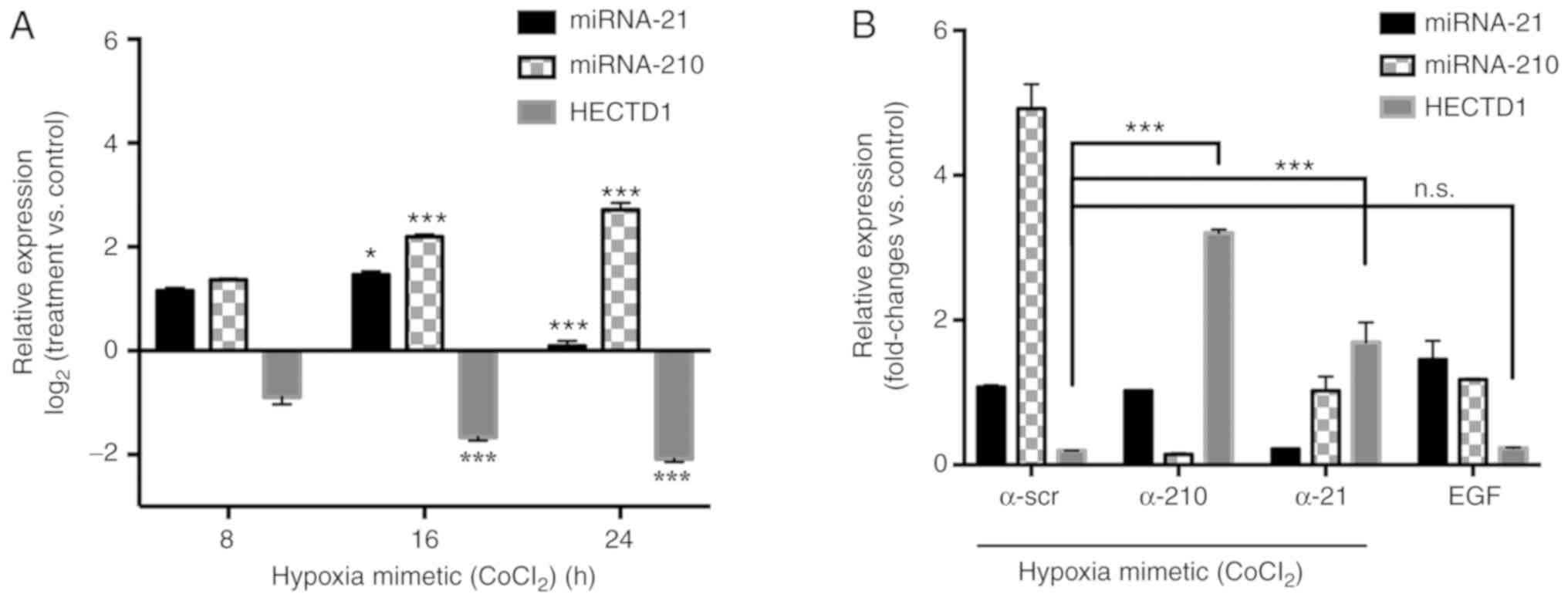
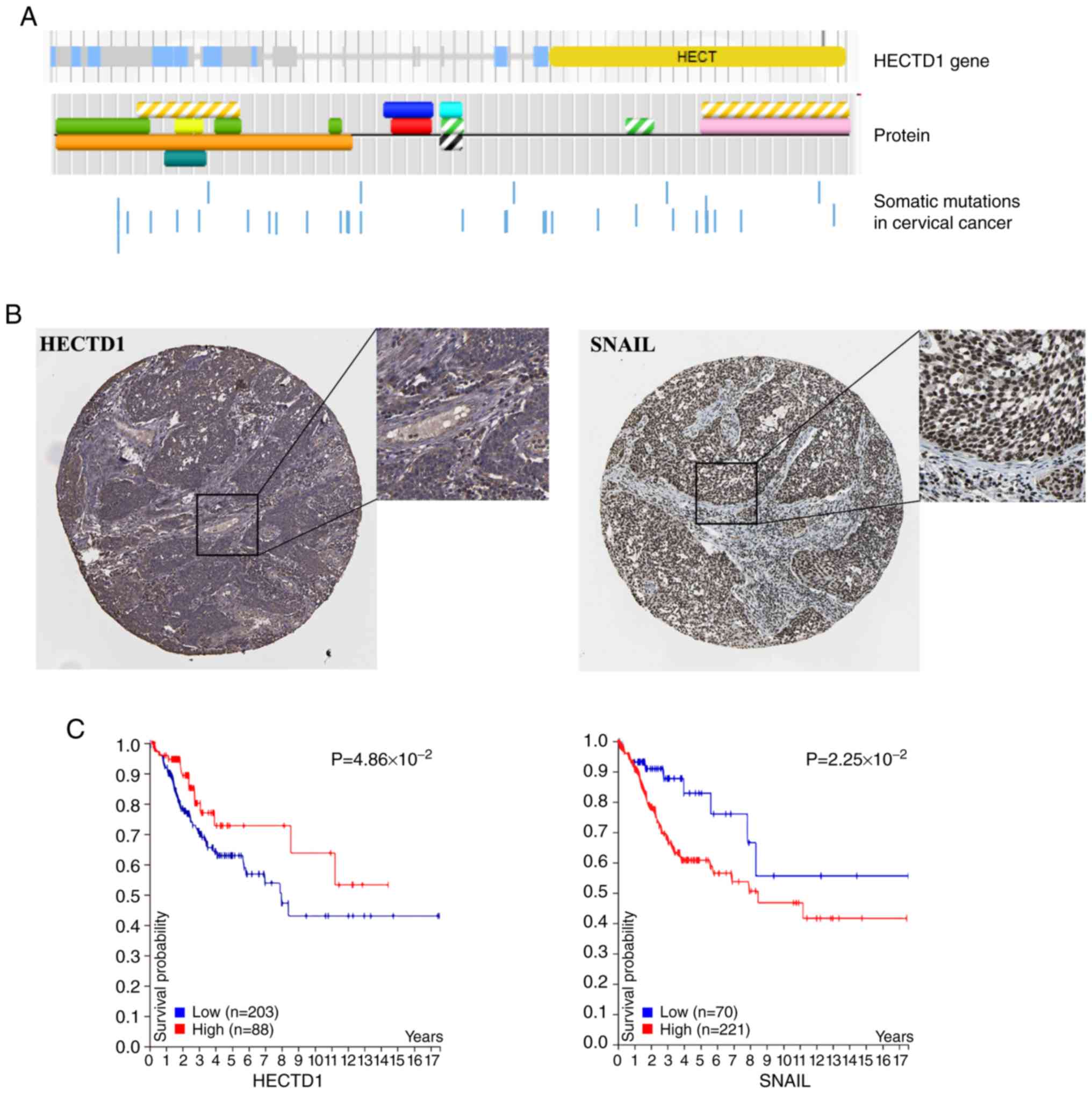
|
1
|
Sahu SK, Tiwari N, Pataskar A, Zhuang Y,
Borisova M, Diken M, Strand S, Beli P and Tiwari VK: FBXO32
promotes microenvironment underlying epithelial-mesenchymal
transition via CtBP1 during tumour metastasis and brain
development. Nat Commun. 8:15232017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Liu Y, Zhou H, Zhu R, Ding F, Li Y, Cao X
and Liu Z: SPSB3 targets SNAIL for degradation in GSK-3beta
phosphorylation-dependent manner and regulates metastasis.
Oncogene. 37:768–776. 2018. View Article : Google Scholar
|
|
3
|
Park SH, Jung EH, Kim GY, Kim BC, Lim JH
and Woo CH: Itch E3 ubiquitin ligase positively regulates TGF-β
signaling to EMT via smad7 ubiquitination. Mol Cells. 38:20–25.
2015. View Article : Google Scholar
|
|
4
|
Van Laar RK: Genomic signatures for
predicting survival and adjuvant chemotherapy benefit in patients
with non-small-cell lung cancer. BMC Med Genomics. 5:302012.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lim SK, Lu SY, Kang SA, Tan HJ, Li Z,
Adrian Wee ZN, Guan JS, Reddy Chichili VP, Sivaraman J, Putti T, et
al: Wnt signaling promotes breast cancer by blocking ITCH-Mediated
degradation of YAP/TAZ transcriptional coactivator WBP2. Cancer
Res. 76:6278–6289. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhou H, Liu Y, Zhu R, Ding F, Wan Y, Li Y
and Liu Z: FBXO32 suppresses breast cancer tumorigenesis through
targeting KLF4 to proteasomal degradation. Oncogene. 36:3312–3321.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Komander D: The emerging complexity of
protein ubiquitination. Biochem Soc Trans. 37:937–953. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pickart CM and Eddins MJ: Ubiquitin:
Structures, functions, mechanisms. Biochim Biophys Acta.
1695:55–72. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Metzger MB, Hristova VA and Weissman AM:
HECT and RING finger families of E3 ubiquitin ligases at a glance.
J Cell Sci. 125:531–537. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Scheffner M and Kumar S: Mammalian HECT
ubiquitin-protein ligases: Biological and pathophysiological
aspects. Biochim Biophys Acta. 1843:61–74. 2014. View Article : Google Scholar
|
|
11
|
D'Alonzo D, Emch FH, Shen X, Bruder E, De
Geyter C and Zhang H: Hectd1 is essential for embryogenesis in
mice. Gene Expr Patterns. 10:1190642019. View Article : Google Scholar
|
|
12
|
Shen X, Jia Z, D'Alonzo D, Wang X, Bruder
E, Emch FH, De Geyter C and Zhang H: HECTD1 controls the protein
level of IQGAP1 to regulate the dynamics of adhesive structures.
Cell Commun Signal. 15:22017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhu S, Si ML, Wu H and Mo YY: MicroRNA-21
targets the tumor suppressor gene tropomyosin 1 (TPM1). J Biol
Chem. 282:14328–14336. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Besnier M, Gasparino S, Vono R, Sangalli
E, Facoetti A, Bollati V, Cantone L, Zaccagnini G, Maimone B,
Fuschi P, et al: MiR-210 enhances the therapeutic potential of
bone-marrow-derived circulating proangiogenic cells in the setting
of limb ischemia. Mol Ther. 26:1694–1705. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fan Q, Hu X, Zhang H, Wang S, Zhang H, You
C, Zhang CY, Liang H, Chen X and Ba Y: MiR-193a-3p is an important
tumour suppressor in lung cancer and directly targets KRAS. Cell
Physiol Biochem. 44:1311–1324. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
17
|
Kaundal R and Raghava GP: RSLpred: An
integrative system for predicting subcellular localization of rice
proteins combining compositional and evolutionary information.
Proteomics. 9:2324–2342. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Nakai K and Horton P: PSORT: A program for
detecting sorting signals in proteins and predicting their
subcellular localization. Trends Biochem Sci. 24:34–36. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Vinas-Castells R, Frias A, Robles-Lanuza
E, Zhang K, Longmore GD, García de Herreros A and Díaz VM: Nuclear
ubiquitination by FBXL5 modulates Snail1 DNA binding and stability.
Nucleic Acids Res. 42:1079–1094. 2014. View Article : Google Scholar :
|
|
20
|
Zheng H, Shen M, Zha YL, Li W, Wei Y,
Blanco MA, Ren G, Zhou T, Storz P, Wang HY and Kang Y: PKD1
phosphory-lation-dependent degradation of SNAIL by SCF-FBXO11
regulates epithelial-mesenchymal transition and metastasis. Cancer
Cell. 26:358–373. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhou BP, Deng J, Xia W, Xu J, Li YM,
Gunduz M and Hung MC: Dual regulation of snail by
GSK-3beta-mediated phosphorylation in control of
epithelial-mesenchymal transition. Nat Cell Biol. 6:931–940. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hutten S and Kehlenbach RH: CRM1-mediated
nuclear export: To the pore and beyond. Trends Cell Biol.
17:193–201. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wolff B, Sanglier JJ and Wang Y:
Leptomycin B is an inhibitor of nuclear export: Inhibition of
nucleo-cytoplasmic translocation of the human immunodeficiency
virus type 1 (HIV-1) Rev protein and Rev-dependent mRNA. Chem Biol.
4:139–147. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chaudhry P, Fabi F, Singh M, Parent S,
Leblanc V and Asselin E: Prostate apoptosis response-4 mediates
TGF-β-induced epithelial-to-mesenchymal transition. Cell Death Dis.
5:e10442014. View Article : Google Scholar
|
|
25
|
Schrevel M, Gorter A, Kolkman-Uljee SM,
Trimbos JB, Fleuren GJ and Jordanova ES: Molecular mechanisms of
epidermal growth factor receptor overexpression in patients with
cervical cancer. Mod Pathol. 24:720–728. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kusewitt DF, Choi C, Newkirk KM, Leroy P,
Li Y, Chavez MG and Hudson LG: Slug/Snai2 is a downstream mediator
of epidermal growth factor receptor-stimulated reepithelialization.
J Invest Dermatol. 129:491–495. 2009. View Article : Google Scholar
|
|
27
|
Zeisberg M and Neilson EG: Biomarkers for
epithelial-mesen-chymal transitions. J Clin Invest. 119:1429–1437.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lee MY, Chou CY, Tang MJ and Shen MR:
Epithelial-Mesenchymal transition in cervical cancer: Correlation
with tumor progression, epidermal growth factor receptor
overexpression, and snail up-regulation. Clin Cancer Res.
14:4743–4750. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gan Y, Shi C, Inge L, Hibner M, Balducci J
and Huang Y: Differential roles of ERK and Akt pathways in
regulation of EGFR-mediated signaling and motility in prostate
cancer cells. Oncogene. 29:4947–4958. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Misra A, Pandey C, Sze SK and Thanabalu T:
Hypoxia activated EGFR signaling induces epithelial to mesenchymal
transition (EMT). PLoS One. 7:e497662012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zavadil J, Narasimhan M, Blumenberg M and
Schneider RJ: Transforming growth factor-beta and microRNA:MRNA
regulatory networks in epithelial plasticity. Cells Tissues Organs.
185:157–161. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ding L, Zhao L, Chen W, Liu T, Li Z and Li
X: MiR-210, a modulator of hypoxia-induced epithelial-mesenchymal
transition in ovarian cancer cell. Int J Clin Exp Med. 8:2299–2307.
2015.PubMed/NCBI
|
|
34
|
Fasanaro P, Greco S, Lorenzi M, Pescatori
M, Brioschi M, Kulshreshtha R, Banfi C, Stubbs A, Calin GA and Ivan
M: An integrated approach for experimental target identification of
hypoxia-induced miR-210. J Biol Chem. 284:35134–35143. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Cicchillitti L, Di Stefano V, Isaia E,
Crimaldi L, Fasanaro P, Ambrosino V, Antonini A, Capogrossi MC,
Gaetano C, Piaggio G and Martelli F: Hypoxia-Inducible factor 1-α
induces miR-210 in normoxic differentiating myoblasts. J Biol Chem.
287:44761–44771. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Forbes SA, Tang G, Bindal N, Bamford S,
Dawson E, Cole C, Kok CY, Jia M, Ewing R, Menzies A, et al: COSMIC
(the Catalogue of Somatic Mutations in Cancer): A resource to
investigate acquired mutations in human cancer. Nucleic Acids Res.
38:D652–D657. 2010. View Article : Google Scholar :
|
|
37
|
Tomczak K, Czerwinska P and Wiznerowicz M:
The cancer genome atlas (TCGA): An immeasurable source of
knowledge. Contemp Oncol (Pozn). 19:A68–A77. 2015.
|
|
38
|
Uhlen M, Zhang C, Lee S, Sjöstedt E,
Fagerberg L, Bidkhori G, Benfeitas R, Arif M, Liu Z, Edfors F, et
al: A pathology atlas of the human cancer transcriptome. Science.
357:63522017. View Article : Google Scholar
|
|
39
|
Lanczky A, Nagy A, Bottai G, Munkácsy G,
Szabó A, Santarpia L and Győrffy B: MiRpower: A web-tool to
validate survival-associated miRNAs utilizing expression data from
2178 breast cancer patients. Breast Cancer Res Treat. 160:439–446.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Rao Q, Shen Q, Zhou H, Peng Y, Li J and
Lin Z: Aberrant microRNA expression in human cervical carcinomas.
Med Oncol. 29:1242–1248. 2012. View Article : Google Scholar
|
|
41
|
Zohn IE, Anderson KV and Niswander L: The
hectd1 ubiquitin ligase is required for development of the head
mesenchyme and neural tube closure. Dev Biol. 306:208–221. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Hugel S, Depping R, Dittmar G, Rother F,
Cabot R, Sury MD, Hartmann E and Bader M: Identification of
importin α 7 specific transport cargoes using a proteomic screening
approach. Mol Cell Proteomics. 13:1286–1298. 2014. View Article : Google Scholar
|
|
43
|
Zada S, Hwang JS, Ahmed M, Lai TH, Pham TM
and Kim DR: Control of the epithelial-to-mesenchymal transition and
cancer metastasis by autophagy-dependent SNAI1 Degradation. Cells.
8:E1292019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Liu G, Rogers J, Murphy CT and Rongo C:
EGF signalling activates the ubiquitin proteasome system to
modulate C. elegans lifespan. EMBO J. 30:2990–3003. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Argenzio E, Bange T, Oldrini B, Bianchi F,
Peesari R, Mari S, Di Fiore PP, Mann M and Polo S: Proteomic
snapshot of the EGF-induced ubiquitin network. Mol Syst Biol.
7:4622011. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Piret JP, Mottet D, Raes M and Michiels C:
CoCl2, a chemical inducer of hypoxia-inducible factor-1, and
hypoxia reduce apop-totic cell death in hepatoma cell line HepG2.
Annals N Y Acad Sci. 973:443–447. 2002. View Article : Google Scholar
|
|
47
|
Djidja MC, Chang J, Hadjiprocopis A,
Schmich F, Sinclair J, Mršnik M, Schoof EM, Barker HE, Linding R,
Jørgensen C and Erler JT: Identification of hypoxia-regulated
proteins using MALDI-mass spectrometry imaging combined with
quantitative proteomics. J Proteome Res. 13:2297–2313. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Lester RD, Jo M, Montel V, Takimoto S and
Gonias SL: UPAR induces epithelial-mesenchymal transition in
hypoxic breast cancer cells. J Cell Biol. 178:425–436. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Duhamel S, Goyette MA, Thibault MP, Filion
D, Gaboury L and Cote JF: The E3 ubiquitin ligase HectD1 suppresses
EMT and metastasis by targeting the +TIP ACF7 for degradation. Cell
Rep. 22:1016–1030. 2018. View Article : Google Scholar : PubMed/NCBI
|