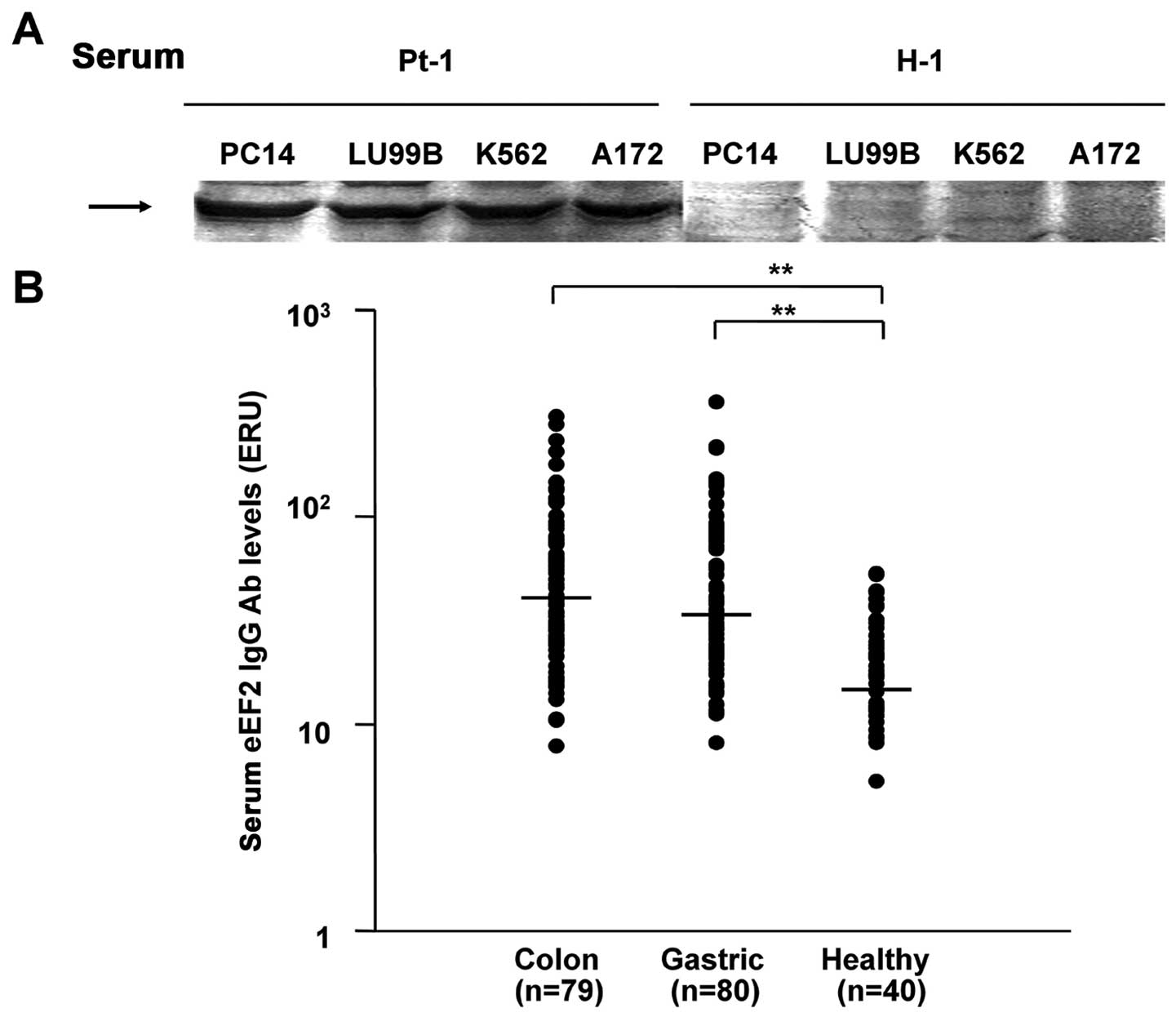
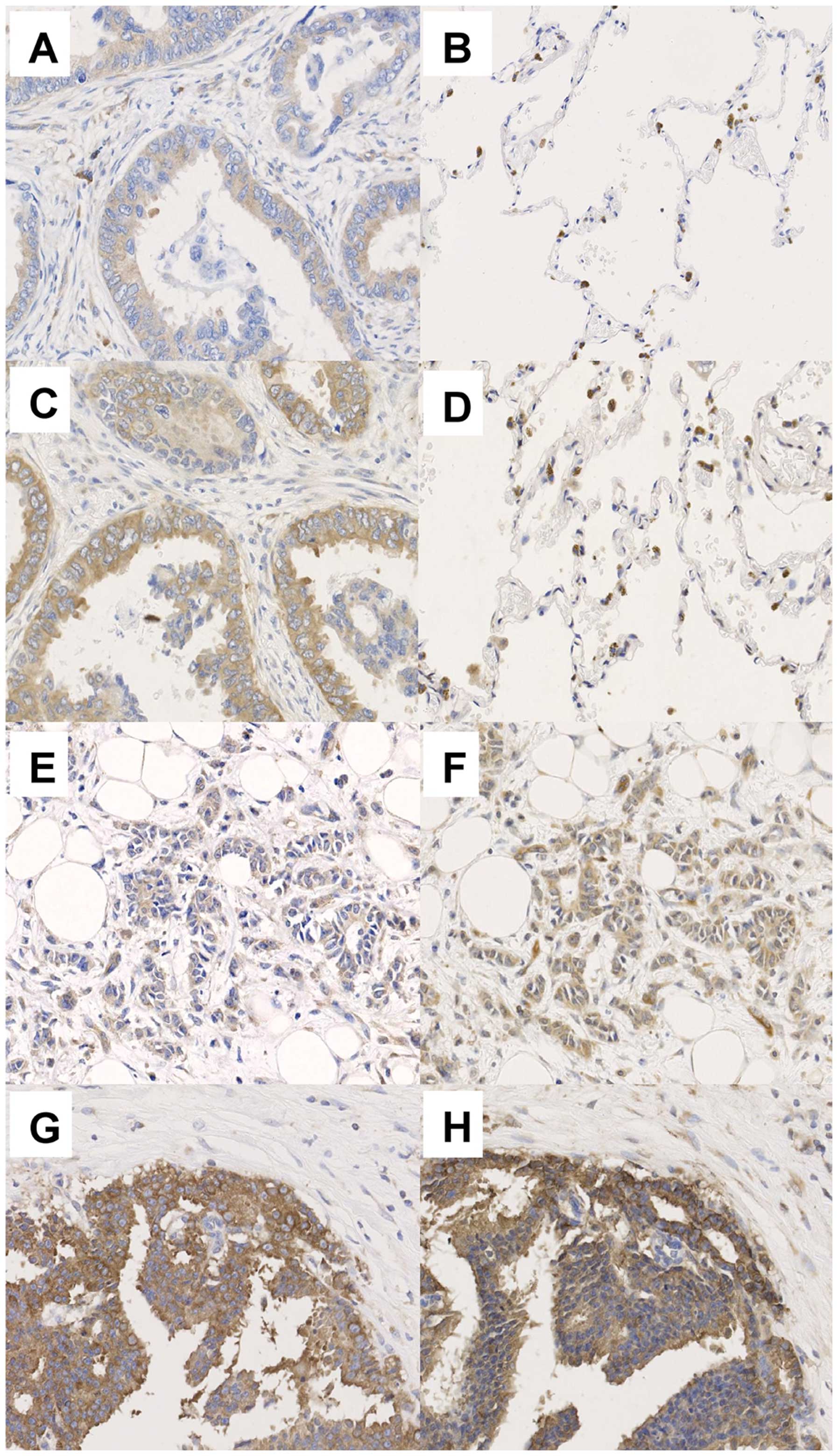
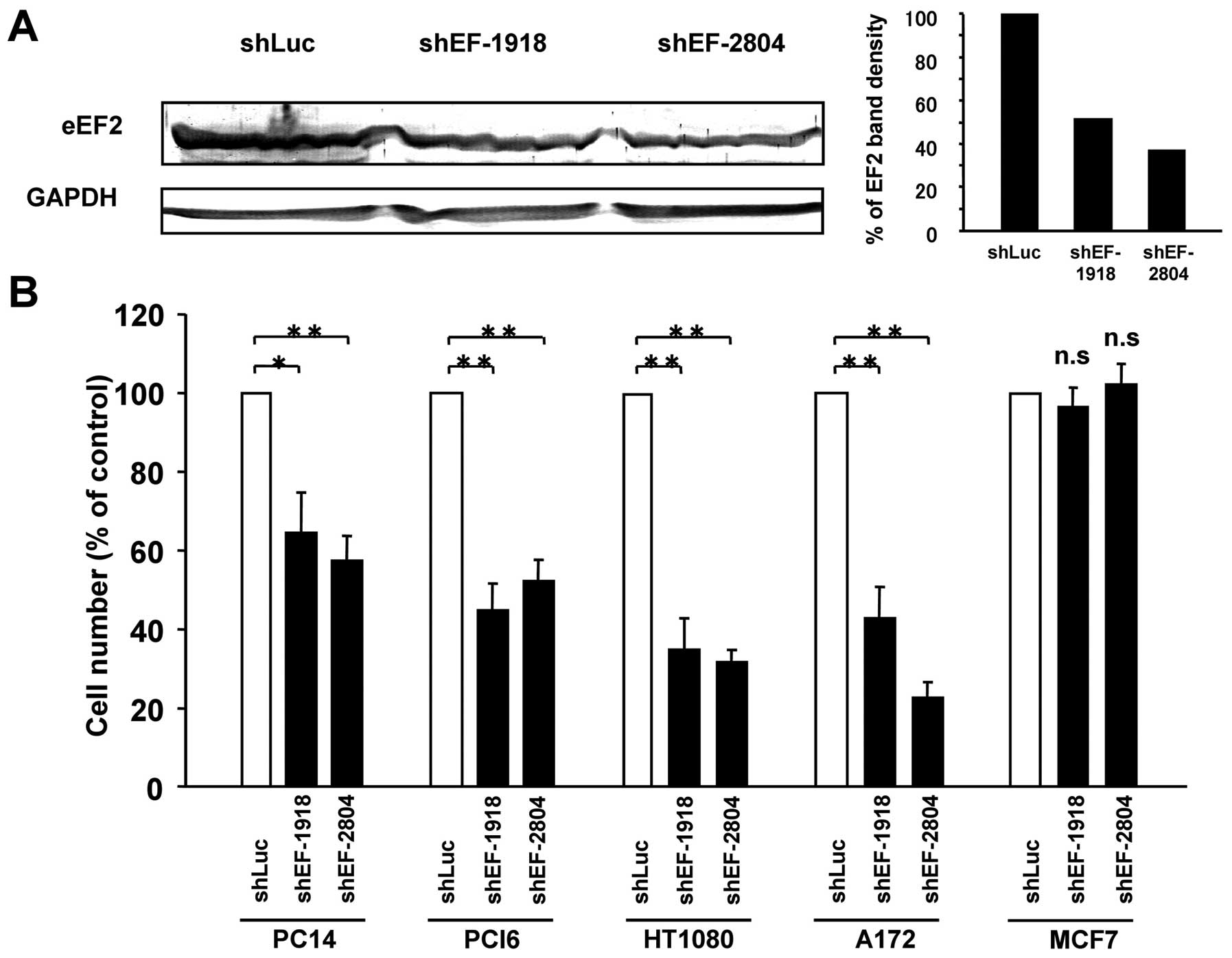
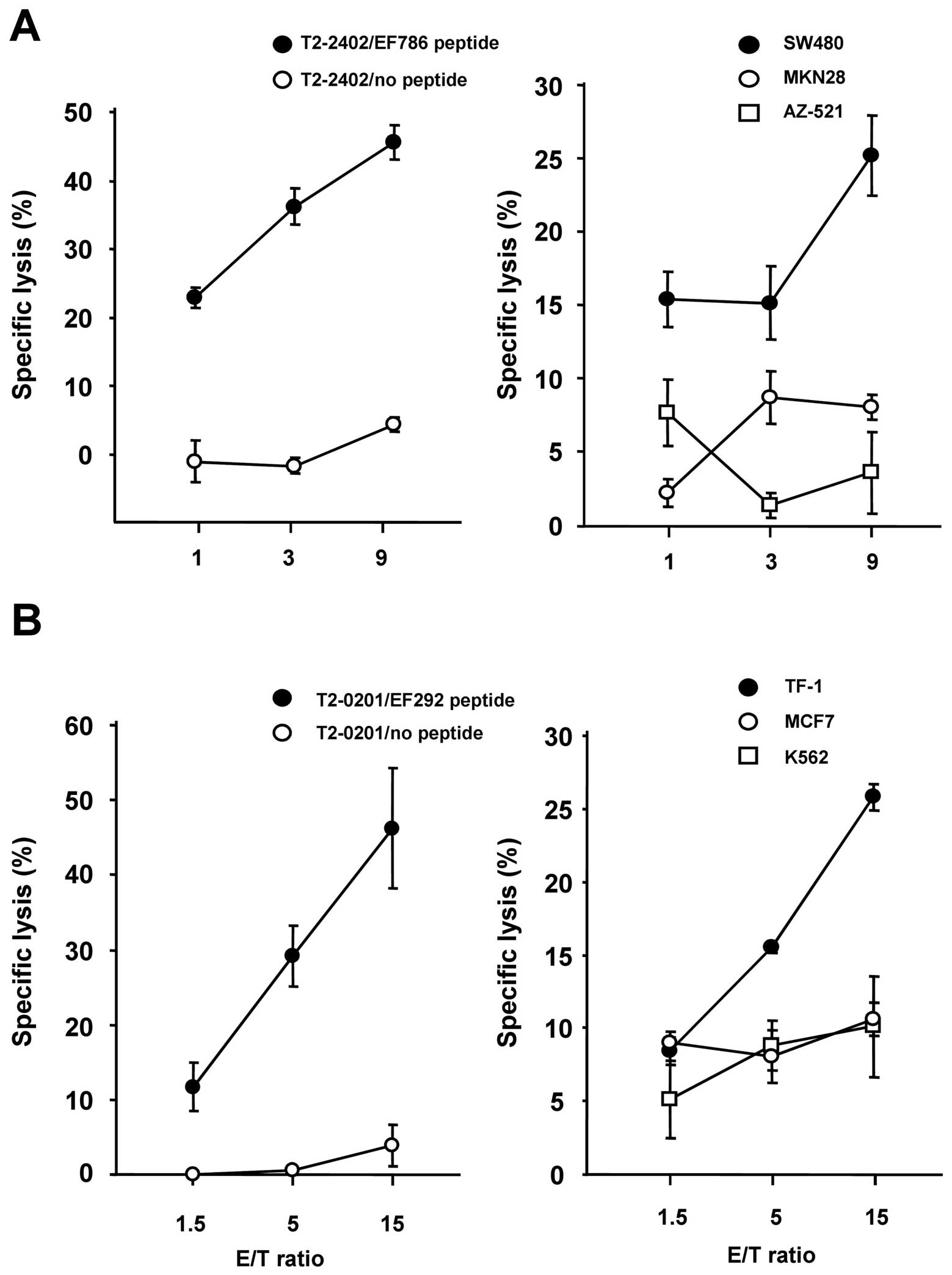
|
1.
|
Lesterhuis WJ, Haanen JB and Punt CJ:
Cancer immunotherapy - revisited. Nat Rev Drug Discov. 10:591–600.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Wright SE: Immunotherapy of breast cancer.
Expert Opin Biol Ther. 12:479–490. 2012. View Article : Google Scholar
|
|
3.
|
Slingluff CL Jr: The present and future of
peptide vaccines for cancer: single or multiple, long or short,
alone or in combination? Cancer J. 17:343–350. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Murala S, Alli V, Kreisel D, Gelman AE and
Krupnick AS: Current status of immunotherapy for the treatment of
lung cancer. J Thorac Dis. 2:237–244. 2010.
|
|
5.
|
Topalian SL, Weiner GJ and Pardoll DM:
Cancer immunotherapy comes of age. J Clin Oncol. 29:4828–4836.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Desmetz C, Mange A, Maudelonde T and
Solassol J: Autoantibody signatures: progress and perspectives for
early cancer detection. J Cell Mol Med. 15:2013–2024. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Murphy MA, O’Leary JJ and Cahill DJ:
Assessment of the humoral immune response to cancer. J Proteomics.
75:4573–4579. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Grzmil M and Hemmings BA: Translation
regulation as a therapeutic target in cancer. Cancer Res.
72:3891–3900. 2012. View Article : Google Scholar
|
|
9.
|
Bilanges B and Stokoe D: Mechanism of
translational deregulation in human tumors and therapeutic
intervention strategies. Oncogene. 26:5973–5990. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Hizli AA, Chi Y, Swanger J, Carter JH,
Liao Y, Welcker M, Ryazanov AG and Clurman BE: Phosphorylation of
eukaryotic elongation factor 2 (eEF2) by cyclin A-cyclin-dependent
kinase 2 regulates its inhibition by eEF2 kinase. Mol Cell Biol.
33:596–604. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
White SJ, Kasman LM, Kelly MM, Lu P,
Spruill L, McDermott PJ and Voelkel-Johnson C: Doxorubicin
generates a proapoptotic phenotype by phosphorylation of EF-2. Free
Radic Biol Med. 43:1313–1321. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Kruiswijk F, Yuniati L, Magliozzi R, Low
TY, Lim R, Bolder R, Mohammed S, Proud CG, Heck AJ, Pagano M and
Guardavaccaro D: Coupled activation and degradation of eEF2K
regulates protein synthesis in response to genotoxic stress. Sci
Signal. 5:ra402012. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Schwer CI, Stoll P, Rospert S, Fitzke E,
Schallner N, Burkle H, Schmidt R and Humar M: Carbon monoxide
releasing molecule-2 CORM-2 represses global protein synthesis by
inhibition of eukaryotic elongation factor eEF2. Int J Biochem Cell
Biol. 45:201–212. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Nakamura J, Aoyagi S, Nanchi I, Nakatsuka
S, Hirata E, Shibata S, Fukuda M, Yamamoto Y, Fukuda I, Tatsumi N,
Ueda T, Fujiki F, Nomura M, Nishida S, Shirakata T, Hosen N, Tsuboi
A, Oka Y, Nezu R, Mori M, Doki Y, Aozasa K, Sugiyama H and Oji Y:
Overexpression of eukaryotic elongation factor eEF2 in
gastrointestinal cancers and its involvement in G2/M
progression in the cell cycle. Int J Oncol. 34:1181–1189.
2009.PubMed/NCBI
|
|
15.
|
Kuzushima K, Hayashi N, Kimura H and
Tsurumi T: Efficient identification of
HLA-A*2402-restricted cytomegalovirus-specific CD8(+)
T-cell epitopes by a computer algorithm and an enzyme-linked
immunospot assay. Blood. 98:1872–1881. 2001.
|
|
16.
|
Oka Y, Elisseeva OA, Tsuboi A, Ogawa H,
Tamaki H, Li H, Oji Y, Kim EH, Soma T, Asada M, Ueda K, Maruya E,
Saji H, Kishimoto T, Udaka K and Sugiyama H: Human cytotoxic T
lymphocyte responses specific for peptides of wild-type Wilms’
tumor gene WT1 product. Immunogenetics. 51:99–107. 2000.
|
|
17.
|
Masuda T, Ide N and Kitabatake N: Effects
of chemical modification of lysine residues on the sweetness of
lysozyme. Chem Senses. 30:253–264. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Oji Y, Kitamura Y, Kamino E, Kitano A,
Sawabata N, Inoue M, Mori M, Nakatsuka S, Sakaguchi N, Miyazaki K,
Nakamura M, Fukuda I, Nakamura J, Tatsumi N, Takakuwa T, Nishida S,
Shirakata T, Hosen N, Tsuboi A, Nezu R, Maeda H, Oka Y, Kawase I,
Aozasa K, Okumura M, Miyoshi S and Sugiyama H: WT1 IgG antibody for
early detection of nonsmall cell lung cancer and as its prognostic
factor. Int J Cancer. 125:381–387. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Morishima S, Akatsuka Y, Nawa A, Kondo E,
Kiyono T, Torikai H, Nakanishi T, Ito Y, Tsujimura K, Iwata K, Ito
K, Kodera Y, Morishima Y, Kuzushima K and Takahashi T:
Identification of an HLA-A24- restricted cytotoxic T lymphocyte
epitope from human papillomavirus type-16 E6: the combined effects
of bortezomib and interferon-gamma on the presentation of a cryptic
epitope. Int J Cancer. 120:594–604. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Chiriva-Internati M, Yu Y, Mirandola L,
D’Cunha N, Hardwicke F, Cannon MJ, Cobos E and Kast WM:
Identification of AKAP-4 as a new cancer/testis antigen for
detection and immunotherapy of prostate cancer. Prostate. 72:12–23.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Qin Z and Blankenstein T: CD4+T
cell-mediated tumor rejection involves inhibition of angiogenesis
that is dependent on IFNγ receptor expression by nonhematopoietic
cells. Immunity. 12:677–686. 2000.
|
|
22.
|
Bogen B, Munthe L, Sollien A, Hofgaard P,
Omholt H, Dagnaes F, Dembic Z and Lauritzsen GF: Naive
CD4+T cells confer idiotype-specific tumor resistance in
the absence of antibodies. Eur J Immunol. 25:3079–3086. 1995.
|
|
23.
|
Lin Y, Fujiki F, Katsuhara A, Oka Y,
Tsuboi A, Aoyama N, Tanii S, Nakajima H, Tatsumi N, Morimoto S,
Tamanaka T, Tachino S, Hosen N, Nishida S, Oji Y, Kumanogoh A and
Sugiyama H: HLA-DPB1*05:01-restricted WT1 332-specific
TCR-transduced CD4+T lymphocytes display a helper
activity for WT1-specific CTL induction and a cytotoxicity against
leukemia cells. J Immunother. 36:159–170. 2013.
|
|
24.
|
Mita AC, Mita MM, Nawrocki ST and Giles
FJ: Survivin: key regulator of mitosis and apoptosis and novel
target for cancer therapeutics. Clin Cancer Res. 14:5000–5005.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Lu CD, Altieri DC and Tanigawa N:
Expression of a novel anti-apoptosis gene, survivin, correlated
with tumor cell apoptosis and p53 accumulation in gastric
carcinomas. Cancer Res. 58:1808–1812. 1998.PubMed/NCBI
|
|
26.
|
Kawasaki H, Altieri DC, Lu CD, Toyoda M,
Tenjo T and Tanigawa N: Inhibition of apoptosis by survivin
predicts shorter survival rates in colorectal cancer. Cancer Res.
58:5071–5074. 1998.PubMed/NCBI
|
|
27.
|
Chakravarti A, Noll E, Black PM,
Finkelstein DF, Finkelstein DM, Dyson NJ and Loeffler JS:
Quantitatively determined survivin expression levels are of
prognostic value in human gliomas. J Clin Oncol. 20:1063–1068.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Wang M, Liu BG, Yang ZY, Hong X and Chen
GY: Significance of survivin expression: Prognostic value and
survival in stage III non-small cell lung cancer. Exp Ther Med.
3:983–988. 2012.PubMed/NCBI
|
|
29.
|
Bria E, Visca P, Novelli F, Casini B,
Diodoro MG, Perrone-Donnorso R, Botti C, Sperduti I, Facciolo F,
Milella M, Cecere FL, Cognetti F and Mottolese M: Nuclear and
cytoplasmic cellular distribution of survivin as survival predictor
in resected non-small-cell lung cancer. Eur J Surg Oncol.
34:593–598. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Tanaka K, Iwamoto S, Gon G, Nohara T,
Iwamoto M and Tanigawa N: Expression of survivin and its
relationship to loss of apoptosis in breast carcinomas. Clin Cancer
Res. 6:127–134. 2000.PubMed/NCBI
|
|
31.
|
Idenoue S, Hirohashi Y, Torigoe T, Sato Y,
Tamura Y, Hariu H, Yamamoto M, Kurotaki T, Tsuruma T, Asanuma H,
Kanaseki T, Ikeda H, Kashiwagi K, Okazaki M, Sasaki K, Sato T,
Ohmura T, Hata F, Yamaguchi K, Hirata K and Sato N: A potent
immunogenic general cancer vaccine that targets survivin, an
inhibitor of apoptosis proteins. Clin Cancer Res. 11:1474–1482.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Kameshima H, Tsuruma T, Torigoe T,
Takahashi A, Hirohashi Y, Tamura Y, Tsukahara T, Ichimiya S,
Kanaseki T, Iwayama Y, Sato N and Hirata K: Immunogenic enhancement
and clinical effect by type-I interferon of anti-apoptotic protein,
survivin-derived peptide vaccine, in advanced colorectal cancer
patients. Cancer Sci. 102:1181–1187. 2011. View Article : Google Scholar
|
|
33.
|
Cebon J, Knights A, Ebert L, Jackson H and
Chen W: Evaluation of cellular immune responses in cancer vaccine
recipients: lessons from NY-ESO-1. Expert Rev Vaccines. 9:617–629.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Kim SH, Lee S, Lee CH, Lee MK, Kim YD,
Shin DH, Choi KU, Kim JY, Park do Y and Sol MY: Expression of
cancer-testis antigens MAGE-A3/6 and NY-ESO-1 in non-small-cell
lung carcinomas and their relationship with immune cell
infiltration. Lung. 187:401–411. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35.
|
Yakirevich E, Sabo E, Lavie O, Mazareb S,
Spagnoli GC and Resnick MB: Expression of the MAGE-A4 and NY-ESO-1
cancer-testis antigens in serous ovarian neoplasms. Clin Cancer
Res. 9:6453–6460. 2003.PubMed/NCBI
|
|
36.
|
Jungbluth AA, Chen YT, Stockert E, Busam
KJ, Kolb D, Iversen K, Coplan K, Williamson B, Altorki N and Old
LJ: Immunohistochemical analysis of NY-ESO-1 antigen expression in
normal and malignant human tissues. Int J Cancer. 92:856–860. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Cheever MA, Allison JP, Ferris AS, Finn
OJ, Hastings BM, Hecht TT, Mellman I, Prindiville SA, Viner JL,
Weiner LM and Matrisian LM: The prioritization of cancer antigens:
a national cancer institute pilot project for the acceleration of
translational research. Clin Cancer Res. 15:5323–5337. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
38.
|
Inoue K, Sugiyama H, Ogawa H, Nakagawa M,
Yamagami T, Miwa H, Kita K, Hiraoka A, Masaoka T, Nasu K, Kyo T,
Dohy H, Nakauchi H, Ishidate T, Akiyama T and Kishimoto T: WT1 as a
new prognostic factor and a new marker for the detection of minimal
residual disease in acute leukemia. Blood. 84:3071–3079.
1994.PubMed/NCBI
|
|
39.
|
Oji Y, Miyoshi S, Maeda H, Hayashi S,
Tamaki H, Nakatsuka S, Yao M, Takahashi E, Nakano Y, Hirabayashi H,
Shintani Y, Oka Y, Tsuboi A, Hosen N, Asada M, Fujioka T, Murakami
M, Kanato K, Motomura M, Kim EH, Kawakami M, Ikegame K, Ogawa H,
Aozasa K, Kawase I and Sugiyama H: Overexpression of the Wilms’
tumor gene WT1 in de novo lung cancers. Int J Cancer. 100:297–303.
2002.
|
|
40.
|
Oji Y, Yamamoto H, Nomura M, Nakano Y,
Ikeba A, Nakatsuka S, Abeno S, Kiyotoh E, Jomgeow T, Sekimoto M,
Nezu R, Yoshikawa Y, Inoue Y, Hosen N, Kawakami M, Tsuboi A, Oka Y,
Ogawa H, Souda S, Aozasa K, Monden M and Sugiyama H: Overexpression
of the Wilms’ tumor gene WT1 in colorectal adenocarcinoma. Cancer
Sci. 94:712–717. 2003.
|
|
41.
|
Oji Y, Nakamori S, Fujikawa M, Nakatsuka
S, Yokota A, Tatsumi N, Abeno S, Ikeba A, Takashima S, Tsujie M,
Yamamoto H, Sakon M, Nezu R, Kawano K, Nishida S, Ikegame K,
Kawakami M, Tsuboi A, Oka Y, Yoshikawa K, Aozasa K, Monden M and
Sugiyama H: Overexpression of the Wilms’ tumor gene WT1 in
pancreatic ductal adenocarcinoma. Cancer Sci. 95:583–587. 2004.
|
|
42.
|
Oji Y, Suzuki T, Nakano Y, Maruno M,
Nakatsuka S, Jomgeow T, Abeno S, Tatsumi N, Yokota A, Aoyagi S,
Nakazawa T, Ito K, Kanato K, Shirakata T, Nishida S, Hosen N,
Kawakami M, Tsuboi A, Oka Y, Aozasa K, Yoshimine T and Sugiyama H:
Overexpression of the Wilms’ tumor gene WT1 in primary astrocytic
tumors. Cancer Sci. 95:822–827. 2004.
|
|
43.
|
Drakos E, Rassidakis GZ, Tsioli P, Lai R,
Jones D and Medeiros LJ: Differential expression of WT1 gene
product in non-Hodgkin lymphomas. Appl Immunohistochem Mol Morphol.
13:132–137. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
44.
|
Schreiber RD, Old LJ and Smyth MJ: Cancer
immunoediting: integrating immunity’s roles in cancer suppression
and promotion. Science. 331:1565–1570. 2011.PubMed/NCBI
|