Introduction
Colorectal cancer (CRC) is the third most common
malignancy among men and women in the United States (1). The 5-year survival rate for all
patients with CRC was 65%, from 2006 to 2012 (2). Tumor stage significantly affects
patient outcome; the 5-year survival rates for localized stage,
regional and distant metastatic disease were 90, 71 and 14%, from
2006 to 2012, respectively (2).
Adjuvant chemotherapy has been demonstrated to improve the outcome
of patients with CRC at the late-stage(3). However, the benefit of adjuvant
chemotherapy in stage II CRC to survival is controversial (4–6). The
mechanism underlying the resistance/sensitivity of CRC cells to
chemotherapeutic agents remains to be fully elucidated. In
addition, identifying novel predictive biomarkers may assist in
predicting the clinical benefit of adjuvant chemotherapy for
CRC.
To date, a series of genetic alterations have been
identified in CRC. The deficient DNA mismatch repair (dMMR) feature
is common in CRC, and is associated with clinical benefit from
postoperative chemotherapy (7).
The survival rates of patients with dMMR tumors were not improved
by fluorouracil (5-Fu)-based adjuvant chemotherapy, compared with
those with proficient mismatch repair (pMMR) tumors (7,8). The
Kirsten rat sarcoma viral oncogene homolog (KRAS) mutation occurs
in ~35–40% of CRC tumors (9,10).
An increased risk of recurrence is observed in CRC tumors with the
KRAS mutation, compared with wild-type tumors (11). The B-raf serine/threonine kinase
proto-oncogene (BRAF) mutation is preferentially observed in dMMR
tumors, and is almost exclusive to the KRAS mutation in CRC
(9). No significant difference in
prognosis was reported between the BRAF mutation and wild-type
tumors in stage II CRC (11),
however, the BRAF mutation was associated with poor disease-free
survival (DFS) rate of stage III CRC (12). Tumor protein 53 (TP53) is
frequently mutated in several types of cancer, including CRC
(13). It has been reported that
TP53 mutations are more frequent in metastatic CRC, compared with
primary CRC (14). However,
evidence that mutations of the KRAS, BRAF or TP53 genes offer
potential as a predictive marker for adjuvant chemotherapeutic
benefit remains limited.
The polo-like kinases (Plks), a family of
serine/threonine protein kinases, are essential in cell cycle
checkpoint and DNA damage signaling (15). Plk1 is essential for cell cycle
regulation, and has been characterized with an oncogenic role in
several types of cancer (16).
However, the exact role of Plk2 in human cancer remains a topic of
debate. A predominate suppression of the expression of Plk2 has
been observed in B-cell lymphoma and ovarian cancer (17,18).
Plk2 inhibited cell proliferation and promoted chemotherapeutic
drug-induced apoptosis in cervical cancer (19). Furthermore, a lower expression of
Plk2 was associated with poor prognosis in breast cancer treated by
irradiation (20). These
observations suggested that Plk2 may serve as a tumor suppressor.
Plk2 has been reported to be essential for promoting survival and
inhibiting apoptosis in other cancer cells, including non-small
cell lung cancer, head and neck carcinoma, and osteosarcoma
(21–23). Previously, Ou et al reported
that the protein levels of Plk2 were significantly elevated in
tumor tissues, and were associated with adverse prognosis in CRC
(24). These data suggested that
the mechanism of Plk2 in carcinogenesis and cancer progression is
more complex than previously expected. Whether Plk2 is involved in
the chemoresistance of CRC remains to be fully elucidated.
In the present study, it was hypothesized that Plk2
is involved in the regulation of chemoresistance in CRC. By
analyzing a public CRC dataset, it was found that a high expression
of Plk2 was associated with dMMR, KRAS and BRAF mutations, and
limited survival benefit for adjuvant chemotherapy in late stage
CRC. In CRC cells, a higher expression of Plk2 was correlated with
increased resistance to chemotherapeutic agents. Experiments
involving the knockdown and exogenous overexpression of Plk2
demonstrated that this gene was involved in regulating the
chemoresistance of CRC cells.
Materials and methods
Gene expression analysis of Plk2 in
patients with CRC
The whole genome gene expression profile and
clinical information of 566 CRC cases were collected from GSE39582
in the Gene Expression Omnibus (GEO, https://www.ncbi.nlm.nih.gov/geo/) (25). The dataset included 310 men and 256
women, with a median age of 69 years. Of these, 233 patients
received standard adjuvant chemotherapy following surgical
resection, 316 patients received surgical treatment alone; the
treatment information for the remaining 17 patients was not
available. The median follow-up time for DFS and overall survival
(OS) were 3.58 and 4.25 years, respectively. For some patients in
the cohort, we did not have DFS or OS information. The number of
patients for which DFS and OS information was available was 560 and
562, respectively for the whole cohort. Similarly, in the adjuvant
chemotherapy subgroup, DFS and OS information was available for 232
and 233 patients, respectively. In the mono-surgery treatment
subgroup, DFS and OS information was available for 312 and 313
patients, respectively. The detailed clinical characteristics,
adjuvant chemotherapy treatment and prognostic information were
downloaded from the GEO website. The clinicopathological
characteristics of the patients are summarized in Table I. The present study was a
retrospective study and all data corresponding to the human tissues
is publicly available in the GEO.
 | Table IAssociation between expression of
Plk2 and clinical variables in colorectal cancer. |
Table I
Association between expression of
Plk2 and clinical variables in colorectal cancer.
| Variable | Parameter | All, n (%) | Expression of Plk
| P-value |
|---|
| Low, n (%) | High, n (%) |
|---|
| Sex | | | | | 0.447 |
| Female | 256 (45.2) | 123 (43.5) | 133 (47) | |
| Male | 310 (54.8) | 160 (56.5) | 150 (53) | |
| Age | | | | | 0.064 |
| <70 years | 301 (53.2) | 162 (57.2) | 139 (49.1) | |
| ≥70 years | 265 (46.8) | 121 (42.8) | 144 (50.9) | |
| Tumor location | | | | |
3.95×10−7 |
| Distal | 342 (60.4) | 201 (71) | 141 (49.8) | |
| Proximal | 224 (39.6) | 82 (29) | 142 (50.2) | |
| Stage | | | | | 0.301 |
| CIS | 4 (0.7) | 4 (1.4) | 0 (0) | |
| I | 37 (6.5) | 20 (7.1) | 17 (6) | |
| II | 258 (45.6) | 128 (45.2) | 130 (45.9) | |
| III | 203 (35.9) | 104 (36.7) | 99 (35) | |
| IV | 61 (10.8) | 27 (9.5) | 34 (12) | |
| NA | 3 (0.5) | 0 (0) | 3 (1.1) | |
| MMR status | | | | |
1.01×10−8 |
| dMMR | 75 (13.3) | 15 (5.3) | 60 (21.2) | |
| pMMR | 444 (78.4) | 251 (88.7) | 193 (68.2) | |
| NA | 47 (8.3) | 17 (6) | 30 (10.6) | |
| TP53 status | | | | |
3.87×10−7 |
| Wt | 161 (28.4) | 57 (20.1) | 104 (36.7) | |
| Mut | 190 (33.6) | 120 (42.4) | 70 (24.7) | |
| NA | 215 (38) | 106 (37.5) | 109 (38.5) | |
| KRAS status | | | | | 0.003 |
| Wt | 328 (58) | 182 (64.3) | 146 (51.6) | |
| Mut | 217 (38.3) | 91 (32.2) | 126 (44.5) | |
| NA | 21 (3.7) | 10 (3.5) | 11 (3.9) | |
| BRAF status | | | | |
5.24×10−7 |
| Wt | 461 (81.4) | 248 (87.6) | 213 (75.3) | |
| Mut | 51 (9) | 8 (2.8) | 43 (15.2) | |
| NA | 54 (9.5) | 27 (9.5) | 27 (9.5) | |
| Adj.Chem. | | | | | 0.185 |
| No | 316 (55.8) | 153 (54.1) | 163 (57.6) | |
| Yes | 233 (41.2) | 127 (44.9) | 106 (37.5) | |
| NA | 17 (3) | 3 (1.1) | 14 (4.9) | |
The gene expression data and the pharmacological
data of certain anticancer drugs of CRC cell lines were also
collected from the Cancer Cell Line Encyclopedia (CCLE) (26). The gene expression profiles of the
CCLE (GSE36133) were downloaded from the GEO database.
The raw data of gene expression profiles ('cel'
files of Affymetrix Human Genome U133 Plus 2.0 microarray) of the
CRC samples or cell lines were downloaded from the GEO database.
The gene expression profiles were log2-transformed and
normalized with the 'RMA' method. 'PMA' callings were detected
using the R package 'affy' (version 1.56.0, https://www.bioconductor.org/packages/release/bioc/html/affy.html).
Probes that were characterized as 'Present' ('PMA' callings) in
>20% of the samples were retained. Probe annotation was
performed based on the R package 'hgu133plus2.db' from Bioconductor
(version 3.2.3, https://www.bioconductor.org/packages/release/data/annotation/html/hgu133plus2.db.html).
The gene expression value of Plk2 (probe set no. 201939_at) was
extracted for subsequent analysis.
Cell culture and treatment
The SW620 cell line (cat. no. ATCC®
CCL-227™) and RKO cell line (cat. no. ATCC® CRL-2577™)
were purchased from the American Type Culture Collection (Manassas,
VA, USA). The HT55 cell line (cat. no. 5061105) was purchased from
the European Collection of Authenticated Cell Cultures
(Sigma-Aldrich, St. Louis, MO, USA). The Colo-678 cell line (cat.
no. ACC194) was purchased from the German Collection of
Microorganisms and Cell Cultures (Leibniz Institute DSMZ,
Braunschweig, Freistaat Sachsen, Germany). The cells were cultured
in DMEM/F12 and RPMI-1640 medium with 10% fetal bovine serum (FBS)
(Gibco/Thermo Fisher Scientific, Waltham, MA, USA), and incubated
at 37°C with 5% CO2. For the cell viability assay, the
cells were seeded in 96-well plates at 37°C at a density of
1.0×105/ml, and treated with oxaliplatin or 5-Fu at 0,
4, 20 32, 100, 160, 500 and 800 nM (6-wells/treatment) for 72 h.
Cell viability was detected with a Cell Counting kit-8 (CCK-8;
Dojindo Molecular Technologies, Inc., Kumamoto, Japan) and the
absorbance at 450 nm was recorded using the iMark microplate reader
(Bio-Rad Laboratories, Inc., Hercules, CA, USA). Dose-response
curves were fitted using the Sigmoid model. The half maximal
inhibitory concentration (IC50) was calculated by the
fitted models.
Cells at ~35% confluence were infected with a
lentivirus packaging different plasmids, including Plk2 short
hairpin RNA (shRNA), scramble control, Plk2 overexpression plasmids
and vector control, and 10 μg/ml polybrene. After 24 h,
cells were selected with fresh media containing puromycin at 2
μg/ml for 48 h. The cells were harvested and used for the
cell viability assay as described above, western blot analysis and
flow cytometric analysis. Lentiviral particles packaging the shRNA
targeting Plk2 (5′-TAGTCAAGTGACGGTGCTG-3′) and the scramble control
(5′-TTCTCCGAACGTGTCACGT-3′) were purchased from GeneChem Co., Ltd.
(Shanghai, China). Lentiviral particles packaging the pGLV-Plk2
expression vector and the empty control vector were purchased from
GenePharma Co., Ltd. (Shanghai, China).
Flow cytometric analysis
The cells were grown at 5×105/well in
6-well plates overnight. The cells were then treated with
oxaliplatin at 1 μM (SW620 cells), or 4 μM (Colo-678
cells), or dimethyl sulfoxide (DMSO) at a final concentration of
0.1% as a control for 16 h. The cells were washed twice with
phosphate-buffered saline. The cells were resuspended in binding
buffer at 1×106 cells/ml. APC-conjugated Annexin V (5
μl) and propidium iodide (5 μl) were added to 100
μl of cell suspension. The samples were incubated for 10 min
at room temperature. Binding buffer (400 μl) was added and
the samples were analyzed using a BD FACSCanto II flow cytometer
(BD Biosciences, Franklin Lakes, NJ, USA).
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
analysis
Total RNA from the CRC cell lines was isolated using
an RNeasy Mini kit (Qiagen GmbH, Düsseldorf, Germany), and then
quantified using a NanoDrop 2000 spectrophotometer (Thermo Fisher
Scientific, Inc., Waltham, MA, USA). The total RNA (100 ng) was
subjected to RT-qPCR analysis with the iTaq Universal SYBR One-Step
kit on the CFX-Connect Real-Time PCR detection system (both from
Bio-Rad Laboratories, Inc., Hercules, CA, USA) following the
manufacturer's instructions. The thermocycling conditions were as
follows: 10 min at 50°C for reverse transcription reaction, 1 min
at 95°C for polymerase activation and DNA denaturation (10 sec at
95°C for denaturation, 30 sec at 60°C for annealing/extension and
plate read) for 35 cycles, and a 0.5°C increment from 65 to 95°C
for melt-curve analysis. The primers used were as follows: Plk2
forward, 5′-GCTGATGTCTGGCTGTTCAT CAG-3′ and reverse sequence,
5′-CTTCCCTGTAGATCTCACA GTG-3′; GAPDH (endogenous control) forward,
5′-ACCCAGAAGACTGTGGATGG-3′ and reverse sequence, 5′-TTCAGC
TCAGGGATGACCTT-3′. The average quantification cycle (Cq) of the
triplicate experiments for each sample was used for the subsequent
analysis. The gene expression was calculated using the
2−ΔΔCq method (27),
where ΔCq = Cqtarget gene − Ctendogenous, and
ΔΔCq = ΔCtindividual sample−ΔCqreference
sample.
Western blot analysis
The cells were lysed with cell lysis buffer (Cell
Signaling Technology, Inc., Danvers, MA, USA) in the presence of
protease inhibitors. Total protein (40 μg) was
electrophoresed on 12% SDS-PAGE and electrophoretically transferred
onto a PVDF membrane, and blocked with 5% skim milk at room
temperature for 1 h. The membranes were then probed with different
primary antibodies overnight at 4°C. The membranes were washed for
5 min three times in TBS with 0.1% Tween-20 and then incubated with
horseradish peroxidase-conjugated mouse (1:10,000; cat. no.
1706516) or rabbit (1:10,000; cat. no. 1706515) (both from Bio-Rad,
Laboratories, Inc.) secondary antibodies at room temperature for 1
h. The membranes were washed 3 times for 5 min in TBS with 0.1%
Tween-20, and then visualized with the Lumi-Light western blotting
substrate (Roche Diagnostics, Basel, Switzerland) on the 5200
Chemiluminescence Imager (Tanon Science & Technology Co., Ltd.,
Shanghai, China). The following primary antibodies were used: Mouse
anti-Plk2 (1:1,000; cat. no. ab137539; Abcam, Cambridge, MA, USA),
mouse anti-cleaved poly(ADP-ribose) polymerase-1 (PARP-1; 1:200;
cat. no. sc56196; Santa Cruz Biotechnology, Inc., Dallas, TX, USA),
mouse anti-GAPDH (1:5,000; cat. no. TA-08; ZSGB-BIO, Beijing,
China).
Statistical analysis
All patients with CRC were grouped into Plk2-low and
Plk2-high expression subgroups by the median Plk2 expression value.
The association between Plk2 subgroups and the clinical/genetic
variables was tested using Pearson's χ2 test with Yates'
continuity correction or Fisher's exact test. The Kaplan-Meier
curve was applied to compare the DFS and OS of patients between
different Plk2 subgroups, and the log-rank test was used to
estimate the significance. Cox's univariate proportional hazards
model was used for Plk2 subgroups and different clinical variables,
based on the DFS and OS. Multivariate Cox analysis was performed
for all clinical variables initially. The final model was selected
by a stepwise selection process with the Akaike information
criterion. To analyze the clinical benefit of adjuvant
chemotherapy, the Kaplan-Meier curve and log-rank test were
performed for DFS and OS between patients with and without adjuvant
chemotherapy. The P-value of the RT-qPCR data of the four cell
lines was calculated using one-way analysis of variance and
multiple comparisons using Tukey's 'Honest Significant Difference'
method. The difference in cell apoptotic rate, determined by flow
cytometry, was calculated using Student's t-test (unpaired). All of
these statistical tests were two-sided, and performed using R
software (version 3.3.0, https://www.r-project.org/). P<0.05 was considered
to indicate a statistically significant difference.
Results
Association between the expression of
Plk2 and clinical/genetic parameters in patients with CRC
The present study investigated the association
between the gene expression of Plk2 and clinical or genetic
variables in the CRC cohort. As shown in Table I, a high expression of Plk2 was
significantly correlated with the proximal location of tumors
(P=3.95×10−7). A significant association was also found
between the high expression of Plk2 and dMMR
(P=1.0×10−8), wild-type TP53 (P=3.87×10−7),
mutant KRAS (P=0.003) and mutant BRAF (P=5.24×10−7).
There was a trend of significance between the expression of Plk2
and patient age (P=0.064). No statistically significant association
was found between the expression of Plk2 and sex (P=0.447) or
tumor-node-metastasis stage (P=0.301). There was also no
statistically significant difference in the expression of Plk2
between patients who received adjuvant chemotherapy and those who
did not receive adjuvant chemotherapy (P=0.185).
Prognostic value of Plk2 in patients with
CRC who received adjuvant chemotherapy
To evaluate the prognostic value of Plk2, all
patients were grouped into Plk2-high and Plk2-low subgroups by the
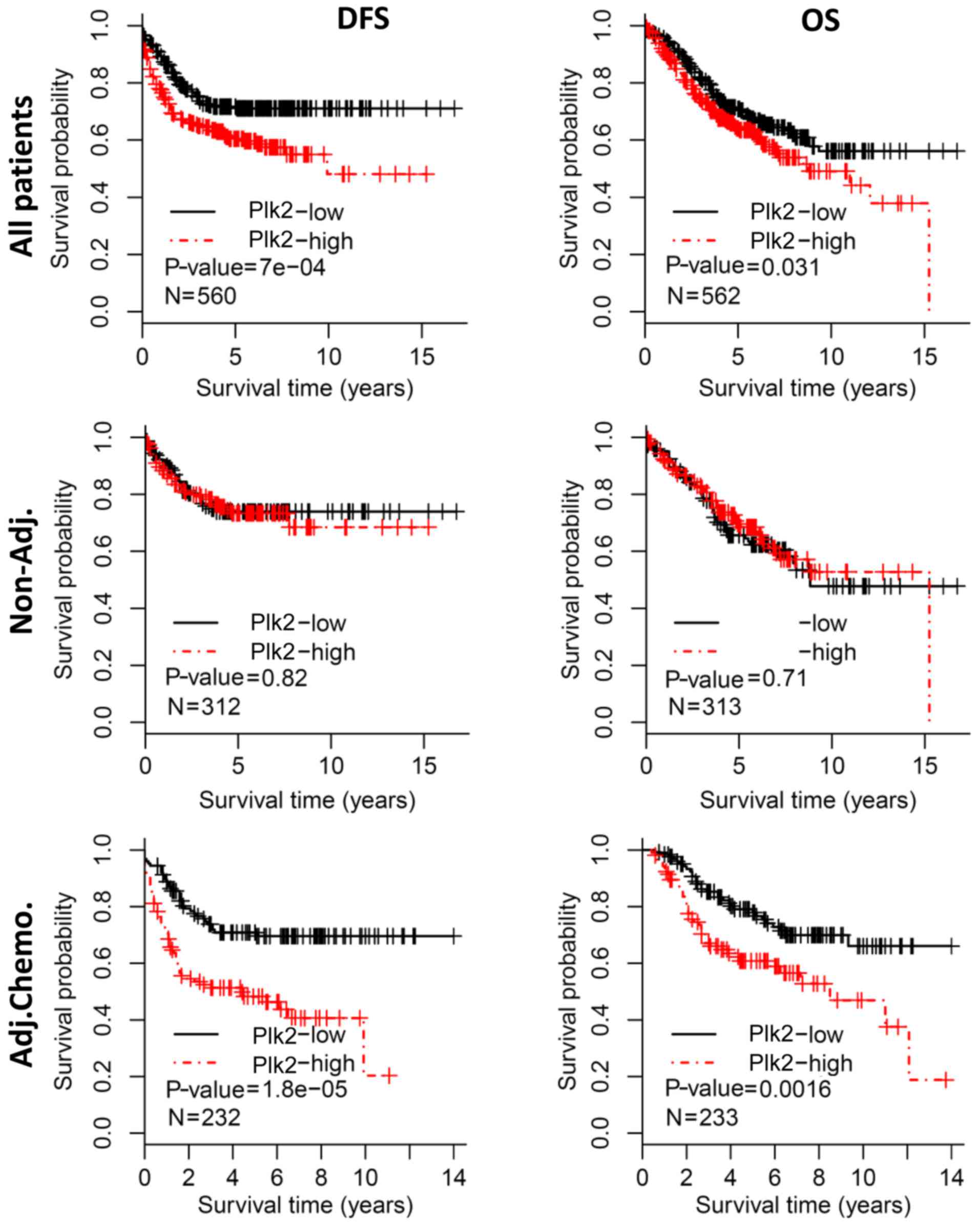
median expression value. Kaplan-Meier analysis revealed that
patients with a high expression of Plk2 had a poorer DFS, compared
with those with a low expression (log-rank test,
P=7.0×10−4) (Fig. 1,
upper left). Additionally, a high expression of Plk2 was associated
with a shorter OS (log-rank test, P=0.031) (Fig. 1, upper right). However, the
prognostic significance of Plk2 was not observed in the subgroup of
patients who did not receive adjuvant chemotherapy, based on either
DFS or OS (log-rank test, P>0.1) (Fig. 1, middle panel). When the patients
who received adjuvant chemotherapy were analyzed, a high expression
of Plk2 was significantly correlated with poorer DFS (log-rank
test, P=1.8×10−5) (Fig.
1, bottom left) and shorter OS (log-rank test, P=0.002)
(Fig. 1, bottom right),
respectively.
Subsequently, the present study investigated whether
Plk2 was an independent prognostic factor in patients with CRC who
received adjuvant chemotherapy. For DFS (Table II), stage IV (HR=3.81, 95% CI:
1.89-7.71, P=0.0002) and a high expression of Plk2 (HR=2.46, 95%
CI: 1.61-3.76, P=3.3×10−5) were significant prognostic
factors, according to univariate Cox analysis. The multivariate Cox
model showed that a high expression of Plk2 (HR=3.18, 95% CI:
1.88-5.35, P=1.43×10−5) was an independent prognostic
factor for DFS (Table II).
Additionally, univariate Cox analysis revealed that stage IV
(HR=9.4, 95% CI: 4.58–19.29, P=1.0×10−9), mutant KRAS
(HR=1.86, 95% CI: 1.17–2.97, P=0.009) and a high expression of Plk2
(HR=2.07, 95% CI: 1.31–3.29, P=0.002) were high risk factors for
poor OS (Table III).
Multivariate analysis showed that a high expression of Plk2
(HR=1.75, 95% CI: 1.08–2.84, P=0.024) was an independent prognostic
factor for OS in the final model (Table III).
 | Table IICox proportional hazards regression
for DFS of patients with colorectal cancer who received adjuvant
chemotherapy. |
Table II
Cox proportional hazards regression
for DFS of patients with colorectal cancer who received adjuvant
chemotherapy.
| Variable | Parameter | Univariate Cox
| Multivariate Cox
|
|---|
| HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Sex | Female | 1 | | | |
| Male | 1.01
(0.67–1.53) | 0.959 | | |
| Age | <70 years | 1 | | | |
| ≥70 years | 0.91
(0.57–1.43) | 0.669 | 0.60
(0.32–1.12) | 0.111 |
| Tumor location | Distal | 1 | | | |
| Proximal | 0.68
(0.43–1.08) | 0.104 | 0.56
(0.31–1.01) | 0.055 |
| Stage | II | 1 | | 1 | |
| III | 1.32
(0.78–2.25) | 0.304 | 1.61
(0.84–3.12) | 0.154 |
| IV | 3.81
(1.89–7.71) |
2.0×10−4 | 4.67
(2.02–10.81) |
3.1×10−4 |
| MMR status | pMMR | 1 | | | |
| dMMR | 0.67
(0.25–1.84) | 0.441 | 0.30
(0.07–1.26) | 0.101 |
| TP53 status | Wt | 1 | | | |
| Mut | 1.10
(0.62–1.95) | 0.736 | | |
| KRAS status | Wt | | | | |
| Mut | 1.27
(0.82–1.96) | 0.278 | | |
| BRAF status | Wt | 1 | | | |
| Mut | 0.60
(0.15–2.45) | 0.476 | | |
| Expression of
Plk2 | Low | 1 | | 1 | |
| High | 2.46
(1.61–3.76) |
3.27×10−5 | 3.18
(1.88–5.35) |
1.43×10−5 |
 | Table IIICox proportional hazards regression
for OS of patients with colorectal carcinoma who received adjuvant
chemotherapy. |
Table III
Cox proportional hazards regression
for OS of patients with colorectal carcinoma who received adjuvant
chemotherapy.
| Variable | Parameter | Univariate Cox
| Multivariate Cox
|
|---|
| HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Sex | Female | 1 | | | |
| Male | 1.22
(0.77–1.94) | 0.397 | | |
| Age | <70 years | 1 | | | |
| ≥70 years | 1.18
(0.72–1.93) | 0.504 | | |
| Tumor location | Distal | 1 | | | |
| Proximal | 1.31
(0.81–2.1) | 0.271 | | |
| Stage | II | 1 | | 1 | |
| III | 1.14
(0.63–2.06) | 0.675 | 1.07
(0.58–1.98) | 0.819 |
| IV | 9.40
(4.58–19.29) |
1.04×10−9 | 9.75
(4.66–20.39) |
1.48×10−9 |
| MMR status | pMMR | 1 | | | |
| dMMR | 0.94
(0.34–2.59) | 0.906 | | |
| TP53 status | Wt | 1 | | | |
| Mut | 1.06
(0.56–2.02) | 0.852 | | |
| KRAS status | Wt | 1 | | 1 | |
| Mut | 1.86
(1.17–2.97) | 0.009 | 1.71
(1.06–2.76) | 0.029 |
| BRAF status | Wt | 1 | | | |
| Mut | 0.38
(0.05–2.73) | 0.335 | | |
| Expression of
Plk2 | Low | 1 | | 1 | |
| High | 2.07
(1.31–3.29) | 0.002 | 1.75
(1.08–2.84) | 0.024 |
Plk2 as a potential predictor of survival
benefit from adjuvant chemotherapy for late stage CRC
Adjuvant chemotherapy was routinely used for the
majority of the late stage CRC cases. The present study aimed to
determine whether the expression of Plk2 affects the survival
benefit of adjuvant chemotherapy in stage III/IV CRC. For all
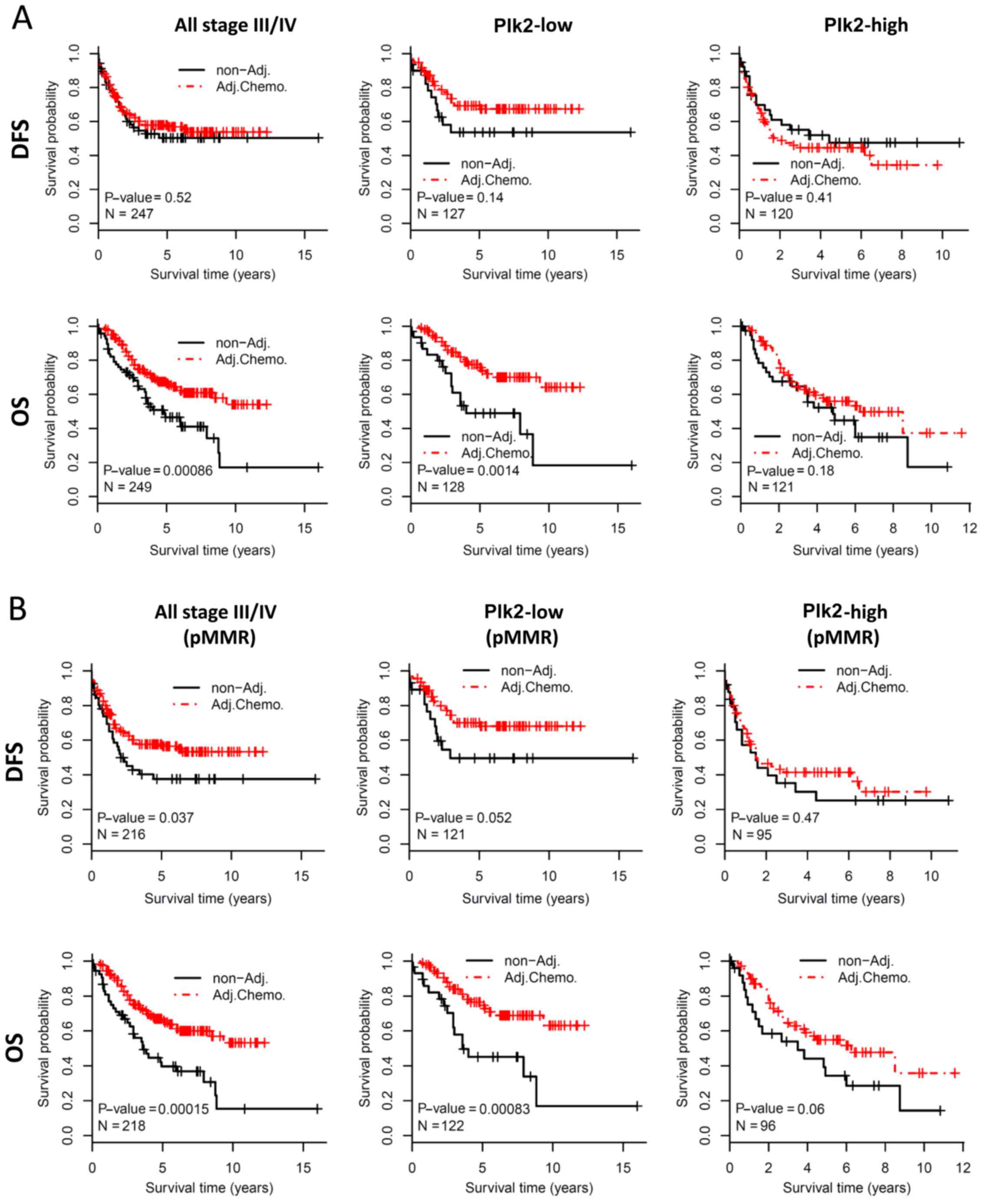
patients with stage III/IVCRC, adjuvant chemotherapy did not
improve DFS in this cohort (log-rank test, P=0.52, Fig. 2A, upper left panel). A trend of
improved DFS for adjuvant chemotherapy was observed in the Plk2-low
subgroup (log-rank test, P=0.14, Fig.
2A, upper middle panel), but not in the Plk2-high subgroup
(log-rank test, P=0.41) (Fig. 2A,
upper right panel). For OS, patients who received adjuvant
chemotherapy had significantly improved prognosis, compared with
those without adjuvant chemotherapy when considering all stage
III/IV patients (log-rank test, P=0.0009) (Fig. 2A, bottom left panel) or the
Plk2-low subgroup (log-rank test, P=0.001) (Fig. 2A, bottom middle panel). However,
adjuvant chemotherapy did not significantly improve OS in the
Plk2-high subgroup (log-rank test, P=0.18) (Fig. 2A, bottom right panel). The dMMR
status is associated with limited chemotherapeutic benefit in CRC.
The present study evaluated the survival effect of Plk2 in the
sub-population with pMMR, which included the majority of the
patients. Similarly, the survival benefit from adjuvant
chemotherapy was preferentially observed in patients with tumors
expressing a low level of Plk2 (Fig.
2B). These data suggested that a high expression of Plk2 may be
associated with limited clinical benefit from adjuvant chemotherapy
in late stage CRC. In addition, the association may be independent
of MMR status.
High expression of Plk2 is associated
with low sensitivity to chemotherapeutic agents in CRC cells
To examine the association between Plk2 and
chemotherapy, the present study detected the expression levels of
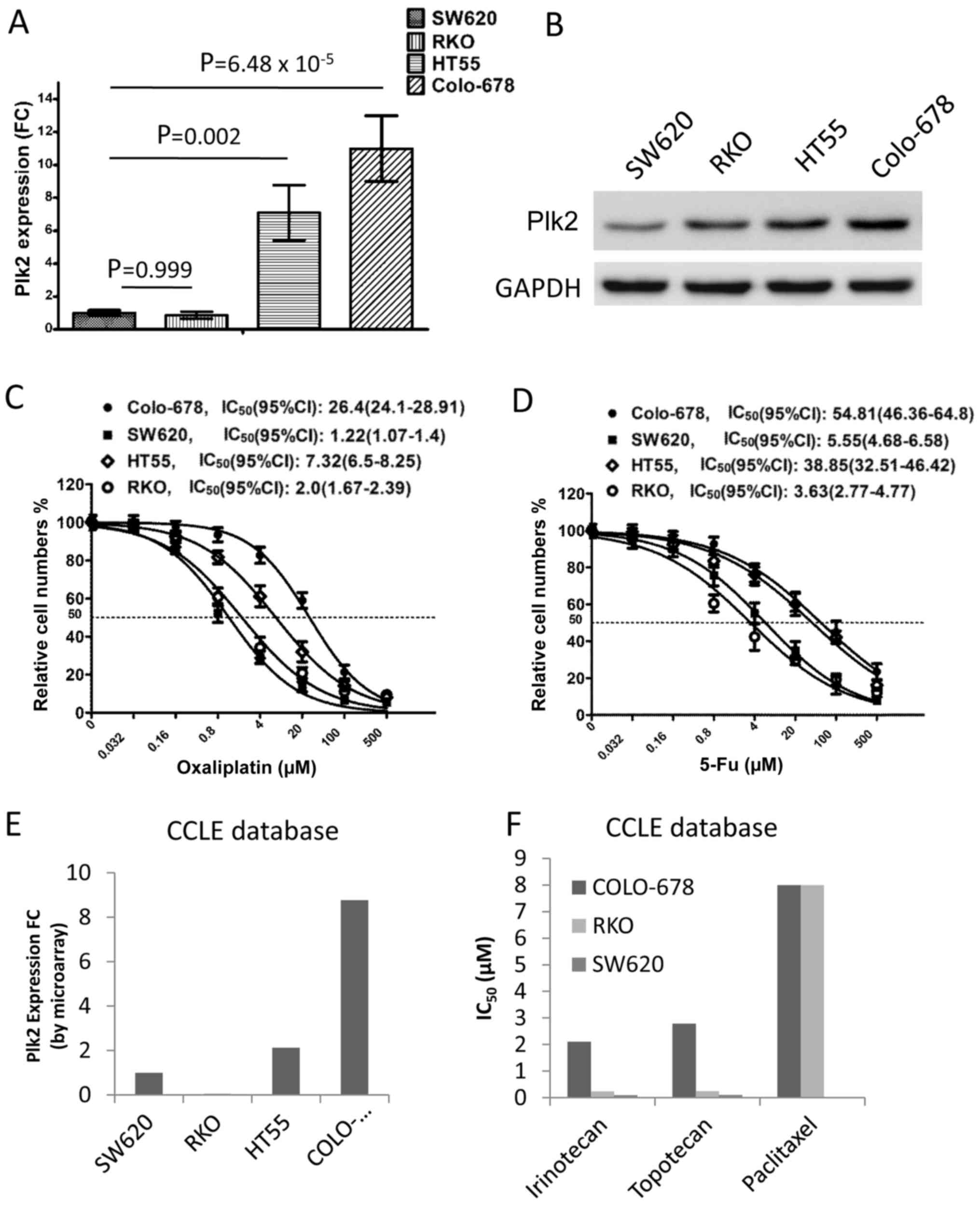
Plk2 in four CRC cell lines. The mRNA level of Plk2 was
significantly higher in the HT-55 and Colo-678 cells, compared with
that in the SW620 and RKO cells (Fig.
3A). In addition, western blot analysis showed elevated protein
levels of Plk2 in the HT-55 and Colo-678 cells, compared with
levels in the SW620 and RKO cells (Fig. 3B). The cell viability assay
revealed that IC50 values to oxaliplatin for the HT-55,
Colo-678, SW620 and RKO cells were 7.32 μM (95% CI:
6.5–8.25), 26.4 μM (95% CI: 24.1–28.91), 1.22 μM (95%
CI: 1.07–1.4) and 2.0 μM (95% CI: 1.67–2.39), respectively
(Fig. 3C). Accordingly, the
IC50 values to 5-Fu, another chemotherapeutic agent used
in CRC treatment, for the HT-55, Colo-678, SW620 and RKO cells were
38.85 μM (95% CI: 32.51–46.42), 54.81 μM (95% CI:
46.36–64.8), 5.55 μM (95% CI: 4.68–6.58) and 3.63 μM
(95% CI: 2.77–4.77), respectively (Fig. 3D). These findings were confirmed
using the CCLE dataset. Consistent with the results of the RT-qPCR
analysis, the mRNA level of Plk2 was higher in the Colo-678 and
HT-55 cells, compared with that in the SW620 and RKO cells in the
microarray (Fig. 3E). The
pharmacological results indicated that Colo-678 cells were more
resistant to certain chemotherapeutic agents, compared with the
SW620 and RKO cells (Fig. 3F). The
pharmacological results of the HT-55 cell line were not available
in the CCLE database. These data suggested that a higher expression
level of Plk2 was associated with increased resistance to
chemotherapeutic agents in the CRC cells.
Plk2 contributes to anti-apoptosis and
chemoresistance in CRC cells
Subsequently, the present study examined whether the
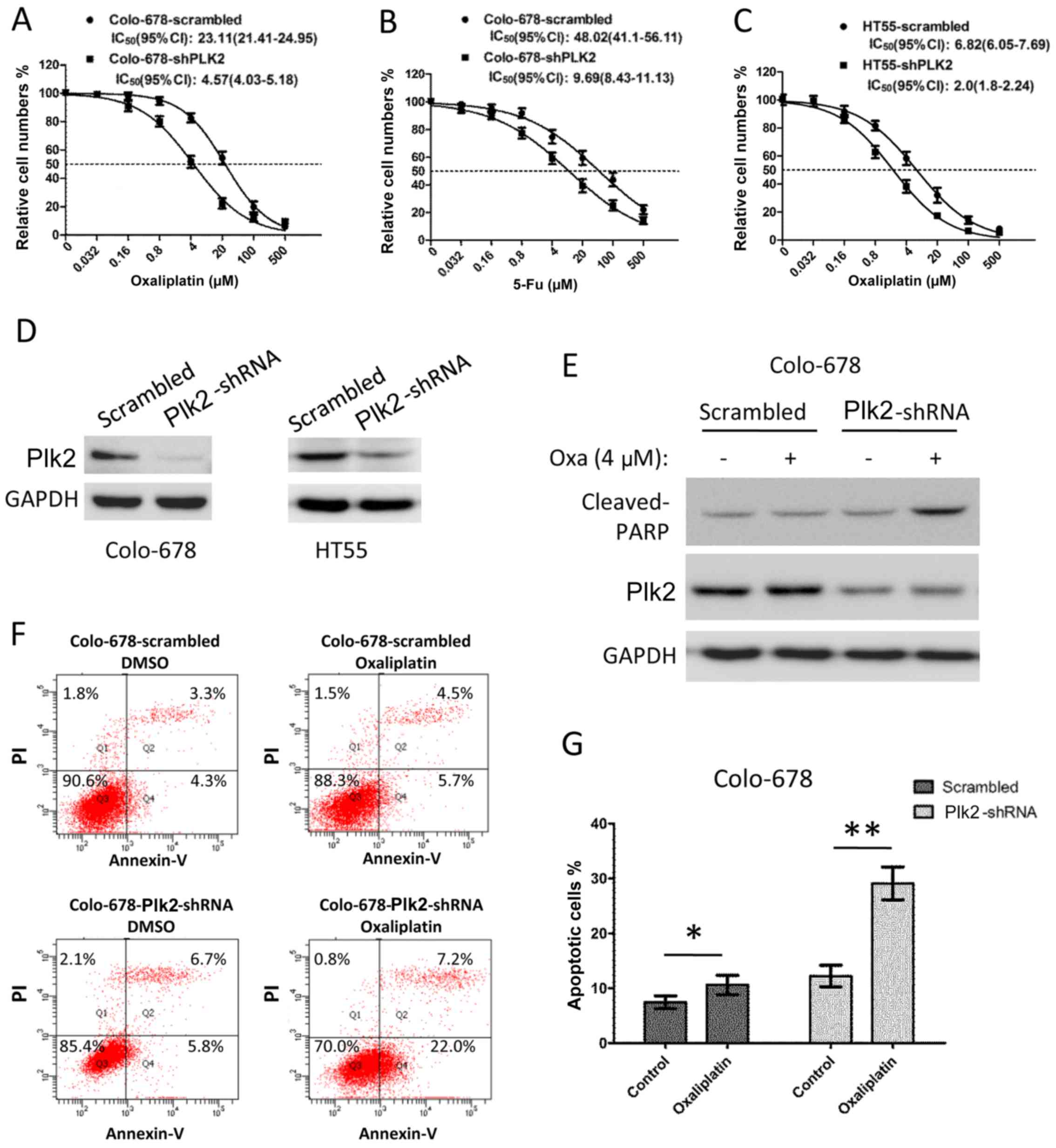
dysregulation of Plk2 affects chemoresistance in CRC cells. The
Colo-678 cells were infected with lentivirus-encoded scramble or
Plk2 shRNA for 48 h to deplete the expression of Plk2. The Colo-678
cells expressing Plk2-shRNA exhibited decreased IC50 to
oxaliplatin (shRNA, vs. scramble, 4.57 μM, 95% CI:
4.03–5.18, vs. 23.11 μM, 95% CI: 21.41–24.95) and decreased
IC50 to 5-Fu (shRNA, vs. scramble, 9.69 μM, 95%
CI: 8.43–11.13, vs. 48.02 μM, 95% CI: 41.1–56.11), as shown
in Fig. 4A and B. Similarly, the
sensitivity to oxaliplatin of the HT55 cells was increased when the
expression of Plk2 was depleted by shRNA (Fig. 4C, IC50 Plk2-shRNA, vs.
scramble, 2.0 μM, 95% CI: 1.8–2.24, vs. 6.82 μM, 95%
CI: 6.05–7.69). Western blot analysis demonstrated a marked
decrease in the protein level of Plk2 in the Plk2-shRNA cells
(Fig. 4D). In addition, an
apoptotic assay was performed to determine whether the decrease in
the expression of Plk2 led to increased apoptosis in response to
chemotherapeutic agents. As shown in Fig. 4E, following treatment with 4
μM oxaliplatin for 16 h, the scramble cells did not show
increased levels of cleaved PARP (a marker of apoptosis). However,
oxaliplatin treatment induced a significant elevation of cleaved
PARP in the Plk2-knockdown cells. Flow cytometric analysis showed
that Colo-678 cells with Plk2 knockdown exhibited increased
apoptosis, compared with the scramble control cells in DMSO.
Oxaliplatin treatment (4 μM for 16 h) induced apoptosis to a
higher level, compared with that in the DMSO control in the
Plk2-knockdown cells. By contrast, oxaliplatin only induced minor
apoptosis over DMSO treatment in the scramble control Colo-678
cells (Fig. 4F and G).
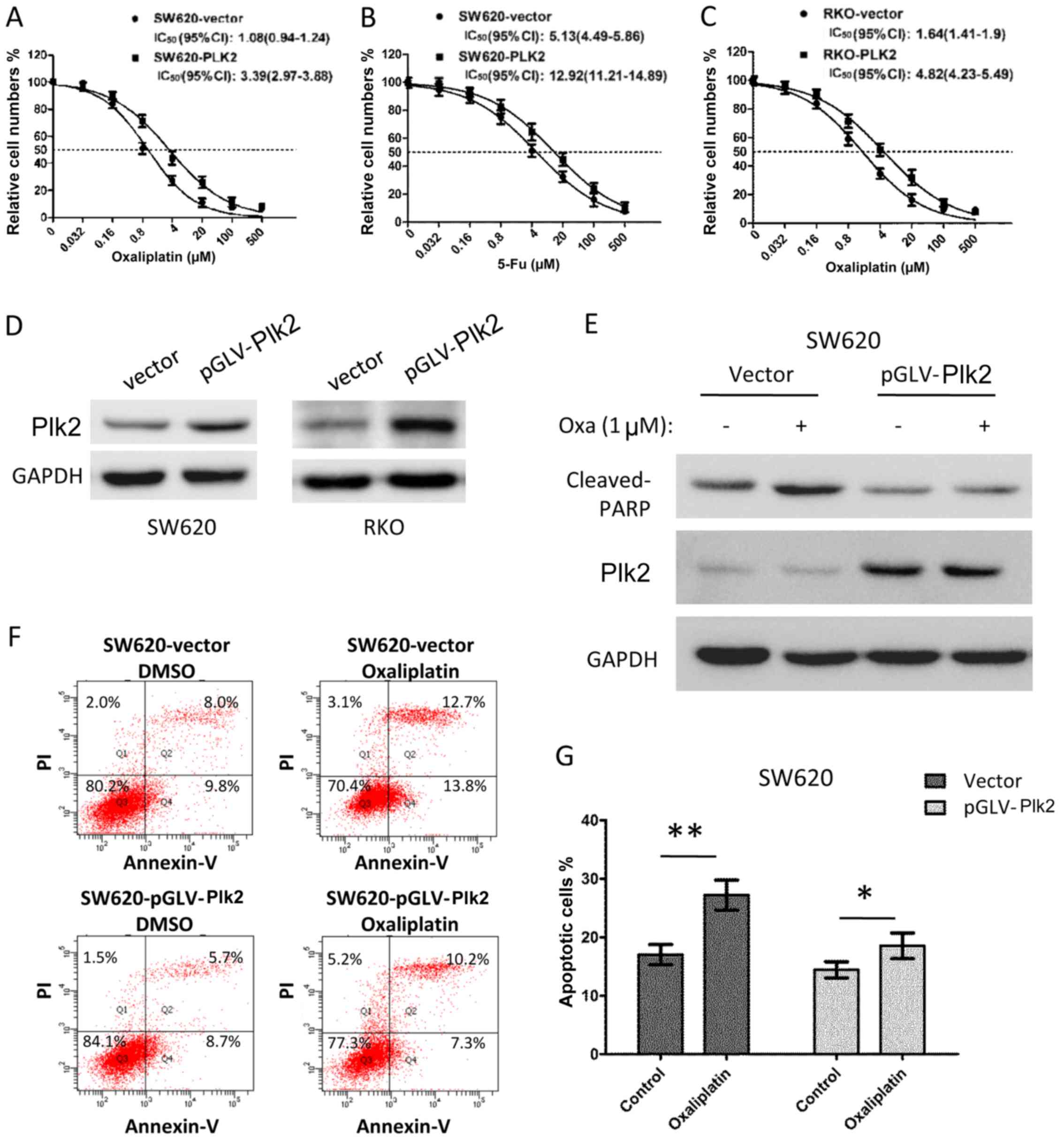
Subsequently, the effect of the overexpression of
Plk2 on chemosensitivity was examined in CRC cells. The
Plk2-overexpressing SW620 cells had an increased IC50 to
oxaliplatin (Plk2, vs. vector, 3.39 μM, 95% CI: 2.97–3.88,
vs. 1.08 μM, 95% CI: 0.94–1.24), and were more resistant to
5-Fu (Plk2, vs. vector, IC50, 12.92 μM, 95% CI:
11.21–14.89, vs. 5.13 μM, 95% CI: 4.49–5.86), as shown in
Fig. 5A and B. Similar results
were observed for RKO cells with exogenous overexpression of Plk2
(Fig. 5C, IC50 to
oxaliplatin, Plk2, vs. vector, 4.82 μM, 95% CI: 4.23–5.49,
vs. 1.64 μM, 95% CI: 1.41–1.9). The results of the western
blot analysis confirmed the overexpression of Plk2 protein in the
Plk2-transfected cells, compared with that in the vector control
cells (Fig. 5D). As expected,
following treatment with 1 μM oxaliplatin for 16 h, the
control cells exhibited increased the levels of cleaved PARP
(Fig. 5E). However, the
overexpression of Plk2 in SW620 cells inhibited the upregulation of
cleaved PARP in response to oxaliplatin (Fig. 5E). Flow cytometric analysis
demonstrated that the Plk2-overexpressing SW620 cells exhibited
decreased apoptosis, compared with the vector control cells in the
DMSO and oxaliplatin treatment conditions. Oxaliplatin treatment (1
μM for 16 h) induced a higher level of apoptosis in the
vector control cells, compared with the DMSO control cells.
However, oxaliplatin induced only minor apoptosis in the
Plk2-overexpressing SW620 cells over DMSO treatment (Fig. 5F and G).
Discussion
In the present study, the clinical association of
Plk2 in patients with CRC was examined and its role in
chemoresistance in CRC cells was investigated. A higher expression
of Plk2 was more frequently observed in proximally located tumors,
and was correlated with pMMR status, BRAF mutation, KRAS mutation
and TP53 wild-type. The expression ofPlk2 was a prognostic factor
for an unfavorable outcome in patients who received adjuvant
chemotherapy, but not in those without. The higher expression of
Plk2 was correlated with increased resistance to chemotherapeutic
agents in CRC cells. The knockdown of Plk2 in cells expressing a
high level of Plk2 resulted in enhanced apoptosis and sensitized
cells to chemotherapeutic drugs. By contrast, the exogenous
expression of Plk2 in cells expressing a low level of Plk2
inhibited the apoptosis induced by chemotherapeutic agents. These
results suggested that Plk2 may be involved in chemoresistance in
CRC through inhibiting apoptosis.
Plk2 has been documented as a tumor suppressor in
B-cell lymphoma and ovarian cancer, and is transcriptionally
silenced in these tumors as a result (17,18).
However, another study demonstrated that Plk2 was significantly
overexpressed in CRC tissues (24); elevated protein levels of Plk2 were
associated with tumor metastasis and poor prognosis for OS and DFS
(24). Consistently, the results
of the present study showed that a higher gene expression of Plk2
in CRC was associated with shorter OS and DFS of patients. It was
also revealed that a higher expression of Plk2 was more likely to
be associated with proximally located tumors, which are reported to
have poorer clinical outcome, compared with distal tumors (28,29).
The present study found that Plk2 was only of prognostic value in
the patients who received adjuvant chemotherapy, but not in those
without adjuvant chemotherapy. However, this finding was not
reported in the previous studies. Multivariate Cox regression
revealed that the expression ofPlk2wasan independent prognostic
factor for OS and DFS in patients who received adjuvant
chemotherapy. In III/IV stage CRC, adjuvant chemotherapy
significantly improved the OS of patients. However, the improvement
of OS by adjuvant chemotherapy was observed only in the Plk2-low
subgroup and not in the Plk2-high subgroup. For stage I/II CRC, the
expression of Plk2 was not able to predict patient outcome in
patients, whether they received adjuvant chemotherapy or not (data
not shown). This was possibly due to only a small number of
patients at the early stage (n=55) receiving adjuvant chemotherapy.
These findings suggested that Plk2 serves as a promising biomarker
for predicting the outcome benefit from adjuvant chemotherapy in
late stage CRC.
The Plk family is essential in cell cycle checkpoint
and DNA damage signaling (15).
Four of the family members (Plk1-4) were present in the CRC dataset
analyzed in the present study. Further investigation found that
high expression levels of Plk1 and Plk4 were protective factors for
favorable prognosis, whereas the overexpression of Plk2 and Plk3
was associated with unfavorable patient outcome (Table IV). Additionally, for patients who
received adjuvant chemotherapy, only Plk1 and Plk2 significantly
affected the prediction of DFS and OS (Table IV). These findings suggested that
the Plk family members may have different roles in the
carcinogenesis of CRC.
 | Table IVUnivariate Cox proportional hazards
regression analysis of Plk family in patients with colorectal
cancer. |
Table IV
Univariate Cox proportional hazards
regression analysis of Plk family in patients with colorectal
cancer.
| Patient group | Plk family | DFS
| OS
|
|---|
| HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| All | Plk1 (high vs.
low) | 0.65
(0.48–0.87) | 0.004 | 0.66
(0.5–0.88) | 0.005 |
| Plk2 (high vs.
low) | 1.68
(1.24–2.27) | 0.0008 | 1.37
(1.03–1.82) | 0.032 |
| Plk3 (high vs.
low) | 1.54
(1.14–2.08) | 0.005 | 1.37
(1.03–1.83) | 0.031 |
| Plk4 (high vs.
low) | 0.50
(0.36–0.68) |
8.67×10−6 | 0.67 (0.5–0.9) | 0.007 |
| Adj.Chemo | Plk1 (high vs.
low) | 0.64
(0.43–0.98) | 0.039 | 0.45
(0.28–0.72) | 0.0009 |
| Plk2 (high vs.
low) | 2.46
(1.61–3.76) |
3.27×10−5 | 2.07
(1.31–3.29) | 0.002 |
| Plk3 (high vs.
low) | 1.26
(0.83–1.9) | 0.28 | 1.5
(0.95–2.37) | 0.085 |
| Plk4 (high vs.
low) | 0.68
(0.45–1.03) | 0.066 | 0.72
(0.46–1.13) | 0.156 |
dMMR is a common genetic alteration in CRCs. A
previous study demonstrated that dMMR tumors were resistant to
5-Fu-based therapy and that the patients benefitted less from
adjuvant chemotherapy (7). The
results of the present study indicated that the tumors with a
higher expression of Plk2 had a higher frequency of dMMR (21.2%),
compared with those with a lower expression ofPlk2 (5.3%). It has
been reported that the BRAF mutation is associated with dMMR status
in CRC (10,30). Accordingly, the present study found
that the upregulation of Plk2 was associated with a higher
frequency of BRAF mutation, compared with the low expression of
Plk2 (15.2 vs. 2.8%, respectively). Although the KRAS mutation is
almost exclusive to the BRAF mutation in CRC (9,10),
the present study found that the expression of Plk2 was also
positively correlated with the KRAS mutation rate (Plk2-high vs.
low: 44.5 vs. 32.2%). These findings suggested that the
upregulation of Plk2 may be more likely to be observed in CRC with
oncogenic mutations. In addition, according to previous reports,
dMMR, BRAF and KRAS mutations are more frequently detected in
proximal/right-sided tumors (9,10).
Consistently, the present study revealed that the proximally
located tumors were associated with a higher expression of Plk2. In
addition, the TP53 mutation was inversely correlated with the
expression ofPlk2, which may due to the expression of Plk2 being
induced by wild-type TP53 (31,32).
From the GDSC database (33), the
present study found that TP53 was mutated in SW620 and HT55 cells,
KRAS mutations were found in SW620 and Colo-678 cells, and BRAF
mutations were observed in RKO cells. Therefore, there was no
marked correlation between the chemosensitivity and the mutant
status of TP53, KRAS and BRAF in the four CRC cells.
A previous study demonstrated that a high expression
of Plk2 was associated with an anti-apoptotic effect in CRC cells
under normal tissue culture conditions (24). However, the downregulation of Plk2
mediated by microRNA-27b has been shown to be associated with
accelerated proliferation and inhibited paclitaxel-induced cell
apoptosis in cervical cancer (19). These results suggest that Plk2 may
have bidirectional effects in the regulation of apoptosis based on
different cell types. In the present study, it was found that a
higher expression of Plk2 indicated a poorer outcome in patients
with CRC who received adjuvant chemotherapy, suggesting an
anti-chemotherapeutic effect of Plk2. A higher expression level of
Plk2 was observed in CRC cells with increased resistance to
chemothereapeutic agents. In line with previous results (24), the present study observed that the
knockdown of Plk2 in Colo-678 cells resulted in increased
apoptosis, whereas the exogenous expression of Plk2 decreased
apoptosis in SW620 cells. It was also found that Plk2 was important
in the response of CRC cells to chemotherapeutic agents. Apoptosis
induced by oxaliplatin was markedly enhanced in the cells with
depressed expression of Plk2. The observation of decreased
apoptosis induced by oxaliplatin in Plk2-overexpressing cells
confirmed the chemoresistant feature of Plk2. Taken together, these
data suggested that a high expression of Plk2 may contribute to
chemoresistance by inhibiting the apoptosis of CRC cells.
There were several possible mechanisms underlying
the anti-chemotherapeutic role of Plk2. The expression of Plk2 is
transcriptionally regulated by TP53, and is involved in DNA
replication and the S-phase checkpoint (21,31).
Plk2 depletion deficiency results in enhanced phosphorylation of
H2AX and DNA damage in response to replication stress (21). The mutant TP53 can be
phosphorylated by Plk2, to result in an improved oncogenic effect
and enhanced chemoresistance in cancer cells (34). However, the TP53 mutation was
identified in the chemoresistant HT-55 cells and the sensitive
SW620 cells, suggesting other mechanisms are involved in the
chemoresistant effect of Plk2.TAp73 is a known tumor suppressor
with a similar structure top 53. The function of TAp73 can be
inhibited by Plk2, which phosphorylates TAp73 at Ser48 and prevents
its nuclear translocation (22).
Suppressing the expression of Plk2 enhances DNA-damaging drug
induced apoptosis through a TAp73-dependent manner (22). In addition, previous studies have
found that TAp73 is involved in the drug-induced apoptosis of CRC
cells (35,36), particularly in p53-deficient cells
(35). Another downstream target
of Plk2 is F-box and WD repeat domain containing 7 (Fbxw7). It is
reported that Plk2 binds to Fbxw7 and mediates its subsequent
degradation, resulting an in anti-apoptotic effect in CRC cells
(24). Accordingly, a low
expression of Fbxw7 was associated with poor patient prognosis and
increased resistance to chemotherapy (37). Collectively, these previous results
indicated that TAp73 and Fbxw7 may serve as the important
downstream targets of Plk2 in the chemoresistance of CRC cells.
In conclusion, the present study found that a high
expression of Plk2 was significantly associated with reduced
benefit from adjuvant chemotherapy in late stage CRC. Furthermore,
experiments involving the knockdown and exogenous overexpression of
Plk2 demonstrated that Plk2 is important in the chemoresistance of
CRC cells. These results suggest that Plk2 may serve as a predictor
marker for the clinical benefit of adjuvant chemotherapy, and that
targeting Plk2 offers a promising novel strategy in CRC
therapy.
Acknowledgments
Not applicable.
Abbreviations:
|
Plk2
|
polo-like kinase 2
|
|
CRC
|
colorectal cancer
|
|
dMMR
|
deficient mismatch repair
|
|
pMMR
|
proficient mismatch repair
|
|
GEO
|
Gene Expression Omnibus
|
|
OS
|
overall survival
|
|
DFS
|
disease-free survival
|
|
IC50
|
half maximal inhibitory
concentration
|
|
FBS
|
fetal bovine serum
|
|
shRNA
|
short hairpin RNA
|
Notes
[1]
Funding
No funding was received.
[2] Authors'
contributions
The contributions of the authors were as follows:
Conception and design: JC; development of methodology: YX, YL and
QL; acquisition of data: YX, YL and QL; analysis and interpretation
of data: YX and YL; writing, review, and/or revision of the
manuscript: YX and JC; administrative, technical or material
support: JC and QL; study supervision: YX and YL. All authors read
and approved the final manuscript.
[3] Availability
of data and materials
The datasets analyzed in the present study are
publicly available in Gene Expression Omnibus (accession nos.
GSE39582 and GSE36133).
[4] Ethics
approval and consent to participate
Not applicable.
[5] Consent for
publication
Not applicable.
[6] Competing
interests
The authors declare that there have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
Statistics, 2017. CA Cancer J Clin. 67:7–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Siegel RL, Miller KD, Fedewa SA, Ahnen DJ,
Meester RGS, Barzi A and Jemal A: Colorectal cancer statistics,
2017. CA Cancer J Clin. 67:177–193. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
André T, Boni C, Mounedji-Boudiaf L,
Navarro M, Tabernero J, Hickish T, Topham C, Zaninelli M, Clingan
P, Bridgewater J, et al Multicenter International Study of
Oxaliplatin/5-Fluorouracil/Leucovorin in the Adjuvant Treatment of
Colon Cancer (MOSAIC) Investigators: Oxaliplatin, fluorouracil, and
leucovorin as adjuvant treatment for colon cancer. N Engl J Med.
350:2343–2351. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
André T, de Gramont A, Vernerey D,
Chibaudel B, Bonnetain F, Tijeras-Raballand A, Scriva A, Hickish T,
Tabernero J, Van Laethem JL, et al: Adjuvant fluorouracil,
leucovorin, and oxaliplatin in stage II to III colon cancer:
Updated 10-year survival and outcomes according to BRAF mutation
and mismatch repair status of the MOSAIC study. J Clin Oncol.
33:4176–4187. 2015. View Article : Google Scholar
|
|
5
|
Gray R, Barnwell J, McConkey C, Hills RK,
Williams NS and Kerr DJ; Quasar Collaborative Group: Adjuvant
chemotherapy versus observation in patients with colorectal cancer:
A randomised study. Lancet. 370:2020–2029. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cao B, Luo L, Feng L, Ma S, Chen T, Ren Y,
Zha X, Cheng S, Zhang K and Chen C: A network-based predictive
gene-expression signature for adjuvant chemotherapy benefit in
stage II colorectal cancer. BMC Cancer. 17:8442017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sargent DJ, Marsoni S, Monges G, Thibodeau
SN, Labianca R, Hamilton SR, French AJ, Kabat B, Foster NR, Torri
V, et al: Defective mismatch repair as a predictive marker for lack
of efficacy of fluorouracil-based adjuvant therapy in colon cancer.
J Clin Oncol. 28:3219–3226. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ribic CM, Sargent DJ, Moore MJ, Thibodeau
SN, French AJ, Goldberg RM, Hamilton SR, Laurent-Puig P, Gryfe R,
Shepherd LE, et al: Tumor microsatellite-instability status as a
predictor of benefit from fluorouracil-based adjuvant chemo-therapy
for colon cancer. N Engl J Med. 349:247–257. 2003. View Article : Google Scholar
|
|
9
|
Gonsalves WI, Mahoney MR, Sargent DJ,
Nelson GD, Alberts SR, Sinicrope FA, Goldberg RM, Limburg PJ,
Thibodeau SN, Grothey A, et al: Patient and tumor characteristics
and BRAF and KRAS mutations in colon cancer, NCCTG/Alliance N0147.
J Natl Cancer Inst. Jun 12–2014.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li W, Qiu T, Zhi W, Shi S, Zou S, Ling Y,
Shan L, Ying J and Lu N: Colorectal carcinomas with KRAS codon 12
mutation are associated with more advanced tumor stages. BMC
Cancer. 15:3402015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hutchins G, Southward K, Handley K, Magill
L, Beaumont C, Stahlschmidt J, Richman S, Chambers P, Seymour M,
Kerr D, et al: Value of mismatch repair, KRAS, and BRAF mutations
in predicting recurrence and benefits from chemotherapy in
colorectal cancer. J Clin Oncol. 29:1261–1270. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sinicrope FA, Mahoney MR, Smyrk TC,
Thibodeau SN, Warren RS, Bertagnolli MM, Nelson GD, Goldberg RM,
Sargent DJ and Alberts SR: Prognostic impact of deficient DNA
mismatch repair in patients with stage III colon cancer from a
randomized trial of FOLFOX-based adjuvant chemotherapy. J Clin
Oncol. 31:3664–3672. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yurgelun MB, Kulke MH, Fuchs CS, Allen BA,
Uno H, Hornick JL, Ukaegbu CI, Brais LK, McNamara PG, Mayer RJ, et
al: Cancer susceptibility gene mutations in individuals with
colorectal cancer. J Clin Oncol. 35:1086–1095. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Vakiani E, Janakiraman M, Shen R, Sinha R,
Zeng Z, Shia J, Cercek A, Kemeny N, D'Angelica M, Viale A, et al:
Comparative genomic analysis of primary versus metastatic
colorectal carcinomas. J Clin Oncol. 30:2956–2962. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Reinhardt HC and Yaffe MB:
Phospho-Ser/Thr-binding domains: Navigating the cell cycle and DNA
damage response. Nat Rev Mol Cell Biol. 14:563–580. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Takai N, Hamanaka R, Yoshimatsu J and
Miyakawa I: Polo-like kinases (Plks) and cancer. Oncogene.
24:287–291. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Syed N, Coley HM, Sehouli J, Koensgen D,
Mustea A, Szlosarek P, McNeish I, Blagden SP, Schmid P, Lovell DP,
et al: Polo-like kinase Plk2 is an epigenetic determinant of
chemosensitivity and clinical outcomes in ovarian cancer. Cancer
Res. 71:3317–3327. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Syed N, Smith P, Sullivan A, Spender LC,
Dyer M, Karran L, O'Nions J, Allday M, Hoffmann I, Crawford D, et
al: Transcriptional silencing of Polo-like kinase 2 (SNK/LK2) is a
frequent event in B-cell malignancies. Blood. 107:250–256. 2006.
View Article : Google Scholar
|
|
19
|
Liu F, Zhang S, Zhao Z, Mao X, Huang J, Wu
Z, Zheng L and Wang Q: MicroRNA-27b up-regulated by human
papillomavirus 16 E7 promotes proliferation and suppresses
apoptosis by targeting polo-like kinase2 in cervical cancer.
Oncotarget. 7:19666–19679. 2016.PubMed/NCBI
|
|
20
|
Gee HE, Buffa FM, Harris AL, Toohey JM,
Carroll SL, Cooper CL, Beith J, McNeil C, Carmalt H, Mak C, et al:
MicroRNA-related DNA repair/cell-cycle genes independently
associated with relapse after radiation therapy for early breast
cancer. Int J Radiat Oncol Biol Phys. 93:1104–1114. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Matthew EM, Yen TJ, Dicker DT, Dorsey JF,
Yang W, Navaraj A and El-Deiry WS: Replication stress, defective
S-phase checkpoint and increased death in Plk2-deficient human
cancer cells. Cell Cycle. 6:2571–2578. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hu Z, Xu Z, Liao X, Yang X, Dong C, Luk K,
Jin A and Lu H: Polo-like kinase 2 acting as a promoter in human
tumor cells with an abundance of TAp73. Onco Targets Ther.
8:3475–3488. 2015.PubMed/NCBI
|
|
23
|
Li J, Ma W, Wang PY, Hurley PJ, Bunz F and
Hwang PM: Polo-like kinase 2 activates an antioxidant pathway to
promote the survival of cells with mitochondrial dysfunction. Free
Radic Biol Med. 73:270–277. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ou B, Zhao J, Guan S, Wangpu X, Zhu C,
Zong Y, Ma J, Sun J, Zheng M, Feng H, et al: Plk2 promotes tumor
growth and inhibits apoptosis by targeting Fbxw7/Cyclin E in
colorectal cancer. Cancer Lett. 380:457–466. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Marisa L, de Reyniès A, Duval A, Selves J,
Gaub MP, Vescovo L, Etienne-Grimaldi MC, Schiappa R, Guenot D,
Ayadi M, et al: Gene expression classification of colon cancer into
molecular subtypes: Characterization, validation, and prognostic
value. PLoS Med. 10:e10014532013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Barretina J, Caponigro G, Stransky N,
Venkatesan K, Margolin AA, Kim S, Wilson CJ, Lehár J, Kryukov GV,
Sonkin D, et al: The cancer cell line encyclopedia enables
predictive modelling of anticancer drug sensitivity. Nature.
483:603–607. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
28
|
Wong HL, Lee B, Field K, Lomax A, Tacey M,
Shapiro J, McKendrick J, Zimet A, Yip D, Nott L, et al: Impact of
primary tumor site on bevacizumab efficacy in metastatic colorectal
cancer. Clin Colorectal Cancer. 15:e9–e15. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Perron L, Daigle JM, Vandal N, Guertin MH
and Brisson J: Characteristics affecting survival after locally
advanced colorectal cancer in Quebec. Curr Oncol. 22:e485–e492.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li W, Zhi W, Zou S, Qiu T, Ling Y, Shan L,
Shi S and Ying J: Distinct clinicopathological patterns of mismatch
repair status in colorectal cancer stratified by KRAS mutations.
PLoS One. 10:e01282022015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Burns TF, Fei P, Scata KA, Dicker DT and
El-Deiry WS: Silencing of the novel p53 target gene Snk/Plk2 leads
to mitotic catastrophe in paclitaxel (taxol)-exposed cells. Mol
Cell Biol. 23:5556–5571. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Shimizu-Yoshida Y, Sugiyama K,
Rogounovitch T, Ohtsuru A, Namba H, Saenko V and Yamashita S:
Radiation-inducible hSNK gene is transcriptionally regulated by p53
binding homology element in human thyroid cells. Biochem Biophys
Res Commun. 289:491–498. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yang W, Soares J, Greninger P, Edelman EJ,
Lightfoot H, Forbes S, Bindal N, Beare D, Smith JA, Thompson IR, et
al: Genomics of drug sensitivity in cancer (GDSC): A resource for
therapeutic biomarker discovery in cancer cells. Nucleic Acids Res.
41:D955–D961. 2013. View Article : Google Scholar :
|
|
34
|
Valenti F, Fausti F, Biagioni F, Shay T,
Fontemaggi G, Domany E, Yaffe MB, Strano S, Blandino G and Di
Agostino S: Mutant p53 oncogenic functions are sustained by Plk2
kinase through an autoregulatory feedback loop. Cell Cycle.
10:4330–4340. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Dabiri Y, Kalman S, Gürth CM, Kim JY,
Mayer V and Cheng X: The essential role of TAp73 in
bortezomib-induced apoptosis in p53-deficient colorectal cancer
cells. Sci Rep. 7:54232017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Herreros-Villanueva M, Muñiz P,
García-Girón C, Cavia-Saiz M and del Corral MJ: TAp73 is one of the
genes responsible for the lack of response to chemotherapy
depending on B-Raf mutational status. J Transl Med. 8:152010.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lorenzi F, Babaei-Jadidi R, Sheard J,
Spencer-Dene B and Nateri AS: Fbxw7-associated drug resistance is
reversed by induction of terminal differentiation in murine
intestinal organoid culture. Mol Ther Methods Clin Dev.
3:160242016. View Article : Google Scholar : PubMed/NCBI
|