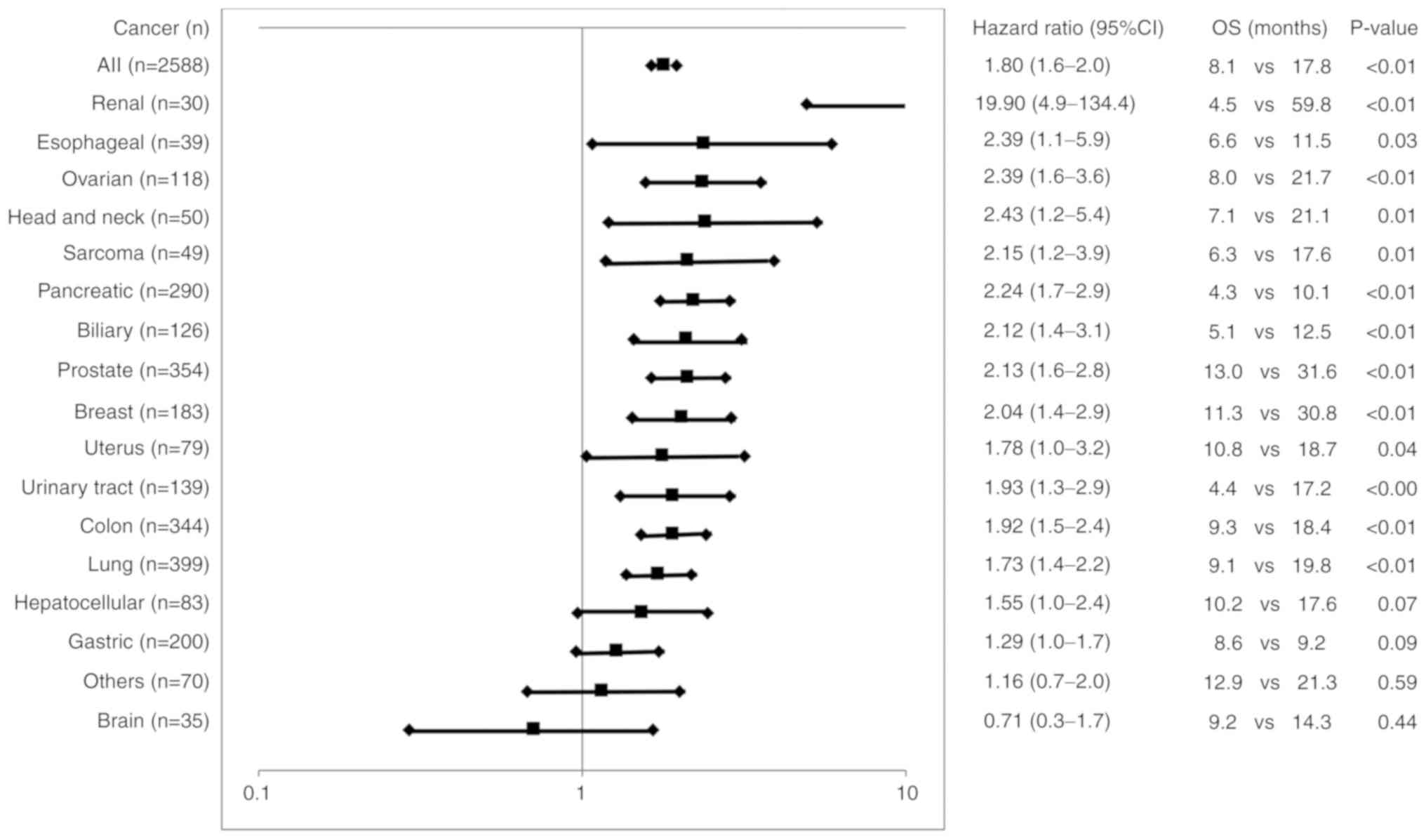
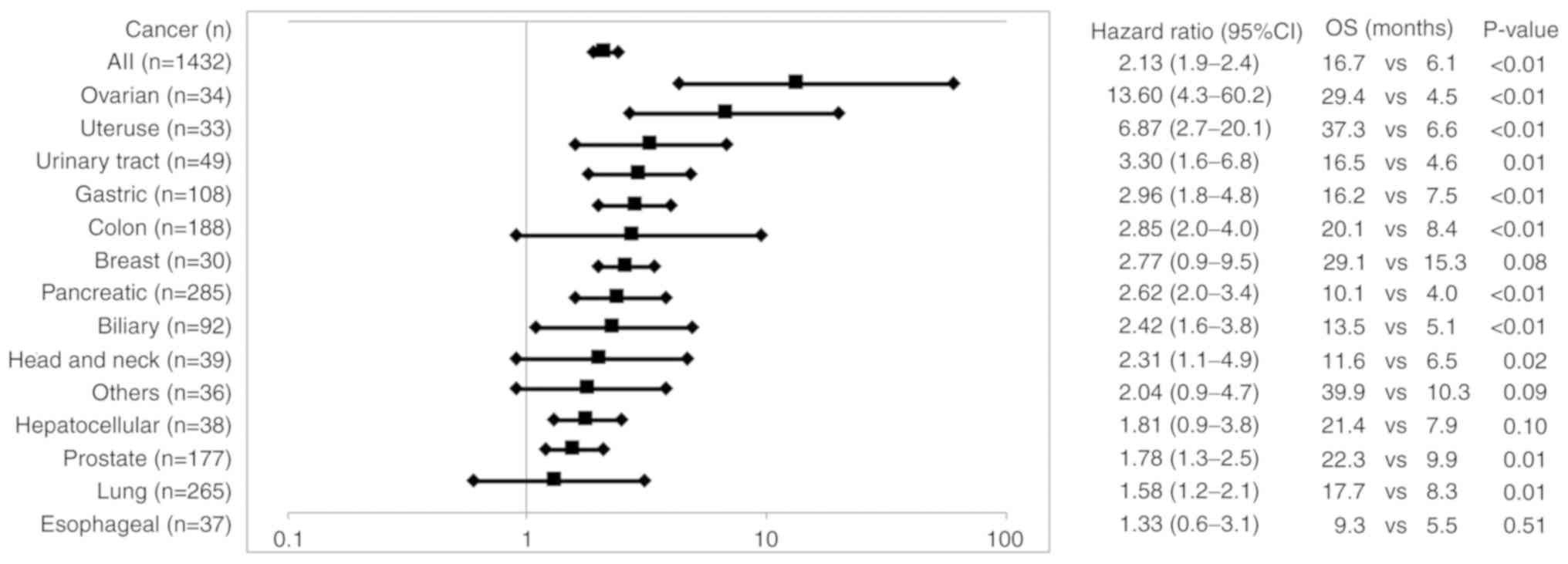
|
1
|
Topalian SL, Hodi FS, Brahmer JR,
Gettinger SN, Smith DC, McDermott DF, Powderly JD, Carvajal RD,
Sosman JA, Atkins MB, et al: Safety, activity, and immune
correlates of anti-PD-1 antibody in cancer. N Eng J Med.
366:2443–2454. 2012. View Article : Google Scholar
|
|
2
|
Brahmer JR, Tykodi SS, Chow LQ, Hwu WJ,
Topalian SL, Hwu P, Drake CG, Camacho LH, Kauh J, Odunsi K, et al:
Safety and activity of anti-PD-L1 antibody in patients with
advanced cancer. N Eng J Med. 366:2455–2465. 2012. View Article : Google Scholar
|
|
3
|
Rosenberg SA, Yang JC, Schwartzentruber
DJ, Hwu P, Marincola FM, Topalian SL, Restifo NP, Dudley ME,
Schwarz SL, Spiess PJ, et al: Immunologic and therapeutic
evaluation of a synthetic peptide vaccine for the treatment of
patients with metastatic melanoma. Nat Med. 4:321–327. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Schwartzentruber DJ, Lawson DH, Richards
JM, Conry RM, Miller DM, Treisman J, Gailani F, Riley L, Conlon K,
Pockaj B, et al: gp100 peptide vaccine and interleukin-2 in
patients with advanced melanoma. N Engl J Med. 364:2119–2127. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bezu L, Kepp O, Cerrato G, Pol J, Fucikova
J, Spisek R, Zitvogel L, Kroemer G and Galluzzi L: Trial watch:
Peptide-based vaccines in anticancer therapy. OncoImmunol.
7:e15115062018. View Article : Google Scholar
|
|
6
|
Noguchi M, Sasada T and Itoh K:
Personalized peptide vaccination: A new approach for advanced
cancer as therapeutic cancer vaccine. Cancer Immunol Immunother.
62:919–929. 2013. View Article : Google Scholar
|
|
7
|
Sasada T, Yamada A, Noguchi M and Itoh K:
Personalized peptide vaccine for treatment of advanced cancer. Curr
Med Chem. 21:2332–2345. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kibe S, Yutani S, Motoyama S, Nomura T,
Tanaka N, Kawahara A, Yamaguchi T, Matsueda S, Komatsu N, Miura M,
et al: Phase II study of personalized peptide vaccination for
previously treated advanced colorectal cancer. Cancer Immunol Res.
2:1154–1162. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Noguchi M, Matsumoto K, Uemura H, Arai G,
Eto M, Naito S, Ohyama C, Nasu Y, Tanaka M, Moriya F, et al: An
open-label, randomized phase II trial of personalized peptide
vaccination in patients with bladder cancer that progressed after
platinum-based chemotherapy. Clin Cancer Res. 22:54–60. 2016.
View Article : Google Scholar
|
|
10
|
Yoshimura K, Minami T, Nozawa M, Kimura T,
Egawa S, Fujimoto H, Yamada A, Itoh K and Uemura H: A phase 2
randomized controlled trial of personalized peptide vaccine
immunotherapy with low-dose dexamethasone versus dexamethasone
alone in chemotherapy-naive castration-resistant prostate cancer.
Eur Urol. 70:35–41. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Noguchi M, Fujimoto K, Arai G, Uemura H,
Hashine K, Matsumoto H, Fukasawa S, Nakatsu H, Takenaka A, Fujisawa
M, et al: Personalized peptide vaccination for castration-resistant
prostate cancer progressing after docetaxel: A randomized,
double-blind, placebo-controlled, phase III trial. J Clin Oncol.
37(Suppl 15): S50332019. View Article : Google Scholar
|
|
12
|
Narita Y, Arakawa Y, Yamasaki F, Nishikawa
R, Aoki T, Kanamori M, Nagane M, Kumabe T, Hirose Y, Ichikawa T, et
al: A randomized, double-blind, phase III trial of personalized
peptide vaccination for recurrent glioblastoma. Neuro Oncol.
21:348–359. 2019. View Article : Google Scholar :
|
|
13
|
Shirahama T, Muroya D, Matsueda S, Yamada
A, Shichijo S, Naito M, Yamashita T, Sakamoto S, Okuda K, Itoh K,
et al: A randomized phase II trial of personalized peptide vaccine
with low dose cyclophosphamide in biliary tract cancer. Cancer Sci.
108:838–845. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Suekane S, Ueda K, Nishihara K, Sasada T,
Yamashita T, Koga N, Yutani S, Shichijo S, Itoh K, Igawa T and
Noguchi M: Personalized peptide vaccination as second-line
treatment for metastatic upper tract urothelial carcinoma. Cancer
Sci. 108:2430–2437. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Takayama K, Sugawara S, Saijo Y, Maemondo
M, Sato A, Takamori S, Harada T, Sasada T, Kakuma T, Kishimoto J,
et al: Randomized phase II study of docetaxel plus personalized
peptide vaccination versus docetaxel plus placebo for patients with
previously treated advanced wild type EGFR non-small-cell lung
cancer. J Immunol Res. 2016:17451082016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Noguchi M, Koga N, Moriya F, Suekane S,
Yutani S, Yamada A, Shichijo S, Kakuma T and Itoh K: Survival
analysis of multiple peptide vaccination for the selection of
correlated peptides in urological cancers. Cancer Sci.
109:2660–2669. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mine T, Sato Y, Noguchi M, Sasatomi T,
Gohara R, Tsuda N, Tanaka S, Shomura H, Katagiri K, Rikimaru T, et
al: Humoral responses to peptides correlate with overall survival
in advanced cancer patients vaccinated with peptides based on
pre-existing, peptide-specific cellular responses. Clin Cancer Res.
10:929–937. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sproston NR and Ashworth JJ: Role of
C-reactive protein at sites of inflammation and infection. Front
Immunol. 9:7542018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Murphy K and Weaver C: Immunobiology
Zayets E (eds) Chapter 9, Garland New York and London. Science
Taylor and Francis Group; pp. 345–398. 2017
|
|
20
|
Komatsu N, Matsueda S, Tashiro K, Ioji T,
Shichijo S, Noguchi M, Yamada A, Doi A, Suekane S, Moriya F, et al:
Gene expression profiles in peripheral blood as a biomarker in
cancer patients receiving peptide vaccination. Cancer.
118:3208–3221. 2012. View Article : Google Scholar
|
|
21
|
Noguchi M, Moriya F, Koga N, Matsueda S,
Sasada T, Yamada A, Kakuma T and Itoh K: A randomized phase II
clinical trial of personalized peptide vaccination with metronomic
low-dose cyclophosphamide in patients with metastatic
castration-resistant prostate cancer. Cancer Immunol Immunother.
65:151–160. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dolan RD, Laird BJA, Horgan PG and
McMillan DC: The prognostic value of the systemic inflammatory
response in randomised clinical trials in cancer: A systematic
review. Crit Rev Oncol Hematol. 132:130–137. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Grivennikov SI, Greten FR and Karin M:
Immunity, inflammation, and cancer. Cell. 140:883–899. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Coffelt SB, Wellenstein MD and de Visser
KE: Neutrophils in cancer: Neutral no more. Nat Rev Cancer.
16:431–446. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shaul ME and Fridlender ZG: Cancer-related
circulating and tumor-associated neutrophils-subtypes, sources and
function. FEBS J. 285:4316–4342. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ritter B and Greten FR: Modulating
inflammation for cancer therapy. J Exp Med. 216:1234–1243. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Matsueda S, Itoh K and Shichijo S:
Antitumor activity of antibody against cytotoxic T lymphocyte
epitope peptide of lymphocyte-specific protein tyrosine kinase.
Cancer Sci. 109:611–617. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dvorak HF: Tumors: Wounds that do not
heal-redux. Cancer Immunol Res. 3:1–11. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Steinman RM, Howiger D and Nussenzweig MC:
Tolerogenic dendritic cells. Annu Rev Immunol. 21:685–711. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Basile D, Garattini SK, Bonotto M, Ongaro
E, Casagrande M, Cattaneo M, Fanotto V, De Carlo E, Loupakis F,
Urbano F, et al: Immunotherapy for colorectal cancer: Where are we
heading? Expert Opin Biol Ther. 17:709–721. 2017. View Article : Google Scholar : PubMed/NCBI
|