Introduction
Prostate cancer (PCa) belongs to the most
heterogeneous malignant tumors, both histologically and clinically.
Initially PCa is slow growing, confined to the prostatic gland and
dependent on testosterone for proliferation. Treatment is not
always necessary and in many cases the only course of action is
simply referred to as watchful observation. About a third of the
patients on watchful observation starts an active treatment within
their first 5-years post diagnosis (1,2), and
the younger men (55–69 years) gain more from active screening and
treatment (3,4). However, in patients receiving first
line therapy a recurrent and more aggressive type of PCa occur in
20–40% of cases within 10 years (5). At this point, the tumor can start to
invade the tissues surrounding the prostate gland, and the
situation requires a more aggressive treatment, preferably a
combination of androgen ablation and local irradiation therapy
(6).
Although the combinational therapy can induce a
clinically stable state for 1.5–3 years, the absence of androgen
and the heterogeneity of the tumor can lead to a clonal selection
of androgen independent cells. Androgen-independent PCa cells can
proliferate and survive in the absence of androgen, also they are
less susceptible to the growth suppressing properties of androgen
ablation. Hence, the recurring PCa is typically
androgen-independent, more likely to metastasize, less responsive
to available treatments and give patients a median survival of
12–24 months, depending on applied treatment (7,8).
Previously, 6 different molecular mechanisms have been identified
as being involved in the development of androgen-independence;
hypersensitive pathway, promiscuous pathway, outlaw pathway, bypass
pathway, balance between coactivators and corepressors and PCa stem
cell regeneration (9). This study
focuses on the outlaw pathway which has the capacity to activate
androgen receptor signalling by ligand-independent mechanisms
involving receptor tyrosine kinase (RTK) (10–12).
Human epidermal growth factor receptor type 2 (HER2)
is one of the actors of the outlaw pathway, and its expression was
found in about 20% of analyzed PCa samples in a study involving
2,000 samples, both primary tumors and metastases (13). In androgen diminished environment,
HER2 promotes cell survival, suppresses apoptosis and increases
cell motility, hence being strongly associated with a more advanced
disease and tumor stage (13,14).
This enables a selective survival for HER2 expressing cells leading
to an accelerated tumor progression and androgen-independence
causing an increased resistance to existing therapy (15,16).
Epidermal growth factor receptor (EGFR) is another member of
HER-family and also plays a role in the outlaw pathway. Since HER2
lacks a natural ligand, it needs to dimerize with another family
member to become active, and its preferable partner is EGFR.
Upregulation of HER2 and EGFR is known to be an important aspect in
the development of different types of cancer (17).
In this study, we therefore speculate that HER2
could be a suitable target for treatment of advanced PCa. Several
therapeutics suppressing HER2 activity in cancer cells are now in
clinical use (18,19). In vivo molecular imaging of
HER2 expression in advanced PCa could stratify patients to
anti-HER2 therapy and further monitor therapy outcome (20). Trastuzumab (Herceptin) is an
approved therapeutic monoclonal antibody that binds to the
extracellular domain of HER2 and downregulates its expression
(21). The drug is today in
clinical use for treatment of HER2 expressing breast cancer (BCa)
and studies including the use of trastuzumab in endometrial
(22), peritoneal, pancreatic,
ovarian, stomach (23) and bladder
(24) tumors are ongoing. Heat
shock protein 90 (Hsp90) is a molecular chaperon essential for the
stability and proper folding of proteins involved in e.g. signal
transduction pathways and cell growth. The expression of Hsp90 in
cancer cells is 10-fold higher than in normal cells, and it is
involved in multiple steps of tumor progression, including the
maintenance of HER2. The drug 17-DMAG
(17-(dimethyl-aminoethylamino)-17-demethoxygeldanamycin) is a
water-soluble Hsp90 inhibitor which prevents the intracellular
production of HER2 and simultaneously disrupts multiple signalling
pathways (25). Cetuximab
(Erbitux) is a monoclonal antibody which is today used in treatment
of EGFR expressing colorectal cancer. The drug binds to the
extracellular domain of EGFR and blocks receptor dimerization and
activation, hence promoting receptor downregulation (26). The aim of this study was to
investigate the effect of different RTK-targeting therapy regimes
on survival of PCa cells in vitro. We also hypothesised that
therapy response could correlate with changes in RTK membranous
expression and could be used for therapy monitoring.
Three PCa cell lines with different properties were
selected for this study: LNCap (lymph node metastasis of PCa,
androgen and estrogen receptor positive), PC3 (bone metastasis of
PCa, androgen sensitive), and DU-145 (brain metastasis of PCa,
hormone insensitive). Together these cell lines might reflect tumor
heterogeneity. Membranous expression of HER2 and EGFR in this cell
lines was documented earlier (27,28).
Cells were treated weekly with drugs influencing HER2 and EGFR
expression. Membranous expression of HER2 and EGFR as well as cell
survival in response to therapy was studied. Trastuzumab and
cetuximab which binds to the extracellular domain of HER2 or EGFR,
respectively, and downregulates their expression, and 17-DMAG Hsp90
inhibitor, were selected as drugs for this study.
Materials and methods
The cell lines LNCap, PC3 and DU-145 from the
American Type Culture Collection were provided by LGC Standards.
The cell lines were evaluated for HER2 and EGFR expression earlier
(27,28). Cells were cultivated in complete
RPMI-media, supplemented with 10% fetal bovine serum (FBS), 2 mM
L-glutamate, 100 IU/ml penicillin and 100 μg/ml of streptomycin.
Media for LNCap cells were also supplemented with Na-pyruvate and
HEPES. All reagents including media and trypsin-EDTA were from
Biochrom AG, except Na-pyruvate (Lonza, BioWhittaker). All plastics
for cell culturing were from Corning, and the cells were cultivated
in a humidified incubator with 5% CO2 at 37°C.
For in vitro treatment trastuzumab
(Herceptin, infusion 21 mg/ml, Roche), cetuximab (Erbitux, infusion
5 mg/ml, Merck Serono), and 17-DMAG (Calbiochem) were used.
For HER2 quantification affibody molecule
ZHER2:2395 (Affibody AB) was used, and for
EGFR-cetuximab coupled to CHX-A″DTPA chelator
([(R)-2-Amino-3-(4-isothiocyanatophenyl)
propyl]-trans-(S,S)-cyclohexane-1,2-diamine-pentaacetic acid
Macrocyclics). Radiolabeling of Z2395 with
technetium-99m was performed using the protocol described by
Ahlgren et al (29).
Cetuximab was labeled with indium-111 according to the method
described earlier (28).
Radioactivity was measured with an automated
gamma-counter with 3-inch NaI(Tl) detector (1480 Wizard, Wallac
Oy). Cells were counted using electronic Scepter™ cell counter from
Millipore.
Student’s t-test was used to evaluate the
significance in changes of proliferation and receptor expression.
P-value below 0.05 was considered as significant (indicated by the
star symbol in respective graphs).
Targeted treatment protocol
The cells were treated weekly according to the
following protocol: cells were seeded at 105
cells/culture flask and next day subjected to a regimen of
trastuzumab, 17-DMAG, cetuximab or different combinations
(trastuzumab/17-DMAG, trastuzumab/cetuximab). One group was used as
a control and treated in the same manner as all other cells, but
not subjected to any drugs. The same procedure was repeated weekly.
For all treatments the drugs were diluted in cell cultivation media
to: 0.05 mg/ml trastuzumab, 0.03 mg/ml cetuximab and 0.003 μg/ml
17-DMAG. Receptor quantification was done for groups treated with
trastuzumab, 17-DMAG and trastuzumab/17-DMAG at the termination of
the experiment (after 8 weeks of treatment). All experiments were
performed in quadruplicates.
Receptor quantification
Cells were seeded at 106 cells/ dish 24 h
prior to the experiments. On the day of the experiment an excess
amount of unlabeled Z2395 (60 nM, for quantification of
HER2) or cetuximab (100 nM, for quantification of EGFR) was added
to half of the wells and the cells were left to incubate for 1 h at
room temperature (RT), followed by addition of 0.6 nM
99mTc-labeled Z2395 or 10 nM
111In-labeled cetuximab to all wells and 1 h of
incubation at RT. The cells were then trypsinized, resuspended,
collected and counted according to protocols described earlier
(27,28). The radioactivity in cell samples
was measured in an automated gamma-counter. All experiments were
performed in triplicates.
Results
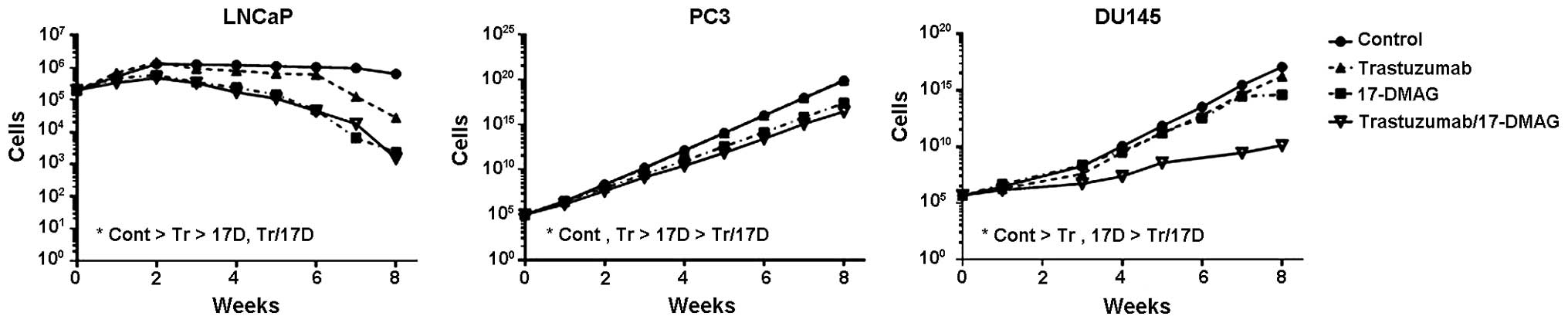
By subjecting cells to a long-term treatment
consisting of sole or combinational drug administration (Fig. 1) we studied the proliferation
inhibitory response. Treated cells varied very much in response
depending on treatment. LNCap cells were not only the most
sensitive to all treatment but also demonstrated inhibited
proliferation in control group, probably due to treatment
protocol.
The HER2 targeting treatment with trastuzumab
demonstrated a significant decrease in proliferation for LNCap and
DU-145 cells, whereas the drug showed a rather pro-proliferative
effect in PC3 cells. Administration of sole Hsp90 inhibitor 17-DMAG
resulted in a significant growth inhibitory effect in all tested
cell lines. Combination therapy using trastuzumab/17-DMAG did not
contribute with any additive growth inhibitory effect in LNCap
cells over sole 17-DMAG; but resulted in an induced growth
inhibition effect for both PC3 and DU-145 cells.
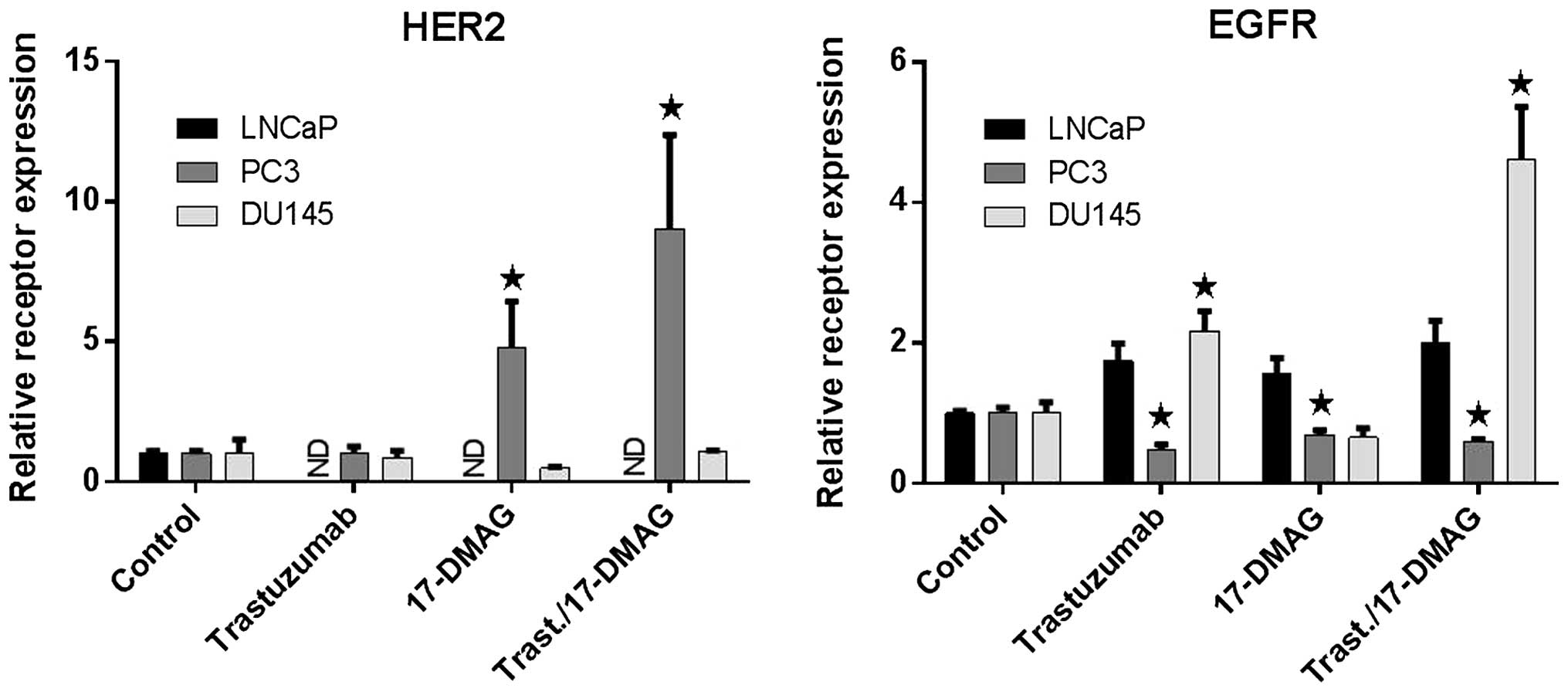
After 8 weeks of treatment, EGFR and HER2 membranous
receptor expression in LNCap, PC3 and DU-145 cells (Fig. 2) were analyzed to evaluate the
response to long-term treatment with trastuzumab, 17-DMAG, and
their combination. As response to all subjected treatments, EGFR
expression in LNCap cells was upregulated: receptor expression
levels were similar for cells subjected to single trastuzumab or
17-DMAG treatment, however, the combination of trastuzumab and
17-DMAG resulted in a 2-fold increase as compared to untreated
cells. The HER2 expression levels were non-detectable in LNCap
cells exposed to drugs. EGFR receptor quantification in PC3 showed
a significant, but rather small, downregulation in expression as a
response to trastuzumab, 17-DMAG and their combination. On the
contrary HER2 expression was upregulated in PC3; 4-fold increase as
a response to 17-DMAG treatment alone, whereas the addition of
trastuzumab to 17-DMAG resulted in a significant 8-fold increase.
In DU-145 cells, a significantly increased expression of EGFR could
be observed for trastuzumab treatment and a significant 3.5-fold
increase for the combination of trastuzumab and 17-DMAG. However,
EGFR expression was not influenced by 17-DMAG sole treatment. HER2
membranous expression in DU-145 cells was not changed in response
to any tested therapies.
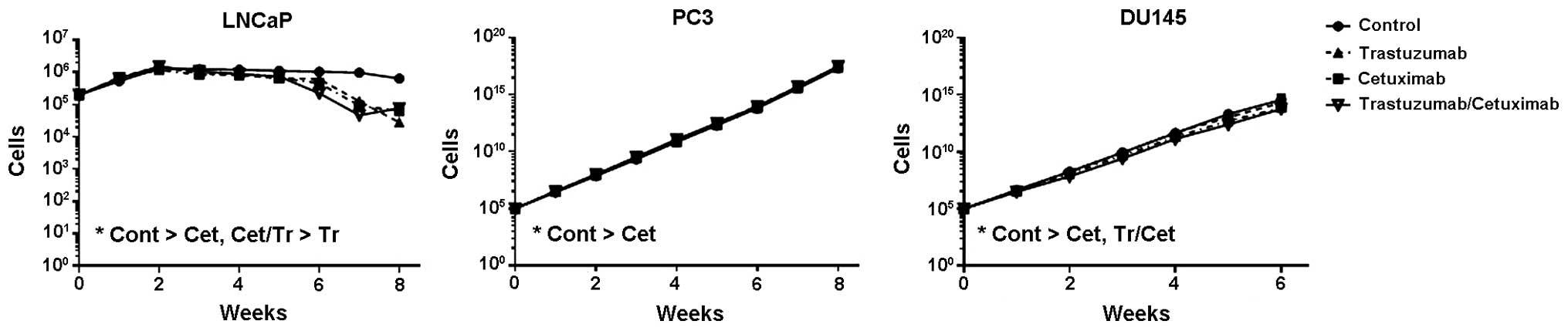
Because of the increased EGFR expression previously
demonstrated in PCa cells as response to trastuzumab/ 17-DMAG
treatment, the cell panel was long-term exposed to sole cetuximab
treatment, or to a trastuzumab/cetuximab combination. LNCap cells
treated with sole cetuximab demonstrated a reduced cell
proliferation in comparison to control cells (Fig. 3). PC3 cells were not affected by
cetuximab treatment, whereas DU-145 cells demonstrated a minor
decrease in cell growth. The addition of trastuzumab showed no or
minor effect in tested cells.
Discussion
HER2 is an RTK which is overexpressed in different
cancer types and is involved in cell division, increased cell
motility and suppression of apoptosis. Previous studies (13,30)
have reported HER2 expression in PCa as well as its role in the
progression of androgen-independent PCa (31). HER2 is therefore considered as a
valid target for treatment of advanced PCa. For this study, we
hypothesized that monitoring of HER2 expression as response to sole
or combinatorial RTK-targeting therapy, could contribute to an
improved personalized therapy.
Tumors are not homogeneous, and the use of one cell
line would not adequately reflect the properties of the disease.
Therefore, three PCa cell lines, LNCap (lymph node metastasis), PC3
(bone metastasis) and DU-145 (brain metastasis), were used in this
study to best represent tumor heterogeneity and disease progression
having different proliferation rates, metastatic potential and rate
of androgen-independence.
To model anti-RTK therapy we selected trastuzumab
targeting HER2 extracellularly, 17-DMAG targeting Hsp90
intracellularly, or their combination. HER2 receptor expression
level in the selected cell lines is 20,000–50,000 receptors/cell
(27) which corresponds to +1 on a
HercepTest (13). This is
considered to be a low expression, as compared to BCa patients
having a strong expression (+3) and receiving anti-HER2 therapy.
However, HER2 expression on the level of +1 is typical for PCa
(13). It has also been taken in
consideration that HER2 expression in PCa is associated with
advanced androgen-independent disease and HER2 as a member of
RTK-family is actively involved in the outlaw pathway of
androgen-resistant PCa. Since HER2 expression in PCa is associated
with the advanced disease stage, its expression cannot be derived
from primary tumor biopsy material. Receptor expression at this
stage could be detected using molecular imaging with PET or SPECT
modalities. Detection of +1 expression in vivo is
challenging, but it has previously been shown that by using highly
sensitive and small protein-based imaging probes (e.g., anti-HER2
affibody molecules), it has become possible to visualize such low
levels of HER2 expression (32–35).
PCa cells receiving trastuzumab treatment alone over
a long period of time demonstrated a different pattern of response
(Fig. 1) in spite of similar HER2
expression in these cell lines (27). The Hsp90 inhibitor 17-DMAG when
used alone affected cell growth significantly in all tested cell
lines. The combination of two treatments, did not result in an
additive decrease in cell proliferation in LNCap cells, but
contributed with an additive effect for PC3 and DU-145 cells. We
can conclude that anti-HER2 treatment using trastuzumab alone did
not result in a strong anti-proliferative effect in tested PCa cell
lines. This is in agreement with therapy efficacy in BCa where
treatment of +1 tumors with trastuzumab is ineffective. However,
combination of trastuzumab and Hsp90 inhibition gave a negative
effect on cell growth in all treated PCa cell lines.
After 8 weeks of treatment HER2 and EGFR expression
of all cells lines were analyzed. We found that even though Hsp90
inhibitor clearly demonstrated a growth reducing effect in all cell
lines, only cells strongly affected by this treatment responded
with downregulation (LNCap) or stable (DU-145) HER2 expression. On
the contrary, PC3 cells that showed a moderate response to 17-DMAG
treatment demonstrated increased HER2 expression. According to the
inhibiting function of 17-DMAG, i.e., prevention of the
intracellular production of HER2 (25,36),
our results contradict literature data. However, previous studies
were made on cell lines with high HER2 expression, and the receptor
levels were estimated using western blot analysis, while we
quantified the membranous expression. Our study set-up also differs
by the drug exposure time, since we studied the receptors
expression after 8 weeks of continuous treatment. We therefore
suggest that cell lines such as PC3 with elevated HER2 expression
post-treatment could be an indication of treatment resistance,
whereas cell lines such as LNCap and DU-145 with increased EGFR
expression and stable or downregulated HER2 expression respond well
to the therapy modality. This observation could be used for therapy
response monitoring.
During long-term treatment with trastuzumab/17-DMAG,
upregulation of EGFR expression was observed in cell lines
responding to all treatments (LNCap and DU-145 cells). This was
especially pronounced in the combination of trastuzumab and
17-DMAG, which resulted in a significant EGFR expression increase
in DU-145 cells (Fig. 2). On the
contrary, PC3 demonstrated a small, but significant decrease in
EGFR expression. EGFR is the preferable partner for HER2
heterodimerization and is also involved in the outlaw pathway of
androgen-independent PCa. The significant upregulation of EGFR
expression in cells responding to anti-HER2 therapy leads us to
speculate on the possibility of using drugs to inhibit the action
from HER2/EGFR complex formation by blockage of several signaling
pathways simultaneously, hence inhibiting PCa cell proliferation.
Monoclonal antibody cetuximab, which is used in treatment of EGFR
expressing colorectal cancer (26), was used in combination with
trastuzumab on our panel of PCa cells, since all cell lines express
both EGFR and HER2 (27,28). The results from EGFR/HER2 targeted
treatment demonstrated that growth of LNCap cells was strongly
affected by sole cetuximab treatment as well as by the addition of
trastuzumab, which also is in agreement with the elevated EGFR
expression as response to trastuzumab treatment. PC3 cells remained
unaffected (Fig. 3). DU-145 cells,
although previously showing an increased EGFR expression, did not
demonstrate any major difference in cell growth when exposed to the
trastuzumab/cetuximab treatments. Moreover, there was no
correlation between initial EGFR expression in tested cell lines
and response to anti-EGFR therapy. LNCap cells having ~40,000
receptors/cells which is 2-fold lower than in PC3 cells and 5-fold
lower than in DU-145 cells (28),
strongly responded to anti-EGFR therapy, when other cells showed
minor response. However, no additive effect from
trastuzumab/cetuximab treatment was observed in any cell line.
This study is a preclinical evaluation using some of
the already available treatments and addresses the fact that all
treatments potentially change the RTK expression profile of the
tumor. Such changes affect the output of the treatment and may also
be used for therapy monitoring.
Simultaneous targeting of different receptors is
already being used in clinics (37) and shows to be beneficial as
compared to single-receptor targeting (38). Concerning PCa, 17-DMAG has been
administered to patients with castrate resistant PCa (39), already on concomitant treatment,
and the outcome was very positive, indicating that combination
treatments could further improve the treatment of late stage of
PCa.
Our data suggest that simultaneous blocking of
action of several receptors and factors involved in the
proliferation of PCa cells can change the phenotypic profile of the
tumor and treatment outcome could be monitored and possibly
improved by observation of such changes. Understanding of molecular
pathways, identification of biomarkers and proper patient
stratification is crucial for the best outcome of targeted
treatment, and we therefore suggest a further investigation into
the significance of RTK expression for patient stratification and
as response to RTK-targeting therapy.
Acknowledgements
This research was financially supported by Swedish
Cancer Society (Cancerfonden) and the Swedish Research Council
(Vetenskapsradet). The authors would like to gratefully acknowledge
Apoteket Farmaci AB, Cytostatikaberedningen Uppsala for providing
this research with trastuzumab and cetuximab, and Affibody AB,
Solna for providing us with Z2395 affibody
molecules.
References
|
1
|
The National Board of Health and Welfare
(Socialstyrelsen). Nationella riktlinjer för bröst-, prostata-,
tjocktarms- och ändtarmscancervård. 2013. ISBN:
978-91-7555-038-12013-2-29 (in Swedish).
|
|
2
|
Sieh W, Lichtensztajn DY, Nelson DO, et
al: Treatment and mortality in men with localized prostate cancer:
a population-based study in California. Open Prost Cancer J. 6:1–9.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Stattin P, Johansson R, Lodnert R, et al:
Geographical variation in incidence of prostate cancer in Sweden.
Scand J Urol Nephrol. 39:372–379. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Carter HB: American Urological Association
(AUA) guideline on prostate cancer detection: process and
rationale. BJU Int. 112:543–547. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kelloff GJ, Choyke P and Coffey DS;
Prostate Cancer Imaging Working Group. Challenges in clinical
prostate cancer: role of imaging. Am J Roentgenol. 192:1455–1470.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Widmark A, Klepp O, Solberg A, et al:
Endocrine treatment, with or without radiotherapy, in locally
advanced prostate cancer (SPCG-7/SFUO-3): an open randomised phase
III trial. Lancet. 373:301–308. 2009. View Article : Google Scholar
|
|
7
|
Tannock IF, de Wit R, Berry WR, et al:
Docetaxel plus prednisone or mitoxantrone plus prednisone for
advanced prostate cancer. New Engl J Med. 351:1502–1512. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Petrylak DP, Tangen CM, Hussain MH, et al:
Docetaxel and estramustine compared with mitoxantrone and
prednisone for advanced refractory prostate cancer. New Engl J Med.
351:1513–1520. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pienta KJ and Bradley D: Mechanisms
underlying the development of androgen-independent prostate cancer.
Clin Cancer Res. 12:1665–1671. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Signoretti S, Montironi R, Manola J, et
al: Her-2-neu expression and progression toward androgen
independence in human prostate cancer. J Natl Cancer Inst.
92:1918–1925. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Craft N, Shostak Y, Carey M and Sawyers
CL: A mechanism for hormone-independent prostate cancer through
modulation of androgen receptor signaling by the HER-2/neu tyrosine
kinase. Nat Med. 5:280–285. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Culig Z, Hobisch A, Cronauer MV, et al:
Androgen receptor activation in prostatic tumor cell lines by
insulin-like growth factor-I, keratinocyte growth factor, and
epidermal growth factor. Cancer Res. 54:5474–5478. 1994.
|
|
13
|
Minner S, Jessen B, Stiedenroth L, et al:
Low level HER2 overexpression is associated with rapid tumor cell
proliferation and poor prognosis in prostate cancer. Clin Cancer
Res. 16:1553–1560. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yarden Y and Sliwkowski MX: Untangling the
ErbB signalling network. Nat Rev Mol Cell Biol. 2:127–137. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
So A, Gleave M, Hurtado-Col A and Nelson
C: Mechanisms of the development of androgen independence in
prostate cancer. World J Urol. 23:1–9. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bacus SS, Altomare DA, Lyass L, et al:
AKT2 is frequently upregulated in HER-2/neu-positive breast cancers
and may contribute to tumor aggressiveness by enhancing cell
survival. Oncogene. 21:3532–3540. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hynes NE and Lane HA: ERBB receptors and
cancer: the complexity of targeted inhibitors. Nat Rev Cancer.
5:341–354. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Vacchelli E, Aranda F, Eggermont A, et al:
Trial Watch: Tumor-targeting monoclonal antibodies in cancer
therapy. Oncoimmunology. 3:e270482014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sharma PS, Sharma R and Tyagi T: Receptor
tyrosine kinase inhibitors as potent weapons in war against
cancers. Curr Pharm Des. 15:758–776. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Altai M, Orlova A and Tolmachev V:
Radiolabeled probes targeting tyrosine-kinase receptors for
personalized medicine. Review invited. Curr Pharm Des.
20:2275–2292. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Albanell J, Codony J, Rovira A, Mellado B
and Gascón P: Mechanism of action of anti-HER2 monoclonal
antibodies: scientific update on trastuzumab and 2C4. Adv Exp Med
Biol. 532:253–268. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
The US National Institutes of Health.
Evaluation of carboplatin/paclitaxel with and without trastuzumab
(Herceptin) in uterine serous cancer. ClinicalTrials.gov.
NCT01367002, 2014-2-13. http://clinicaltrials.gov/ct2/show/NCT01367002?term=NCT01367002&rank=1.
|
|
23
|
The US National Institutes of Health.
Safety study of 212Pb-TCMC-trastuzumab radio
immunotherapy. ClinicalTrials.gov. NCT01384253, 2014-2-13.
http://clinicaltrials.gov/show/NCT01384253.
|
|
24
|
The US National Institutes of Health.
Paclitaxel and radiation therapy with or without trastuzumab in
treating patients who have undergone surgery for bladder cancer.
ClinicalTrials.gov. NCT00238420, 2014-2-13. http://clinicaltrials.gov/ct2/show/study/NCT00238420.
|
|
25
|
Zuehlke A and Johnson JL: Hsp90 and
co-chaperones twist the functions of diverse client proteins.
Biopolymers. 93:211–217. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wheeler DL, Huang S, Kruser TJ, et al:
Mechanisms of acquired resistance to cetuximab: role of HER (ErbB)
family members. Oncogene. 27:3944–3956. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Malmberg J, Tolmachev V and Orlova A:
Imaging agents for in vivo molecular profiling of disseminated
prostate cancer: Cellular processing of [(111)In]-labeled
CHX-A″DTPA-trastuzumab and anti-HER2 ABY-025 Affibody in prostate
cancer cell lines. Exp Ther Med. 2:523–528. 2011.
|
|
28
|
Malmberg J, Tolmachev V and Orlova A:
Imaging agents for in vivo molecular profiling of
disseminated prostate cancer targeting EGFR receptors in prostate
cancer: Comparison of cellular processing of
[111In]-labeled affibody molecule ZEGFR:2377
and cetuximab. Int J Oncol. 38:1137–1143. 2011.
|
|
29
|
Ahlgren S, Orlova A, Wållberg H, et al:
Targeting of HER2-expressing tumors using 111In-ABY-025, a
second-generation affibody molecule with a fundamentally
reengineered scaffold. J Nucl Med. 51:1131–1138. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Baek KH, Hong ME, Jung YY, et al:
Correlation of AR, EGFR, and HER2 expression levels in prostate
cancer: Immunohistochemical analysis and chromogenic in situ
hybridization. Cancer Res Treat. 44:50–56. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Carrion-Salip D, Panosa C, Menendez JA, et
al: Androgen-independent prostate cancer cells circumvent EGFR
inhibition by overexpression of alternative HER receptors and
ligands. Int J Oncol. 41:1128–1138. 2012.PubMed/NCBI
|
|
32
|
Malmberg J, Perols A, Varasteh Z, et al:
Comparative evaluation of synthetic anti-HER2 Affibody molecules
site-specifically labelled with 111In using N-terminal
DOTA, NOTA and NODAGA chelators in mice bearing prostate cancer
xenografts. Eur J Nucl Med Mol Imaging. 39:481–492. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wållberg H, Orlova A, Altai M, et al:
Molecular design and optimization of 99mTc-labeled recombinant
affibody molecules improves their biodistribution and imaging
properties. J Nucl Med. 52:461–469. 2011.PubMed/NCBI
|
|
34
|
Baum RP, Prasad V, Müller D, et al:
Molecular imaging of HER2-expressing malignant tumors in breast
cancer patients using synthetic 111In- or 68Ga-labeled
Affibody molecules. J Nucl Med. 51:892–897. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Sörensen J, Sandberg D, Sandström M, et
al: First-in-human molecular imaging of HER2 expression in breast
cancer metastases using the 111In-ABY-025 Affibody
molecule. J Nucl Med. 55:730–735. 2014.PubMed/NCBI
|
|
36
|
Kramer-Marek G, Kiesewetter DO and Capala
J: Changes in HER2 expression in breast cancer xenografts after
therapy can be quantified using PET and (18)F-labeled affibody
molecules. J Nucl Med. 50:1131–1139. 2009. View Article : Google Scholar
|
|
37
|
Clinic H: Hallwang Clinic Treatments.
2014-2-13. http://www.germancancertreatments.com/treatments-at-hallwang-clinic/.
|
|
38
|
Andersson K: Bringing time into molecular
and cellular biology. J Anal Oncol. 2:652013.
|
|
39
|
Pacey S, Wilson RH, Walton M, et al: A
phase I study of the heat shock protein 90 inhibitor alvespimycin
(17-DMAG) given intravenously to patients with advanced solid
tumors. Clin Cancer Res. 17:1561–1570. 2011. View Article : Google Scholar : PubMed/NCBI
|