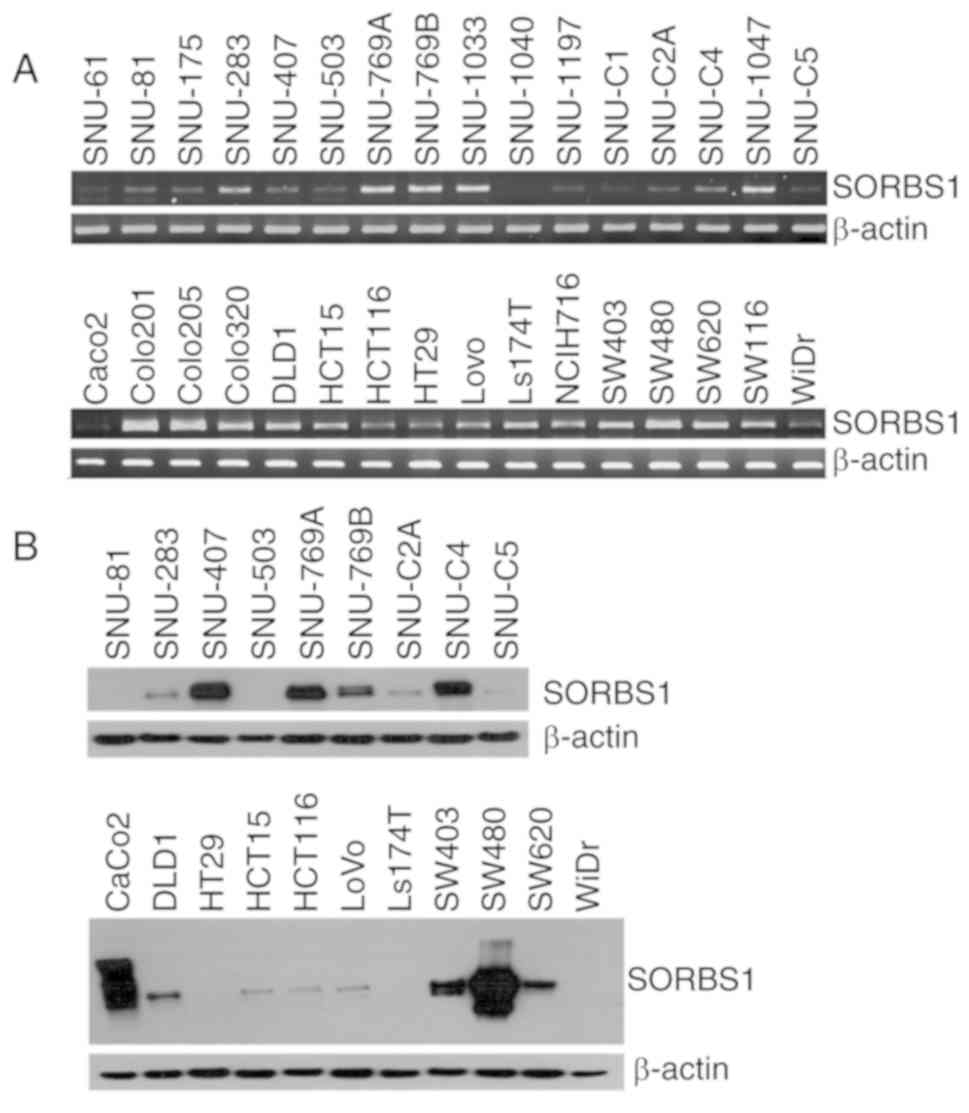
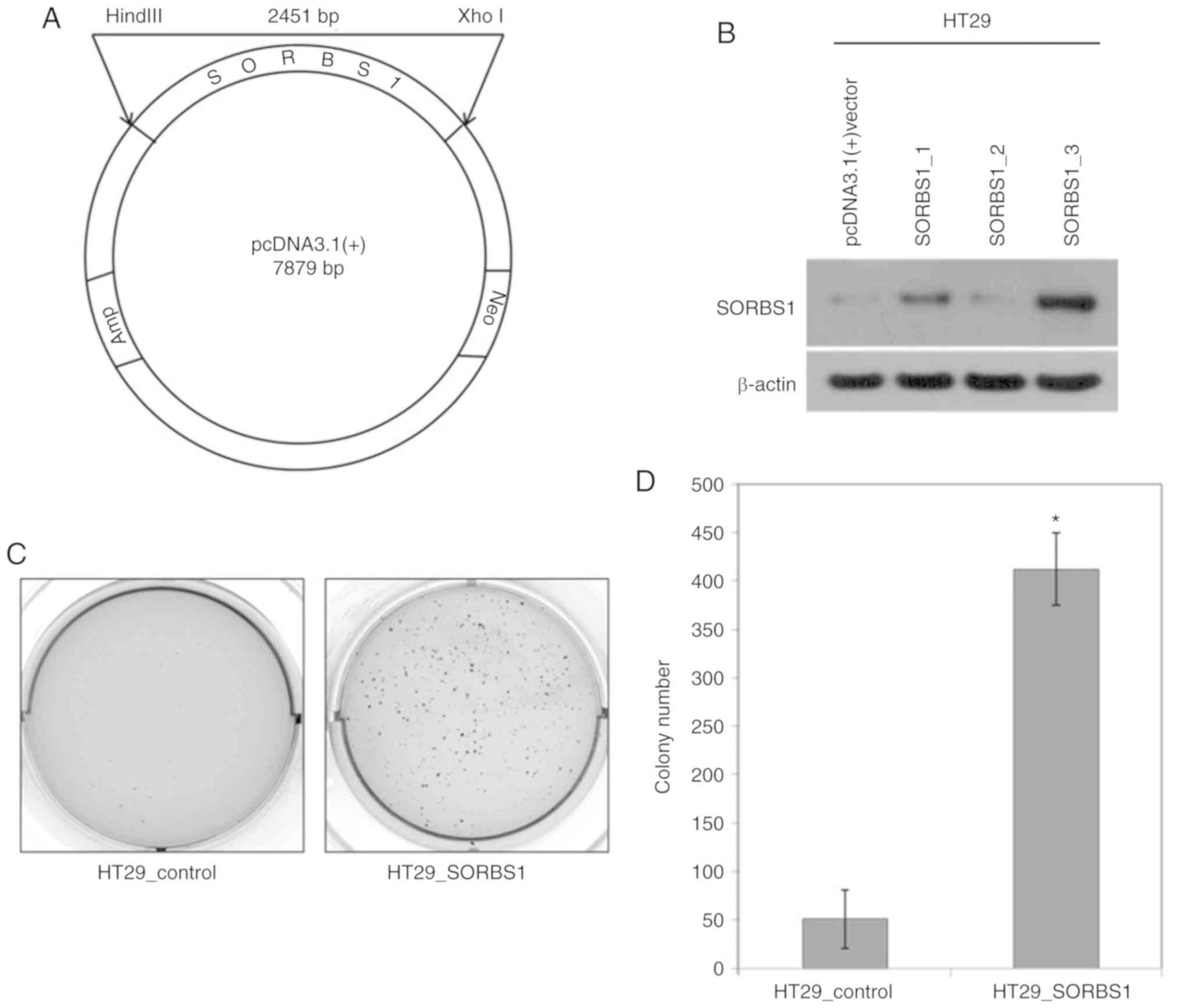
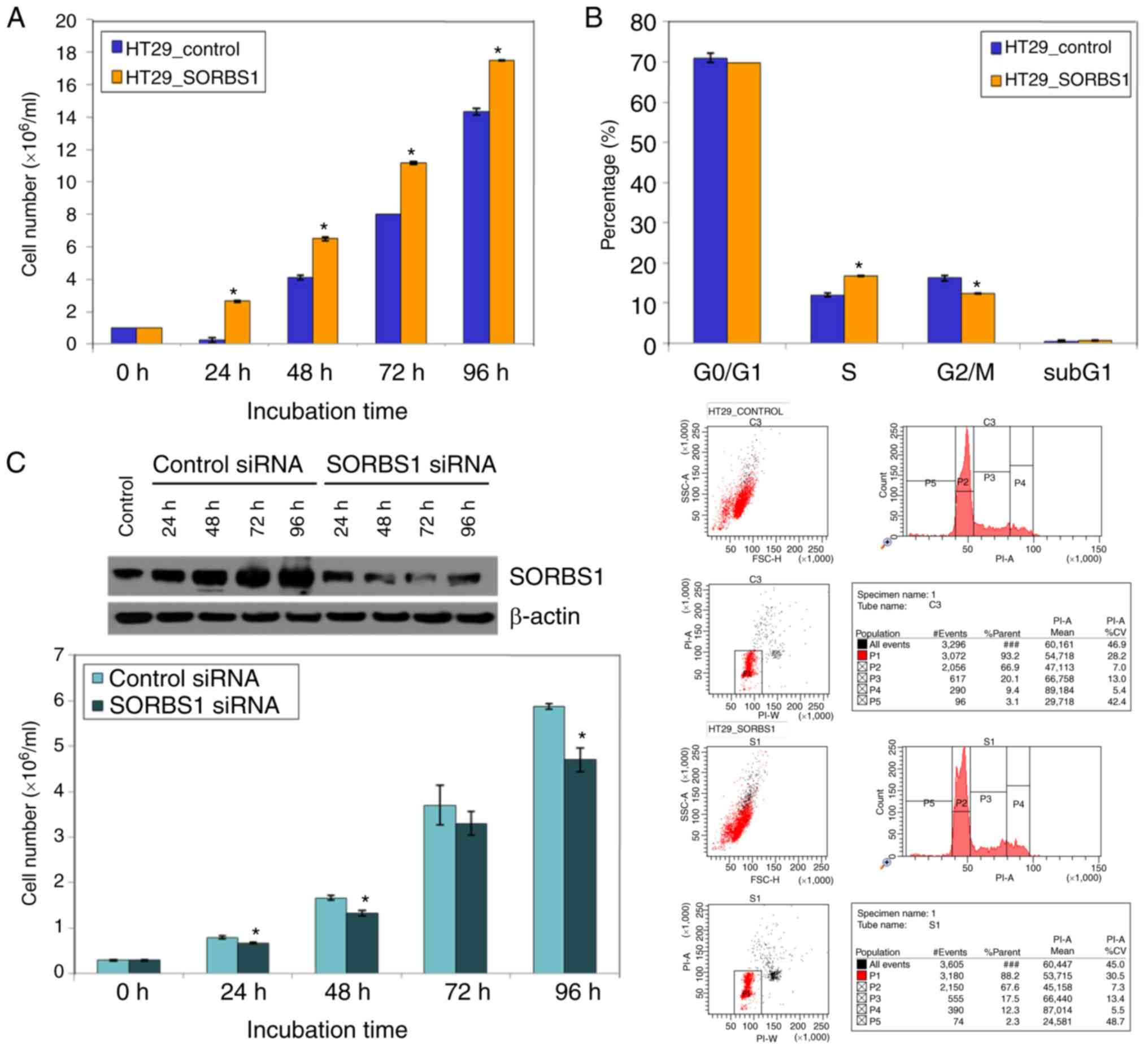
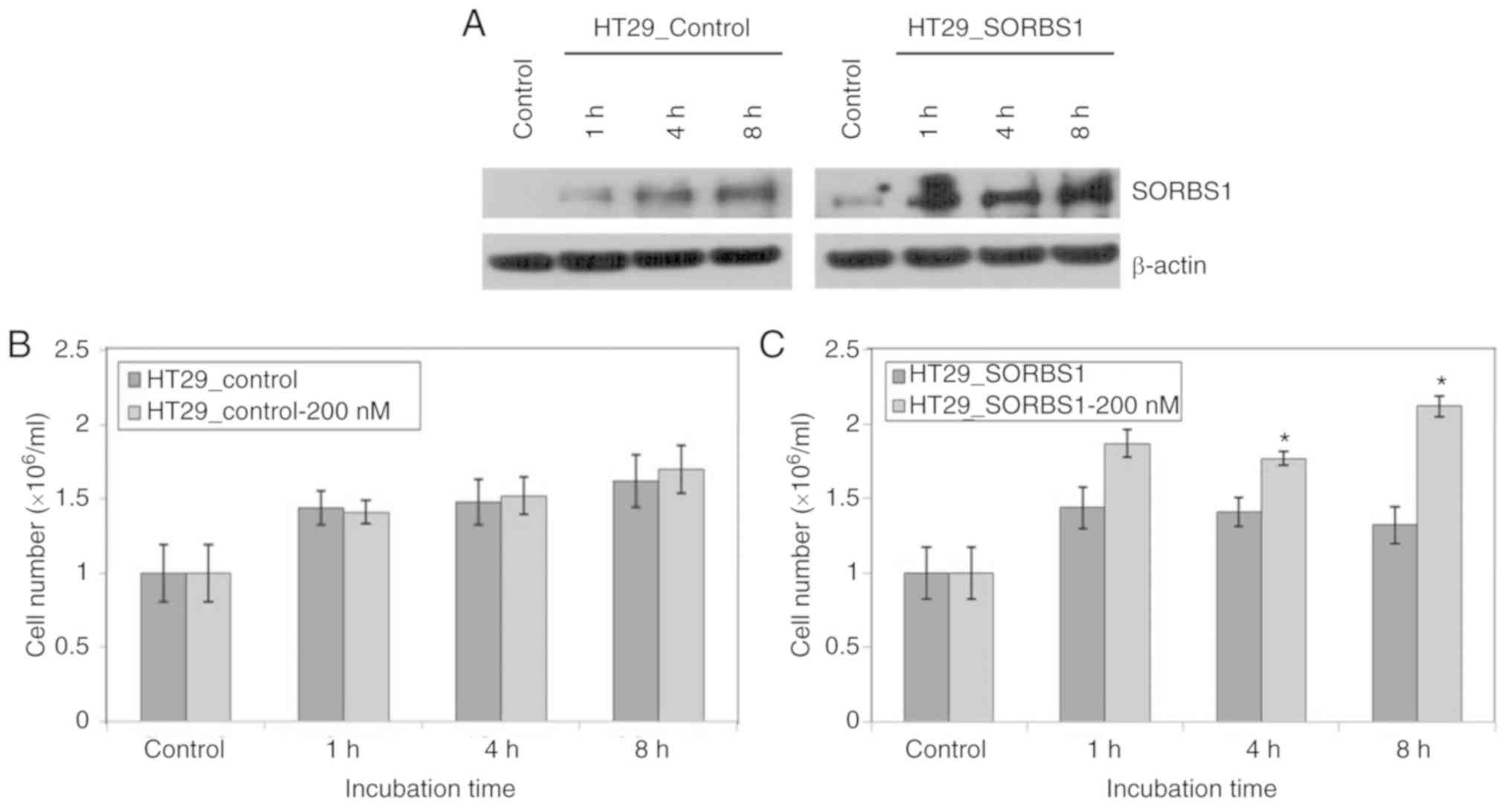
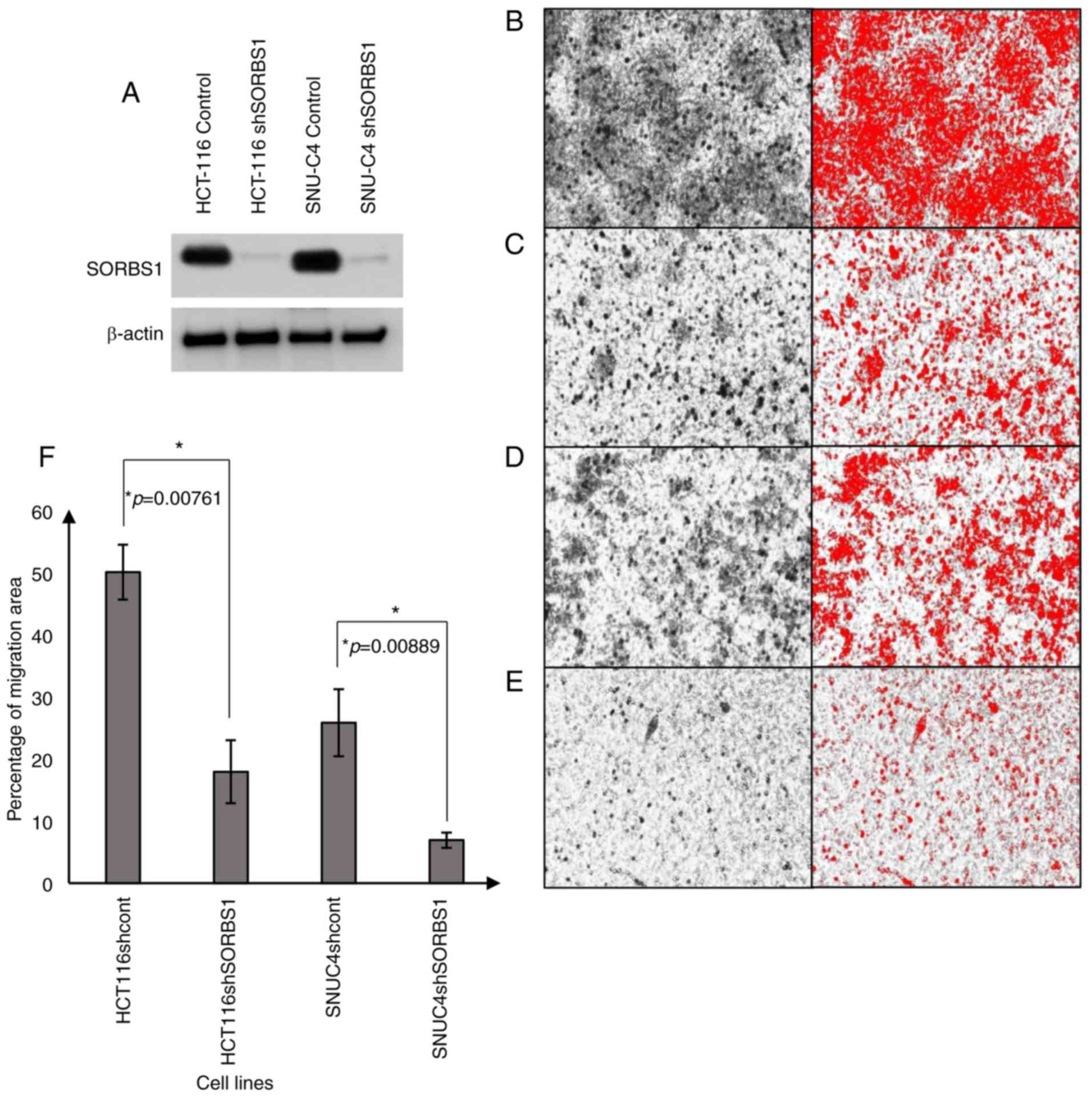
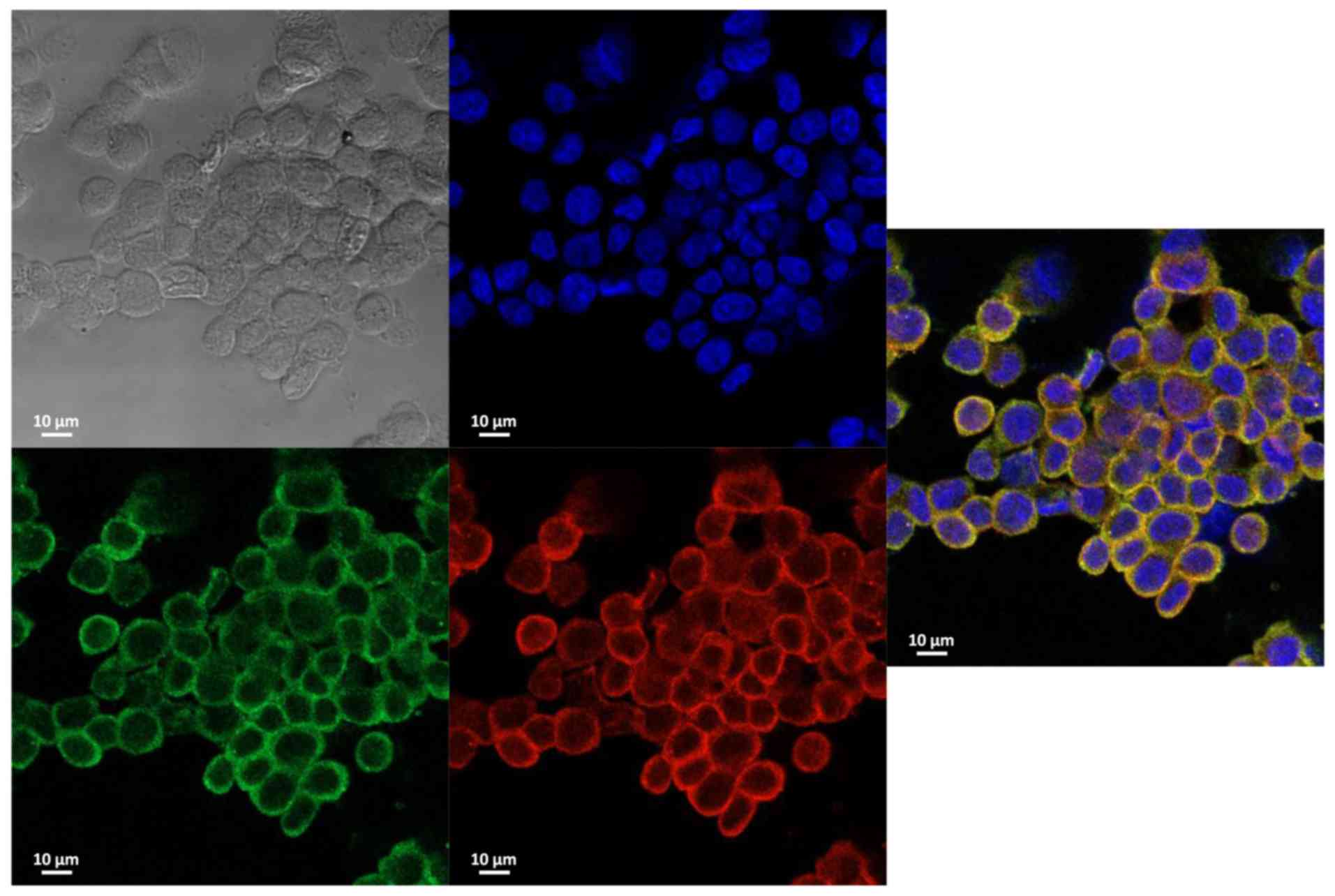
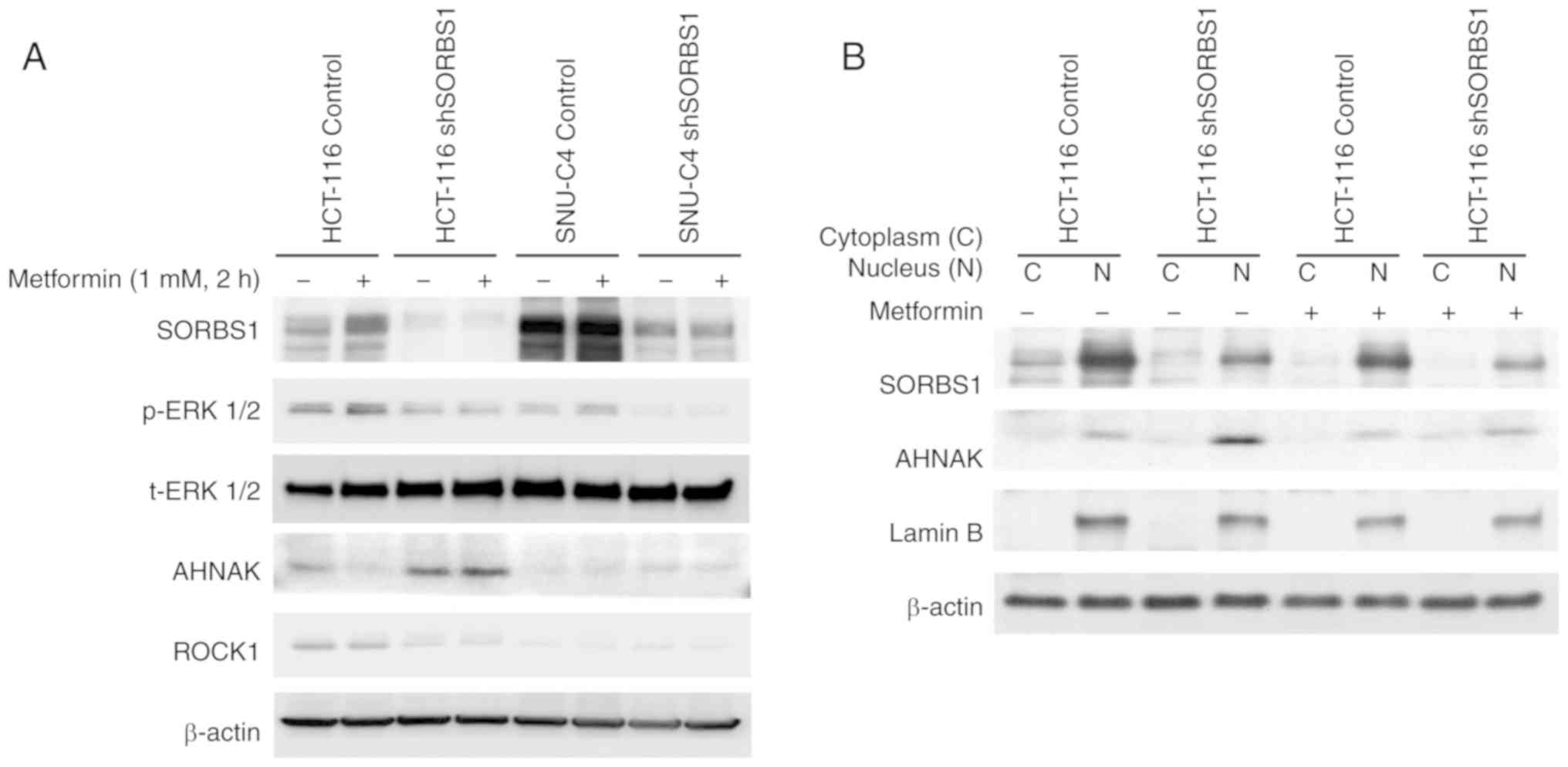
|
1
|
Ahn MY, Katsanakis KD, Bheda F and Pillay
TS: Primary and essential role of the adaptor protein APS for
recruitment of both c-Cbl and its associated protein CAP in insulin
signaling. J Biol Chem. 279:21526–21532. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Baumann CA, Ribon V, Kanzaki M, Thurmond
DC, Mora S, Shigematsu S, Bickel PE, Pessin JE and Saltiel AR: CAP
defines a second signalling pathway required for insulin-stimulated
glucose transport. Nature. 407:202–207. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Collier CA, Bruce CR, Smith AC, Lopaschuk
G and Dyck DJ: Metformin counters the insulin-induced suppression
of fatty acid oxidation and stimulation of triacylglycerol storage
in rodent skeletal muscle. Am J Physiol Endocrinol Metab.
291:E182–E189. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rojas LB and Gomes MB: Metformin: An old
but still the best treatment for type 2 diabetes. Diabetol Metab
Syndr. 5:62013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mandai K, Nakanishi H, Satoh A, Takahashi
K, Satoh K, Nishioka H, Mizoguchi A and Takai Y: Ponsin/SH3P12: An
l-afadin- and vinculin-binding protein localized at cell-cell and
cell-matrix adherens junctions. J cell Biol. 144:1001–1017. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kioka N, Ueda K and Amachi T: Vinexin,
CAP/ponsin, ArgBP2: A novel adaptor protein family regulating
cytoskeletal organi-zation and signal transduction. Cell Struct
Funct. 27:1–7. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ribon V, Printen JA, Hoffman NG, Kay BK
and Saltiel AR: A novel, multifunctional c-Cbl binding protein in
insulin receptor signaling in 3T3-L1 adipocytes. Mol Cell Biol.
18:872–879. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ribon V, Herrera R, Kay BK and Saltiel AR:
A role for CAP, a novel, multifunctional Src homology 3
domain-containing protein in formation of actin stress fibers and
focal adhesions. J Biol Chem. 273:4073–4080. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kim SC, Hong CW, Jang SG, Kim YA, Yoo BC,
Shin YK, Jeong SY, Ku JL and Park J: Establishment and
characterization of paired primary and peritoneal seeding human
colorectal cancer cell lines: Identification of genes that mediate
metastatic potential. Transl Oncol. 11:1232–1243. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang WS, Lee WJ, Huang KC, Lee KC, Chao
CL, Chen CL, Tai TY and Chuang LM: mRNA levels of the
insulin-signaling molecule SORBS1 in the adipose depots of
nondiabetic women. Obes Res. 11:586–590. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Blake TJ, Heath KG and Langdon WY: The
truncation that generated the v-cbl oncogene reveals an ability for
nuclear transport, DNA binding and acute transformation. EMBO J.
12:2017–2026. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Blake TJ, Shapiro M, Morse HC III and
Langdon WY: The sequences of the human and mouse c-cbl
proto-oncogenes show v-cbl was generated by a large truncation
encompassing a proline-rich domain and a leucine zipper-like motif.
Oncogene. 6:653–657. 1991.PubMed/NCBI
|
|
13
|
Andoniou CE, Thien CB and Langdon WY:
Tumour induction by activated abl involves tyrosine phosphorylation
of the product of the cbl oncogene. EMBO J. 13:4515–4523. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Buday L, Khwaja A, Sipeki S, Farago A and
Downward J: Interactions of Cbl with two adapter proteins, Grb2 and
Crk, upon T cell activation. J Biol Chem. 271:6159–6163. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fukazawa T, Reedquist KA, Trub T, Soltoff
S, Panchamoorthy G, Druker B, Cantley L, Shoelson SE and Band H:
The SH3 domain-binding T cell tyrosyl phosphoprotein p120.
Demonstration of its identity with the c-cbl protooncogene product
and in vivo complexes with Fyn, Grb2, and phosphati-dylinositol
3-kinase. J Biol Chem. 270:19141–19150. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Odai H, Sasaki K, Iwamatsu A, Hanazono Y,
Tanaka T, Mitani K, Yazaki Y and Hirai H: The proto-oncogene
product c-Cbl becomes tyrosine phosphorylated by stimulation with
GM-CSF or Epo and constitutively binds to the SH3 domain of
Grb2/Ash in human hematopoietic cells. J Biol Chem.
270:10800–10805. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rivero-Lezcano OM, Sameshima JH, Marcilla
A and Robbins KC: Physical association between Src homology 3
elements and the protein product of the c-cbl proto-oncogene. J
Biol Chem. 269:17363–17366. 1994.PubMed/NCBI
|
|
18
|
Zhang X, Ozawa Y, Lee H, Wen YD, Tan TH,
Wadzinski BE and Seto E: Histone deacetylase 3 (HDAC3) activity is
regulated by interaction with protein serine/threonine phosphatase
4. Genes Dev. 19:827–839. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Myung JK, Yeo SG, Kim KH, Baek KS, Shin D,
Kim JH, Cho JY and Yoo BC: Proteins that interact with calgranulin
B in the human colon cancer cell line HCT-116. Oncotarget.
8:6819–6832. 2017. View Article : Google Scholar :
|
|
20
|
Uhlen M, Fagerberg L, Hallström BM,
Lindskog C, Oksvold P, Mardinoglu A, Sivertsson Å, Kampf C,
Sjöstedt E, Asplund A, et al: Proteomics. Tissue-based map of the
human proteome. Science. 347:12604192015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang M, Liu J, Cheng A, Deyoung SM, Chen
X, Dold LH and Saltiel AR: CAP interacts with cytoskeletal proteins
and regulates adhesion-mediated ERK activation and motility. EMBO
J. 25:5284–5293. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cestra G, Toomre D, Chang S and De Camilli
P: The Abl/Arg substrate ArgBP2/nArgBP2 coordinates the function of
multiple regulatory mechanisms converging on the actin
cytoskeleton. Proc Natl Acad Sci USA. 102:1731–1736. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Liu J, Kimura A, Baumann CA and Saltiel
AR: APS facilitates c-Cbl tyrosine phosphorylation and GLUT4
translocation in response to insulin in 3T3-L1 adipocytes. Mol Cell
Biol. 22:3599–3609. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Davis TA, Loos B and Engelbrecht AM:
AHNAK: The giant jack of all trades. Cell Signal. 26:2683–2693.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Dumitru CA, Bankfalvi A, Gu X, Zeidler R,
Brandau S and Lang S: AHNAK and inflammatory markers predict poor
survival in laryngeal carcinoma. PLoS One. 8. pp. e564202013,
View Article : Google Scholar
|
|
26
|
Lee IH, Sohn M, Lim HJ, Yoon S, Oh H, Shin
S, Shin JH, Oh SH, Kim J, Lee DK, et al: Ahnak functions as a tumor
suppressor via modulation of TGFβ/Smad signaling pathway. Oncogene.
33:4675–4684. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Shankar J, Messenberg A, Chan J, Underhill
TM, Foster LJ and Nabi IR: Pseudopodial actin dynamics control
epithelial-mesen-chymal transition in metastatic cancer cells.
Cancer Res. 70:3780–3790. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Xi ZW, Xin SY, Zhou LQ, Yuan HX, Wang Q
and Chen KX: Downregulation of rho-associated protein kinase 1 by
miR-124 in colorectal cancer. World J Gastroenterol. 21:5454–5464.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Mebratu Y and Tesfaigzi Y: How ERK1/2
activation controls cell proliferation and cell death: Is
subcellular localization the answer? Cell Cycle. 8:1168–1175. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hu C, Zhou H, Liu Y, Huang J, Liu W, Zhang
Q, Tang Q, Sheng F, Li G and Zhang R: ROCK1 promotes migration and
invasion of nonsmallcell lung cancer cells through the
PTEN/PI3K/FAK pathway. Int J Oncol. 55:833–844. 2019.PubMed/NCBI
|