Introduction
Pancreatic cancer is one of the most common
malignancies worldwide (1). The
prognosis of patients with pancreatic cancer remains poor even
after curative resection and more than half of patients develop
tumor recurrence at distant or local sites, with an estimated
5-year survival rate of only 20% (2–4).
Chemotherapy plays an important role in the treatment of pancreatic
cancer. Gemcitabine (GEM), a cell cycle specific inhibitor of DNA
synthesis and a ribonucleotide reductase, has become the gold
standard chemotherapeutic agent for pancreatic cancer (5,6).
However, the response rate to GEM is <20%, indicating that the
outcome remains unsatisfactory (5). We reported that the expression of
ribonucleotide reductase M1 subunit (RRM1) was significantly
associated with the response to GEM in pancreatic cancer cell lines
and clinical specimens (7,8). However, the clinical response to GEM
based on the expression of RRM1 cannot be predicted satisfactorily.
Therefore, it is necessary to find novel biological markers that
can accurately predict the clinical response to GEM.
Recently, microRNA (miRNA) has emerged as a critical
class of negative regulators of gene expression through modulation
of the post-transcriptional activity of its multiple target mRNAs
by repression of translation or direct cleavage (9,10).
MiRNAs control a wide array of biological processes, including cell
proliferation, differentiation and apoptosis. Aberrant expression
of miRNAs is widely reported in human cancers with both up- and
downregulation detected in cancer cells compared with their normal
counterparts (11,12). Employing gene manipulation
protocols, the present study was designed to identify the miRNA
linked to the response of pancreatic cancer cells to GEM through
the modulation of Wnt/β-catenin signaling pathway. The results
showed a significant relationship between miR-29a and response to
GEM in pancreatic cancer cells. Additional experiments using Wnt3a,
a Wnt/β-catenin signaling activator, demonstrated that the
miR-29a-induced resistance to GEM correlated significantly with the
activation of Wnt/β-catenin signaling in pancreatic cancer cell
lines.
Materials and methods
Pancreatic cancer cell line and clinical
samples
Four human pancreatic carcinoma cell lines
(MIAPaCa-2, PSN-1, BxPC-3 and Panc-1) were used in the present
study (8). MIAPaCa-2 and PSN-1
cell lines were obtained from the Japanese Collection of Research
Bioresources (JCRB, Tokyo, Japan). BxPC-3 and Panc-1 cell lines
were obtained from the American Type Culture Collection (ATCC,
Rockville, MD, USA). These cells were maintained in Dulbecco’s
modified Eagle’s medium supplemented with 10% fetal bovine serum,
100 U/ml penicillin and 100 mg/ml streptomycin at 37°C in a
humidified incubator under 5% CO2-95% air.
Drugs and reagents
GEM was purchased from Eli Lilly and Co.
(Indianapolis, IN, USA). Polyclonal rabbit anti-human DKK1 antibody
(Cell Signaling Technology, Beverly, MA, USA), polyclonal rabbit
anti-human sFRP2 antibody (Abcam Inc., Cambridge, MA, USA),
polyclonal mouse anti-human Kremen2 antibody (Abcam Inc.) and
polyclonal rabbit anti-human β-actin (Sigma-Aldrich Co., St. Louis,
MO, USA) were used for western blot analysis. Recombinant human
Wnt3a (R&D Systems, Minneapolis, MN, USA) was used as a
Wnt/β-catenin signaling activator. In this study, Wnt3a was used at
50 ng/ml based on the protocol described in a previous study
(13).
Transfection
Antisense miR-29a inhibitor (anti-miR-29a), miR-29a
precursor (pre-miR-29a) and their negative control oligonucleotides
were obtained from Ambion Inc. (Austin, TX, USA). These were used
to transfect pancreatic cancer cells by using siPORT NeoFx (Ambion
Inc.) according to the instructions provided by the manufacturer.
The transected cells were resuspended and cultured in regular
culture medium for 24–72 h before analysis.
RNA extraction
Total RNA and miRNA fractions were isolated from
tissue samples and cell lines by TRIzol agent (Invitrogen,
Carlsbad, CA, USA) and the quality of the RNA was assessed with a
NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies,
Wilmington, DE, USA) at 260 and 280 nm (A260/280).
Real-time quantitative reverse
transcription-polymerase chain reaction for miRNA expression
Reverse transcription reaction and real-time
quantitative reverse transcription-polymerase chain reaction
(qRT-PCR) were performed using TaqMan human miRNA assay kit
(Applied Biosystems, Foster City, CA, USA) according to the
instructions supplied by the manufacturer. The expression of the
target miRNA was normalized relative to that of the internal
control; RNU48. Data were analyzed according to the comparative Ct
method (14).
Real-time qRT-PCR for mRNA
expression
RT reaction was performed with SuperScript II
(Invitrogen) based on the protocol provided by the manufacturer,
followed by qRT-PCR. The expression of the target gene was
normalized relative to the expression of porphobilinogen
deaminase (PBGD), which was used as an internal control. The
designed PCR primers were as follows; AXIN2 forward primer,
5′-GGTGTTTGAGGAGATCTGGG-3′; AXIN2 reverse primer,
5′-TGCTCACAGCCAAGACAGTT-3′; CCND1 forward primer,
5′-AAGGCCTGAACCTGAGGAG-3′; CCND1 reverse primer,
5′-CTTGACTCCAGCAGGGCTT-3′; MYC forward primer,
5′-AAGAGGACTTGTTGCGGAAA-3′; MYC reverse primer,
5′-CTCAGCCAAGGTTGTGAGGT-3′; TACSTD1 forward primer,
5′-TCCAGAAAGAAGAGAATGGCA-3′; TACSTD1 reverse primer,
5′-AAAGATGTCTTCGTCCCACG-3′; TCF3 forward primer,
5′-ATCTGTGTCCCATGTCCCAG-3′; TCF3 reverse primer,
5′-CCAGGGTAGGAGACTTGCAG-3′; and PBGD forward primer,
5′-TGTCTGGTAACGGCAATGCGGCTGCAAC-3′; PBGD reverse primer,
5′-TCAATGTTGCCACCACACTGTCCGTCT-3′.
Western blot analysis
Cells grown to semiconfluence were lysed in RIPA
buffer [25 mM Tris (pH 7.5), 50 mM NaCl, 0.5% sodium deoxycholate,
2% Nonidet P-40, 0.2% sodium dodecyl sulfate, 1 mM
phenylmethylsulphonyl fluoride and 500 KIE/ml Trasylol, proteinase
inhibitor (Bayer, Leverkusen, Germany)]. Western blot analysis was
carried out as described previously (15,16).
Growth-inhibitory assay
Inhibition of cell growth in the presence of
chemotherapeutic agents was assessed by the
3-(4-,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide
(MTT) (Sigma-Aldrich Co.) assay as described previously (15,17).
Briefly, the cells were incubated for 72 h under various
concentrations of GEM. After re-incubation for 4 h in MTT solution,
acid-isopropanol was added to dissolve the resultant formazan
crystals. The absorbance of the plate was measured in a microplate
reader at a wavelength of 570 nm with a 650-nm reference and the
results were expressed as the percentage of absorbance relative to
untreated controls.
Annexin V assay
The binding of Annexin V was used as a sensitive
method for measuring apoptosis, as described previously (15). Twenty-four hours after treatment,
cells were stained with Annexin V-FITC and propidium iodide (PI)
(BioVision Research Products, Mountain View, CA, USA) and analyzed
on a FACSCalibur (BD Biosciences, Franklin Lakes, NJ, USA). For the
assessment of apoptosis, Annexin V-positive and PI-negative cells
and Annexin V-positive and PI-positive cells were considered as
early apoptotic cells and late apoptotic cells, respectively.
Cell cycle analysis
Cell cycle analysis was performed based on flow
cytometric analysis, as described previously (16,17).
Briefly, PI and RNase (Sigma-Aldrich Co.) were added and data were
acquired on the FACSCalibur (BD Biosciences). The cell cycle was
analyzed using ModFIT software (BD Biosciences).
Luciferase reporter assay
To evaluate the activity of the Wnt/β-catenin
signaling pathway, TCF/LEF transcriptional activity was examined.
For the examination, the reporter assay kit (SA Biosciences,
Frederick, MD, USA) was used according to the instructions provided
by the manufacturer. In brief, cells were transiently transfected
with the transcription factor-responsive reporter or negative
control by the Lipofectamine 2000 reagent (Invitrogen). After the
transfection, the cells were transfected with anti-miR-29a or its
negative control oligonucleotide. After 48 h, luciferase activity
was measured with the Dual-Luciferase Assay System (Promega,
Madison, WI, USA) using luminometer, Lumat LB9507 (Berthold
Technologies, Calmbacher, Germany). The Firefly luciferase
activity, indicating TCF-dependent transcription, was normalized to
the Renilla luciferase activity as an internal control to obtain
the relative luciferase activity.
Statistical analysis
Data were expressed as mean ± SD.
Clinicopathological parameters were compared using the
χ2 test and continuous variables were compared using the
Student’s t-test. A p<0.05 denoted the presence of a
statistically significant difference. Statistical analysis was
performed using the StatView software (SAS Institute Inc., Cary,
NC, USA).
Results
Transfection of anti-miR-29a reduces
resistance to GEM
All the four cell lines used in the present study
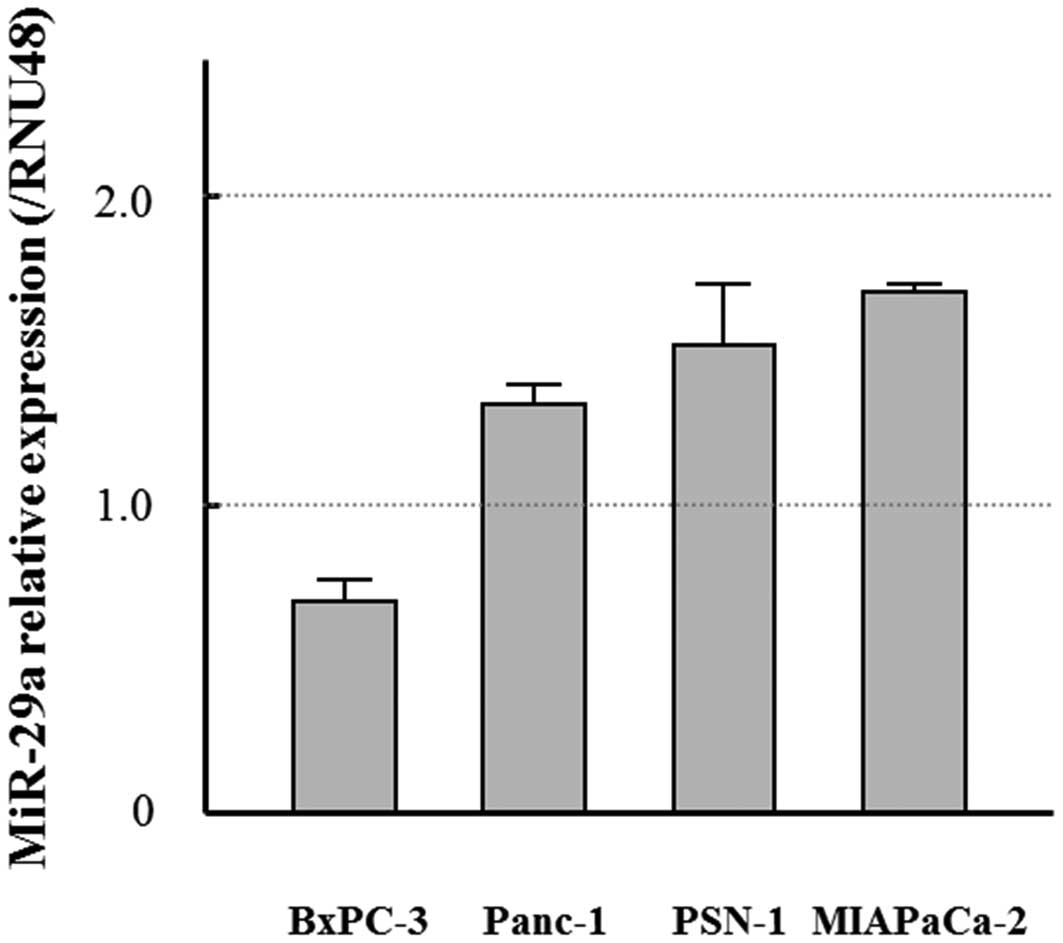
expressed miR-29a though the level varied among the cells (Fig. 1). The relative expression of
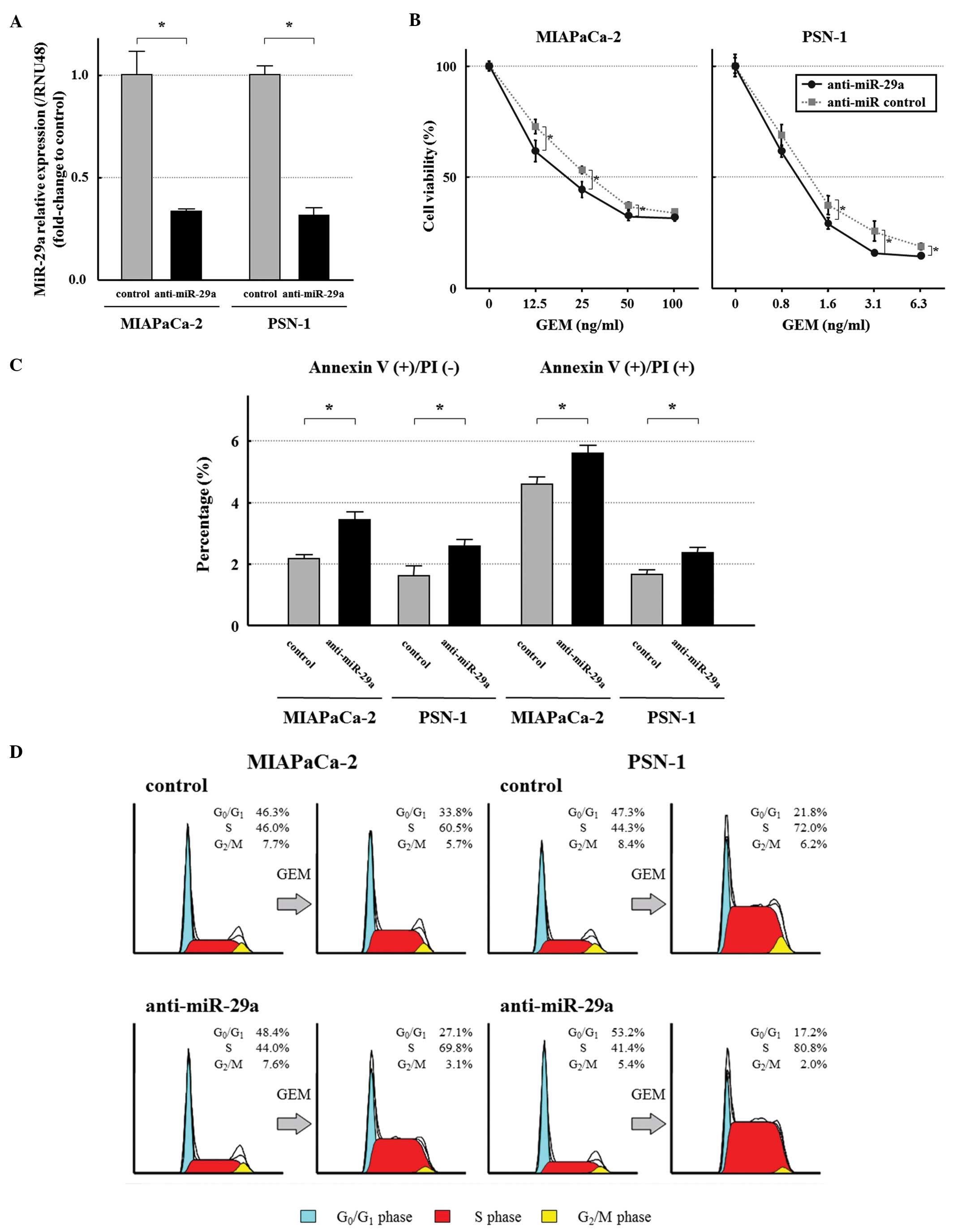
miR-29a was significantly lower in the transfected cells by qRT-PCR
(Fig. 2A). To evaluate the effect
of miR-29a on the response to GEM, we transfected anti-miR-29a in
MIAPaCa-2 and PSN-1 cells, which showed higher miR-29a expression
levels than the other cell lines. The MTT assay showed that cells
transfected with anti-miR-29a were significantly less resistant to
GEM compared to control cells (Fig.
2B). Next, we evaluated the extent of apoptosis of these cells
at 24 h after treatment with GEM (MIAPaCa-2; 40 ng/ml, PSN-1; 2
ng/ml) by the Annexin V assay. The percentages of early and late
apoptotic cells were significantly higher in the two cancer cell
lines transfected with anti-miR-29a than in the control cells
(Fig. 2C).
We also examined the influence of miR-29a on the
cell cycle in MIAPaCa-2 and PSN-1. The distribution of cells in
G0/G1 phase, S phase and G2/M
phase was similar between the miR-29a-suppressed cells and the
control cells in the absence of GEM (Fig. 2D). However, 24 h after GEM
treatment (MIAPaCa-2; 20 ng/ml, PSN-1; 1 ng/ml), the percentage of
cells at the S phase among the miR-29a-suppressed cells was higher
than the control cells (Fig.
2D).
MiR-29a activates Wnt/β-catenin signaling
pathway
We examined next the expression levels of these
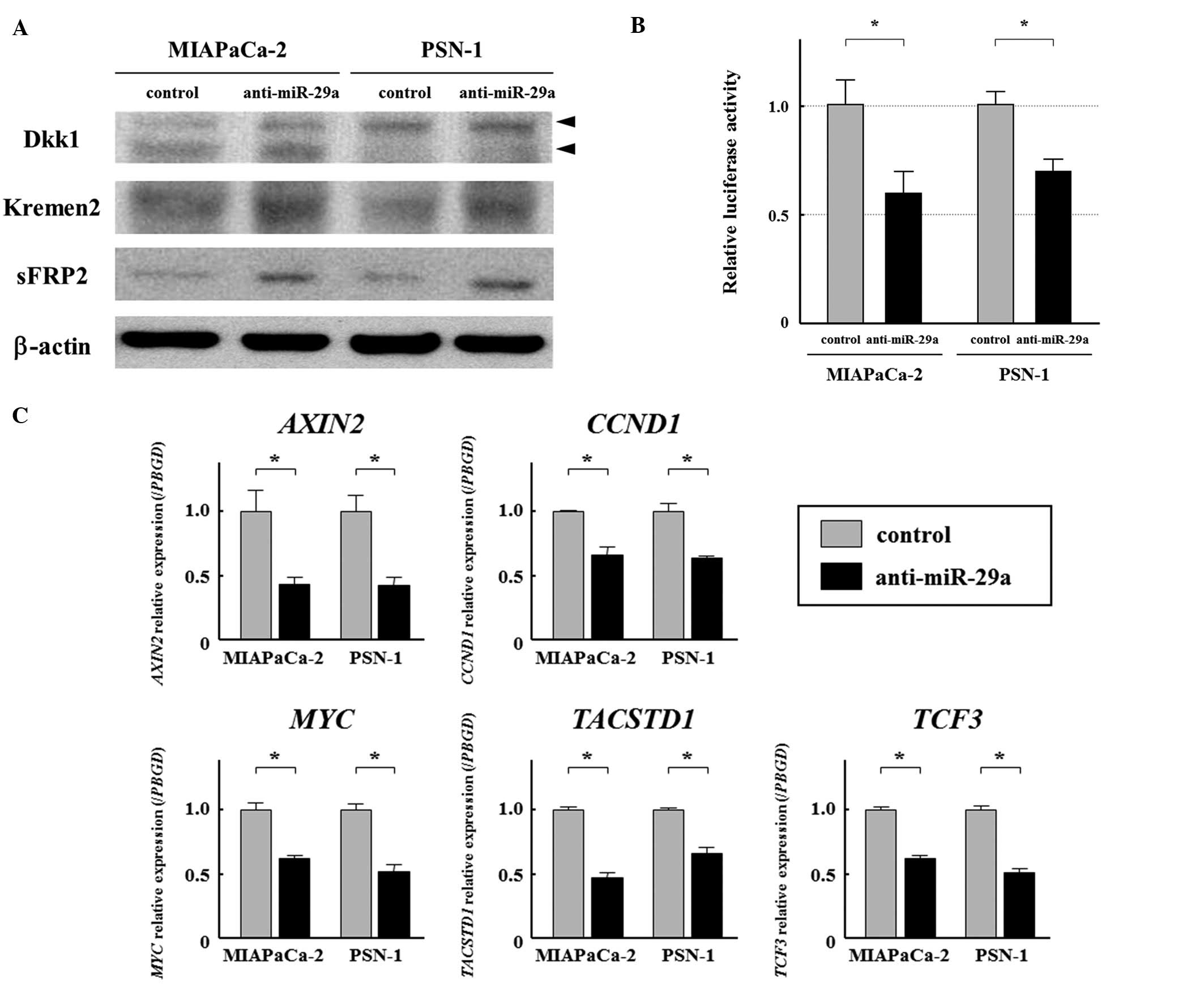
molecules in MIAPaCa-2 and PSN-1. Western blot analysis showed
significantly higher protein expression levels of these molecules
in the anti-miR-29a-transfected cells compared with the control
cells (Fig. 3A). Furthermore, the
luciferase reporter assay showed that TCF/LEF transcriptional
activity, representing the activity of the Wnt/β-catenin signaling
pathway, was significantly lower in the miR-29a-suppressed cells
than in the control cells (Fig.
3B). We also examined the expression of five Wnt/β-catenin
signaling targeted genes (AXIN2, CCND1, MYC, TACSTD1 and
TCF3) in anti-miR-29a-transfected MIAPaCa-2 and PSN-1 cell
lines by qRT-PCR. Suppression of miR-29a significantly reduced the
mRNA expression of the targeted genes (Fig. 3C). Taken together, the results
suggest that miR-29a activates the Wnt/β-catenin signaling pathway
through the suppression of Dkk1, Kremen2 and sFRP2.
Transfection of pre-miR-29a induces
resistance to GEM
To further assess the effects of miR-29a,
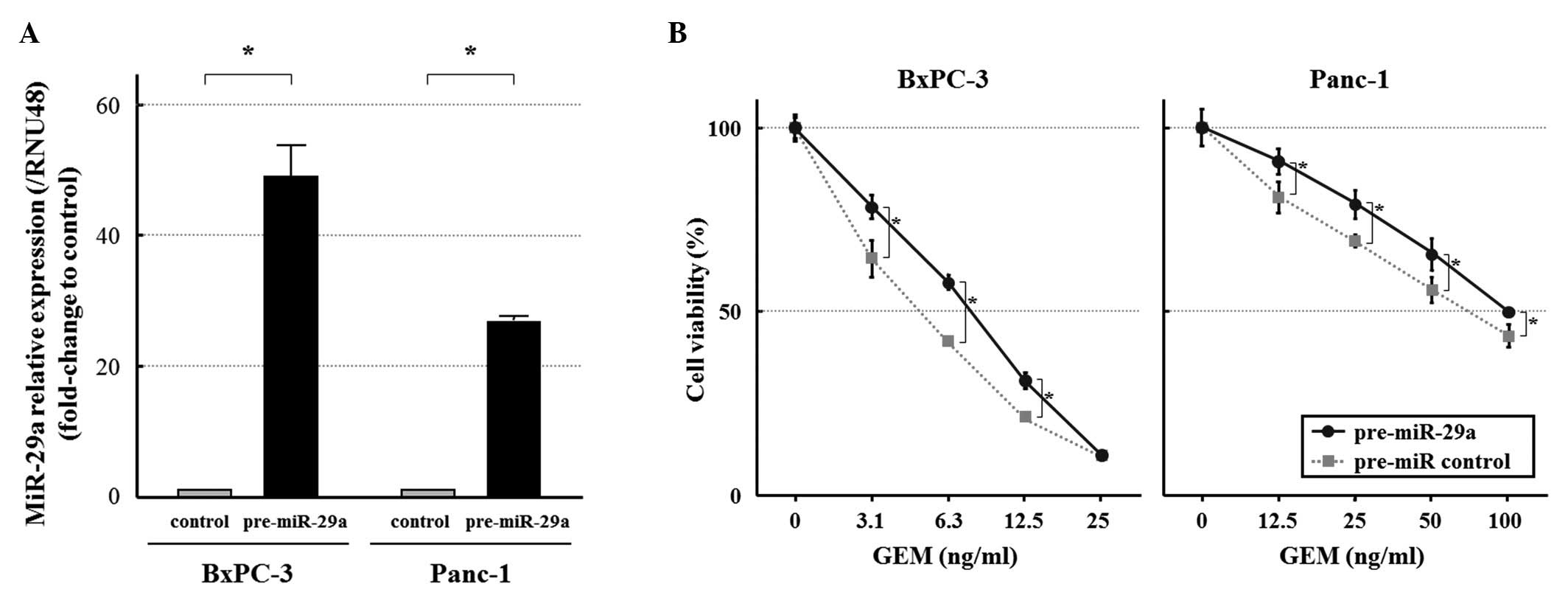
pre-miR-29a was transfected into BxPC-3 and Panc-1, which expressed
lower levels of miR-29a than the other cell lines (Fig. 1). Transfection of cells with
pre-miR-29a increased miR-29a level compared to the control cells
(Fig. 4A). The
miR-29a-overexpressing cells were significantly more resistant to
GEM than the control cells, as evident by the MTT assay (Fig. 4B).
MiR-29a-induced resistance is mediated by
Wnt/β-catenin signaling activation
Finally, we analyzed the mechanism responsible for
the miR-29a-induced resistance to GEM. We focused on the
Wnt/β-catenin signaling pathway, based on the results reported by
Kapinas et al (13). The
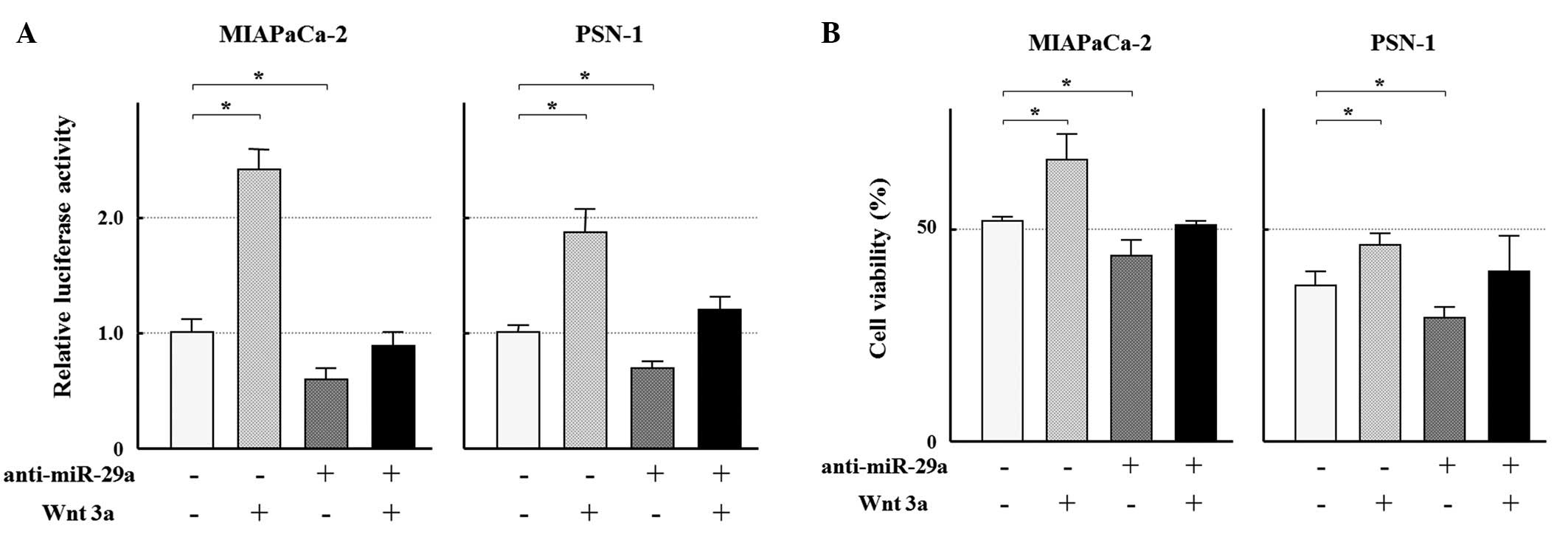
addition of Wnt3a to the cultures of MIAPaCa-2 and PSN-1 resulted
in the activation of Wnt/β-catenin signal in the cell lines
(Fig. 5A). Furthermore, the MTT
assay showed the Wnt3a-treated cells were more resistant to GEM
(MIAPaCa-2; 25 ng/ml, PSN-1; 1.6 ng/ml, Fig. 5B). In addition, both the
inactivated Wnt/β-catenin signal and the augmented
growth-inhibitory effect by the afore-mentioned anti-miR-29a
transfection were weakened after the addition of Wnt3a (Fig. 5). These findings suggest that
activation of the Wnt/β-catenin signaling mediates, at least in
part, the miR-29a-induced resistance to GEM.
Discussion
The present study demonstrated that the expression
of miR-29a correlated significantly with the growth-inhibitory
effect of GEM and that the Wnt/β-catenin signal mediates the
miR-29a-induced resistance to GEM in pancreatic cancer cell lines.
Kapinas et al reported previously that miR-29a activates the
Wnt/β-catenin signal through direct regulation of the negative
regulators of the signal, Dkk1, Kremen2 and sFRP2 (13). Other studies indicated that
activation of the Wnt/β-catenin-signaling, which is observed in 65%
of pancreatic cancer cases, also plays an important role in the
proliferation and differentiation of stem cells and that some
chemotherapeutic drugs often induce tumor cell death, but not
cancer stem cells (18–22). Moreover, the Wnt/β-catenin signal
was reported to correlate significantly with chemoresistance
(16,23–25).
Thus, the results of the present study are in agreement with the
above previous reports.
On the other hand, the Wnt/β-catenin signaling
pathway, which plays important roles in the development of various
malignancies, cell proliferation and differentiation, has been also
reported to correlate with chemoresistance (23–25).
In fact, we reported previously the activation of the Wnt/β-catenin
signaling pathway in HCC with poor response to interferon and
5-fluorouracil therapy (16).
Recently, Kapinas et al (13) reported that miR-29a activates the
Wnt/β-catenin signal by directly regulating Dikkopf-1 (Dkk1),
Kremen2 and secreted frizzled related protein 2 (sFRP2), which are
negative regulators of the signal transduction, suggesting that
miR-29a induces chemoresistance to chemotherapeutic agents through
the activation of the Wnt/β-catenin signaling pathway.
We reported previously that RRM1 expression
correlates significantly with the response to GEM (7,8).
Therefore, in the present study, we also investigated the effects
of anti-miR-29a transfection on RRM1 expression. The result showed
no significant change in RRM1 expression after anti-miR-29a
transfection (data not shown). Several investigators also reported
that the expression of miRNAs correlates significantly with
chemoresistance in several types of cancers (15,26,27).
For example, we reported that the expression of miR-21 is
associated with resistance to interferon and 5-fluorouracil in
hepatocellular carcinoma (HCC) (13), while others indicated that such
expression is associated with response to GEM in pancreatic cancer
and cholangiocarcinoma cells (15,26,27).
Furthermore, since miRNA is associated with the
response to GEM, miR-21 has been reported in some studies of
pancreatic and other cancers (15,26,27).
Therefore, in the present study, the effects of transfection of
anti-miR-29a on miR-21 expression were also examined and the
results showed no significant changes in miR-21 expression (data
not shown). These results suggest that the chemoresistance induced
by miR-29a is different from that related to RRM1 and miR-21.
To date, evidence suggests that miR-29a acts as an
oncomiRNA as well as an anti-oncomiRNA (28–31).
Xiong et al (28) reported
that miR-29a promotes apoptosis and represses tumorigenicity in HCC
cells, while the present study showed contradictory results. The
reason for this contradiction remains unresolved, but it is
speculated that miR-29a can act as either an oncomiRNA or an
anti-oncomiRNA, depending on the tumor circumstances, suggesting
that the exact role of miR-29a in cancer is still unclear and needs
to be fully investigated in the future.
Several studies have reported that miR-29a is
detected in the sera of patients with ovarian and colorectal
cancers, suggesting its potential use as a biomarker for cancer
detection (32,33). Confirmation of the present findings
in larger population multicenter studies may allow the measurement
of plasma levels of miR-29a to predict the clinical response to GEM
in patients with pancreatic cancer.
In conclusion, the present study demonstrated a
significant association between miR-29a expression and the response
to GEM in pancreatic cancer cell lines by genetic manipulation
experiments. The results showed that the miR-29a-induced resistance
to GEM is mediated by activation of the Wnt/β-catenin signaling
pathway. These findings suggest that miR-29a could be potentially
used as a marker for the prediction of the clinical response to GEM
and serves as a potential target for therapy against pancreatic
cancer.
References
|
1.
|
Warshaw AL and Fernandez-del Castillo C:
Pancreatic carcinoma. N Engl J Med. 326:455–465. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Li D, Xie K, Wolff R and Abbruzzese JL:
Pancreatic cancer. Lancet. 363:1049–1057. 2004. View Article : Google Scholar
|
|
3.
|
Neoptolemos JP, Stocken DD, Friess H, et
al: A randomized trial of chemoradiotherapy and chemotherapy after
resection of pancreatic cancer. N Engl J Med. 350:1200–1210. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Sener SF, Fremgen A, Menck HR and
Winchester DP: Pancreatic cancer: a report of treatment and
survival trends for 100,313 patients diagnosed from 1985–1995,
using the National Cancer Database. J Am Coll Surg. 189:1–7.
1999.PubMed/NCBI
|
|
5.
|
Burris HA III, Moore MJ, Andersen J, et
al: Improvements in survival and clinical benefit with gemcitabine
as first-line therapy for patients with advanced pancreas cancer: a
randomized trial. J Clin Oncol. 15:2403–2413. 1997.PubMed/NCBI
|
|
6.
|
Oettle H, Post S, Neuhaus P, et al:
Adjuvant chemotherapy with gemcitabine vs observation in patients
undergoing curative-intent resection of pancreatic cancer: a
randomized controlled trial. JAMA. 297:267–277. 2007. View Article : Google Scholar
|
|
7.
|
Akita H, Zheng Z, Takeda Y, et al:
Significance of RRM1 and ERCC1 expression in resectable pancreatic
adenocarcinoma. Oncogene. 28:2903–2909. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Nakahira S, Nakamori S, Tsujie M, et al:
Involvement of ribo-nucleotide reductase M1 subunit overexpression
in gemcitabine resistance of human pancreatic cancer. Int J Cancer.
120:1355–1363. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Calin GA and Croce CM: MicroRNA-cancer
connection: the beginning of a new tale. Cancer Res. 66:7390–7394.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Bartel DP: MicroRNAs: genomics,
biogenesis, mechanism and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Croce CM and Calin GA: miRNAs, cancer and
stem cell division. Cell. 122:6–7. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Calin GA and Croce CM: MicroRNA signatures
in human cancers. Nat Rev Cancer. 6:857–866. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Kapinas K, Kessler C, Ricks T, Gronowicz G
and Delany AM: miR-29 modulates Wnt signaling in human osteoblasts
through a positive feedback loop. J Biol Chem. 285:25221–25231.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Schmittgen TD, Jiang J, Liu Q and Yang L:
A high-throughput method to monitor the expression of microRNA
precursors. Nucleic Acids Res. 32:e432004. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Tomimaru Y, Eguchi H, Nagano H, et al:
MicroRNA-21 induces resistance to the anti-tumor effect of
interferon-α/5-fluorouracil in hepatocellular carcinoma cells. Br J
Cancer. 103:1617–1626. 2010.PubMed/NCBI
|
|
16.
|
Noda T, Nagano H, Takemasa I, et al:
Activation of Wnt/beta-catenin signalling pathway induces
chemoresistance to interferon-alpha/5-fluorouracil combination
therapy for hepato-cellular carcinoma. Br J Cancer. 100:1647–1658.
2009. View Article : Google Scholar
|
|
17.
|
Eguchi H, Nagano H, Yamamoto H, et al:
Augmentation of antitumor activity of 5-fluorouracil by interferon
alpha is associated with up-regulation of p27Kip1 in human
hepatocellular carcinoma cells. Clin Cancer Res. 6:2881–2890.
2000.PubMed/NCBI
|
|
18.
|
Jamieson CH, Ailles LE, Dylla SJ, et al:
Granulocyte-macrophage progenitors as candidate leukemic stem cells
in blast-crisis CML. N Engl J Med. 351:657–667. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Klaus A and Birchmeier W: Wnt signalling
and its impact on development and cancer. Nat Rev Cancer.
8:387–398. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Pasca di Magliano M, Biankin AV, Heiser
PW, et al: Common activation of canonical Wnt signaling in
pancreatic adenocarcinoma. PLoS One. 2:e11552007.PubMed/NCBI
|
|
21.
|
Zeng G, Germinaro M, Micsenyi A, et al:
Aberrant Wnt/beta-catenin signaling in pancreatic adenocarcinoma.
Neoplasia. 8:279–289. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Barker N and Clevers H: Mining the Wnt
pathway for cancer therapeutics. Nat Rev Drug Discov. 5:997–1014.
2006. View
Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Yang W, Yan HX, Chen L, et al:
Wnt/beta-catenin signaling contributes to activation of normal and
tumorigenic liver progenitor cells. Cancer Res. 68:4287–4295. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Su HY, Lai HC, Lin YW, et al: Epigenetic
silencing of SFRP5 is related to malignant phenotype and
chemoresistance of ovarian cancer through Wnt signaling pathway.
Int J Cancer. 127:555–567. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Flahaut M, Meier R, Coulon A, et al: The
Wnt receptor FZD1 mediates chemoresistance in neuroblastoma through
activation of the Wnt/beta-catenin pathway. Oncogene. 28:2245–2256.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Giovannetti E, Funel N, Peters GJ, et al:
MicroRNA-21 in pancreatic cancer: correlation with clinical outcome
and pharmacologic aspects underlying its role in the modulation of
gemcitabine activity. Cancer Res. 70:4528–4538. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Ali S, Ahmad A, Banerjee S, et al:
Gemcitabine sensitivity can be induced in pancreatic cancer cells
through modulation of miR-200 and miR-21 expression by curcumin or
its analogue CDF. Cancer Res. 70:3606–3617. 2010. View Article : Google Scholar
|
|
28.
|
Xiong Y, Fang JH, Yun JP, et al: Effects
of microRNA-29 on apoptosis, tumorigenicity and prognosis of
hepatocellular carcinoma. Hepatology. 51:836–845. 2010.PubMed/NCBI
|
|
29.
|
Fabbri M, Ivan M, Cimmino A, Negrini M and
Calin GA: Regulatory mechanisms of microRNAs involvement in cancer.
Expert Opin Biol Ther. 7:1009–1019. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Gebeshuber CA, Zatloukal K and Martinez J:
miR-29a suppresses tristetraprolin, which is a regulator of
epithelial polarity and metastasis. EMBO Rep. 10:400–405. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
31.
|
Han YC, Park CY, Bhagat G, et al:
microRNA-29a induces aberrant self-renewal capacity in
hematopoietic progenitors, biased myeloid development and acute
myeloid leukemia. J Exp Med. 207:475–489. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Resnick KE, Alder H, Hagan JP, Richardson
DL, Croce CM and Cohn DE: The detection of differentially expressed
microRNAs from the serum of ovarian cancer patients using a novel
real-time PCR platform. Gynecol Oncol. 112:55–59. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33.
|
Huang Z, Huang D, Ni S, Peng Z, Sheng W
and Du X: Plasma microRNAs are promising novel biomarkers for early
detection of colorectal cancer. Int J Cancer. 127:118–126. 2010.
View Article : Google Scholar : PubMed/NCBI
|