|
1.
|
Ferlay J, Shin HR, Bray F, Forman D,
Mathers C and Parkin DM: Estimates of worldwide burden of cancer in
2008: GLOBOCAN 2008. Int J Cancer. 127:2893–2917. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Husain AN: Lung tumors. Robbins Basic
Pathology. Kumar V, Abbas AK and Aster JC: Elsevier Saunders;
Philadelphia, PA: pp. 505–510. 2013
|
|
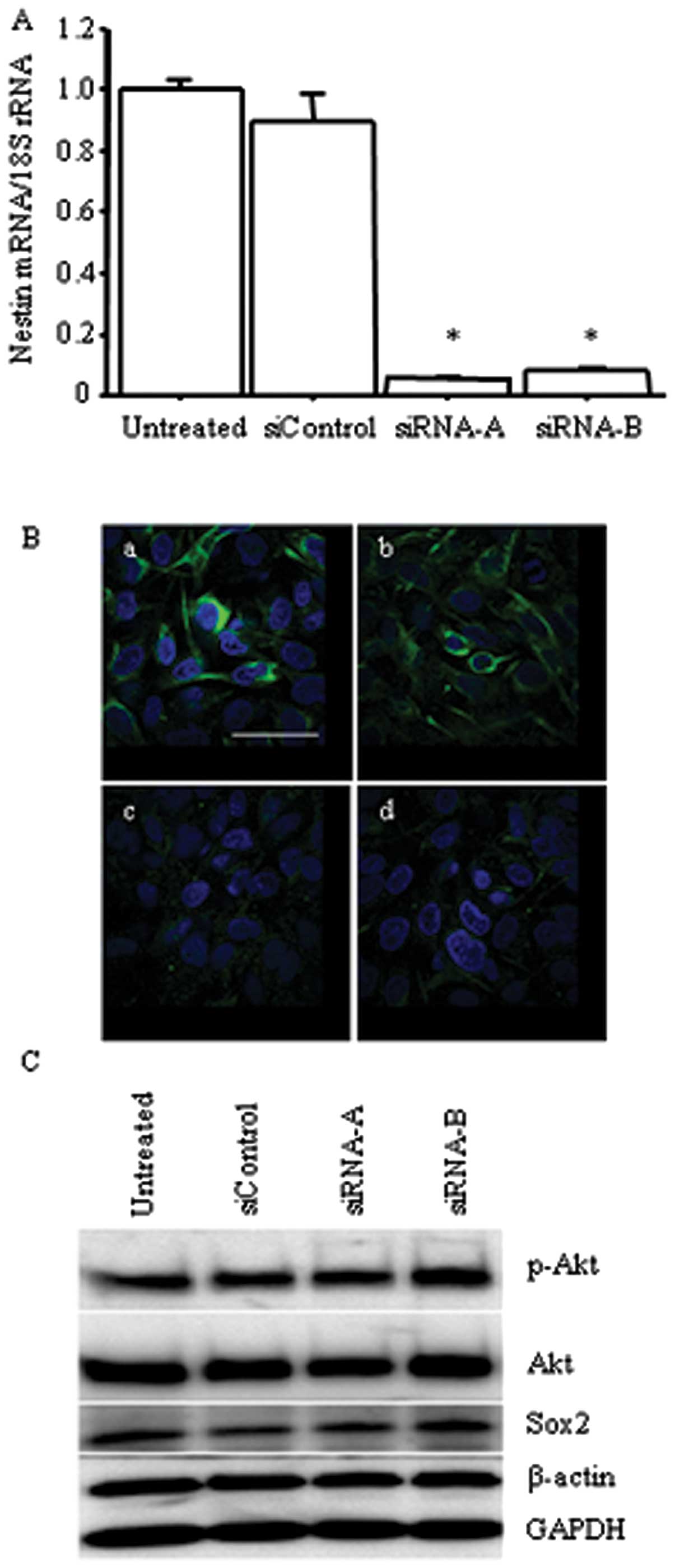
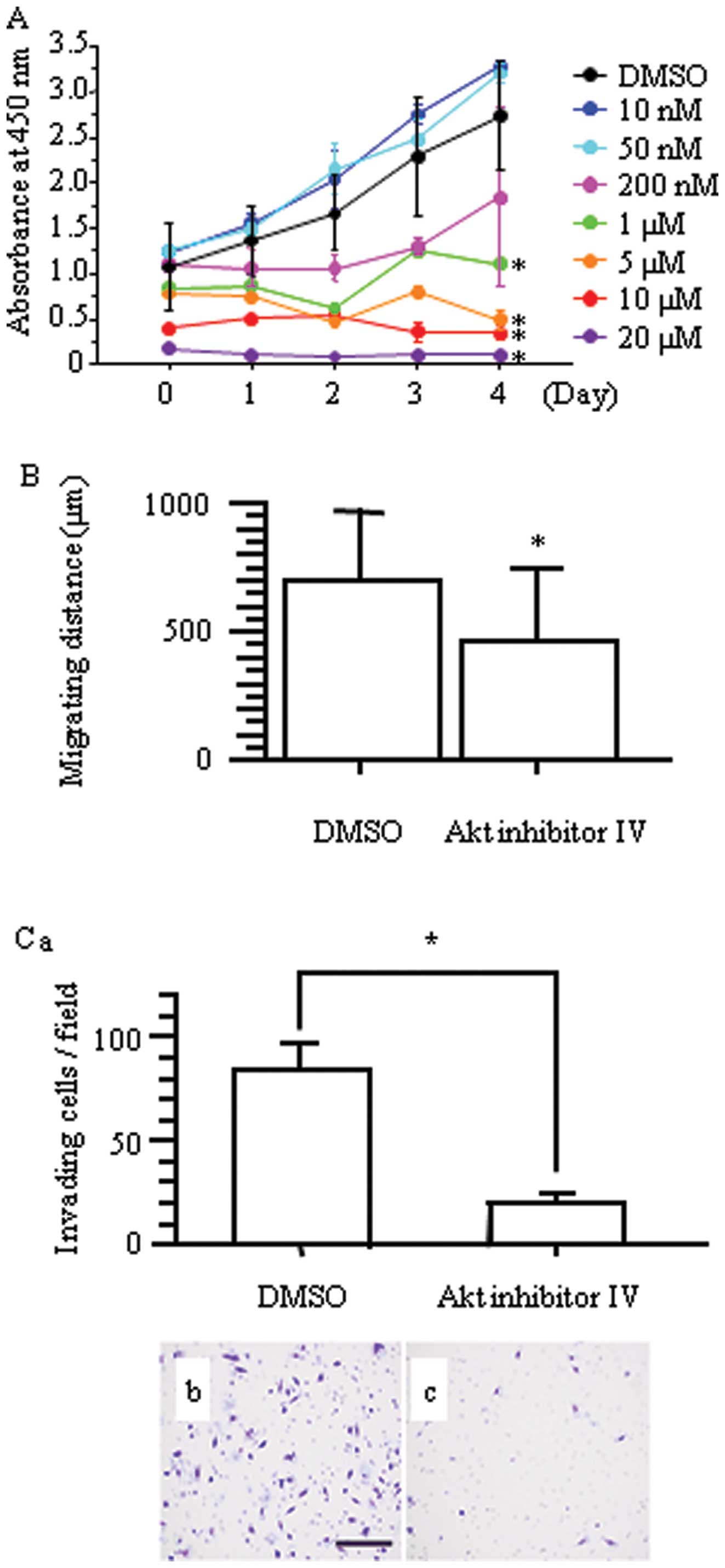
3.
|
Non-small Cell Lung Cancer Collaborative
Group: Chemotherapy in non-small cell lung cancer: a meta-analysis
using updated data on individual patients from 52 randomised
clinical trials. BMJ. 311:899–909. 1995. View Article : Google Scholar : PubMed/NCBI
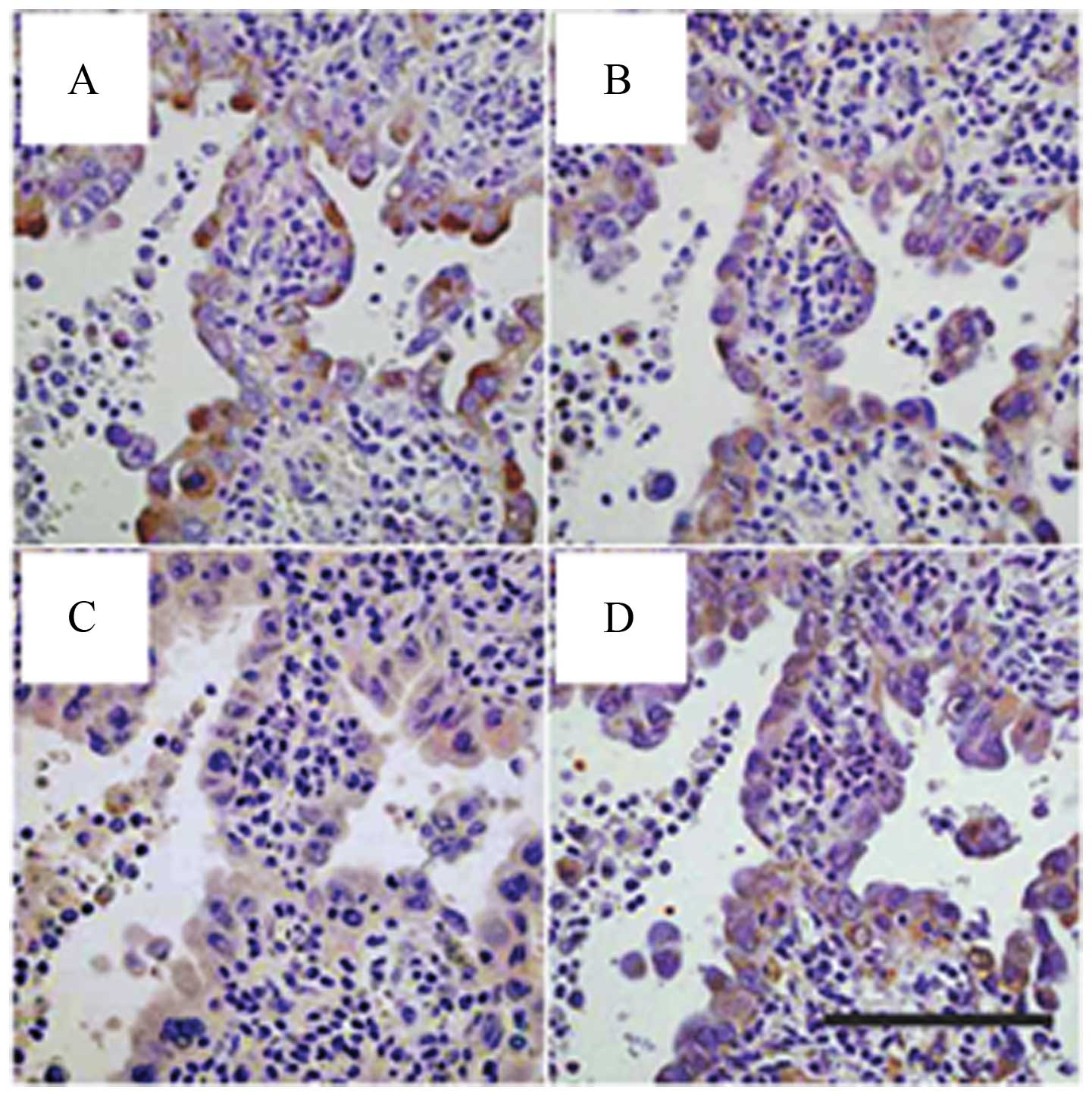
|
|
4.
|
Maemondo M, Inoue A, Kobayashi K, et al:
Gefitinib or chemotherapy for non-small-cell lung cancer with
mutated EGFR. N Engl J Med. 362:2380–2388. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Devesa SS, Bray F, Vizcaino AP and Parkin
DM: International lung cancer trends by histologic type:
male:female differences diminishing and adenocarcinoma rates
rising. Int J Cancer. 117:294–299. 2005. View Article : Google Scholar : PubMed/NCBI
|
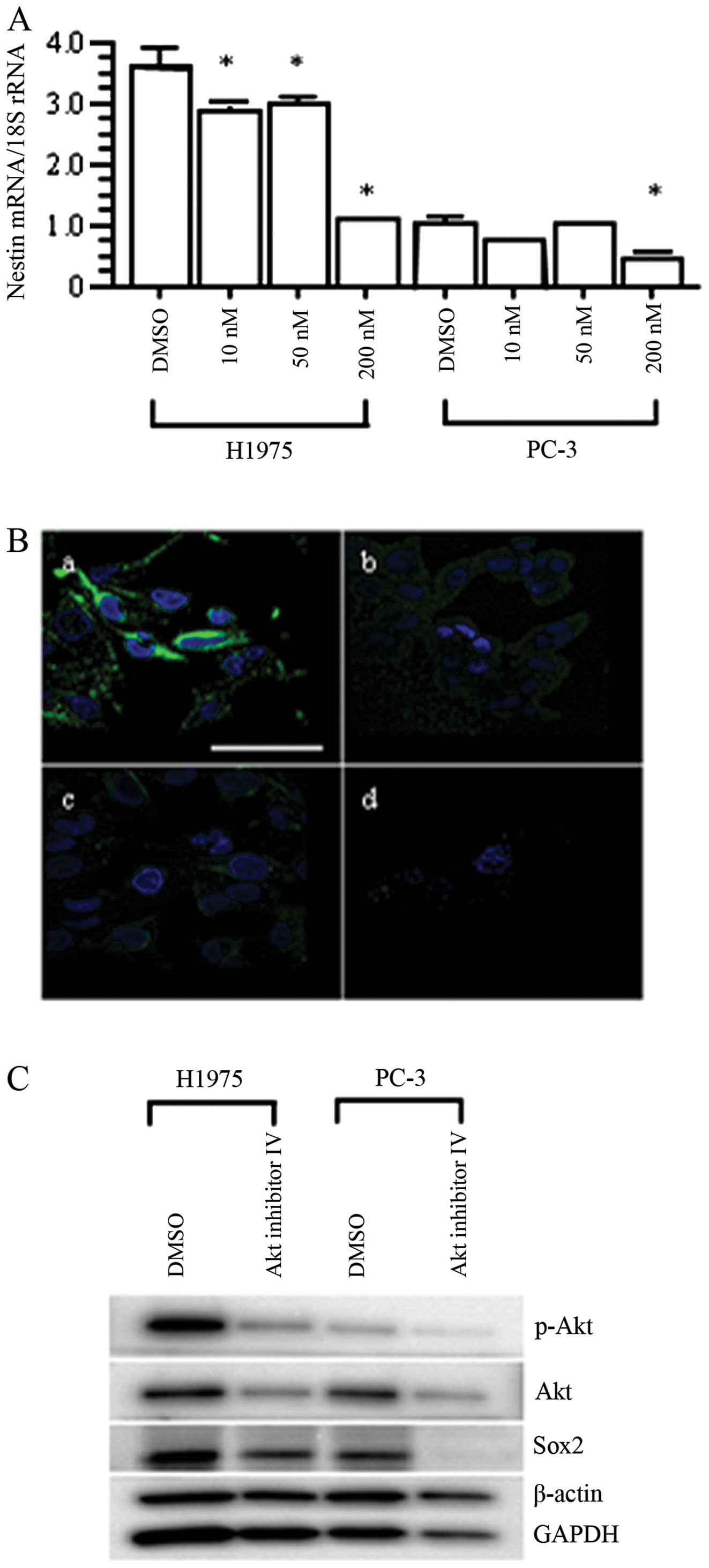
|
6.
|
Mitsudomi T, Morita S, Yatabe Y, et al:
Gefitinib versus cisplatin plus docetaxel in patients with
non-small-cell lung cancer harbouring mutations of the epidermal
growth factor receptor (WJTOG3405): an open label, randomised phase
3 trial. Lancet Oncol. 11:121–128. 2010.
|
|
7.
|
Shaw AT, Kim DW, Nakagawa K, et al:
Crizotinib versus chemotherapy in advanced ALK-positive lung
cancer. N Engl J Med. 368:2385–2394. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Choi YL, Soda M, Yamashita Y, et al:
EML4-ALK mutations in lung cancer that confer resistance to ALK
inhibitors. N Engl J Med. 363:1734–1739. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Kobayashi S, Boggon TJ, Dayaram T, et al:
EGFR mutation and resistance of non-small-cell lung cancer to
gefitinib. N Engl J Med. 352:786–792. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Engelman JA, Zejnullahu K, Mitsudomi T, et
al: MET amplification leads to gefitinib resistance in lung cancer
by activating ERBB3 signaling. Science. 316:1039–1043. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Marvin MJ, Dahlstrand J, Lendahl U and
McKay RD: A rod end deletion in the intermediate filament protein
nestin alters its subcellular localization in neuroepithelial cells
of transgenic mice. J Cell Sci. 111:1951–1961. 1998.PubMed/NCBI
|
|
12.
|
Sjöberg G, Jiang WQ, Ringertz NR, Lendahl
U and Sejersen T: Colocalization of nestin and vimentin/desmin in
skeletal muscle cells demonstrated by three-dimensional
fluorescence digital imaging microscopy. Exp Cell Res. 214:447–458.
1994.PubMed/NCBI
|
|
13.
|
Lendahl U, Zimmerman LB and McKay RD: CNS
stem cells express a new class of intermediate filament protein.
Cell. 60:585–595. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Esni F, Stoffers DA, Takeuchi T and Leach
SD: Origin of exocrine pancreatic cells from nestin-positive
precursors in developing mouse pancreas. Mech Dev. 121:15–25. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Ishiwata T, Teduka K, Yamamoto T, Kawahara
K, Matsuda Y and Naito Z: Neuroepithelial stem cell marker nestin
regulates the migration, invasion and growth of human gliomas.
Oncol Rep. 26:91–99. 2011.PubMed/NCBI
|
|
16.
|
Akiyama M, Matsuda Y, Ishiwata T, Naito Z
and Kawana S: Inhibition of the stem cell marker nestin reduces
tumor growth and invasion of malignant melanoma. J Invest Dermatol.
133:1384–1387. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Kawamoto M, Ishiwata T, Cho K, et al:
Nestin expression correlates with nerve and retroperitoneal tissue
invasion in pancreatic cancer. Hum Pathol. 40:189–198. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Matsuda Y, Naito Z, Kawahara K, Nakazawa
N, Korc M and Ishiwata T: Nestin is a novel target for suppressing
pancreatic cancer cell migration, invasion and metastasis. Cancer
Biol Ther. 11:512–523. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Kleeberger W, Bova GS, Nielsen ME, et al:
Roles for the stem cell associated intermediate filament Nestin in
prostate cancer migration and metastasis. Cancer Res. 67:9199–9206.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Ishiwata T, Matsuda Y and Naito Z: Nestin
in gastrointestinal and other cancers: effects on cells and tumor
angiogenesis. World J Gastroenterol. 17:409–418. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
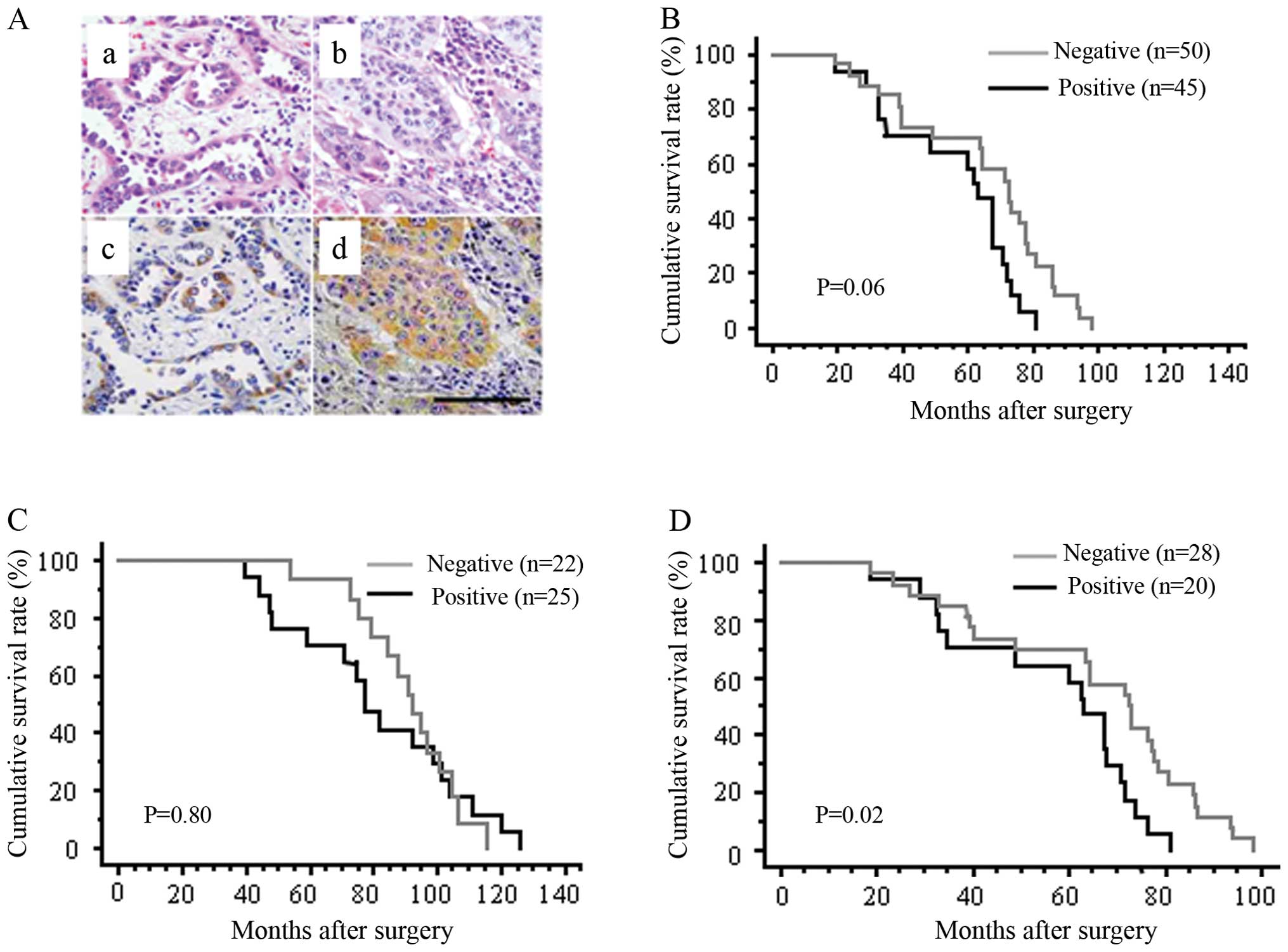
Ryuge S, Sato Y, Wang GQ, et al:
Prognostic significance of nestin expression in resected non-small
cell lung cancer. Chest. 139:862–869. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Ryuge S, Sato Y, Jiang SX, et al:
Prognostic impact of nestin expression in resected large cell
neuroendocrine carcinoma of the lung. Lung Cancer. 77:415–420.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Skarda J, Kolar Z, Janikova M, et al:
Analysis of the prognostic impact of nestin expression in non-small
cell lung cancer. Biomed Pap Med Fac, Univ Palacky Olomouc, Czech
Repub. 156:135–142. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Chen Z, Wang T, Luo H, et al: Expression
of nestin in lymph node metastasis and lymphangiogenesis in
non-small cell lung cancer patients. Hum Pathol. 41:737–744. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Janikova M, Skarda J, Dziechciarkova M, et
al: Identification of CD133+/nestin+ putative
cancer stem cells in non-small cell lung cancer. Biomed Pap Med Fac
Univ Palacky Olomouc Czech Repub. 154:321–326. 2010.
|
|
26.
|
Takakuwa O, Maeno K, Kunii E, et al:
Involvement of intermediate filament nestin in cell growth of
small-cell lung cancer. Lung Cancer. 81:174–179. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Goldstraw P, Crowley J, Chansky K, et al:
The IASLC Lung Cancer Staging Project: proposals for the revision
of the TNM stage groupings in the forthcoming (seventh) edition of
the TNM Classification of malignant tumours. J Thorac Oncol.
2:706–714. 2007. View Article : Google Scholar
|
|
28.
|
Ishiwata T, Matsuda Y, Yamamoto T, Uchida
E, Korc M and Naito Z: Enhanced expression of fibroblast growth
factor receptor 2 IIIc promotes human pancreatic cancer cell
proliferation. Am J Pathol. 180:1928–1941. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Matsuda Y, Ishiwata T, Yamahatsu K, et al:
Overexpressed fibroblast growth factor receptor 2 in the invasive
front of colorectal cancer: a potential therapeutic target in
colorectal cancer. Cancer Lett. 309:209–219. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Yamahatsu K, Matsuda Y, Ishiwata T, Uchida
E and Naito Z: Nestin as a novel therapeutic target for pancreatic
cancer via tumor angiogenesis. Int J Oncol. 40:1345–1357.
2012.PubMed/NCBI
|
|
31.
|
Matsuda Y, Hagio M, Seya T and Ishiwata T:
Fibroblast growth factor receptor 2 IIIc as a therapeutic target
for colorectal cancer cells. Mol Cancer Ther. 11:2010–2020. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Kawase R, Ishiwata T, Matsuda Y, et al:
Expression of fibroblast growth factor receptor 2 IIIc in human
uterine cervical intraepithelial neoplasia and cervical cancer. Int
J Oncol. 36:331–340. 2010.PubMed/NCBI
|
|
33.
|
Arai K, Sakamoto R, Kubota D and Kondo T:
Proteomic approach toward molecular backgrounds of drug resistance
of osteosarcoma cells in spheroid culture system. Proteomics.
13:2351–2360. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Ushijima H and Maeda M: cAMP-dependent
proteolysis of GATA-6 is linked to JNK-signaling pathway. Biochem
Biophys Res Commun. 423:679–683. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35.
|
Huang YL, Wu CM, Shi GY, et al: Nestin
serves as a prosurvival determinant that is linked to the
cytoprotective effect of epidermal growth factor in rat vascular
smooth muscle cells. J Biochem. 146:307–315. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36.
|
Xue XJ and Yuan XB: Nestin is essential
for mitogen-stimulated proliferation of neural progenitor cells.
Mol Cell Neurosci. 45:26–36. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Brognard J, Clark AS, Ni Y and Dennis PA:
Akt/protein kinase B is constitutively active in non-small cell
lung cancer cells and promotes cellular survival and resistance to
chemotherapy and radiation. Cancer Res. 61:3986–3997.
2001.PubMed/NCBI
|
|
38.
|
Guo Y, Du J and Kwiatkowski DJ: Molecular
dissection of AKT activation in lung cancer cell lines. Mol Cancer
Res. 11:282–293. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39.
|
Singh S, Trevino J, Bora-Singhal N, et al:
EGFR/Src/Akt signaling modulates Sox2 expression and self-renewal
of stem-like side-population cells in non-small cell lung cancer.
Mol Cancer. 11:732012. View Article : Google Scholar : PubMed/NCBI
|
|
40.
|
Tanaka S, Kamachi Y, Tanouchi A, Hamada H,
Jing N and Kondoh H: Interplay of SOX and POU factors in regulation
of the Nestin gene in neural primordial cells. Mol Cell Biol.
24:8834–8846. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
41.
|
Dobashi Y, Kimura M, Matsubara H, Endo S,
Inazawa J and Ooi A: Molecular alterations in AKT and its protein
activation in human lung carcinomas. Hum Pathol. 43:2229–2240.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
42.
|
Liu K, Lin B, Zhao M, et al: The multiple
roles for Sox2 in stem cell maintenance and tumorigenesis. Cell
Signal. 25:1264–1271. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
43.
|
Nakatsugawa M, Takahashi A, Hirohashi Y,
et al: SOX2 is overexpressed in stem-like cells of human lung
adenocarcinoma and augments the tumorigenicity. Lab Invest.
91:1796–1804. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44.
|
Sholl LM, Barletta JA, Yeap BY, Chirieac
LR and Hornick JL: Sox2 protein expression is an independent poor
prognostic indicator in stage I lung adenocarcinoma. Am J Surg
Pathol. 34:1193–1198. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
45.
|
Noro R, Gemma A, Kosaihira S, et al:
Gefitinib (IRESSA) sensitive lung cancer cell lines show
phosphorylation of Akt without ligand stimulation. BMC Cancer.
6:2772006. View Article : Google Scholar : PubMed/NCBI
|
|
46.
|
Pao W, Miller VA, Politi KA, et al:
Acquired resistance of lung adenocarcinomas to gefitinib or
erlotinib is associated with a second mutation in the EGFR kinase
domain. PLoS Med. 2:e732005. View Article : Google Scholar : PubMed/NCBI
|
|
47.
|
Nolte SM, Venugopal C, McFarlane N, et al:
A cancer stem cell model for studying brain metastases from primary
lung cancer. J Natl Cancer Inst. 105:551–562. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
48.
|
Clarke MF, Dick JE, Dirks PB, et al:
Cancer stem cells - perspectives on current status and future
directions: AACR Workshop on cancer stem cells. Cancer Res.
66:9339–9344. 2006. View Article : Google Scholar
|
|
49.
|
Eramo A, Lotti F, Sette G, et al:
Identification and expansion of the tumorigenic lung cancer stem
cell population. Cell Death Differ. 15:504–514. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
50.
|
Reya T, Morrison SJ, Clarke MF and
Weissman IL: Stem cells, cancer, and cancer stem cells. Nature.
414:105–111. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
51.
|
Ho MM, Ng AV, Lam S and Hung JY: Side
population in human lung cancer cell lines and tumors is enriched
with stem-like cancer cells. Cancer Res. 67:4827–4833. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
52.
|
Liang D and Shi Y: Aldehyde
dehydrogenase-1 is a specific marker for stem cells in human lung
adenocarcinoma. Med Oncol. 29:633–639. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
53.
|
Alamgeer M, Peacock CD, Matsui W, Ganju V
and Watkins DN: Cancer stem cells in lung cancer: Evidence and
controversies. Respirology. 18:757–764. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
54.
|
Bombí JA, Martínez A, Ramírez J, et al:
Ultrastructural and molecular heterogeneity in non-small cell lung
carcinomas: study of 110 cases and review of the literature.
Ultrastruct Pathol. 26:211–218. 2002.PubMed/NCBI
|
|
55.
|
Kau TR, Schroeder F, Ramaswamy S, et al: A
chemical genetic screen identifies inhibitors of regulated nuclear
export of a Forkhead transcription factor in PTEN-deficient tumor
cells. Cancer Cell. 4:463–476. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
56.
|
Lam EW, Francis RE and Petkovic M: FOXO
transcription factors: key regulators of cell fate. Biochem Soc
Trans. 34:722–726. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
57.
|
Sangodkar J, Dhawan NS, Melville H, et al:
Targeting the FOXO1/KLF6 axis regulates EGFR signaling and
treatment response. J Clin Invest. 122:2637–2651. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
58.
|
Zhang X, Yalcin S, Lee DF, et al: FOXO1 is
an essential regulator of pluripotency in human embryonic stem
cells. Nat Cell Biol. 13:1092–1099. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
59.
|
DeFeo-Jones D, Barnett SF, Fu S, et al:
Tumor cell sensitization to apoptotic stimuli by selective
inhibition of specific Akt/PKB family members. Mol Cancer Ther.
4:271–279. 2005.PubMed/NCBI
|