Introduction
Pancreatic cancer is the 8th and 9th leading cause
of cancer-related deaths worldwide in men and women. More than 50%
of patients are diagnosed with advanced disease and have
meta-static disease at presentation, 10 to 15% will be resectable,
and the remainder will be locally advanced unresectable disease as
well as an incidence of unrecognized metastases. The majority of
patients have a median survival with treatment of less than 6
months. Even for those seemingly fortunate enough to have
early-stage local and resectable disease, the 5-year survival is
only 20% after resection (1).
Therefore, pancreatic cancer is a refractory cancer having poor
prognosis.
MUC5AC is a member of the secreted mucin family and
is expressed as secretory mucin from goblet cells of the stomach,
airways and cervical secretion. These cover the epithelium and
provide a lubricating barrier that protects the mucosal surface. On
the other hand, it is overexpressed as a membrane-bound type in the
ductal region of human pancreatic cancer, while remaining
undetectable in the normal pancreas. However, there have been few
reports on the function of MUC5AC in pancreatic cancer (2–4).
The dominant view of polymorphonuclear neutrophils
(PMNs) is that they are of the most abundant and short-lived in
total circulating blood leukocytes and their cytoplasm has highly
developed cytomatrices and granules containing microbicidal
proteins and digestive enzymes. They provide the first-line of
defense against various infections, are potent effectors of
inflammation, and release chemotactic factors that lead to the
recruitment of non-specific and specific immune effectors (5). Studies on the immunological
mechanisms have focused mainly on the function of lymphocytes and
monocytes/macrophages as important mediators of the host response.
However, PMNs are increasingly recognized as an important effector
cell population for the rejection of malignant tumors in
vivo, including Fas ligand-mediated apoptosis,
antibody-dependent cellular cytotoxicity (ADCC), direct cell
killing by H2O2 and superoxide and
calprotectin (6,7).
The antitumor molecule TNF-related
apoptosis-inducing ligand (TRAIL) has been reported previously
(8,9) and is part of the TNF superfamily
member, is expressed in a broad range of cells including activated
T cells, B cells, natural killer cells, dendritic cells, monocytes
and activated neutrophils, and exerts great antitumor activity. It
is known that TRAIL is highly expressed on neutrophils selectively
inducing apoptosis. TRAIL exerts its activity by interacting with a
complex system of 2 death receptors (DRs) (DR4/TRAIL-R1 and
DR5/TRAIL-R2) and 3 decoy receptors (DcRs) [DcR1/TRAIL-R3,
DcR2/TRAIL-R4 and osteoprotegerin (OPG)]. Although these receptors
are characterized by high sequence homology in their extracellular
domains, only DR4/TRAIL-R1 and DR5/TRAIL-R2 contain a functionally
active death domain that allows an apoptotic response by TRAIL
stimulation (10,11). Some correlations between TRAIL
sensitivity and receptor expression have been reported in various
tumor cells. TRAIL only acts on cancer cells, it does not act on
normal cells. Although there is a theory that this reason is due to
a difference in expression levels of death receptors and decoy
receptors on these cells, the detailed mechanisms are not known. In
our previous study, when MUC5AC-expressing pancreatic cancer cells
were knocked down for MUC5AC by specific siRNA, cell survival,
proliferation, and morphology in vitro were the same as
control cells. However, their tumor growth in in vivo
xenograft studies was significantly lower than that of control
cells and most infiltrated leukocytes, particularly neutrophils and
B cells, were observed to be accumulated into its tumor challenged
site. Moreover, a greater number of antibodies against cancer cells
were found in the blood of SW1990/si-MUC5AC cell-challenged mice
than that of SW1990/si-mock cell-challenged mice. As a result, it
appeared that the tumorigenicity and growth of MUC5AC knockdown
cells were significantly lower than that of control cells (12).
In the present study, we assessed the mechanisms of
MUC5AC on these immune cells using the neutrophil depletion model
by anti-Gr-1 mAb and SCID mice in which B and T cells were
depleted. Furthermore, we focused on the anti-tumor effects of
neutrophils and hypothesized that MUC5AC was able to inhibit
apoptosis via TRAIL signaling pathways. To investigate the function
of MUC5AC in pancreatic cancer, we evaluated whether or not MUC5AC
knockdown would increase TRAIL responsiveness such as the
expression of TRAIL receptor and the effects on apoptosis signaling
pathways in pancreatic cancer cells.
Materials and methods
Reagents
Recombinant human TRAIL was purchased from PeproTech
GmbH (Paris, France). The tetrapeptide caspase inhibitor z-VAD-fmk
was obtained from R&D Systems (Minneapolis, MN). The rat
anti-mouse Gr-1 monoclonal antibody used for deplete murine
neutrophils was from BD Pharmingen Inc. (San Diego, CA). GeneJuice
used for siRNA transfection was purchased from Merck (Darmstadt,
Germany).
Animals
Specific-pathogen-free female BALB/c-nu/nu mice
(nude mice) and C.B17-scid/scid mice (SCID mice) purchased from
Charles River Japan Inc. (Kanagawa, Japan) were acclimatized and
then used in experiments at the age of six weeks. The experimental
design was approved by the Ethics Committee on Animal Experiments
of the Biomedical Research Laboratories of Kureha Corp., and mice
were treated in accordance with the guidelines of the committee.
All animals were allowed free access to sterilized CE-2 food
(Oriental Yeast, Tokyo, Japan) and sterilized tap water. Mice were
bred at 25±2°C, a humidity of 55±7%, laminar flow, and under a 12 h
light/12 h dark cycle at 150–300 lux. To maintain a uniform
environment, noise was carefully avoided and only experimental
staff and keepers were allowed into the animal room.
Cell culture conditions
SW1990 was cultured in Dulbecco’s modified Eagle’s
medium (DMEM; Invitrogen Corporation, San Diego, CA) supplemented
with 10% fetal bovine serum (FBS; Biowest, Nuaill, France), 50
IU/ml penicillin and 50 μg/ml streptomycin. Cells were grown
at 37°C with 5% CO2 in a humidified atmosphere and
passaged before they reached confluency using 0.25% (w/v) trypsin
solution containing 0.04% (w/v) EDTA.
Construction of siRNA-MUC5AC and
establishing the stable expression cell line
The MUC5AC siRNA target sequence
5′-TTTGAGAGACGAAGGATAC-3′ was cloned to generate a stable siRNA
expressing construct into the pSilencer 3.1-H1 neo vector
(Ambion Inc., Austin, TX) as described previously (12). SW1990 cells were transfected with
pSilencer/si-MUC5AC as a target or pSilencer/si-mock
as a control using GeneJuice according to the manufacturer’s
instructions. SW1990 cells were selected by culturing in the
presence of geneticin at 600 μg/ml. The efficiency of
MUC5AC-knockdown was tested through RT-PCR and FACS analysis (data
not shown).
Preparation of neutrophil-depleted
mice
Neutrophils were depleted from nude mice using
anti-Gr-1 mAb. Anti-Gr-1 mAb was treated at single doses of 100
μg/mice via i.p. injection on days -1, 4, 9, 14, 19, 24, 29,
34 and 39. The depletion of neutrophils was determined before tumor
cell implantation by a flow cytometer (13).
Tumorigenicity assay in xenograft
models
Cells were implanted subcutaneously (s.c.) at
1×107/mice on the flank of nude mice or SCID mice. Tumor
volumes were measured at least once a week. For the determination
of tumor volume, two bisecting diameters of each tumor were
measured by slide calipers and tumor volumes were calculated using
the following formula: tumor volume = length × (width)2
× 0.5236. Eight mice were used for each cell line. Tumor growth
curves were plotted as the mean volume ± standard error (SE).
Apoptosis assays
SW1990/si-MUC5AC cells and SW1990/si-mock cells
(2×103 cells/100 μl) were cultured in 96-well
plates in medium containing recombinant human TRAIL at fixed
concentrations. Cell proliferation was evaluated by the MTT assay
as previously described (12). The
pan-caspase inhibitor z-VAD-fmk was dosed at 10 μM before 1
h of TRAIL treatment.
Human apoptosis array
The expression profile of apoptosis-related proteins
was detected and analyzed using a Proteome Profiler™
(R&D Systems Inc.) according to the manufacturer’s
instructions. Briefly, protein lysates (500 μg) from
SW1990/si-MUC5AC cells and SW1990/si-mock cells were loaded onto an
array membrane blocked with blocking reagent. The membrane was
incubated overnight at 4°C, washed thrice with TBST, and then
incubated with a detection antibody cocktail for 1 h. After three
TBST washes, spots were visualized by a chemiluminescence assay and
the average density of duplicate spots was recorded.
Production of IL-8
SW1990/si-MUC5AC cells and SW1990/si-mock cells
(104 cells/100 μl) were cultured in 96-well
plates for 2 or 4 days. Culture supernatants were collected and
measured using a human IL-8 ELISA kit (RayBiotech Inc., Norcross,
GA), as directed by the manufacturer.
Statistical analysis
All data are expressed as the means ± SE.
Statistical significance was determined by the Student’s t-test.
P-values <0.05 were considered significant.
Results
Effects of MUC5AC on immunocytes
Previous studies have demonstrated that the tumor
growth of MUC5AC-knockdown cells is markedly suppressed when they
are subcutaneously implanted into a nude mouse. Furthermore,
neutrophils and B cells accumulated in the tumor (12). Thus, in the present study, we
investigated the effects of neutrophils and B cells on the tumor
growth of MUC5AC-expressing cells and the immune evasion mechanism
through MUC5AC.
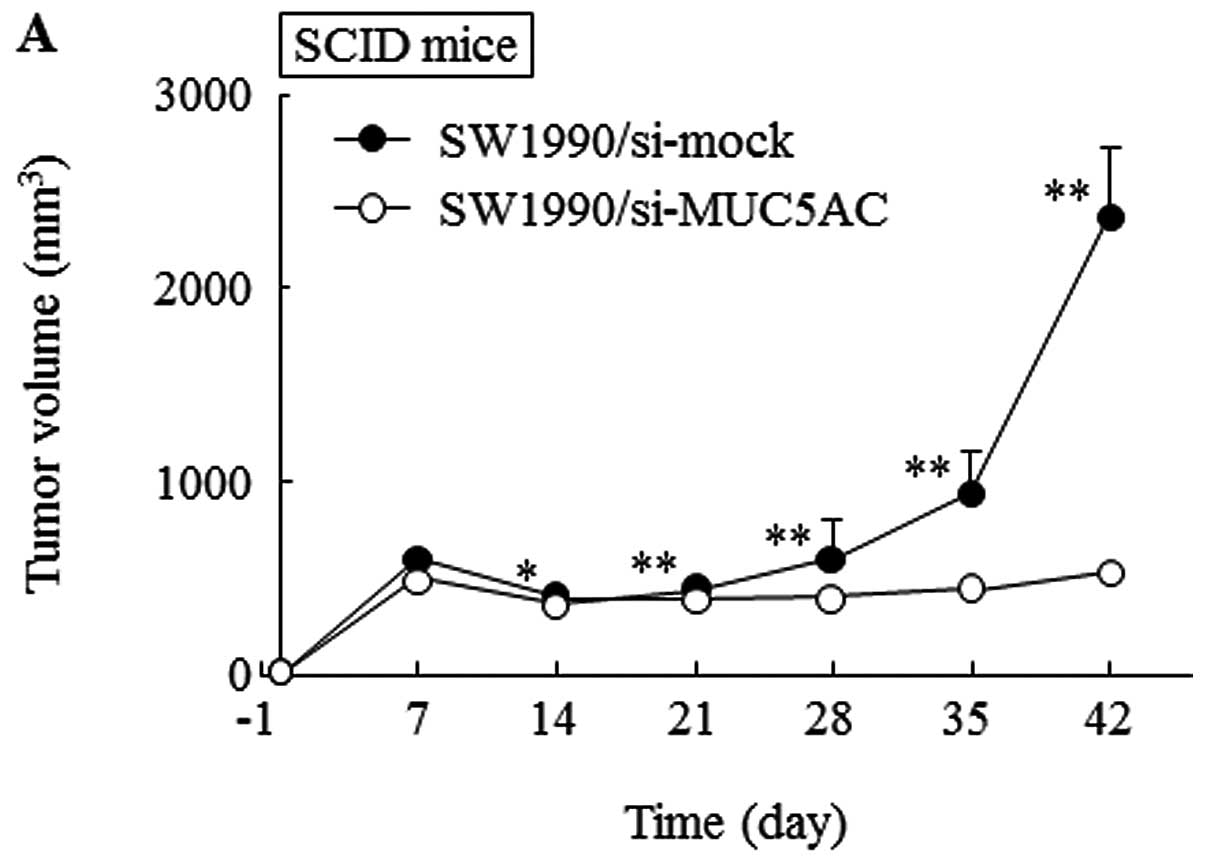
First, to examine the involvement of B cells on the
tumor growth of MUC5AC-expressing cells, we investigated the tumor
growth of MUC5AC-expressing and MUC5AC-knockdown cells in SCID
mice. SW1990/si-MUC5AC or SW1990/si-mock cells were implanted
subcutaneously into SCID mice at 1×107 cells/mice and
then tumor volume was assessed. If B cells act directly on the
antitumor effects of MUC5AC-knockdown-challenged mice,
MUC5AC-knockdown cells should grow comparably to MUC5AC-expressing
cells. However, the tumor volume of SW1990/si-mock cells increased
over time, while that of SW1990/si-MUC5AC cells barely increased
and little tumor growth was observed (Fig. 1A).
Subsequently, to examine the involvement of MUC5AC
on the antitumor effects of neutrophils, 100 μg of anti-Gr-1
antibody was administered once every five days to a nude mouse to
deplete neutrophils. Neutrophil depletion was confirmed by FACS
before tumor implantation (data not shown). SW1990/si-MUC5AC or
SW1990/si-mock cells were implanted subcutaneously at
1×107 cells/nude mouse, and then tumor volume was
assessed. The tumor growth of SW1990/si-MUC5AC cells was barely
observed in the presence of neutrophils (Fig. 1B). However, tumor growth recovered
to the same level as SW1990/si-mock cells when neutrophils were
depleted with an anti-Gr-1 antibody (Fig. 1C).
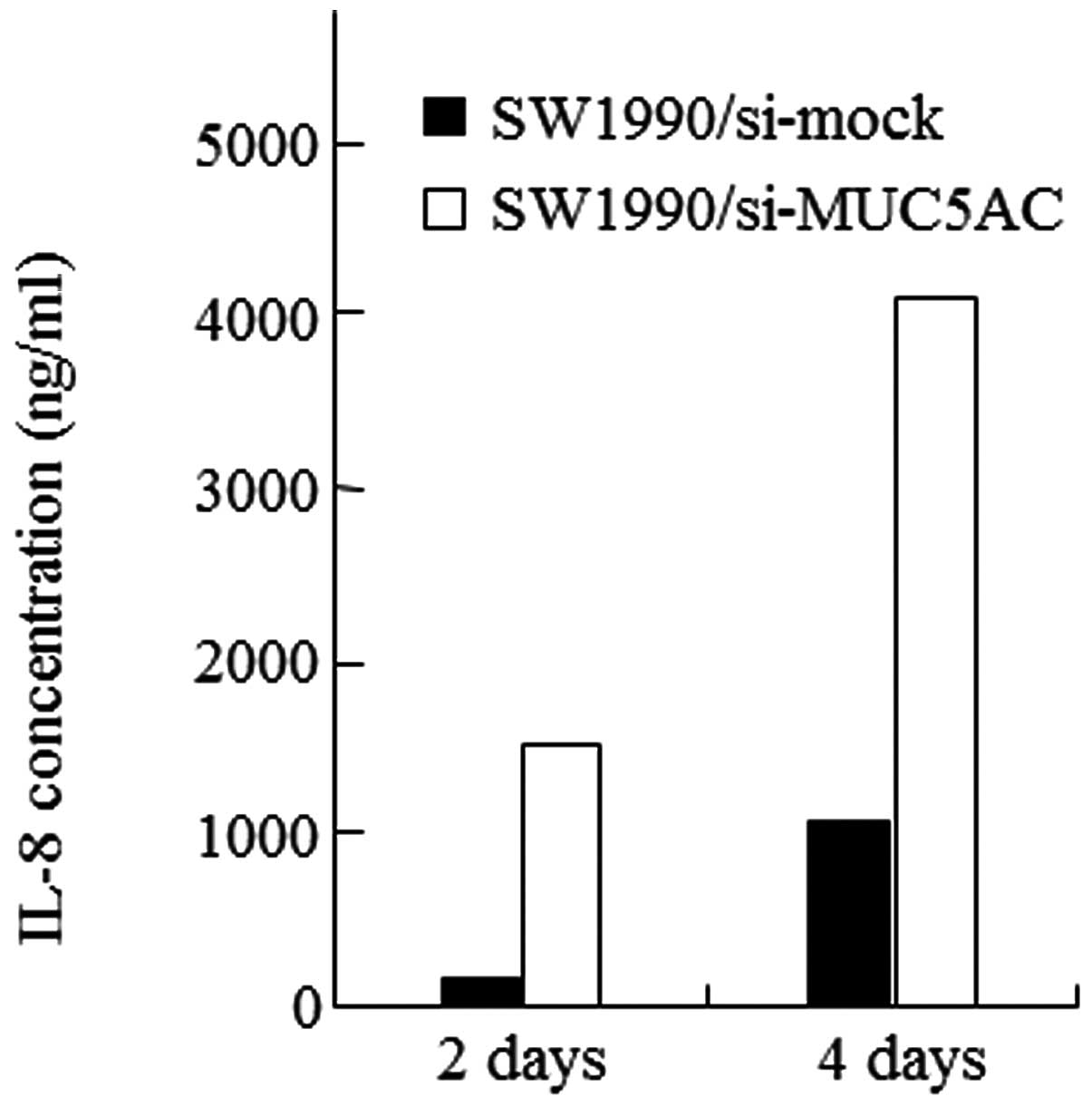
MUC5AC suppresses IL-8 production of
tumor cells
IL-8 produced by peripheral tissue cells and
activated neutrophils is involved in the migration of neutrophils
into the tissue (8). Hence, the
amounts of in vitro IL-8 production were determined in
SW1990/si-MUC5AC and SW1990/si-mock cells. IL-8 concentrations in
the supernatants of SW1990/si-MUC5AC and SW1990/si-mock cells were
1,529 and 179 ng/ml on day 2 and 4,100 and 1,094 ng/ml on day 4,
respectively. Suppressed MUC5AC expression significantly increased
IL-8 production (Fig. 2).
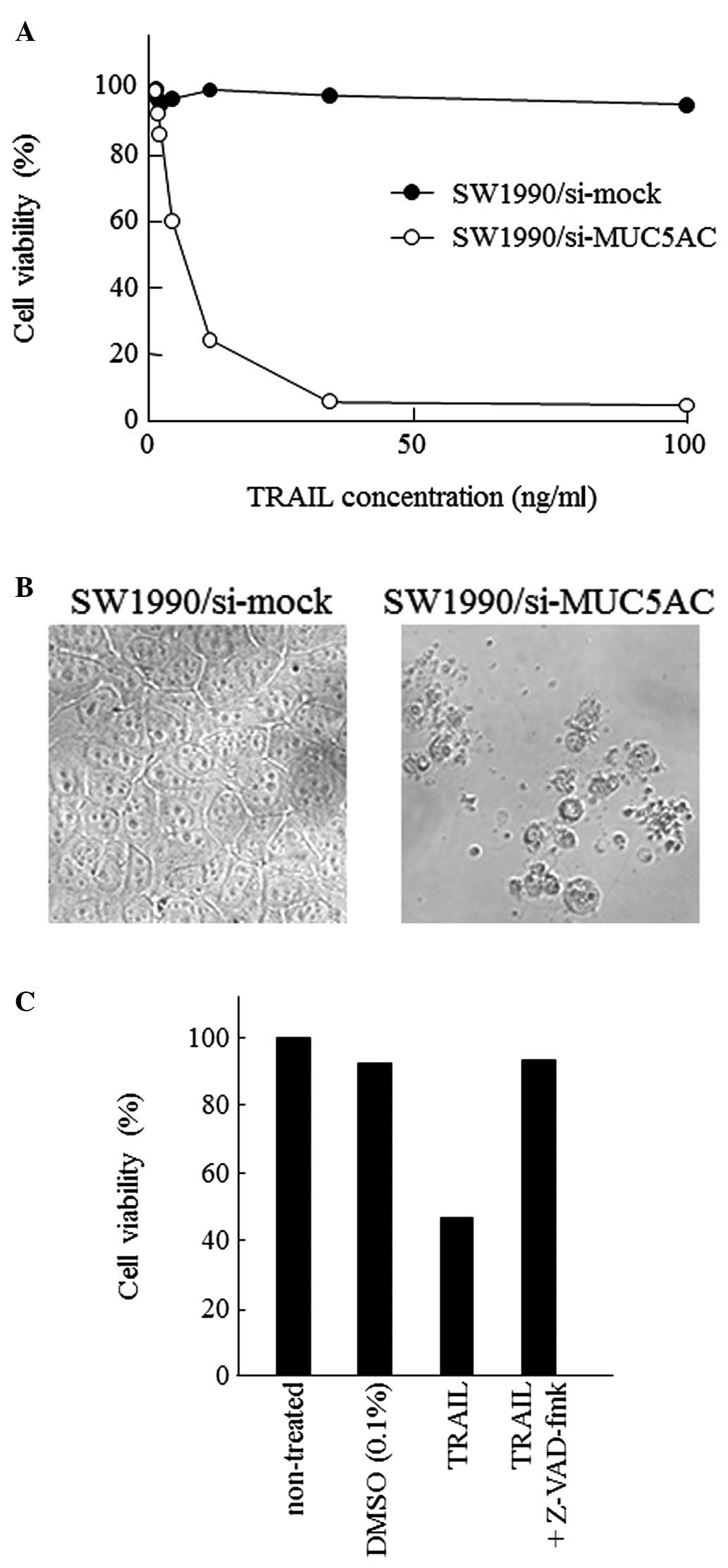
MUC5AC inhibits TRAIL-induced
apoptosis
It is known that activated neutrophils produced
TRAIL, which in turn, induced apoptosis of cancer cells via its
death receptors. Therefore, we evaluated cell viability after TRAIL
treatment by the MTT assay. The correlation between the dose of
TRAIL and cell growth inhibition is illustrated in Fig. 3A. Our previous reports show that no
difference in cell proliferation was observed between
SW1990/si-MUC5AC cells and SW1990/si-mock cells under normal
conditions in vitro(12).
After 12 h of TRAIL treatment, cell death was induced in
SW1990/si-MUC5AC cells in a dose-dependent manner. In this
condition, the IC50 value of TRAIL was 5.8 ng/ml. In
contrast, treatment of SW1990/si-mock cells at a concentration of
TRAIL up to 100 ng/ml resulted in <10% cell death. Concerning
cell morphologic changes, SW1990/si-MUC5AC cells were rounded and
floating in the medium, while SW1990/si-mock cells still formed a
typical epithelioid monolayer at a TRAIL concentration of 100 ng/ml
for 4 h (Fig. 3B). Consequently,
to examine whether caspases were involved in TRAIL-induced
apoptosis in SW1990/si-MUC5AC cells, either the pan-caspase
inhibitor z-VAD-fmk (10 μM) or a vehicle control was added
to the culture 1 h before TRAIL treatment (10 ng/ml, 4 h). The
results indicated that z-VAD-fmk completely blocked TRAIL-induced
apoptosis and caspase activation is a required signal event for
TRAIL-induced apoptosis. As shown in Fig. 3C, the percentage of cell viability
in SW1990/si-MUC5AC cells by the DMSO vehicle control was 92 and
TRAIL treatment was 47% in SW1990/si-MUC5AC cells. In contrast,
pretreatment of SW1990/si-MUC5AC cells with z-VAD-fmk followed by
TRAIL treatment significantly increased cell viability to 93%.
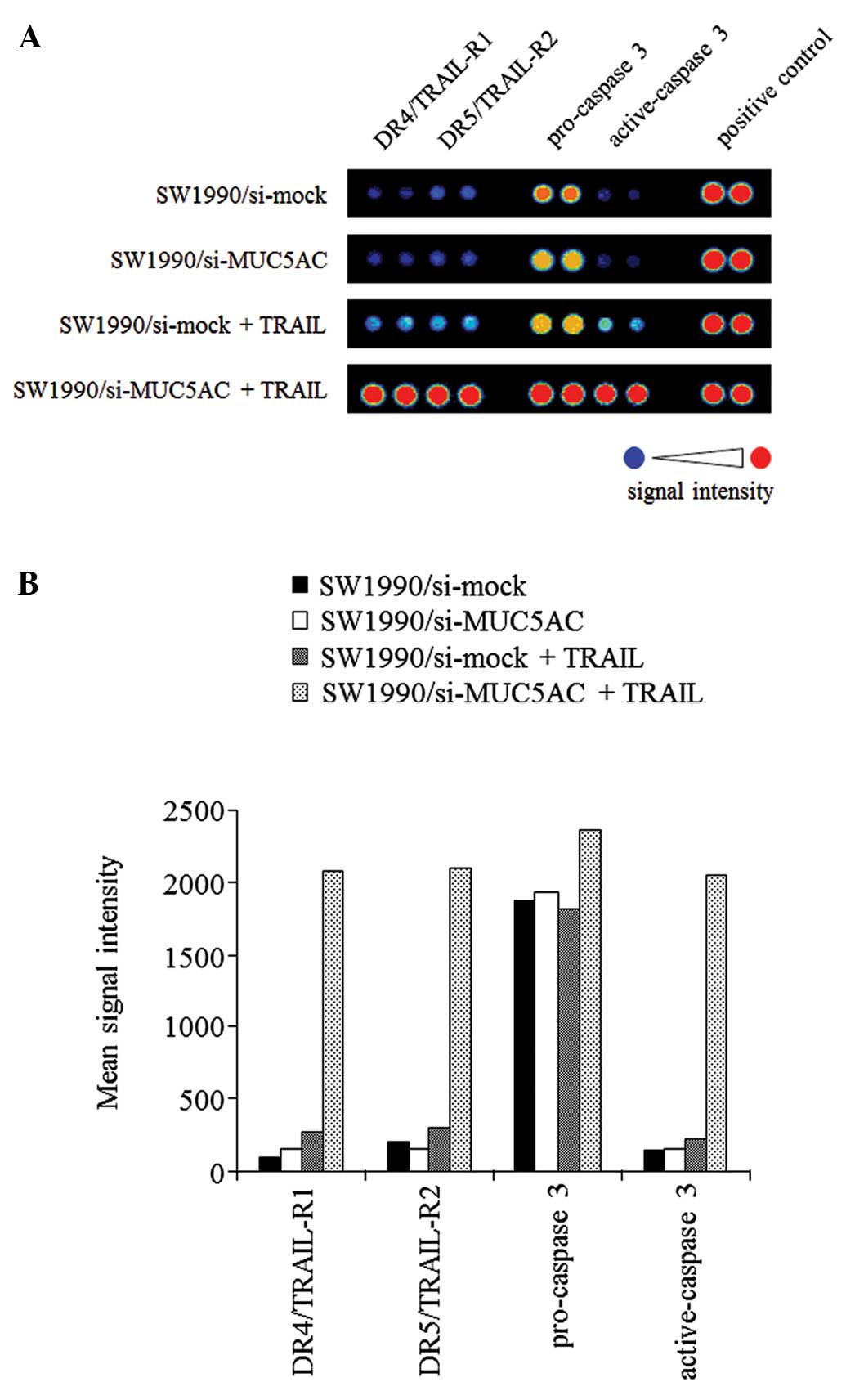
Analysis of the apoptosis suppression
mechanism of MUC5AC using an apoptosis array
As described above, MUC5AC knockdown with siRNA
induced apoptosis mediated by TRAIL. Then, to examine differences
in the apoptotic signal between SW1990/si-MUC5AC and SW1990/si-mock
cells, protein levels relating to apoptosis after the addition of
TRAIL to the cells were analyzed using an apoptosis array. Cells
were treated with 10 ng/ml of TRAIL for 4 h. The signal intensities
in the apoptosis array (Fig. 4A)
were digitized and normalized for the positive control value
(Fig. 4B). The vehicle control for
SW1990/si-MUC5AC cells showed weak expression of TRAIL receptors
(DR4/TRAIL-R1 and DR5/TRAIL-R2) and active-caspase 3 and strong
expression of pro-caspase 3 (active-caspase 3 precursor). The same
expression patterns were observed for SW1990/si-mock cells.
However, the addition of TRAIL induced an enhancement in the
expression of DR4/TRAIL-R1 and DR5/TRAIL-R2 in SW1990/si-MUC5AC
cells. Additionally, a significant increase in active-caspase 3
expression was observed. On the other hand, TRAIL had no effect on
DR4/TRAIL-R1 and DR5/TRAIL-R2 expressions in SW1990/si-mock
cells.
Discussion
Mucins are classified generally into two groups: a
secretory type and membrane-bound type. Secretory mucins include
MUC2, MUC5AC, MUC5B and MUC6. These mucins are called 11p15.5 mucin
because they are located on human chromosome 11p15.5 and are
structurally similar. On the other hand, membrane-bound mucins
include MUC1, MUC3A, MUC3B, MUC4, MUC11, MUC12 and MUC13. All these
mucins have transmembrane domains. MUC3A, MUC3B, MUC11 and MUC12
are located on chromosome 7q22, while MUC4 and MUC13 are located on
chromosome 3q29. MUC7 and MUC8 belong to neither of the above
groups and have no mutual similarity (3,14).
The core protein of a mucin molecule has a characteristic
repetitive sequence. The repetitive sequence, rich in Thr and Ser,
is frequently bound to a mucin-type sugar chain. Mucin-type sugar
chains generally consist of N-acetylgalactosamine,
N-acetylglucosamine, galactose, fucose and sialic acid. Sugar
chains account for 50–80% or above of the molecular weight of
mucin, giving it various properties including viscosity, a
water-holding capacity and proteinase resistance. Thus, the sugar
chain structure of mucin, which is varied quantitatively and
qualitatively with malignant transformations, is likely to be
involved in altered adhesion during metastasis and evasion from
immune cells. However, the detailed mechanisms remain unclear.
We established MUC5AC-knockdown cells using siRNA
and have previously reported analysis of the functions of MUC5AC in
cancer cells (12). A stable human
pancreatic cancer cell line, SW1990/si-MUC5AC, obtained by
introducing si-MUC5AC into a parent SW1990, was almost equal to the
control cells, SW1990/si-mock, regarding in vitro
morphology, proliferation and infiltration capacity. However, an
in vivo subcutaneously implanted model yielded results
completely different between SW1990/si-MUC5AC and SW1990/si-mock
cells. SW1990/si-mock cells caused continuous rapid tumor growth,
whereas SW1990/si-MUC5AC cells caused only slight tumor growth when
they were subcutaneously implanted into nude mice. Furthermore, the
amounts of immune cells, including neutrophils and B cells, existed
in the tumor derived from SW1990/si-MUC5AC cells. Tumor-specific
antibodies also existed in the serum. This suggests that
MUC5AC-expressing cells may suppress immune cells to evade the
immune system, thereby playing an important role in creating an
environment to facilitate cancer cell survival.
Neutrophils are characterized by immunity against
bacteria, such as direct phagocytosis of bacteria and
opsonophagocytosis of encapsulated bacteria. According to previous
reports, neutrophils also have antitumor effects. The antitumor
effects of neutrophils are reportedly caused by a number of
mechanisms including ADCC activity through a tumor-specific
antibody, apoptosis induction mediated by TRAIL (15–19).
Furthermore, inhibition of tumor growth by IL-12 was reduced when
neutrophils were depleted in a mouse model of prostate cancer
(20). PMNs are the predominant
effector cell population for the killing of breast cancer cells in
the presence of HER-2/neu monoclonal antibody (6). In addition, PMN-mediated ADCC has
been reported to contribute to the efficacy of the antitumoral
antibody rituximab and trastuzumab (13,21,22).
Additionally, TRAIL is expressed at significantly
higher levels in neutrophils than in other immune cells. TRAIL was
isolated in 1995 as a cytokine that induces apoptosis and has a
TNF-family analogous sequence (23). TRAIL is ∼30 kDa type II cell
surface protein consisting of 281 amino acids and is about 28%
homologous to the Fas ligand. TRAIL induces apoptosis in various
malignant tumors, but is non-toxic to normal cells. These functions
of TRAIL may lead to the promise of a molecular target agent. TRAIL
receptors are classified into TRAIL-R1 (death receptor 4, DR4);
TRAIL-R2 (death receptor 5, DR5); TRAIL-R3 (decoy receptor 1,
DcR1); TRAIL-R4 (decoy receptor 2, DcR2) and osteoprotegerin (OPG).
DRs 4 and 5 have an intracellular death domain to induce apoptosis
through initiator and effector caspases, whereas DcRs 1 and 2 are
decoy receptors that do not induce apoptosis (10). Differences in TRAIL receptor
expressions possibly result in apoptosis induction only in
malignant tumors. However, its detailed mechanism remains
unclear.
The present study aimed to identify the immune cells
on which MUC5AC acts using an immune cell-depleted animal model and
analyze the suppression mechanism. Then, we investigated the tumor
growth of MUC5AC-knockdown cells using nude mice depleted of
neutrophils with an anti-Gr-1 antibody and SCID mice depleted of B
and T cells.
To examine whether B cells act directly on the
rejection of implanted MUC5AC-knockdown cells, we investigated the
tumor growth of MUC5AC-knockdown and MUC5AC-expressing cells in
SCID mice. The growth of MUC5AC-knockdown cells did not recover
(Fig. 1A). In SCID mice, an
investigation in the double implantation system demonstrated that
the tumor growth of MUC5AC-expressing cells was increased
irrespective of whether a primary tumor was present or absent
(unpublished observation). However, such growth was not observed in
nude mice (unpublished observation). This suggests that MUC5AC
suppresses memory B-cell immune reactions that function during
prolonged antitumor reactions or cell metastasis.
In an experiment with neutrophil-depleted model
mice, the tumor growth of MUC5AC-knockdown cells, barely observed
in the presence of neutrophils, was recovered to the same level as
MUC5AC-expressing cells when neutrophils were depleted (Fig. 1C). This suggests that MUC5AC plays
an important role in directly suppressing the antitumor effects of
neutrophils. Additionally, IL-8 production, barely observed in
MUC5AC-expressing cells, was significantly increased in
MUC5AC-knockdown cells (Fig. 2).
According to these results, neutrophil infiltration into a tumor
inducing by IL-8 was found to exert antitumor effects. However, the
mechanism by which MUC5AC-knockdown increases IL-8 production by
tumor cells still remains unknown and should be investigated in a
future study.
An apoptosis array was employed to examine why
MUC5AC-expressing cells differed from MUC5AC-knockdown cells in
their susceptibilities to apoptosis mediated by TRAIL. DR4/TRAIL-R1
and DR5/TRAIL-R2 were weakly expressed in the routine cultures of
both cells. However, the addition of TRAIL increased the
expressions of DR4/TRAIL-R1, DR5/TRAIL-R2 and active-caspase 3
(effector caspase) in MUC5AC-knockdown cells (Fig. 4). Future studies will be aimed at
investigating how TRAIL increases death receptor expressions in
MUC5AC-knockdown cells.
The caspase family of proteases is the ultimate
effector of programmed cell death. Under ordinary circumstances,
caspases are kept in check by the inhibitor of apoptosis proteins
(IAPs) such as cIAP1, cIAP2, XIAP, NAIP, livin/ML-IAP,
BRUCE/Apollon and survivin, which bind to and inactive caspases
until they are needed. Caspases are overexpressed in tumors, but
IAPs likewise are overexpressed. Therefore, failure to activate
caspases could create resistance to apoptosis (24,25).
Several studies show that MUC4 and ErbB2 are coexpressed in some
tumor types such as the breast, non-small cell lung cancer, and
pancreas. The possibility that MUC4 could elicit its anti-apoptotic
effects by ErbB2, engaging a variety of signaling cascades to
elicit cellular responses, such as proliferation and survival, has
been suggested (26,27). Prostaglandin E2
(PGE2) synthesized by cyclooxygenase-2 (COX-2),
overproduced in various malignancies, has also been reported to be
associated with anti-apoptotic effects and increase survivin
expression (28,29). It is believed that MUC5AC may have
promoted the expression or action of IAPs in some way or MUC5AC may
have formed a complex with a certain molecule and promoted tumor
growth via suppression of tumor cell apoptosis.
In the present study, we established
MUC5AC-knockdown cells using siRNA to elucidate the functions of
MUC5AC, whose expression is increased in pancreatic cancer. IL-8
production was promoted in MUC5AC-knockdown cells. In addition,
cell death was induced by TRAIL through the apoptosis pathway,
suggesting reduced tumorigenicity in vivo. It was suggested
that induction of neutrophil migration is weak in normal
MUC5AC-producing pancreatic cancer cells because IL-8 production is
low and apoptosis induction by neutrophil-derived TRAIL is blocked
due to the presence of MUC5AC, and these conditions promote
pancreatic cancer cell proliferation and growth in vivo,
being involved in aggressive tumor formation. Our observations
suggest that the very potent anti-apoptotic effects of MUC5AC allow
tumor cells to escape key barriers to tumor progression. These
studies add to the knowledge on the significance of MUC5AC
expression in cancer cells.
Acknowledgements
This study was conducted at Kureha
Corporation, Tokyo and Osaka City University, Osaka, Japan.
Hirotaka Hoshi, Motoyuki Uchida and Hiroko Iijima are employees of
Kureha Corporation, but the study presents no conflict of
interest.
References
|
1
|
Wang Z, Song W, Aboukameel A, Mohammad M,
Wang G, Banerjee S, Kong D, Wang S, Sarkar FH and Mohammad RM:
TW-37, a small-molecule inhibitor of Bcl-2, inhibits cell growth
and invasion in pancreatic cancer. Int J Cancer. 123:958–966. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sheehan JK, Brazeau C, Kutay S, Pigeon H,
Kirkham S, Howard M and Thornton DJ: Physical characterization of
the MUC5AC mucin: a highly oligomeric glycoprotein whether isolated
from cell culture or in vivo from respiratory mucous secretions.
Biochem J. 347:37–44. 2000. View Article : Google Scholar
|
|
3
|
Hollingsworth MA and Swanson BJ: Mucins in
cancer: protection and control of the cell surface. Nat Rev Cancer.
4:45–60. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ho JJL, Crawley S, Pan PL, Farrelly ER and
Kim YS: Secretion of MUC5AC mucin from pancreatic cancer cells in
response to forskolin and VIP. Biochem Biophys Res Commun.
294:680–686. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tecchio C, Huber V, Scapini P, Calzetti F,
Margotto D, Todeschini G, Pilla L, Martinelli G, Pizzolo G,
Rivoltini L and Cassatella MA: IFNalpha-stimulated neutrophils and
monocytes release a soluble form of TNF-related apoptosis-inducing
ligand (TRAIL/Apo-2 ligand) displaying apoptotic activity on
leukemic cells. Blood. 103:3837–3844. 2004. View Article : Google Scholar
|
|
6
|
Stockmeyer B, Beyer T, Neuhuber W, Repp R,
Kalden JR, Valerius T and Herrmann M: Polymorphonuclear
granulocytes induce antibody-dependent apoptosis in human breast
cancer cells. J Immunol. 171:5124–5129. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Challacombe JM, Suhrbier A, Parsons PG,
Jones B, Hampson P, Kavanagh D, Rainger GE, Morris M, Lord JM, Le
TT, Hoang-Le D and Ogbourne SM: Neutrophils are a key component of
the antitumor efficacy of topical chemotherapy with
ingenol-3-angelate. J Immunol. 177:8123–8132. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Koga Y, Matsuzaki A, Suminoe A, Hattori H
and Hara T: Neutrophil-derived TNF-related apoptosis-inducing
ligand (TRAIL): a novel mechanism of antitumor effect by
neutrophils. Cancer Res. 64:1037–1043. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kemp TJ, Ludwig AT, Earel JK, Moore JM,
Vanoosten RL, Moses B, Leidal K, Nauseef WM and Griffith TS:
Neutrophil stimulation with Mycobacterium bovis bacillus
Calmette-Guérin (BCG) results in the release of functional soluble
TRAIL/Apo-2L. Blood. 106:3474–3482. 2005.
|
|
10
|
de Vries EG, Gietema JA and de Jong S:
Tumor necrosis factor-related apoptosis-inducing ligand pathway and
its therapeutic implications. Clin Cancer Res. 12:2390–2393.
2006.
|
|
11
|
Voortman J and Resende TP: TRAIL therapy
in non-small cell lung cancer cells: sensitization to death
receptor-mediated apoptosis by proteasome inhibitor bortezomib. Mol
Cancer Ther. 6:2103–2112. 2007. View Article : Google Scholar
|
|
12
|
Hoshi H, Sawada T, Uchida M, Saito H,
Iijima H, Toda-Agetsuma M, Wada T, Yamazoe S, Tanaka H, Kimura K,
Kakehashi A, Wei M, Hirakawa K and Wanibuchi H: Tumor-associated
MUC5AC stimulates in vivo tumorigenicity of human pancreatic
cancer. Int J Oncol. 38:619–627. 2011.PubMed/NCBI
|
|
13
|
Hernandez-Ilizaliturri FJ, Jupudy V,
Ostberg J, Oflazoglu E, Huberman A, Repasky E and Czuczman MS:
Neutrophils contribute to the biological antitumor activity of
rituximab in a non-Hodgkin’s lymphoma severe combined
immunodeficiency mouse model. Clin Cancer Res. 9:5866–5873.
2003.PubMed/NCBI
|
|
14
|
Singh PK and Hollingsworth MA: Cell
surface-associated mucins in signal transduction. Trends Cell Biol.
16:467–476. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lane D, Côté M, Grondin R, Couture MC and
Piché A: Acquired resistance to TRAIL-induced apoptosis in human
ovarian cancer cells is conferred by increased turnover of mature
caspase-3. Mol Cancer Ther. 5:509–521. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lane D, Robert V, Grondin R, Rancourt C
and Piché A: Malignant ascites protect against TRAIL-induced
apoptosis by activating the PI3K/Akt pathway in human ovarian
carcinoma cells. Int J Cancer. 121:1227–1237. 2007. View Article : Google Scholar
|
|
17
|
Di Carlo E, Forni G, Lollini P, Colombo
MP, Modesti A and Musiani P: The intriguing role of
polymorphonuclear neutrophils in antitumor reactions. Blood.
97:339–345. 2001.PubMed/NCBI
|
|
18
|
Alvarez MJ, Prada F, Salvatierra E, Bravo
AI, Lutzky VP, Carbone C, Pitossi FJ, Chuluyan HE and Podhajcer OL:
Secreted protein acidic and rich in cysteine produced by human
melanoma cells modulates polymorphonuclear leukocyte recruitment
and antitumor cytotoxic capacity. Cancer Res. 65:5123–5132. 2005.
View Article : Google Scholar
|
|
19
|
Chen YL, Chen SH, Wang JY and Yang BC: Fas
ligand on tumor cells mediates inactivation of neutrophils. J
Immunol. 171:1183–1191. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sanford MA, Yan Y, Canfield SE, Hassan W,
Selleck WA, Atkinson G, Chen SH and Hall SJ: Independent
contributions of GR-1+ leukocytes and Fas/FasL interactions to
induce apoptosis following interleukin-12 gene therapy in a
metastatic model of prostate cancer. Hum Gene Ther. 12:1485–1498.
2001.PubMed/NCBI
|
|
21
|
Niitsu N, Khori M, Hayama M, Kajiwara K,
Higashihara M and Tamaru J: Phase I/II study of the rituximab-EPOCT
regimen in combination with granulocyte colony-stimulating factor
in patients with relapsed or refractory follicular lymphoma
including evaluation of its cardiotoxicity using B-type natriuretic
peptide and troponin T levels. Clin Cancer Res. 11:697–702.
2005.
|
|
22
|
Otten MA, Rudolph E, Dechant M, Tuk CW,
Reijmers RM, Beelen RH, van de Winkel JG and van Egmond M: Immature
neutrophils mediate tumor cell killing via IgA but not IgG Fc
receptors. J Immunol. 174:5472–5480. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wiley SR, Schooley K, Smolak PJ, Din WS,
Huang CP, Nicholl JK, Sutherland GR, Smith TD, Rauch C, Smith CA
and Goodwin RG: Identification and characterization of a new member
of the TNF family that induces apoptosis. Immunity. 3:673–682.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Schimmer AD: Inhibition of apoptosis
proteins: translating basic knowledge into clinical practice.
Cancer Res. 64:7183–7190. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wei Y, Fan T and Yu M: Inhibitor of
apoptosis proteins and apoptosis. Acta Biochim Biophys Sin
(Shanghai). 40:278–288. 2009. View Article : Google Scholar
|
|
26
|
Komatsu M, Jepson S, Arango ME, Carraway
CAC and Carraway KL: Muc4/sialomucin complex, an intramembrane
modulator of ErbB2/HER2/Neu, potentiates primary tumor growth and
suppresses apoptosis in a xenotransplanted tumor. Oncogene.
20:461–470. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Workman HC, Sweeney C and Carraway KL III:
The membrane mucin Muc4 inhibits apoptosis induced by multiple
insults via ErbB2-dependent and ErbB2-independent mechanisms.
Cancer Res. 69:2845–2852. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Baratelli F, Krysan K, Heuzé-Vourc’h N,
Zhu L, Escuadro B, Sharma S, Reckamp K, Dohadwala M and Dubinett
SM: PGE2 confers survivin-dependent apoptosis resistance in human
monocyte-derived dendritic cells. J Leukoc Biol. 78:555–564. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Leone V, di Palma A, Ricchi P, Acquaviva
F, Giannouli M, Di Prisco AM, Iuliano F and Acquaviva AM: PGE2
inhibits apoptosis in human adenocarcinoma Caco-2 cell line through
Ras-PI3K association and cAMP-dependent kinase A activation. Am J
Physiol Gastrointest Liver Physiol. 293:G673–G681. 2007. View Article : Google Scholar : PubMed/NCBI
|