Introduction
Breast cancer is the most common female malignancy
and the second most common cause of cancer-associated fatality in
the world (1). Approximately 70%
of patients with breast cancer are estrogen receptor-positive
(ER+) (2). Apart from
surgery, endocrine therapy (including tamoxifen, fulvestrant and
letrozole) has improved the overall survival and quality of life
for patients with breast cancer (2-4).
Among all endocrine therapies, tamoxifen is the most extensively
used hormone therapy and functions as an estrogen antagonist in
breast cancer (5,6). Although the majority of patients with
ER+ breast cancer benefit from tamoxifen therapy, many tumors
eventually recur because of tamoxifen resistance (7,8).
Tamoxifen resistance can arise via several mechanisms, including
loss of ERα, induction of abnormal estra-diol levels and
alterations of coregulatory proteins, including amplified in breast
cancer 1 and histone deacetylase (9-11).
An increasing number of long non-coding (lnc)RNAs in
the human genome have been identified, and have provided new
directions in cancer research (12). lncRNA, a class of non-protein
coding transcripts with >200 nucleotides, regulates
protein-coding genes during transcription and post-transcription in
a sequence-specific manner (13-15).
Importantly, lncRNAs in cancer cells are associated with the
formation of tamoxifen resistance (16-18).
However, only a few lncRNAs have been proposed to be clinically
relevant biomarkers for tamoxifen resistance, such as H19 and
homeobox antisense intergenic RNA (19-21).
Searching for appropriate lncRNAs is valuable for the management of
tamoxifen-resistance.
Out of the numerous cancer-associated lncRNAs,
lncRNA urothelial carcinoma-associated 1 (UCA1) serves an important
oncogenic role in several cancer types, including bladder cancer,
colorectal cancer and gastric cancer (22). UCA1 has three exons that encode a
1.4-kb isoform and a 2.2-kb isoform (23). It was originally identified as a
urine marker (the 1.4-kb isoform) in bladder cancer (24). Tuo et al (25) demonstrated that UCA1 can modulate
breast cancer cell growth and apoptosis through downregulation of
the tumor suppressor microRNA (miR)-143. Huang et al
(23) reported that UCA1 can
promote breast tumor growth by suppressing the level of p27. UCA1
is also associated with the poor prognosis of cancer. Bian et
al (26) demonstrated that
patients with colorectal cancer and higher UCA1 expression had a
significantly poorer prognosis. Furthermore, it was reported that
UCA1 expression was correlated with a reduction in recurrence-free
survival in breast cancer (27).
These findings highlight the important role of UCA1 in cancer
development.
The polycomb group protein enhancer of zeste homolog
2 (EZH2) is a critical regulator of tumorigenesis (28,29).
It has been demonstrated that the level of EZH2 is elevated in
human bladder cancer, breast cancer, colon cancer and prostate
cancer (30). Furthermore, the
expression and mutation of EZH2 can regulate the level of H3K27me3
(31). In hepatocellular
carcinoma, UCA1 repressed p27 expression through its association
with EZH2, which suppresses p27Kip1 through H3K27me3 on the p27Kip1
promoter (32). However, the
effects of UCA1 on EZH2 expression and the underlying molecular
mechanisms in breast cancer are not fully understood.
The phosphoinositide 3-kinase (PI3K)/protein kinase
B (AKT) signaling pathway is the most frequently altered pathway in
human cancer, and previous studies have demonstrated that UCA1
regulates the cell cycle progression of bladder carcinoma cells via
PI3K/AKT-dependent signaling (33,34).
Notably, the knockdown of UCA1 inhibits AKT phosphorylation in
breast cancer cells (35).
Additionally, activation of the PI3K/AKT signaling pathway has been
demonstrated to confer resistance to antiestrogens in
tamoxifen-resistant breast cancer cells (36). Therefore, it would be useful to
determine whether UCA1 is involved in the PI3K/AKT signaling
pathway and if it induces tamoxifen resistance in breast cancer
cells.
In the present study, the level of UCA1 expression
was investigated in tamoxifen-resistant cells and compared with
tamoxifen-sensitive cells. Induction of UCA1 overexpression in
MCF-7 and T47D breast cancer cells and silencing of UCA1 in LLC2
and LLC9 breast cancer cells was performed to assess the drug
sensitivity of the cells to tamoxifen. Furthermore, it was explored
whether UCA1 was physically associated with EZH2. In addition, it
was investigated whether UCA1 regulates tamoxifen resistance
through a EZH2/p21 axis and the PI3K/AKT signaling pathway in
breast cancer.
Materials and methods
Patients and specimens
A total of 10 hormone receptor-positive breast
cancer specimens and 10 non-tumor specimens were randomly selected
from the First Hospital of Jilin University (Changchun, China)
between April 2015 and April 2017. All these participants were
female. The breast cancer specimens were histologically diagnosed
as breast carcinoma using ultrasound-guided core needle biopsy of
the breast. In the 10 breast cancer specimens, 1 was at stage I, 5
were at stage II and 4 were at stage III. The age range of the 10
patients was from 37-68 years old, with a median age of 51.
Evidence of bilateral disease and pregnancy concomitant with the
diagnosis of breast cancer resulted in exclusion from the study.
All samples were collected prior to tamoxifen therapy and stored in
liquid nitrogen (−196°C) until use. Permission to use the clinical
samples for research purposes was obtained and approved by the
Ethics Committee of the First Hospital of Jilin University.
Informed consents were obtained from all patients.
Cell culture
Human breast cancer cell lines MCF-7
(tamoxifen-sensitive), T47D (tamoxifen-sensitive), LCC2
(tamoxifen-resistant) and LCC9 (tamoxifen-resistant), were
purchased from American Type Culture Collection (Manassas, VA,
USA). All cancer cells were maintained in Dulbecco's modified
Eagle's medium (DMEM, Gibco; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS,
HyClone, Logan, UT, USA) 2 mM glutamine (Gibco; Thermo Fisher
Scientific, Inc.), 100 U/ml penicillin and 100 µg/ml
streptomycin (HyClone). Cells were cultured at 37°C in an incubator
with a humidified atmosphere containing 5% CO2.
Overexpression and knockdown of UCA1 in
breast cancer cells
To induce the overexpression of UCA1 in breast
cancer cells, the cDNA encoding UCA1 was polymerase chain reaction
(PCR)-amplified. The primer sequences were as follows: UCA1,
forward 5′-CGCGGATCCTTTATCAGGCATATTAG CTTTAA-3′ (BamHI) and
reverse 5′-GCGAATTCTGACATTC TTCTGGACAATG-3′ (EcoRI).
Following this, the PCR product was subcloned into the pGreen.puro
lentivirus vector (SBI, Palo Alto, CA, USA) with BamHI and
EcoRI restriction sites (Takara Biotechnology Co. Ltd.,
Dalian, China). Viral particles were harvested at 48 h
post-cotransfection of the pGreen-UCA1-puro constructs with the
packaging plasmid ps-PAX2 and the envelope plasmid pMD2G (SBI) into
293T cells using Lipofectamine 3000 (Invitrogen; Thermo Fisher
Scientific, Inc.). The empty vector was used as the control
(lv-NC). MCF-7 and T47D cells were infected with the lentiviral
particles (5×107 TU/ml; lv-UCA1 or lv-NC) plus 6
µg/ml polybrene (Sigma-Aldrich; Merck KGaA, Darmstadt,
Germany). The virus titers in the control and experimental groups
were nearly the same as above (~5×107 TU/ml).
For the knockdown of UCA1, the small interfering
(si)RNA targeting UCA1 (si-UCA1) and the scramble non-target
control siRNA (si-NC) were synthesized by Shanghai GenePharma Co.,
Ltd., (Shanghai, China). si-UCA1 and si-NC sequences were as
follows (37): si-UCA1,
5′-GTTAATCCAGGAGACAA AGA-3′; and si-NC, 5′-TTCTCCGAACGTGTCACGT-3′.
LCC2 and LCC9 cells were transfected with equal amounts (100 nM) of
si-UCA1 and si-NC using Lipofectamine 3000. All the following
cellular or molecular experiments were carried out at 48 h
post-transfection (38,39).
RNA extraction, reverse transcription-PCR
(RT-PCR) and RT-quantitative PCR (RT-qPCR)
Total RNA was extracted from breast cancer tissues
and cells using TRIzol reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's instructions.
cDNA was reverse transcribed using 1 µg of total RNA and the
SuperScript III First-Stand Synthesis Kit (Invitrogen; Thermo
Fisher Scientific, Inc.). The expression level of UCA1 were
determined on a PCR thermal cycler (T100, Bio-Rad Laboratories,
Inc., Hercules, CA, USA) using 2X Taq PCR StarMix buffer (GeneStar,
Beijing, China) or on a real-time PCR thermal cycler (ABI PRISM
7500, Applied Biosystems; Thermo Fisher Scientific, Inc.) using
SYBR Green Master Mix (Applied Biosystems; Thermo Fisher
Scientific, Inc.). The endogenous control gene was
glyceraldehyde-3-phosphate dehydrogenase (GAPDH). The RT-PCR
amplification process was as follows: 1 cycle at 98°C for 2 min and
32 cycles at 95°C for 20 sec, 62°C for 15 sec, followed by 72°C for
15 sec; ending with an extension cycle at 72°C for 5 min. The qPCR
amplification process consisted of 1 cycle at 95°C for 10 min,
followed by 40 cycles at 95°C for 10 sec and 58°C for 30 sec. The
results of RT-PCR were visual-ized using a 3% agarose gel and qPCR
was performed using the 2−ΔΔCq method. All the
oligonucleotide primers were synthesized by Takara. The primer
sequences used were as follows (33): UCA1, forward 5′-CTTCTGCATAGGATCTG
CAATCAG-3′ and reverse 5′-TTTTGTCCCCATTTTCCATCA TACG-3′; GAPDH,
forward 5′-AGGTCGGAGTCAACGG ATTTG-3′ and reverse
5′-GTGATGGCATGGACTGTGGT-3′.
WST-1 assay to assess cell viability
Tamoxifen was purchased from Sigma-Aldrich; Merck
KGaA. The stock solution of tamoxifen (500 µM) was prepared
in 100% MeOH and maintained at 4°C. Working standard solutions at
different concentrations were prepared by dilution in DMEM
supplemented with 10% FBS. Solutions were added into the 96-well
plate at a final concentration of 0, 0.01, 0.1, 1, 10 or 100
µM and incubated with cells for 24 h. Following this, 10
µl WST-1 (Roche Diagnostics, Shanghai, China) was added into
each well and the cells were incubated at 37°C in the dark for 2 h.
The absorbances of 450 and 630 nm were monitored. The relative cell
viability percentage in each group was calculated by comparison to
that of the control group.
Flow cytometry for cell cycle and
apoptosis analysis
LCC2 cells transfected with si-UCA1 (si-UCA1 LCC2)
were treated with 10 µM tamoxifen for 24 h, trypsinized,
collected and washed with PBS. For cell cycle analysis, cells were
fixed in pre-cold 70% ethanol for 20 min and stored at -20°C.
Following this, cells were washed with PBS and stained with a
solution containing 3.5 µM Tris-HCl (pH 7.6), 10 mM NaCl, 50
µg/ml propidium iodide (PI) (Sigma-Aldrich; Merck KGaA), 20
µg/ml RNase and 0.1% igepal CA-630 (Sigma-Aldrich; Merck
KGaA) for 20 min on ice to label DNA. Subsequently, LCC2 cells were
analyzed using a FACSCaliber flow cytometer with a FlowJo software
(version 10.0, BD Biosciences, Franklin Lakes, NJ, USA). The
percentages of cells at different phases were calculated from three
independent experiments.
For cell apoptosis analysis, cells were stained
using an Annexin V-FITC/PI double staining apoptosis detection kit
(BD Biosciences, San Jose, CA, USA) according to the manufacturer's
instructions, and analyzed with a FACSCaliber flow cytometer. The
cells in the different portions represented the different cell
states as follows: The late-apoptotic cells were present in the
upper right portion, the viable cells were present in the lower
left portion and the early apoptotic cells were present in the
lower right portion.
RNA immunoprecipitation (RIP) assay
The RIP experiment was performed in LCC2 cells using
the Magna RIP R NA-Binding Protein Immunoprecipitation Kit (EMD
Millipore, Billerica, MA, USA) according to the manufacturer's
instructions. EZH2 antibody for the RIP assay was purchased from
Abcam (1:500; #ab186006; Shanghai, China). Samples were treated
with proteinase K (Thermo Fisher Scientific, Inc.) to digest the
protein for 1 h at 37°C and the immunoprecipitated RNA was
isolated. Final analysis of co-precipitated RNA was performed using
qPCR and demonstrated as fold enrichment of UCA1.
Chromatin immunoprecipitation (ChIP)
assay
The ChIP experiment was performed in LCC2 cells
using the EZ ChIP Chromatin Immunoprecipitation Kit (#17-371, EMD
Millipore) according to the manufacturer's instructions. Briefly,
LCC2 cells were incubated with formaldehyde for 10 min to generate
DNA-protein cross-links; the crosslinked chromatin DNAs were
sonicated into 200 to 1,000-bp-sized fragments. Subsequently,
immunoprecipitation was performed using anti-EZH2 antibody
(1:1,000; #07-689, EMD Millipore) and anti-H3K27me3 antibody
(1:1,000; #17-622, EMD Millipore), or normal IgG (1:200, EZ ChIP
Chromatin Immunoprecipitation Kit) as control. Precipitated
chromatin DNA was recovered and analyzed by qPCR. The primer
sequences of p21 promoter were as follows: Forward (40), 5′-AGACCATGTGGACCTGTCACTG-3′ and
reverse 5′-GTTTGGAGTGGTAGAAATCTGTC-3′.
Western blot analysis
Cell samples were lysed using
radioimmunoprecipitation assay lysis buffer (Beyotime Institute of
Biotechnology, Shanghai, China) containing protease inhibitor. The
total protein concentration was determined using a BCA protein
assay kit (Beyotime Institute of Biotechnology). A total of 20
µg of total protein was loaded per lane and separated by
SDS-PAGE (10 or 12% gels) and transferred to polyvinylidene
fluoride membrane (Roche). The membranes were blocked in 5% skimmed
milk diluted with Tris-buffered saline/Tween-20 (Tris-HCl 20
mmol/l, NaCl 150 mmol/l, 0.1% Tween-20, pH 7.5) at room temperature
for 1 h and subsequently incubated overnight at 4°C with primary
antibodies: Anti-AKT (1:1,000, #ab8805, Abcam), anti-phospho(p)-AKT
(1:2,000, #ab8933, Abcam), CAMP responsive element binding protein
(CREB, 1:2,000, #ab178322, Abcam), anti-p-CREB (1:1,000, #ab10564,
Abcam), anti-GAPDH (1:2,000, #ab181603, AbMart Bio-tech Co. Ltd.,
Shanghai, China), anti-B cell lymphoma/leukemia-2 (Bcl-2, 1:2,000,
#ab196495, Abcam), anti-cleaved caspase-3 (1:1,000, #9661, Cell
Signaling Technology, Inc., Danvers, MA, USA), anti-cleaved
caspase-9 (1:1,000, #52873, Cell Signaling Technology, Inc.),
anti-cyclin D1 (1:1,000, #2978, Cell Signaling Technology, Inc.)
and anti-p21 (1:1,000, #2947, Cell Signaling Technology, Inc.).
Subsequently, the membranes were incubated with anti-mouse
(1:5,000, SAB3701214, Sigma-Aldrich; Merck KGaA) or rabbit
(1:5,000, SAB3700852, Sigma-Aldrich; Merck KGaA) horseradish
peroxidase-conjugated secondary antibodies at 37°C for 1 h. The
immunoreactive bands were visualized using the ECL western blot
substrate (Promega Corporation, Madison, WI, USA) and the relative
band density was analyzed by Quantity-one software (version 4.6,
Bio-Rad Laboratories, Inc.).
Suppression of the PI3K signaling
pathway
The PI3K signaling pathway was suppressed by the
PI3K inhibitor LY294002 (Cell Signaling Technology, Inc.). LCC2
cells were treated with 50 µM LY294002 for 24 h in DMEM
supplemented with 10% FBS. Subsequent qPCR and western blot
analysis were conducted at 24 h post-inhibition.
Statistical analysis
Data were presented as the mean ± standard error of
the mean of at least three independent experiments. Statistical
significance between two groups was determined using one-way
analysis of variance followed by an LSD or Dunnett's post hoc test
or the Student's t-test. P<0.05 was considered to indicate a
statistically significant difference. All analyses were performed
using SPSS 16.0 software (SPSS Inc., Chicago, IL, USA).
Results
UCA1 expression is upregulated in
tamoxifen-resistant breast cancer cells
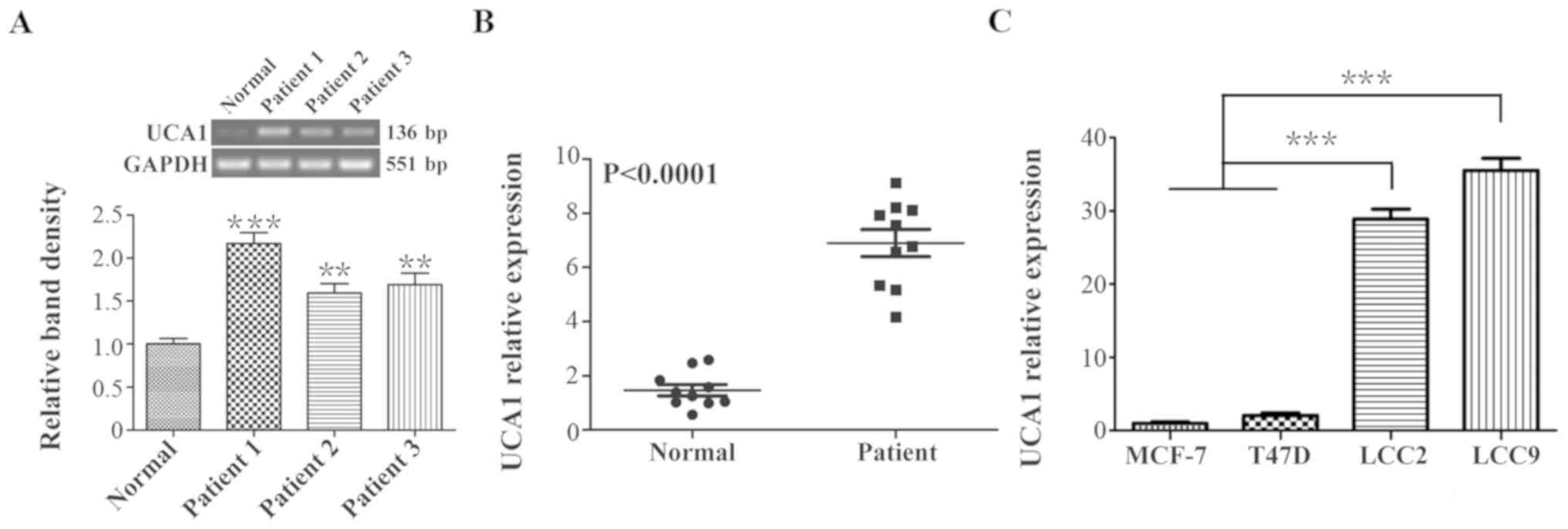
Firstly, 1 normal breast tissue and 3 breast cancer
tissues were randomly selected from the 20 samples, and the level
of UCA1 expression was detected using RT-PCR. The PCR results
revealed that the level of UCA1 expression was significantly
increased in breast cancer tissues compared with normal tissues
(P<0.001 and P<0.01; Fig.
1A). Following this, the UCA1 expression levels in all the 10
normal breast tissues and 10 breast cancer tissues were assessed by
qPCR. As indicated in Fig. 1B, the
mean expression level of UCA1 in the breast cancer group was
4.68-fold greater when compared with that in the normal control
group (P<0.0001). These data indicated a positive association
between breast cancer and the expression of UCA1.
According to these results, the expression levels of
UCA1 in tamoxifen-sensitive cells, MCF-7 and T47D, and in the
tamoxifen-resistant cells, LCC2 and LCC9, were assessed using qPCR
(Fig. 1C). It was revealed that
the level of UCA1 expression in LCC2 and LCC9 cells was >20-fold
greater when compared with that in MCF-7 and T47D cells
(P<0.001), suggesting a positive association between tamoxifen
resistance and UCA1 expression in breast cancer cells.
UCA1 affects the cell viability of breast
cancer cells treated with tamoxifen
In order to further confirm the contribution of UCA1
to tamoxifen resistance, the WST-1 assay was performed to detect
the cell survival rate following UCA1 knockdown or overexpression
in breast cancer cells.
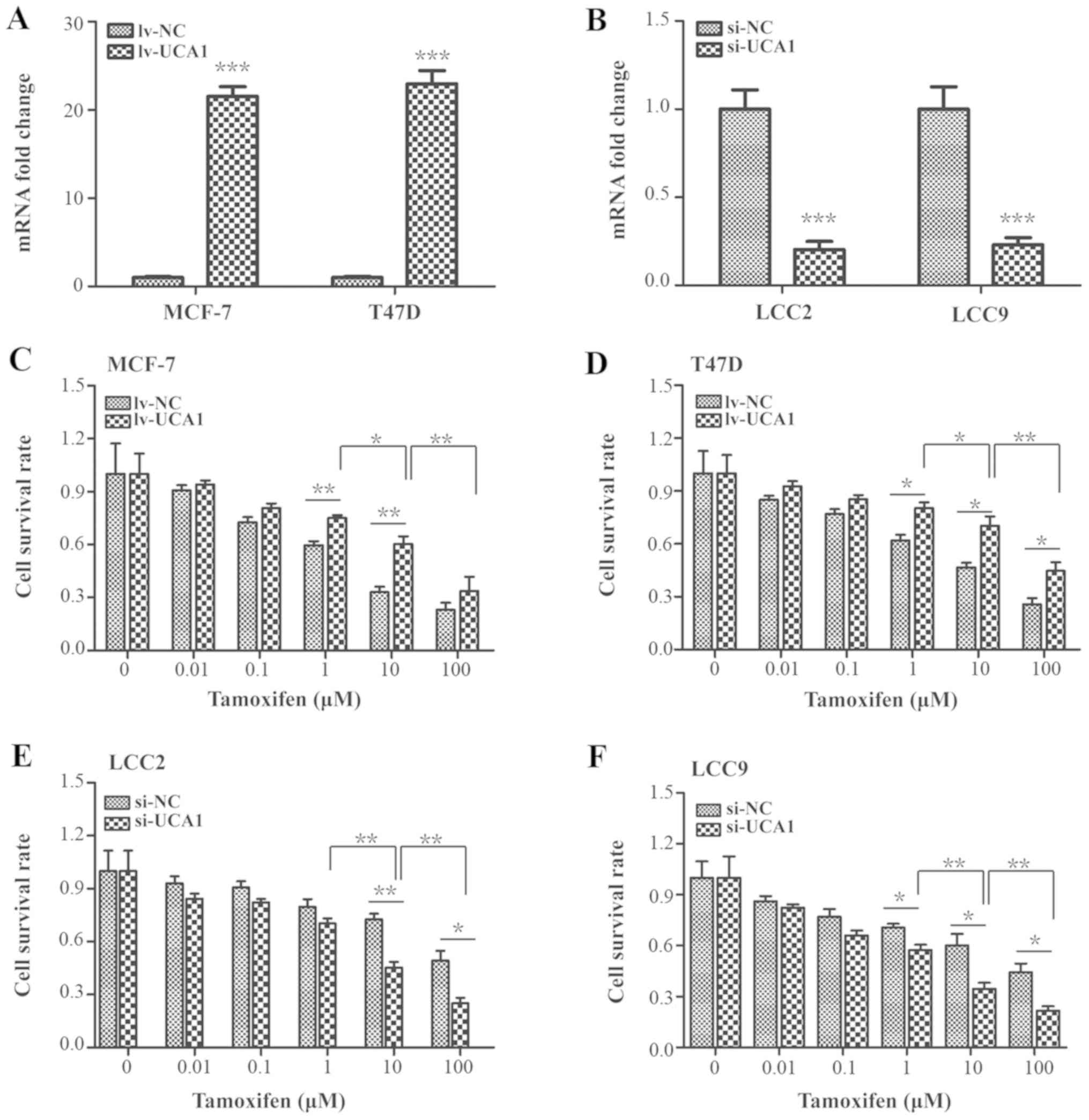
The delivery efficiencies of the lentivirus carrying
UCA1 DNA and the siRNA were assessed. As indicated in Fig. 2A, UCA1 expression was significantly
elevated by 21.67- and 22.97-fold in lentivirus-transduced MCF-7
and T47D cells compared with the lv-NC group, respectively
(P<0.001). Furthermore, UCA1 expression was significantly
downregulated to 0.2-fold and 0.23-fold in the UCA1-siRNA
transfected LCC2 and LCC9 cells when compared with the si-NC group,
respectively (Fig. 2B;
P<0.001).
Following treatment with increasing concentrations
of tamoxifen (0, 0.01, 0.1, 1, 10 and 100 µM), it was
observed that the cell survival rates of UCA1-overexpressed cells
were significantly increased compared with the lv-NC group in the
presence of 1 or 10 µM tamoxifen in MCF-7 cells and in the
presence of 1, 10 or 100 µM tamoxifen in T47D cells
(Fig. 2C and D; P<0.05 and
P<0.01). Conversely, UCA1 silencing significantly decreased the
cell survival rate compared with the si-NC group in the presence of
10 or 100 µM tamoxifen in LCC2 cells or in the presence of
1, 10 or 100 µM tamoxifen in LCC9 cells (Fig. 2E and F; P<0.05 and P<0.01).
Specifically, the cell survival rates significantly changed in the
10 µM tamoxifen treatment group compared with the 1
µM tamoxifen treatment group (Fig. 2C-F; P<0.05 or P<0.01).
However, in the 100 µM tamoxifen treatment group, the cell
survival rates of the control and the experimental group were
significantly decreased compared with the 10 µM tamoxifen
treatment group, indicating that a high concentration of tamoxifen
promoted non-specific cytotoxicity (Fig. 2C-F; P<0.01).
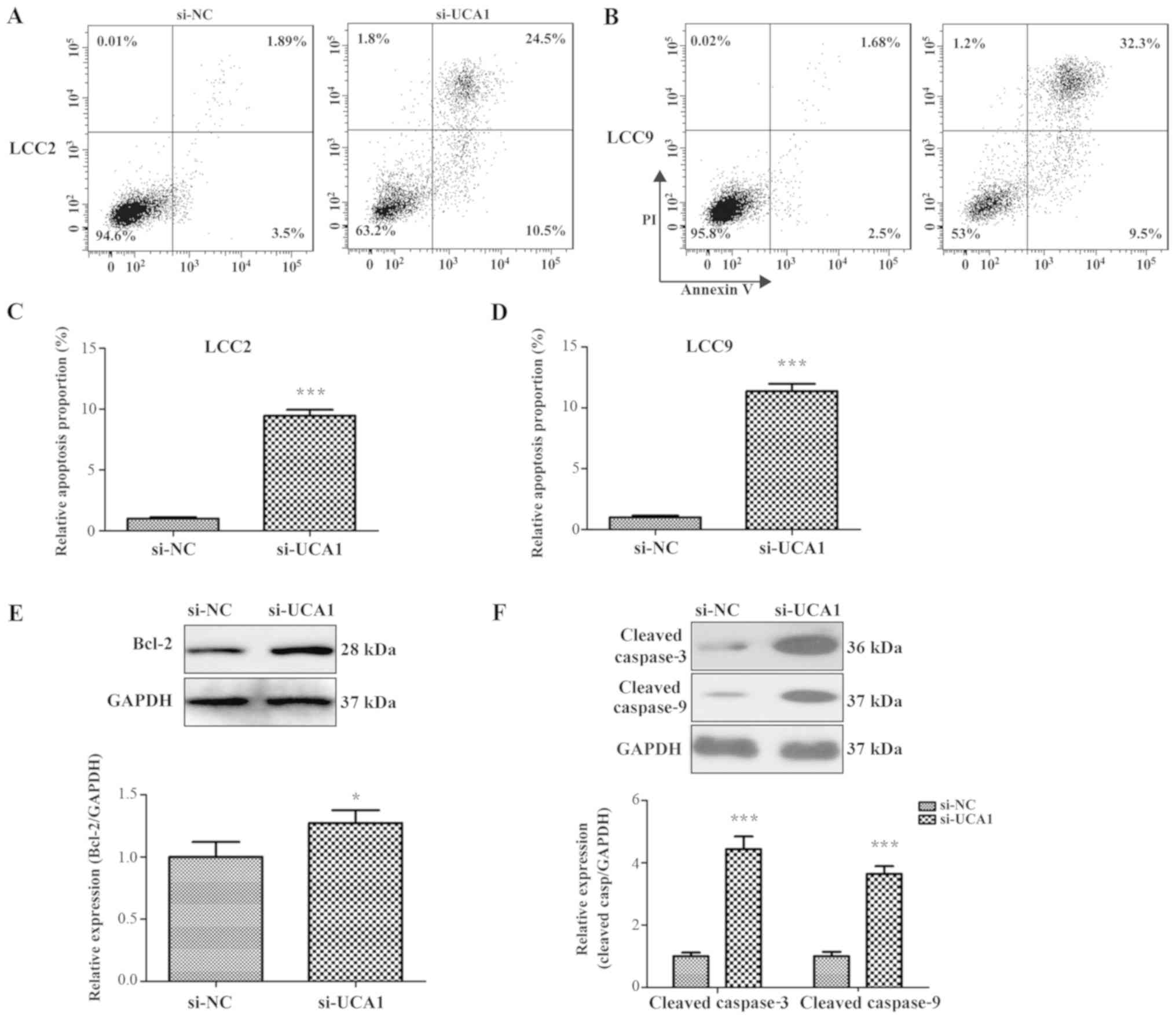
Flow cytometry results indicated that the cell
apoptosis rate of si-UCA1 LCC2 cells (35%) and si-UCA1 LCC9 cells
41.8%) was significantly increased following 10 µM tamoxifen
treatment when compared with the negative control (si-NC, 5.39% and
4.18%; Fig. 3A-D; P<0.001).
Several apoptosis-associated factors were also measured by western
blot analysis. Results indicated that the expression levels of
Bcl-2, cleaved caspase-3 and cleaved caspase-9 were significantly
increased in si-UCA1 LCC2 cells (Fig.
3E and F; P<0.05 and P<0.001). These data suggest that
UCA1 contributed to the tamoxifen resistance in breast cancer
cells.
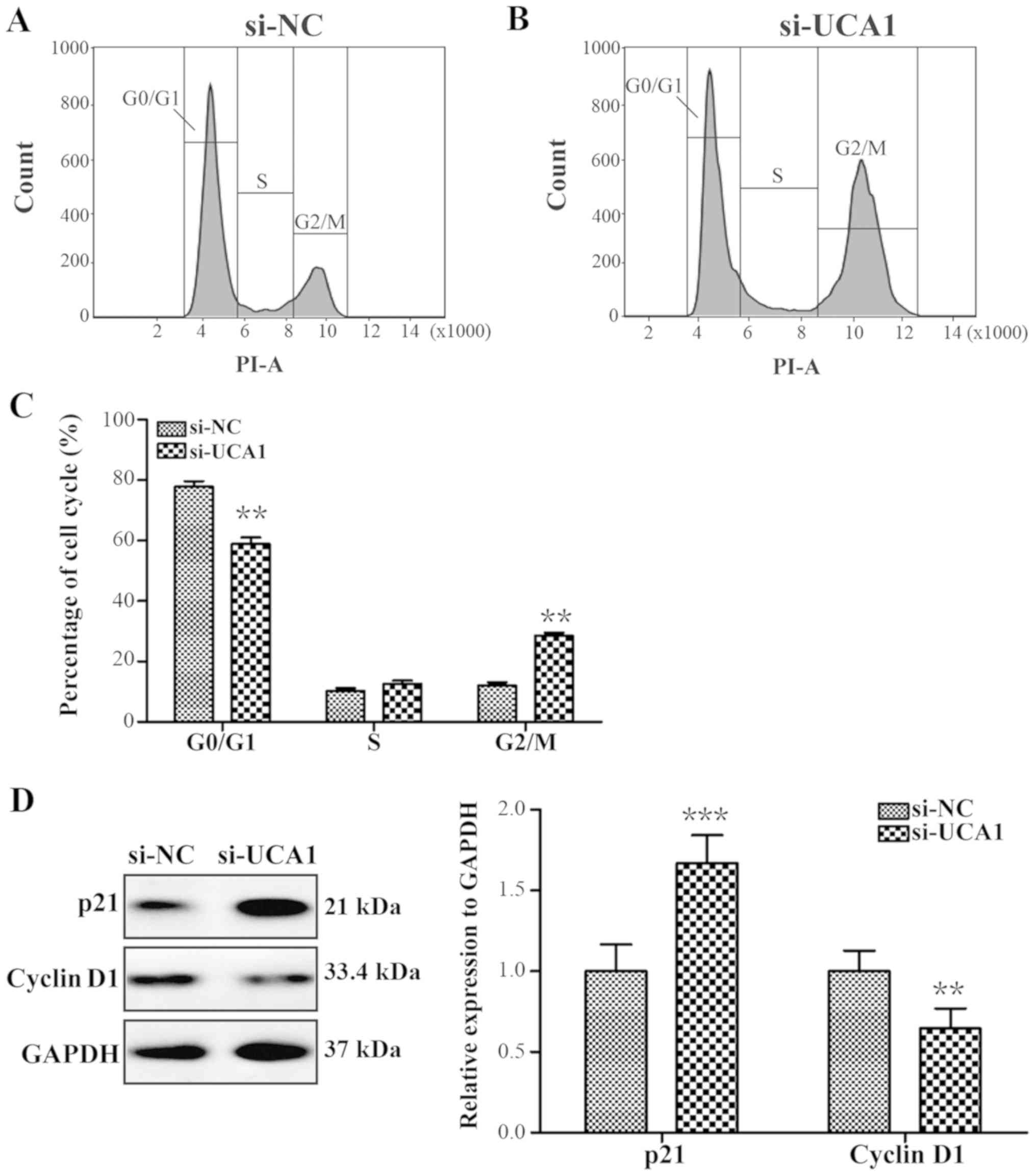
UCA1 silencing promotes G2/M
phase cell cycle arrest following tamoxifen treatment
A previous study have demonstrated that UCA1 could
promote bladder cancer progression (33). Therefore, the cell cycle
distribution in LCC2 cells post-UCA1 knockdown was assessed.
si-UCA1 LCC2 cells treated with 10 µM tamoxifen for 24 h
exhibited significant G2/M phase arrest (Fig. 4A-C; P<0.01), and the expression
level of cell cycle-associated factor p21 was significantly
upregulated and the expression level of cyclin D1 was significantly
downregulated (Fig. 4D; P<0.001
and P<0.01).
UCA1 recruits EZH2 to the p21 promoter
and represses p21 expression
It was reported that EZH2 could inhibit the
expression of p21 and that p21 is a target of UCA1 (41). It was speculated in the present
cell model that p21 may also be suppressed by UCA1 through the
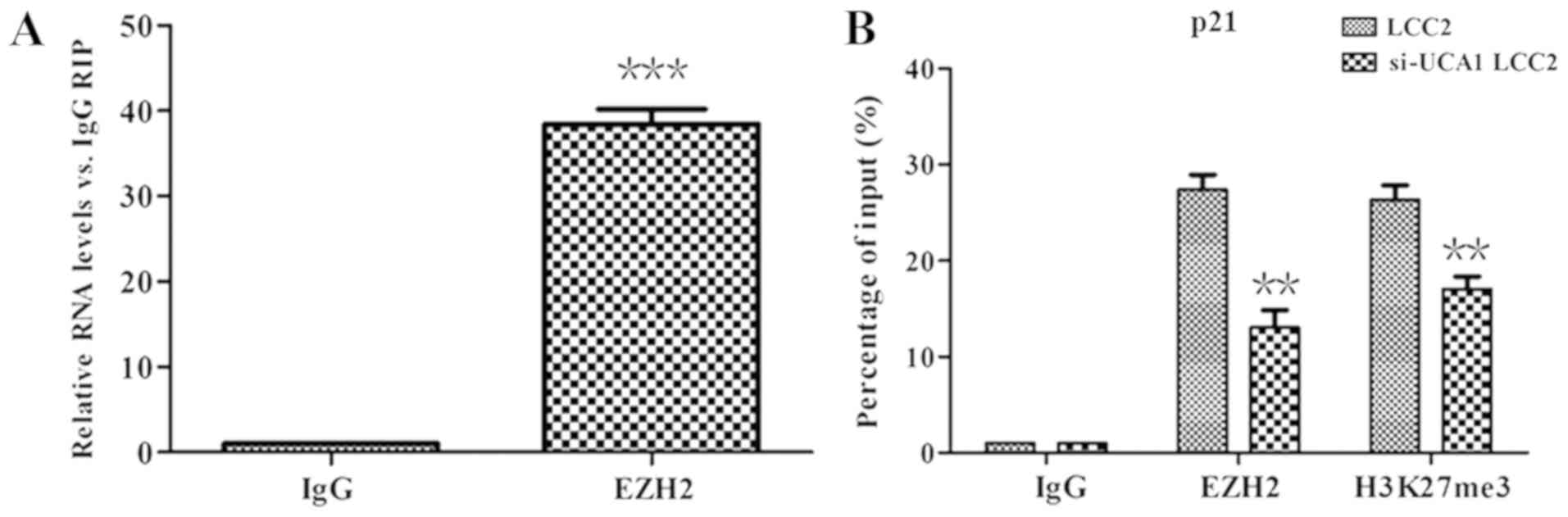
recruitment of EZH2 on the p21 promoter. Therefore, RIP analysis
was performed. The results indicated that, compared with the IgG
control antibody, UCA1 was significantly enriched by EZH2 antibody
(Fig. 5A; P<0.01).
ChIP analysis was further performed to demonstrate
whether UCA1 inhibited p21 expression by interacting with EZH2. As
indicated in Fig. 5B, EZH2 and
H3K27me3 could bind to the p21 promoter region directly. However,
in the si-UCA1 LCC2 cells, the binding of EZH2 and H3K27me3 to the
p21 promoter region was significantly weakened (P<0.01). This
finding suggested that UCA1 repressed the expression of p21 via the
recruitment of EZH2 and H3K27me3.
UCA1 contributes to tamoxifen resistance
in breast cancer cells through the PI3K/AKT signaling pathway
CREB-binding protein, a key nuclear transcription
factor in the PI3K/AKT signaling pathway, serves an important role
in cell cycle progression (42). A
previous study demonstrated that cell cycle progression was greatly
arrested in UCA1 knockdown cells, and CREB expression levels were
significantly downregulated simultaneously (33). In the present study, it was
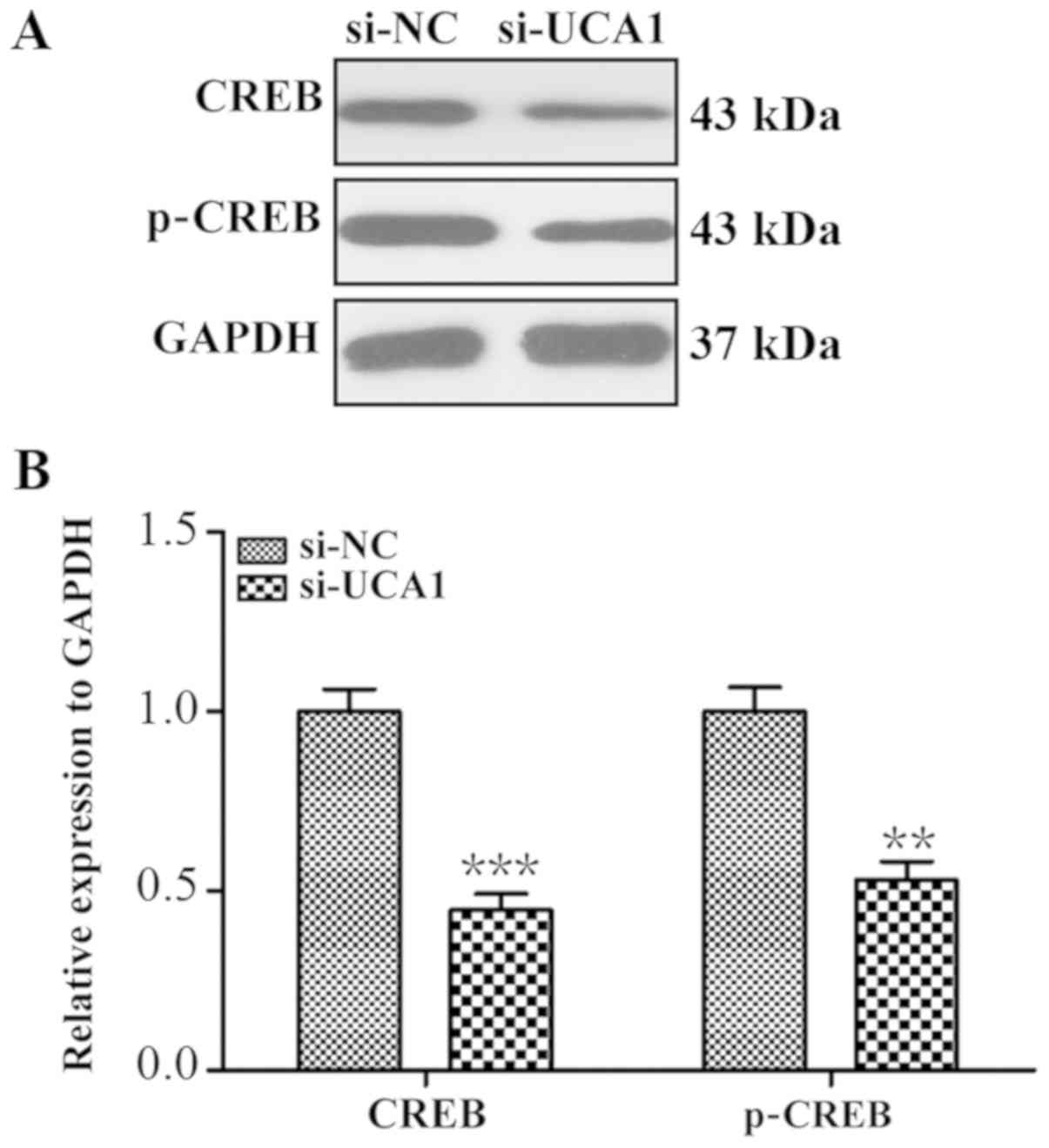
investigated whether UCA1 could influence the expression of CREB.
As indicated in Fig. 6A, CREB and
p-CREB expression levels were reduced in si-UCA1 LCC2 cells. Band
density analysis revealed that the level of CREB and the p-CREB
expression significantly decreased 3.06-fold and 2.1-fold when
compared with the control group (Fig.
6B; P<0.001 and P<0.01).
Considering that the PI3K/AKT signaling pathway is
pivotal for the maintenance of normal cell cycle progression and is
associated with CREB expression (43,44),
it was further assessed whether the PI3K/AKT signal pathway could
regulate the expression of CREB in si-UCA1 LCC2 cells in the
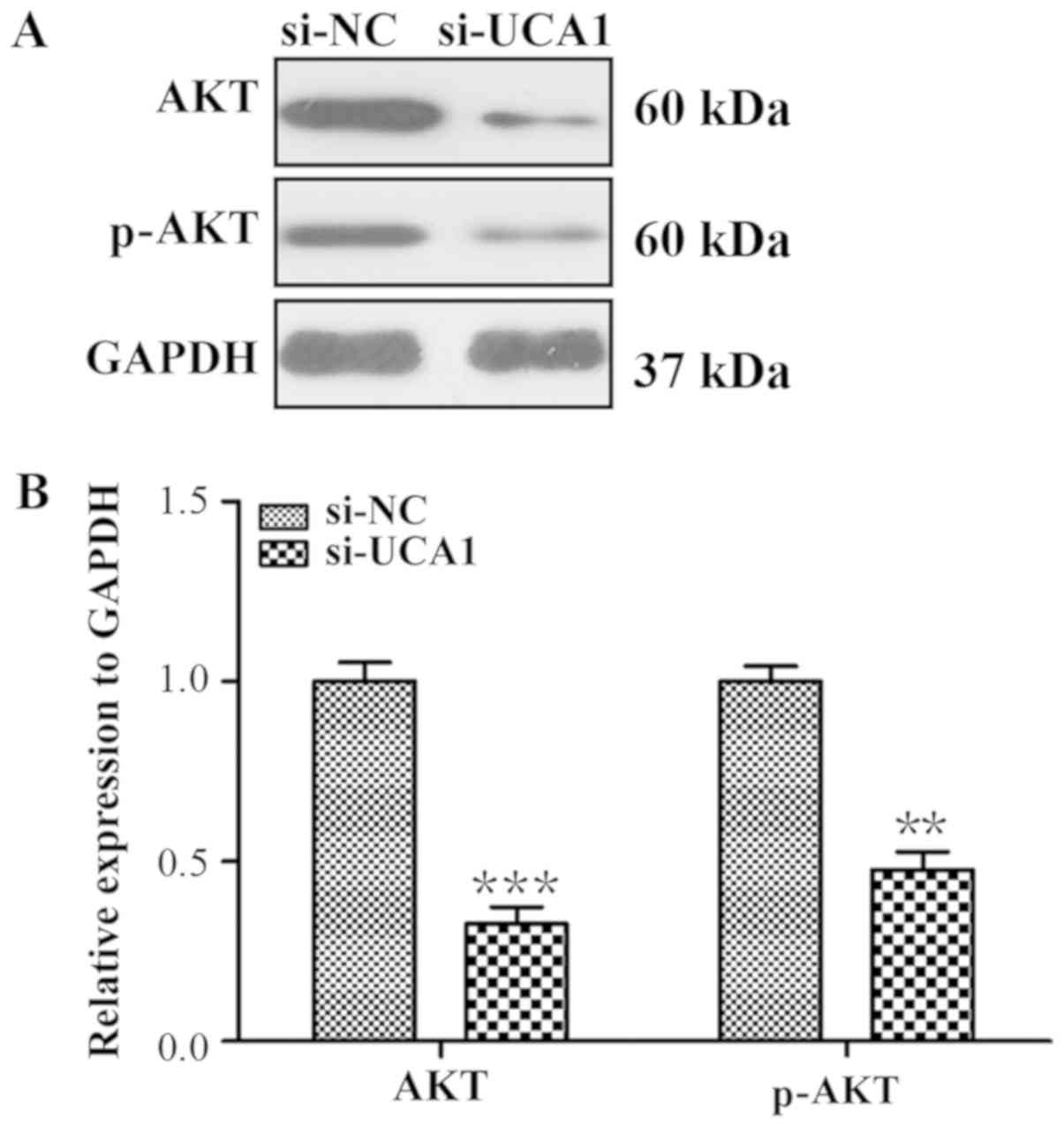
present study. As indicated in Fig.
7, the expression levels of AKT and p-AKT were significantly
reduced in si-UCA1 LCC2 cells (P<0.0001 and P<0.01),
suggesting that UCA1 was involved in the activation of AKT.
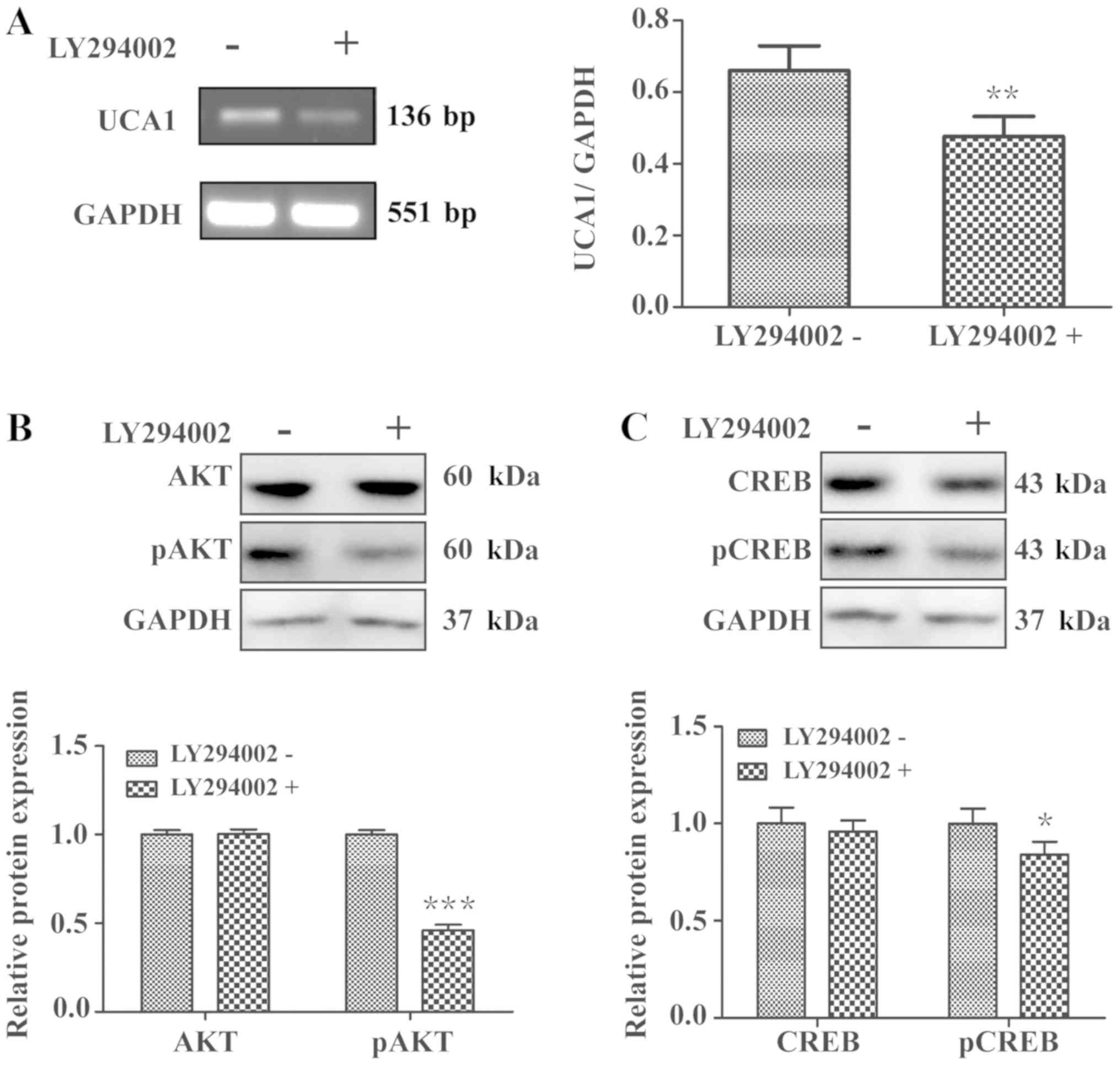
In order to further verify whether UCA1 could
regulate CREB through the PI3K/AKT signaling pathway, LCC2 cells
were treated with the PI3K inhibitor LY294002 for 24 h. qPCR
analysis revealed that LCC2 cells treated with LY294002 exhibited
significantly decreased UCA1 expression levels (Fig. 8A; P<0.01). Furthermore, the
phosphorylation of CREB and AKT was also significantly repressed in
LCC2 cells were treated with LY294002 (Fig. 8B and C; P<0.001 and P<0.05).
Taken together, these results further indicated that UCA1 regulated
the activation of CREB and impacted cell cycle progression through
PI3K/AKT-dependent signaling.
Discussion
Breast cancer currently remains the most common
female malignancy in the world (45). Tamoxifen is the most frequently
used endocrinotherapy for ER+ breast cancer (46). Despite great treatment advances in
improving the survival rate of patients with breast cancer, almost
30% of patients treated with tamoxifen may develop resistance to
the drug (47). Numerous studies
have focused on the function of lncRNA, and emerging evidence has
demonstrated that lncRNAs significantly contribute to various
aspects of cancer biology and have been identified as critical
players of drug resistance in cancer therapy (44). However, the underlying mechanisms
for tamoxifen resistance are largely unknown. In the present study,
it was indicated that UCA1 expression was significantly increased
in tamoxifen-resistant breast cancer compared with
tamoxifen-sensitive breast cancer. Following the knockdown of UCA1,
breast cancer cells exhibited a significant increase in
G2/M phase cell cycle arrest.
UCA1 has been reported to be upregulated and to
exert its oncogenic activity and enhance chemoresistance in several
cancer types (23,26,35,48).
It has been reported that UCA1 can increase chemosensitivity
through a CREB-miR-196a-5p paradigm in bladder cancer (49). Various studies have demonstrated
that UCA1 expression is elevated in breast cancer. For example, Liu
et al (50) revealed that
UCA1 regulates tamoxifen resistance through the Wnt/β-catenin
signaling pathway in breast cancer. Consistent with these reports,
in the present study it was demonstrated that UCA1 was
significantly increased in tamoxifen-resistant breast cancer.
Following treatment with tamoxifen, the expression levels of Bcl-2
and cleaved caspase-3 and -9 were increased in si-UCA1 LCC2 and
si-UCA1 LCC9 cells, which demonstrated that UCA1 contributed to
tamoxifen drug resistance in breast cancer cells. Bcl-2 protein is
a critical component in cell apoptotic signaling. It blocks the
increased permeability of the mitochondrial membrane and prevents
the release of cytochrome c (51). Several studies have reported
lncRNA-mediated sequestering of miR expression, whereas some miRs
can directly target Bcl-2 and affect the function of Bcl-2
(52-54). It was presumed that UCA1 regulated
Bcl-2 through a similar manner. However, the exact reason for this
change remains to be further studied.
The PI3K/AKT signaling pathway serves an important
role in cell growth, cell cycle distribution, apoptosis and
survival of human cancer (55).
AKT and CREB are two key molecules in this pathway. lncRNA may
regulate the activation of the PI3K/AKT signaling pathway and
affect tumorigenesis and drug sensitivity. For example, miR-21 can
modulate tamoxifen sensitivity of breast cancer cells through the
PI3K/AKT/mTOR signaling pathway (56). In the present study, it was
demonstrated that knockdown of UCA1 in LCC2 cells induced an
apparent G2/M phase arrest and altered the expression of
p21 and cyclin D1.
A previous study reported that p21 transcription
could be repressed through recruitment of EZH2, which was mediated
by UCA1 in renal cell carcinoma cells (40). EZH2 is a histone methyltransferase
that catalyzes the trimethylation of H3K27me3 of target genes. The
levels of EZH2 are frequently elevated in breast cancer (30). The present study indicated that p21
transcription was repressed by EZH2 through H3K27me3, which was
mediated by UCA1 in breast cancer cells. These data demonstrated
that UCA1 could modulate the cell cycle through EZH2 and H3K27me3
in breast cancer cells.
CREB, a proto-oncogenic transcription factor, is
crucial in cell cycle regulation of breast cancer cells (57). In the present study, the
association between the expression of UCA1 with the expression of
CREB was assessed by western blot analysis. Results demonstrated
that CREB and p-CREB expression levels were significantly decreased
when UCA1 was suppressed. CREB is mediated by various protein
kinases, including AKT and PI3K (58). Likewise, it was indicated in the
present study that AKT expression was positively associated with
UCA1 expression. A previous study reported that CREB could be
positively regulated by AKT kinase activity (33). Furthermore, the present results
confirmed that the expression levels of p-AKT and p-CREB were
inhibited by the PI3K inhibitor, LY294002, and this was consistent
with a previous report (33).
These data demonstrated that UCA1 could regulate CREB through AKT
via PI3K/AKT signaling.
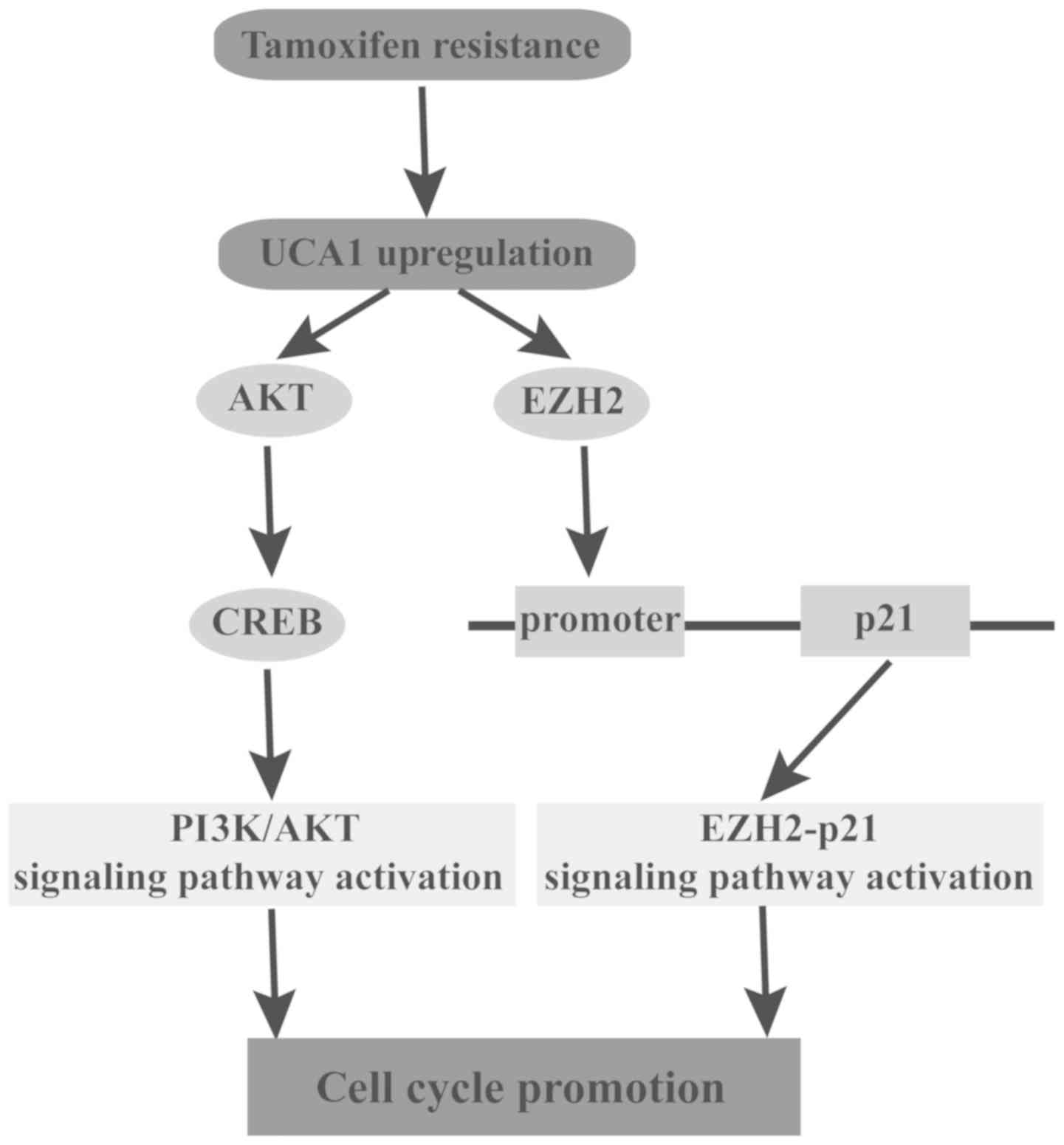
In conclusion, to the best of our knowledge the
present study demonstrated for the first time that UCA1 regulates
tamoxifen resistance through the EZH2/p21 axis and the PI3K/AKT
signaling pathway in breast cancer (Fig. 9). Based on the present results,
UCA1 may be considered a novel biomarker of poor response to
tamoxifen and a potential therapeutic intervention target of breast
cancer endocrinotherapy.
Funding
The present study was supported in part by grants
from the Natural Science Foundation of Jilin University (Bethune
plan B) (grant no. 2015311 was awarded to ZL).
Availability of data and materials
The datasets used and analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
SL contributed to the design of the study and wrote
the manuscript. ZL and DY performed the experiments and analyzed
the data. HL and YL helped perform the analysis with constructive
discussions. All authors have read and approved this
manuscript.
Ethics approval and consent to
participate
Permission to use the clinical samples for research
purposes was obtained and approved by the Ethics Committee of the
First Hospital of Jilin University.
Patient consent for publication
Informed consents were obtained from all
patients.
Competing interests
The authors declare no conflict of interest.
Acknowledgments
Not applicable.
References
|
1
|
Beiki O, Hall P, Ekbom A and Moradi T:
Breast cancer incidence and case fatality among 4.7 million women
in relation to social and ethnic background: A population-based
cohort study. Breast Cancer Res. 14:R52012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lim E, Metzger-Filho O and Winer EP: The
natural history of hormone receptor-positive breast cancer.
Oncology (Williston Park). 26:pp. 688–694. pp. 6962012
|
|
3
|
Regan MM, Neven P, Giobbie-Hurder A,
Goldhirsch A, Ejlertsen B, Mauriac L, Forbes JF, Smith I, Láng I,
Wardley A, et al: BIG 1-98 Collaborative Group; International
Breast Cancer Study Group (IBCSG): Assessment of letrozole and
tamoxifen alone and in sequence for postmenopausal women with
steroid hormone receptor-positive breast cancer: The BIG 1-98
randomised clinical trial at 8·1 years median follow-up. Lancet
Oncol. 12:1101–1108. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Colditz GA: Relationship between estrogen
levels, use of hormone replacement therapy, and breast cancer. J
Natl Cancer Inst. 90:814–823. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lumachi F, Brunello A, Maruzzo M, Basso U
and Basso SM: Treatment of estrogen receptor-positive breast
cancer. Curr Med Chem. 20:596–604. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Higgins MJ and Stearns V: CYP2D6
polymorphisms and tamoxifen metabolism: Clinical relevance. Curr
Oncol Rep. 12:7–15. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Raha P, Thomas S and Munster PN:
Epigenetic modulation: A novel therapeutic target for overcoming
hormonal therapy resistance. Epigenomics. 3:451–470. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hurvitz SA and Pietras RJ: Rational
management of endocrine resistance in breast cancer: A
comprehensive review of estrogen receptor biology, treatment
options, and future directions. Cancer. 113:2385–2397. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Anzick SL, Kononen J, Walker RL, Azorsa
DO, Tanner MM, Guan XY, Sauter G, Kallioniemi OP, Trent JM and
Meltzer PS: AIB1, a steroid receptor coactivator amplified in
breast and ovarian cancer. Science. 277:965–968. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ravdin PM, Fritz NF, Tormey DC and Jordan
VC: Endocrine status of premenopausal node-positive breast cancer
patients following adjuvant chemotherapy and long-term tamoxifen.
Cancer Res. 48:1026–1029. 1988.PubMed/NCBI
|
|
11
|
Paridaens R, Sylvester RJ, Ferrazzi E,
Legros N, Leclercq G and Heuson JC: Clinical significance of the
quantitative assessment of estrogen receptors in advanced breast
cancer. Cancer. 46(Suppl): 2889–2895. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Di Gesualdo F, Capaccioli S and Lulli M: A
pathophysiological view of the long non-coding RNA world.
Oncotarget. 5:10976–10996. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li Z, Shen J, Chan MT and Wu WK: TUG1: A
pivotal oncogenic long non-coding RNA of human cancers. Cell
Prolif. 49:471–475. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Han P and Chang CP: Long non-coding RNA
and chromatin remodeling. RNA Biol. 12:1094–1098. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kornienko AE, Guenzl PM, Barlow DP and
Pauler FM: Gene regulation by the act of long non-coding RNA
transcription. BMC Biol. 11:592013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang HY, Liang F, Zhang JW, Wang F, Wang
L and Kang XG: Effects of long noncoding RNA-ROR on tamoxifen
resistance of breast cancer cells by regulating microRNA-205.
Cancer Chemother Pharmacol. 79:327–337. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li X, Wu Y, Liu A and Tang X: Long
non-coding RNA UCA1 enhances tamoxifena resistance in breast cancer
cells through a miR-18a-HIF1α feedback regulatory loop. Tumor Biol.
37:14733–14743. 2016. View Article : Google Scholar
|
|
18
|
Cai Y, He J and Zhang D: Suppression of
long non-coding RNA CCAT2 improves tamoxifen-resistant breast
cancer cells' response to tamoxifen. Mol Biol. 50:821–827. 2016.In
Russian.
|
|
19
|
Chen G, Wang Z, Wang D, Qiu C, Liu M, Chen
X, Zhang Q, Yan G and Cui Q: lncRNADisease: A database for
long-non-coding RNA-associated diseases. Nucleic Acids Res.
41D:D983–D986. 2013.
|
|
20
|
Lottin S, Adriaenssens E, Dupressoir T,
Berteaux N, Montpellier C, Coll J, Dugimont T and Curgy JJ:
Overexpression of an ectopic H19 gene enhances the tumorigenic
properties of breast cancer cells. Carcinogenesis. 23:1885–1895.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gupta RA, Shah N, Wang KC, Kim J, Horlings
HM, Wong DJ, Tsai MC, Hung T, Argani P, Rinn JL, et al: Long
non-coding RNA HOTAIR reprograms chromatin state to promote cancer
metastasis. Nature. 464:1071–1076. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xue M, Chen W and Li X: Urothelial cancer
associated 1: A long noncoding RNA with a crucial role in cancer. J
Cancer Res Clin Oncol. 142:1407–1419. 2016. View Article : Google Scholar
|
|
23
|
Huang J, Zhou N, Watabe K, Lu Z, Wu F, Xu
M and Mo YY: Long non-coding RNA UCA1 promotes breast tumor growth
by suppression of p27 (Kip1). Cell Death Dis. 5:e10082014.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang XS, Zhang Z, Wang HC, Cai JL, Xu QW,
Li MQ, Chen YC, Qian XP, Lu TJ, Yu LZ, et al: Rapid identification
of UCA1 as a very sensitive and specific unique marker for human
bladder carcinoma. Clin Cancer Res. 12:4851–4858. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tuo YL, Li XM and Luo J: Long noncoding
RNA UCA1 modulates breast cancer cell growth and apoptosis through
decreasing tumor suppressive miR-143. Eur Rev Med Pharmacol Sci.
19:3403–3411. 2015.PubMed/NCBI
|
|
26
|
Bian Z, Jin L, Zhang J, Yin Y, Quan C, Hu
Y, Feng Y, Liu H, Fei B, Mao Y, et al: lncRNA-UCA1 enhances cell
proliferation and 5-fluorouracil resistance in colorectal cancer by
inhibiting miR-204-5p. Sci Rep. 6:238922016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li GY, Wang W, Sun JY, Xin B, Zhang X,
Wang T, Zhang QF, Yao LB, Han H, Fan DM, et al: Long non-coding
RNAs AC026904.1 and UCA1: A 'one-two punch' for TGF-β-induced SNAI2
activation and epithelial-mesenchymal transition in breast cancer.
Theranostics. 8:2846–2861. 2018. View Article : Google Scholar :
|
|
28
|
Ringrose L and Paro R: Epigenetic
regulation of cellular memory by the Polycomb and Trithorax group
proteins. Annu Rev Genet. 38:413–443. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Haupt Y, Alexander WS, Barri G, Klinken SP
and Adams JM: Novel zinc finger gene implicated as myc collaborator
by retro-virally accelerated lymphomagenesis in E mu-myc transgenic
mice. Cell. 65:753–763. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sauvageau M and Sauvageau G: Polycomb
group proteins: Multi-faceted regulators of somatic stem cells and
cancer. Cell Stem Cell. 7:299–313. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yap DB, Chu J, Berg T, Schapira M, Cheng
SW, Moradian A, Morin RD, Mungall AJ, Meissner B, Boyle M, et al:
Somatic mutations at EZH2 Y641 act dominantly through a mechanism
of selectively altered PRC2 catalytic activity, to increase H3K27
trimethylation. Blood. 117:2451–2459. 2011. View Article : Google Scholar :
|
|
32
|
Hu JJ, Song W, Zhang SD, Shen XH, Qiu XM,
Wu HZ, Gong PH, Lu S, Zhao ZJ, He ML, et al: HBx-upregulated lncRNA
UCA1 promotes cell growth and tumorigenesis by recruiting EZH2 and
repressing p27Kip1/CDK2 signaling. Sci Rep. 6:235212016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yang C, Li X, Wang Y, Zhao L and Chen W:
Long non-coding RNA UCA1 regulated cell cycle distribution via CREB
through PI3-K dependent pathway in bladder carcinoma cells. Gene.
496:8–16. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Xie X, Pan J, Wei L, Wu S, Hou H, Li X and
Chen W: Gene expression profiling of microRNAs associated with UCA1
in bladder cancer cells. Int J Oncol. 48:1617–1627. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chen S, Shao C, Xu M, Ji J, Xie Y, Lei Y
and Wang X: Macrophage infiltration promotes invasiveness of breast
cancer cells via activating long non-coding RNA UCA1. Int J Clin
Exp Pathol. 8:9052–9061. 2015.PubMed/NCBI
|
|
36
|
You D, Jung SP, Jeong Y, Bae SY, Lee JE
and Kim S: Fibronectin expression is upregulated by PI-3K/Akt
activation in tamoxifen-resistant breast cancer cells. BMB Rep.
50:615–620. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Xue M, Li X, Li Z and Chen W: Urothelial
carcinoma associated 1 is a hypoxia-inducible factor-1α-targeted
long noncoding RNA that enhances hypoxic bladder cancer cell
proliferation, migration, and invasion. Tumour Biol. 35:6901–6912.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Motawi TK, Abdelazim SA, Darwish HA, Elbaz
EM and Shouman SA: Modulation of tamoxifen cytotoxicity by caffeic
acid phenethyl ester in MCF-7 breast cancer cells. Oxid Med Cell
Longev. 2016:30171082016. View Article : Google Scholar
|
|
39
|
Grigsby JG, Parvathaneni K, Almanza MA,
Botello AM, Mondragon AA, Allen DM and Tsin AT: Effects of
tamoxifen versus raloxifene on retinal capillary endothelial cell
proliferation. J Ocul Pharmacol Ther. 27:225–233. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lu Y, Liu WG, Lu JH, Liu ZJ, Li HB, Liu
GJ, She HY, Li GY and Shi XH: lncRNA UCA1 promotes renal cell
carcinoma proliferation through epigenetically repressing p21
expression and negatively regulating miR-495. Tumour Biol.
39:10104283177016322017. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Chen WM, Huang MD, Sun DP, Kong R, Xu TP,
Xia R, Zhang EB and Shu YQ: Long intergenic non-coding RNA 00152
promotes tumor cell cycle progression by binding to EZH2 and
repressing p15 and p21 in gastric cancer. Oncotarget. 7:9773–9787.
2016.PubMed/NCBI
|
|
42
|
Giordano A and Avantaggiati ML: p300 and
CBP: Partners for life and death. J Cell Physiol. 181:218–230.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Du J, Tong A, Wang F, Cui Y, Li C, Zhang Y
and Yan Z: The Roles of PI3K/AKT/mTOR and MAPK/ERK Signaling
Pathways in Human Pheochromocytomas. Int J Endocrinol.
2016:52869722016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Liang MH, Wendland JR and Chuang DM:
Lithium inhibits Smad3/4 transactivation via increased CREB
activity induced by enhanced PKA and AKT signaling. Mol Cell
Neurosci. 37:440–453. 2008. View Article : Google Scholar
|
|
45
|
Plantamura I, Cosentino G and Cataldo A:
MicroRNAs and DNA-damaging drugs in breast cancer: Strength in
numbers. Front Oncol. 8:3522018. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Rocca A, Maltoni R, Bravaccini S, Donati C
and Andreis D: Clinical utility of fulvestrant in the treatment of
breast cancer: A report on the emerging clinical evidence. Cancer
Manag Res. 10:3083–3099. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Xia H and Hui KM: Mechanism of cancer drug
resistance and the involvement of noncoding RNAs. Curr Med Chem.
21:3029–3041. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Cheng N, Cai W, Ren S, Li X, Wang Q, Pan
H, Zhao M, Li J, Zhang Y, Zhao C, et al: Long non-coding RNA UCA1
induces non-T790M acquired resistance to EGFR-TKIs by activating
the AKT/mTOR pathway in EGFR-mutant non-small cell lung cancer.
Oncotarget. 6:23582–23593. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Pan J, Li X, Wu W, Xue M, Hou H, Zhai W
and Chen W: Long non-coding RNA UCA1 promotes cisplatin/gemcitabine
resistance through CREB modulating miR-196a-5p in bladder cancer
cells. Cancer Lett. 382:64–76. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Liu H, Wang G, Yang L, Qu J, Yang Z and
Zhou X: Knockdown of long non-coding RNA UCA1 Increases the
Tamoxifen Sensitivity of Breast cancer cells through inhibition of
Wnt/β-catenin pathway. PLoS One. 11:e01684062016. View Article : Google Scholar
|
|
51
|
Adams JM and Cory S: The Bcl-2 protein
family: Arbiters of cell survival. Science. 281:1322–1326. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Deng J, Deng H, Liu C, Liang Y and Wang S:
Long non-coding RNA OIP5-AS1 functions as an oncogene in lung
adenocar-cinoma through targeting miR-448/Bcl-2. Biomed
Pharmacother. 98:102–110. 2018. View Article : Google Scholar
|
|
53
|
Taylor MA, Sossey-Alaoui K, Thompson CL,
Danielpour D and Schiemann WP: TGF-β upregulates miR-181a
expression to promote breast cancer metastasis. J Clin Invest.
123:150–163. 2013. View Article : Google Scholar
|
|
54
|
Srivastava N, Manvati S, Srivastava A, Pal
R, Kalaiarasan P, Chattopadhyay S, Gochhait S, Dua R and Bamezai
RN: miR-24-2 controls H2AFX expression regardless of gene copy
number alteration and induces apoptosis by targeting antiapoptotic
gene BCL-2: A potential for therapeutic intervention. Breast Cancer
Res. 13:R392011. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Vanhaesebroeck B, Leevers SJ, Ahmadi K,
Timms J, Katso R, Driscoll PC, Woscholski R, Parker PJ and
Waterfield MD: Synthesis and function of 3-phosphorylated inositol
lipids. Annu Rev Biochem. 70:535–602. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Yu X, Li R, Shi W, Jiang T, Wang Y, Li C
and Qu X: Silencing of microRNA-21 confers the sensitivity to
tamoxifen and fulvestrant by enhancing autophagic cell death
through inhibition of the PI3K-AKT-mTOR pathway in breast cancer
cells. Biomed Pharmacother. 77:37–44. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
de Groot RP, Ballou LM and Sassone-Corsi
P: Positive regulation of the cAMP-responsive activator CREM by the
p70 S6 kinase: An alternative route to mitogen-induced gene
expression. Cell. 79:81–91. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Linnerth NM, Greenaway JB, Petrik JJ and
Moorehead RA: cAMP response element-binding protein is expressed at
high levels in human ovarian adenocarcinoma and regulates ovarian
tumor cell proliferation. Int J Gynecol Cancer. 18:1248–1257. 2008.
View Article : Google Scholar : PubMed/NCBI
|