Introduction
Cancer metastasis is a highly coordinated and
sequential process. An epithelial-mesenchymal transition (EMT) is
important for disseminating cancer cells by endowing them with
greater motility and invasiveness (1,2).
Transformed cells downregulate epithelial cell marker E-cadherin
whilst upregulating mesenchymal markers N-cadherin and vimentin
(3–6).
MicroRNAs (miRNAs) are a class of small, endogenous
non-coding RNAs that regulate target gene expression through
pairing to its 3′-UTR (7).
Accumulating evidence demonstrates that miRNAs are integral to the
progression and metastasis of many human malignancies (8,9),
including cholangiocarcinoma (CCA).
miR-21 is a major oncogenic miRNA, which is
associated with several biomarkers and therapeutic targets in a
variety of tumor types, such as renal (10), bladder (11), colorectal cancer (12) and others. miR-21 is an important
critical regulator of the biological behavior of cancer cells and
plays a role in apoptosis (10)
and proliferation (13).
Increasing evidence has demonstrated that miR-21 is implicated with
induction and regulation of the EMT program (14–17).
In addition, recent studies have suggested that miR-21 increases
the proliferation, migration and invasion of cancer cells of
various origin through activation of the phosphatidylinositol
3-kinase (PI3K)/protein kinase B (Akt) and mitogen-activated
protein kinase (MAPK)/extracellular signal regulated kinase1/2
(ERK1/2) pathways (18,19), both of which are dysregulated
during the maintenance of EMT (20–22).
Notably, Yan et al (23)
found that PI3K/AKT inhibition by miR-21 knockdown reversed EMT in
breast cancer in vitro.
We previously demonstrated that miR-21 is involved
in the EMT of CCA cells (24).
Krüppel-like factor 4 (KLF4) has been considered to be critical for
EMT (25), however, the
interactions between miR-21 and KLF4 that trigger the EMT of CCA
cells remain unclear. The present study aimed to identify if
miR-21, jointly with KLF4, is involved in the maintenance of EMT of
CCA cells via AKT and ERK1/2 pathways.
Materials and methods
Reagents and antibodies
Human hsa-miR-21 (MIMAT 0000076) antagomir, mimics,
scramble control and negative control were purchased from Guangzhou
Land Unicomed Biotechnology, Co., Ltd. (Guangzhou, China). PCR
primers were synthesized by Sangon Biotech, Co., Ltd. (Shanghai,
China). Real-time PCR assay kits were purchased from Takara Bio
(Dalian, China). Specific siKLF4 duplexes and a scramble control
siRNA sequence (SsiKLF4) were purchased from Guangzhou Land
Unicomed Biotechnology. LY294002 and U0126 were purchased from
Sigma-Aldrich (St. Louis, MO, USA). Lipofectamine 2000 was
purchased from Invitrogen (Carlsbad, CA, USA). Rabbit anti-human
raised against E-cadherin, N-cadherin, vimentin, KLF4, Akt,
phospho-Akt (p-Akt), ERK1/2, phospho-ERK1/2 (p-ERK1/2), GAPDH and
an HRP-conjugated anti-rabbit IgG secondary antibody were purchased
from Cell Signaling Technology (Danvers, MA, USA).
Cell lines and xenografts
Human QBC939 cholangiocarcinoma cells were purchased
from the Cell Bank of the Chinese Academy of Sciences. Cells were
maintained in high-glucose Dulbecco's modified Eagle's medium
(DMEM; Gibco, Waltham, MA, USA), supplemented with 10% fetal bovine
serum (FBS; Zhejiang Tianhang Biotechnology, Co. Ltd., Huzhou,
China), 100 units/ml penicillin, 100 mg/ml streptomycin and 1%
glutamine (Invitrogen) and were grown at 37°C in a humidified
atmosphere of 95% air and 5% CO2. Xenografts, procured
from our previous study (24),
were embedded within consecutive paraffin sections and sliced prior
to immunohistochemical analysis.
Vectors and transfection
Specific siKLF-4 duplexes and a scramble control
siRNA sequence (SsiKLF-4) were designed, synthesized and annealed
by Sigma-Aldrich. The selected RNA duplex (siKLF-4) corresponded to
nucleotide sequences of KLF-4 mRNA sense: 5′-CCC ATC TTT CTC CAC
GTT CG-3′ and antisense: 5′-TGC TTG ATC TTG GGG CAC GT-3′. LY294002
(inhibitor of PI3K/Akt) and U0126 (inhibitor of ERK1/2) were
purchased from Sigma-Aldrich. Human miR-21 mimics, inhibitor and
negative control were purchased from Guangzhou RiboBio, Co., Ltd.
(Guangzhou, China). QBC939 cells were transfected with either: i)
miR-21 mimics (mimic group); ii) miR-21 negative control (control
group); or ii) miR-21 inhibitor (inhibitor group) using
Lipofectamine 2000 transfection reagent (Invitrogen) following the
manufacturer's guidelines. Briefly, 5×105 cells were
seeded into 6-well plates and grown to 60% confluence. Human miR-21
antagomir or its negative control were transfected directly into
QBC939 cells at a final concentration of 50 nmol/l. Human miR-21
mimics or its negative control were allowed to form transfection
complexes with Lipofectamine™ 2000 in Opti-MEMH I serum-free medium
(Invitrogen) at a final concentration of 40 nmol/l. Transfections
were performed in triplicate. The transfection efficiency was
determined by fluorescent microscopy 24 h post transfection.
Finally, the transfected cells were harvested for total RNA and
protein extraction.
Real-time RT-PCR analysis
We investigated the expression of miR-21, KLF4 and
ERK in transfected QBC939 CCA cells. Initially, total RNA was
extracted using TRIzol reagent (Invitrogen) following the
manufacturer's guidelines. Next, RNA was reverse transcribed into
cDNA using a PrimeScripts RT reagent kit (Takara Bio). For the
reverse transcription of miR-21, a miR-21 reverse transcription
(RT) primer (5′-GTC GTA TCC AGT GCA GGG TCC GAG GTA TTC GCA CTG GAT
ACG ACT CAA CA-3′) and quantitative real-time RT-PCR (qPCR) primers
(forward, 5′-GCC CGC TAG CTT ATC AGA CTG ATG-3′ and reverse, 5′-GTG
CAG GGT CCG AGG T-3′) were used. qPCR reactions were carried out
using SYBRH Premix Ex Taq™ II (Takara Bio) using an Applied
Biosystems real-time PCR system (Life Technologies, Carlsbad, CA,
USA). U6 small nuclear RNA was used as an internal control for
miR-21. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) mRNA was
used as an internal control for KLF4 and ERK genes. The following
primers were used: miR-21: forward, 5′-ACA CTC CAG CTG GGT AGC TTA
TCA GAC TGA TG-3′ and reverse, 5′-CTC AAC TGG TGT CGT GGA-3′; U6:
forward, 5′-CTC GCT TCG GCA GCA CA-3′ and reverse, 5′-AAC GCT TCA
CGA ATT TGC GT-3′. PCR reactions were done in triplicate. All
samples were normalized to internal controls and fold changes were
calculated using the relative quantification method
(2−ΔΔCT).
Western blotting
Western blotting of five proteins was conducted as
described in our previous study (24). Briefly, cells and xenografts were
washed in phosphate-buffered saline (PBS) and incubated in lysis
buffer (Sangon Biotech, Co., Ltd., Shanghai, China). Protein was
separated by 12% SDS-PAGE and transferred to a PVDF membrane
(Sangon Biotech). Non-specific binding sites were blocked by
incubation with TBST containing 5% (w/v) non-fat dried milk. Next,
the membrane was incubated with rabbit anti-human Akt, p-Akt,
KLF-4, ERK or p-ERK (1:1,000 dilution) primary antibodies at 37°C
for 1 h. Subsequently, membranes were incubated overnight with an
HRP-conjugated anti-rabbit IgG secondary antibody (1:1,000
dilution) at 4°C. Signals were visualized by an ECL
chemiluminescence detection kit and semi-quantitated using ImageJ
software. Equal protein loading was assessed by the expression of
GAPDH.
Immunohistochemical staining
Tissues were obtained from mouse xenografts as
previously described (24), and
were fixed in formalin, embedded in paraffin and sectioned (2
μm). Slides were stained with KLF-4, p-Akt and p-ERK
antibodies, and developed with a streptavidin-peroxidase (S-P) kit
(Fuzhou Maixin Biotechnology Development, Co., Ltd., Fuzhou,
China). The proportion of positive cells was determined using five
×200 magnification fields per slide.
Migration and invasion assays
Migration and invasion of cells were determined with
the Transwell chamber assay (8-μm pore size; Millipore,
Billerica, MA, USA) completed as per the manufacturer's
instructions. To determine invasion, the chamber was coated with 80
μl Matrigel (BD Biosciences, San Jose, CA, USA) hydrated
with 50 μl serum-free medium. Next, 200 μl
transfected cell suspension (1×105 cells/ml) was added
to the upper chamber, and media supplemented with 30% fetal bovine
serum (FBS) was added to the bottom wells. After culturing for 24
h, cells were fixed for 15 min in 4% formaldehyde and stained with
1% crystal violet. Cell numbers were counted under an optical
microscope. Cell migration was also evaluated using the Transwell
chamber assay with the exception that the chamber was not coated
with Matrigel. Each experiment was repeated at least three
times.
Construction and transfection of
siKLF4
Based on the human KLF4 mRNA sequence (NM_000314),
specific siRNA duplexes were designed, synthesized and annealed by
Land Unicomed Biotechnology. The selected RNA duplex (siKLF4)
corresponding to nucleotides 1567-1585 of KLF4 mRNA was defined as
sense: 5′-GCU ACC UGU UAA AGA AUC AdTdT-3′ and antisense: 5′-UGA
UUC UUU AAC AGG UAG CdTdT-3′. The scramble-control siRNA sequence
(SsiKLF4) was also designed by Land Unicomed Biotechnology, and has
no significant homology to any known human gene sequence. Prior to
transfection, 5×105 cells were seeded into 6-well plates
and grown to 70% confluence. Human siKLF4 or SsiKLF4 (final
concentration of 50 nmol/l) was allowed to form transfection
complexes with the Lipofectamine 2000 reagent (Invitrogen) in
Opti-MEMH I serum-free medium (Invitrogen) as per the
manufacturer's guidelines.
Scratch wound migration assay
For the scratch wound migration assay,
1×106 cells were seeded into 6-well plates and grown to
60% confluence. Next, the cells were transfected with a miR-21
mimic and co-incubated with LY294002 or U0126 inhibitors. A 'wound'
was then created by scratching the cell cultures with a plastic
filter tip. The cells were rinsed three times with PBS and
incubated at 37°C. The average distance that cells migrated after
0, 24 and 48 h was determined using an inverted microscope with ×8
vision. Migration distance was calculated by image processing using
Prolog software. From the data, the migration rate = (1-distance at
indicated time/distance at 0 h)%. Experiments were performed in
triplicate.
Statistical analysis
All data are expressed as the mean ± SD. Data were
analyzed using SPSS V20.0 software. The Student's t-test was used
to determine statistical differences between treatment groups. A
P≤0.05 was considered statistically significant.
Results
Transfected miR-21 and its inhibitor
change the expression of miR-21 in QBC939 cells
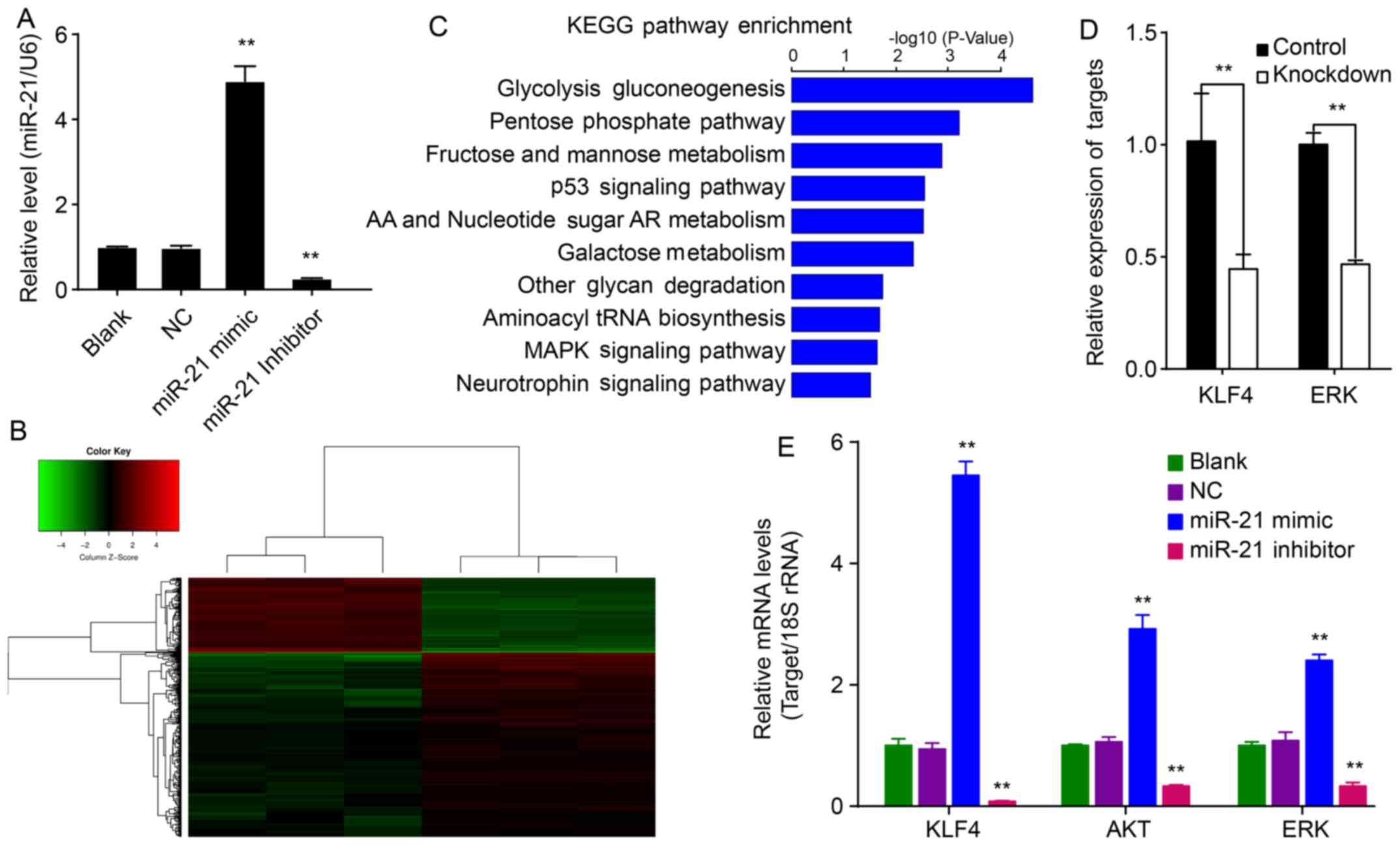
To evaluate whether miR-21 mimics and inhibitors can
modify the expression of miR-21, QBC939 cells were transfected with
a miR-21 mimic or inhibitor for 48 h after which the relative
expression of miR-21 was measured by RT-PCR. The relative
expression of miR-21 was significantly upregulated in QBC939/miR-21
cells relative to NC cells (P<0.01; Fig. 1A). However, the relative expression
of miR-21 was significantly downregulated in QBC939/miR-21
inhibitor cells relative to NC cells (P<0.01; Fig. 1A). These results suggested that
hsa-miR-21 upregulates expression of miR-21 within QBC939 cells,
whereas the hsa-miR-21 inhibitor could downregulate miR-21 in
QBC939/miR-21 inhibitor cells.
Inhibition of miR-21 downregulates KLF4
in QBC939 cells
To investigate the role of miR-21 on EMT, QBC939
cells transfected with the hsa-miR-21 inhibitor or control
differentially expressed genes and EMT-related genes were analyzed
by Human GeneChip PrimeView. The data showed that 400 genes were
differentially expressed (115 upregulated and 285 downregulated
genes) in miR-21-knockdown cells compared to NC cells (Fig. 1B). These differentially expressed
genes were mainly enriched for the glycolysis gluconeogenesis,
pentose phosphate, fructose and mannose metabolism and p53
signaling pathways (Fig. 1C).
Further analysis of these genes using IPA software showed that
genes KLF4 and ERK are related to EMT. KLF4 and ERK
expression were verified by qPCR (Fig.
1E).
miR-21 augments levels of KLF4, Akt and
ERK in QBC939 cells
To investigate the mechanism of miR-21 on EMT in
QBC939 cells, the relative mRNA expression of KLF4, Akt and ERK was
determined. miR-21 increased mRNAs levels of KLF4, Akt and ERK
relative to corresponding control cells (P<0.01; Fig. 1E). Conversely, the miR-21 inhibitor
decreased KLF4, Akt and ERK levels (P<0.01). These results
suggest that miR-21 affects mRNA expression of KLF4, Akt and
ERK.
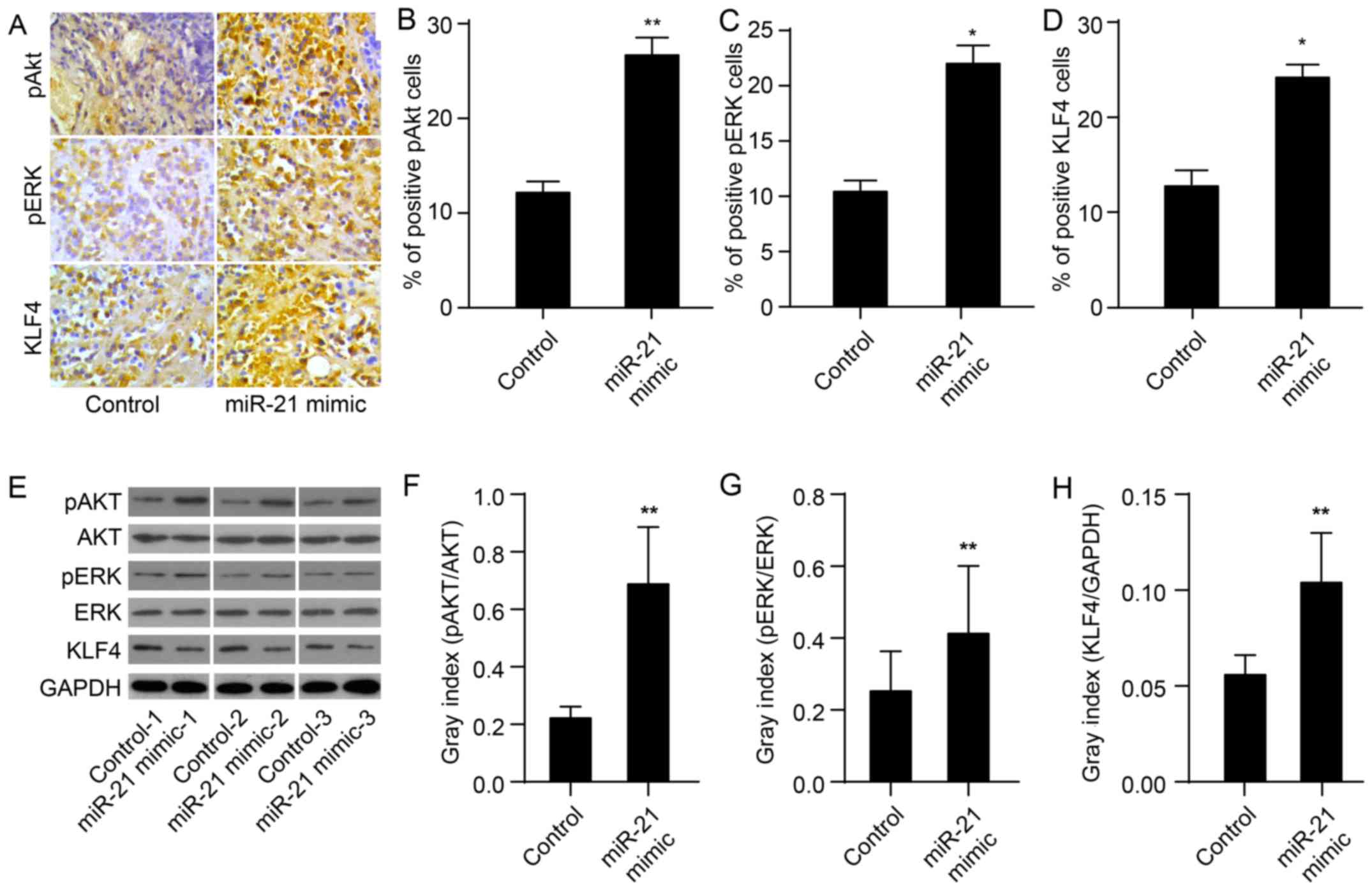
miR-21 increases expression of KLF4, and
Akt and ERK activation in xenografts
To evaluate whether miR-21 upregulates the
expression of KLF4 and of Akt and ERK activation, we used
immunohistochemistry and western blotting to determine miR-21
protein levels in tissues obtained from mouse xeno-grafts as
previously described (24). As
expected, the protein expression of KLF4 in the miR-21 mimic group
was markedly upregulated compared to the control group (P<0.01;
Fig. 2A, D and I). In the miR-21
mimic group, levels of pAkt and pERK were also significantly
upregulated (P<0.01; Fig. 2A–C and
E–G). These results demonstrate that miR-21 mimics upregulate
the expression of KLF4 and activate Akt and ERK.
miR-21 antagonism accompanies KLF4
knockdown inactivated AKT and ERK1/2
Akt and ERK1/2 are two major components of signaling
pathways that are involved in the regulation of cell proliferation,
migration and survival. To determine the signaling molecules
involved in the antagonism of KLF4 and miR-21-induced EMT
phenotype, protein levels of phosphorylated Akt (p-Akt), Akt,
phosphorylated ERK1/2 (p-ERK1/2) and ERK1/2 were determined by
western blot analysis. The protein levels of p-Akt and p-ERK1/2 in
QBC939/anti-KLF4/miR-21 inhibitor cells were markedly lower
compared to levels in anti-KLF4 cells (P<0.01; Fig. 3A–C). miR-21 antagonism, in
combination with KLF4 knockdown, could reverse EMT via suppressing
Akt and ERK1/2 activation.
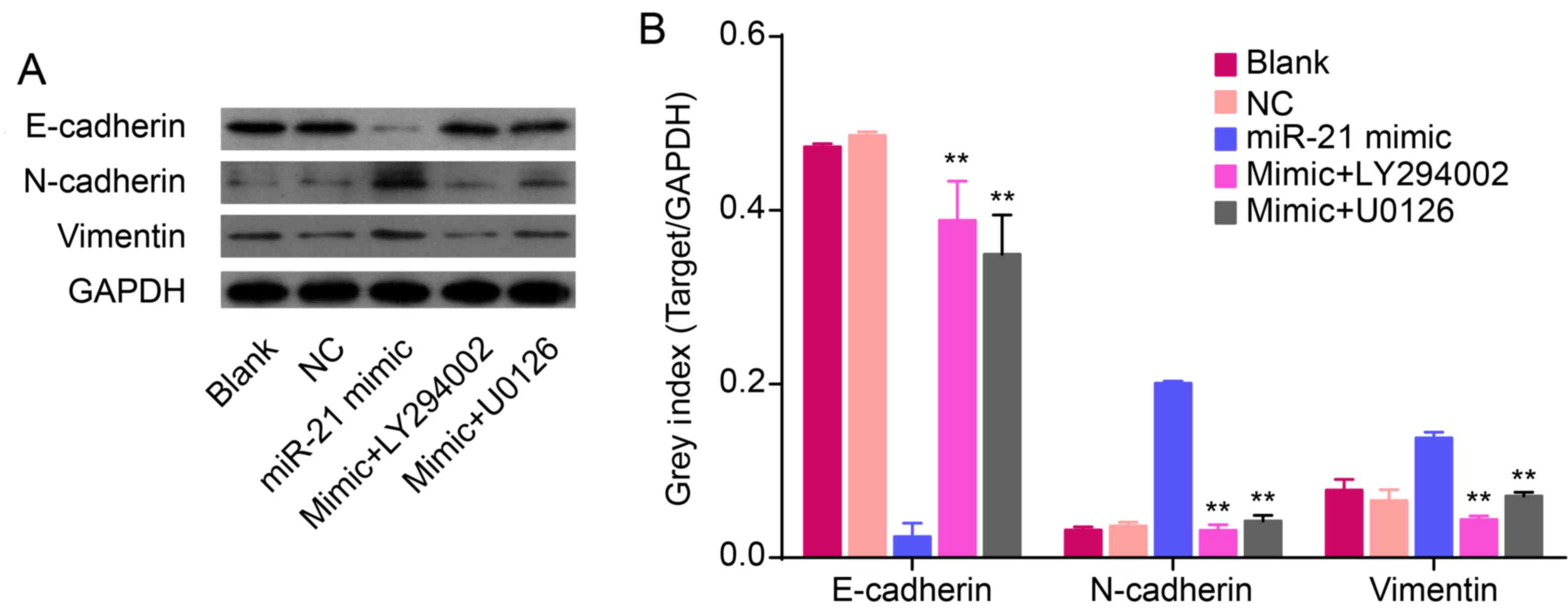
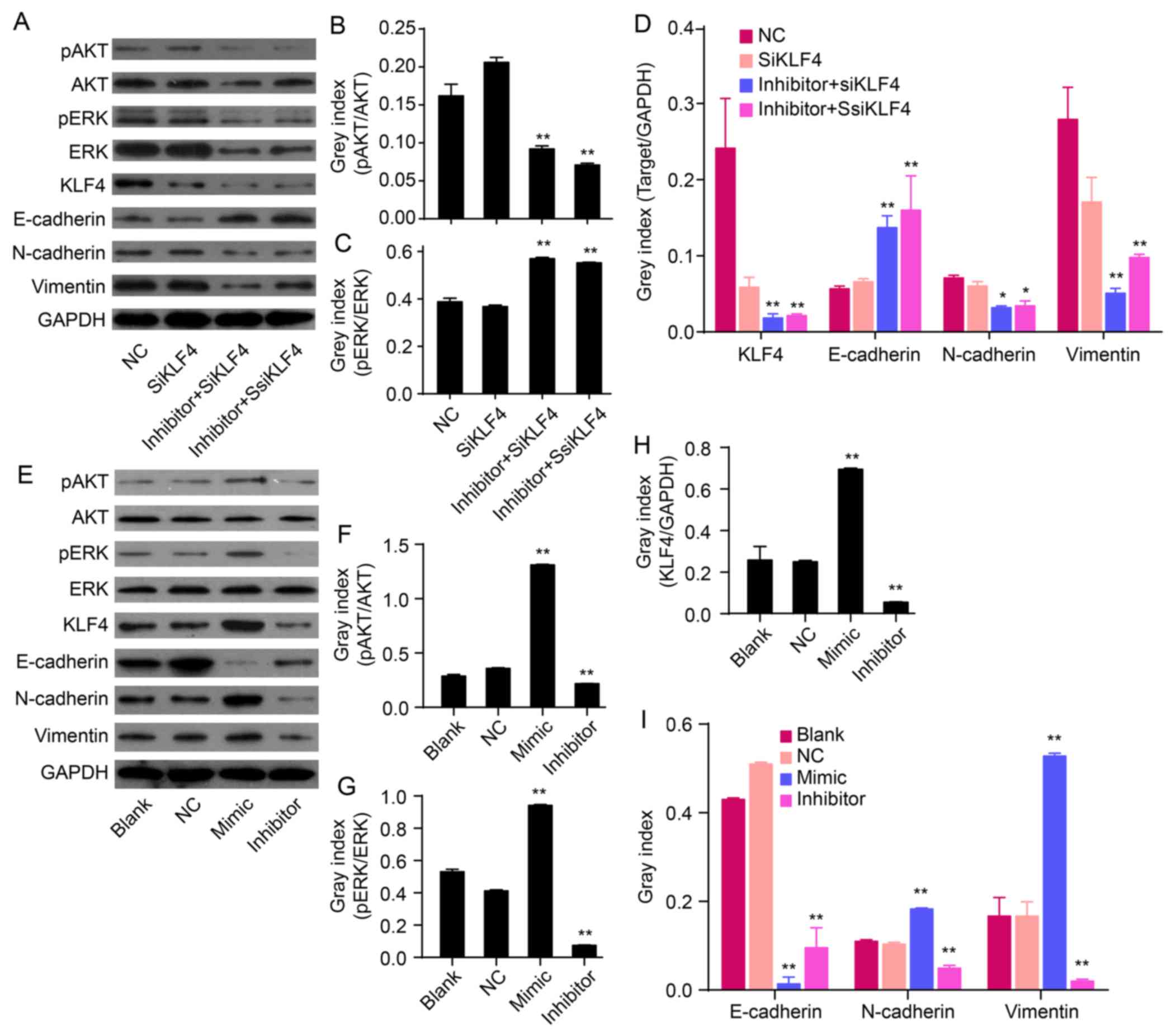 | Figure 3Effects of miR-21 on the expression of
KLF4, E-cadherin, N-cadherin, vimentin and/or Akt/ERK1/2 pathways.
(A) The relative levels of p-Akt, Akt, p-ERK, ERK, KLF4 and EMT
marker proteins were measured by western blot analysis. (B-D) Bands
were semi-quantified using Quantity One software. (E) Western blot
analysis showing protein levels of p-Akt, Akt, p-ERK1/2, ERK1/2,
KLF4, mesenchymal markers (N-cadherin and vimentin) and epithelial
cell marker (E-cadherin). (F-I) Bands were semi-quantified using
Quantity One software. GAPDH was used as loading control
(**P<0.01). |
Overexpression of miR-21 induces an EMT
phenotype accompanied with upregulation of KLF4 and activation of
AKT and ERK1/2
To confirm the role of miR-21 in regulating an EMT
phenotype, hsa-miR-21 mimic or inhibitor were transfected into
QBC939 cells. The protein expression of EMT biomarkers (N-cadherin,
vimentin and E-cadherin), KLF4, p-Akt, Akt, p-ERK1/2 and ERK1/2
were measured by western blot analysis. Compared to NC cells,
overexpression of miR-21 increased the protein expression of
N-cadherin and vimentin, but decreased the expression of E-cadherin
(P<0.01; Fig. 3E and I). In
addition, low expression of miR-21, induced by the miR-21
inhibitor, decreased the levels of N-cadherin and vimentin and
increased the expression of E-cadherin (P<0.01; Fig. 3E and I). This suggests that
overexpression of miR-21 induces an EMT phenotype in QBC939 cells.
Overexpression of miR-21 increased the expression of KLF4
(P<0.01; Fig. 3E and H). In
addition, re-expression of miR-21 increased the expression of p-Akt
and p-ERK1/2 in QBC939 cells (P<0.01; Fig. 3F and G), thereby suggesting that an
overexpression of miR-21 could activate Akt and ERK1/2 pathways.
Together, these data imply that miR-21 regulates EMT and is related
to alterations in KLF4 expression and Akt/ERK1/2 pathways.
miR-21 regulates an EMT phenotype and
increases invasion and migration through AKT and ERK1/2
pathways
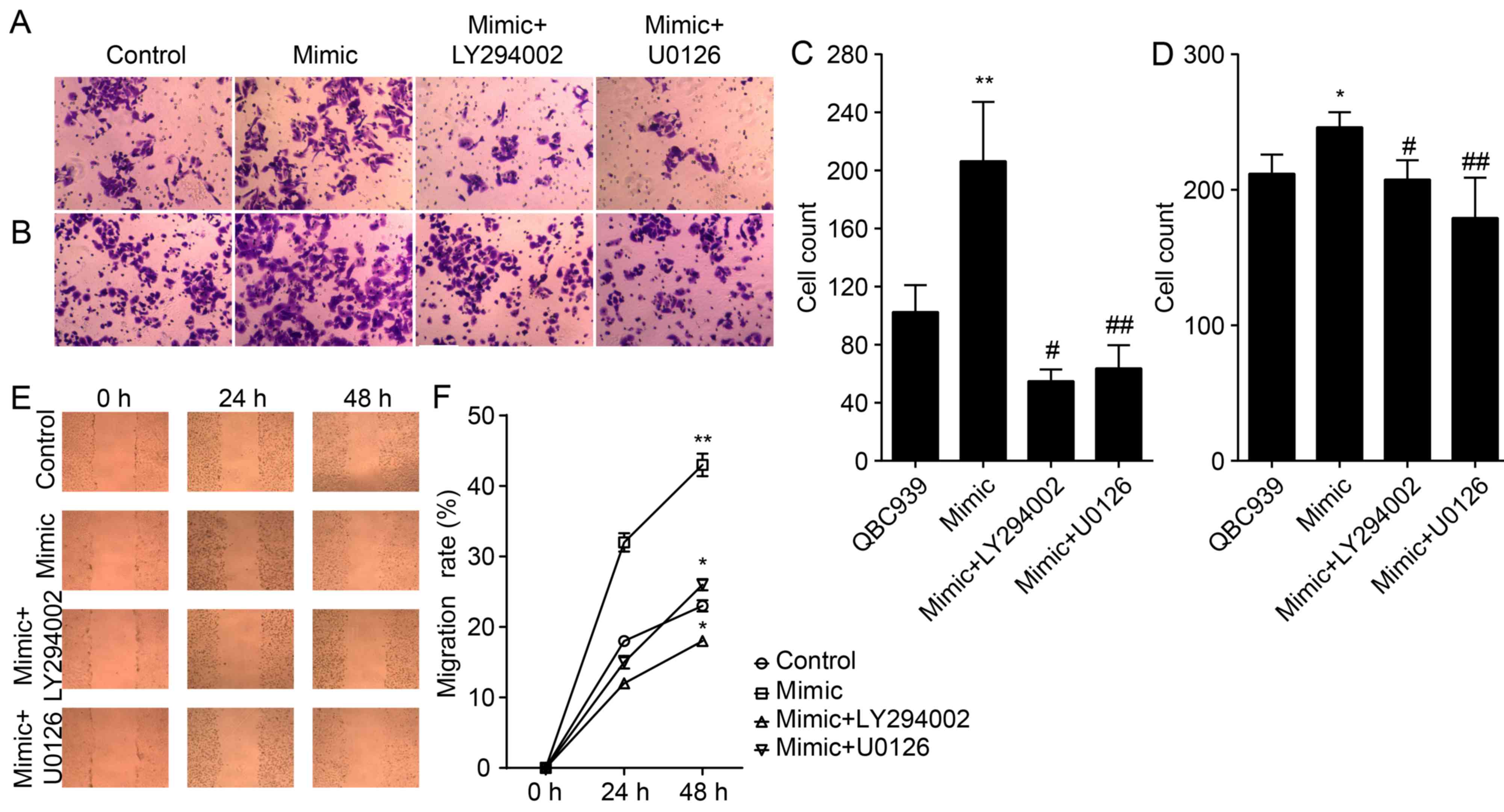
To further investigate the involvement of Akt and
ERK1/2 pathways in miR-21 regulating an EMT, QBC939 cells were
transfected with miR-21 mimics for 72 h. Cells were treated for 24
h with either the LY294002 inhibitor of Akt or the U0126 inhibitor
of ERK1/2. Our data confirm that treatment with LY294002 and U0126
resulted in abolished overexpression of miR-21, induced inhibition
of E-cadherin, and activation of N-cadherin and vimentin
(P<0.01; Fig. 4). Thus, we have
demonstrated that miR-21 overexpression and its ability to induce
the EMT phenotype were inhibited by LY294002 or U0126 in QBC939
cells.
We also studied the effect of antagonizing miR-21 on
the migration and invasion of CCA cells (Fig. 5A, B and E). The number of migrated
and invaded QBC939/anti-miR-21 cells were significantly lower
compared to that of NC cells (P<0.01; Fig. 5C, D and F), thereby suggesting that
miR-21 antagonists reduced the migration and invasion of CCA
cells.
Discussion
Increasing evidence suggests that the expression of
miR-21 is upregulated in various cancer types, including
cholangiocarcinoma (CCA), where it is frequently associated with
EMT (15,16,26).
Overexpression of miR-21 in CCA biopsies is predictive for a poor
outcome (24), and is accompanied
with low expression of E-cadherin and high expression of N-cadherin
and vimentin (27–29). In our previous study, we showed
that miR-21 complicates the EMT of CCA cells. We also showed that
that CCA xenografts have high levels of N-cadherin and vimentin and
low levels of E-cadherin (24). In
the present study, we first identified genes that are
differentially expressed in a CCA xenograft model transfected with
miR-21 knockdown or control lentivirus, and found that KLF4
and ERK were significantly downregulated. Both KLF4
and ERK genes are associated with EMT. Tiwari et al
(25) previously showed that
KLF4 was significantly downregulated during EMT of breast
cancer cells. However, the role of KLF4 in the EMT of CCA cells has
not been elucidated. Accordingly, we studied the effect of miR-21
on the expression of KLF4, its role in the EMT, and investigated
the potential underlying mechanisms of action. We used
immunohistochemistry and western blot analysis to show that, in
QBC939 CCA cells, overexpression of miR-21 promoted upregulation of
KLF4 and expression of Akt and ERK proteins. Therefore, our data
suggest that miR-21 and KLF4 regulate EMT through AKT/ERK1/2
pathways.
To investigate the roles of miR-21 and KLF4 in
regulating the EMT phenotype of CCA cells, miR-21 mimics or
inhibitors were transfected into QBC939 cells. In addition, KLF4
was silenced by using siKLF4. The data showed that the EMT
phenotype was maintained by miR-21 mimics and reversed by miR-21
inhibitor. The results are consistent with previous studies, in
which was shown that miR-21 facilitated EMT of pancreatic cancers
(15,30), thyroid (16) and CCA origin and decreased
activation of Akt and ERK1/2 pathways (24). We also demonstrated that the
downregulation of KLF4 by using RNAi reversed the EMT and
suppressed the activation of Akt and ERK1/2 signaling. Studies have
demonstrated that Akt and/or ERK1/2 pathways are required for
augmenting the EMT of several different cancer cell lines,
including MCF-7 breast cancer cells (20), breast epithelial cells (21,22),
cervical cancer cells (20),
ovarian carcinoma cells (31) and
colon cancer cells (32). These
results demonstrate that miR-21 and KLF4 play an important role in
mediating EMT of CCA cells through the AKT and ERK1/2 pathway.
Finally, we evaluated the invasion and migration of
QBC939 cells. Overexpression of miR-21 by miR-21 mimics promoted
the activation of Akt and ERK1/2 pathways and resulted in an
increase in invasive ability and migration rate. However, these
effects were suppressed by treatment of the cells with PI3K-Akt or
the ERK1/2 inhibitor LY294002 or U0126.
In summary, our data demonstrate that miR-21 and
KLF4 synergistically regulated EMT of QBC939 CCA cells through Akt
and ERK1/2 pathways. The present study illustrated a mechanism by
which miR-21 and KLF4 maintain an EMT within CCA cells, which may
be beneficial for the development of novel therapies to treat
cancer.
Acknowledgments
The present study was supported by the National
Natural Science Foundation of China (no. 81272397) and the Research
Project of Anhui Natural Science for Colleges and Universities
(KJ2013Z143).
References
|
1
|
Tsai JH and Yang J: Epithelial-mesenchymal
plasticity in carcinoma metastasis. Genes Dev. 27:2192–2206. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wells A, Chao YL, Grahovac J, Wu Q and
Lauffenburger DA: Epithelial and mesenchymal phenotypic switchings
modulate cell motility in metastasis. Front Biosci (Landmark Ed).
16:815–837. 2011. View
Article : Google Scholar
|
|
3
|
Zeisberg M and Neilson EG: Biomarkers for
epithelial-mesenchymal transitions. J Clin Invest. 119:1429–1437.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Thiery JP and Sleeman JP: Complex networks
orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell
Biol. 7:131–142. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Christiansen JJ and Rajasekaran AK:
Reassessing epithelial to mesenchymal transition as a prerequisite
for carcinoma invasion and metastasis. Cancer Res. 66:8319–8326.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial-mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wen KC, Sung PL, Yen MS, Chuang CM, Liou
WS and Wang PH: MicroRNAs regulate several functions of normal
tissues and malignancies. Taiwan J Obstet Gynecol. 52:465–469.
2013. View Article : Google Scholar
|
|
8
|
Olson P, Lu J, Zhang H, Shai A, Chun MG,
Wang Y, Libutti SK, Nakakura EK, Golub TR and Hanahan D: MicroRNA
dynamics in the stages of tumorigenesis correlate with hallmark
capabilities of cancer. Genes Dev. 23:2152–2165. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Farazi TA, Horlings HM, Ten Hoeve JJ,
Mihailovic A, Halfwerk H, Morozov P, Brown M, Hafner M, Reyal F,
van Kouwenhove M, et al: MicroRNA sequence and expression analysis
in breast tumors by deep sequencing. Cancer Res. 71:4443–4453.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhou X, Zhang J, Jia Q, Ren Y, Wang Y, Shi
L, Liu N, Wang G, Pu P, You Y, et al: Reduction of miR-21 induces
glioma cell apoptosis via activating caspase 9 and 3. Oncol Rep.
24:195–201. 2010.PubMed/NCBI
|
|
11
|
Zhang HH, Qi F, Cao YH, Zu XB and Chen MF:
Expression and clinical significance of microRNA-21, maspin and
vascular endothelial growth factor-C in bladder cancer. Oncol Lett.
10:2610–2616. 2015.PubMed/NCBI
|
|
12
|
Fukushima Y, Iinuma H, Tsukamoto M,
Matsuda K and Hashiguchi Y: Clinical significance of microRNA-21 as
a biomarker in each Dukes' stage of colorectal cancer. Oncol Rep.
33:573–582. 2015.
|
|
13
|
Zhou X, Ren Y, Moore L, Mei M, You Y, Xu
P, Wang B, Wang G, Jia Z, Pu P, et al: Downregulation of miR-21
inhibits EGFR pathway and suppresses the growth of human
glioblastoma cells independent of PTEN status. Lab Invest.
90:144–155. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kothari AN, Mi Z, Zapf M and Kuo PC: Novel
clinical therapeutics targeting the epithelial to mesenchymal
transition. Clin Transl Med. 3:352014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bao B, Wang Z, Ali S, Kong D, Li Y, Ahmad
A, Banerjee S, Azmi AS, Miele L and Sarkar FH: Notch-1 induces
epithelial-mesenchymal transition consistent with cancer stem cell
phenotype in pancreatic cancer cells. Cancer Lett. 307:26–36. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Braun J, Hoang-Vu C, Dralle H and
Hüttelmaier S: Down-regulation of microRNAs directs the EMT and
invasive potential of anaplastic thyroid carcinomas. Oncogene.
29:4237–4244. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kumarswamy R, Volkmann I, Jazbutyte V,
Dangwal S, Park DH and Thum T: Transforming growth factor-β-induced
endothelial-to-mesenchymal transition is partly mediated by
microRNA-21. Arterioscler Thromb Vasc Biol. 32:361–369. 2012.
View Article : Google Scholar
|
|
18
|
Weng LP, Smith WM, Brown JL and Eng C:
PTEN inhibits insulin-stimulated MEK/MAPK activation and cell
growth by blocking IRS-1 phosphorylation and IRS-1/Grb-2/Sos
complex formation in a breast cancer model. Hum Mol Genet.
10:605–616. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Han M, Liu M, Wang Y, Chen X, Xu J, Sun Y,
Zhao L, Qu H, Fan Y and Wu C: Antagonism of miR-21 reverses
epithelial-mesenchymal transition and cancer stem cell phenotype
through AKT/ERK1/2 inactivation by targeting PTEN. PLoS One.
7:e395202012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li J and Zhou BP: Activation of β-catenin
and Akt pathways by Twist are critical for the maintenance of EMT
associated cancer stem cell-like characters. BMC Cancer. 11:492011.
View Article : Google Scholar
|
|
21
|
Iliopoulos D, Polytarchou C,
Hatziapostolou M, Kottakis F, Maroulakou IG, Struhl K and Tsichlis
PN: MicroRNAs differentially regulated by Akt isoforms control EMT
and stem cell renewal in cancer cells. Sci Signal. 2:ra622009.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Irie HY, Pearline RV, Grueneberg D, Hsia
M, Ravichandran P, Kothari N, Natesan S and Brugge JS: Distinct
roles of Akt1 and Akt2 in regulating cell migration and
epithelial-mesenchymal transition. J Cell Biol. 171:1023–1034.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yan LX, Liu YH, Xiang JW, Wu QN, Xu LB,
Luo XL, Zhu XL, Liu C, Xu FP, Luo DL, et al: PIK3R1 targeting by
miR-21 suppresses tumor cell migration and invasion by reducing
PI3K/AKT signaling and reversing EMT, and predicts clinical outcome
of breast cancer. Int J Oncol. 48:471–484. 2016.
|
|
24
|
Liu Z, Jin ZY, Liu CH, Xie F, Lin XS and
Huang Q: MicroRNA-21 regulates biological behavior by inducing EMT
in human cholangiocarcinoma. Int J Clin Exp Pathol. 8:4684–4694.
2015.PubMed/NCBI
|
|
25
|
Tiwari N, Meyer-Schaller N, Arnold P,
Antoniadis H, Pachkov M, van Nimwegen E and Christofori G: Klf4 is
a transcriptional regulator of genes critical for EMT, including
Jnk1 (Mapk8). PLoS One. 8:e573292013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zheng G, Li N, Jia X, Peng C, Luo L, Deng
Y, Yin J, Song Y, Liu H, Lu M, et al: MYCN-mediated miR-21
overexpression enhances chemo-resistance via targeting CADM1 in
tongue cancer. J Mol Med (Berl). 94:1129–1141. 2016. View Article : Google Scholar
|
|
27
|
Huang Q, Liu L, Liu CH, You H, Shao F, Xie
F, Lin XS, Hu SY and Zhang CH: MicroRNA-21 regulates the invasion
and metastasis in cholangiocarcinoma and may be a potential
biomarker for cancer prognosis. Asian Pac J Cancer Prev.
14:829–834. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu CZ, Liu W, Zheng Y, Su JM, Li JJ, Yu
L, He XD and Chen SS: PTEN and PDCD4 are bona fide targets of
microRNA-21 in human cholangiocarcinoma. Chin Med Sci J. 27:65–72.
2012.PubMed/NCBI
|
|
29
|
Selaru FM, Olaru AV, Kan T, David S, Cheng
Y, Mori Y, Yang J, Paun B, Jin Z, Agarwal R, et al: MicroRNA-21 is
overexpressed in human cholangiocarcinoma and regulates programmed
cell death 4 and tissue inhibitor of metalloproteinase 3.
Hepatology. 49:1595–1601. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bao B, Ali S, Kong D, Sarkar SH, Wang Z,
Banerjee S, Aboukameel A, Padhye S, Philip PA and Sarkar FH:
Anti-tumor activity of a novel compound-CDF is mediated by
regulating miR-21, miR-200, and PTEN in pancreatic cancer. PLoS
One. 6:e178502011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Latifi A, Abubaker K, Castrechini N, Ward
AC, Liongue C, Dobill F, Kumar J, Thompson EW, Quinn MA, Findlay
JK, et al: Cisplatin treatment of primary and metastatic epithelial
ovarian carcinomas generates residual cells with mesenchymal stem
cell-like profile. J Cell Biochem. 112:2850–2864. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang YK, Zhu YL, Qiu FM, Zhang T, Chen ZG,
Zheng S and Huang J: Activation of Akt and MAPK pathways enhances
the tumorigenicity of CD133+ primary colon cancer cells.
Carcinogenesis. 31:1376–1380. 2010. View Article : Google Scholar : PubMed/NCBI
|