Introduction
Osteosarcoma (OS), a malignant primary tumor, is one
of the most common primary bone tumors and mainly generates in
teenagers (1). OS originates from
primitive bone-forming mesenchymal stem cells and is usually
associated with an aggressive behavior and growth speed, a high
local recurrence rate and a high metastatic potential (2,3).
With the improvement of medical technology, the 5-year survival
rate for patients with local OS remains at 70%, while for patients
with metastasis it remains at merely 20% (4). Over the past decades, no marked
improvements have been made in OS therapy (at least to the best of
our knowledge) and this is mainly due to local recurrence or
distant metastasis (5). Thus, the
study of the molecular mechanisms of OS tumorigenesis is
imperative, as well as the exploration of novel biomarkers for the
early diagnosis of OS.
The Ski oncogene was first isolated from the avian
Sloan-Kettering viruses, and its expression can induce the
transformation of chicken embryo fibroblasts, and its oncogenic
activity is achieved through the inhibition of the transforming
growth factor-β(TGF-β)/Smad signaling pathway (6-8). A
number of studies have proven that Ski expression is notably
upregulated in some human cancer cell lines and is associated with
a number of advanced stages tumors, including Barrett's esophagus
(9), colorectal cancer (10), gastric cancer (11), pancreatic cancer (12) and melanoma (13,14).
However, the exact function of Ski in human OS and the underlying
mechanisms in tumorigenesis remain unclear.
In this study, we concentrated on revealing the
roles of Ski in OS and the underlying mechanisms. We found that Ski
was over expression in OS tissues and cell lines. Moreover, the
knockdown of Ski decreased OS cell proliferative and migratory
abilities, and these effects may be mediated via the inhibition of
the PI3K/Akt signaling pathway. These findings indicate that Ski
may prove to be a potential therapeutic target in the treatment of
OS.
Materials and methods
OS samples
The study protocol was specifically approved by the
Ethics Committee of West China Hospital of Sichuan University
(Chengdu, China) and complied with the Declaration of Helsinki. A
total of 6 OS tissue samples and 6 osteochondroma tissue samples
were collected during surgery from patients between October 1, 2017
and December 31, 2018, and the patient information is presented in
Table I. No patients had received
radiotherapy or chemotherapy prior to surgery. All patients
provided written informed consent and agreed to participate in this
research. All collected tissues were frozen instantly in liquid
nitrogen following resection and were preserved in an ultralow
temperature refrigerator prior to use in the experiments.
 | Table IClinical profles of the 12 patients
with osteosarcoma and osteochondroma. |
Table I
Clinical profles of the 12 patients
with osteosarcoma and osteochondroma.
| Clinicopathological
parameters | No. of osteosarcoma
patients (n=6) | No. of
osteochondroma patients (n=6) |
|---|
| Sex | | |
| Male | 3 | 4 |
| Female | 3 | 2 |
| Age, years | | |
| Median | 38 | 17 |
| Range | 10-49 | 13-52 |
| Tumor location | | |
| Femur | 3 | 4 |
| Tibia and
fibula | 2 | 2 |
| Others | 1 | 0 |
| Enneking stage | | |
| I/II A | 2 | - |
| IIB/III | 4 | - |
| Metastasis | | |
| Yes | 4 | - |
| No | 2 | - |
Immunohistochemistry (IHC) analysis
The expression of Ski in both OS tissues and
osteochondroma tissues was detected by IHC under the standard
immunoperoxidase staining procedure. In brief, the tissues were
embedded with paraffin and conventionally sliced into
4-µm-thick sections. The sections were baked for 2 h at 60°C
before being dewaxed in xylene, and were then rehydrated through
graded ethanol, and subsequently placed in sodium citrate buffer
(pH 6.0) and heated up to 100°C for 5 min for antigen repair. After
cooling down, the sections were incubated with 5% goat serum
(Solarbio) at 37°C for 30 min, and the sections were then incubated
with rabbit anti-human Ski monoclonal antibodies (1:200; sc-33693,
Santa Cruz Biotechnology) at 4°C overnight, and then incubated with
the secondary antibody (1:1,000; biotinylated goat anti-mouse lgG,
ZB-2305; ZSBIO) at 37°C for 1 h. The sections were developed with
diaminobenzidine (DAB; Beyotime Biotechnology) to detect the bound
antibody. After staining, the sections were sealed up with balsam
before observation using an optical microscope (Olympus Optical).
The brownish-yellow presented in cytoplasm or cytomembrane
indicated positive results, or otherwise indicated negative
results. Five fields were randomly selected for detection to
calculate the positive expression. The experiment was performed 3
times.
Cell lines and cell culture
All the cell lines were purchased from the Cellular
Center of Institute of Basic Medical Sciences. Human OS cell lines,
including MG63 and U2OS were cultured in Eagle's minimum essential
medium containing 10% fetal bovine serum (FBS; Gibco; Thermo Fisher
Scientific). The human normal osteoblasts (hFOB1.19) were cultured
in Dulbecco's modified Eagle's medium/F12 (DMEM/F12; HyClone)
containing 10% FBS (Gibco; Thermo Fisher Scientific) and 1%
streptomycin and penicillin (Gibco; Thermo Fisher Scientific). The
cells were cultured in a humidified atmosphere in CO2
cell incubator (37°C, 5% CO2).
Western blot analysis
The protein samples were extracted from the tissues
or cells using RIPA lysis buffer (990 µl RIPA and 10
µl phenylmethylsulfonyl fluoride; Beyotime) on the ice. The
supernatants were then transferred into a new 1.5 ml centrifuge
tube and centrifuged in a low temperature environment for 15 min at
17,900 × g, and insoluble material was discarded. The sample
concentration was detected using the BCA kit (Beyotime). The
protein samples (20 µg) were separated by 10 or 12%
SDS-PAGE, and the proteins were then transferred onto PVDF
membranes (0.45 µm; Millipore) using a transfer unit
(Bio-Rad). Thereafter, the membranes were blocked in TBS solution
containing 5% BSA for 2 h at room temperature. The membranes were
then incubated with primary antibodies including anti-Ski (mouse,
1:200, sc-33693; Santa Cruz Biotechnology), proliferating cell
nuclear antigen (PCNA; 1:1,000, 10205-2-AP, rabbit; Proteintech),
CDK4 (sc-23896), cyclin D1 (sc-450) (both mouse, 1:500; Santa Cruz
Biotechnology), matrix metalloproteinase (MMP)2 (4022S), MMP9
(3852S) (both rabbit, 1:1,000; Cell Signaling Technology), PI3K
(4292S), p-PI3K (4228S), Akt (9272S) (all rabbit, 1:1,000; Cell
Signaling Technology), p-Akt (4060S, rabbit, 1:2,000; Cell
Signaling Technology) and β-actin (TA09, mouse, 1:1,000; ZSBIO)
overnight at 4°C. Following washing with TBST, the membranes were
then incubated with secondary antibodies (ZB-2301 and ZB2305,
1:10,000; ZSBIO) for 1 h. After washing with TBST 3 times, the
proteins on the membranes were visualized by ECL, and the results
were scanned by Quantity One software (Bio-Rad).
Immunofluorescence staining
The cells were cultured on small coverslips and
allowed to adhere. Thereafter, the cells were washed with PBS
solution for 5 min each, then fixed with 4% paraformaldehyde for 30
min, and washed with PBS for 5 min each. The cells were then
permeabilized with 0.3% Triton X-100 for 20 min, and washed in PBS
for 5 min each. The cells were blocked for 30 min with 10% goat
serum at 37°C, and thereafter, anti-Ski (sc-33693, anti-human
1:300; Santa Cruz Biotechnology); antibodies were added followed by
incubation overnight at 4°C. The following day, the cells were
washed in PBS 3 times, and then incubated with secondary antibodies
(Alexa Fluor 488-conjugated anti-mouse, 1:300; A21202; Invitrogen;
Thermo Fisher Scientific) for 2 h at room temperature. Finally, the
samples were incubated with DAPI for 10 min at room temperature.
The images were acquired with a fluorescence microscope (Zeiss;
Carl Zeiss).
Transfection assay
Ski specific siRNA (Ski-siRNA) and scrambled
sequence siRNA (negative control siRNA, NC-siRNA) were purchased
from Thermo Fisher Scientific. The target sequence of Ski-siRNA was
5′-CGGACCTTGGCTGGTTCCTCCAATA-3′, and the sequence of
5′-TTCTCCGAACGTGTCACGT-3′ was considered to be NC-siRNA. The MG-63
and U2OS cells were transfected with Ski-siRNA or NC-siRNA using
Lipofectamine® RNAiMAX Reagent (Thermo Fisher
Scientific) in accordance with the manufacture's protocol.
Following 48 h of transfection, the cells were collected for the
used in the following experiments. Western blot analyses were used
to assess the silencing effect of Ski-siRNA. The cells were
pre-treated with basic culture medium DMEM/F12 with or without 50
µM LY294002 (a specific PI3K inhibitor, S1737, Beyotime) for
6 h prior to transfection.
Proliferation assay in vitro
Cell Counting kit-8 (CCK-8) assays (Dojindo) were
used to assess cell proliferation. In brief, at 48 h following
transfectiong with Ski-siRNA and NC-siRNA, the cells were
trypsinized, and then replanted into 96-well plates with
3×103 cells/well for the cell proliferation assay. This
was followed by the addition of DMEM (100 µl) containing 10
µl of CCK-8 working solution to each well at corresponding
time-points and incubation at 37°C for 3 h in an incubator.
Following incubation, cell viability was determined by measuring
the absorbance at 450 nm using a microplate reader (Bio-Rad).
The EdU reagent kit (Ribobio) was also used to
evaluate cell proliferation based on our previous study (15). Briefly, the OS cells were
transfected with siRNA for 48 h and the medium was then changed
with culture medium to continue culture for 2 and 4 days. The cells
were then incubated with complete medium solution containing 50 M
EdU for 1 h at corresponding time-points. The following procedure
was carried out in accordance with the manufacture's protocol.
Finally, images were acquired with a fluorescence microscope
(Zeiss; Carl Zeiss).
Cell cycle analysis
The OS cells were planted in a cell culture plate
(35 mm) with 3×105 cells and then incubated overnight
until 50-70% confluent. Prior to transfection, the cells were
synchronized for 6 h in serum-free DMEM culture medium. At 48 h
following transfection with siRNA, the cells were trypsinized and
transferred to a 10 ml centrifuge tube (centrifugation at 200 × g
for 5 min at 4°C, washed with cold PBS solution twice, fixed with
70% pre-cold ethanol overnight at −20°C. Subsequently, 190
µl EDTA and 10 µl Rnase A (1 mg/ml) were added to
tubes for 5 min at room temperature, and 18 µl propidium
iodide and 97 µl PBS were then added for 10 min at 4°C in
the dark, respectively. The volume was adjusted to 1 ml with PBS.
Subsequently, the percentage of cells cycle was analyzed using a BD
FACSCan (BD Biosciences).
Wound healing assay
Wound healing assay was performed to estimate the OS
cell migratory activities. The transfected OS cells were plated
into a 6-well plate at a density of 5×105 cells/well and
two horizontal lines were marked on the back of the well plate.
When the confluence of the cells reached 100%, the cells were
scratched with a sterile 100 µl pipet tip, and the cells
debris was then removed using PBS solution. Following the addition
of serum-free medium containing 2% FBS, the cells were placed back
into the incubator (37°C, 5% CO2) for 48 h. The
migration of the cells was photographed under a microscope with Zen
Imaging software at 0, 12, 24 and 48 h after scratching. The
percentage of wound healing was determined as described in our
previous study (16).
Statistical analysis
Statistical analysis was performed using SPSS 21.0
software (IBM). All data are expressed as the means ± SD.
Statistical significance between two groups were analyzed with the
Student's t-test. The significance among multiple groups was using
one-way ANOVA followed by Tukey's post hoc test. P-values <0.05
were considered to indicate statistically significant differences.
Each experiment was carried out in triplicate.
Results
Ski is highly expressed in OS tissues and
cell lines
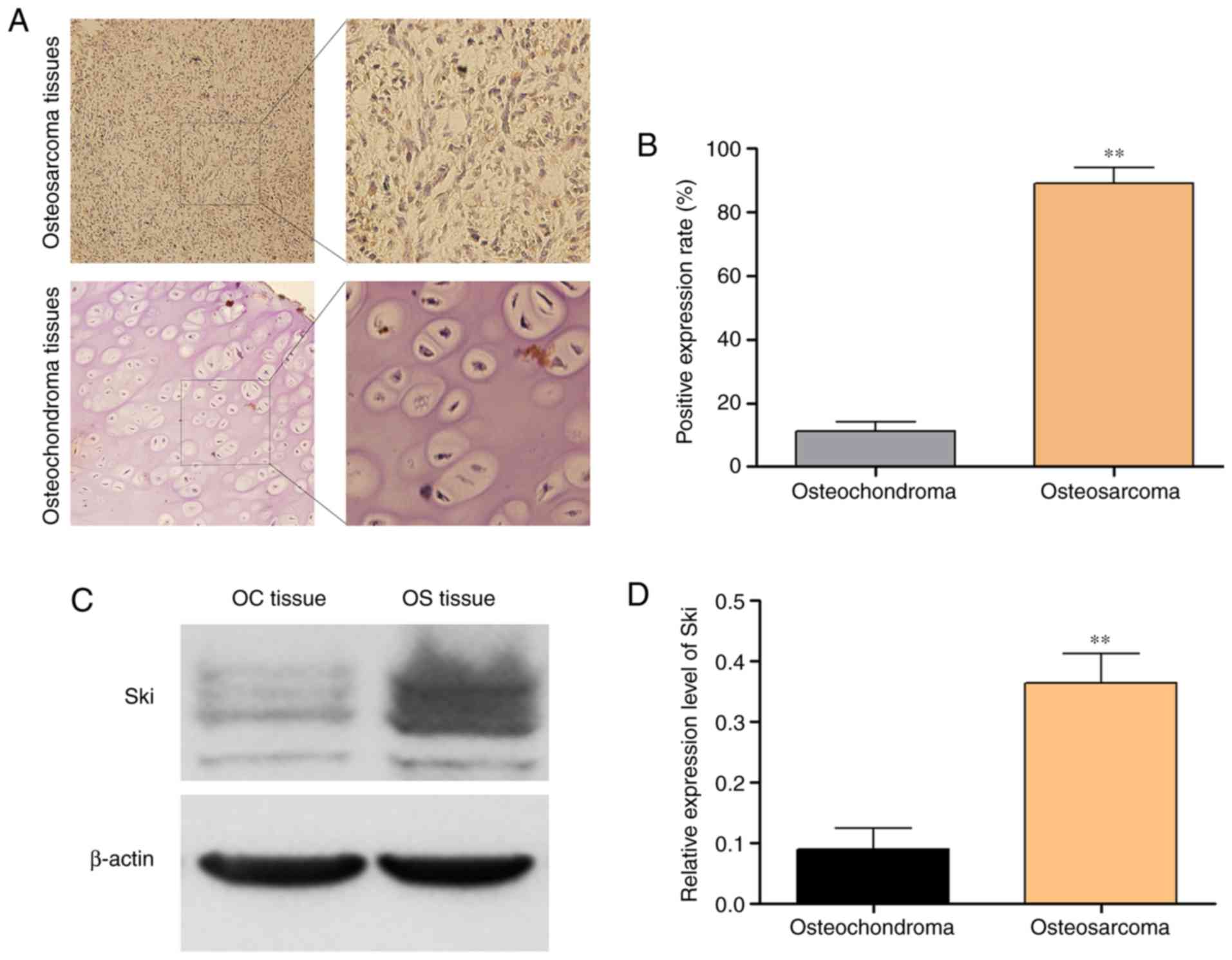
Compared with the osteochondroma tissues, Ski was
more highly expressed in the OS tissues. As shown in Fig. 1A and B, Ski was overexpressed in OS
tissues, whereas its expression was hardly detected in the
osteochondroma tissues by immunohistochemistry. The results of
western blot analysis also demonstrated that Ski (the molecular
weight of Ski varies between 90 and 132 kDa) increased
significantly in the OS sample tissues compared with the
osteochondroma samples (Fig. 1C and
D). These results indicated that Ski may play an essential role
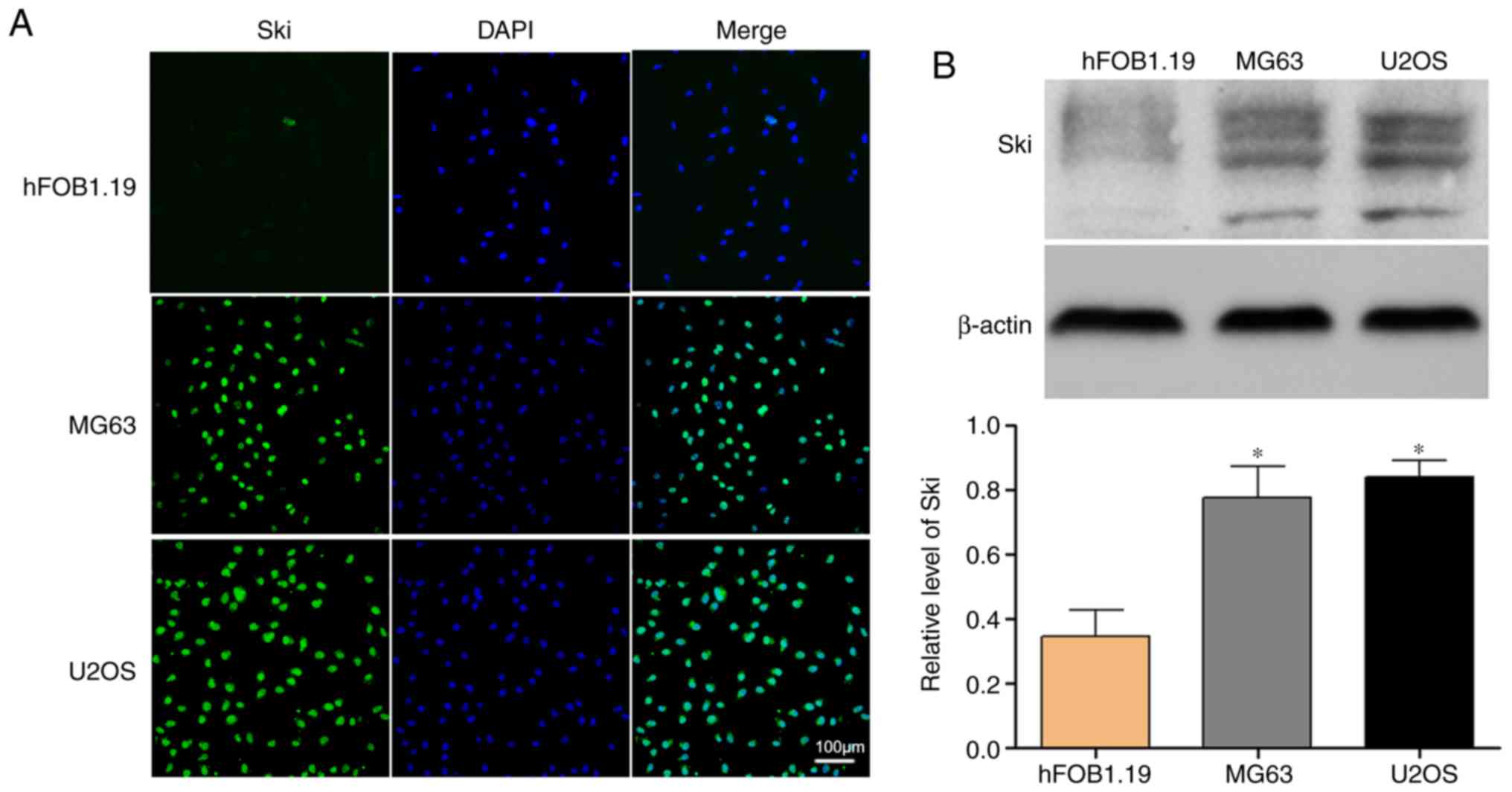
in the tumorigenesis of OS. Thus, we compared Ski expression level
in OS cell lines (MG63 and U2OS) with hFOB1.19 normal osteoblast
cells. The results of immunofluorescence staining revealed that Ski
expression was more abundant in the OS cell lines compared with the
osteoblasts (Fig. 2A).
Furthermore, western blot analysis confirmed the results again
(Fig. 2B).
Ski knockdown decreases the proliferation
of OS cells
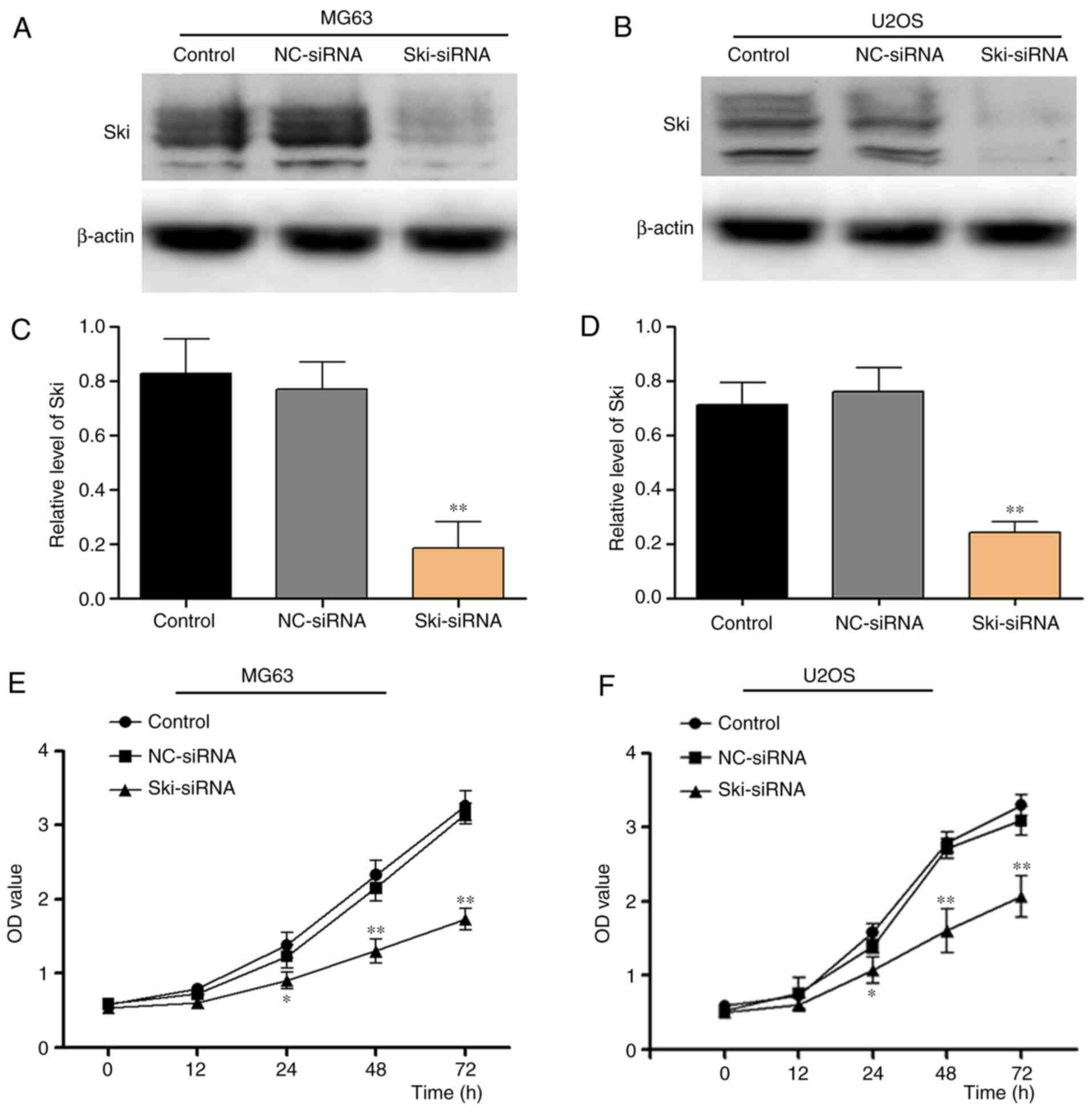
In order to examine the role of Ski in OS cells, a
specific siRNA (Ski-siRNA) was applied in the OS cell lines, MG63
and U2OS cells. The results indicated that Ski expression was
inhibited efficiently in the MG63 and U2OS cells (Fig. 3A and D). Subsequently, CCK-8 assay
was performed to investigate the effects of Ski-siRNA on OS cell
proliferation. The results revealed that OS cell proliferation was
markedly suppressed following transfection with Ski-siRNA at 48 and
72 h in the MG63 cells (P<0.001) compared with the control group
and NC-siRNA group (Fig. 3E), and
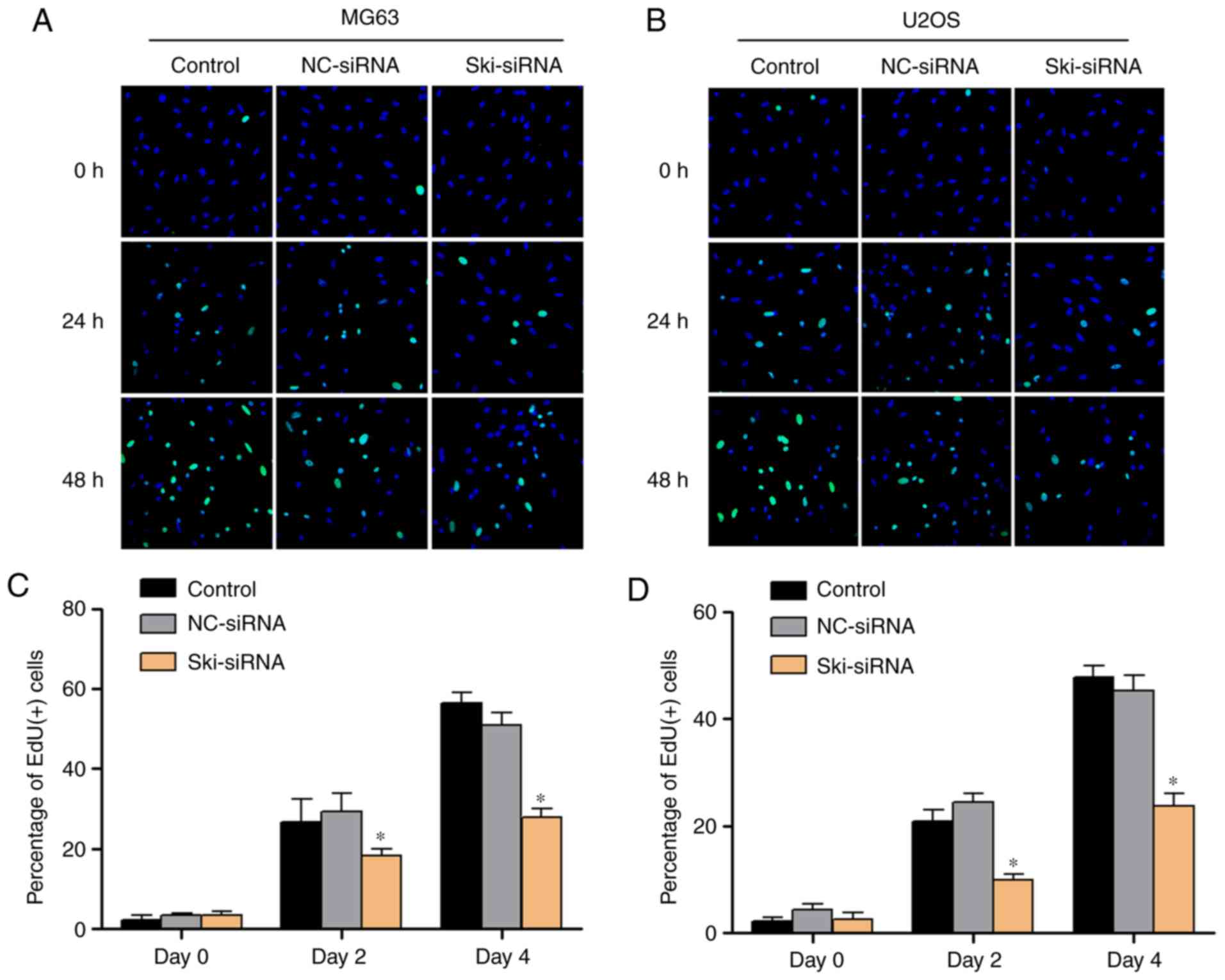
similar results were obtained with the U2-OS cells (Fig. 3F). In addition, EdU kit assay was
applied to investigate the OS cell proliferative ability, and the
results indicated that the MG63 and U2-OS cell proliferative
ability was inhibited significantly following the knockdown of Ski
(Fig. 4A-D). Moreover, to further
confirm the results mentioned above, the proportions of cell cycle
profiles were investigated following transfection with Ski-siRNA by
flow cytometry. The results revealed that, the percentage of MG63
cells at the G1 phase increased significantly, while the percentage
at the G2/M phase decreased in the Ski-siRNA group compared with
the NC-siRNA group (Fig. 4E and
G); however, no significant difference was observed in the
cells in the S phase. Similar results were obtained for the U2OS
cells (Fig. 4F and H).
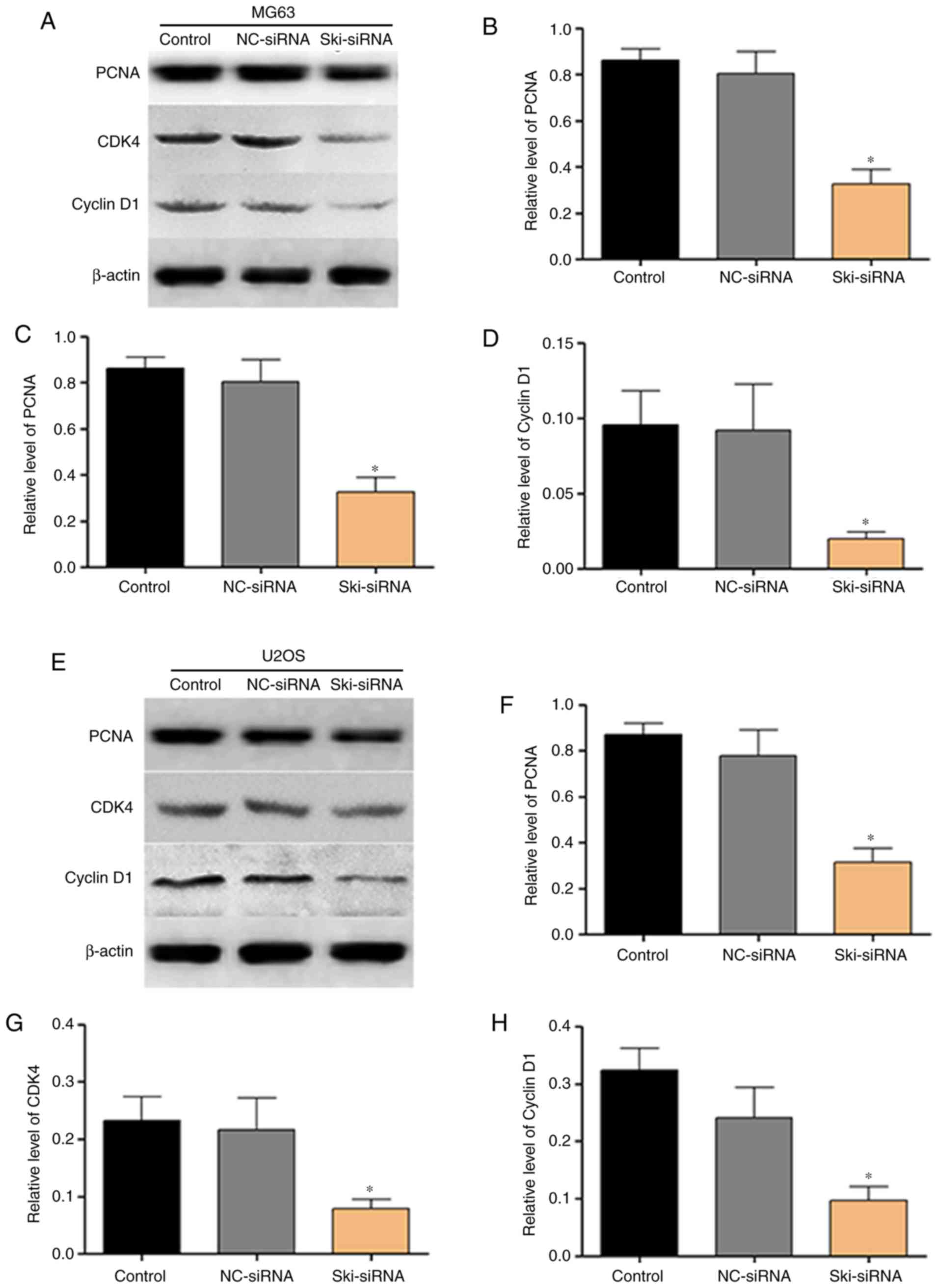
Furthermore, the results of western blot analysis revealed that the
expression of proliferation-related proteins, including PCNA, CDK4
and cyclin D1 decreased significantly following transfection with
Ski-siRNA in OS cells (Fig. 5).
All the above-mentioned results strongly indicate that Ski plays a
crucial role in the proliferation of OS cells.
Ski knockdown decreases the migration of
OS cells
In addition to proliferation, OS cell migration also
plays a critical role in tumor metastasis. Thus, in this study, a
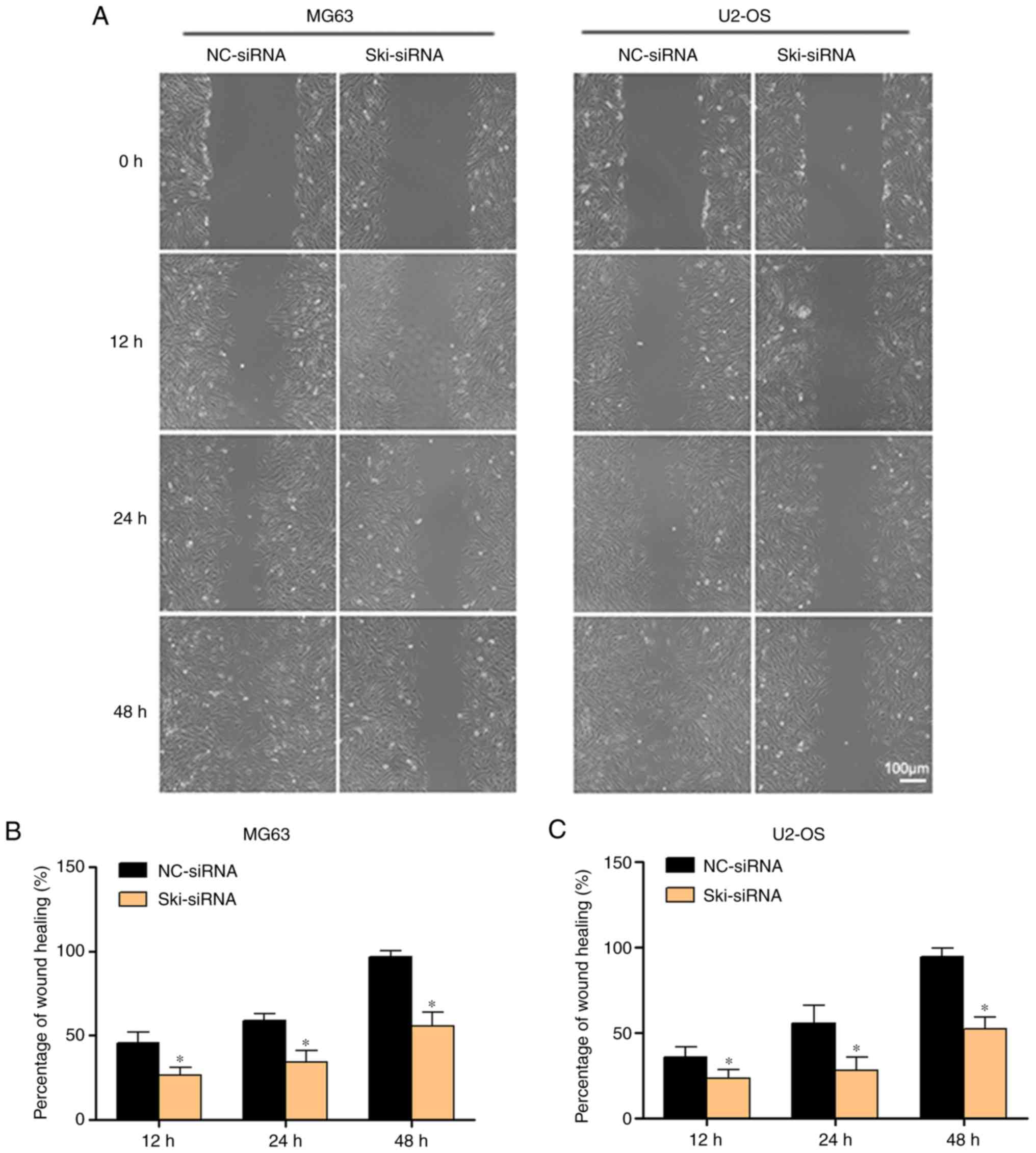
wound healing assay was performed to assess the effects of Ski
knockdown on MG63 and U2OS cell migration. Following the knockdown
of Ski, the percentages of MG63 cells after wound healing at 48 h
in the NC-siRNA group and Ski-siRNA group were 94.47±5.36 and
52.53±6.57%, respectively, and the rate of wound healing in the
Ski-siRNA group was much lower than that of the NC-siRNA group
(Fig. 6A and B); similar results
were obtained for the U2OS cells as well (Fig. 6A and C). In addition, the results
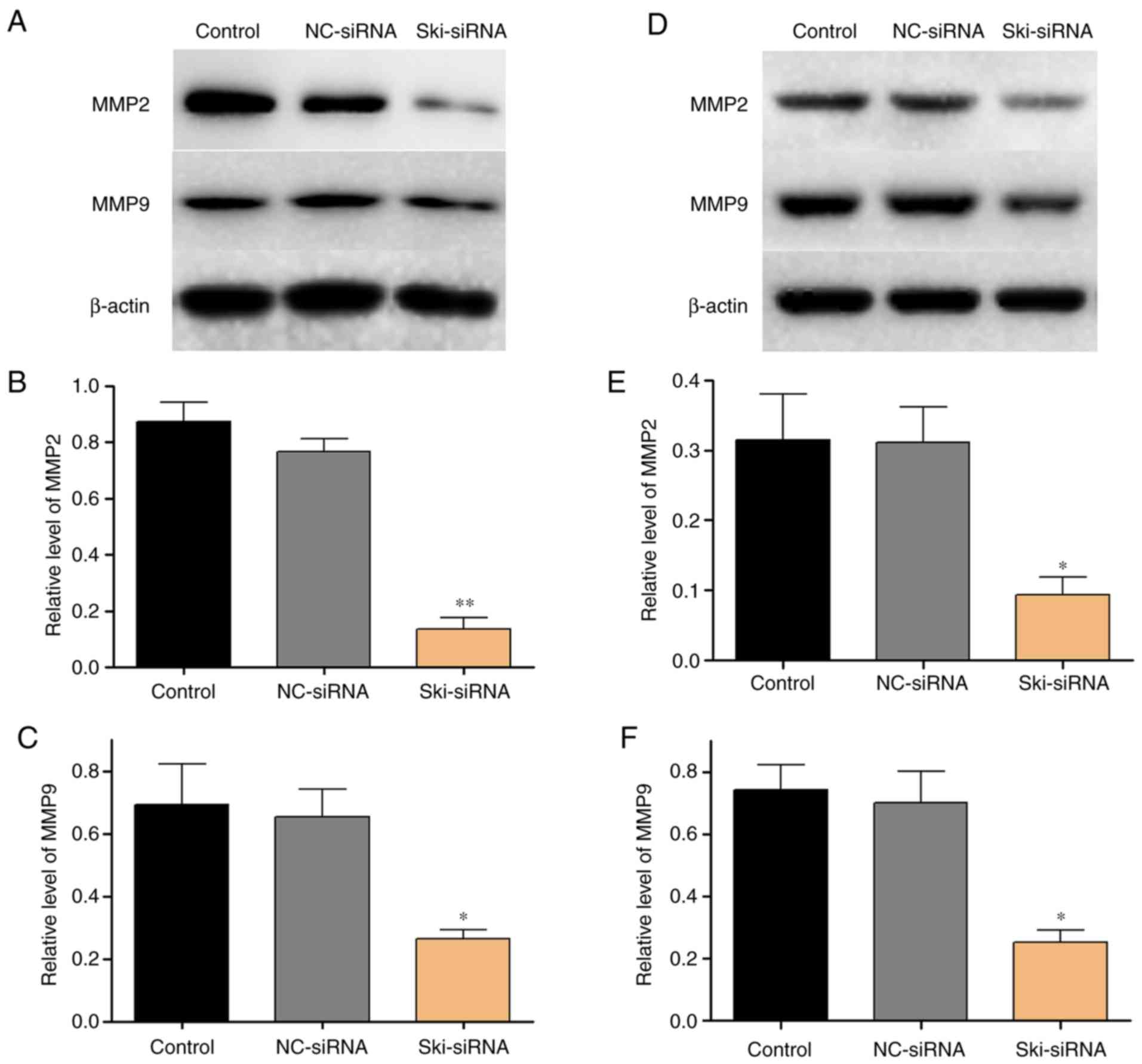
of western blot analysis revealed that the expression of
migration-related proteins, including MMP2 and MMP9 markedly
decreased following the knockdown of Ski (Fig. 7).
Ski knockdown suppresses the activation
of the PI3K/Akt signaling pathway in OS cells
The PI3K/Akt signaling pathway plays an essential
role in tumorigenesis, which has been well expounded as an
essential pathway for the proliferation and migration of OS cells.
Thus, in this study, the effects of Ski on the certain molecules
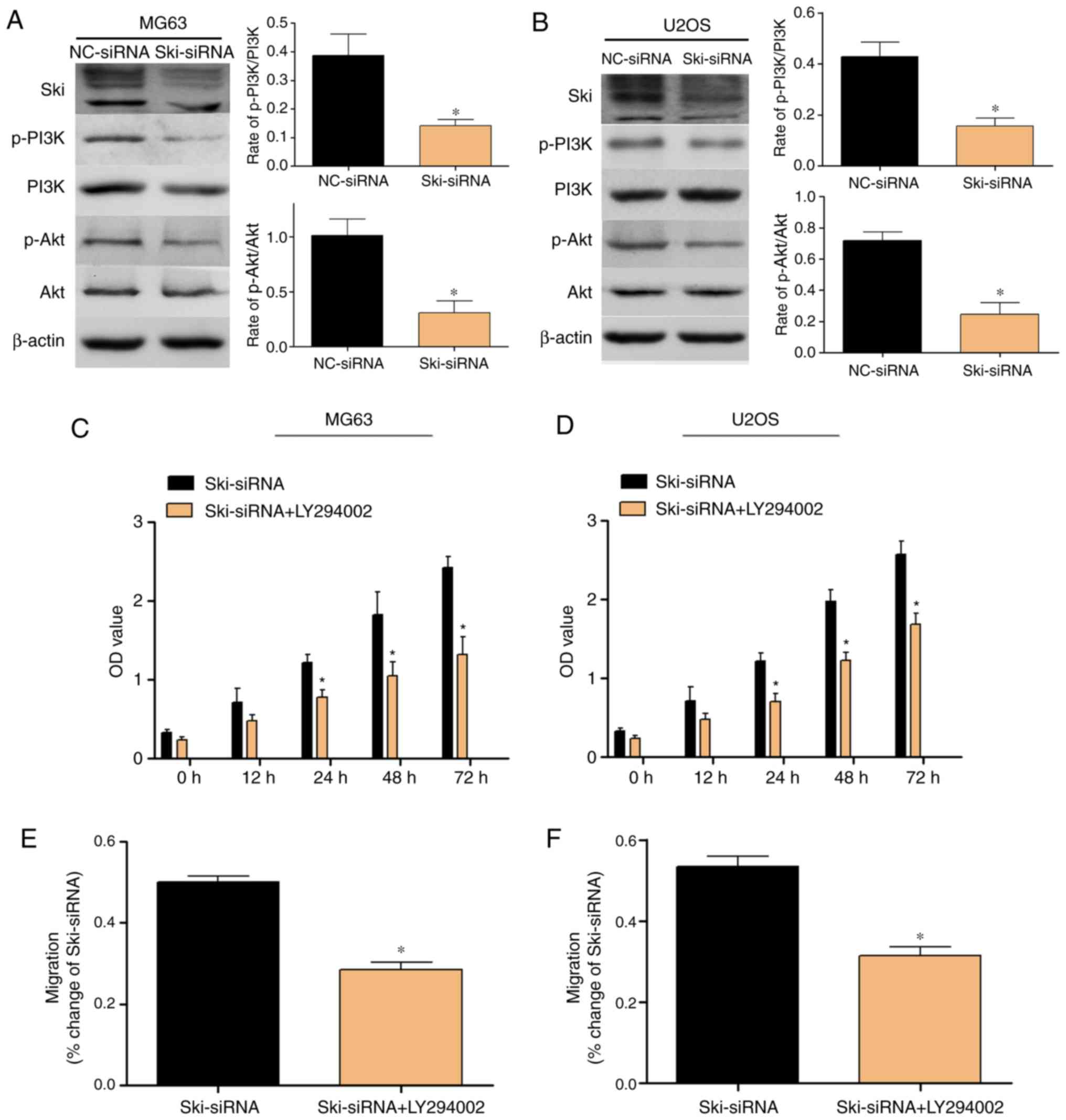
which are involved in the PI3K/Akt signaling pathway in MG63 and
U2-OS cells were examined. The results revealed that the knockdown
of Ski decreased the PI3K and Akt phosphorylation levels
significantly in the MG63 cells compared with the control and
NC-siRNA groups (Fig. 8A). Similar
results were obtained in the U2OS cells (Fig. 8B). Furthermore, to verify the
function of the PI3K/Akt pathway, we examined the effects of the
LY294002 on the Ski-mediated proliferation and migration of OS
cells. The results revealed that LY294002 significantly enhanced
the inhibitory effects of Ski-siRNA on OS cell proliferation and
migration, and these results were further verified by CCK8 assay
(Fig. 8C and D) and wound healing
assay (Fig. 8E and F).
Discussion
OS, as a malignant bone tumor, mainly occurs in
children and young individuals (17,18).
With the development of new therapeutic technologies, the survival
rate of patients with OS has markedly increased. However, the
overall outcome of patients with OS remains poor due to drug or
multidrug resistance (19,20). Thus, the study of the mechanisms
responsible for the development of OS and the identification of
novel effective treatable methods for patients with OS is
crucial.
Ski, as an evolutionary conserved protein, has been
reported to exist in various tissues and species, and participates
in diverse cellular processes, such as cell proliferation,
metastasis, transformation and tumor progressions (6). Previous studies have demonstrated
that Ski plays an essential role in certain pathophysiological
processes, such as wound healing and astrocyte proliferation
(15,21), vascular smooth muscle cell
proliferation (22,23), muscle differentiation (24,25)
and liver regeneration (26).
Notably, Ski, as a novel therapeutic target, has been found over
expression in the development of solid tumors (27). However, the function and roles of
Ski in human OS remain largely unknown. The present study focused
on investigating Ski expression and its roles in OS cell lines.
In this study, we have found that Ski expressed much
more in OS tissues sample compared with osteochondroma samples.
Similarly, Ski expression was also more abundant in MG63 and U2OS
cell lines compared with normal osteoblast cell line. However, the
low sample size was a limitation to the present study. A previous
study demonstrated that the Ski expression level ws markedly
increased during human melanoma tumor progression (13,28).
Wang et al demonstrated that Ski was overexpressed in
pancreatic cancer cell lines and that Ski may act as a tumor
proliferation-promoting factor in pancreatic cancer (29). Combined with our results,
therefore, we highly suspected that Ski may play a vital role in
the pathological process of OS.
To the best of our knowledge, the definite role of
Ski in OS has not yet been extensively studied. This study provides
the first evidence that Ski definitely plays an important role in
OS. In the present study, we demonstrated that the knockdown of Ski
decreased OS cell line proliferation verified by CCK8 assay and EdU
staining assay. Moreover, the expression of
proliferation-association proteins, including PCNA, CDK4 and cyclin
D1 was downregulated in OS cells following transfection with
Ski-siRNA. Furthermore, OS cell cycle arrest in the G1/G0 phase
occurred following the knockdown of Ski. Atanasoski et al
demonstrated that Ski controls the proliferation of Schwann cell
and myelination process (30).
Zhao et al also revealed that Ski plays a vital role in the
proliferation of astrocyte and astrogliosis process (16). Both of these studies demonstrated
that Ski participates in several types of cell proliferation
biological properties. Combined with the findings of the present
study, it is thus proven that Ski is positively associated with the
proliferation of OS cell lines.
OS cell proliferation plays a vital role in tumor
metastasis, while migration is also a critical step for tumor
metastasis (31). In the present
study, OS cell migration markedly decreased following the knockdown
of the Ski gene, as shown by wound healing assays. Therefore, the
results revealed that Ski knockdown suppressed OS cell metastasis.
In addition, MMPs are considered to play an essential role in
collagen degradation, and can promote the migration and invasion of
cancer cells (32,33), thereby exerting a profound effect
on tumor metastasis. In this study, it was found that MMP2 and MMP9
expression levels were significantly decreased following the
knockdown of Ski. Qin et al reported that PAD1 promotes
breast cancer cell metastasis by regulating the ERK1/2/MMP2
signaling pathway (34). Li et
al found that the knockdown of TKTL1 decreased ESCC cell
metastasis by downregulating MMP2 and MMP9 expression (35). Moreover, Arndt et al
reported that Fussel-15, a new member of the Ski family, plays a
vital role in fibroblast migration (36). In this study, it was found that the
knockdown of the Ski gene markedly suppressed the migration in OS
cells, and that the expression of MMP2 and MMP9 decreased
significantly. The above-mentioned data demonstrated that Ski
knockdown significantly decreased OS cell migration by suppressing
MMP2 and MMP9 expression.
The PI3K/Akt pathway plays a critical regulatory
role in tumorigenesis by regulating cell proliferation and
metastasis (37,38). There is evidence to indicate that
the PI3K/Akt pathway is activated in the pathological process of OS
(39,40). The activation of Akt further
phosphorylates multiple proteins that regulate cellular
proliferation and migration (41).
Therefore, inhibiting the phosphorylation of the PI3K/Akt pathway
represents a potential treatment method for OS (42,43).
Thus, inhibiting the phosphorylation of the PI3K/Akt pathway, by
various means, disrupts OS progression. It has been reported that
the PI3K-specific inhibitor, LY294002, markedly suppresses OS cell
proliferation and migration by downregulating the activity of the
PI3K/Akt pathway (44-46). Jiang et al found that the
knockdown of the DDX46 gene inhibited the tumorigenesis of OS cells
by suppressing the phosphorylation of the PI3K/Akt signaling
pathway (47). Chen et al
found that isoliquiritigenin suppressed OS cell proliferation by
down-regulating the PI3K/Akt pathway (48). Similarly, it has been revealed that
TROP promotes OS cell proliferation and migration by activating the
PI3K/Akt signaling pathway (40).
Of note, a previous study demonstrated that there may be a
potential connection between Ski and the PI3K/Akt signaling
pathway; for example, Ski can be phosphorylated by Akt and this
phosphorylation is elevated by the activation of the PI3K/Akt
pathway (49). However, whether
the roles of Ski in OS cells are regulated by the PI3K/Akt pathway
remain unclear.
In the present study, the underlying mechanisms of
the biological functions of Ski, including the proliferation and
migration of OS cell lines were investigated. It was found that Ski
knockdown significantly inhibited the phosphorylation levels of
both PI3K and Akt in OS cell lines. In order to confirm the
association between Ski and the PI3K/Akt signaling pathway in OS
cell lines, an inhibitor of PI3K (LY 294002) was used. In this
study, it was found that LY 294002 significantly enhanced the
inhibitory effects of Ski-siRNA on MG63 cell proliferation and
migration, and similar results were obtained with the U2OS cells.
The above-mentioned results strongly demonstrated that Ski
knockdown notably inhibited OS proliferation and migration by
blocking the PI3K/Akt signaling pathway. To further confirm the
function of Ski in OS, further studies on cell proliferation and
migration trends following Ski gene transfection are warranted and
our team aims to continue to study its role in OS cells.
In conclusion, this study demonstrates for the first
time, to the best of our knowledge, that the expression of Ski was
markedly increased in OS tissues and cell lines, and that Ski
knockdown decreased OS cell proliferation and migration, which was
performed by blocking the PI3K/Akt signaling pathway. It can thus
be concluded that Ski may become a potential therapeutic target
molecule in the treatment of OS.
Funding
This study was supported by The National Natural
Science Foundation of China (grant no. 81672165) and the Science
And Technology Program of Sichuan Province (grant no.
2019YFS0123).
Availability of data and materials
The datasets used and analyzed in the present study
are available from the corresponding author on reasonable
request.
Authors' contributions
XZ and YF were involved in the design of the study,
performed the experiments and drafted the manuscript. XW performed
the experiments. ZY and DL designed and analyzed the
immunohistochemistry data. PK and MT were involved in the design of
the study, and in the critical appraisal of the manuscript. All
authors have read and approved the final version of the manuscript
to be published.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of West China Hospital of Sichuan University (Chengdu,
China). Written informed consent was obtained from all
patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
The authors would like to thank the Research Core
Facility of West China Hospital of Sichuan University for providing
support with the experimental techniques.
References
|
1
|
Lei Z, Duan H, Zhao T, Zhang Y, Li G, Meng
J, Zhang S and Yan W: PARK2 inhibits osteosarcoma cell growth
through the JAK2/STAT3/VEGF signaling pathway. Cell Death Dis.
9:3752018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Morrow JJ and Khanna C: Osteosarcoma
genetics and epigenetics: Emerging biology and candidate therapies.
Crit Rev Oncog. 20:173–197. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Na KY, Bacchini P, Bertoni F, Kim YW and
Park YK: Syndecan-4 and fibronectin in osteosarcoma. Pathology.
44:325–330. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lilic V, Lilic G, Filipovic S, Milosevic
J, Tasic M and Stojiljkovic M: Modern treatment of invasive
carcinoma of the uterine cervix. J BUON. 14:587–592. 2009.
|
|
5
|
Carnelio S, Pai K, Rao N, Solomon M and
Ahasan A: Metastatic osteosarcoma to the maxilla: A case report and
a review of the literature. Quintessence Int. 33:397–399.
2002.PubMed/NCBI
|
|
6
|
Bonnon C and Atanasoski S: c-Ski in health
and disease. Cell Tissue Res. 347:51–64. 2012. View Article : Google Scholar
|
|
7
|
Li Y, Turck CM, Teumer JK and Stavnezer E:
Unique sequence, ski, in sloankettering avian retroviruses with
properties of a new cell-derived oncogene. J Virol. 57:1065–1072.
1986.PubMed/NCBI
|
|
8
|
Luo K, Stroschein SL, Wang W, Chen D,
Martens E, Zhou S and Zhou Q: The Ski oncoprotein interacts with
the Smad proteins to repress TGFbeta signaling. Genes Dev.
13:2196–2206. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Villanacci V, Bellone G, Battaglia E,
Rossi E, Carbone A, Prati A, Verna C, Niola P, Morelli A, Grassini
M and Bassotti G: Ski/SnoN expression in the sequence
metaplasia-dysplasia-adenocarcinoma of Barrett's esophagus. Hum
Pathol. 39:403–409. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Buess M, Terracciano L, Reuter J,
Ballabeni P, Boulay JL, Laffer U, Metzger U, Herrmann R and
Rochlitz C: Amplification of SKI is a prognostic marker in early
colorectal cancer. Neoplasia. 6:207–212. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Nakao T, Kurita N, Komatsu M, Yoshikawa K,
Iwata T, Utsunomiya T and Shimada M: Expression of thrombospondin-1
and Ski are prognostic factors in advanced gastric cancer. Int J
Clin Oncol. 16:145–152. 2011. View Article : Google Scholar
|
|
12
|
Heider TR, Lyman S, Schoonhoven R and
Behrns KE: Ski promotes tumor growth through abrogation of
transforming growth factor-beta signaling in pancreatic cancer. Ann
Surg. 246:61–68. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chen D, Lin Q, Box N, Roop D, Ishii S,
Matsuzaki K, Fan T, Hornyak TJ, Reed JA, Stavnezer E, et al: SKI
knockdown inhibits human melanoma tumor growth in vivo. Pigment
Cell Melanoma Res. 22:761–772. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fumagalli S, Doneda L, Nomura N and
Larizza L: Expression of the c-ski proto-oncogene in human melanoma
cell lines. Melanoma Res. 3:23–27. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhao X, Zhou KS, Li ZH, Nan W, Wang J, Xia
YY and Zhang HH: Knockdown of Ski decreased the reactive astrocytes
proliferation in vitro induced by oxygen-glucose
deprivation/reoxygenation. J Cell Biochem. 119:4548–4558. 2018.
View Article : Google Scholar
|
|
16
|
Zhao X, Wang XW, Zhou KS, Nan W, Guo YQ,
Kou JL, Wang J, Xia YY and Zhang HH: Expression of Ski and its role
in astro-cyte proliferation and migration. Neuroscience. 362:1–12.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang Z, Sun X, Bao Y, Mo J, Du H, Hu J and
Zhang X: E2F1 silencing inhibits migration and invasion of
osteosarcoma cells via regulating DDR1 expression. Int J Oncol.
51:1639–1650. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Jiang W, Yu Y, Liu J, Zhao Q, Wang J,
Zhang J and Dang X: Downregulation of Cdc6 inhibits tumorigenesis
of osteosarcoma in vivo and in vitro. Biomed Pharmacother.
115:1089492019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen R, Huang LH, Gao YY, Yang JZ and Wang
Y: Identification of differentially expressed genes in MG63
osteosarcoma cells with drug-resistance by microarray analysis. Mol
Med Rep. 19:1571–1580. 2019.
|
|
20
|
Chen R, Wang G, Zheng Y, Hua Y and Cai Z:
Drug resistance-related microRNAs in osteosarcoma: Translating
basic evidence into therapeutic strategies. J Cell Mol Med.
23:2280–2292. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kokura K, Kim H, Shinagawa T, Khan MM,
Nomura T and Ishii S: The Ski-binding protein C184M negatively
regulates tumor growth factor-beta signaling by sequestering the
Smad proteins in the cytoplasm. J Biol Chem. 278:20133–20139. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li J, Li P, Zhang Y, Li GB, Zhou YG, Yang
K and Dai SS: c-Ski inhibits the proliferation of vascular smooth
muscle cells via suppressing Smad3 signaling but stimulating p38
pathway. Cell Signal. 25:159–167. 2013. View Article : Google Scholar
|
|
23
|
Li J, Zhao L, He X, Yang T and Yang K:
MiR-21 inhibits c-Ski signaling to promote the proliferation of rat
vascular smooth muscle cells. Cell Signal. 26:724–729. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Colmenares C and Stavnezer E: The ski
oncogene induces muscle differentiation in quail embryo cells.
Cell. 59:293–303. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang H and Stavnezer E: Ski regulates
muscle terminal differentiation by transcriptional activation of
Myog in a complex with Six1 and Eya3. J Biol Chem. 284:2867–2879.
2009. View Article : Google Scholar :
|
|
26
|
Macias-Silva M, Li W, Leu JI, Crissey MA
and Taub R: Up-regulated transcriptional repressors SnoN and Ski
bind Smad proteins to antagonize transforming growth factor-beta
signals during liver regeneration. J Biol Chem. 277:28483–28490.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang L, Hou Y, Sun Y, Zhao L, Tang X, Hu
P, Yang J, Zeng Z, Yang G, Cui X and Liu M: c-Ski activates
cancer-associated fibroblasts to regulate breast cancer cell
invasion. Mol Oncol. 7:1116–1128. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Reed JA, Lin Q, Chen D, Mian IS and
Medrano EE: SKI pathways inducing progression of human melanoma.
Cancer Metastasis Rev. 24:265–272. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang P, Chen Z, Meng ZQ, Fan J, Luo JM,
Liang W, Lin JH, Zhou ZH, Chen H, Wang K, et al: Dual role of Ski
in pancreatic cancer cells: Tumor-promoting versus
metastasis-suppressive function. Carcinogenesis. 30:1497–1506.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Atanasoski S, Notterpek L, Lee HY,
Castagner F, Young P, Ehrengruber MU, Meijer D, Sommer L, Stavnezer
E, Colmenares C and Suter U: The protooncogene Ski controls schwann
cell proliferation and myelination. Neuron. 43:499–511. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Huang J, Dey R, Wang Y, Jakoncic J,
Kurinov I and Huang XY: Structural insights into the induced-fit
inhibition of fascin by a small-molecule inhibitor. J Mol Biol.
430:1324–1335. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Cao LL, Pei XF, Qiao X, Yu J, Ye H, Xi CL,
Wang PY and Gong ZL: SERPINA3 silencing inhibits the migration,
invasion, and liver metastasis of colon cancer cells. Dig Dis Sci.
63:2309–2319. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
de Oliveira Poswar F, de Carvalho Fraga
CA, Gomes ES, Farias LC, Souza LW, Santos SH, Gomez RS, de-Paula AM
and Guimarães AL: Protein expression of MMP-2 and MT1-MMP in
actinic keratosis, squamous cell carcinoma of the skin, and basal
cell carcinoma. Int J Surg Pathol. 23:20–25. 2015. View Article : Google Scholar
|
|
34
|
Qin H, Liu X, Li F, Miao L, Li T, Xu B, An
X, Muth A, Thompson PR, Coonrod SA and Zhang X: PAD1 promotes
epithelial-mesenchymal transition and metastasis in triple-negative
breast cancer cells by regulating MEK1-ERK1/2-MMP2 signaling.
Cancer Lett. 409:30–41. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Li J, Zhu SC, Li SG, Zhao Y, Xu JR and
Song CY: TKTL1 promotes cell proliferation and metastasis in
esophageal squamous cell carcinoma. Biomed Pharmacother. 74:71–76.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Arndt S, Schmidt J, Wacker E, Karrer S and
Bosserhoff AK: Fussel-15, a new player in wound healing, is
deregulated in keloid and localized scleroderma. Am J Pathol.
178:2622–2631. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Brader S and Eccles SA: Phosphoinositide
3-kinase signalling pathways in tumor progression, invasion and
angiogenesis. Tumori. 90:2–8. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhang J, Yu XH, Yan YG, Wang C and Wang
WJ: PI3K/Akt signaling in osteosarcoma. Clin Chim Acta.
444:182–192. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Cohen-Solal KA, Boregowda RK and Lasfar A:
RUNX2 and the PI3K/AKT axis reciprocal activation as a driving
force for tumor progression. Mol Cancer. 14:1372015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Gu QZ, Nijiati A, Gao X, Tao KL, Li CD,
Fan XP and Tian Z: TROP2 promotes cell proliferation and migration
in osteosarcoma through PI3K/AKT signaling. Mol Med Rep.
18:1782–1788. 2018.PubMed/NCBI
|
|
41
|
Zhang Y, Cheng H, Li W, Wu H and Yang Y:
Highly-expressed P2X7 receptor promotes growth and metastasis of
human HOS/MNNG osteosarcoma cells via PI3K/Akt/GSK3β/β-catenin and
mTOR/HIF1α/VEGF signaling. Int J Cancer. 145:1068–1082. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ma J, Huang H, Han Z, Zhu C and Yue B:
RLN2 is a positive regulator of Akt-2-induced gene expression
required for osteosarcoma cells invasion and chemoresistance.
Biomed Res Int. 2015:1474682015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wang H, Luo QF, Peng AF, Long XH, Wang TF,
Liu ZL, Zhang GM, Zhou RP, Gao S, Zhou Y and Chen WZ: Positive
feedback regulation between Akt phosphorylation and fatty acid
synthase expression in osteosarcoma. Int J Mol Med. 33:633–639.
2014. View Article : Google Scholar
|
|
44
|
Liu T, Zhou W, Cai B, Chu J, Shi G, Teng
H, Xu J, Xiao J and Wang Y: IRX2-mediated upregulation of MMP-9 and
VEGF in a PI3K/AKT-dependent manner. Mol Med Rep. 12:4346–4351.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Long XH, Zhong ZH, Peng AF, Zhu LB, Wang
H, Zhang GM and Liu ZL: LY294002 suppresses the malignant phenotype
and sensitizes osteosarcoma cells to pirarubicin chemotherapy. Mol
Med Rep. 10:2967–2972. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhou Y, Zhu LB, Peng AF, Wang TF, Long XH,
Gao S, Zhou RP and Liu ZL: LY294002 inhibits the malignant
phenotype of osteosarcoma cells by modulating the
phosphatidylinositol 3-kinase/Akt/fatty acid synthase signaling
pathway in vitro. Mol Med Rep. 11:1352–1357. 2015. View Article : Google Scholar
|
|
47
|
Jiang F, Zhang D, Li G and Wang X:
Knockdown of DDX46 inhibits the invasion and tumorigenesis in
osteosarcoma cells. Oncol Res. 25:417–425. 2017. View Article : Google Scholar
|
|
48
|
Chen J, Liu C, Yang QQ, Ma RB, Ke Y, Dong
F and Wu XE: Isoliquiritigenin suppresses osteosarcoma U2OS cell
proliferation and invasion by regulating the PI3K/Akt signalling
pathway. Chemotherapy. 63:155–161. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Band AM, Björklund M and Laiho M: The
phosphatidylinositol 3-kinase/Akt pathway regulates transforming
growth factor-{beta} signaling by destabilizing ski and inducing
Smad7. J Biol Chem. 284:35441–35449. 2009. View Article : Google Scholar : PubMed/NCBI
|