Introduction
Prostate cancer is the second most common cancer
with the fifth highest age-standardized mortality rate in men
worldwide (1). Unlike localized
disease, which is curable via a number of therapeutic strategies,
advanced prostate cancer that has metastasized and is
hormone-resistant has limited therapeutic options that offer only
temporary disease control (2).
In 2005, the FDA approved docetaxel for the
treatment of metastatic castration-resistant prostate cancer after
two randomized clinical trials demonstrated improved survival over
then standard-of-care mitoxantrone and prednisone therapy (3,4).
Docetaxel chemotherapy has become standard treatment and the
building block for novel therapeutic approaches to metastatic,
hormone-refractory disease. Docetaxel acts by stabilizing
microtubules in mitotic spindles, directly inhibiting the cell
division process (5,6). Because docetaxel is cytotoxic to all
dividing cells, it induces myelosuppression and neutropenia at
therapeutic doses (7,8), limiting its therapeutic
potential.
Immunotherapies offer a potential means to enhance
the clinical efficacy of docetaxel by inducing antitumor immune
responses in conjunction with docetaxel's direct antitumor effects.
Several immunotherapies are currently under development or in use
in cancer treatment. The dendritic cell vaccine sipuleucel-T
(Provenge®) was approved by the FDA in 2010 after a
phase III trial showed a 38% increase in 3-year overall survival
with a median survival advantage of 4.1 months compared to placebo
in men with metastatic castration-resistant prostate cancer (CRPC)
(9). Clinical trials combining
vaccine therapy and docetaxel are in progress (10). A second even more recently
FDA-approved immunotherapeutic drug, ipilimumab
(Yervoy®), a humanized anti-CTLA-4 drug, showed improved
survival in melanoma patients and several trials to assess its
efficacy in prostate cancer have been done or are underway
(11–15). These promising immunotherapies
support the hypothesis that combining pharmacological agents with
novel immune therapies can lead to tumor suppression and survival
benefit.
Polysaccharide-K (PSK) is a pharmaceutical grade
extract of the mushroom Trametes versicolor used throughout
Asia as an adjunctive cancer treatment due to its reported direct
antitumor effects, antimetastatic actions and immune modulatory
properties (16). PSK has recently
been shown to have TLR2-agonist activities and to significantly
inhibit breast cancer growth in neu transgenic mice in a
CD8+ T cell- and NK cell-dependent manner (17). A role of NK cells in the antitumor
actions of PSK is further supported by results from an earlier
study showing that PSK lengthened tumor doubling times and
increased mean survival times in T cell-deficient, but not
NK-deficient mice bearing human prostate tumors (18). A number of randomized prospective
clinical trials testing oral administration of PSK combined with
chemotherapy demonstrated improved survival for lung (19,20),
breast (21), gastric (22,23),
and colorectal (24) cancers. PSK
in combination with docetaxel has shown enhanced antitumor activity
against human pancreatic (25) and
gastric (26) cancer cells, and in
murine xenograft models of gastric cancer (26,27).
These findings together suggest that combining PSK with docetaxel
chemotherapy may enhance docetaxel's tumor suppressive actions and
further suggest that PSK addition may increase T cell and NK
cell-dependent antitumor responses.
Despite these promising preliminary data, the
effects of oral administration of PSK on docetaxel-induced
antitumor responses in an immunocompetent prostate cancer setting
are as yet unknown. This study was conducted to investigate whether
combining orally administered PSK with docetaxel is safe and
whether it enhances docetaxel-induced therapeutic responses and
antitumor responses in TRAMP-C2 prostate tumor-bearing mice.
Materials and methods
Reagents
PSK was donated by Kureha Chemical Industry (Tokyo,
Japan) and docetaxel obtained from Sanofi-Aventis USA (Bridgewater,
NJ).
Antibodies and staining reagents
CD4 and CD8 primary Abs were obtained from Santa
Cruz Biotechnology (Santa Cruz, CA). Anti-Ki67 was obtained from
Dako (Copenhagen, Denmark). The TUNEL staining kit was obtained
from R&D Systems (Minneapolis, MN). The ABC Vector staining kit
(Vector Labs, Burlingame, CA) was used for fluorescent secondary
staining in some cases. Rabbit polyclonal IgG against cleaved
caspase-3 (Asp 175) (specific for mouse, rat and human cleaved
caspase-3) was obtained from Cell Signaling Technology (Beverley,
MA).
Murine prostate cancer model and
therapy
Mouse prostate tumor (TRAMP-C2) cells (American Type
Culture Collection, Manassas, VA) were injected orthotopically into
C57BL/6 mice (Charles River, Wilmington, MA) following a study
protocol approved by the University of Minnesota Institutional
Animal Care and Use Committee. This procedure was performed under
general anesthesia (isoflurane). Tumors were allowed to grow for
approximately two weeks. Tumor growth was monitored by palpation of
the prostate; established tumors present as hardened masses at the
injection site. After this period, mice were randomly assigned to
one of four treatment groups: 1) saline control; 2) docetaxel only;
3) PSK only; 4) docetaxel plus PSK. Control mice received saline
daily through oral gavage, as well as twice weekly saline
intraperitoneal (i.p.) injections. The docetaxel only treatment
group received docetaxel (5 mg/kg) injected i.p. twice weekly and
saline vehicle control daily by oral gavage. The PSK only treatment
group received PSK (300 mg/kg) daily by oral gavage and i.p.
injections of saline twice weekly. The docetaxel + PSK treatment
group received PSK (300 mg/kg) daily by oral gavage and docetaxel
(5 mg/kg) i.p. injections twice weekly. Injections were
administered in 100 μl volumes. Mice were weighed every other day.
Groups were treated for 11–13 days before mice were sacrificed and
specific organs harvested for end-point monitoring. Prostate tumors
were excised and weighed, and tumors and spleens were used to
determine concentration and activity of different immune cell
types. Excised tumors were also analyzed for proliferation and
apoptosis. Blood was also collected to determine white blood cell
counts and assay hepatic and renal function for safety
assessment.
Hematoxylin and eosin staining of
tissue
To confirm the presence of tumor, frozen sections of
tumor tissue (4 micron) were subjected to hematoxylin and eosin
staining. Sections were placed into distilled water and nuclei
stained with alum hematoxylin for 4 minutes (min), then rinsed with
water. Sections were then treated with 0.3% acid alcohol and
rinsed. Slides were then treated with eosin for 2 min, rinsed,
dehydrated, mounted and examined for tumor presence.
Immunohistochemistry for Ki67 and TUNEL
apoptosis assay
Unstained serial frozen sections of tumor were cut
to 4-micron thickness and placed into phosphate buffered saline
(PBS). Sections were exposed to 1–1,000 dilution of Ki67 (Dako) and
incubated for 1 hour (h) at 37°C. All slides were incubated with
the appropriate biotinylated secondary antibody: goat anti-rabbit
IgG (Vector), for 20 min at 37°C. The cells were then counted as
described below. Other nearby sections were used for
immuno-fluorescent staining to detect the presence of apoptosis
using the TUNEL apoptosis assay kit (R&D Systems) according to
the manufacturer's protocol. Briefly, slides were treated with
proteinase K for 30 min at 37°C and incubated with a terminal
deoxynucleotidyl transferase end labeling cocktail (terminal
deoxynucleotidyl transferase buffer, biotin-dUTP, and terminal
deoxynucleotidyl transferase at a ratio of 90:5:5) for 120 min at
37°C. This was followed by overlaying an avidin-FITC (green)
solution (50 μl) and incubating in the dark for 60 min at 37°C.
Determination of WBC counts
At the time of sacrifice, mice were sedated and
heart punctured with 25 gauge needle and 500–1,000 μl blood removed
for WBC analysis using a Coulter Counter (Beckman Coulter, Brea,
CA).
Liver and renal function analysis
To assess toxicity of the different treatments,
plasma samples obtained from mice (N=4) in each treatment group at
time of sacrifice (11–13 days post-treatment) were tested for
standard parameters of liver and renal function, including alanine
transaminase (ALT), aspartate aminotransferase (AST), creatinine
(Cr), blood urea nitrogen (BUN), albumin and total protein
levels.
Cytokine and cytolytic gene and Foxp3
mRNA expression
To assess expression of IFN-γ, IL-2, TNF-α, TGF-β,
perforin, granzyme B and FoxP3 genes, ribonucleic acid (RNA) was
isolated from tumors using the RNAqueous4PCR kit (Applied
Biosystems/Ambion, Austin, TX). The integrity of RNA was tested
using an Agilent Bioanalyzer (Agilent Technologies, Palo Alto, CA).
Complementary deoxyribonucleic acid (cDNA) was generated from 5 μg
of RNA using Superscript III reverse transcriptase (Invitrogen, San
Diego, CA) with oligo-dT as primers according to the manufacturer's
protocol. The cDNA (5 μl) diluted 1:40 was used as the template for
real-time PCR analysis. The primers and probes (FAM-MGB) for
TaqMan-based gene expression assays were purchased from Applied
Biosystems (Foster City, CA). Real-time PCR was performed in 384
well thin-wall PCR plates using ABI PRISM 7900HT under the
following conditions: initial denaturation at 95°C for 10 min,
followed by 40 cycles of denaturation at 95°C for 15 sec and a
combined annealing/extension step at 60°C for 1 min. Data analysis
was performed using SDS 2.21 (Applied Biosystems). The messenger
RNA (mRNA) expression level of the target gene was normalized to
β-actin using the ΔCT method. Level of
expression=2−ΔCT, where ΔCT=CT target
gene−CT actin. CT is the cycle threshold at which
the fluorescence signal crosses an arbitrary value.
Flow cytometry for immunophenotyping and
apoptosis analysis of spleen cells
After sacrifice, spleens were removed and each
spleen rendered to a single cell suspension using standard
techniques (28). Red blood cells
were lysed with hypotonic media (ACK lysing buffer, Lonza, Inc.
Walkersville, MD) for 3–5 min and then mononuclear cells were
washed with PBS two times. Approximately
105–106 cells were stained on ice with Fc
block (BD Pharmingen, San Diego, CA) for 15 min, followed by
addition of fluorochrome-labeled mAbs to specified cell surface
markers for immunophenotyping, and annexin V and propidium iodide
for apoptosis analysis. Cells were washed and resuspended and
analyzed by a FACSCanto flow cytometer and FlowJo software (BD
Biosciences, San Jose, CA).
Dual fluorescence immunohistochemistry
for caspase-3 and CD4/CD8
Sections of frozen tumors (6–8 from each group) were
cut to 4 micron thickness. Immediately prior to immunolabeling,
sections were re-fixed in cold acetone, equilibrated in PBS-saponin
and incubated with 5% normal goat (or donkey) serum for 1 h at
37°C. After washing, they were incubated with rabbit anti-caspase-3
(1:75) for 48–60 h at 4°C, followed by addition of goat anti-rabbit
IgG biotin (1:100; Jackson ImmunoResearch Laboratories, West Grove,
PA) for 1 h at 37°C. The anti-caspase-3 Ab detects only the large
fragment of activated caspase-3 (17–19 kDa) that results from
cleavage after Asp 175 and does not recognize other caspases.
Sections were routinely washed in excess PBS, pH 7.5, containing
0.3% saponin (Sigma-Aldrich, St. Louis, MO) at the end of each
incubation step, and the same buffer was used as a diluent in all
immunohistochemical reagents for caspase-3. These sections were
incubated with streptavidin Texas red (1:100; Jackson
ImmunoResearch Laboratories) for 1 h at 37°C. Each section was then
incubated with rat antibodies to either CD4 or CD8. Sections were
then washed with PBS and exposed to goat anti-rat secondary
antibodies and pig IgG-FITC (1:50; Jackson ImmunoResearch
Laboratories). Sections were then washed and prepared for
fluorescence microscopy. Slides were examined with a Nikon
TE2,000-U fluorescent microscope (Nikon Corp., Tokyo, Japan).
Images were digitized by Photoshop 8.0. Ten high powered fields
(×100) from 3–4 slides from each of 6–8 tumors per treatment group
were viewed for the presence of fluorescently labeled cells: red
(cleaved caspase-3), green (CD4 or CD8 in 2 separate slide sets) or
dual-expressing yellow. Numbers of positive cells were counted and
mean cell number ± standard error of the mean reported. The
individual counting the cells was blinded to the treatment group
from which sections were taken.
NK cytolytic activity
At the time of sacrifice, splenocytes were isolated
and assessed for NK cell cytolytic activity against YAC-1 tumor
cells by a standard flow cytometric assay. YAC-1 cells labeled with
3,3′-dioctadecyloxacarbocyanine [DiOC18(3)] purchased from Sigma were cultured
with splenocytes for 4 h in triplicate at the effector to target
cell (E:T) ratios of 50:1, 25:1, 12.5:1 and 6.25:1. After 4 h,
cells were stained with propidium iodide (PI) and washed prior to
flow cytometric analysis. Dead target cells were detected as
DiOC+PI+ cells. NK cell activity, defined as
percent specific cytotoxicity, was calculated at each E:T ratio
using the following formula: (% DiOC+, PI+
cells) Splenocytes+YAC-1 - (% DiOC+,
PI+ cells) YAC-1 alone for each replicate
sample. Data were analyzed for significant differences using a
linear mixed model to analyze data from 17 mice.
Statistical analysis
Continuous variables, unless otherwise noted, were
analyzed using the Student's t-test. Non-continuous variables
(tumor weights) were analyzed using the Mann-Whitney U test.
Statistical significance was set at p≤0.05.
Results
PSK enhances docetaxel-induced tumor
suppression and apoptosis of tumor cells
To determine if PSK enhances tumor suppression
induced by docetaxel, groups of mice (n=10) bearing established
TRAMP-C2 tumors were treated with saline control; subtherapeutic
doses of docetaxel (5 mg/kg i.p. twice weekly); PSK (300 mg/kg
orally by gavage once daily) or a combination of the docetaxel plus
PSK (following the same dosing regimens as single agent
treatments). A subtherapeutic dose of docetaxel was used to allow
observation of enhancing effects of the combination treatment,
consistent with previous studies that showed direct antitumor
effects of adding intraperitoneally administered PSK to docetaxel
treatment in mouse models of gastric cancer (26,27).
Doses of up to 1,000 mg/kg PSK have been used in other studies with
no reported toxicities (29). The
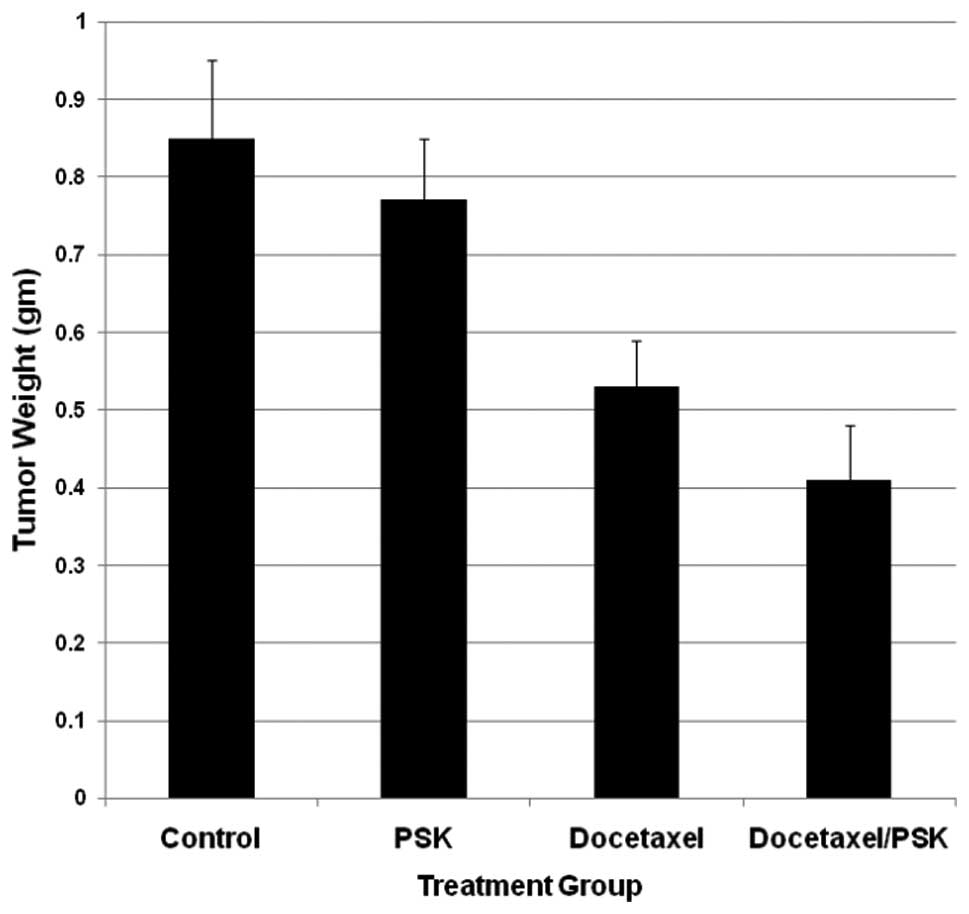
combination of docetaxel plus PSK resulted in significantly smaller
tumors, relative to saline control, docetaxel or PSK alone
(Fig. 1) (p<0.05), though
PSK-treated tumors were not significantly different than
control.
In order to assess potential mechanisms by which PSK
enhances the effect of docetaxel induced tumor regression, tumors
from treated mice were stained for Ki67 (proliferation) and annexin
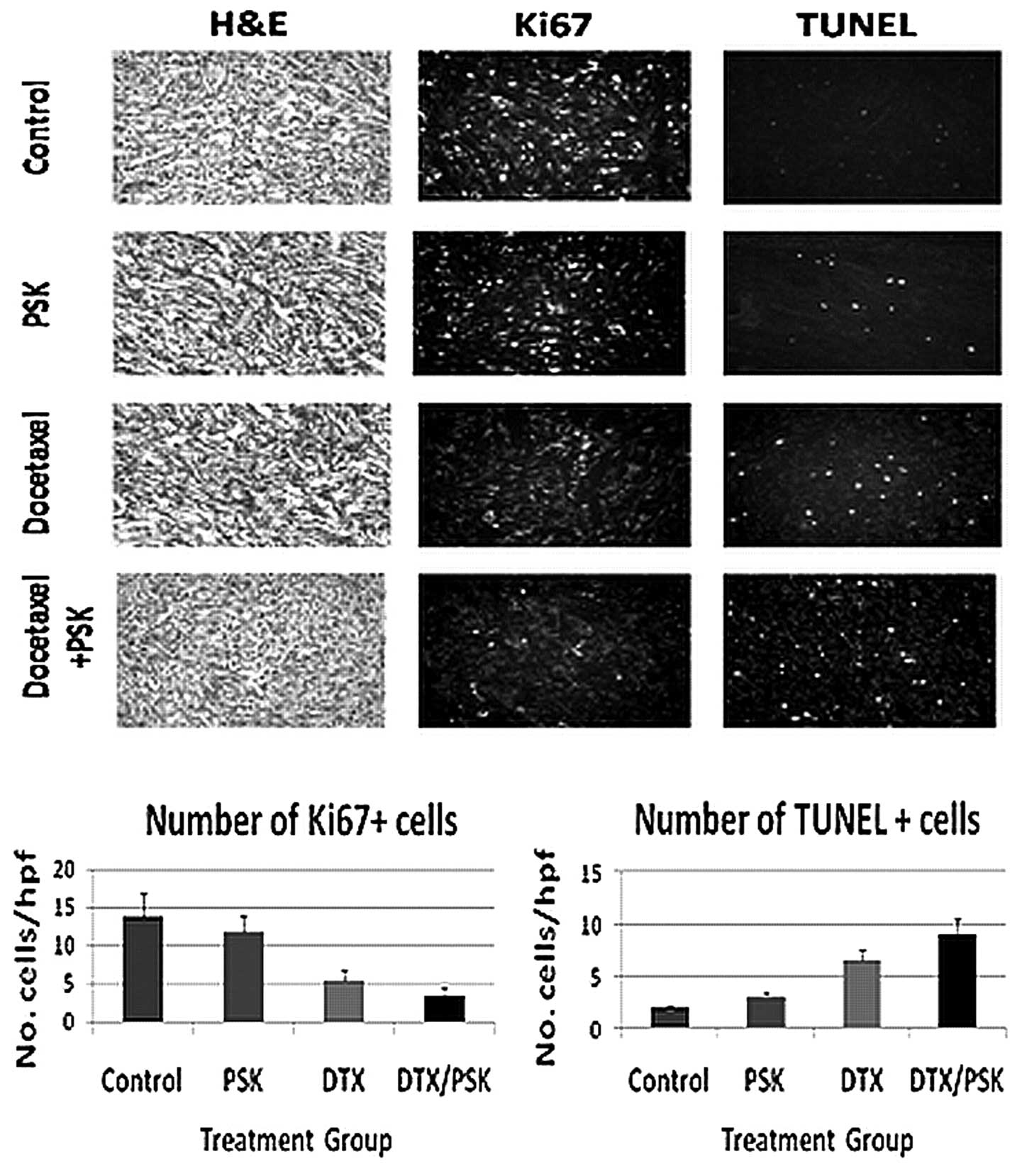
V (apoptosis). Combining PSK with docetaxel resulted in a reduction
of Ki67 positive cells compared to docetaxel alone (p<0.05), and
induced a 3-fold increase in the number of apoptotic, TUNEL
positive cells compared to therapy with PSK (p<0.01), and a
1.5-fold increase compared to docetaxel alone (p<0.05) (Fig. 2).
Combined docetaxel and PSK treatment does
not cause adverse effects
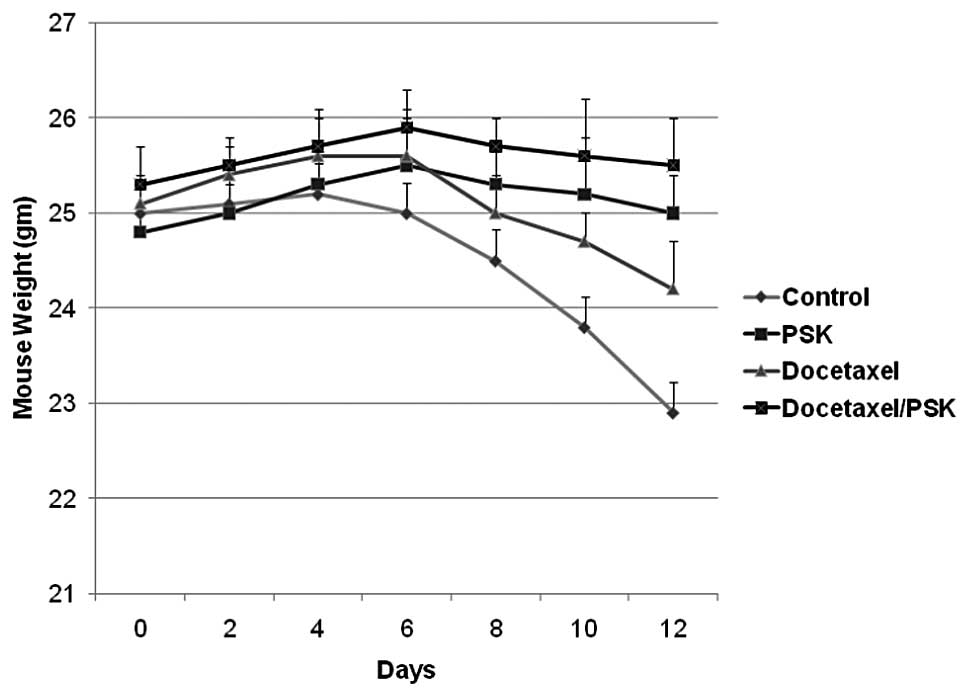
Given the apoptosis-inducing effects that we
observed, we investigated weight, hepatic and renal function in
mice treated with PSK or docetaxel alone or in combination. No
significant differences in liver and kidney function were observed
between the treatment groups (Table
I). Interestingly, weight was actually maintained with addition
of PSK to the docetaxel regimen as compared to either the control
or docetaxel groups (Fig. 3),
suggesting a protective effect of the combined treatment against
tumor-induced weight loss. Thus, no significant toxicity was
observed with the PSK and docetaxel combination therapy. We next
asked whether the combined therapy could allay observed
immunosuppressive effects of docetaxel.
 | Table IAnalysis of plasma parameters of
liver and renal function in treated TRAMP-C2 mice. |
Table I
Analysis of plasma parameters of
liver and renal function in treated TRAMP-C2 mice.
| Treatment | ALT (U/l) | AST (U/l) | Cr (mg/dl) | BUN (mg/dl) | Albumin (g/dl) | Total protein
(g/dl) |
|---|
| Control | 40±12 | 150±88 | 0.22±0.10 | 22.8±7.2 | 1.83±0.2 | 4.26±0.4 |
| DTX | 36±11 | 112±32 | 0.20±0.10 | 20.3±1.2 | 1.95±0.3 | 4.40±0.5 |
| PSK | 45±10 | 178±188 | 0.19±0.15 | 17.0±2.0 | 2.00±0.2 | 4.50±0.4 |
| PSK + DTX | 43±12 | 143±56 | 0.20±0.10 | 19.0±2.5 | 1.92±0.2 | 4.33±0.4 |
PSK reduces docetaxel-induced immune
suppression
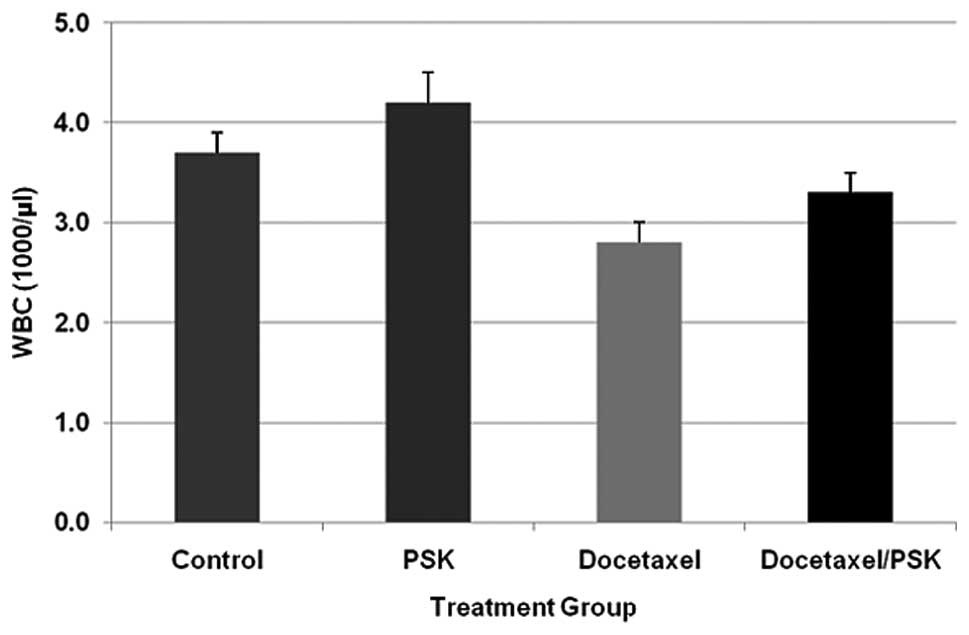
To determine the impact of PSK on docetaxel-induced
immunosuppression, WBC counts were determined. Docetaxel alone
induced a 25% reduction in WBC, and addition of PSK decreased this
suppressive effect by ~50%. The difference in WBC level observed in
response to PSK + docetaxel treatment compared to docetaxel alone
was statistically significant (p=0.03) (Fig. 4). Whether this effect is protective
(inhibits chemotherapy-induced lymphatic suppression) or
restorative (increases WBC levels after therapy) remains to be
determined.
Combining PSK with docetaxel enhances
tumor infiltrating lymphocyte (TIL) numbers
Since PSK enhanced antitumor responses in a Her2/neu
breast cancer mouse model (17),
we investigated whether combining PSK with docetaxel could enhance
measures of antitumor immunity in the TRAMP-C2 model. First we
asked whether numbers of TILs changed in response to the individual
and combined treatments. Tumor sections were stained for expression
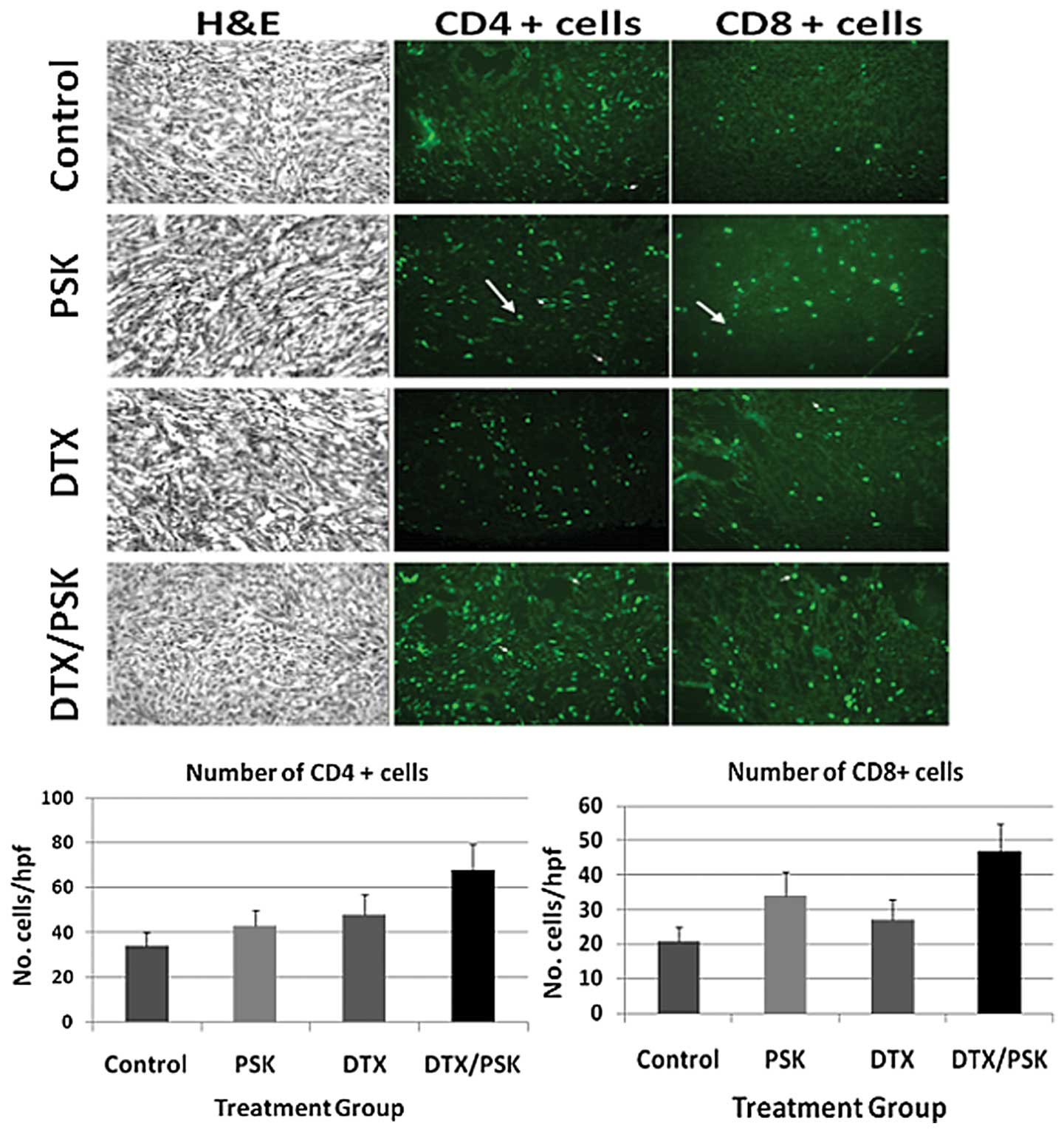
of tumor infiltrating CD4+ and CD8+ T cells.
The PSK and docetaxel treatments alone resulted in higher numbers
of TILs than in control mice, and the addition of PSK to docetaxel
increased both tumor infiltrating CD4+ T cells compared
to PSK or docetaxel alone (p<0.05) and CD8+ T cell
numbers compared to docetaxel alone (p<0.05) (Fig. 5), suggesting an enhancing effect of
the combined treatment on tumor infiltrating T cells.
Effects of PSK and docetaxel treatments
on mRNA expression of cytokine and cytolytic genes and FoxP3
Given the observation of PSK and docetaxel-induced
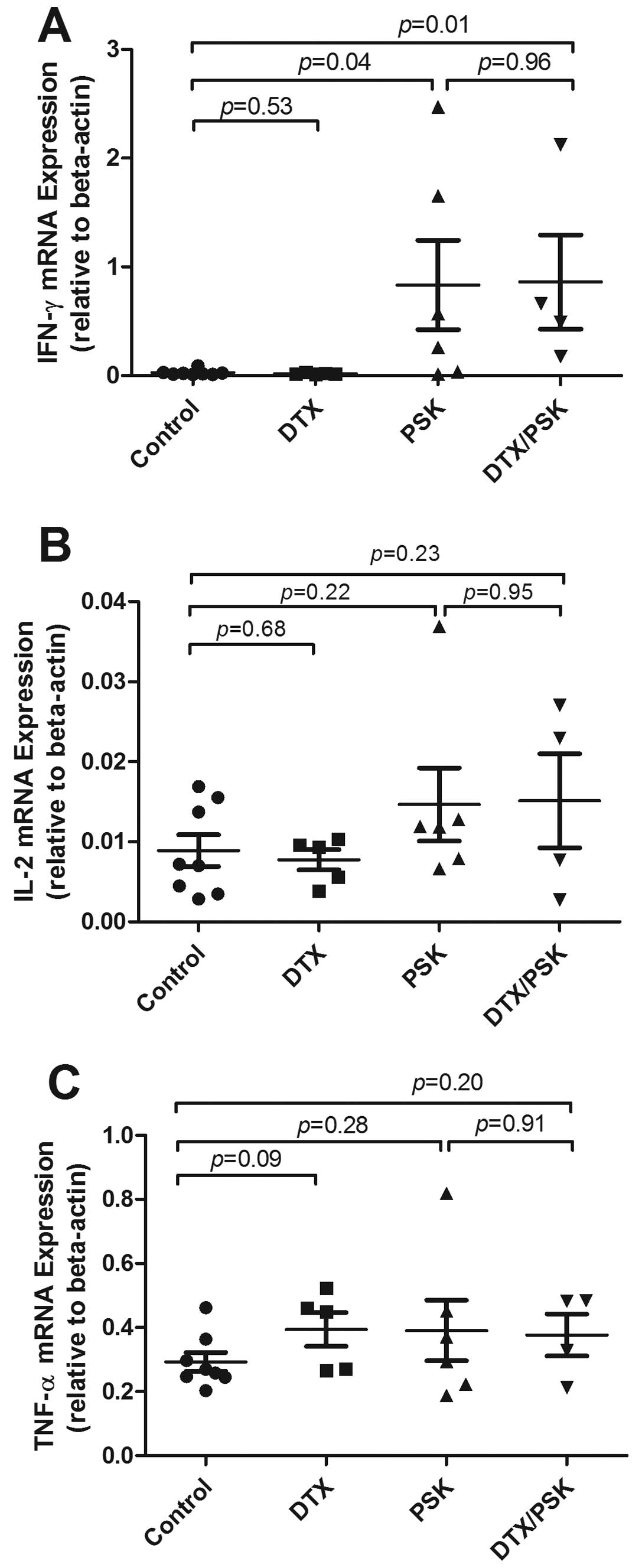
TIL enhancement, we further analyzed whether any changes in IFN-γ,
IL-2, TNF-α, TGF-β, perforin, granzyme B and FoxP3 mRNA expression
in the tumor micro-environment could be observed in response to the
treatments. TRAMP-C2 prostate tumors from the 4 different treatment
groups of mice were assessed using RT-PCR for the presence of mRNA
of cytokines and other antitumor immune response markers, as well
as for the T-regulatory cell marker, FoxP3. Docetaxel treatment
alone did not induce IFN-γ. Significant induction of interferon
gamma (IFN-γ) mRNA expression in TRAMP-C2 tumors occurred in mice
treated with PSK, either alone (p=0.04) or with docetaxel (p=0.01)
compared to saline control (Fig.
6A). No significant differences between the treatment groups
were observed for the other markers examined. However, mean IL-2
mRNA expression in response to combined PSK and docetaxel treatment
was higher than docetaxel treatment alone (Fig. 6B). Mean levels of tumor necrosis
factor-alpha (TNF-α) mRNA in response to docetaxel or PSK treatment
alone or in combination trended toward an increase compared to
control (p=0.09) (Fig. 6C). No
significant differences in perforin, granzyme B, TGF-β or FoxP3
mRNA expression were observed between treatment groups (data not
shown).
PSK and docetaxel treatments do not
affect splenic immune cell population percentages or apoptosis of
splenic T cells
Since PSK enhanced docetaxel-induced apoptosis of
tumor cells, we next investigated whether this effect was specific
to tumor cells or if PSK treatment also modulates apoptosis
induction of splenic immune cell sub-sets. Immunophenotyping was
used to analyze distribution of splenic lymphocyte sub-populations
alone and with annexin V, a marker of apoptosis. No significant
differences in distribution of splenic T cells, NK cells, B cells
or dendritic cells (DC) between treatment groups were observed
(data not shown). Further, levels of apoptotic cells among the
splenic sub-populations were not significantly different in
response to treatments compared to control. Thus, the PSK and
docetaxel treatments alone or combined do not appear to induce
splenic cell apoptosis.
We attempted to take a similar approach to assess
the effect of PSK and docetaxel treatments on apoptosis in
tumor-infiltrating T cells. We were unable to disassociate the
tumors specimens sufficiently to create single cell suspensions
needed for flow cytometry. As an alternative strategy, 8 frozen
tumor sections from mice in each treatment group were processed for
dual immunohistochemistry, staining for the caspase-3 apoptosis
marker and CD4 or CD8 T cell markers. Relatively little apoptosis
among CD4 or CD8 cells was observed, regardless of type of
treatment (data not shown).
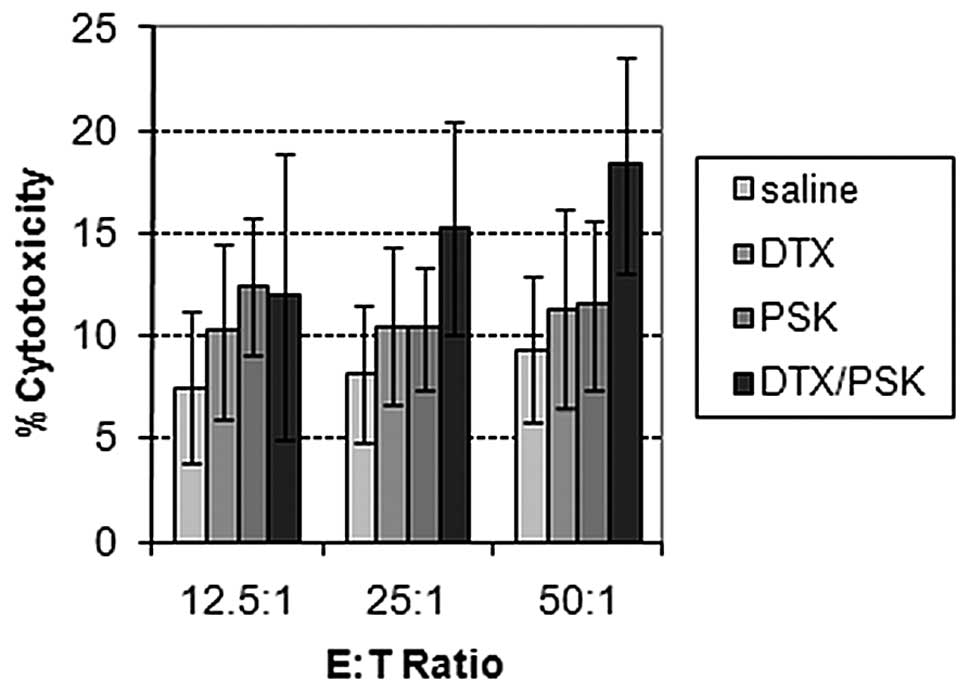
PSK combined with docetaxel enhances
splenic NK cell cytolytic activity
Spleens were collected and preserved from additional
mice from each treatment group to assess the influence of PSK alone
and combined with docetaxel on NK cell activity against YAC-1 tumor
target cells. PSK and docetaxel combination therapy was found to
have the greatest NK cell-inducing activity (Fig. 7). The splenic NK cell activity in
mice treated with the combination of PSK and docetaxel was
significantly higher compared to control mice at the 50:1 E:T ratio
(p=0.045).
Discussion
The TRAMP C2 tumor-bearing mouse model has
frequently been used as a model to develop novel prostate cancer
cytostatic and immunotherapy approaches (30–33).
In this study we used this model to assess the modulatory effects
of combination therapy with PSK and docetaxel treatment. Our
results demonstrate that the addition of PSK to docetaxel has
several beneficial effects. Addition of PSK to standard
chemotherapy (docetaxel) enhanced tumor regression, prostate
carcinoma apoptosis and proliferation compared to docetaxel alone.
The combination of PSK and docetaxel was also immunoprotective, in
that animals treated with the combination had less of a decrease in
WBC compared to those treated with docetaxel alone. This was
accompanied by an increase in the numbers of CD4+ and
CD8+ T cells within the tumor. PSK treatment alone or
with docetaxel significantly enhanced IFN-γ mRNA expression in
tumors compared to the near absence in tumors from control and
docetaxel groups. PSK combined with docetaxel also enhanced splenic
NK cell cytolytic activity against tumor target cells.
These results are consistent with published studies
showing the capacity of PSK to enhance gene expression of cytokines
(34) and to induce TLR2-dependent
activation pathways driving antitumor immune responses (17). PSP, a similar proteopolysaccharide
extract isolated from T. versicolor has been reported to
induce IFN-γ production and enhance NK cell tumoricidal activity in
experimental animal cancer models (35). IFN-γ enhancement by
polysaccharide-containing extracts from Ganoderma lucidum
(Reishi) and Grifola frondosa (Maitake) mushroom species has
also been reported (36–38). In this study we show that when
given in conjunction with traditional, front-line docetaxel
therapy, PSK administration was associated with smaller tumor size,
more adenocarcinoma apoptosis and higher amounts of TILs.
Collectively, these results support the clinical testing of the
combination of PSK with taxol based therapy, such as docetaxol.
Studies in other cancer models have reported
PSK-enhancing effects of docetaxel-induced tumor cell apoptosis
(26,27). These studies of apoptosis induction
mechanisms suggest that this apoptosis-enhancing effect may be a
result of PSK inhibiting NFκB-induced expression of survivin,
leading to a higher level of docetaxel-induced caspase-3 activation
in tumor cells (25,26). These studies did not assess the
effects of PSK addition to docetaxel on apoptosis induction in
immune cells, so whether these effects are tumor-specific is
unknown. In this study, we were unable to detect significant
changes in splenic immune cell sub-population percentages of total
mononuclear cells (data not shown). We therefore conclude that this
combination appears to not affect normal lymphocytes. Moreover, the
T cell apoptosis within tumor samples was low from all treatment
groups. Thus, the ability of PSK to protect against apoptosis of
tumor infiltrating lymphocytes was unclear. Docetaxel has been
reported to induce apoptosis in mitogen-activated human peripheral
blood mononuclear cells (PBMC) ex vivo without affecting Th1
cytokine production (39). Further
studies examining this question in more depth are needed to
determine whether the apoptosis-inducing effects of combined PSK
and docetaxel treatment are tumor-specific.
Immunomodulatory effects of docetaxel have also been
reported. Docetaxel is known to induce myelosuppression and reduce
WBC counts in a time and dose-dependent fashion (7,8,40)
and data from this study suggest that PSK may mitigate these
effects while enhancing antitumor responses. With respect to direct
immunological effects, docetaxel treatment in mice bearing
syngeneic transplanted tumors induced a significant increase in T
cell and NK cell infiltration into tumors (41). In this study, docetaxel induced an
increase in both CD4+ and CD8+ TILs over
saline treatment, an effect that was enhanced by the addition of
PSK. Markasz et al reported that docetaxel at therapeutic
concentrations effectively inhibited NK cell-mediated killing
without affecting NK cell viability (42). We did not observe inhibition of NK
cell lytic activity against tumor cells in response to docetaxel
treatment alone in the TRAMP-C2 tumor model. However, the ability
of PSK to enhance NK cell activity raises the possibility that PSK
administration may counter docetaxel-induced NK cell
inhibition.
The dose of PSK (300 mg/kg) used in combination with
docetaxel in these murine experiments is somewhat higher than has
been used in human clinical trials (~75 mg/kg) (43–45).
At these higher doses, we observed no toxicities of daily oral PSK
administration. Other investigators have tested oral administration
of up to 1,000 mg/kg PSK in a murine lung cancer model and found
fewer treatment-related deaths in PSK-treated animals (29). Furthermore, higher doses of
Trametes versicolor preparations (up to 9 gm daily) have
been well tolerated in a human trial in breast cancer patients
(Standish, unpublished data). Collectively, the results of this
study in a TRAMP-C2 murine model of human prostate cancer suggest
that the combination regimen of PSK and docetaxel warrants further
study in humans to determine if this combination therapy is of
clinical benefit in the treatment of advanced prostate cancer. A
phase Ib dose escalation clinical trial to determine the maximum
tolerated dose of PSK in combination with docetaxel is
underway.
Acknowledgements
The authors would like to acknowledge Kureha Corp.
for providing PSK and funding for this study, and Dr Masa Sasagawa
and Lauren Procopio for manuscript preparation assistance. This
research was also supported by the National Center for
Complementary and Alternative Medicine (NCCAM) at the National
Institutes of Health grants U19-AT019980 and U19-AT006028.
Abbreviations:
|
ALT
|
alanine transaminase
|
|
AST
|
aspartate amino-transferase
|
|
BUN
|
blood urea nitrogen
|
|
CDNA
|
complementary deoxyribonucleic
acid
|
|
Cr
|
creatinine
|
|
CRPC
|
castration-resistant prostate
cancer
|
|
IFN-γ
|
interferon-gamma
|
|
IgG
|
immunoglobulin G
|
|
IL-2
|
interleukin-2
|
|
i.p.
|
intraperitoneal
|
|
mRNA
|
messenger ribonucleic acid
|
|
NK
|
natural killer
|
|
PBMC
|
peripheral blood mononuclear cells
|
|
PCR
|
polymerization chain reaction
|
|
PSK
|
polysaccharide-K
|
|
TGF-β
|
transforming growth factor beta
|
|
TIL
|
tumor infiltrating lymphocytes
|
|
TLR
|
toll-like receptor
|
|
TNF-α
|
tumor necrosis factor alpha
|
|
TRAMP
|
transgenic adenocarcinoma of the mouse
prostate
|
|
WBC
|
white blood cell
|
References
|
1
|
Ferlay J, Shin HR, Bray F, Forman D,
Mathers C and Parkin DM: Estimates of worldwide burden of cancer in
2008: GLOBOCAN 2008. Int J Cancer. 127:2893–2917. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lassi K and Dawson NA: Update on
castrate-resistant prostate cancer: 2010. Curr Opin Oncol.
22:263–267. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Petrylak DP, Tangen CM, Hussain MH, et al:
Docetaxel and estramustine compared with mitoxantrone and
prednisone for advanced refractory prostate cancer. N Engl J Med.
351:1513–1520. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tannock IF, De Wit R, Berry WR, et al:
Docetaxel plus prednisone or mitoxantrone plus prednisone for
advanced prostate cancer. N Engl J Med. 351:1502–1512. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gueritte-Voegelein F, Guenard D, Lavelle
F, Le Goff MT, Mangatal L and Potier P: Relationships between the
structure of taxol analogues and their antimitotic activity. J Med
Chem. 34:992–998. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bissery MC, Guenard D, Gueritte-Voegelein
F and Lavelle F: Experimental antitumor activity of taxotere (RP
56976, NSC 628503), a taxol analogue. Cancer Res. 51:4845–4852.
1991.PubMed/NCBI
|
|
7
|
Mike S, Harrison C, Coles B, Staffurth J,
Wilt TJ and Mason MD: Chemotherapy for hormone-refractory prostate
cancer. Cochrane Database Syst Rev. CD005247.2006.PubMed/NCBI
|
|
8
|
Quartino AL, Friberg LE and Karlsson MO: A
simultaneous analysis of the time-course of leukocytes and
neutrophils following docetaxel administration using a
semi-mechanistic myelosuppression model. Invest New Drugs. Dec
14–2010.(E-pub ahead of print).
|
|
9
|
Kantoff PW, Higano CS, Shore ND, et al:
Sipuleucel-T immunotherapy for castration-resistant prostate
cancer. N Engl J Med. 363:411–422. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rozkova D, Tiserova H, Fucikova J, et al:
FOCUS on FOCIS: combined chemo-immunotherapy for the treatment of
hormone-refractory metastatic prostate cancer. Clin Immunol.
131:1–10. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Fong L, Kwek SS, O'Brien S, et al:
Potentiating endogenous antitumor immunity to prostate cancer
through combination immunotherapy with CTLA4 blockade and GM-CSF.
Cancer Res. 69:609–615. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hodi FS, O'Day SJ, McDermott DF, et al:
Improved survival with ipilimumab in patients with metastatic
melanoma. N Engl J Med. 363:711–723. 2010. View Article : Google Scholar
|
|
13
|
Small EJ, Tchekmedyian NS, Rini BI, Fong
L, Lowy I and Allison JP: A pilot trial of CTLA-4 blockade with
human anti-CTLA-4 in patients with hormone-refractory prostate
cancer. Clin Cancer Res. 13:1810–1815. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bristol-Myers Squibb. Phase 3 Study of
Immunotherapy to Treat Advanced Prostate Cancer. ClinicalTrialgov
[Internet] Bethesda (MD): National Library of Medicine (US); 2000,
[cited 20110824]. Available from: http://clinicaltrials.gov/ct2/show/NCT01057810.
NCT01057810.
|
|
15
|
Bristol-Myers Squibb. Study of
Immunotherapy to Treat Advanced Prostate Cancer. ClinicalTrialsgov
[Internet] Bethesda (MD): National Library of Medicine (US); 2000,
[cited 20110824]. Available from: http://clinicaltrials.gov/ct2/show/NCT00861614.
NCT00861614.
|
|
16
|
Kobayashi H, Matsunaga K and Oguchi Y:
Antimetastatic effects of PSK (Krestin), a protein-bound
polysaccharide obtained from basidiomycetes: an overview. Cancer
Epidemiol Biomarkers Prev. 4:275–281. 1995.PubMed/NCBI
|
|
17
|
Lu H, Yang Y, Gad E, et al: Polysaccharide
krestin is a novel TLR2 agonist that mediates inhibition of tumor
growth via stimulation of CD8 T cells and NK cells. Clin Cancer
Res. 17:67–76. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mickey DD, Bencuya PS and Foulkes K:
Effects of the immunomodulator PSK on growth of human prostate
adenocarcinoma in immunodeficient mice. Int J Immunopharmacol.
11:829–838. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hayakawa K, Mitsuhashi N, Saito Y, et al:
Effect of Krestin as adjuvant treatment following radical
radiotherapy in non-small cell lung cancer patients. Cancer Detect
Prev. 21:71–77. 1997.PubMed/NCBI
|
|
20
|
Hayakawa K, Mitsuhashi N, Saito Y, et al:
Effect of krestin (PSK) as adjuvant treatment on the prognosis
after radical radiotherapy in patients with non-small cell lung
cancer. Anticancer Res. 13:1815–1820. 1993.PubMed/NCBI
|
|
21
|
Iino Y, Yokoe T, Maemura M, et al:
Immunochemotherapies versus chemotherapy as adjuvant treatment
after curative resection of operable breast cancer. Anticancer Res.
15:2907–2911. 1995.PubMed/NCBI
|
|
22
|
Oba K, Teramukai S, Kobayashi M, Matsui T,
Kodera Y and Sakamoto J: Efficacy of adjuvant immunochemotherapy
with polysaccharide K for patients with curative resections of
gastric cancer. Cancer Immunol Immunother. 56:905–911. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Nakazato H, Koike A, Saji S, Ogawa N and
Sakamoto J: Efficacy of immunochemotherapy as adjuvant treatment
after curative resection of gastric cancer. Study Group of
Immunochemotherapy with PSK for Gastric Cancer. Lancet.
343:1122–1126. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sakamoto J, Morita S, Oba K, et al:
Efficacy of adjuvant immunochemotherapy with polysaccharide K for
patients with curatively resected colorectal cancer: a
meta-analysis of centrally randomized controlled clinical trials.
Cancer Immunol Immunother. 55:404–411. 2006. View Article : Google Scholar
|
|
25
|
Zhang H, Morisaki T, Nakahara C, et al:
PSK-mediated NF-kappaB inhibition augments docetaxel-induced
apoptosis in human pancreatic cancer cells NOR-P1. Oncogene.
22:2088–2096. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kinoshita J, Fushida S, Harada S, et al:
PSK enhances the efficacy of docetaxel in human gastric cancer
cells through inhibition of nuclear factor-kappaB activation and
survivin expression. Int J Oncol. 36:593–600. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yamasaki A, Shoda M, Iijima H, et al: A
protein-bound polysaccharide, PSK, enhances tumor suppression
induced by docetaxel in a gastric cancer xenograft model.
Anticancer Res. 29:843–850. 2009.PubMed/NCBI
|
|
28
|
Vremec D and Shortman K: Dendritic cell
subtypes in mouse lymphoid organs: cross-correlation of surface
markers, changes with incubation, and differences among thymus,
spleen, and lymph nodes. J Immunol. 159:565–573. 1997.
|
|
29
|
Katoh R, Takenoshita S, Shimizu Y, Tanaka
S, Yajima Y and Nagamachi Y: Effects on host of modified PVP
therapy combined with biological response modifiers in lung cancer.
Oncol Rep. 5:103–107. 1998.PubMed/NCBI
|
|
30
|
Castelo-Branco P, Passer BJ, Buhrman JS,
et al: Oncolytic herpes simplex virus armed with xenogeneic
homologue of prostatic acid phosphatase enhances antitumor efficacy
in prostate cancer. Gene Ther. 17:805–810. 2010. View Article : Google Scholar
|
|
31
|
Dzojic H, Loskog A, Totterman TH and
Essand M: Adenovirus-mediated CD40 ligand therapy induces tumor
cell apoptosis and systemic immunity in the TRAMP-C2 mouse prostate
cancer model. Prostate. 66:831–838. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Reddy KR, Guan Y, Qin G, Zhou Z and Jing
N: Combined treatment targeting HIF-1alpha and Stat3 is a potent
strategy for prostate cancer therapy. Prostate. 71:1796–1809. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yi H, Yu X, Guo C, Manjili MH, Repasky EA
and Wang XY: Adoptive cell therapy of prostate cancer using female
mice-derived T cells that react with prostate antigens. Cancer
Immunol Immunother. 60:349–360. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kato M, Hirose K, Hakozaki M, et al:
Induction of gene expression for immunomodulating cytokines in
peripheral blood mononuclear cells in response to orally
administered PSK, an immunomodulating protein-bound polysaccharide.
Cancer Immunol Immunother. 40:152–156. 1995. View Article : Google Scholar
|
|
35
|
Ng TB: A review of research on the
protein-bound polysaccharide (polysaccharopeptide, PSP) from the
mushroom Coriolus versicolor (Basidiomycetes: Polyporaceae). Gen
Pharmacol. 30:1–4. 1998. View Article : Google Scholar
|
|
36
|
Kodama N, Mizuno S, Nanba H and Saito N:
Potential antitumor activity of a low-molecular-weight protein
fraction from Grifola frondosa through enhancement of cytokine
production. J Med Food. 13:20–30. 2010. View Article : Google Scholar
|
|
37
|
Masuda Y, Inoue M, Miyata A, Mizuno S and
Nanba H: Maitake beta-glucan enhances therapeutic effect and
reduces myelosupression and nephrotoxicity of cisplatin in mice.
Int Immunopharmacol. 9:620–626. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Rubel R, Dalla Santa HS, Bonatto SJ, et
al: Medicinal mushroom Ganoderma lucidum (Leyss: Fr) Karst.
triggers immunomodulatory effects and reduces nitric oxide
synthesis in mice. J Med Food. 13:142–148. 2010. View Article : Google Scholar
|
|
39
|
Si MS, Imagawa DK, Ji P, et al:
Immunomodulatory effects of docetaxel on human lymphocytes. Invest
New Drugs. 21:281–290. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Suzuki Y and Tokuda Y: Docetaxel. Gan to
Kagaku Ryoho. 28:1363–1367. 2001.PubMed/NCBI
|
|
41
|
Mason K, Staab A, Hunter N, et al:
Enhancement of tumor radioresponse by docetaxel: involvement of
immune system. Int J Oncol. 18:599–606. 2001.PubMed/NCBI
|
|
42
|
Markasz L, Skribek H, Uhlin M, et al:
Effect of frequently used chemotherapeutic drugs on cytotoxic
activity of human cytotoxic T-lymphocytes. J Immunother.
31:283–293. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Ogden CL, Fryar CD, Carroll MD and Flegal
KM: Mean body weight, height, and body mass index, United States
1960–2002. Adv Data. 1–17. 2004.
|
|
44
|
Hirono M, Yoshinaka K, Matsuki K, et al:
Surgical treatment of Borrmann type 4 (diffuse) gastric cancer. Gan
No Rinsho. 30:717–723. 1984.PubMed/NCBI
|
|
45
|
Kidd PM: The use of mushroom glucans and
proteoglycans in cancer treatment. Altern Med Rev. 5:4–27.
2000.PubMed/NCBI
|