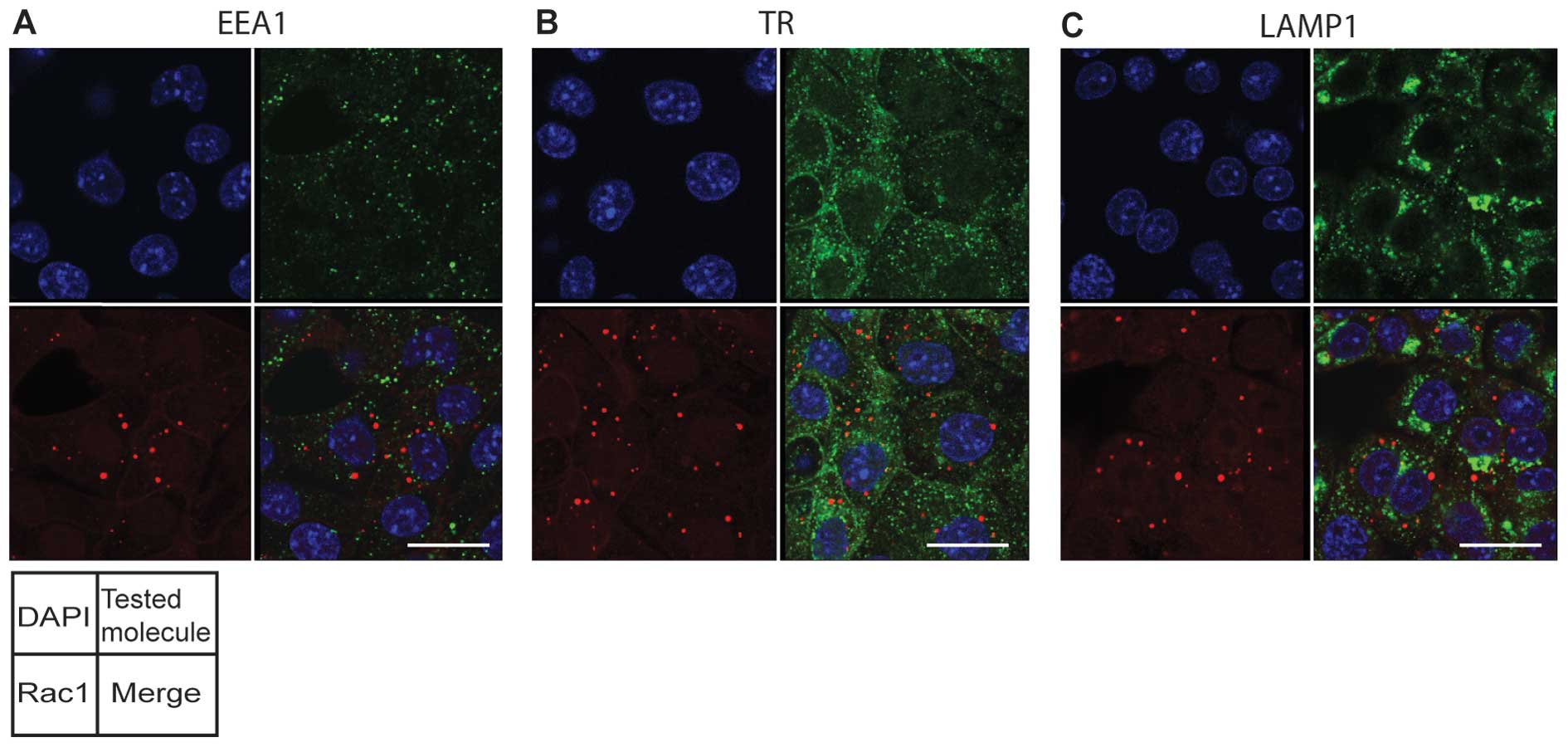
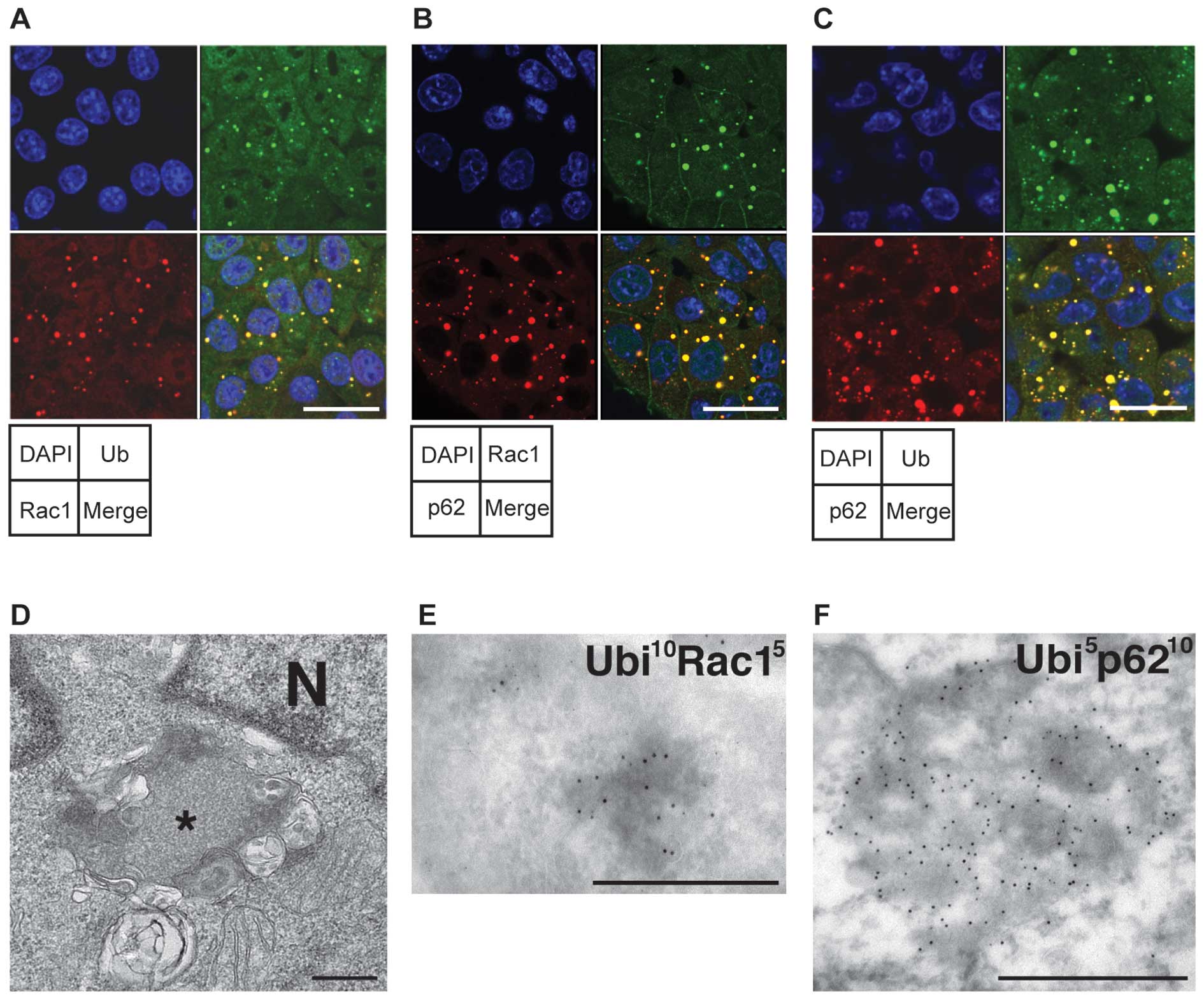
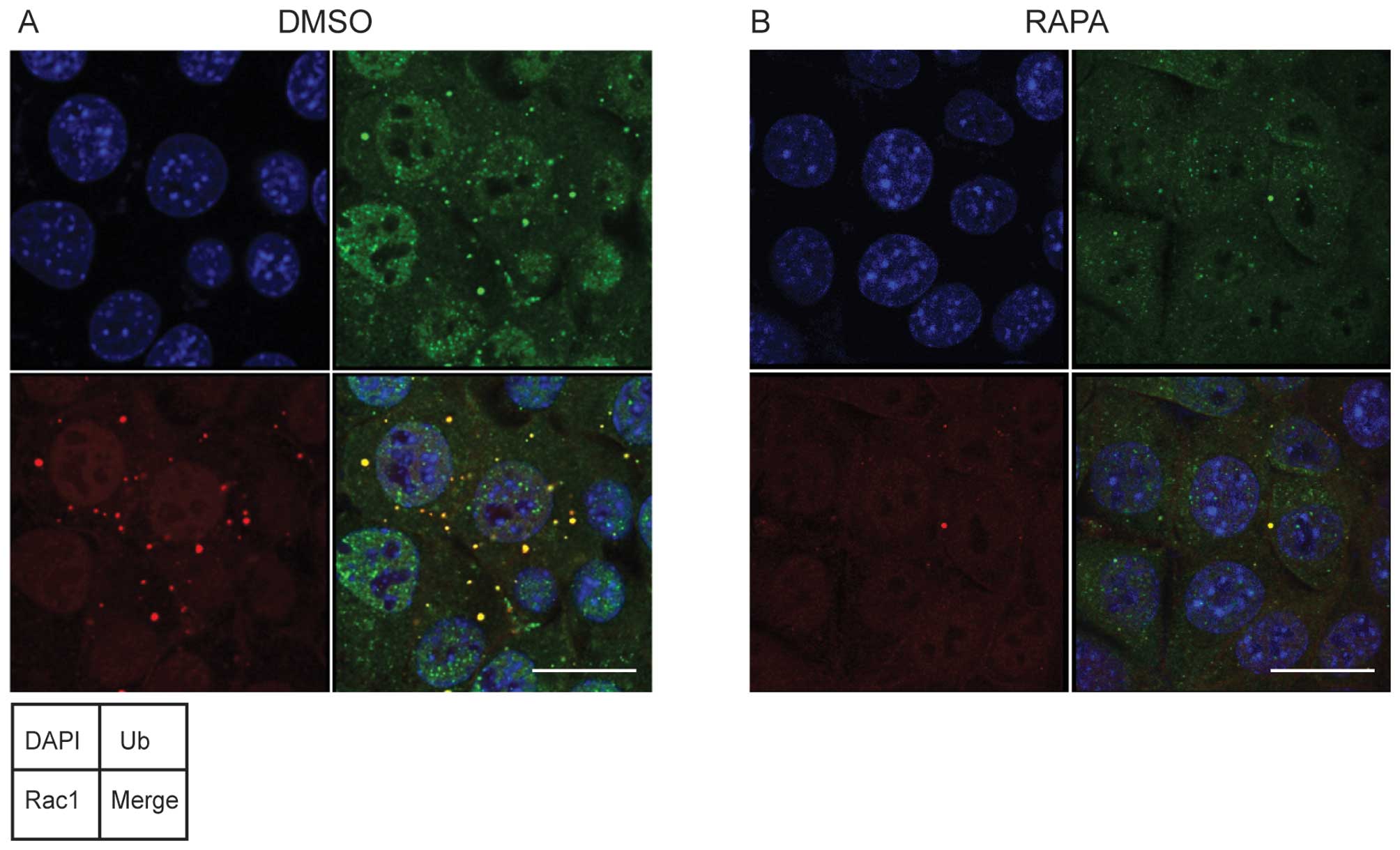
|
1.
|
Inoki K, Li Y, Xu T and Guan KL: Rheb
GTPase is a direct target of TSC2 GAP activity and regulates mTOR
signaling. Genes Dev. 17:1829–1834. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Nellist M, van Slegtenhorst MA, Goedbloed
M, van den Ouweland AM, Halley DJ and van der Sluijs P:
Characterization of the cytosolic tuberin-hamartin complex. Tuberin
is a cytosolic chaperone for hamartin. J Biol Chem.
274:35647–35652. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Adriaensen ME, Schaefer-Prokop CM, Stijnen
T, Duyndam DA, Zonnenberg BA and Prokop M: Prevalence of
subependymal giant cell tumors in patients with tuberous sclerosis
and a review of the literature. Eur J Neurol. 16:691–696. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Nabbout R, Santos M, Rolland Y, Delalande
O, Dulac O and Chiron C: Early diagnosis of subependymal giant cell
astrocytoma in children with tuberous sclerosis. J Neurol Neurosurg
Psychiatry. 66:370–375. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Guertin DA and Sabatini DM: An expanding
role for mTOR in cancer. Trends Mol Med. 11:353–361. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Krueger DA, Care MM, Holland K, et al:
Everolimus for subependymal giant-cell astrocytomas in tuberous
sclerosis. N Engl J Med. 363:1801–1811. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Guertin DA and Sabatini DM: Defining the
role of mTOR in cancer. Cancer Cell. 12:9–22. 2007. View Article : Google Scholar
|
|
8.
|
Sarbassov DD, Ali SM and Sabatini DM:
Growing roles for the mTOR pathway. Curr Opin Cell Biol.
17:596–603. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Loewith R, Jacinto E, Wullschleger S, et
al: Two TOR complexes, only one of which is rapamycin sensitive,
have distinct roles in cell growth control. Mol Cell. 10:457–468.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Jacinto E, Loewith R, Schmidt A, et al:
Mammalian TOR complex 2 controls the actin cytoskeleton and is
rapamycin insensitive. Nat Cell Biol. 6:1122–1128. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Hosokawa N, Hara T, Kaizuka T, et al:
Nutrient-dependent mTORC1 association with the ULK1-Atg13-FIP200
complex required for autophagy. Mol Biol Cell. 20:1981–1991. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Levine B and Kroemer G: Autophagy in the
pathogenesis of disease. Cell. 132:27–42. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Mathew R, Karp CM, Beaudoin B, et al:
Autophagy suppresses tumorigenesis through elimination of p62.
Cell. 137:1062–1075. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Ross CA and Poirier MA: Protein
aggregation and neurodegenerative disease. Nat Med. 10(Suppl):
S10–S17. 2004. View
Article : Google Scholar
|
|
15.
|
Maiuri MC, Tasdemir E, Criollo A, et al:
Control of autophagy by oncogenes and tumor suppressor genes. Cell
Death Differ. 16:87–93. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Ng S, Wu YT, Chen B, Zhou J and Shen HM:
Impaired autophagy due to constitutive mTOR activation sensitizes
TSC2-null cells to cell death under stress. Autophagy. 7:1173–1186.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Yu J, Parkhitko A and Henske EP:
Autophagy: An ‘Achilles’ heel of tumorigenesis in TSC and LAM.
Autophagy. 7:1400–1401. 2011.
|
|
18.
|
Burridge K and Wennerberg K: Rho and Rac
take center stage. Cell. 116:167–179. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Etienne-Manneville S and Hall A: Rho
GTPases in cell biology. Nature. 420:629–635. 2002. View Article : Google Scholar
|
|
20.
|
Goncharova E, Goncharov D, Noonan D and
Krymskaya VP: TSC2 modulates actin cytoskeleton and focal adhesion
through TSC1-binding domain and the Rac1 GTPase. J Cell Biol.
167:1171–1182. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Larson Y, Liu J, Stevens PD, et al:
Tuberous sclerosis complex 2 (TSC2) regulates cell migration and
polarity through activation of CDC42 and RAC1. J Biol Chem.
285:24987–24998. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Hernandez-Negrete I, Carretero-Ortega J,
Rosenfeldt H, et al: P-Rex1 links mammalian target of rapamycin
signaling to Rac activation and cell migration. J Biol Chem.
282:23708–23715. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Yoshizawa M, Hoshino M, Sone M and
Nabeshima Y: Expression of stef, an activator of Rac1, correlates
with the stages of neuronal morphological development in the mouse
brain. Mech Dev. 113:65–68. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Yoshizawa M, Kawauchi T, Sone M, et al:
Involvement of a Rac activator, P-Rex1, in neurotrophin-derived
signaling and neuronal migration. J Neurosci. 25:4406–4419. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Piao X, Kobayashi T, Wang L, et al:
Regulation of folliculin (the BHD gene product) phosphorylation by
Tsc2-mTOR pathway. Biochem Biophys Res Commun. 389:16–21. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Cao Y, Kamioka Y, Yokoi N, et al:
Interaction of FoxO1 and TSC2 induces insulin resistance through
activation of the mammalian target of rapamycin/p70 S6K pathway. J
Biol Chem. 281:40242–40251. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Fukuda T, Tani Y, Kobayashi T, Hirayama Y
and Hino O: A new Western blotting method using polymer
immunocomplexes: detection of Tsc1 and Tsc2 expression in various
cultured cell lines. Anal Biochem. 285:274–276. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Mori Y, Koike M, Moriishi E, et al: Human
herpesvirus-6 induces MVB formation, and virus egress occurs by an
exosomal release pathway. Traffic. 9:1728–1742. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Guo F, Debidda M, Yang L, Williams DA and
Zheng Y: Genetic deletion of Rac1 GTPase reveals its critical role
in actin stress fiber formation and focal adhesion complex
assembly. J Biol Chem. 281:18652–18659. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Hall A: Rho GTPases and the actin
cytoskeleton. Science. 279:509–514. 1998. View Article : Google Scholar
|
|
31.
|
Rubinsztein DC: The roles of intracellular
protein-degradation pathways in neurodegeneration. Nature.
443:780–786. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Moscat J and Diaz-Meco MT: p62 at the
crossroads of autophagy, apoptosis, and cancer. Cell.
137:1001–1004. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33.
|
Bjorkoy G, Lamark T, Brech A, et al:
p62/SQSTM1 forms protein aggregates degraded by autophagy and has a
protective effect on huntingtin-induced cell death. J Cell Biol.
171:603–614. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Yu L, McPhee CK, Zheng L, et al:
Termination of autophagy and reformation of lysosomes regulated by
mTOR. Nature. 465:942–946. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35.
|
Vega FM and Ridley AJ: Rho GTPases in
cancer cell biology. FEBS Lett. 582:2093–2101. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
36.
|
Lozano E, Betson M and Braga VM: Tumor
progression: small GTPases and loss of cell-cell adhesion.
Bioessays. 25:452–463. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Ng J, Nardine T, Harms M, et al: Rac
GTPases control axon growth, guidance and branching. Nature.
416:442–447. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
38.
|
Hoeller D and Dikic I: Targeting the
ubiquitin system in cancer therapy. Nature. 458:438–444. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
39.
|
Hershko A and Ciechanover A: The ubiquitin
system. Annu Rev Biochem. 67:425–479. 1998. View Article : Google Scholar
|
|
40.
|
Nethe M and Hordijk PL: The role of
ubiquitylation and degradation in RhoGTPase signalling. J Cell Sci.
123:4011–4018. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
41.
|
Sanchez P, De Carcer G, Sandoval IV,
Moscat J and Diaz-Meco MT: Localization of atypical protein kinase
C isoforms into lysosome-targeted endosomes through interaction
with p62. Mol Cell Biol. 18:3069–3080. 1998.PubMed/NCBI
|