Mendonca EF:
Effect of low-level laser therapy on chemoradiotherapy-induced oral
mucositis and salivary inflammatory mediators in head and neck
cancer patients. Lasers Surg Med. 47:296–305. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Shields LB, Shelton BJ, Shearer AJ, Chen
L, Sun DA, Parsons S, Bourne TD, LaRocca R and Spalding AC:
Dexamethasone administration during definitive radiation and
temozolomide renders a poor prognosis in a retrospective analysis
of newly diagnosed glioblastoma patients. Radiat Oncol. 10:2222015.
View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Thabet NM and Moustafa EM: Synergistic
effect of Ebselen and gamma radiation on breast cancer cells. Int J
Radiat Biol. 93:784–792. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Wunderlich R, Ernst A, Rodel F, Fietkau R,
Ott O, Lauber K, Frey B and Gaipl US: Low and moderate doses of
ionizing radiation up to 2 Gy modulate transmigration and
chemotaxis of activated macrophages, provoke an anti-inflammatory
cytokine milieu, but do not impact upon viability and phagocytic
function. Clin Exp Immunol. 179:50–61. 2015. View Article : Google Scholar
|
|
126
|
Zhao X, Hu S, Zeng L, Liu X, Song Y, Zhang
Y, Chen Q, Bai Y, Zhang J, Zhang H, et al: Irradiation combined
with PD-L1(−/−) and autophagy inhibition enhances the antitumor
effect of lung cancer via cGAS-STING-mediated T cell activation.
iScience. 25:1046902022. View Article : Google Scholar
|
|
127
|
Straub JM, New J, Hamilton CD, Lominska C,
Shnayder Y and Thomas SM: Radiation-induced fibrosis: Mechanisms
and implications for therapy. J Cancer Res Clin Oncol.
141:1985–1994. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Li R, Chen G, Zhou L, Xu H, Tang F, Lan J,
Tong R, Deng L, Xue J and Lu Y: The fatty acid amide hydrolase
inhibitor URB937 ameliorates radiation-induced lung injury in a
mouse model. Inflammation. 40:1254–1263. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Suzuki K, Ogura M, Abe Y, Suzuki T,
Tobinai K, Ando K, Taniwaki M, Maruyama D, Kojima M, Kuroda J, et
al: Phase 1 study in Japan of siltuximab, an anti-IL-6 monoclonal
antibody, in relapsed/refractory multiple myeloma. Int J Hematol.
101:286–294. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Vanpouille-Box C, Pilones KA, Wennerberg
E, Formenti SC and Demaria S: In situ vaccination by radiotherapy
to improve responses to anti-CTLA-4 treatment. Vaccine.
33:7415–7422. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Palata O, Hradilova Podzimkova N,
Nedvedova E, Umprecht A, Sadilkova L, Palova Jelinkova L, Spisek R
and Adkins I: Radiotherapy in combination with cytokine treatment.
Front Oncol. 9:3672019. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Lemke G: How macrophages deal with death.
Nat Rev Immunol. 19:539–549. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Shi Z, Yu P, Lin WJ, Chen S, Hu X, Chen S,
Cheng J, Liu Q, Yang Y, Li S, et al: Microglia drive transient
insult-induced brain injury by chemotactic recruitment of CD8(+) T
lymphocytes. Neuron. 111:696–710.e9. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Lodermann B, Wunderlich R, Frey S, Schorn
C, Stangl S, Rodel F, Keilholz L, Fietkau R, Gaipl US and Frey B:
Low dose ionising radiation leads to a NF-kappaB dependent
decreased secretion of active IL-1beta by activated macrophages
with a discontinuous dose-dependency. Int J Radiat Biol.
88:727–734. 2012. View Article : Google Scholar
|
|
135
|
Prakash H, Klug F, Nadella V, Mazumdar V,
Schmitz-Winnenthal H and Umansky L: Low doses of gamma irradiation
potentially modifies immunosuppressive tumor microenvironment by
retuning tumor-associated macrophages: lesson from insulinoma.
Carcinogenesis. 37:301–313. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Wu Q, Allouch A, Paoletti A, Leteur C,
Mirjolet C, Martins I, Voisin L, Law F, Dakhli H, Mintet E, et al:
NOX2-dependent ATM kinase activation dictates pro-inflammatory
macrophage phenotype and improves effectiveness to radiation
therapy. Cell Death Differ. 24:1632–1644. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Teresa Pinto A, Laranjeiro Pinto M,
Patricia Cardoso A, Monteiro C, Teixeira Pinto M, Filipe Maia A,
Castro P, Figueira R, Monteiro A, Marques M, et al: Ionizing
radiation modulates human macrophages towards a pro-inflammatory
phenotype preserving their pro-invasive and pro-angiogenic
capacities. Sci Rep. 6:187652016. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Stary V, Wolf B, Unterleuthner D, List J,
Talic M, Laengle J, Beer A, Strobl J, Stary G, Dolznig H and
Bergmann M: Short-course radiotherapy promotes pro-inflammatory
macrophages via extracellular vesicles in human rectal cancer. J
Immunother Cancer. 8:e0006672020. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Crittenden MR, Cottam B, Savage T, Nguyen
C, Newell P and Gough MJ: Expression of NF-kappaB p50 in tumor
stroma limits the control of tumors by radiation therapy. PLoS One.
7:e392952012. View Article : Google Scholar
|
|
140
|
Rodriguez-Ruiz ME, Vitale I, Harrington
KJ, Melero I and Galluzzi L: Immunological impact of cell death
signaling driven by radiation on the tumor microenvironment. Nat
Immunol. 21:120–134. 2020. View Article : Google Scholar
|
|
141
|
Choi JY, Seok HJ, Lee DH, Kwon J, Shin US,
Shin I and Bae IH: miR-1226-5p is involved in radioresistance of
colorectal cancer by activating M2 macrophages through suppressing
IRF1. J Transl Med. 22:9802024. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Chiang Y, Tsai YC, Wang CC, Hsueh FJ,
Huang CY, Chung SD, Chen CH, Pu YS and Cheng JC: Tumor-Derived C-C
motif ligand 2 induces the recruitment and polarization of
tumor-associated macrophages and increases the metastatic potential
of bladder cancer cells in the postirradiated microenvironment. Int
J Radiat Oncol Biol Phys. 114:321–333. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Lee HL, Tsai YC, Pikatan NW, Yeh CT, Yadav
VK, Chen MY and Tsai JT: Tumor-Associated macrophages affect the
tumor microenvironment and radioresistance via the upregulation of
CXCL6/CXCR2 in hepatocellular carcinoma. Biomedicines. 11:20812023.
View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Li W, Xing X, Shen C and Hu C: Tumor
cell-derived exosomal miR-193b-3p promotes tumor-associated
macrophage activation to facilitate nasopharyngeal cancer cell
invasion and radioresistances. Heliyon. 10:e308082024. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Gu X, Shi Y, Dong M, Jiang L, Yang J and
Liu Z: Exosomal transfer of tumor-associated macrophage-derived
hsa_ circ_0001610 reduces radiosensitivity in endometrial cancer.
Cell Death Dis. 12:8182021. View Article : Google Scholar
|
|
146
|
Sheng Y, Zhang B, Xing B, Liu Z, Chang Y,
Wu G and Zhao Y: Cancer-associated fibroblasts exposed to high-dose
ionizing radiation promote M2 polarization of macrophages, which
induce radiosensitivity in cervical cancer. Cancers (Basel).
15:16202023. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Meng Y, Beckett MA, Liang H, Mauceri HJ,
van Rooijen N, Cohen KS and Weichselbaum RR: Blockade of tumor
necrosis factor alpha signaling in tumor-associated macrophages as
a radiosensitizing strategy. Cancer Res. 70:1534–1543. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Fu E, Liu T, Yu S, Chen X, Song L, Lou H,
Ma F, Zhang S, Hussain S, Guo J, et al: M2 macrophages reduce the
radiosensitivity of head and neck cancer by releasing HB-EGF. Oncol
Rep. 44:698–710. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Klug F, Prakash H, Huber PE, Seibel T,
Bender N, Halama N, Pfirschke C, Voss RH, Timke C, Umansky L, et
al: Low-dose irradiation programs macrophage differentiation to an
iNOS(+)/M1 phenotype that orchestrates effective T cell
immunotherapy. Cancer Cell. 24:589–602. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Zhu L, Hu S, Chen Q, Zhang H, Fu J, Zhou
Y, Bai Y, Pan Y and Shao C: Macrophage contributes to
radiation-induced anti-tumor abscopal effect on transplanted breast
cancer by HMGB1/TNF-α signaling factors. Int J Biol Sci.
17:926–941. 2021. View Article : Google Scholar
|
|
151
|
Zhan S, Cao Z, Li J, Chen F, Lai X, Yang
W, Teng Y, Li Z, Zhang W and Xie J: Iron oxide nanoparticles induce
macrophage secretion of ATP and HMGB1 to enhance irradiation-led
immunogenic cell death. Bioconjug Chem. 36:80–91. 2025. View Article : Google Scholar :
|
|
152
|
Wan C, Sun Y, Tian Y, Lu L, Dai X, Meng J,
Huang J, He Q, Wu B, Zhang Z, et al: Irradiated tumor cell-derived
microparticles mediate tumor eradication via cell killing and
immune reprogramming. Sci Adv. 6:eaay97892020. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Connolly KA, Belt BA, Figueroa NM, Murthy
A, Patel A, Kim M, Lord EM, Linehan DC and Gerber SA: Increasing
the efficacy of radiotherapy by modulating the CCR2/CCR5 chemokine
axes. Oncotarget. 7:86522–86535. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Xu J, Escamilla J, Mok S, David J,
Priceman S, West B, Bollag G, McBride W and Wu L: CSF1R signaling
blockade stanches tumor-infiltrating myeloid cells and improves the
efficacy of radiotherapy in prostate cancer. Cancer Res.
73:2782–2794. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Park I, Yang H, Park JS, Koh GY and Choi
EK: VEGF-Grab enhances the efficacy of radiation therapy by
blocking VEGF-A and treatment-induced PlGF. Int J Radiat Oncol Biol
Phys. 102:609–618. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Fulda S: Smac mimetics as IAP antagonists.
Semin Cell Dev Biol. 39:132–138. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Feng Y, Mu R, Wang Z, Xing P, Zhang J,
Dong L and Wang C: A toll-like receptor agonist mimicking microbial
signal to generate tumor-suppressive macrophages. Nat Commun.
10:22722019. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Rodell CB, Arlauckas SP, Cuccarese MF,
Garris CS, Li R, Ahmed MS, Kohler RH, Pittet MJ and Weissleder R:
TLR7/8-agonist-loaded nanoparticles promote the polarization of
tumour-associated macrophages to enhance cancer immunotherapy. Nat
Biomed Eng. 2:578–588. 2018. View Article : Google Scholar :
|
|
159
|
Zhang Y, Feng Z and Liu J, Li H, Su Q,
Zhang J, Huang P, Wang W and Liu J: Polarization of
tumor-associated macrophages by TLR7/8 conjugated radiosensitive
peptide hydrogel for overcoming tumor radioresistance. Bioact
Mater. 16:359–371. 2022.PubMed/NCBI
|
|
160
|
Zanganeh S, Hutter G, Spitler R, Lenkov O,
Mahmoudi M, Shaw A, Pajarinen JS, Nejadnik H, Goodman S, Moseley M,
et al: Iron oxide nanoparticles inhibit tumour growth by inducing
pro-inflammatory macrophage polarization in tumour tissues. Nat
Nanotechnol. 11:986–994. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Hu Y, Sun Y, Liao Z, An D, Liu X, Yang X,
Tian Y, Deng S, Meng J, Wang Y, et al: Irradiated engineered tumor
cell-derived microparticles remodel the tumor immune
microenvironment and enhance antitumor immunity. Mol Ther.
32:411–425. 2024. View Article : Google Scholar
|
|
162
|
Shang Q, Zhang P, Lei X, Du L and Qu B:
Insights into CSF-1/CSF-1R signaling: the role of macrophage in
radiotherapy. Front Immunol. 16:15308902025. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
De Ruysscher D, Niedermann G, Burnet NG,
Siva S, Lee AWM and Hegi-Johnson F: Radiotherapy toxicity. Nat Rev
Dis Primers. 5:132019. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Makale MT, McDonald CR, Hattangadi-Gluth
JA and Kesari S: Mechanisms of radiotherapy-associated cognitive
disability in patients with brain tumours. Nat Rev Neurol.
13:52–64. 2017. View Article : Google Scholar
|
|
165
|
Zhang P, Kishimoto Y, Grammatikakis I,
Gottimukkala K, Cutler RG, Zhang S, Abdelmohsen K, Bohr VA, Misra
Sen J, Gorospe M and Mattson MP: Senolytic therapy alleviates
Abeta-associated oligodendrocyte progenitor cell senescence and
cognitive deficits in an Alzheimer's disease model. Nat Neurosci.
22:719–728. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Ji J, Ding K, Cheng B, Zhang X, Luo T,
Huang B, Yu H, Chen Y, Xu X, Lin H, et al: Radiotherapy-Induced
astrocyte senescence promotes an immunosuppressive microenvironment
in glioblastoma to facilitate tumor regrowth. Adv Sci (Weinh).
11:e23046092024. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Xu A, Li R, Ren A, Jian H, Huang Z, Zeng
Q, Wang B, Zheng J, Chen X, Zheng N, et al: Regulatory coupling
between long noncoding RNAs and senescence in irradiated microglia.
J Neuroinflammation. 17:3212020. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Luo N, Zhu W, Li X, Fu M, Zhang Y, Yang F,
Zhang Y, Chen Z, Zhang Q, Peng B, et al: Defective autophagy of
pericytes enhances radiation-induced senescence promoting radiation
brain injury. Neuro Oncol. 26:2288–2304. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Kalm M, Fukuda A, Fukuda H, Ohrfelt A,
Lannering B, Bjork-Eriksson T, Blennow K, Marky I and Blomgren K:
Transient inflammation in neurogenic regions after irradiation of
the developing brain. Radiat Res. 171:66–76. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Blomstrand M, Kalm M, Grander R,
Bjork-Eriksson T and Blomgren K: Different reactions to irradiation
in the juvenile and adult hippocampus. Int J Radiat Biol.
90:807–815. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Ungvari Z, Podlutsky A, Sosnowska D,
Tucsek Z, Toth P, Deak F, Gautam T, Csiszar A and Sonntag WE:
Ionizing radiation promotes the acquisition of a
senescence-associated secretory phenotype and impairs angiogenic
capacity in cerebromicrovascular endothelial cells: role of
increased DNA damage and decreased DNA repair capacity in
microvascular radiosensitivity. J Gerontol A Biol Sci Med Sci.
68:1443–1457. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Brincat SL and Miller EK:
Frequency-specific hippocampal-prefrontal interactions during
associative learning. Nat Neurosci. 18:576–581. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Wang Y, Zhou K, Li T, Xu Y, Xie C, Sun Y,
Rodriguez J, Zhang S, Song J, Wang X, et al: Selective neural
deletion of the Atg7 gene reduces irradiation-induced cerebellar
white matter injury in the juvenile mouse brain by ameliorating
oligodendrocyte progenitor cell loss. Front Cell Neurosci.
13:2412019. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Leeman DS, Hebestreit K, Ruetz T, Webb AE,
McKay A, Pollina EA, Dulken BW, Zhao X, Yeo RW, Ho TT, et al:
Lysosome activation clears aggregates and enhances quiescent neural
stem cell activation during aging. Science. 359:1277–1283. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Shakhbazau A, Danilkovich N, Seviaryn I,
Ermilova T and Kosmacheva S: Effects of minocycline and rapamycin
in gamma-irradiated human embryonic stem cells-derived cerebral
organoids. Mol Biol Rep. 46:1343–1348. 2019. View Article : Google Scholar
|
|
176
|
Juricic P, Lu YX, Leech T, Drews LF,
Paulitz J, Lu J, Nespital T, Azami S, Regan JC, Funk E, et al:
Long-lasting geroprotection from brief rapamycin treatment in early
adulthood by persistently increased intestinal autophagy. Nat
Aging. 2:824–836. 2022. View Article : Google Scholar
|
|
177
|
Rao E, Hou Y, Huang X, Wang L, Wang J,
Zheng W, Yang H, Yu X, Yang K, Bugno J, et al: All-trans retinoic
acid overcomes solid tumor radioresistance by inducing inflammatory
macrophages. Sci Immunol. 6:eaba84262021. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Martin N, Ma X and Bernard D: Regulation
of cellular senescence by retinoid X receptors and their partners.
Mech Ageing Dev. 183:1111312019. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Zhang Z, Jiang J, He Y, Cai J, Xie J, Wu
M, Xing M, Zhang Z, Chang H, Yu P, et al: Pregabalin mitigates
microglial activation and neuronal injury by inhibiting HMGB1
signaling pathway in radiation-induced brain injury. J
Neuroinflammation. 19:2312022. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Lastra Romero A, Seitz T, Zisiadis GA,
Jeffery H and Osman AM: EDA2R reflects the acute brain response to
cranial irradiation in liquid biopsies. Neuro Oncol. 26:1617–1627.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Bilgic SN, Domaniku A, Toledo B, Agca S,
Weber BZC, Arabaci DH, Ozornek Z, Lause P, Thissen JP, Loumaye A
and Kir S: EDA2R-NIK signalling promotes muscle atrophy linked to
cancer cachexia. Nature. 617:827–834. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Jia N, Jia Y, Yang F and Du W: Knockdown
of EDA2R alleviates hyperoxia-induced lung epithelial cell injury
by inhibiting NF-ĸB pathway. Allergol Immunopathol (Madr).
50:84–90. 2022. View Article : Google Scholar
|
|
183
|
Colonna M and Butovsky O: Microglia
function in the central nervous system during health and
neurodegeneration. Annu Rev Immunol. 35:441–468. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Kalm M, Lannering B, Bjork-Eriksson T and
Blomgren K: Irradiation-induced loss of microglia in the young
brain. J Neuroimmunol. 206:70–75. 2009. View Article : Google Scholar
|
|
185
|
Osman AM, Sun Y, Burns TC, He L, Kee N,
Oliva-Vilarnau N, Alevyzaki A, Zhou K, Louhivuori L, Uhlen P, et
al: Radiation triggers a dynamic sequence of transient microglial
alterations in juvenile brain. Cell Rep. 31:1076992020. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Monje ML, Toda H and Palmer TD:
Inflammatory blockade restores adult hippocampal neurogenesis.
Science. 302:1760–1765. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Trott KR, Herrmann T and Kasper M: Target
cells in radiation pneumopathy. Int J Radiat Oncol Biol Phys.
58:463–469. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Rube CE, Wilfert F, Palm J, Konig J,
Burdak-Rothkamm S, Liu L, Schuck A, Willich N and Rube C:
Irradiation induces a biphasic expression of pro-inflammatory
cytokines in the lung. Strahlenther Onkol. 180:442–448. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Ozturk B, Egehan I, Atavci S and Kitapci
M: Pentoxifylline in prevention of radiation-induced lung toxicity
in patients with breast and lung cancer: A double-blind randomized
trial. Int J Radiat Oncol Biol Phys. 58:213–219. 2004. View Article : Google Scholar
|
|
190
|
Kasmann L, Dietrich A, Staab-Weijnitz CA,
Manapov F, Behr J, Rimner A, Jeremic B, Senan S, De Ruysscher D,
Lauber K and Belka C: Radiation-induced lung toxicity-cellular and
molecular mechanisms of pathogenesis, management, and literature
review. Radiat Oncol. 15:2142020. View Article : Google Scholar
|
|
191
|
Venkatesulu BP, Mahadevan LS, Aliru ML,
Yang X, Bodd MH, Singh PK, Yusuf SW, Abe JI and Krishnan S:
Radiation-induced endothelial vascular injury: A review of possible
mechanisms. JACC Basic Transl Sci. 3:563–572. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Cytlak UM, Dyer DP, Honeychurch J,
Williams KJ, Travis MA and Illidge TM: Immunomodulation by
radiotherapy in tumour control and normal tissue toxicity. Nat Rev
Immunol. 22:124–138. 2022. View Article : Google Scholar
|
|
193
|
Liu X, Zhang T, Zhou J, Xiao Z, Li Y,
Zhang Y, Yue H, Li Z and Tian J: β-Catenin/Lin28/let-7 regulatory
network determines type II alveolar epithelial stem cell
differentiation phenotypes following thoracic irradiation. J Radiat
Res. 62:119–132. 2021. View Article : Google Scholar :
|
|
194
|
House IG, Savas P, Lai J, Chen AXY, Oliver
AJ, Teo ZL, Todd KL, Henderson MA, Giuffrida L, Petley EV, et al:
Macrophage-derived CXCL9 and CXCL10 are required for antitumor
immune responses following immune checkpoint blockade. Clin Cancer
Res. 26:487–504. 2020. View Article : Google Scholar
|
|
195
|
Ni J, Guo T, Zhou Y, Jiang S, Zhang L and
Zhu Z: STING signaling activation modulates macrophage polarization
via CCL2 in radiation-induced lung injury. J Transl Med.
21:5902023. View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Kishore A and Petrek M: Roles of
macrophage polarization and macrophage-derived miRNAs in pulmonary
fibrosis. Front Immunol. 12:6784572021. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Zhou BW, Liu HM, Xu F and Jia XH: The role
of macrophage polarization and cellular crosstalk in the pulmonary
fibrotic microenvironment: A review. Cell Commun Signal.
22:1722024. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Inoue T, Fujishima S, Ikeda E, Yoshie O,
Tsukamoto N, Aiso S, Aikawa N, Kubo A, Matsushima K and Yamaguchi
K: CCL22 and CCL17 in rat radiation pneumonitis and in human
idiopathic pulmonary fibrosis. Eur Respir J. 24:49–56. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Strandberg J, Louie A, Lee S, Hahn M,
Srinivasan P, George A, De La Cruz A, Zhang L, Hernandez Borrero L,
Huntington KE, et al: TRAIL agonists rescue mice from
radiation-induced lung, skin, or esophageal injury. J Clin Invest.
135:e1736492025. View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Ban Y, Markowitz GJ, Zou Y, Ramchandani D,
Kraynak J, Sheng J, Lee SB, Wong STC, Altorki NK, Gao D and Mittal
V: Radiation-activated secretory proteins of Scgb1a1(+) club cells
increase the efficacy of immune checkpoint blockade in lung cancer.
Nat Cancer. 2:919–931. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Palma DA, Senan S, Oberije C, Belderbos J,
de Dios NR, Bradley JD, Barriger RB, Moreno-Jimenez M, Kim TH,
Ramella S, et al: Predicting esophagitis after chemoradiation
therapy for non-small cell lung cancer: An individual patient data
meta-analysis. Int J Radiat Oncol Biol Phys. 87:690–696. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Gomez DR, Tucker SL, Martel MK, Mohan R,
Balter PA, Lopez Guerra JL, Liu H, Komaki R, Cox JD and Liao Z:
Predictors of high-grade esophagitis after definitive
three-dimensional conformal therapy, intensity-modulated radiation
therapy, or proton beam therapy for non-small cell lung cancer. Int
J Radiat Oncol Biol Phys. 84:1010–1016. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Bradley JD, Bae K, Graham MV, Byhardt R,
Govindan R, Fowler J, Purdy JA, Michalski JM, Gore E and Choy H:
Primary analysis of the phase II component of a phase I/II dose
intensification study using three-dimensional conformal radiation
therapy and concurrent chemotherapy for patients with inoperable
non-small-cell lung cancer: RTOG 0117. J Clin Oncol. 28:2475–2480.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Kitamura H, Tanigawa T, Kuzumoto T,
Nadatani Y, Otani K, Fukunaga S, Hosomi S, Tanaka F, Kamata N,
Nagami Y, et al: Interferon-alpha exerts proinflammatory properties
in experimental radiation-induced esophagitis: Possible involvement
of plasmacytoid dendritic cells. Life Sci. 289:1202152022.
View Article : Google Scholar
|
|
205
|
Fries PN and Griebel PJ: Mucosal dendritic
cell diversity in the gastrointestinal tract. Cell Tissue Res.
343:33–41. 2011. View Article : Google Scholar
|
|
206
|
Fitzgerald-Bocarsly P, Dai J and Singh S:
Plasmacytoid dendritic cells and type I IFN: 50 years of convergent
history. Cytokine Growth Factor Rev. 19:3–19. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Reizis B: Plasmacytoid dendritic cells:
Development, regulation, and function. Immunity. 50:37–50. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Vanneste BG, Van De Voorde L, de Ridder
RJ, Van Limbergen EJ, Lambin P and van Lin EN: Chronic radiation
proctitis: Tricks to prevent and treat. Int J Colorectal Dis.
30:1293–1303. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Lu W, Xie Y, Huang B, Ma T, Wang H, Deng
B, Zou S, Wang W, Tang Q, Yang Z, et al: Platelet-derived growth
factor C signaling is a potential therapeutic target for radiation
proctopathy. Sci Transl Med. 13:eabc23442021. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Ge Z, Chen C, Chen J, Jiang Z, Chen L, Wei
Y, Chen H, He L, Zou Y, Long X, et al: Gut microbiota-derived
3-hydroxybutyrate blocks GPR43-Mediated IL6 signaling to ameliorate
radiation proctopathy. Adv Sci (Weinh). 11:e23062172024. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Lei R, Li J, Liu F, Li W, Zhang S, Wang Y,
Chu X and Xu J: HIF-1α promotes the keloid development through the
activation of TGF-beta/Smad and TLR4/MyD88/NF-ĸB pathways. Cell
Cycle. 18:3239–3250. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Ramakrishnan S, Anand V and Roy S:
Vascular endothelial growth factor signaling in hypoxia and
inflammation. J Neuroimmune Pharmacol. 9:142–160. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Bedini N, Cicchetti A, Palorini F, Magnani
T, Zuco V, Pennati M, Campi E, Allavena P, Pesce S, Villa S, et al:
Evaluation of mediators associated with the inflammatory response
in prostate cancer patients undergoing radiotherapy. Dis Markers.
2018:91281282018. View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Hamade DF, Epperly MW, Fisher R, Hou W,
Shields D, van Pijkeren JP, Mukherjee A, Yu J, Leibowitz BJ, Vlad
AM, et al: Release of Interferon-β (IFN-β) from probiotic
limosilactobacillus reuteri-IFN-β (LR-IFN-β) mitigates
gastrointestinal acute radiation syndrome (GI-ARS) following whole
abdominal irradiation. Cancers (Basel). 15:16702023. View Article : Google Scholar
|
|
215
|
Xie LW, Cai S, Lu HY, Tang FL, Zhu RQ,
Tian Y and Li M: Microbiota-derived I3A protects the intestine
against radiation injury by activating AhR/IL-10/Wnt signaling and
enhancing the abundance of probiotics. Gut Microbes.
16:23477222024. View Article : Google Scholar : PubMed/NCBI
|
|
216
|
Romesser PB, Cahlon O, Scher ED, Hug EB,
Sine K, DeSelm C, Fox JL, Mah D, Garg MK, Han-Chih Chang J and Lee
NY: Proton beam reirradiation for recurrent head and neck cancer:
Multi-institutional report on feasibility and early outcomes. Int J
Radiat Oncol Biol Phys. 95:386–395. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Lin L, Yao JJ, Zhou GQ, Guo R, Zhang F,
Zhang Y, Xu L, Zhang LL, Lin AH, Ma J and Sun Y: The efficacy and
toxicity of individualized intensity-modulated radiotherapy based
on the tumor extension patterns of nasopharyngeal carcinoma.
Oncotarget. 7:20680–20690. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Elad S, Yarom N, Zadik Y, Kuten-Shorrer M
and Sonis ST: The broadening scope of oral mucositis and oral
ulcerative mucosal toxicities of anticancer therapies. CA Cancer J
Clin. 72:57–77. 2022.
|
|
219
|
Potrich AR, So BB, Schuch LF, Wagner VP,
Silveira FM, de Abreu Alves F, Prado-Ribeiro AC, Santos-Silva AR,
Treister NS, Martins MD and Martins MAT: Impact of
photobiomodulation for prevention of oral mucositis on the quality
of life of patients with head and neck cancer: A systematic review.
Lasers Med Sci. 39:12023. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Sonis ST: The pathobiology of mucositis.
Nat Rev Cancer. 4:277–284. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Logan RM, Stringer AM, Bowen JM, Gibson
RJ, Sonis ST and Keefe DM: Serum levels of NFkappaB and
pro-inflammatory cytokines following administration of mucotoxic
drugs. Cancer Biol Ther. 7:1139–1145. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Vasconcelos RM, Sanfilippo N, Paster BJ,
Kerr AR, Li Y, Ramalho L, Queiroz EL, Smith B, Sonis ST and Corby
PM: Host-Microbiome cross-talk in oral mucositis. J Dent Res.
95:725–733. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Thaiss CA, Zmora N, Levy M and Elinav E:
The microbiome and innate immunity. Nature. 535:65–74. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Bonan PR, Kaminagakura E, Pires FR, Vargas
PA and De Almeida OP: Histomorphometry and immunohistochemical
features of grade I (WHO) oral radiomucositis. Oral Diseases.
13:170–176. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Imam T, Park S, Kaplan MH and Olson MR:
Effector T helper cell subsets in inflammatory bowel diseases.
Front Immunol. 9:12122018. View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Krebs CF, Schmidt T, Riedel JH and Panzer
U: T helper type 17 cells in immune-mediated glomerular disease.
Nat Rev Nephrol. 13:647–659. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Oronsky B, Goyal S, Kim MM, Cabrales P,
Lybeck M, Caroen S, Oronsky N, Burbano E, Carter C and Oronsky A: A
review of clinical radioprotection and chemoprotection for oral
mucositis. Transl Oncol. 11:771–778. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Lozano A, Marruecos J, Rubio J, Farre N,
Gomez-Millan J, Morera R, Planas I, Lanzuela M, Vazquez-Masedo MG,
Cascallar L, et al: Randomized placebo-controlled phase II trial of
high-dose melatonin mucoadhesive oral gel for the prevention and
treatment of oral mucositis in patients with head and neck cancer
undergoing radiation therapy concurrent with systemic treatment.
Clin Transl Oncol. 23:1801–1810. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Giralt J, Tao Y, Kortmann RD, Zasadny X,
Contreras-Martinez J, Ceruse P, Arias de la Vega F, Lalla RV,
Ozsahin EM, Pajkos G, et al: Randomized phase 2 trial of a novel
clonidine mucoadhesive buccal tablet for the amelioration of oral
mucositis in patients treated with concomitant chemoradiation
therapy for head and neck cancer. Int J Radiat Oncol Biol Phys.
106:320–328. 2020. View Article : Google Scholar
|
|
230
|
Flanders CA, Rocke AS, Edwardson SA,
Baillie JK and Walsh TS: The effect of dexmedetomidine and
clonidine on the inflammatory response in critical illness: A
systematic review of animal and human studies. Crit Care.
23:4022019. View Article : Google Scholar : PubMed/NCBI
|
|
231
|
Marques LJ, Zheng L, Poulakis N, Guzman J
and Costabel U: Pentoxifylline inhibits TNF-alpha production from
human alveolar macrophages. Am J Respir Crit Care Med. 159:508–511.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
232
|
Sayed R, El Wakeel L, Saad AS, Kelany M
and El-Hamamsy M: Pentoxifylline and vitamin E reduce the severity
of radiotherapy-induced oral mucositis and dysphagia in head and
neck cancer patients: A randomized, controlled study. Med Oncol.
37:82019. View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Wei H, Wei J and Dong X: A prospective
interventional study of recombinant human interleukin-11 mouthwash
in chemotherapy-induced oral mucositis. BMC Oral Health.
22:3132022. View Article : Google Scholar : PubMed/NCBI
|
|
234
|
Zhang Y, Li Y, He A, Wang J, Zhang P, Lei
B, Huang Z, Zhang L, Zhao W and Ma X: Efficacy of recombinant human
interleukin-11 in preventing and treating oral mucositis after
chemotherapy for patients with acute leukemia. BMC Oral Health.
23:4762023. View Article : Google Scholar : PubMed/NCBI
|
|
235
|
Aggarwal BB, Gupta SC and Sung B:
Curcumin: An orally bioavailable blocker of TNF and other
pro-inflammatory biomarkers. Br J Pharmacol. 169:1672–1692. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
236
|
Thomas PL, Kang HK and Rishi KS:
Randomized control study of the effects of turmeric mouthwash on
oral health status, treatment-induced mucositis, and associated
oral dysfunctions among patients with head and neck cancer. Cancer
Nurs. 46:36–44. 2023. View Article : Google Scholar
|
|
237
|
Ramezani V, Ghadirian S, Shabani M,
Boroumand MA, Daneshvar R and Saghafi F: Efficacy of curcumin for
amelioration of radiotherapy-induced oral mucositis: A preliminary
randomized controlled clinical trial. BMC Cancer. 23:3542023.
View Article : Google Scholar : PubMed/NCBI
|
|
238
|
Wagner VP, Curra M, Webber LP, Nor C,
Matte U, Meurer L and Martins MD: Photobiomodulation regulates
cytokine release and new blood vessel formation during oral wound
healing in rats. Lasers Med Sci. 31:665–671. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
239
|
Zadik Y, Arany PR, Fregnani ER, Bossi P,
Antunes HS, Bensadoun RJ, Gueiros LA, Majorana A, Nair RG, Ranna V,
et al: Systematic review of photobiomodulation for the management
of oral mucositis in cancer patients and clinical practice
guidelines. Support Care Cancer. 27:3969–3983. 2019.PubMed/NCBI
|
|
240
|
Elad S, Cheng KKF, Lalla RV, Yarom N, Hong
C, Logan RM, Bowen J, Gibson R, Saunders DP, Zadik Y, et al:
MASCC/ISOO clinical practice guidelines for the management of
mucositis secondary to cancer therapy. Cancer. 126:4423–4431. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
241
|
Robijns J, Nair RG, Lodewijckx J, Arany P,
Barasch A, Bjordal JM, Bossi P, Chilles A, Corby PM, Epstein JB, et
al: Photobiomodulation therapy in management of cancer
therapy-induced side effects: WALT position paper 2022. Front
Oncol. 12:9276852022. View Article : Google Scholar : PubMed/NCBI
|
|
242
|
Cox PB and Gupta R: Contemporary
computational applications and tools in drug discovery. ACS Med
Chem Lett. 13:1016–1029. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
243
|
Pushpakom S, Iorio F, Eyers PA, Escott KJ,
Hopper S, Wells A, Doig A, Guilliams T, Latimer J, McNamee C, et
al: Drug repurposing: progress, challenges and recommendations. Nat
Rev Drug Discov. 18:41–58. 2019. View Article : Google Scholar
|
|
244
|
Kim J and Jung Y: Radiation-induced liver
disease: Current understanding and future perspectives. Exp Mol
Med. 49:e3592017. View Article : Google Scholar : PubMed/NCBI
|
|
245
|
Guha C and Kavanagh BD: Hepatic radiation
toxicity: Avoidance and amelioration. Semin Radiat Oncol.
21:256–263. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
246
|
Fischietti M, Fratini E, Verzella D,
Vecchiotti D, Capece D, Di Francesco B, Esposito G, Balata M,
Ioannuci L, Sykes P, et al: Low radiation environment switches the
overgrowth-induced cell apoptosis toward autophagy. Front Public
Health. 8:5947892021. View Article : Google Scholar : PubMed/NCBI
|
|
247
|
Xu B, Jiang M, Chu Y, Wang W, Chen D, Li
X, Zhang Z, Zhang D, Fan D, Nie Y, et al: Gasdermin D plays a key
role as a pyroptosis executor of non-alcoholic steatohepatitis in
humans and mice. J Hepatol. 68:773–782. 2018. View Article : Google Scholar
|
|
248
|
Lv X, Chen J, He J, Hou L, Ren Y, Shen X,
Wang Y, Ji T and Cai X: Gasdermin D-mediated pyroptosis suppresses
liver regeneration after 70% partial hepatectomy. Hepatol Commun.
6:2340–2353. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
249
|
Li J, Zhao J, Xu M, Li M, Wang B, Qu X, Yu
C, Hang H, Xia Q, Wu H, et al: Blocking GSDMD processing in innate
immune cells but not in hepatocytes protects hepatic
ischemia-reperfusion injury. Cell Death Dis. 11:2442020. View Article : Google Scholar : PubMed/NCBI
|
|
250
|
Chen G, Zhao Q, Yuan B, Wang B, Zhang Y,
Li Z, Du S and Zeng Z: ALKBH5-Modified HMGB1-STING activation
contributes to radiation induced liver disease via innate immune
response. Int J Radiat Oncol Biol Phys. 111:491–501. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
251
|
Dong A, Wei G, Liang Z, Di Y, Tang Y, Ling
Y, Li S, Chen Y, Zhou Y, Wang X and Peng Z: Gasdermin D aggravates
a mouse model of radiation-induced liver disease by promoting
chemokine secretion and neutrophil recruitment. Nat Commun.
16:60642025. View Article : Google Scholar : PubMed/NCBI
|
|
252
|
Deng W, Yang Z, Yue H, Ou Y, Hu W and Sun
P: Disulfiram suppresses NLRP3 inflammasome activation to treat
peritoneal and gouty inflammation. Free Radic Biol Med. 152:8–17.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
253
|
Hu S, Wang L, Xu Y, Li F and Wang T:
Disulfiram attenuates hypoxia-induced pulmonary hypertension by
inhibiting GSDMD cleavage and pyroptosis in HPASMCs. Respir Res.
23:3532022. View Article : Google Scholar : PubMed/NCBI
|
|
254
|
Wu J, Zhang J, Zhao J, Chen S, Zhou T and
Xu J: Treatment of severe acute pancreatitis and related lung
injury by targeting gasdermin D-Mediated pyroptosis. Front Cell Dev
Biol. 9:7801422021. View Article : Google Scholar : PubMed/NCBI
|
|
255
|
Liu M, Liu D, Yu C, Fan HH, Zhao X, Wang
H, Zhang C, Zhang M, Bo R, He S, et al: Caffeic acid, but not
ferulic acid, inhibits macrophage pyroptosis by directly blocking
gasdermin D activation. MedComm (2020). 4:e2552023. View Article : Google Scholar : PubMed/NCBI
|
|
256
|
Chatzipapas KP, Papadimitroulas P,
Emfietzoglou D, Kalospyros SA, Hada M, Georgakilas AG and Kagadis
GC: Ionizing radiation and complex DNA damage: Quantifying the
radiobiological damage using monte carlo simulations. Cancers
(Basel). 12:7992020. View Article : Google Scholar : PubMed/NCBI
|
|
257
|
Taysi S, Tascan AS, Ugur MG and Demir M:
Radicals, oxidative/nitrosative stress and preeclampsia. Mini Rev
Med Chem. 19:178–193. 2019. View Article : Google Scholar
|
|
258
|
Taysi S, Algburi FS, Mohammed ZR, Ali OA
and Taysi ME: Thymoquinone: A review on its pharmacological
importance, and its association with oxidative stress, COVID-19,
and radiotherapy. Mini Rev Med Chem. 22:1847–1875. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
259
|
Dent P, Yacoub A, Fisher PB, Hagan MP and
Grant S: MAPK pathways in radiation responses. Oncogene.
22:5885–5896. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
260
|
Du SS, Qiang M, Zeng ZC, Ke AW, Ji Y,
Zhang ZY, Zeng HY and Liu Z: Inactivation of kupffer cells by
gadolinium chloride protects murine liver from radiation-induced
apoptosis. Int J Radiat Oncol Biol Phys. 76:1225–1234. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
261
|
Mossanen JC, Krenkel O, Ergen C, Govaere
O, Liepelt A, Puengel T, Heymann F, Kalthoff S, Lefebvre E, Eulberg
D, et al: Chemokine (C-C motif) receptor 2-positive monocytes
aggravate the early phase of acetaminophen-induced acute liver
injury. Hepatology. 64:1667–1682. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
262
|
Zigmond E, Samia-Grinberg S, Pasmanik-Chor
M, Brazowski E, Shibolet O, Halpern Z and Varol C: Infiltrating
monocyte-derived macrophages and resident kupffer cells display
different ontogeny and functions in acute liver injury. J Immunol.
193:344–353. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
263
|
Yuan D, Huang S, Berger E, Liu L, Gross N,
Heinzmann F, Ringelhan M, Connor TO, Stadler M, Meister M, et al:
Kupffer cell-derived tnf triggers cholangiocellular tumorigenesis
through JNK due to chronic mitochondrial dysfunction and ROS.
Cancer Cell. 31:771–789 e6. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
264
|
Su L, Li N, Tang H, Lou Z, Chong X, Zhang
C, Su J and Dong X: Kupffer cell-derived TNF-alpha promotes
hepatocytes to produce CXCL1 and mobilize neutrophils in response
to necrotic cells. Cell Death Dis. 9:3232018. View Article : Google Scholar
|
|
265
|
Natarajan V, Harris EN and Kidambi S: SECs
(Sinusoidal Endothelial Cells), liver microenvironment, and
fibrosis. Biomed Res Int. 2017:40972052017. View Article : Google Scholar : PubMed/NCBI
|
|
266
|
Chen YX, Zeng ZC, Sun J, Zeng HY, Huang Y
and Zhang ZY: Mesenchymal stem cell-conditioned medium prevents
radiation-induced liver injury by inhibiting inflammation and
protecting sinusoidal endothelial cells. J Radiat Res. 56:700–708.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
267
|
Yuan B, Chen Y, Wu Z, Zhang L, Zhuang Y,
Zhao X, Niu H, Cheng JC and Zeng Z: Proteomic profiling of human
hepatic stellate cell line LX2 responses to irradiation and TGF-β1.
J Proteome Res. 18:508–521. 2019.
|
|
268
|
Seong J, Kim SH, Chung EJ, Lee WJ and Suh
CO: Early alteration in TGF-beta mRNA expression in irradiated rat
liver. Int J Radiat Oncol Biol Phys. 46:639–643. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
269
|
Meng XM, Nikolic-Paterson DJ and Lan HY:
TGF-β: The master regulator of fibrosis. Nat Rev Nephrol.
12:325–338. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
270
|
Wu Y, Wang W, Peng XM, He Y, Xiong YX,
Liang HF, Chu L, Zhang BX, Ding ZY and Chen XP: Rapamycin
upregulates connective tissue growth factor expression in hepatic
progenitor cells through TGF-β-Smad2 dependent signaling. Front
Pharmacol. 9:8772018. View Article : Google Scholar
|
|
271
|
Du SS, Qiang M, Zeng ZC, Zhou J, Tan YS,
Zhang ZY, Zeng HY and Liu ZS: Radiation-induced liver fibrosis is
mitigated by gene therapy inhibiting transforming growth factor-β
signaling in the rat. Int J Radiat Oncol Biol Phys. 78:1513–1523.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
272
|
Zhu W, Zhang X, Yu M, Lin B and Yu C:
Radiation-induced liver injury and hepatocyte senescence. Cell
Death Discov. 7:2442021. View Article : Google Scholar : PubMed/NCBI
|
|
273
|
Marampon F, Gravina GL, Festuccia C, Popov
VM, Colapietro A, Sanita P, Musio D, De Felice F, Lenzi A, Jannini
EA, et al: Vitamin D protects endothelial cells from
irradiation-induced senescence and apoptosis by modulating
MAPK/SirT1 axis. J Endocrinol Invest. 39:411–422. 2016. View Article : Google Scholar
|
|
274
|
Chen YX, Zeng ZC, Sun J, Zhang ZY, Zeng HY
and Hu WX: Radioprotective effect of kupffer cell depletion on
hepatic sinusoidal endothelial cells. Radiat Res. 183:563–570.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
275
|
Sun YW, Zhang YY, Ke XJ, Wu XJ, Chen ZF
and Chi P: Pirfenidone prevents radiation-induced intestinal
fibrosis in rats by inhibiting fibroblast proliferation and
differentiation and suppressing the TGF-β1/Smad/CTGF signaling
pathway. Eur J Pharmacol. 822:199–206. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
276
|
Xie Y, Zhang J, Xu Y and Shao C: SirT1
confers hypoxia-induced radioresistance via the modulation of c-Myc
stabilization on hepatoma cells. J Radiat Res. 53:44–50. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
277
|
Mahmoud Moustafa E, Rashed ER, Rashed RR
and Omar NN: Piceatannol promotes hepatic and renal
AMPK/SIRT1/PGC-1alpha mitochondrial pathway in rats exposed to
reserpine or gamma-radiation. Int J Immunopathol Pharmacol.
35:205873842110161942021. View Article : Google Scholar
|
|
278
|
Srinivasan M, Sudheer AR, Rajasekaran KN
and Menon VP: Effect of curcumin analog on gamma-radiation-induced
cellular changes in primary culture of isolated rat hepatocytes in
vitro. Chem Biol Interact. 176:1–8. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
279
|
Li W, Jiang L, Lu X, Liu X and Ling M:
Curcumin protects radiation-induced liver damage in rats through
the NF-ĸB signaling pathway. BMC Complement Med Ther. 21:102021.
View Article : Google Scholar
|
|
280
|
Zhang J, Zhou S, Zhou Y, Feng F, Wang Q,
Zhu X, Ai H, Huang X and Zhang X: Hepatocyte growth factor
gene-modified adipose-derived mesenchymal stem cells ameliorate
radiation induced liver damage in a rat model. PLoS One.
9:e1146702014. View Article : Google Scholar : PubMed/NCBI
|
|
281
|
Lee YH, Tai D, Yip C, Choo SP and Chew V:
Combinational immunotherapy for hepatocellular carcinoma:
Radiotherapy, immune checkpoint blockade and beyond. Front Immunol.
11:5687592020. View Article : Google Scholar : PubMed/NCBI
|
|
282
|
Yi M, Li T, Niu M, Mei Q, Zhao B, Chu Q,
Dai Z and Wu K: Exploiting innate immunity for cancer
immunotherapy. Mol Cancer. 22:1872023. View Article : Google Scholar : PubMed/NCBI
|
|
283
|
De Ridder M, Jiang H, Van Esch G, Law K,
Monsaert C, Van den Berge DL, Verellen D, Verovski VN and Storme
GA: IFN-gamma+ CD8+ T lymphocytes: possible link between immune and
radiation responses in tumor-relevant hypoxia. Int J Radiat Oncol
Biol Phys. 71:647–651. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
284
|
He K, Puebla-Osorio N, Barsoumian HB,
Sezen D, Rafiq Z, Riad TS, Hu Y, Huang A, Voss TA, Leyton CSK, et
al: Novel engineered IL-2 Nemvaleukin alfa combined with PD1
checkpoint blockade enhances the systemic anti-tumor responses of
radiation therapy. J Exp Clin Cancer Res. 43:2512024. View Article : Google Scholar : PubMed/NCBI
|
|
285
|
Vanpouille-Box C, Alard A, Aryankalayil
MJ, Sarfraz Y, Diamond JM, Schneider RJ, Inghirami G, Coleman CN,
Formenti SC and Demaria S: DNA exonuclease Trex1 regulates
radiotherapy-induced tumour immunogenicity. Nat Commun.
8:156182017. View Article : Google Scholar : PubMed/NCBI
|
|
286
|
Wang Y, Chen Z, Liang K, Wang W, Hu Z, Mao
Y, Liang X, Jiang L, Liu Z and Ma Z: AGO2 mediates immunotherapy
failure via suppressing tumor IFN-gamma response-dependent CD8(+) T
cell immunity. Cell Rep. 44:1154452025. View Article : Google Scholar
|
|
287
|
Trovo M, Giaj-Levra N, Furlan C, Bortolin
MT, Muraro E, Polesel J, Minatel E, Tedeschi R, Filippi AR, Alongi
F and Ricardi U: Stereotactic body radiation therapy and intensity
modulated radiation therapy induce different plasmatic cytokine
changes in non-small cell lung cancer patients: A pilot study. Clin
Transl Oncol. 18:1003–1010. 2016. View Article : Google Scholar
|
|
288
|
Mills BN, Connolly KA, Ye J, Murphy JD,
Uccello TP, Han BJ, Zhao T, Drage MG, Murthy A, Qiu H, et al:
Stereotactic body radiation and interleukin-12 combination therapy
eradicates pancreatic tumors by repolarizing the immune
microenvironment. Cell Rep. 29:406–421 e5. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
289
|
Jiang K, Yin X, Zhang Q, Yin J, Tang Q, Xu
M, Wu L, Shen Y, Zhou Z, Yu H and Yan S: STC2 activates PRMT5 to
induce radioresistance through DNA damage repair and ferroptosis
pathways in esophageal squamous cell carcinoma. Redox Biol.
60:1026262023. View Article : Google Scholar : PubMed/NCBI
|
|
290
|
Berquist BR and Wilson DM III: Pathways
for repairing and tolerating the spectrum of oxidative DNA lesions.
Cancer Lett. 327:61–72. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
291
|
Mladenov E, Magin S, Soni A and Iliakis G:
DNA double-strand-break repair in higher eukaryotes and its role in
genomic instability and cancer: Cell cycle and
proliferation-dependent regulation. Semin Cancer Biol. 37-38:51–64.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
292
|
Zong C, Zhu T, He J, Huang R, Jia R and
Shen J: PARP mediated DNA damage response, genomic stability and
immune responses. Int J Cancer. 150:1745–1759. 2022. View Article : Google Scholar
|
|
293
|
Li T, Cheng H, Yuan H, Xu Q, Shu C, Zhang
Y, Xu P, Tan J, Rui Y, Li P and Tan X: Antitumor activity of cGAMP
via stimulation of cGAS-cGAMP-STING-IRF3 mediated innate immune
response. Sci Rep. 6:190492016. View Article : Google Scholar : PubMed/NCBI
|
|
294
|
Zhao X, Ma Y, Li J, Sun X, Sun Y, Qu F,
Shi X, Xie Y, Liu S, Ma Y, et al: The AEG-1-USP10-PARP1 axis
confers radioresistance in esophageal squamous cell carcinoma via
facilitating homologous recombination-dependent DNA damage repair.
Cancer Lett. 577:2164402023. View Article : Google Scholar : PubMed/NCBI
|
|
295
|
Liu W, Miao C, Zhang S, Liu Y, Niu X, Xi
Y, Guo W, Chu J, Lin A, Liu H, et al: VAV2 is required for DNA
repair and implicated in cancer radiotherapy resistance. Signal
Transduct Target Ther. 6:3222021. View Article : Google Scholar : PubMed/NCBI
|
|
296
|
Liu YG, Chen JK, Zhang ZT, Ma XJ, Chen YC,
Du XM, Liu H, Zong Y and Lu GC: NLRP3 inflammasome activation
mediates radiation-induced pyroptosis in bone marrow-derived
macrophages. Cell Death Dis. 8:e25792017. View Article : Google Scholar : PubMed/NCBI
|
|
297
|
Ortiz F, Acuna-Castroviejo D, Doerrier C,
Dayoub JC, Lopez LC, Venegas C, Garcia JA, Lopez A, Volt H,
Luna-Sanchez M and Escames G: Melatonin blunts the
mitochondrial/NLRP3 connection and protects against
radiation-induced oral mucositis. J Pineal Res. 58:34–49. 2015.
View Article : Google Scholar
|
|
298
|
Feldmeyer L, Keller M, Niklaus G, Hohl D,
Werner S and Beer HD: The inflammasome mediates UVB-induced
activation and secretion of interleukin-1beta by keratinocytes.
Curr Biol. 17:1140–1145. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
299
|
Anand PK, Malireddi RK and Kanneganti TD:
Role of the nlrp3 inflammasome in microbial infection. Front
Microbiol. 2:122011. View Article : Google Scholar : PubMed/NCBI
|
|
300
|
Francois A, Milliat F, Guipaud O and
Benderitter M: Inflammation and immunity in radiation damage to the
gut mucosa. Biomed Res Int. 2013:1232412013. View Article : Google Scholar : PubMed/NCBI
|
|
301
|
DeBo RJ, Lees CJ, Dugan GO, Caudell DL,
Michalson KT, Hanbury DB, Kavanagh K, Cline JM and Register TC:
Late effects of total-body gamma irradiation on cardiac structure
and function in male rhesus macaques. Radiat Res. 186:55–64. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
302
|
De Palma M, Coukos G and Hanahan D: A new
twist on radiation oncology: Low-dose irradiation elicits
immunostimulatory macrophages that unlock barriers to tumor
immunotherapy. Cancer Cell. 24:559–561. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
303
|
Ratikan JA, Micewicz ED, Xie MW and Schaue
D: Radiation takes its toll. Cancer Lett. 368:238–245. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
304
|
Curti B, Crittenden M, Seung SK, Fountain
CB, Payne R, Chang S, Fleser J, Phillips K, Malkasian I, Dobrunick
LB and Urba WJ: Randomized phase II study of stereotactic body
radiotherapy and interleukin-2 versus interleukin-2 in patients
with metastatic melanoma. J Immunother Cancer. 8:e0007732020.
View Article : Google Scholar : PubMed/NCBI
|
|
305
|
Ng SSW, Zhang H, Wang L, Citrin D and
Dawson LA: Association of pro-inflammatory soluble cytokine
receptors early during hepatocellular carcinoma stereotactic
radiotherapy with liver toxicity. NPJ Precis Oncol. 4:172020.
View Article : Google Scholar : PubMed/NCBI
|
|
306
|
McBride WH and Schaue D: In situ tumor
ablation with radiation therapy: Its effect on the tumor
microenvironment and anti-tumor immunity. Tumor ablation: Effects
on systemic and local anti-tumor immunity and on other
tumor-microenvironment interactions. Keisari Y: Springer;
Dordrecht: pp. 109–119. 2013, View Article : Google Scholar
|
|
307
|
Seung SK, Curti BD, Crittenden M, Walker
E, Coffey T, Siebert JC, Miller W, Payne R, Glenn L, Bageac A and
Urba WJ: Phase 1 study of stereotactic body radiotherapy and
interleukin-2-tumor and immunological responses. Sci Transl Med.
4:137ra742012. View Article : Google Scholar
|
|
308
|
Hughson AL, Hannon G, Salama NA, Vrooman
TG, Stockwell CA, Mills BN, Garrett-Larsen J, Qiu H, Katerji R,
Benoodt L, et al: Integrating IL-12 mRNA nanotechnology with SBRT
eliminates T cell exhaustion in preclinical models of pancreatic
cancer. Mol Ther Nucleic Acids. 35:1023502024. View Article : Google Scholar
|