Introduction
Gastric cancer ranks as the second most common
malignant disease and the third leading cause of cancer-associated
mortality in developing countries (1). Chemotherapy is one of the principal
therapeutic approaches used in the treatment of cancer.
Chemoresistance has been acknowledged as a major obstacle in
successful cancer treatment. Paclitaxel is widely used as a
front-line chemotherapeutic agent for gastric cancer; however,
resistance limits the effectiveness of chemotherapy and results in
treatment failure in the majority of cases (2,3). An
improved understanding of the mechanism of chemoresistance to
paclitaxel may provide novel therapeutic approaches for gastric
cancer therapy; however, the underlying mechanisms of paclitaxel
resistance in gastric cancer are not well understood.
Exosomes are a subset of extracellular microvesicles
with a size ranging from 40-100 nm and are released by all types of
cells (4). Increasing evidence has
demonstrated that exosomes are intercellular mediators that
contribute to the hallmarks of cancer by influencing tumor growth,
metastasis, angiogenesis, immune regulation (5-7).
Recently, increasing attention to the role of exosomes in
chemoresistance has been gained (8-10).
It is well known that exosomes are nanosized vesicles with a lipid
bilayer membrane and can carry a variety of cell-of-origin cargo,
including DNA, RNA and proteins (11). Chemotherapy may alter the exosomal
composition of tumor cells (12-14);
the potential role of exosomes in chemoresistance regulation may be
determined by analyzing exosomal cargo.
Among the molecular components present as exosomal
cargo, microRNAs (miRNAs/miRs), as a type of noncoding small RNA,
have been extensively studied in the regulation of chemoresistance
(15,16); however, the role and mechanism of
exosomal miRNAs from chemoresistant cancer cells in the variations
of phenotypic chemoresistance remain unclear. miRNA expression
profile analysis of chemoresistant cancer cell-derived exosomes has
been conducted in recent years. Bioinformatics analysis has
revealed that target genes regulated by these dysregulated exosomal
miRNAs were associated with drug resistance (17-19).
Experimental evidence has demonstrated that exosomal miRNAs could
be shuttled from chemoresistant to chemosensitive cancer cells to
transmit chemoresistance (19-21).
Recently, researchers have revealed that exosomal
miR-155-5p served an important role in the regulation of
chemoresistance. Challagundla et al (22) firstly reported that exosomal
miR-155-5p mediated cross-talk between monocyte and neuroblastoma
cells to promote cancer cell chemoresistance. In addition, Patel
et al (23) and Mikamori
et al (24) revealed that
miR-155-5p expression levels were upregulated in cancer cells and
their exosomes following exposure to gemcitabine. Exosomes derived
from gemcitabine-treated pancreatic cancer cells mediated the
acquisition of chemo-resistance via the delivery of miR-155-5p into
the sensitive cells (23,24). Additionally, Santos et al
(25) reported that doxorubicin
(DOX)- and paclitaxel-resistant breast cancer cells transmitted
chemoresistance to neighboring cancer cells by exosomal delivery of
miR-155-5p. These findings suggested that exosomal miR-155-5p may
be a very important signaling molecule to transmit chemoresistance
from drug-resistant to drug-sensitive cancer cells; however, the
role and mechanism of chemoresistant cancer cell-derived exosomal
miR-155-5p in this process require further investigation. Whether
exosomal miR-155-5p mediates the transmission of paclitaxel
resistance in gastric cancer cells remains unknown.
In the present study, a paclitaxel-resistant gastric
cancer cell line MGC-803 (MGC-803R) was established, and the
cellular morphological characteristics and miR-155-5p expression
levels between MGC-803R cells and sensitive (MGC-803S) cells were
compared. Cancer cell-derived exosomes were then isolated and
characterized, followed by analysis of the role and mechanism of
exosomal miR-155-5p in transmitting a chemoresistance phenotype
from paclitaxel-resistant to paclitaxel-sensitive gastric cancer
cells.
Materials and methods
Establishment of a paclitaxel-resistant
MGC-803 cell line
The human gastric cancer cell line MGC-803 was
obtained from the Cell Bank of Type Culture Collection of Chinese
Academy of Sciences (Shanghai, China). The cells were cultured in
Dulbecco’s modified Eagle’s medium (DMEM; Gibco; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) supplemented with 10% fetal
bovine serum (FBS; Gibco; Thermo Fisher Scientifics, Inc.) and
incubated at 37°C in a humidified incubator with 5% CO2.
Paclitaxel-resistant MGC-803R cells were established by continuous
exposure to stepwise-increasing concentrations of paclitaxel
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany). MGC-803 cells were
initially cultured in DMEM containing a low concentration of
paclitaxel (1 µg/l). The cells were then sub-cultured every
2 weeks in DMEM with increasing concentrations of the drug, a 25%
increase each time. Finally, cells that were viable in the cell
culture medium with a high concentration of paclitaxel (100
µg/l) were designated as paclitaxel-resistant MGC-803R
cells. These cells were maintained in the drug-containing medium
following induction, but were cultured in drug-free medium for 1
week at 37°C prior to subsequent experimentation. Parental cells,
denoted as MGC-803S, were cultured under the same conditions
without treatment.
Cell proliferation analysis
MGC-803R and MGC-803S cells were seeded in 96-well
plates (5,000 cells/well) and exposed to increasing concentrations
of paclitaxel for 48 h at 37°C. The concentrations of paclitaxel
used for the drug dose-response curve analysis of MGC-803R cells
were 0, 100, 200, 300, 400, 500, 600 and 700 µg/l, while the
concentration of drug used for MGC-803S cells was 0, 2, 6, 8, 10,
12 and 14 µg/l. The proliferative ability of cells was
determined with a Cell Counting Kit-8 (CCK-8) kit (MedChemExpress,
Monmouth Junction, NJ, USA) according to the manufacturer’s
protocols and presented as cell viability (%). The absorbance was
measured at 450 nm using a Cytation 5 cell imaging multi-mode
reader (BioTek Instruments, Inc., Winooski, VT, USA). The
half-maximal inhibitory concentration (IC50) of drugs
was calculated using GraphPad Prism version 5.0 software (GraphPad
Software, Inc., La Jolla, CA, USA). The resistant index (RI) was
calculated utilizing the following formula: RI = IC50 of
resistant cells/IC50 of sensitive cells. Exosomes were
isolated from MGC-803S cells, MGC-803R cells and cancer cells
following transfection with oligonucleotides (described below). For
exosome treatment analysis, each well in a 96-well plate was seeded
with 1,000 cells and loaded with exosomes at 100 µg/ml for
48 h for functional analysis at 37°C; untreated cells served as the
control. The cell culture medium was then removed and fresh medium
containing the IC50 concentration of paclitaxel was
added to each well for 48 h. At the end of treatment, cell
proliferation was measured. The inhibition rate (%) of cells by
drug was calculated as 1-cell viability (%).
Exosome isolation and
characterization
Gastric cancer cells were cultured in freshly
prepared DMEM containing exosome-free FBS for 48 h until cells had
reached 90% confluence. Cell culture supernatants were collected
and filtered using 0.22-µm pore filters (Merck KGaA),
followed by differential centrifugation at 4°C: 300 × g for 10 min
to remove cells, 2,000 × g for 15 min to remove cell debris and
10,000 × g for 30 min to remove large particles. The cell culture
supernatants were then concentrated by ultrafiltration with 30 kDa
Amicon filter membranes (Merck KGaA) to remove a large amount of
small soluble proteins. Finally, the exosomes were isolated using
ExoQuick exosome precipitation solution (SBI System Biosciences,
Palo Alto, CA, USA) as previously described (26). Exosomes were fixed in 1%
glutaraldehyde for 5 min at room temperature. The morphology of
exosomes was examined via transmission electron microscopy
(×30,000) in a total of 6 fields. The exosomal protein
concentration was measured by a Bicinchoninic Acid colorimetric
method and exosome-associated protein cluster of differentiation 9
(CD9) expression was analyzed by western blotting (described
below). The number and size distribution of exosomes were detected
by a Nanosight NS300 system (Nanosight™ Technology; Malvern
Panalytical Ltd., Malvern, UK) and analyzed using Nanoparticle
Tracking Analysis (NTA) software (version 3.1, Malvern Panalytical
Ltd.).
Exosome labeling, internalization and
immunofluorescence staining
Exosomes (25 µg/µl) from MGC-803S
cells and MGC-803R cells were pre-labeled with the red fluorescent
dye CM-Dil (Invitrogen; Thermo Fisher Scientific, Inc.) at 37°C for
1 h, washed with PBS and centrifuged at 110,000 × g at 4°C for 70
min to remove excess dye; 8×104 of MGC-803S cells were
seeded in 12-well plates and incubated at 37°C with the labeled
exosomes for 4 h. Unlabeled exosomes were used as a negative
control. The cells were fixed with 4% paraformaldehyde at room
temperature for 1 h, permeabilized with 0.1% Triton X-100 and
blocked with 5% bovine serum albumin (BSA) (Wuhan Boster Biological
Technology, Ltd., Wuhan, China) at room temperature for 20 min. The
cells were then incubated with rabbit monoclonal anti-α-smooth
muscle actin (α-SMA) antibody (cat. no. A03744, dilution: 1:100,
Wuhan Boster Biological Technology Co. Ltd.) at 4°C overnight,
followed by incubation with Alexa Fluor® 488-conjugated
anti-rabbit secondary antibody (cat. no. A-11034, dilution:
1:1,000, Invitrogen; Thermo Fisher Scientific, Inc.) at 37°C for 1
h; the nuclei were stained with Hoechst 33342 dye at room
temperature for 10 min. Incorporation of exosomes into target cells
was visualized ≥6 field by fluorescence microscopy (magnification,
×400, Zeiss AG, Oberkochen, Germany).
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
analysis
Total RNA from cells and exosomes was extracted
using an miRNeasy Mini Kit (Qiagen, Inc., Hilden, Germany). miRNAs
and mRNAs were reverse transcribed using the miScript II RT Kit
(Qiagen, Inc.) and detected by miScript SYBR Green PCR Kit (Qiagen,
Inc.) according to the manufacturer’s protocols. The amplification
of fluorescence signals was detected by a fluorescence thermal
cycler (Bio-Rad Laboratories, Inc., Hercules, CA, USA). RNU6B and
β-actin were set as internal controls for cellular miRNAs and
mRNAs, respectively. The relative expression levels of exosomal
miRNA were normalized to miR-16-5p. The 2−ΔΔCq method
was used for miRNAs and mRNAs quantification (27); miScript primers for miRNAs and
RNU6B (cat. no. 218300) were obtained from Qiagen, Inc. The primers
sequences for mRNAs detection and the thermocycling conditions
applied are listed in Table I;
experiments were conducted ≥3 times.
 | Table IPrimers sequences and amplification
conditions for reverse transcription-quantitative polymerase chain
reaction of mRNAs. |
Table I
Primers sequences and amplification
conditions for reverse transcription-quantitative polymerase chain
reaction of mRNAs.
| Genes | Primers sequences
(5′-3′) | Annealing
temperatures (°C) | Product length
(bp) |
|---|
| β-actin | For:
CACGAAACTACCTTCAACTCC | 56 | 265 |
| Rev:
CATACTCCTGCTTGCTGATC | | |
| E-cadherin | For:
CGCATTGCCACATACACTCT | | |
| Rev:
TTGGCTGAGGATGGTGTAAG | 60 | 252 |
| Vimentin | For:
GAGCTGCAGGAGCTGAATG | | |
| Rev:
AGGTCAAGACGTGCCAGAG | 60 | 344 |
| GATA3 | For:
ATCTGACCGAGCAGGTCGTA | | |
| Rev:
GGCGACGACTCTGCAATTCT | 63 | 145 |
| TP53INP1 | For:
CTTCCTCCAACCAAGAAC | | |
| Rev:
CTCCTCCATTGGACATGAC | 55 | 249 |
Western blot analysis
Cellular proteins were extracted with
radioimmunoprecipitation buffer (Vazyme, Piscataway, NJ, USA),
supplemented with protease inhibitors. Protein concentration was
determined by Bicinchoninic Acid protein quantification kit
(Vazyme). A total of 40 µg of protein for each group was
separated on 12% SDS-PAGE gels and transferred to 0.45 µm
polyvinylidene difluoride membranes (Merck KGaA). Membranes were
then blocked with 5% BSA and incubated with primary antibodies
against E-cadherin (CDH1 polyclonal antibody, dilution: 1:1,000,
cat. no. A3044, ABclonal Biotechnology Co., Ltd., Wuhan, China),
Vimentin (vimentin mouse monoclonal antibody, dilution: 1:800, cat.
no. MB9006, Bioworld Technology, Inc., St. Louis Park, MN, USA) and
CD9 (CD9 polyclonal antibody, dilution: 1:1,000, cat. no. BS60359,
Bioworld Technology, Inc.) followed by incubation with
peroxidase-conjugated secondary antibodies [goat anti-rabbit IgG
(H+L)-horseradish peroxidase (HRP), dilution: 1:2,500, cat. no.
BS13278; goat anti-mouse IgG (H+L)-HRP, dilution: 1:2,000, cat. no.
BS50350, Bioworld Technology, Inc.) at 37°C for 1 h. β-actin
[β-actin (I102) poly-clonal antibody, dilution: 1:5,000, cat. no.
AP0060, Bioworld Technology, Inc.] was used as the loading control
for cellular protein. Bands were visualized by chemiluminescence
using immobilon ECL Ultra Western HRP Substrate (Merck KGaA)
according to the manufacturer’s protocols.
Cell transfection
Oligonucleotides, including miR-155-5p mimics,
mimics negative control (MNC), miR-155-5p inhibitor (inhibitor) and
inhibitor negative control (INC) were synthesized and purified by
Shanghai GenePharma Co., Ltd. (Shanghai, China). The sequences of
the oligonucleotides were described as previously (28). Cells were seeded at a density of
1.0×105 cells/well in 6-well plates and incubated
overnight, followed by transfection with 5 nM mimics or 100 nM
inhibitor using Lipofectamine® 2000 (Invitrogen; Thermo
Fisher Scientific, Inc.); correspondingly, MNC and INC were used as
controls for mimics and inhibitor, respectively. Following
transfection for 24 h, the cells were collected, seeded into
96-well plates at a density of 5,000 cells per well and treated
with IC50 level of paclitaxel at 37°C for 48 h. Cell
proliferation was then measured as aforementioned. Successful
transfection was determined via RT-qPCR. Following transfection for
48 h, additional cells were collected for RNA and protein
extraction. For exosome isolation, cells were refreshed with DMEM
containing exosome-free FBS after transfection for 4-6 h. Following
culture for 48 h, the cell culture medium was collected for exosome
isolation.
Luciferase reporter construction and
analysis
The potential binding sites of miR-155-5p to the 3′
untranslated region (UTR) of GATA binding protein 3 (GATA3) and
tumor protein p53-inducible nuclear protein 1 (TP53INP1) mRNAs were
predicted by TargetScan analysis (http://www.targetscan.org, release 7.2). The 3′UTR
fragments of the two mRNAs containing the binding sites were
synthesized and purified by Invitrogen (Thermo Fisher Scientific,
Inc.); corresponding mutant fragments were also designed and
synthesized. These oligonucleotides were annealed and inserted
between SacI and SalI restriction sites of the
pmirGLO dual-luciferase miRNA target expression vectors (Promega
Corporation, Madison, WI, USA). Sequences of these oligonucleotides
are listed in Table II. For miRNA
functional analysis, MGC-803S cells were seeded into 24-well plates
overnight and then co-transfected with 5 nM of mimics, MNC and 250
ng of reporter plasmids using Lipofectamine 2000. For exosome
analysis, MGC-803S cells were transfected with luciferase reporter
vectors for 4-6 h and then incubated with exosomes at 37°C for 48
h. Luciferase activities were quantified using the Dual Glo
Luciferase Assay System (Promega Corporation) and normalized to
activity of Renilla luciferase (Promega Corporation).
 | Table IISequences of the synthesized
oligonucleotides employed for the dual-luciferase assay. |
Table II
Sequences of the synthesized
oligonucleotides employed for the dual-luciferase assay.
|
Oligonucleotides | Sequences
|
|---|
| Sense (5′-3′) | Antisense
(5′-3′) |
|---|
| GATA3 mRNA |
CTAGCGGCCGCTAGTCAGTTGTT |
TCGACTTTATTTTCTTTTAATGCATCAA |
| 3′UTR containing
the binding sites (wild-type) |
TGATGCATTAAAAGAAAATAAAG |
ACAACTGACTAGCGGCCGCTAGAGCT |
| GATA3 mRNA |
CTAGCGGCCGCTAGTCAGTTGTTT |
TCGACTTTATTTTCTTCGCCGATATCAA |
| 3′UTR containing
the mutant sites (mutant type) |
GATATCGGCGAAGAAAATAAAG |
ACAACTGACTAGCGGCCGCTAGAGCT |
| TP53INP1 mRNA |
CTAGCGGCCGCTAGTCACACTAA |
TCGACCTATCACCTGTTAATGCTAATGT |
| 3′UTR containing
the binding sites (wild-type) |
CATTAGCATTAACAGGTGATAGG |
TAGTGTGACTAGCGGCCGCTAGAGCT |
| TP53INP1 mRNA |
CTAGCGGCCGCTAGTCACACTAA |
TCGACCTATCACCTGGCCGCTAGAATGT |
| 3′UTR containing
the mutant sites (mutant type) |
CATTCTAGCGGCCAGGTGATAGG |
TAGTGTGACTAGCGGCCGCTAGAGCT |
Statistical analysis
All data are presented as the mean ± standard error
of the mean from at least three independent experiments. A
Student’s t-test, one-way analysis of variance and a Tukey’s post
hoc test were performed using GraphPad Prism 5 software. P<0.05
was considered to indicate a statistically significant
difference.
Results
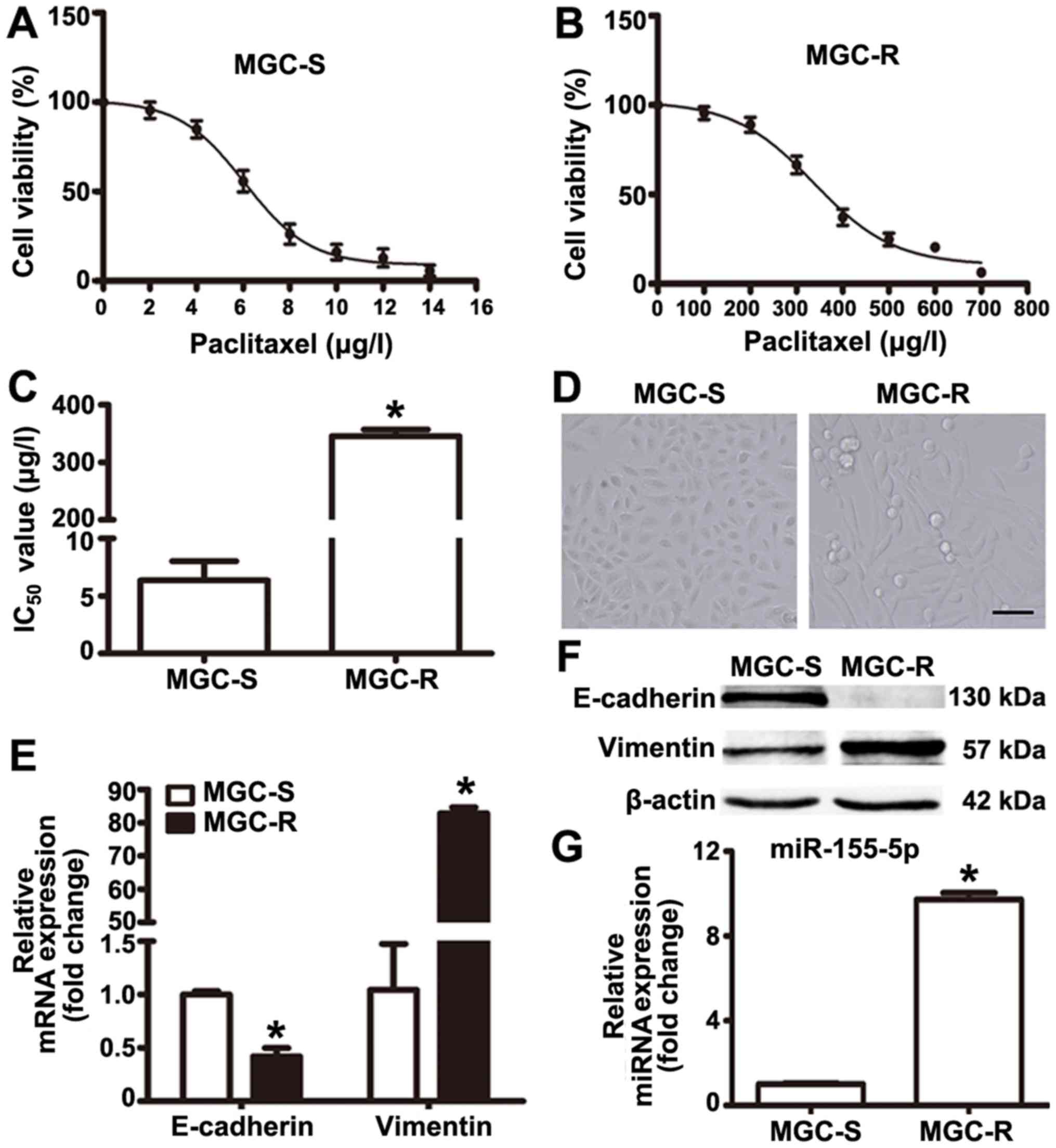
Paclitaxel-resistant gastric cancer cell
line MGC-803R exhibits epithelial-mesenchymal transition (EMT) and
upregulated levels of miR-155-5p
Paclitaxel-resistant MGC-803R cells were generated
via the treatment of paclitaxel-sensitive parental MGC-803S cells
with gradually increasing doses of paclitaxel in successive
passages. MGC-803R and MGC-803S cells were exposed to various
concentrations of paclitaxel for 48 h to determine
paclitaxel-induced-cytotoxicity. The drug dose-response curves for
the two cell lines are presented based on cell proliferation as
determined by CCK-8 assays (Fig. 1A
and B). The IC50 values for MGC-803R and MGC-803S
cells were 345.4±11.67 and 6.06±1.68 µg/l, respectively
(Fig. 1C). Compared with MGC-803S
cells, the RI of MGC-803R cells was determined to be 57.5 (Fig. 1C).
In addition, it was observed that MGC-803R cells
became elongated with a mesenchymal-like morphology, which was
markedly different from the epithelial-like morphology of MGC-803S
cells (Fig. 1D). To confirm that
these morphological changes were indicative of EMT in MGC-803R
cells, the mRNA and protein expression levels of E-cadherin and
Vimentin were determined by RT-qPCR and western blotting. The
results revealed notably increased levels of Vimentin and reduced
levels of E-cadherin protein in MGC-803R cells compared with in
MGC-803S cells (Fig. 1E); however,
a significant increase and decrease in the expression levels of
E-cadherin and Vimentin were respectively reported in MGC-803R
cells compared with MGC-803S cells (Fig. 1F). miRNA detection analysis
demonstrated that the expression levels of miR-155-5p were
significantly higher in MGC-803R cells than in MGC-803S cells
(Fig. 1G). These results indicate
that MGC-803 cells exhibit EMT phenotype and increased levels of
miR-155-5p following induction by paclitaxel treatment.
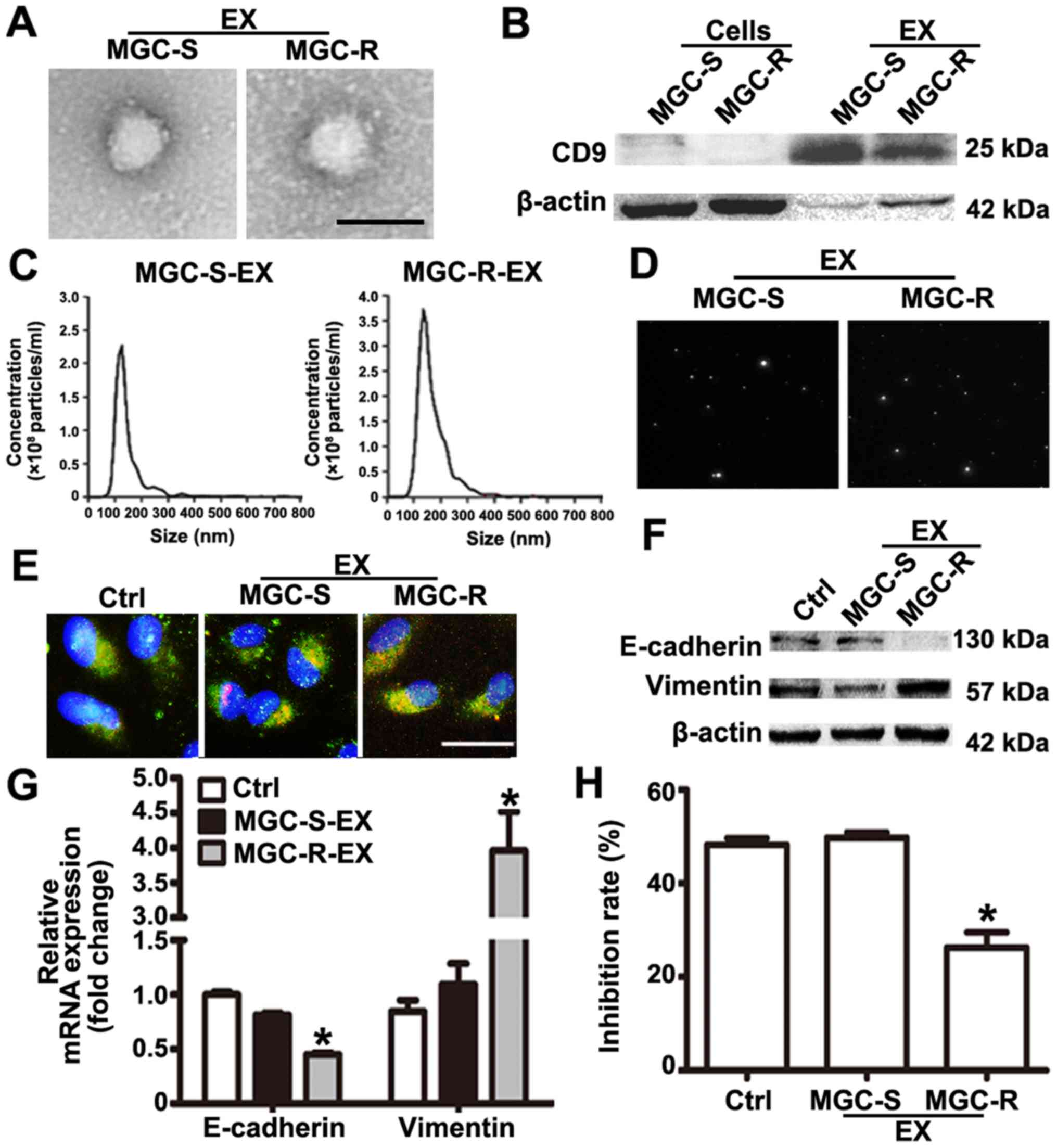
MGC-803R-exosomes induce EMT and a
paclitaxel-resistant phenotype in MGC-803S cells
Exosomes act as an important mediator of
intercellular communication (11).
Studies have indicated that chemoresistant tumor cells can release
exosomes that transmit drug resistance during tumorigenesis
(12-14). To analyze whether MGC-803R
cell-derived exosomes may confer a malignant phenotype on
paclitaxel-sensitive cancer cells, exosomes from the culture medium
of MGC-803R and MGC-803S cells were isolated. Purified exosomes
from the cultures exhibited typical cup-shaped morphology with
positive expression of exosomal marker CD9 (Fig. 2A and B). The mean size distribution
and concentration of exosomes were determined with a Nanosight™
system. The mean size of MGC-803S-exosomes was 150±12.7 nm, while
that of MGC-803R-exosomes was 168±13.2 nm (Fig. 2C). According to the size
distribution of tumor exosomes detected by NTA, these results
indicated that the detected size corresponded with the expected
size of exosomes (29). Typical
images of two cell-derived exosomes demonstrated the presence of
particles with a concentration of more than 2.0×108
particles/ml (Fig. 2D). There was
no notable difference in exosome quantities extracted from MGC-803R
and MGC-803S cells. To demonstrate that exosomes could be taken up
by the recipient cells, MGC-803S cells were incubated with
CM-Dil-labeled exosomes. As presented in Fig. 2E, CM-Dil red fluorescence signals
were visible around the nuclei and were also in the cytoplasm of
MGC-803S cells following exposure to MGC-803R-derived exosomes or
MGC-803S-exosomes; however, the negative control did not exhibit
any red fluorescence. This suggested the uptake of CM-Dil-labeled
exosomes by MGC-803S cells.
To investigate whether MGC-803R-exosomes were
responsible for the spread of chemoresistance, MGC-803S cells were
incubated with MGC-803R-exosomes or MGC-803S-exosomes for 48 h. The
treated MGC-803S cells were then collected for the analysis of
E-cadherin and Vimentin mRNA and protein expression. The protein
expression levels of E-cadherin in MGC-803R-exosome-treated cells
were notably lower and those of Vimentin were markedly increased
compared with the control (Fig.
2F). Compared with MGC-803S-exosome treatment and the untreated
control groups, MGC-803R-exosomes significantly reduced the mRNA
expression levels of E-cadherin and increased that of Vimentin in
MGC-803S cells (Fig. 2G).
Furthermore, MGC-803S cells were also collected and treated with
paclitaxel for chemosensitivity analysis following incubation with
exosomes. CCK-8 assays revealed that MGC-803R-exosome treatment
significantly reduced the chemosensitivity of MGC-803S cells, while
MGC-803S-exosomes had no such effect compared with the control
(Fig. 2H). These data suggested
that the induction of EMT and a paclitaxel-resistant phenotype in
MGC-803S cells may be ascribed to MGC-803R-exosomes.
Overexpression of miR-155-5p in MGC-803S
cells exhibits similar effects to treatment with
MGC-803R-exosomes
Previous studies revealed that exosomal miR-155-5p
may serve an important role in chemoresistance (22-24).
The present study reported that miR-155-5p expression levels were
significantly elevated in MGC-803R cells. MGC-803R-exosome
treatment was proposed to confer EMT and chemoresistant phenotypes
to MGC-803S cells in the present study. To investigate whether
miR-155-5p was selectively compartmen-talized into
MGC-803R-exosomes, RT-qPCR was performed to detect miR-155-5p
expression levels in MGC-803R- and MGC-803S-exosomes. The results
revealed that miR-155-5p was significantly enriched in
MGC-803R-exosomes compared with MGC-803S-exosomes (Fig. 3A). In addition, miR-155-5p
expression levels in MGC-803S cells following treatment with
exosomes were determined. The expression levels of miR-155-5p were
significantly increased in MGC-803S cells following incubation with
MGC-803R-exosomes compared with the control (Fig. 3B). These results indicated that
miR-155-5p was enriched in MGC-803R-exosomes and could be delivered
to MGC-803S cells via exosomal transfer.
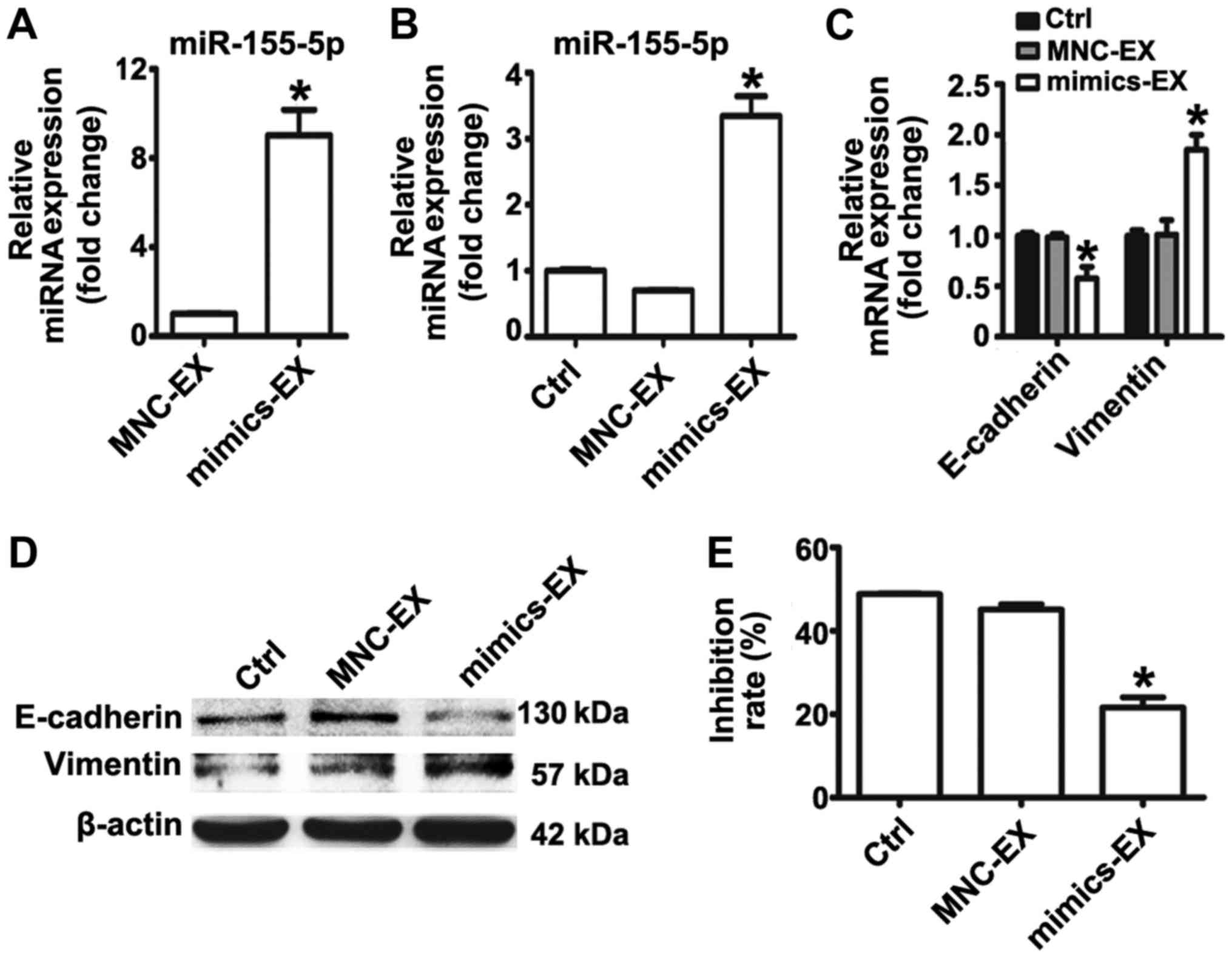
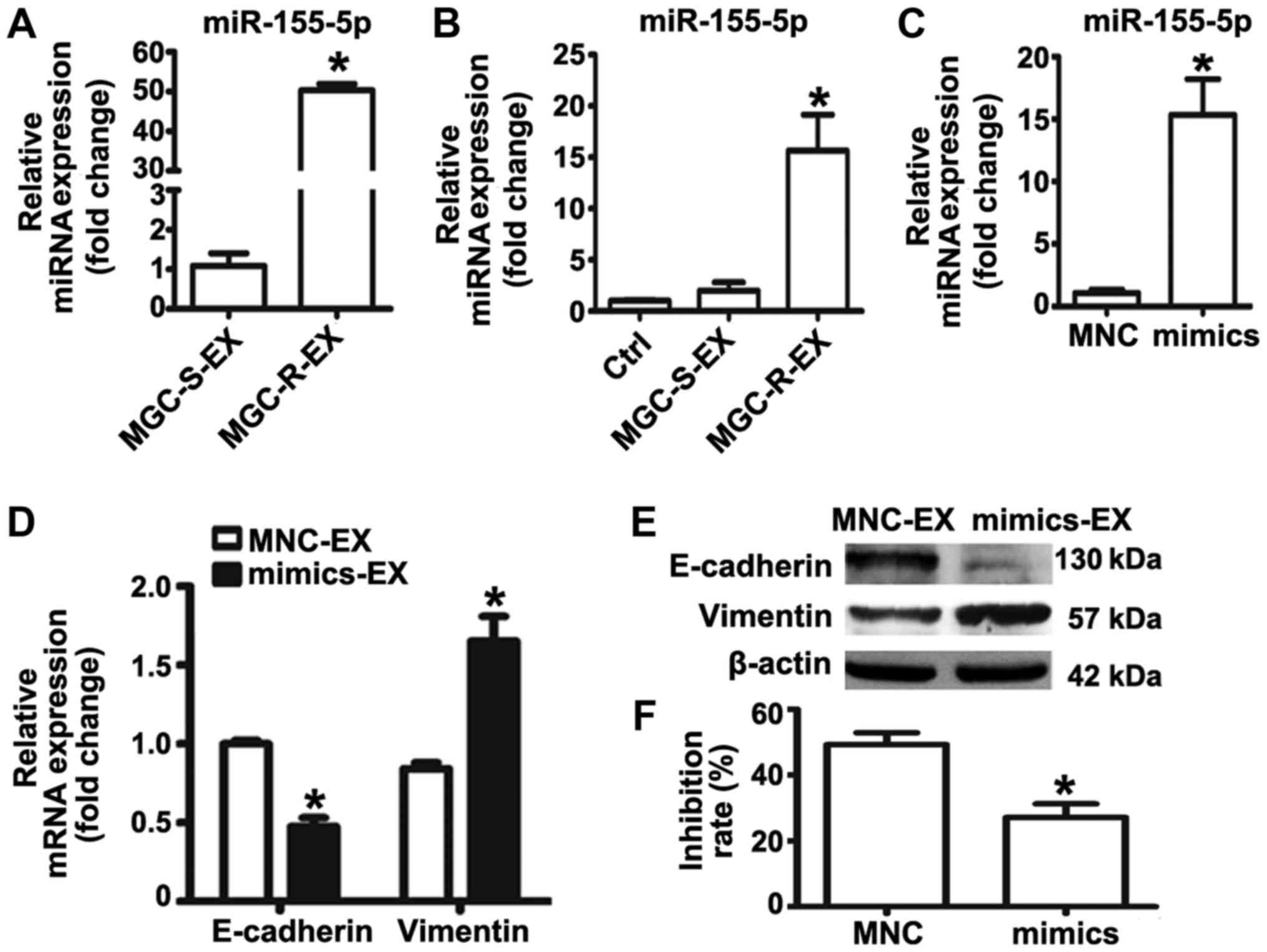 | Figure 3miR-155-5p overexpression confers
epithelial-mesenchymal transition and paclitaxel-resistant
phenotypes on MGC-803S cells. (A) miR-155-5p expression levels in
MGC-S-EX and MGC-R-EX were detected by RT-qPCR; (B) Analysis of
miR-155-5p expression levels in MGC-803S cells following incubation
with MGC-S-EX or MGC-R-EX. (C) MGC-803S cells were transfected with
miR-155-5p mimics or MNC. Following transfection for 48 h,
miR-155-5p expression levels were measured by RT-qPCR. (D)
Alterations in the mRNA expression levels of E-cadherin and
Vimentin in MGC-803S cells after transfection; (E) E-cadherin and
Vimentin protein expression levels as analyzed by western blotting.
(F) Analysis of the proliferation of MGC-803S cells following
transfection. *P<0.05 vs. MGC-S-EX, Ctrl, MNC-EX or
MNC. Ctrl, control; EX, exosome; miR, microRNA; MNC, mimics
negative control; MGC-R, paclitaxel-resistant; MGC-S,
paclitaxel-sensitive; RT-qPCR, reverse transcription-quantitative
polymerase chain reaction. |
To further investigate the role of miR-155-5p in EMT
and the regulation of paclitaxel chemoresistance, miRNA mimics were
used to overexpress miR-155-5p in MGC-803S cells. miR-155-5p
expression was observed to be significantly upregulated in the
mimics group compared with the control (Fig. 3C); the miR-155-5p expression levels
were similar to that of MGC-803S cells following MGC-803R-exosome
treatment (Fig. 3B). Detection of
mRNA and protein expression levels of EMT markers revealed that
transfection of miR-155-5p mimics resulted in significantly reduced
E-cadherin and increased Vimentin expression compared with the
control (Fig. 3D and E). MGC-803S
cells transfected with miRNA mimics were then exposed to
paclitaxel. Compared with MNC-transfected cells, those transfected
with miR-155-5p mimics exhibited significantly lower proliferative
abilities in response to paclitaxel treatment (Fig. 3F). The results of the present study
indicated that MGC-803R-exosomes may induce EMT and a
paclitaxel-resistant phenotype in MGC-803S cells via the transfer
of miR-155-5p.
Exosomal transfer of miR-155-5p promotes
EMT and paclitaxel chemoresistance in MGC-803S cells
To further validate that exosomal miR-155-5p is
involved in EMT and the induction of a paclitaxel-resistant
phenotype, exosomes from MGC-803S cells following transfection with
miRNA mimics or MNC were isolated. RT-qPCR analysis demonstrated
that miR-155-5p was significantly enriched in exosomes isolated
from mimic-transfected MGC-803S cells (Fig. 4A). MGC-803S cells were then treated
with mimics or MNC-transfected-MGC-803S cell-derived exosomes
(mimics-exosomes or MNC-exosomes, respectively). RT-qPCR analysis
revealed that miR-155-5p expression levels were consistently
elevated in MGC-803S cells following incubation with
mimics-exosomes (Fig. 4B). This
suggested that miR-155-5p may be delivered into MGC-803S cells via
exosomal transfer.
To evaluate whether mimics-exosomes treatment could
replicate the effect of MGC-803R-exosomes on MGC-803S cells,
alterations in the expression of EMT markers and the
chemosensitivity of MGC-803S cells following incubation with
mimics-exosomes were determined. The results revealed that
mimics-exosome treatment significantly inhibited E-cadherin
expression and enhanced Vimentin expression at the mRNA level;
notable differences at the protein level were observed (Fig. 4C and D). Additionally, the
proliferation of MGC-803S cells was significantly reduced by
mimics-exosomes treatment following incubation with paclitaxel
compared with MNC-exosomes and the control (Fig. 4E). These results indicated that
exosomal transfer of miR-155-5p may be involved in EMT and the
induction of a paclitaxel-resistant phenotype.
Exosomal miR-155-5p suppresses GATA3 and
TP53INP1 by directly targeting their 3′ UTRs
To determine the potential mechanism of exosomal
miR-155-5p in acquiring a malignant phenotype, the potential
targets of miR-155-5p were predicted by TargetScan. Among the list
of predicted targets, GATA3 and TP53INP1 have been reported to be
associated with the regulation of chemoresistance (30,31).
Therefore, GATA3 and TP53INP1 were selected as candidate targets
for further validation in the present study. According to the
predicted binding sites in the 3′UTR of GATA3 and TP53INP1 mRNAs,
the corresponding wild-type and mutant-type luciferase reporter
vectors were constructed, which were then co-transfected with
mimics or MNC into MGC-803S cells (Fig. 5A and B). Luciferase activity
analysis demonstrated that miR-155-5p mimics significantly
suppressed the relative luciferase activity of the wild-type
luciferase reporter vector groups compared with the MNC and mutant
groups (Fig. 5C and D).
Conversely, the relative luciferase activity was not affected by
mimics in the mutant-type luciferase reporter vector groups. The
results suggested that GATA3 and TP53INP1 were targets of
miR-155-5p. To further investigate the effect of MGC-803R-exosomes
and mimics-exosomes on relative luciferase activity, MGC-803S cells
were transfected with luciferase reporter vectors and then
incubated with MGC-803R-exosomes or mimics-exosomes. Compared with
the control, MGC-803R-exosomes and mimics-exosome treatment
significantly reduced the relative luciferase activity in wild-type
luciferase reporter vector groups; significant alterations in
luciferase activity in the mutant-type luciferase reporter vector
groups were not observed (Fig. 5E and
F). RT-qPCR analysis revealed that MGC-803R-exosome and
mimics-exosome treatments significantly downregulated GATA3 and
TP53INP1 mRNA levels compared with in the control group (Fig. 5G and H). Furthermore, the mRNA
expression levels of the two target genes were also compared
between MGC-803S and MGC-803R cells. As expected, TP53INP1 and
GATA3 mRNA expression levels were significantly suppressed in
MGC-803R cells compared with MGC-803S cells (Fig. 5I). The results suggested that
exosomal transfer of miR-155-5p may induce chemoresistance via the
suppression of GATA3 and TP53INP1 expression.
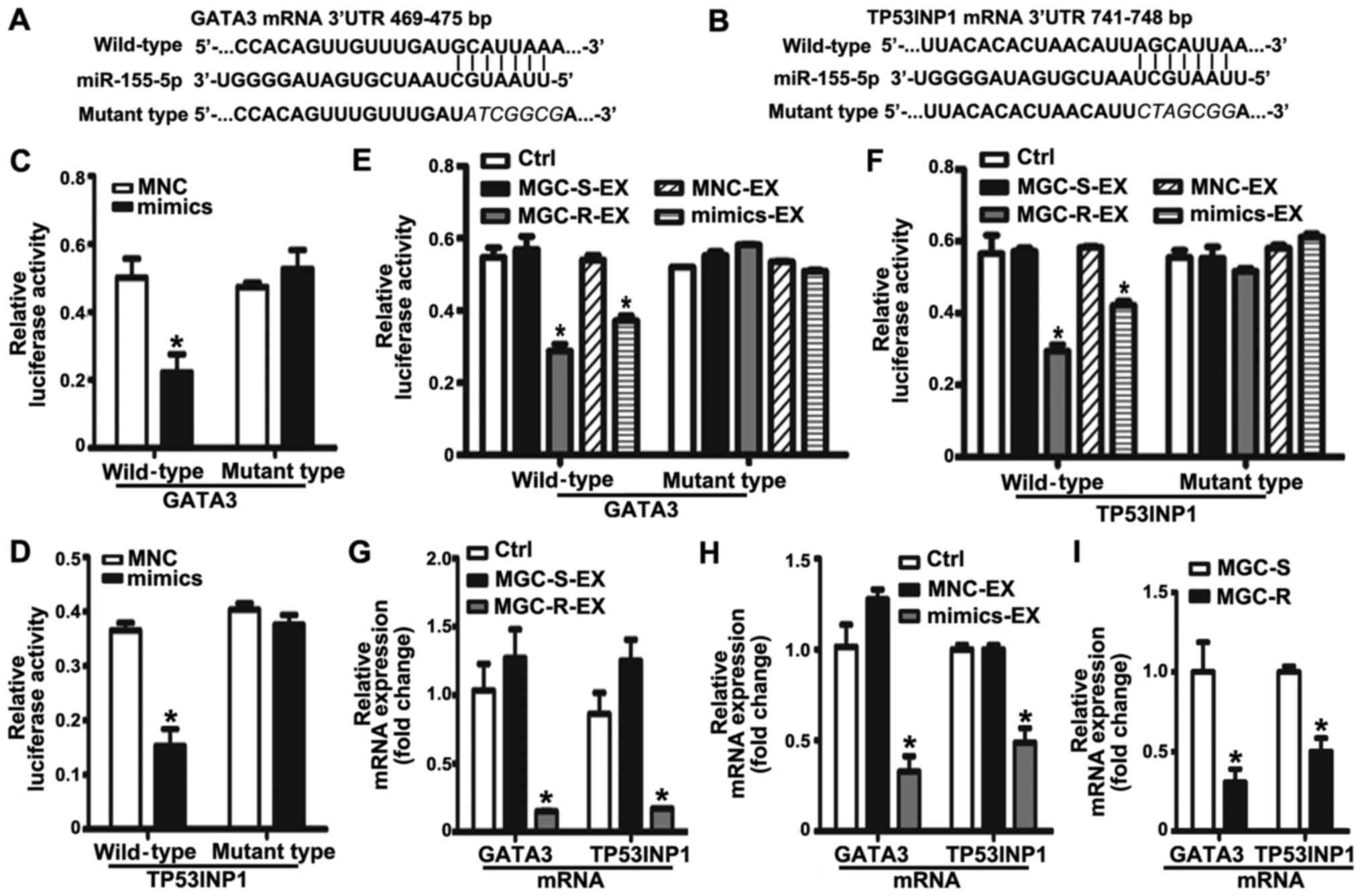 | Figure 5Exosomal miR-155-5p suppresses GATA3
and TP53INP1 by directly targeting their 3′-UTRs. The position and
sequences of the binding sites in the 3′UTR of target gene mRNAs,
(A) GATA3 and (B) TP53INP1 were predicted by TargetScan. DNA
fragments containing the wild-type or mutant binding sites were
designed and inserted into pmirGLO dual-luciferase miRNA target
expression vectors. (C and D) MGC-803S cells were co-transfected
with reporter vectors, and mimics or MNC. Cells were then collected
for luciferase activity assay. (E and F) The effects of exosomes on
the luciferase activity in MGC-803S cells. (G) GATA3 and TP53INP1
mRNA expression levels in MGC-803S cells following treatment with
MGC-R-EX and MGC-S-EX. (H) GATA3 and TP53INP1 mRNA expression
levels in MGC-803S cells after co-culturing with MNC-EX and
mimics-EX. (I) Comparison of GATA3 and TP53INP1 mRNA expression
levels between MGC-803S and MGC-803R cells. *P<0.05
vs. Ctrl, MNC, MNC-EX, MGC-S-EX or MGC-S. EX, exosome; GATA3, GATA
binding protein 3; MGC-R, paclitaxel-resistant; MGC-S,
paclitaxel-sensitive; TP53INP1, tumor protein p53-inducible nuclear
protein 1; 3′UTR, 3′-untranslated region. |
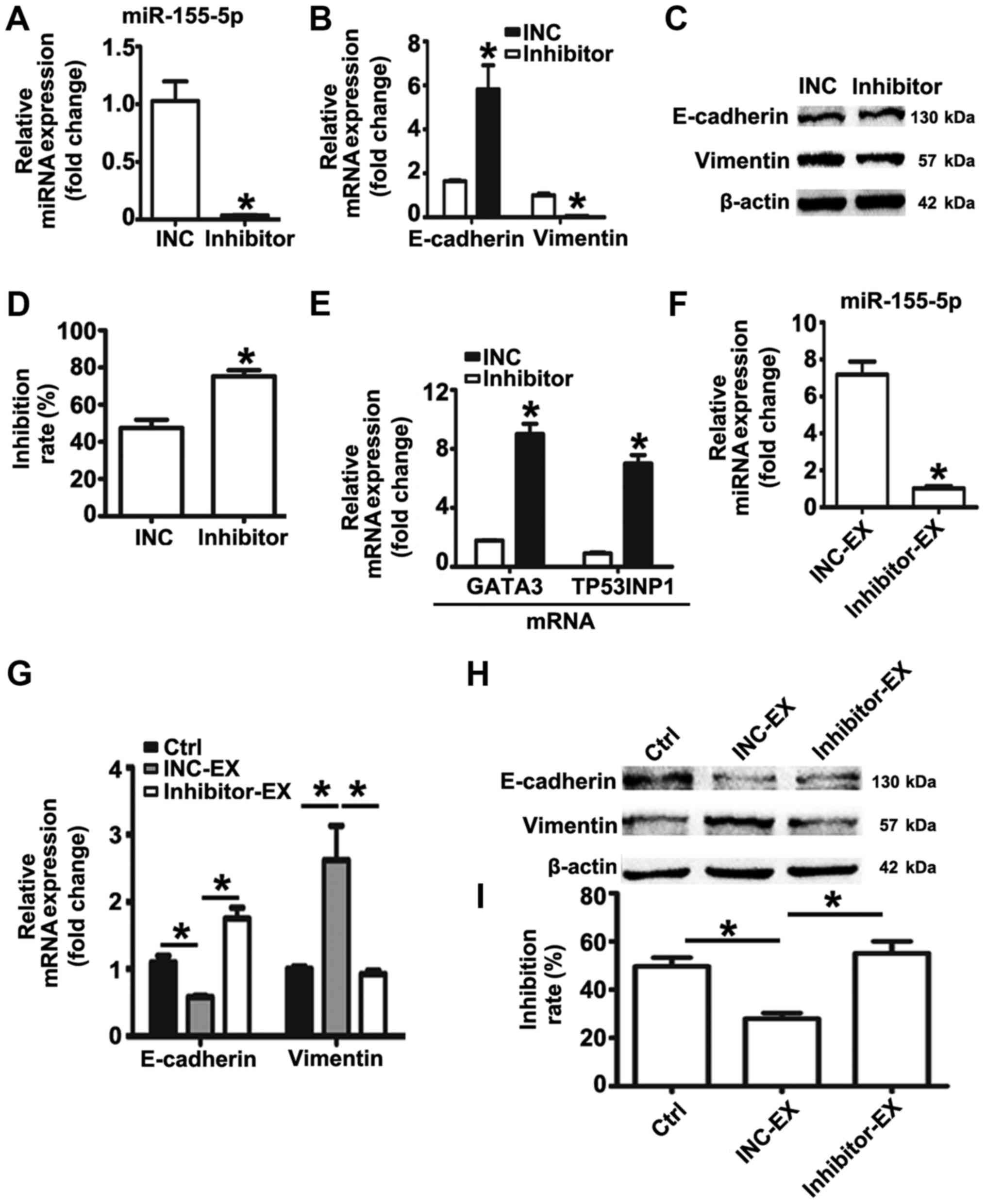
miR-155-5p inhibition reverses EMT and
the chemoresistant phenotype of MGC-803R cells, and suppresses the
induction of malignant phenotypes by MGC-803R-exosomes
The expression levels of miR-155-5p in MGC-803R
cells were significantly higher than in MGC-803S cells.
MGC-803R-exosomes was proposed to deliver miR-155-5p into
chemosensitive cancer cells, inducing EMT and paclitaxel-resistant
phenotypes in the present study. Based on these data, it was
suggested that knockdown of miR-155-5p may dysregulate the
malignant phenotypes of gastric cancer cells. Therefore, an miRNA
inhibitor was employed to down-regulate the expression levels of
miR-155-5p in MGC-803R cells. RT-qPCR detection revealed that
miR-155-5p expression levels were significantly suppressed by miRNA
inhibitor compared with the control (Fig. 6A). Compared with the INC group,
significantly increased mRNA expression levels of E-cadherin, and
reduced mRNA and protein levels of Vimentin were observed in
MGC-803R cells following transfection with miRNA inhibitor
(Fig. 6B and C); the protein
expression levels of E-cadherin were not significantly altered.
MGC-803R cells transfected with miRNA inhibitor or INC were then
subjected to paclitaxel treatment. Knockdown of miR-155-5p
significantly increased the chemosensitivity of MGC-803R cells to
paclitaxel compared with the INC group (Fig. 6D), which was accompanied with
significantly increased expression levels of GATA3 and TP53INP1
mRNA (Fig. 6E).
Furthermore, exosomes from MGC-803R cells
transfected with INC or inhibitor were isolated. RT-qPCR analysis
revealed that miR-155-5p levels were significantly reduced in
exosomes derived from inhibitor-transfected MGC-803R cells compared
with the exosomes secreted by INC-transfected MGC-803R cells
(Fig. 6F). To analyze whether
miR-155-5p suppression influenced MGC-803R-exosomes spreading the
malignant phenotypes to sensitive cells, MGC-803S cells were
incubated with inhibitor-exosomes or INC-exosomes. The results
revealed that INC-exosomes treatment induced the EMT phenotype with
markedly reduced levels of E-cadherin and increased levels of
Vimentin, and significantly increased the paclitaxel resistance of
MGC-803S cells compared with the untreated control group, while
inhibitor-exosomes exhibited no such effect (Fig. 6G-I).
Discussion
Paclitaxel has been reported to possess notable
antitumor activity; the development of chemoresistance is commonly
observed in patients with gastric cancer (2,3). At
present, paclitaxel resistance in tumors has been reported to be
associated with underlying mechanisms, including increased drug
efflux, alteration of intercellular signaling, tubulin mutation and
over-expression of β-tubulin isotype composition (32,33).
Recently, it has been demonstrated that drug-resistant cancer cells
could transmit chemoresistant phenotypes to chemosensitive cancer
cells via exosomes (8). Whether
the acquisition of paclitaxel resistance by gastric cancer cells
occurs in this way remains unknown. In the present study, the
paclitaxel-resistant gastric cancer cell line MGC-803R was
generated; it was proposed that MGC-803R cells could confer EMT
characteristics and a paclitaxel-resistant phenotype on parental
cells by exosomal transfer of miR-155-5p.
Increasing evidence suggests that tumor cell-derived
exosomes provide critical signals in the development of
chemoresistance (8-10). Several studies have revealed that
delivery of key molecules by exosomes to tumor cells could lead to
chemoresistance (34,35). Colon cancer cell exosomes highly
enriched in N-terminal truncated isoforms of p73 mRNA were able to
confer drug resistance on recipient cells (34). Exosomes from glioblastoma cancer
cells harboring a protein tyrosine phosphatase receptor type Z1-MET
fusion conferred temozolomide resistance on parental cells
(35). It has been demonstrated
that chemotherapeutic agents can also alter exosomal cargo
components of cancer cells (12,14,19).
Kreger et al (14) reported
that paclitaxel treatment stimulated the secretion of specific
exosomes from breast cancer cells, which were highly enriched with
survivin protein. Bandari et al (12) observed that chemotherapy notably
promoted exosome secretion in myeloma and resulted in a distinct
exosomal proteome profile. miRNA microarray analysis revealed that
a total of 11 miRNAs were upregulated in cisplatin (DDP)-resistant
A549 cells and in A549/DDP-exosomes compared with A549 cells and
their exosomes (19). These tumor
cell-exosomes could be taken up by tumor cells, altering their
behavior in ways that enhanced tumor survival and progression
(19). Additionally,
chemotherapeutic agents also enhanced exosome release from cancer
cells and were also exported into exosomes (36). This finding suggests that cancer
cells may protect themselves from the cytotoxicity of therapeutic
drugs by secluding them in exosomes.
To improve understanding of the underlying
mechanisms of chemoresistance, chemoresistant cancer cells may be
an ideal cell model for investigation. The role of exosomes
secreted from chemoresistant cancer cells in the induction of
chemoresistance has been studied. Adriamycin (ADM/ADR)-resistant
breast cancer cells (MCF7/ADM) exhibited increased
expression levels of drug-resistance-associated proteins,
including ubiquitin carboxyl-terminal hydrolase-L1 and
P-glycoprotein (P-gp) (13). These
proteins could be sorted into MCF7/ADM cell-derived exosomes, which
transferred the chemoresistant phenotype into ADM-sensitive breast
cancer cells (13). ADR-resistant
breast cancer cells (MCF-7/ADR)-derived exosomes were reported to
contain the drug-resistance-associated gene multidrug resistance-1
and P-gp. MCF-7/ADR cell-derived exosomes induced a drug resistance
phenotype in MCF-7 parental cells (37). These findings demonstrated that
exosomes could transfer intercellular drug resistance from
drug-resistant to drug-sensitive cancer cells. To investigate the
mechanism of paclitaxel resistance in gastric cancer cells, the
paclitaxel-resistant gastric cancer cell line MGC-803R was
established in the present study. Consistently, it was demonstrated
that MGC-803R derived-exosomes conferred a paclitaxel-resistant
phenotype in MGC-803S cells.
It has been observed that exosomal miRNAs shuttled
from drug-resistant to drug-sensitive tumor cells were widely
involved in the spread of chemoresistance (38,39).
Exosomal miR-222-3p from the gemcitabine-resistant lung cancer line
A549 enhanced gemcitabine resistance in parental sensitive cells
(20). A549/DDP-exosomes conferred
resistance to DDP in recipient cells depending on exosomal
miR-100-5p (19). Thus, it was
noted that miRNAs contained in chemoresistant cancer cell-derived
exosomes may determine the degree of chemoresistance acquired
mediated by exosomes between cancer cells. Identifying the specific
miRNA cargo of exosomes may clarify their function and mechanism in
the transfer of chemoresistance. Among these exosomal miRNAs,
miR-155-5p has been reported to mediate transmission of a
chemoresistant phenotype in breast cancer and pancreatic cancer
(23-25). Notably, exosomal miR-155-5p levels
were notably increased in paclitaxel-resistant breast cancer
cell-derived exosomes (25).
Exosomal delivery of miR-155-5p altered the response of sensitive
cancer cells to treatment with chemotherapeutic agents (25). miR-155-5p as an oncogenic miRNAs is
significantly elevated in gastric cancer and drives therapy
resistance in numerous types of tumor (40-42).
Additionally, anti-miR-155-5p increased the chemosensitivity of
cancer cells to paclitaxel (43).
Therefore, the present study determined whether miR-155-5p was
involved in inducing the paclitaxel-resistant phenotype between
gastric cancer cells. As expected, miR-155-5p was highly expressed
in MGC-803R cells and was enriched in their exosomes compared with
MGC-803S cells and their corresponding exosomes. MGC-803R
cell-exosomes were effectively internalized by MGC-803S cells,
which resulted in increased levels of miR-155-5p in the recipient
cells. This suggested that miR-155-5p could be delivered into
MGC-803S cells via exosomes. Increased miR-155-5p expression by
miRNA mimics resulted in similar effects to those of MGC-803R
cell-exosomes on MGC-803S cells. Furthermore, overexpression of
miR-155-5p increased the content of miR-155-5p in MGC-803S cell
exosomes, which acquired the capacity to reduce paclitaxel
chemosensitivity in the parental cells. The results of the present
study indicated that the paclitaxel-resistant phenotype may be
transmitted from paclitaxel-resistant gastric cancer cells to
sensitive cells via exosomal miR-155-5p.
EMT is characterized by loss of epithelial
characteristics and the acquisition of a mesenchymal phenotype.
Acquisition of an EMT phenotype has been demonstrated in numerous
types of chemoresistant cancer cells (25,44,45).
Consistent with previous findings, the present study reported
morphological alterations in accordance with the characteristics of
EMT in MGC-803R cells compared with the parental cells. EMT has
been demonstrated to be associated with enhanced migration and
invasion of cancer cells and has also been proposed as an important
contributor to drug resistance (46). Several studies revealed that the
acquisition of EMT was important for chemoresistance, but not
indispensable for metastasis (47,48).
Cancer cells undergoing EMT have been considered to exhibit
increased resistance to chemotherapeutic agents (49). Therefore, the detection of the EMT
phenotype may aid the determination of alterations in the response
of cancer cells to chemotherapeutic agents. In the present study,
MGC-803R exosomes induced EMT in MGC-803S cells. Overexpression of
miR-155-5p and exosomal miR-155-5p also promoted EMT in MGC-803S
cells. EMT induction in parental cells was an indicator of
increased resistance to paclitaxel. miR-155-5p has been validated
as an important regulator of EMT induction in cancer cells
(50), which suggests that
exosomal transfer of miR-155-5p conferred the EMT phenotype and
increased paclitaxel resistance in MGC-803S cells. The mechanisms
by which EMT induces chemoresistance are complex. Evidence has
indicated that the contribution of EMT to chemoresis-tance may be
associated with the development of cancer stem cells (51,52).
miR-155-5p has been reported to target the regulation of TP53INP1
to promote cancer cell EMT and a cancer stem cell phenotype
(50). In addition, morin
increased the chemosensitivity of prostate cancer cells to
paclitaxel by suppressing the expression of miR-155-5p and
upregulating its target GATA3 (43). To determine the mechanism of
exosomal miR-155-5p in the present study, the two aforementioned
proteins were selected as potential targets for investigation. We
reported that MGC-803R-exosomes and exosomes secreted by MGC-803S
cells following transfection with miR-155-5p mimics suppressed
GATA3 and TP53INP1 expression by directly targeting their 3′UTRs.
Additionally, knockdown of miR-155-5p reversed EMT and the
paclitaxel-resistant phenotype, which was accompanied with elevated
mRNA expression levels of GATA3 and TP53INP1. Furthermore, compared
with the sensitive cells, the mRNA expression levels of the two
targets were significantly reduced in MGC-803R cells. These data
suggested that miR-155-5p loaded in exosomes was functional.
MGC-803R-derived miR-155-5p in exosomes may therefore suppress the
expression of GATA3 and TP53INP1 to confer EMT and a chemoresistant
phenotype on MGC-803S cells. Whether the two targets are
indispensable for exosomal miR-155-5p-mediated transfer of
malignant phenotypes requires further investigation in the future.
In addition, EMT mechanisms are very complex. A hallmark of EMT is
the functional loss of E-cadherin and the overexpression of
mesenchymal markers, including N-cadherin, Vimentin and Fibronectin
(53). In the present study,
alterations in the expression of two classical markers, E-cadherin
and Vimentin, were detected to determine whether gastric cancer
cells acquire an EMT phenotype, which may be not adequate;
EMT-associated transcription factors and signaling pathways require
further investigation.
In conclusion, the present study successfully
generated the paclitaxel-resistant gastric cancer cell line
MGC-803R and demonstrated that miR-155-5p expression levels were
significantly elevated in MGC-803R cells, which was selectively
sorted into MGC-803R cell-derived exosomes. Exosomal delivery of
miR-155-5p may induce EMT and chemoresistance phenotypes from
paclitaxel-resistant gastric cancer cells to sensitive cancer cells
by suppressing the expression of GATA3 and TP53INP1. Therefore,
targeting miR-155-5p may be a novel strategy to overcome paclitaxel
resistance in gastric cancer.
Acknowledgments
Not applicable.
Funding
The present study was supported by the Scientific
research project of ‘six one project’ for high-level health
personnel from Jiangsu Provincial Health and Family Planning
Commission (grant no. LGY 2016026); The project of the Nanjing
Science and Technology Commission (grant no. 201605005); Key and
general program of Jiangsu Cancer Hospital (grant no. ZK201604);
Project funded by China Postdoctoral Science Foundation (grant no.
2017M611740); Jiangsu province postdoctoral research funding scheme
(grant no. 1701027A); Jiangsu province social development key
research and development plan (grant no. BE2017694), and Science
Foundation for Doctorate Research of the Affiliated Hospital of
Jiangsu University (grant no. jdfyRC2016001).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors’ contributions
MW and BS made substantial contributions to the
design of the study and wrote the paper. MW, RQ and SY performed
the experiments. XX, GL and RG analyzed and interpreted the data.
CT, WZ and BS revised the manuscript for critically important
intellectual content and edited the manuscript. All authors read
and approved the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Guo Z, Wang X, Lin R, Chen L, Fan N, Chen
Y, Lin J and Yu J: Paclitaxel-based regimens as first-line
treatment in advanced gastric cancer. J Chemother. 27:94–98. 2015.
View Article : Google Scholar
|
|
3
|
Zhang D and Fan D: Multidrug resistance in
gastric cancer: Recent research advances and ongoing therapeutic
challenges. Expert Rev Anticancer Ther. 7:1369–1378. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhang J, Li S, Li L, Li M, Guo C, Yao J
and Mi S: Exosome and exosomal microRNA: Trafficking, sorting, and
function. Genomics Proteomics Bioinformatics. 13:17–24. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Meehan K and Vella LJ: The contribution of
tumour-derived exosomes to the hallmarks of cancer. Crit Rev Clin
Lab Sci. 53:121–131. 2016. View Article : Google Scholar
|
|
6
|
Ruivo CF, Adem B, Silva M and Melo SA: The
biology of cancer exosomes: Insights and new perspectives. Cancer
Res. 77:6480–6488. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Brinton LT, Sloane HS, Kester M and Kelly
KA: Formation and role of exosomes in cancer. Cell Mol Life Sci.
72:659–671. 2015. View Article : Google Scholar
|
|
8
|
Sharma A: Chemoresistance in cancer cells:
Exosomes as potential regulators of therapeutic tumor
heterogeneity. Nanomedicine (Lond). 12:2137–2148. 2017. View Article : Google Scholar
|
|
9
|
Butera G, Pacchiana R and Donadelli M:
Autocrine mechanisms of cancer chemoresistance. Semin Cell Dev
Biol. 78:3–12. 2018. View Article : Google Scholar
|
|
10
|
Sousa D, Lima RT and Vasconcelos MH:
Intercellular transfer of cancer drug resistance traits by
extracellular vesicles. Trends Mol Med. 21:595–608. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Colombo M, Raposo G and Théry C:
Biogenesis, secretion, and intercellular interactions of exosomes
and other extracellular vesicles. Annu Rev Cell Dev Biol.
30:255–289. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bandari SK, Purushothaman A, Ramani VC,
Brinkley GJ, Chandrashekar DS, Varambally S, Mobley JA, Zhang Y,
Brown EE, Vlodavsky I, et al: Chemotherapy induces secretion of
exosomes loaded with heparanase that degrades extracellular matrix
and impacts tumor and host cell behavior. Matrix Biol. 65:104–118.
2018. View Article : Google Scholar
|
|
13
|
Ning K, Wang T, Sun X, Zhang P, Chen Y,
Jin J and Hua D: UCH-L1-containing exosomes mediate
chemotherapeutic resistance transfer in breast cancer. J Surg
Oncol. 115:932–940. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kreger BT, Johansen ER, Cerione RA and
Antonyak MA: The enrichment of survivin in exosomes from breast
cancer cells treated with paclitaxel promotes cell survival and
chemoresistance Cancers (Basel). 8. pp. 82016
|
|
15
|
Liu X, Fu Q, Du Y, Yang Y and Cho WC:
MicroRNA as regulators of cancer stem cells and chemoresistance in
colorectal cancer. Curr Cancer Drug Targets. 16:738–754. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cui SY, Wang R and Chen LB: MicroRNAs: Key
players of taxane resistance and their therapeutic potential in
human cancers. J Cell Mol Med. 17:1207–1217. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhong S, Chen X, Wang D, Zhang X, Shen H,
Yang S, Lv M, Tang J and Zhao J: MicroRNA expression profiles of
drug-resistance breast cancer cells and their exosomes. Oncotarget.
7:19601–19609. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Qin X, Yu S, Xu X, Shen B and Feng J:
Comparative analysis of microRNA expression profiles between A549,
A549/DDP and their respective exosomes. Oncotarget. 8:42125–42135.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Qin X, Yu S, Zhou L, Shi M, Hu Y, Xu X,
Shen B, Liu S, Yan D and Feng J: Cisplatin-resistant lung cancer
cell-derived exosomes increase cisplatin resistance of recipient
cells in exosomal miR-100-5p-dependent manner. Int J Nanomedicine.
12:3721–3733. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wei F, Ma C, Zhou T, Dong X, Luo Q, Geng
L, Ding L, Zhang Y, Zhang L, Li N, et al: Exosomes derived from
gemcitabine-resistant cells transfer malignant phenotypic traits
via delivery of miRNA-222-3p. Mol Cancer. 16:1322017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen WX, Liu XM, Lv MM, Chen L, Zhao JH,
Zhong SL, Ji MH, Hu Q, Luo Z, Wu JZ, et al: Exosomes from
drug-resistant breast cancer cells transmit chemoresistance by a
horizontal transfer of microRNAs. PLoS One. 9:e952402014.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Challagundla KB, Wise PM, Neviani P, Chava
H, Murtadha M, Xu T, Kennedy R, Ivan C, Zhang X, Vannini I, et al:
Exosome-mediated transfer of microRNAs within the tumor
microenvironment and neuroblastoma resistance to chemotherapy. J
Natl Cancer Inst. 107:1072015. View Article : Google Scholar
|
|
23
|
Patel GK, Khan MA, Bhardwaj A, Srivastava
SK, Zubair H, Patton MC, Singh S, Khushman M and Singh AP: Exosomes
confer chemoresistance to pancreatic cancer cells by promoting ROS
detoxification and miR-155-mediated suppression of key
gemcitabine-metabolising enzyme, DCK. Br J Cancer. 116:609–619.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mikamori M, Yamada D, Eguchi H, Hasegawa
S, Kishimoto T, Tomimaru Y, Asaoka T, Noda T, Wada H, Kawamoto K,
et al: MicroRNA-155 controls exosome synthesis and promotes
gemcitabine resistance in pancreatic ductal adenocarcinoma. Sci
Rep. 7:423392017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Santos JC, Lima NDS, Sarian LO, Matheu A,
Ribeiro ML and Derchain SFM: Exosome-mediated breast cancer
chemoresistance via miR-155 transfer. Sci Rep. 8:8292018.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang M, Zhao C, Shi H, Zhang B, Zhang L,
Zhang X, Wang S, Wu X, Yang T, Huang F, et al: Deregulated
microRNAs in gastric cancer tissue-derived mesenchymal stem cells:
Novel biomarkers and a mechanism for gastric cancer. Br J Cancer.
110:1199–1210. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
28
|
Zhu M, Wang M, Yang F, Tian Y, Cai J, Yang
H, Fu H, Mao F, Zhu W, Qian H, et al: miR-155-5p inhibition
promotes the transition of bone marrow mesenchymal stem cells to
gastric cancer tissue derived MSC-like cells via NF-κB p65
activation. Oncotarget. 7:16567–16580. 2016.PubMed/NCBI
|
|
29
|
Whitehead B, Wu L, Hvam ML, Aslan H, Dong
M, Dyrskjøt L, Ostenfeld MS, Moghimi SM and Howard KA: Tumour
exosomes display differential mechanical and complement activation
properties dependent on malignant state: Implications in
endothelial leakiness. J Extracell Vesicles. 4:296852015.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bockhorn J, Dalton R, Nwachukwu C, Huang
S, Prat A, Yee K, Chang YF, Huo D, Wen Y, Swanson KE, et al:
MicroRNA-30c inhibits human breast tumour chemotherapy resistance
by regulating TWF1 and IL-11. Nat Commun. 4:13932013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yu SJ, Yang L, Hong Q, Kuang XY, Di GH and
Shao ZM: MicroRNA-200a confers chemoresistance by antagonizing
TP53INP1 and YAP1 in human breast cancer. BMC Cancer. 18:742018.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Vergara D, Tinelli A, Iannone A and Maffia
M: The impact of proteomics in the understanding of the molecular
basis of Paclitaxel-resistance in ovarian tumors. Curr Cancer Drug
Targets. 12:987–997. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kavallaris M: Microtubules and resistance
to tubulin-binding agents. Nat Rev Cancer. 10:194–204. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Soldevilla B, Rodríguez M, San Millán C,
García V, Fernández-Periañez R, Gil-Calderón B, Martín P,
García-Grande A, Silva J, Bonilla F, et al: Tumor-derived exosomes
are enriched in ∆Np73, which promotes oncogenic potential in
acceptor cells and correlates with patient survival. Hum Mol Genet.
23:467–478. 2014. View Article : Google Scholar
|
|
35
|
Zeng AL, Yan W, Liu YW, Wang Z, Hu Q, Nie
E, Zhou X, Li R, Wang XF, Jiang T, et al: Tumour exosomes from
cells harbouring PTPRZ1-MET fusion contribute to a malignant
phenotype and temozolomide chemoresistance in glioblastoma.
Oncogene. 36:5369–5381. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang J, Yeung BZ, Cui M, Peer CJ, Lu Z,
Figg WD, Guillaume Wientjes M, Woo S and Au JL: Exosome is a
mechanism of inter-cellular drug transfer: Application of
quantitative pharmacology. J Control Release. 268:147–158. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang X, Xu C, Hua Y, Sun L, Cheng K, Jia
Z, Han Y, Dong J, Cui Y and Yang Z: Exosomes play an important role
in the process of psoralen reverse multidrug resistance of breast
cancer. J Exp Clin Cancer Res. 35:1862016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Bach DH, Hong JY, Park HJ and Lee SK: The
role of exosomes and miRNAs in drug-resistance of cancer cells. Int
J Cancer. 141:220–230. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhao L, Liu W, Xiao J and Cao B: The role
of exosomes and ‘exosomal shuttle microRNA’ in tumorigenesis and
drug resistance. Cancer Lett. 356B:339–346. 2015. View Article : Google Scholar
|
|
40
|
Qu Y, Zhang H, Sun W, Han Y, Li S, Qu Y,
Ying G and Ba Y: MicroRNA-155 promotes gastric cancer growth and
invasion by negatively regulating transforming growth factor-β
receptor 2. Cancer Sci. 109:618–628. 2018. View Article : Google Scholar :
|
|
41
|
Bayraktar R and Van Roosbroeck K: miR-155
in cancer drug resistance and as target for miRNA-based
therapeutics. Cancer Metastasis Rev. 37:33–44. 2018. View Article : Google Scholar
|
|
42
|
Van Roosbroeck K, Fanini F, Setoyama T,
Ivan C, Rodriguez-Aguayo C, Fuentes-Mattei E, Xiao L, Vannini I,
Redis RS, D’Abundo L, et al: Combining anti-mir-155 with
chemotherapy for the treatment of lung cancers. Clin Cancer Res.
23:2891–2904. 2017. View Article : Google Scholar
|
|
43
|
Li B, Jin X, Meng H, Hu B, Zhang T, Yu J,
Chen S, Guo X, Wang W, Jiang W, et al: Morin promotes prostate
cancer cells chemosensitivity to paclitaxel through miR-155/GATA3
axis. Oncotarget. 8:47849–47860. 2017.PubMed/NCBI
|
|
44
|
Chen D, Lin X, Zhang C, Liu Z, Chen Z, Li
Z, Wang J, Li B, Hu Y, Dong B, et al: Dual PI3K/mTOR inhibitor
BEZ235 as a promising therapeutic strategy against
paclitaxel-resistant gastric cancer via targeting PI3K/Akt/mTOR
pathway. Cell Death Dis. 9:1232018. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wang Z, Li Y, Kong D, Banerjee S, Ahmad A,
Azmi AS, Ali S, Abbruzzese JL, Gallick GE and Sarkar FH:
Acquisition of epithelial-mesenchymal transition phenotype of
gemcitabine-resistant pancreatic cancer cells is linked with
activation of the notch signaling pathway. Cancer Res.
69:2400–2407. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Iwatsuki M, Mimori K, Yokobori T, Ishi H,
Beppu T, Nakamori S, Baba H and Mori M: Epithelial-mesenchymal
transition in cancer development and its clinical significance.
Cancer Sci. 101:293–299. 2010. View Article : Google Scholar
|
|
47
|
Fischer KR, Durrans A, Lee S, Sheng J, Li
F, Wong ST, Choi H, El Rayes T, Ryu S, Troeger J, et al:
Epithelial-to-mesenchymal transition is not required for lung
metastasis but contributes to chemoresistance. Nature. 527:472–476.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Zheng X, Carstens JL, Kim J, Scheible M,
Kaye J, Sugimoto H, Wu CC, LeBleu VS and Kalluri R:
Epithelial-to-mesenchymal transition is dispensable for metastasis
but induces chemoresistance in pancreatic cancer. Nature.
527:525–530. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Du B and Shim JS: Targeting
epithelial-mesenchymal transition (EMT) to overcome drug resistance
in cancer. Molecules. 21:212016. View Article : Google Scholar
|
|
50
|
Liu F, Kong X, Lv L and Gao J: TGF-β1 acts
through miR-155 to down-regulate TP53INP1 in promoting
epithelial-mesenchymal transition and cancer stem cell phenotypes.
Cancer Lett. 359:288–298. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan
A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M, et al: The
epithelial-mesenchymal transition generates cells with properties
of stem cells. Cell. 133:704–715. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Jiang ZS, Sun YZ, Wang SM and Ruan JS:
Epithelial-mesenchymal transition: Potential regulator of ABC
transporters in tumor progression. J Cancer. 8:2319–2327. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Zeisberg M and Neilson EG: Biomarkers for
epithelial-mesenchymal transitions. J Clin Invest. 119:1429–1437.
2009. View Article : Google Scholar : PubMed/NCBI
|