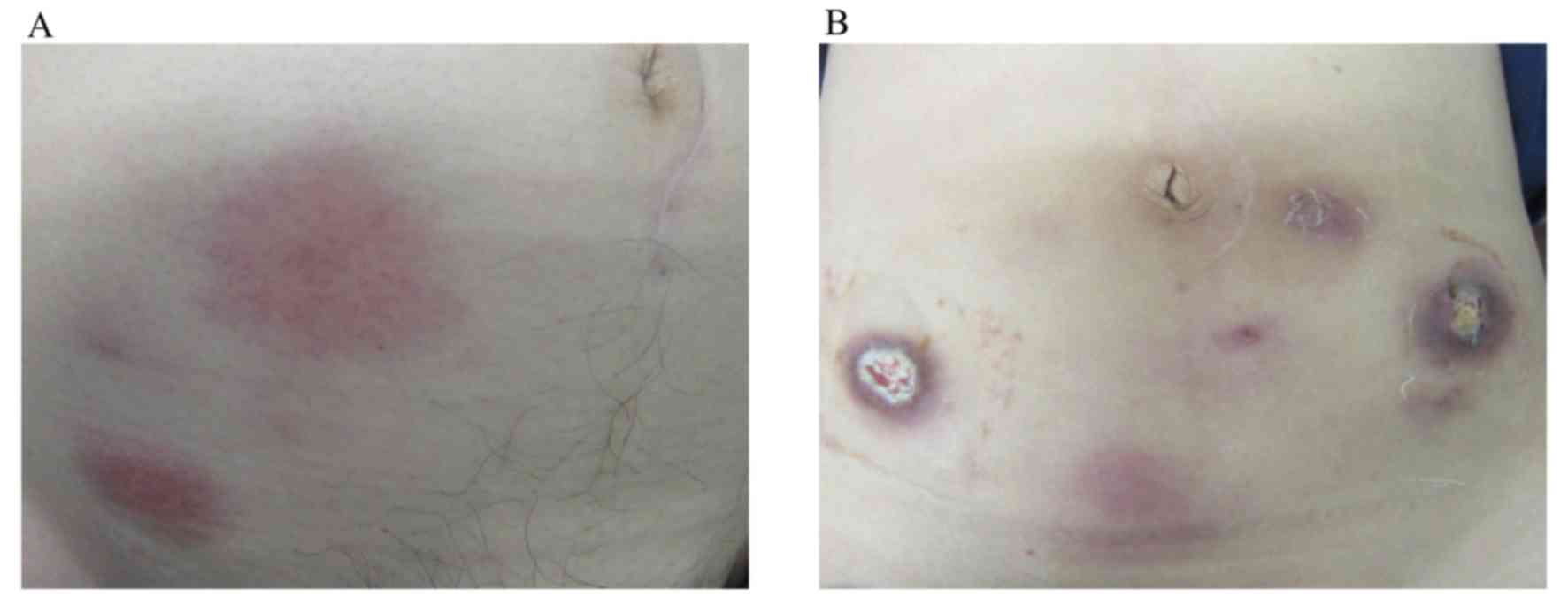
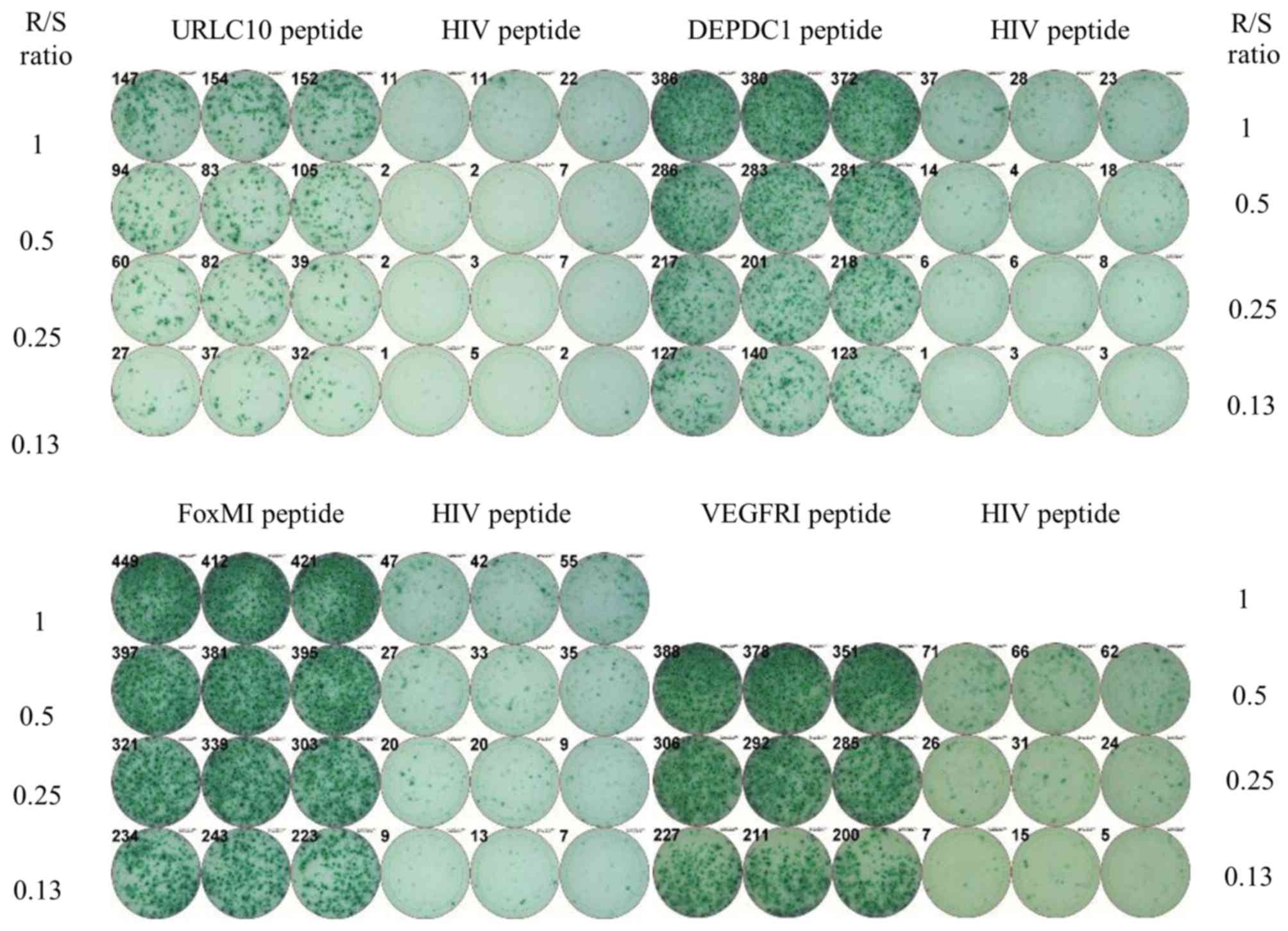
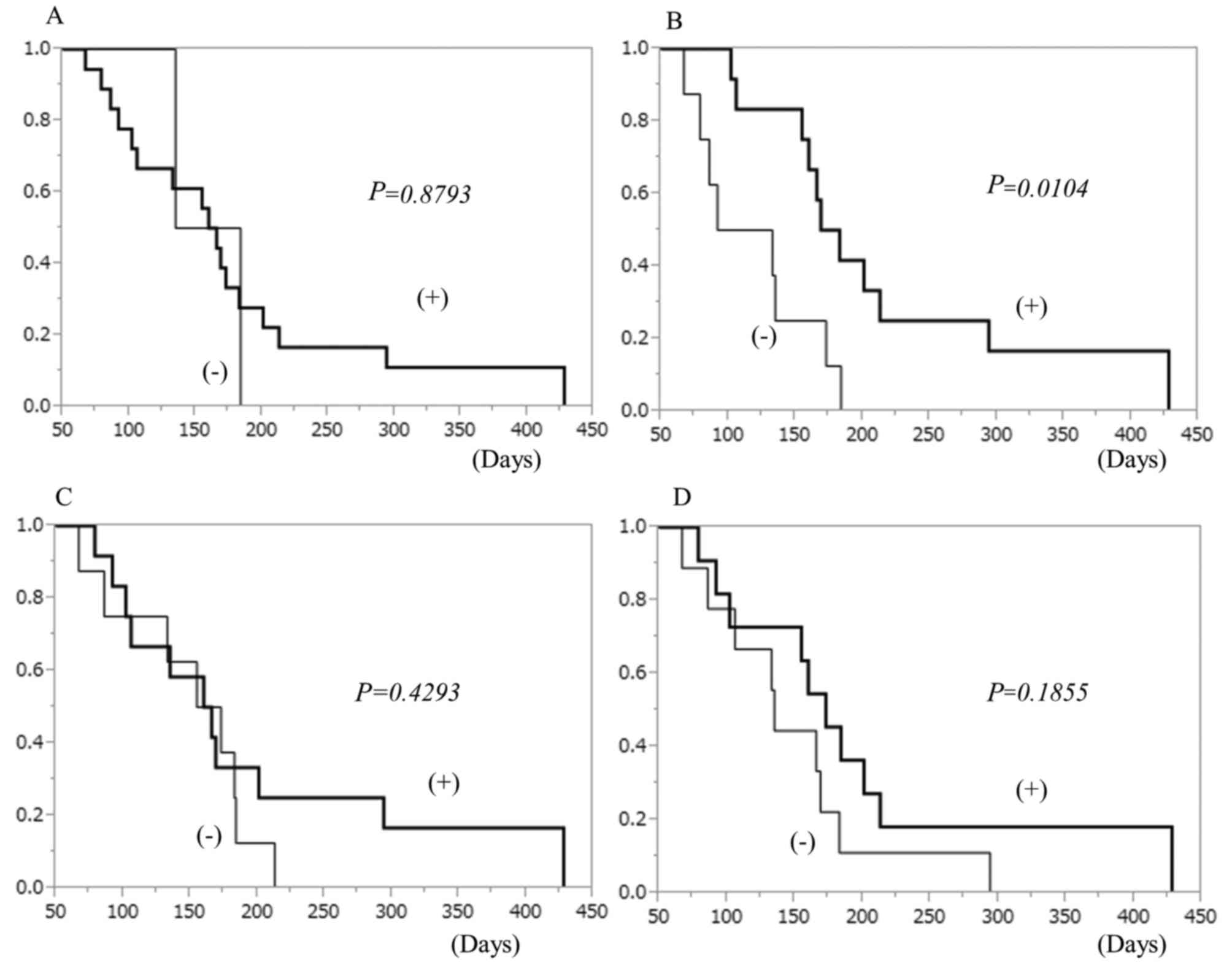
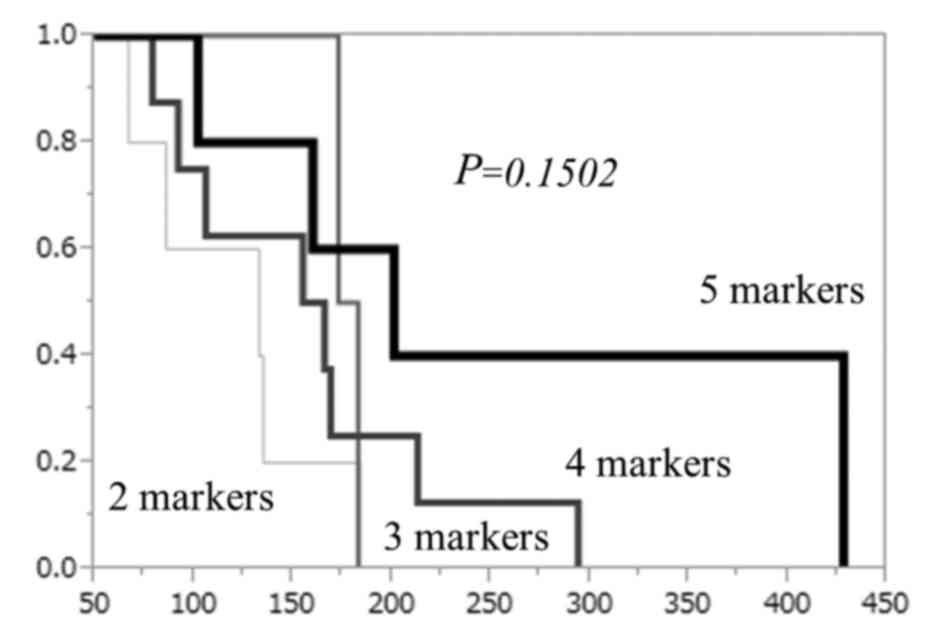
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ajani JA, Fairweather J, Dumas P, Patt YZ,
Pazdur R and Mansfield PF: Phase II study of Taxol in patients with
advanced gastric carcinoma. Cancer J Sci Am. 4:269–274.
1998.PubMed/NCBI
|
|
3
|
Einzig AI, Neuberg D, Remick SC, Karp DD,
O'Dwyer PJ, Stewart JA and Benson AB III: Phase II trial of
docetaxel (Taxotere) in patients with adenocarcinoma of the upper
gastrointestinal tract previously untreated with cytotoxic
chemotherapy: The Eastern Cooperative Oncology Group (ECOG) results
of protocol E1293. Med Oncol. 13:87–93. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Futatsuki K, Wakui A, Nakao I, Sakata Y,
Kambe M, Shimada Y, Yoshino M, Taguchi T and Ogawa N: Late phase II
study of irinotecan hydrochloride (CPT-11) in advanced gastric
cancer. CPT-11 Gastrointestinal Cancer Study Group. Gan To Kagaku
Ryoho. 21:1033–1038. 1994.In Japanese. PubMed/NCBI
|
|
5
|
Sakata Y, Ohtsu A, Horikoshi N, Sugimachi
K, Mitachi Y and Taguchi T: Late phase II study of novel oral
fluoropyrimidine anticancer drug S-1 (1 M tegafur-0.4 M gimestat-1
M otastat potassium) in advanced gastric cancer patients. Eur J
Cancer. 34:1715–1720. 1998. View Article : Google Scholar
|
|
6
|
Taguchi T, Tsukagoshi S, Furue H, Niitani
H and Noda K: Phase I clinical study of oxaliplatin. Gan To Kagaku
Ryoho. 25:1899–1907. 1998.In Japanese. PubMed/NCBI
|
|
7
|
Liu C, Sun Q, Hang X, Zhong B and Wang D:
Multicenter phase II study of capecitabine plus oxaliplatin as a
first-line therapy in Chinese patients with advanced gastric
cancer. Anticancer Drugs. 19:825–831. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Koizumi W, Narahara H, Hara T, Takagane A,
Akiya T, Takagi M, Miyashita K, Nishizaki T, Kobayashi O, Takiyama
W, et al: S-1 plus cisplatin versus S-1 alone for first-line
treatment of advanced gastric cancer (SPIRITS trial): A phase III
trial. Lancet Oncol. 9:215–221. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Japanese Gastric Cancer Association:
Japanese classification of gastric carcinoma: 3rd English edition.
Gastric Cancer. 14:101–112. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bang YJ, Van Cutsem E, Feyereislova A,
Chung HC, Shen L, Sawaki A, Lordick F, Ohtsu A, Omuro Y, Satoh T,
et al: ToGA Trial Investigators: Trastuzumab in combination with
chemotherapy versus chemotherapy alone for treatment of
HER2-positive advanced gastric or gastro-oesophageal junction
cancer (ToGA): A phase 3, open-label, randomized controlled trial.
Lancet. 376:687–697. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wilke H, Muro K, Van Cutsem E, Oh SC,
Bodoky G, Shimada Y, Hironaka S, Sugimoto N, Lipatov O, Kim TY, et
al RAINBOW Study Group: Ramucirumab plus paclitaxel versus placebo
plus paclitaxel in patients with previously treated advanced
gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): A
double-blind, randomised phase 3 trial. Lancet Oncol. 15:1224–1235.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Boon T, De Plaen E, Lurquin C, Van den
Eynde B, van der Bruggen P, Traversari C, Amar-Costesec A and Van
Pel A: Identification of tumour rejection antigens recognized by T
lymphocytes. Cancer Surv. 13:23–37. 1992.PubMed/NCBI
|
|
13
|
Rosenberg SA, Yang JC and Restifo NP:
Cancer immunotherapy: Moving beyond current vaccines. Nat Med.
10:909–915. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kantoff PW, Higano CS, Shore ND, Berger
ER, Small EJ, Penson DF, Redfern CH, Ferrari AC, Dreicer R, Sims
RB, et al IMPACT Study Investigators: Sipuleucel-T immunotherapy
for castration-resistant prostate cancer. N Engl J Med.
363:411–422. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Schwartzentruber DJ, Lawson DH, Richards
JM, Conry RM, Miller DM, Treisman J, Gailani F, Riley L, Conlon K,
Pockaj B, et al: gp100 peptide vaccine and interleukin-2 in
patients with advanced melanoma. N Engl J Med. 364:2119–2127. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Weber JS, O'Day S, Urba W, Powderly J,
Nichol G, Yellin M, Snively J and Hersh E: Phase I/II study of
ipilimumab for patients with metastatic melanoma. J Clin Oncol.
26:5950–5956. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Teh MT, Wong ST, Neill GW, Ghali LR,
Philpott MP and Quinn AG: FOXM1 is a downstream target of Gli1 in
basal cell carcinomas. Cancer Res. 62:4773–4780. 2002.PubMed/NCBI
|
|
18
|
Wierstra I and Alves J: FOXM1, a typical
proliferation-associated transcription factor. Biol Chem.
388:1257–1274. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang IC, Chen YJ, Hughes D, Petrovic V,
Major ML, Park HJ, Tan Y, Ackerson T and Costa RH: Forkhead box M1
regulates the transcriptional network of genes essential for
mitotic progression and genes encoding the SCF (Skp2-Cks1)
ubiquitin ligase. Mol Cell Biol. 25:10875–10894. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Laoukili J, Kooistra MR, Brás A, Kauw J,
Kerkhoven RM, Morrison A, Clevers H and Medema RH: FoxM1 is
required for execution of the mitotic programme and chromosome
stability. Nat Cell Biol. 7:126–136. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kalinichenko VV, Major ML, Wang X,
Petrovic V, Kuechle J, Yoder HM, Dennewitz MB, Shin B, Datta A,
Raychaudhuri P, et al: Foxm1b transcription factor is essential for
development of hepatocellular carcinomas and is negatively
regulated by the p19ARF tumor suppressor. Genes Dev. 18:830–850.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wonsey DR and Follettie MT: Loss of the
forkhead transcription factor FoxM1 causes centrosome amplification
and mitotic catastrophe. Cancer Res. 65:5181–5189. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kalin TV, Wang IC, Ackerson TJ, Major ML,
Detrisac CJ, Kalinichenko VV, Lyubimov A and Costa RH: Increased
levels of the FoxM1 transcription factor accelerate development and
progression of prostate carcinomas in both TRAMP and LADY
transgenic mice. Cancer Res. 66:1712–1720. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liu M, Dai B, Kang SH, Ban K, Huang FJ,
Lang FF, Aldape KD, Xie TX, Pelloski CE, Xie K, et al: FoxM1B is
overexpressed in human glioblastomas and critically regulates the
tumorigenicity of glioma cells. Cancer Res. 66:3593–3602. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chan DW, Yu SY, Chiu PM, Yao KM, Liu VW,
Cheung AN and Ngan HY: Overexpression of FOXM1 transcription factor
is associated with cervical cancer progression and pathogenesis. J
Pathol. 215:245–252. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Uddin S, Ahmed M, Hussain A, Abubaker J,
Al-Sanea N, Abduljabbar A, Ashari LH, Alhomoud S, Al-Dayel F, Jehan
Z, et al: Genome-wide expression analysis of Middle Eastern
colorectal cancer reveals FOXM1 as a novel target for cancer
therapy. Am J Pathol. 178:537–547. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kim IM, Ackerson T, Ramakrishna S,
Tretiakova M, Wang IC, Kalin TV, Major ML, Gusarova GA, Yoder HM,
Costa RH, et al: The Forkhead Box m1 transcription factor
stimulates the proliferation of tumor cells during development of
lung cancer. Cancer Res. 66:2153–2161. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Okada K, Fujiwara Y, Takahashi T, Nakamura
Y, Takiguchi S, Nakajima K, Miyata H, Yamasaki M, Kurokawa Y, Mori
M, et al: Overexpression of forkhead box M1 transcription factor
(FOXM1) is a potential prognostic marker and enhances
chemo-resistance for docetaxel in gastric cancer. Ann Surg Oncol.
20:1035–1043. 2013. View Article : Google Scholar
|
|
29
|
Higashihara Y, Kato J, Nagahara A, Izumi
K, Konishi M, Kodani T, Serizawa N, Osada T and Watanabe S: Phase I
clinical trial of peptide vaccination with URLC10 and VEGFR1
epitope peptides in patients with advanced gastric cancer. Int J
Oncol. 44:662–668. 2014.PubMed/NCBI
|
|
30
|
Murahashi M, Hijikata Y, Yamada K, Tanaka
Y, Kishimoto J, Inoue H, Marumoto T, Takahashi A, Okazaki T and
Takeda K: Phase I clinical trial of a five-peptide cancer vaccine
combined with cyclophosphamide in advanced solid tumors. Clin
Immunol. 166–167. 48–58. 2016.
|
|
31
|
Suzuki N, Hazama S, Ueno T, Matsui H,
Shindo Y, Iida M, Yoshimura K, Yoshino S, Takeda K and Oka M: A
phase I clinical trial of vaccination with KIF20A-derived peptide
in combination with gemcitabine for patients with advanced
pancreatic cancer. J Immunother. 37:36–42. 2014. View Article : Google Scholar
|
|
32
|
Yoshimura K, Minami T, Nozawa M and Uemura
H: Phase I clinical trial of human vascular endothelial growth
factor receptor 1 peptide vaccines for patients with metastatic
renal cell carcinoma. Br J Cancer. 108:1260–1266. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Therasse P, Arbuck SG, Eisenhauer EA,
Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van
Oosterom AT, Christian MC, et al: New guidelines to evaluate the
response to treatment in solid tumors. European Organization for
Research and Treatment of Cancer, National Cancer Institute of the
United States, National Cancer Institute of Canada. J Natl Cancer
Inst. 92:205–216. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Iinuma H, Fukushima R, Inaba T, Tamura J,
Inoue T, Ogawa E, Horikawa M, Ikeda Y, Matsutani N, Takeda K, et
al: Phase I clinical study of multiple epitope peptide vaccine
combined with chemoradiation therapy in esophageal cancer patients.
J Transl Med. 12:842014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Obara W, Ohsawa R, Kanehira M, Takata R,
Tsunoda T, Yoshida K, Takeda K, Katagiri T, Nakamura Y and Fujioka
T: Cancer peptide vaccine therapy developed from oncoantigens
identified through genome-wide expression profile analysis for
bladder cancer. Jpn J Clin Oncol. 42:591–600. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Asahara S, Takeda K, Yamao K, Maguchi H
and Yamaue H: Phase I/II clinical trial using HLA-A24-restricted
peptide vaccine derived from KIF20A for patients with advanced
pancreatic cancer. J Transl Med. 11:2912013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Masuzawa T, Fujiwara Y, Okada K, Nakamura
A, Takiguchi S, Nakajima K, Miyata H, Yamasaki M, Kurokawa Y, Osawa
R, et al: Phase I/II study of S-1 plus cisplatin combined with
peptide vaccines for human vascular endothelial growth factor
receptor 1 and 2 in patients with advanced gastric cancer. Int J
Oncol. 41:1297–1304. 2012.PubMed/NCBI
|
|
38
|
Kono K, Iinuma H, Akutsu Y, Tanaka H,
Hayashi N, Uchikado Y, Noguchi T, Fujii H, Okinaka K, Fukushima R,
et al: Multicenter, phase II clinical trial of cancer vaccination
for advanced esophageal cancer with three peptides derived from
novel cancer-testis antigens. J Transl Med. 10:1412012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Finn OJ, Khleif SN and Herberman RB: The
FDA guidance on therapeutic cancer vaccines: The need for revision
to include preventive cancer vaccines or for a new guidance
dedicated to them. Cancer Prev Res (Phila). 8:1011–1016. 2015.
View Article : Google Scholar
|