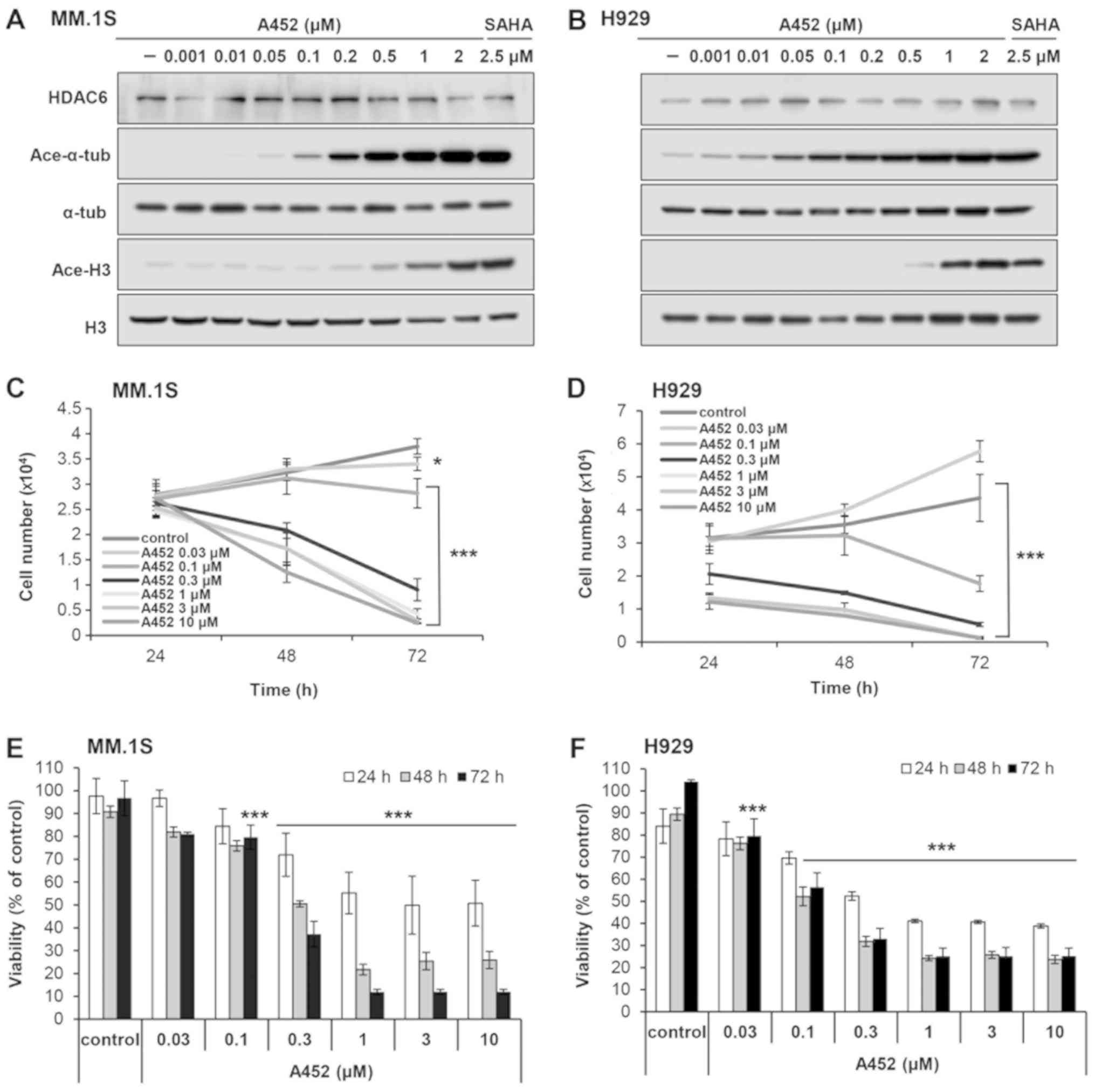
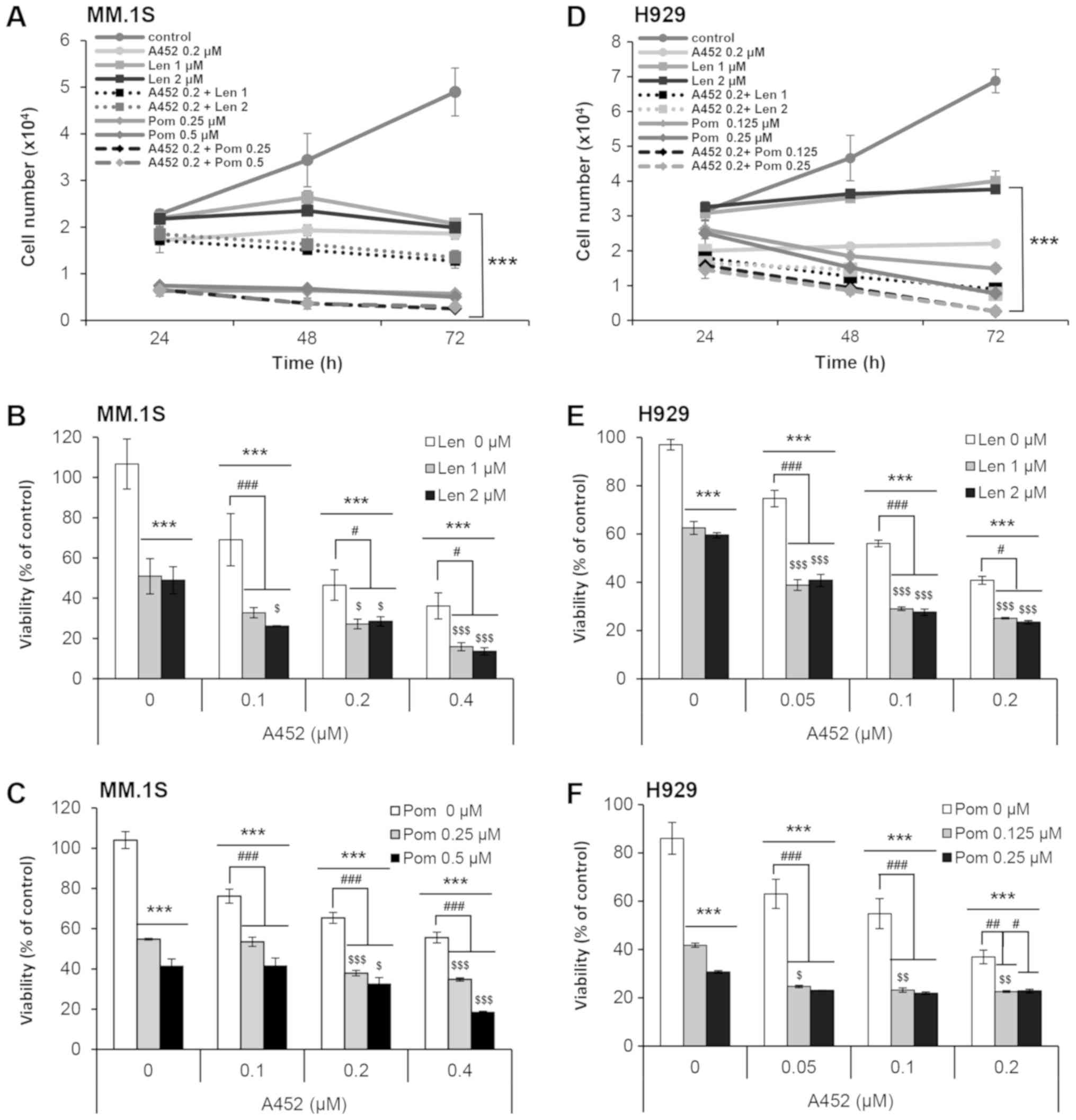
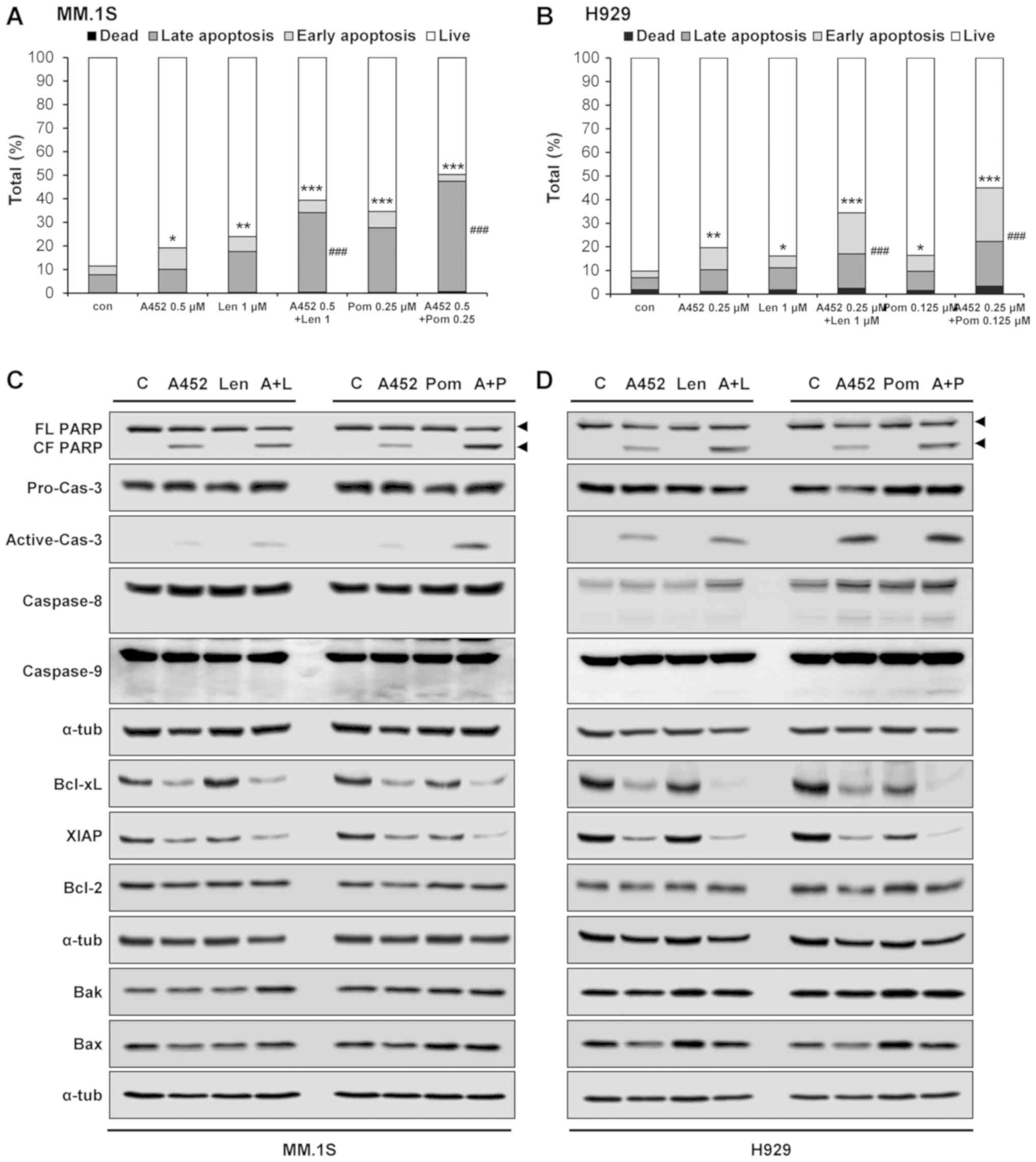
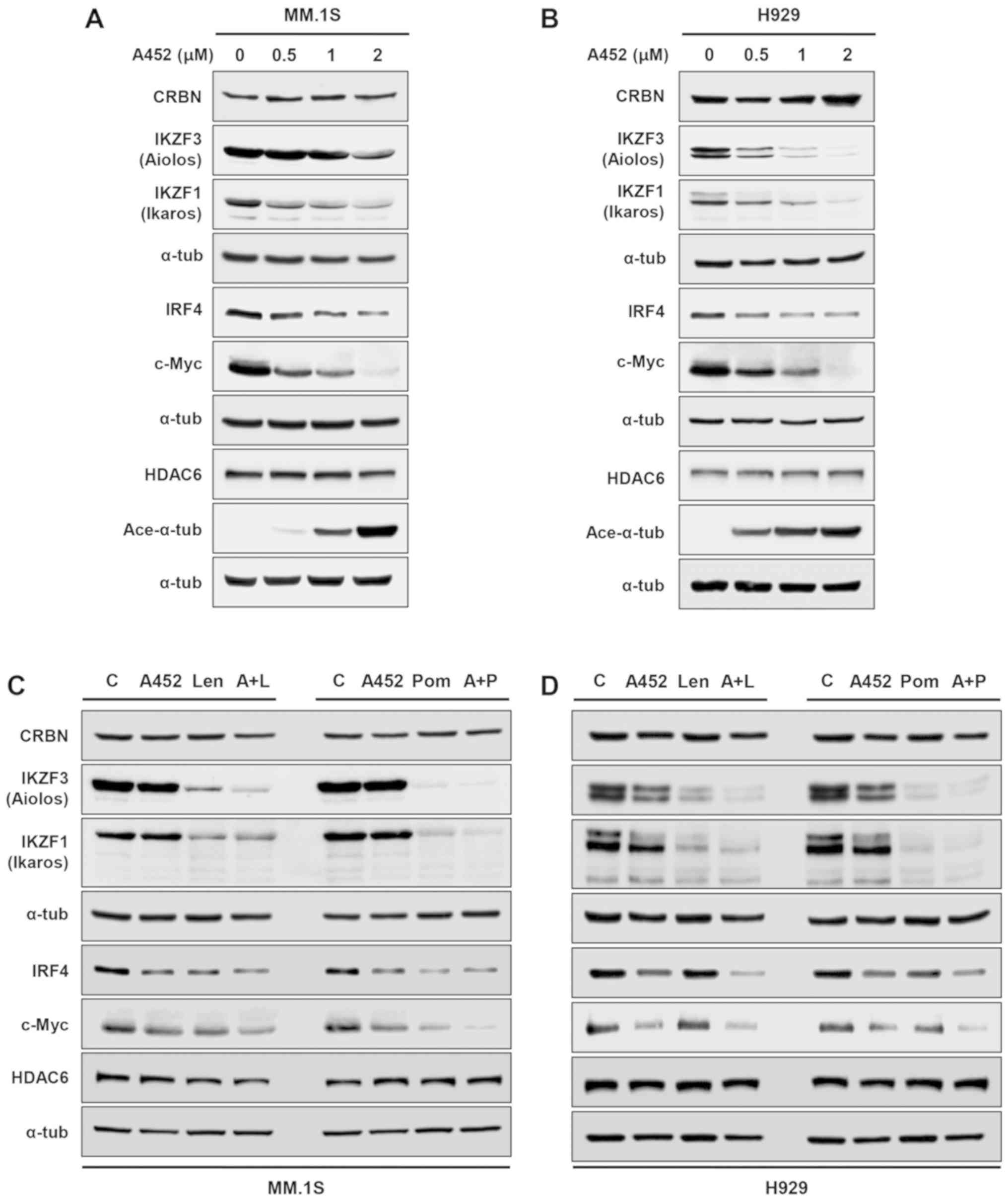
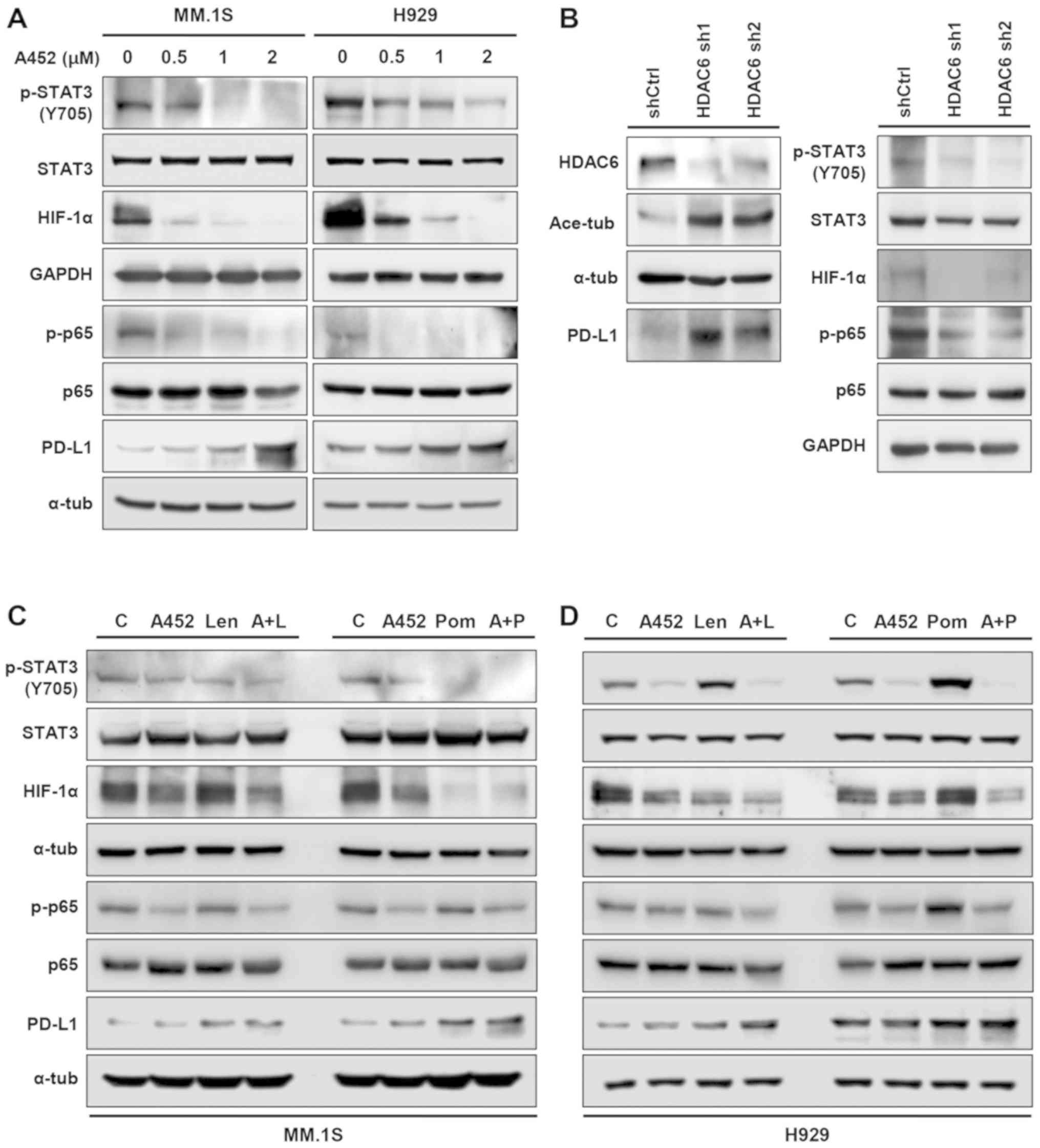
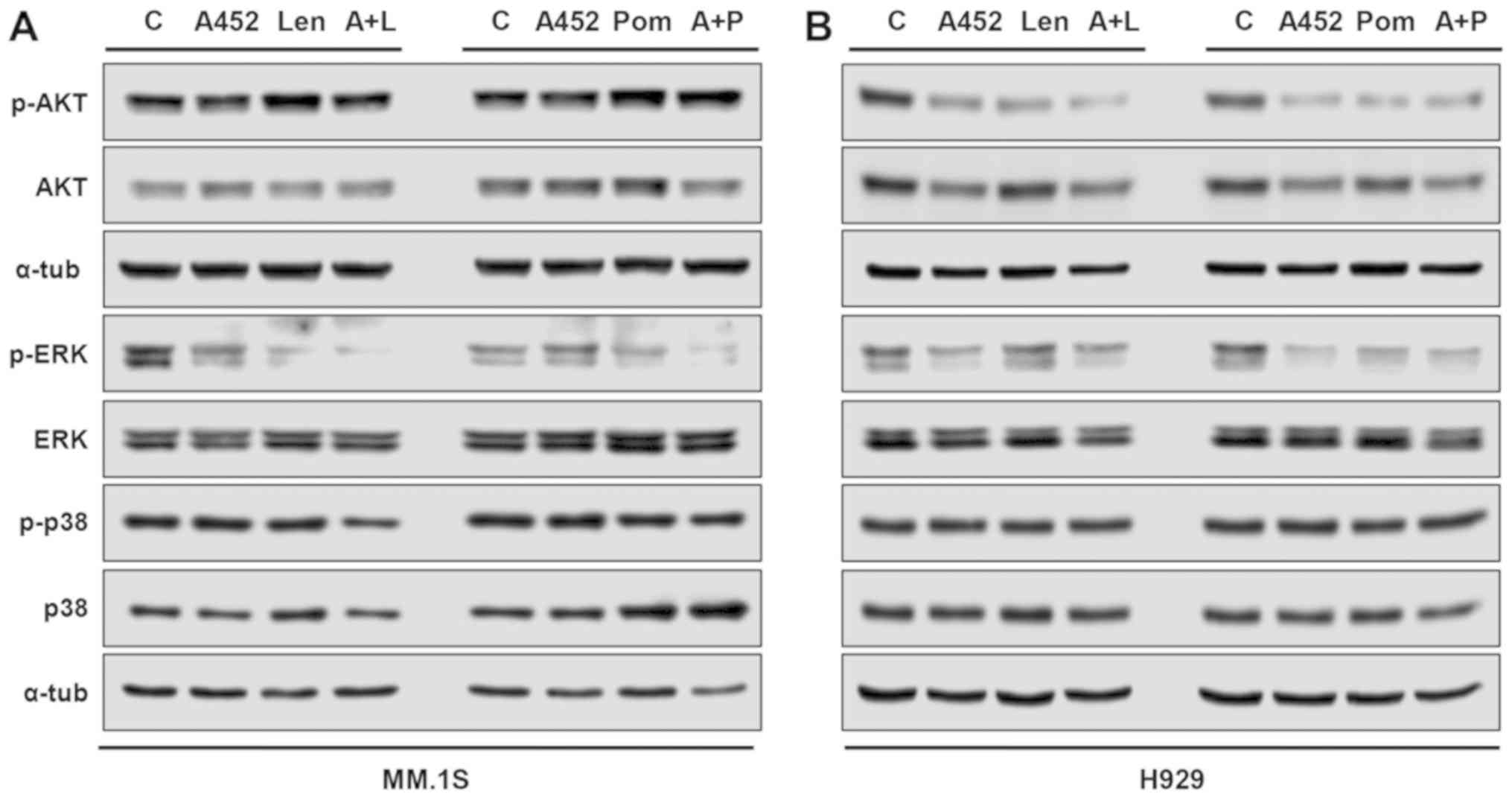
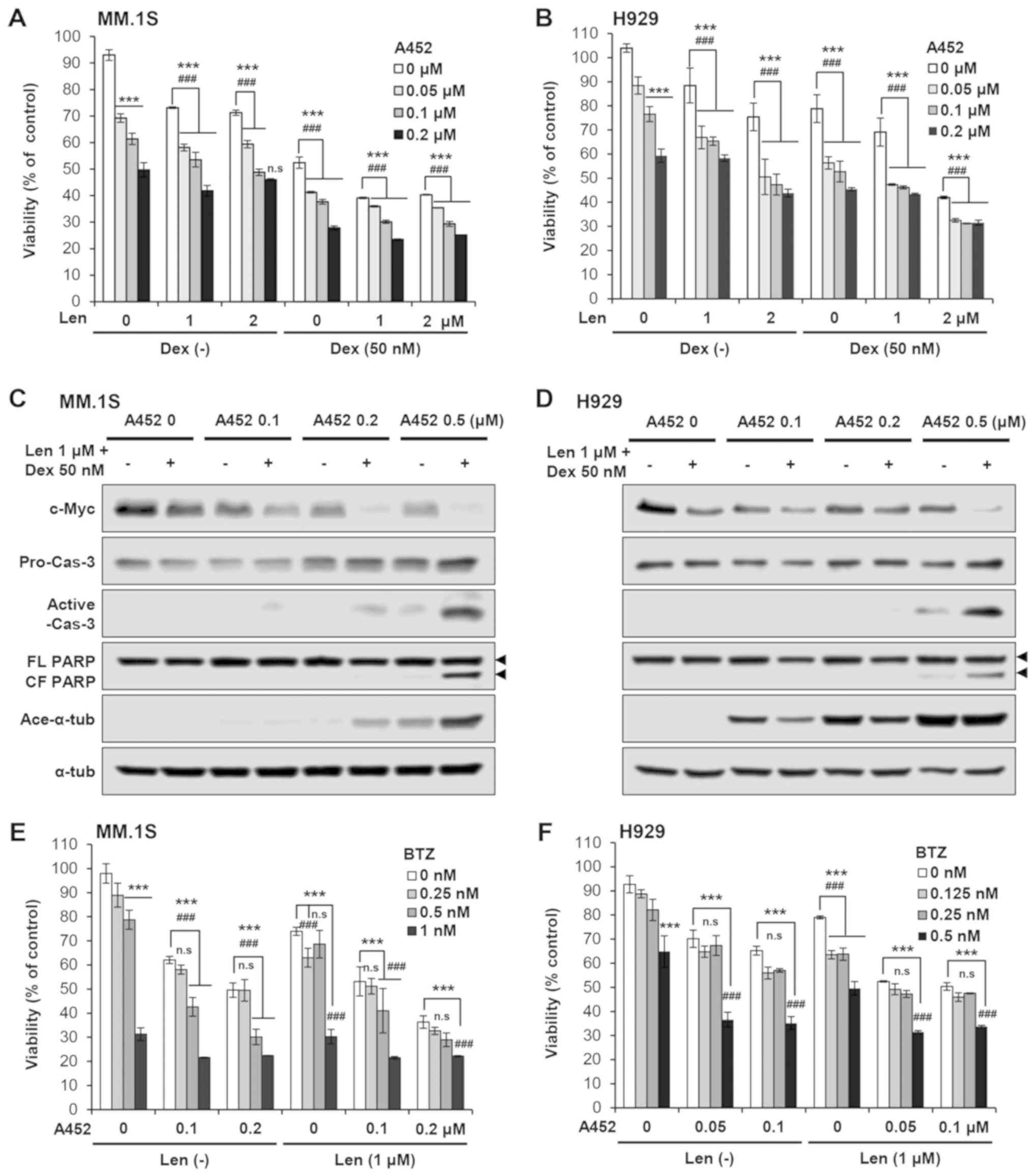
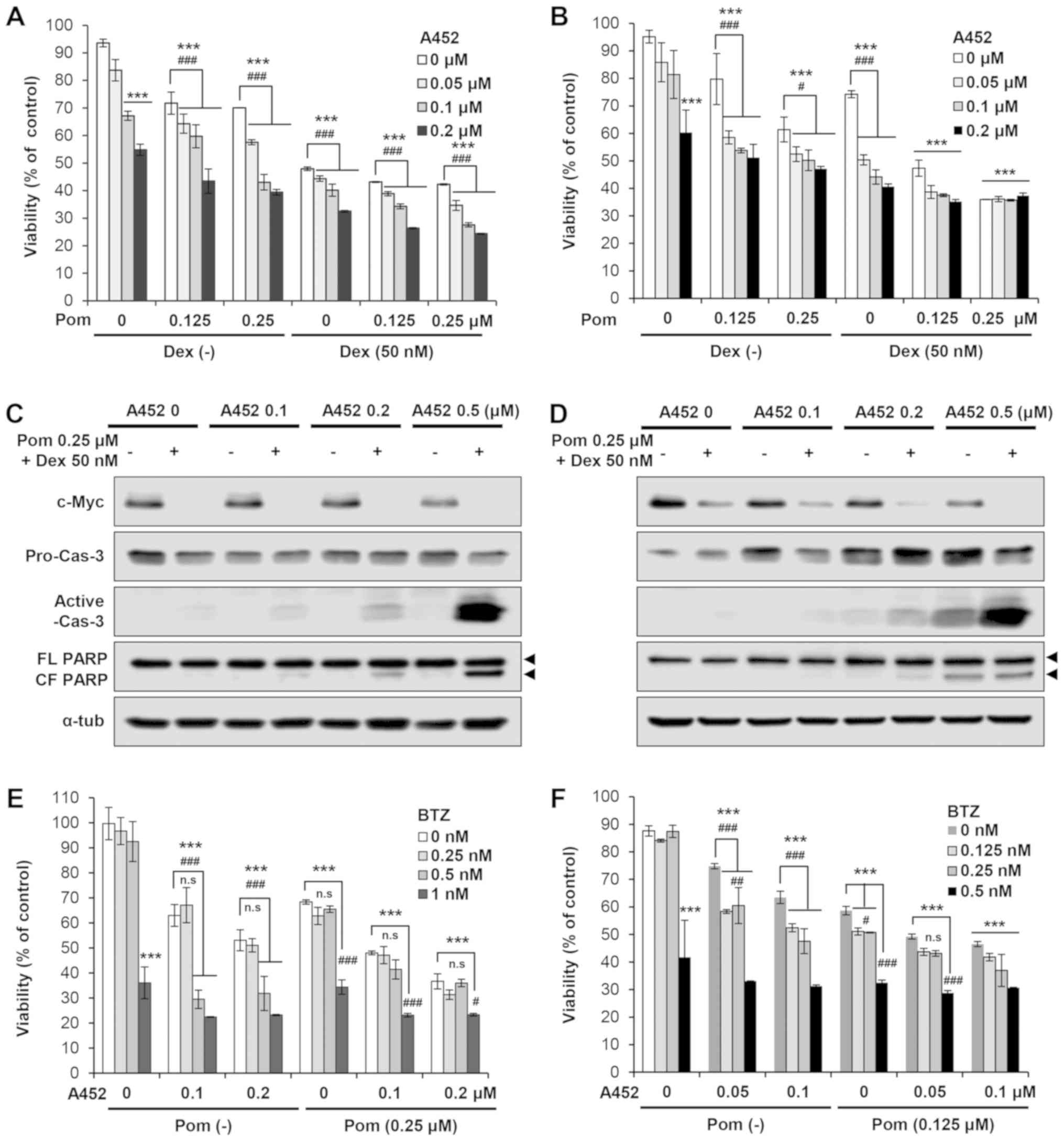
|
1
|
Palumbo A and Anderson K: Multiple
myeloma. N Engl J Med. 364:1046–1060. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Morgan GJ, Walker BA and Davies FE: The
genetic architecture of multiple myeloma. Nat Rev Cancer.
12:335–348. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Offidani M, Corvatta L, Caraffa P, Leoni
P, Pautasso C, Larocca A and Palumbo A: Pomalidomide for the
treatment of relapsed-refractory multiple myeloma: A review of
biological and clinical data. Expert Rev Anticancer Ther.
14:499–510. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Moehler T and Goldschmidt H: Therapy of
relapsed and refractory multiple myeloma. Recent Results Cancer
Res. 183:239–271. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Laubach JP, Mitsiades CS, Mahindra A,
Luskin MR, Rosenblatt J, Ghobrial IM, Schlossman RL, Avigan D, Raje
N, Munshi NC, et al: Management of relapsed and relapsed/refractory
multiple myeloma. J Natl Compr Canc Netw. 9:1209–1216. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lonial S, Mitsiades CS and Richardson PG:
Treatment options for relapsed and refractory multiple myeloma.
Clin Cancer Res. 17:1264–1277. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Dredge K, Horsfall R, Robinson SP, Zhang
LH, Lu L, Tang Y, Shirley MA, Muller G, Schafer P, Stirling D, et
al: Orally administered lenalidomide (CC-5013) is anti-angiogenic
in vivo and inhibits endothelial cell migration and Akt
phosphorylation in vitro. Microvasc Res. 69:56–63. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Davies FE, Raje N, Hideshima T, Lentzsch
S, Young G, Tai YT, Lin B, Podar K, Gupta D, Chauhan D, et al:
Thalidomide and immunomodulatory derivatives augment natural killer
cell cytotoxicity in multiple myeloma. Blood. 98:210–216. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Dredge K, Marriott JB, Macdonald CD, Man
HW, Chen R, Muller GW, Stirling D and Dalgleish AG: Novel
thalidomide analogues display anti-angiogenic activity
independently of immunomodulatory effects. Br J Cancer.
87:1166–1172. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Reddy N, Hernandez-Ilizaliturri FJ, Deeb
G, Roth M, Vaughn M, Knight J, Wallace P and Czuczman MS:
Immunomodulatory drugs stimulate natural killer-cell function,
alter cytokine production by dendritic cells, and inhibit
angiogenesis enhancing the anti-tumour activity of rituximab in
vivo. Br J Haematol. 140:36–45. 2008.
|
|
11
|
Verhelle D, Corral LG, Wong K, Mueller JH,
Moutouh-de Parseval L, Jensen-Pergakes K, Schafer PH, Chen R,
Glezer E, Ferguson GD, et al: Lenalidomide and CC-4047 inhibit the
proliferation of malignant B cells while expanding normal
CD34+ progenitor cells. Cancer Res. 67:746–755. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ito T, Ando H, Suzuki T, Ogura T, Hotta K,
Imamura Y, Yamaguchi Y and Handa H: Identification of a primary
target of thalidomide teratogenicity. Science. 327:1345–1350. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lopez-Girona A, Mendy D, Ito T, Miller K,
Gandhi AK, Kang J, Karasawa S, Carmel G, Jackson P, Abbasian M, et
al: Cereblon is a direct protein target for immunomodulatory and
antiproliferative activities of lenalidomide and pomalidomide.
Leukemia. 26:2326–2335. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Aldana-Masangkay GI, Rodriguez-Gonzalez A,
Lin T, Ikeda AK, Hsieh YT, Kim YM, Lomenick B, Okemoto K, Landaw
EM, Wang D, et al: Tubacin suppresses proliferation and induces
apoptosis of acute lymphoblastic leukemia cells. Leuk Lymphoma.
52:1544–1555. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fecteau JF, Corral LG, Ghia EM, Gaidarova
S, Futalan D, Bharati IS, Cathers B, Schwaederle M, Cui B,
Lopez-Girona A, et al: Lenalidomide inhibits the proliferation of
CLL cells via a cereblon/p21(WAF1/Cip1)-dependent mechanism
independent of functional p53. Blood. 124:1637–1644. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Schey S and Ramasamy K: Pomalidomide
therapy for myeloma. Expert Opin Investig Drugs. 20:691–700. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mehnert JM and Kelly WK: Histone
deacetylase inhibitors: Biology and mechanism of action. Cancer J.
13:23–29. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Richon VM, Garcia-Vargas J and Hardwick
JS: Development of vorinostat: Current applications and future
perspectives for cancer therapy. Cancer Lett. 280:201–210. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ropero S and Esteller M: The role of
histone deacetylases (hdacs) in human cancer. Mol Oncol. 1:19–25.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xu WS, Parmigiani RB and Marks PA: Histone
deacetylase inhibitors: Molecular mechanisms of action. Oncogene.
26:5541–5552. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Campas-Moya C: Romidepsin for the
treatment of cutaneous T-cell lymphoma. Drugs Today (Barc).
45:787–795. 2009. View Article : Google Scholar
|
|
22
|
Duvic M, Olsen EA, Breneman D, Pacheco TR,
Parker S, Vonderheid EC, Abuav R, Ricker JL, Rizvi S, Chen C, et
al: Evaluation of the long-term tolerability and clinical benefit
of vorinostat in patients with advanced cutaneous T-cell lymphoma.
Clin Lymphoma Myeloma. 9:412–416. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Marks PA and Breslow R: Dimethyl sulfoxide
to vorinostat: Development of this histone deacetylase inhibitor as
an anticancer drug. Nat Biotechnol. 25:84–90. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Siegel DS, Richardson P, Dimopoulos M,
Moreau P, Mitsiades C, Weber D, Houp J, Gause C, Vuocolo S, Eid J,
et al: Vorinostat in combination with lenalidomide and
dexamethasone in patients with relapsed or refractory multiple
myeloma. Blood Cancer J. 4:e2022014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ocio EM, Vilanova D, Atadja P, Maiso P,
Crusoe E, Fernández-Lázaro D, Garayoa M, San-Segundo L,
Hernández-Iglesias T, de Alava E, et al: In vitro and in vivo
rationale for the triple combination of pano- binostat (LBH589) and
dexamethasone with either bortezomib or lenalidomide in multiple
myeloma. Haematologica. 95:794–803. 2010. View Article : Google Scholar
|
|
26
|
San-Miguel JF, Richardson PG, Günther A,
Sezer O, Siegel D, Bladé J, LeBlanc R, Sutherland H, Sopala M,
Mishra KK, et al: Phase Ib study of panobinostat and bortezomib in
relapsed or relapsed and refractory multiple myeloma. J Clin Oncol.
31:3696–3703. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sivaraj D, Green MM and Gasparetto C:
Panobinostat for the management of multiple myeloma. Future Oncol.
13:477–488. 2017. View Article : Google Scholar
|
|
28
|
Bergman JA, Woan K, Perez-Villarroel P,
Villagra A, Sotomayor EM and Kozikowski AP: Selective histone
deacetylase 6 inhibitors bearing substituted urea linkers inhibit
melanoma cell growth. J Med Chem. 55:9891–9899. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Butler LM, Agus DB, Scher HI, Higgins B,
Rose A, Cordon-Cardo C, Thaler HT, Rifkind RA, Marks PA and Richon
VM: Suberoylanilide hydroxamic acid, an inhibitor of histone
deacetylase, suppresses the growth of prostate cancer cells in
vitro and in vivo. Cancer Res. 60:5165–5170. 2000.PubMed/NCBI
|
|
30
|
Inks ES, Josey BJ, Jesinkey SR and Chou
CJ: A novel class of small molecule inhibitors of HDAC6. ACS Chem
Biol. 7:331–339. 2012. View Article : Google Scholar :
|
|
31
|
Santo L, Hideshima T, Kung AL, Tseng JC,
Tamang D, Yang M, Jarpe M, van Duzer JH, Mazitschek R, Ogier WC, et
al: Preclinical activity, pharmacodynamic, and pharmacokinetic
properties of a selective HDAC6 inhibitor, ACY-1215, in combination
with bortezomib in multiple myeloma. Blood. 119:2579–2589. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Smil DV, Manku S, Chantigny YA, Leit S,
Wahhab A, Yan TP, Fournel M, Maroun C, Li Z, Lemieux AM, et al:
Novel HDAC6 isoform selective chiral small molecule histone
deacetylase inhibitors. Bioorg Med Chem Lett. 19:688–692. 2009.
View Article : Google Scholar
|
|
33
|
Hideshima T, Cottini F, Ohguchi H,
Jakubikova J, Gorgun G, Mimura N, Tai YT, Munshi NC, Richardson PG
and Anderson KC: Rational combination treatment with histone
deacetylase inhibitors and immunomodulatory drugs in multiple
myeloma. Blood Cancer J. 5:e3122015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yee AJ, Bensinger WI, Supko JG, Voorhees
PM, Berdeja JG, Richardson PG, Libby EN, Wallace EE, Birrer NE,
Burke JN, et al: Ricolinostat plus lenalidomide, and dexamethasone
in relapsed or refractory multiple myeloma: A multicentre phase 1b
trial. Lancet Oncol. 17:1569–1578. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Raje N, Vogl DT, Hari PN, et al: ACY-1215,
a selective histone deacetylase (HDAC) 6 inhibitor: Interim results
of combination therapy with bortezomib in patients with multiple
myeloma (MM). Blood. 122:7592013.
|
|
36
|
Choi E, Lee C, Park JE, Seo JJ, Cho M,
Kang JS, Kim HM, Park SK, Lee K and Han G: Structure and property
based design, synthesis and biological evaluation of γ-lactam based
HDAC inhibitors. Bioorg Med Chem Lett. 21:1218–1221. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ryu HW, Shin DH, Lee DH, Won HR and Kwon
SH: A potent hydroxamic acid-based, small-molecule inhibitor A452
preferentially inhibits HDAC6 activity and induces cytotoxicity
toward cancer cells irrespective of p53 status. Carcinogenesis.
39:72–83. 2018. View Article : Google Scholar
|
|
38
|
Lee DH, Won HR, Ryu HW, Han JM and Kwon
SH: The HDAC6 inhibitor ACY-1215 enhances the anticancer activity
of oxali- platin in colorectal cancer cells. Int J Oncol.
53:844–854. 2018.PubMed/NCBI
|
|
39
|
Chou TC: Drug combination studies and
their synergy quantification using the Chou-Talalay method. Cancer
Res. 70:440–446. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Witter DJ, Harrington P, Wilson KJ,
Chenard M, Fleming JC, Haines B, Kral AM, Secrist JP and Miller TA:
Optimization of biaryl Selective hdac1&2 Inhibitors (SHI-1:2).
Bioorg Med Chem Lett. 18:726–731. 2008. View Article : Google Scholar
|
|
41
|
López R, Cuca LE and Delgado G:
Antileishmanial and immunomodulatory activity of Xylopia discreta.
Parasite Immunol. 31:623–630. 2009. View Article : Google Scholar
|
|
42
|
Kronke J, Udeshi ND, Narla A, Grauman P,
Hurst SN, McConkey M, Svinkina T, Heckl D, Comer E, Li X, et al:
Lenalidomide causes selective degradation of IKZF1 and IKZF3 in
multiple myeloma cells. Science. 343:301–305. 2014. View Article : Google Scholar :
|
|
43
|
Lu G, Middleton RE, Sun H, Naniong M, Ott
CJ, Mitsiades CS, Wong KK, Bradner JE and Kaelin WG Jr: The myeloma
drug lenalidomide promotes the cereblon-dependent destruction of
Ikaros proteins. Science. 343:305–309. 2014. View Article : Google Scholar :
|
|
44
|
Zhu YX, Braggio E, Shi CX, Bruins LA,
Schmidt JE, Van Wier S, Chang XB, Bjorklund CC, Fonseca R,
Bergsagel PL, et al: Cereblon expression is required for the
antimyeloma activity of lenalidomide and pomalidomide. Blood.
118:4771–4779. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
M L, P PV, T K, M P, E S, J P, K v W, C L,
F C, S D, et al: Essential role of HDAC6 in the regulation of PD-L1
in melanoma. Mol Oncol. 10:735–750. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Chen J, Jiang CC, Jin L and Zhang XD:
Regulation of PD-L1: A novel role of pro-survival signalling in
cancer. Ann Oncol. 27:409–416. 2016. View Article : Google Scholar
|
|
47
|
Hideshima T, Bradner JE, Wong J, Chauhan
D, Richardson P, Schreiber SL and Anderson KC: Small-molecule
inhibition of proteasome and aggresome function induces synergistic
antitumor activity in multiple myeloma. Proc Natl Acad Sci USA.
102:8567–8572. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Hideshima T, Mazitschek R, Santo L, Mimura
N, Gorgun G, Richardson PG, Raje N and Anderson KC: Induction of
differential apoptotic pathways in multiple myeloma cells by
class-selective histone deacetylase inhibitors. Leukemia.
28:457–460. 2014. View Article : Google Scholar
|
|
49
|
Desplanques G, Giuliani N, Delsignore R,
Rizzoli V, Bataille R and Barillé-Nion S: Impact of XIAP protein
levels on the survival of myeloma cells. Haematologica. 94:87–93.
2009. View Article : Google Scholar :
|
|
50
|
Bjorklund CC, Lu L, Kang J, Hagner PR,
Havens CG, Amatangelo M, Wang M, Ren Y, Couto S, Breider M, et al:
Rate of CRL4(CRBN) substrate Ikaros and Aiolos degradation
underlies differential activity of lenalidomide and pomalidomide in
multiple myeloma cells by regulation of c-Myc and IRF4. Blood
Cancer J. 5:e3542015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Broyl A, Kuiper R, van Duin M, van der
Holt B, el Jarari L, Bertsch U, Zweegman S, Buijs A, Hose D,
Lokhorst HM, et al: Dutch-Belgian HOVON group; German gmmg Group.
High cereblon expression is associated with better survival in
patients with newly diagnosed multiple myeloma treated with
thalidomide maintenance. Blood. 121:624–627. 2013. View Article : Google Scholar
|
|
52
|
Heintel D, Rocci A, Ludwig H, Bolomsky A,
Caltagirone S, Schreder M, Pfeifer S, Gisslinger H, Zojer N, Jäger
U, et al: High expression of cereblon (CRBN) is associated with
improved clinical response in patients with multiple myeloma
treated with lenalidomide and dexamethasone. Br J Haematol.
161:695–700. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Richardson PG, Xie W, Jagannath S,
Jakubowiak A, Lonial S, Raje NS, Alsina M, Ghobrial IM, Schlossman
RL, Munshi NC, et al: A phase 2 trial of lenalidomide, bortezomib,
and dexamethasone in patients with relapsed and relapsed/refractory
myeloma. Blood. 123:1461–1469. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Richardson PG, Weller E, Lonial S,
Jakubowiak AJ, Jagannath S, Raje NS, Avigan DE, Xie W, Ghobrial IM,
Schlossman RL, et al: Lenalidomide, bortezomib, and dexamethasone
combination therapy in patients with newly diagnosed multiple
myeloma. Blood. 116:679–686. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Nooka AK, Kaufman JL, Muppidi S, Langston
A, Heffner LT, Gleason C, Casbourne D, Saxe D, Boise LH and Lonial
S: Consolidation and maintenance therapy with lenalidomide,
bortezomib and dexamethasone (RVD) in high-risk myeloma patients.
Leukemia. 28:690–693. 2014. View Article : Google Scholar
|
|
56
|
Ashjian E and Redic K: Multiple myeloma.
Updates for pharmacists in the treatment of relapsed and refractory
disease. J Oncol Pharm Pract. 22:289–302. 2016. View Article : Google Scholar
|
|
57
|
Messaoudi K, Ali A, Ishaq R, Palazzo A,
Sliwa D, Bluteau O, Souquére S, Muller D, Diop KM, Rameau P, et al:
Critical role of the HDAC6-cortactin axis in human megakaryocyte
maturation leading to a proplatelet-formation defect. Nat Commun.
8:17862017. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Badros A, Hyjek E, Ma N, Lesokhin A, Dogan
A, Rapoport AP, Kocoglu M, Lederer E, Philip S, Milliron T, et al:
Pembrolizumab, pomalidomide, and low-dose dexamethasone for
relapsed/refractory multiple myeloma. Blood. 130:1189–1197. 2017.
View Article : Google Scholar : PubMed/NCBI
|