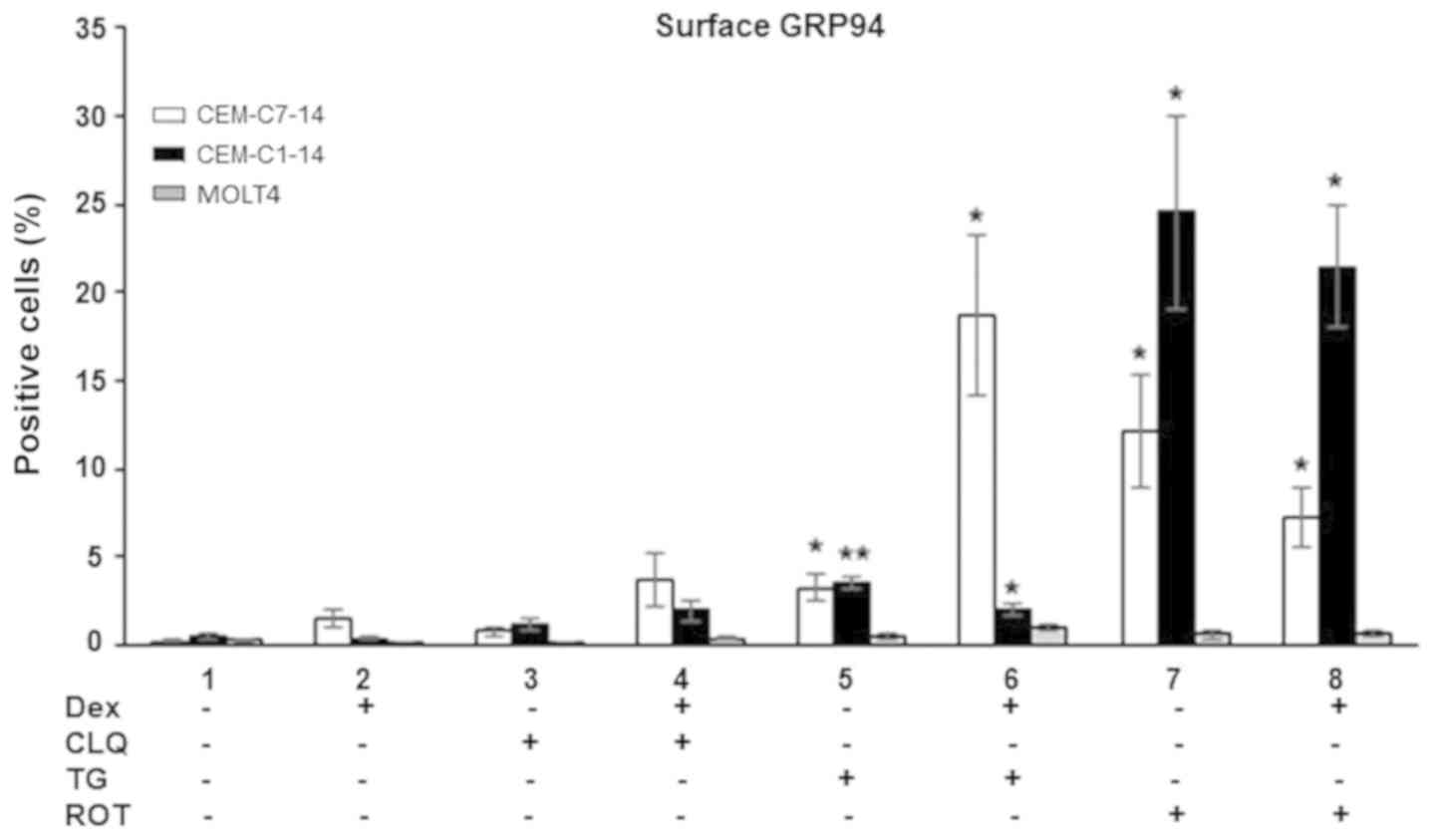
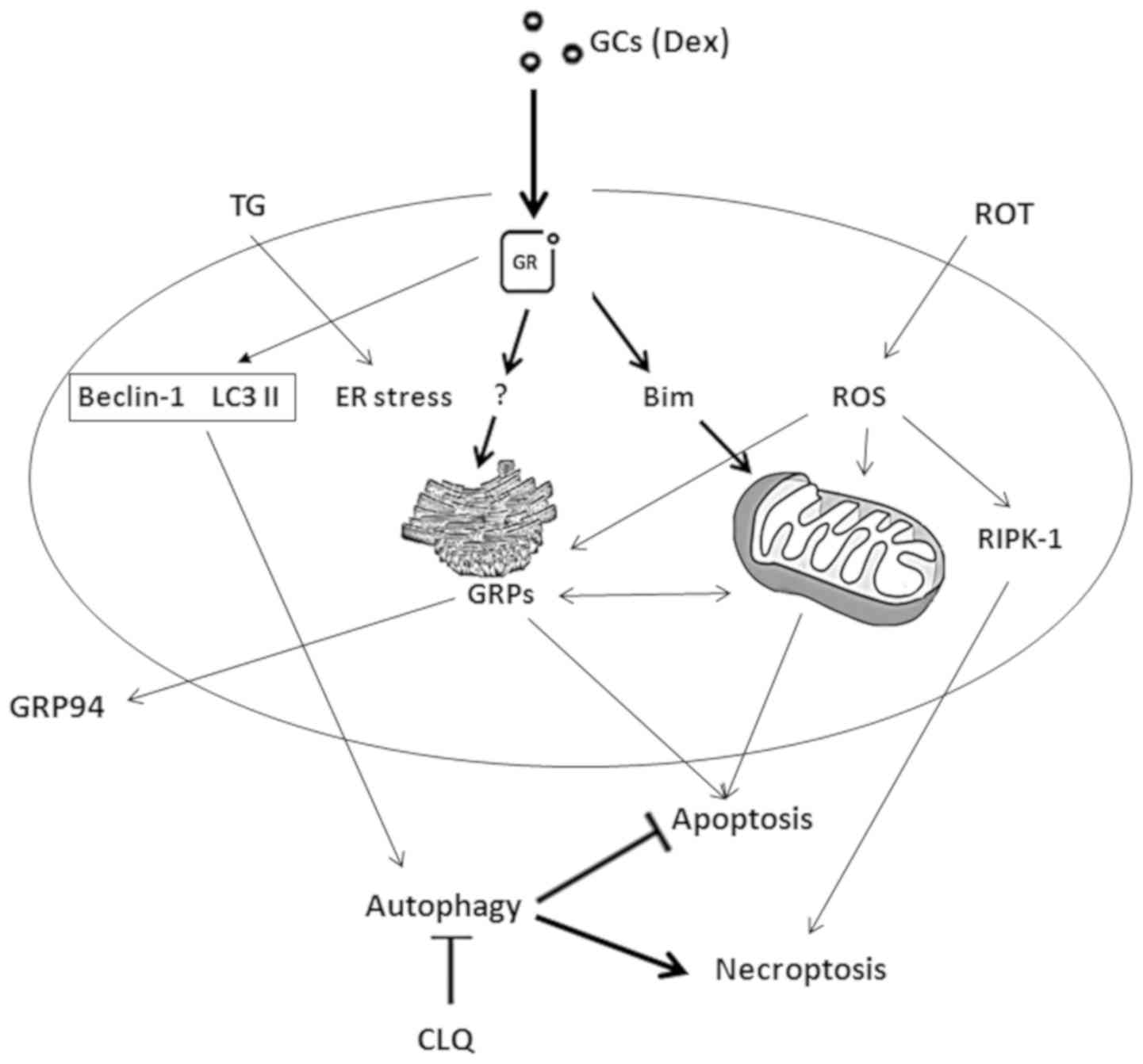
|
1
|
Arlt W and Stewart PM: Adrenal
corticosteroid biosynthesis, metabolism, and action. Endocrinol
Metab Clin North Am. 34:293–313. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Galon J, Franchimont D, Hiroi N, Frey G,
Boettner A, Ehrhart-Bornstein M, O'Shea JJ, Chrousos GP and
Bornstein SR: Gene profiling reveals unknown enhancing and
suppressive actions of glucocorticoids on immune cells. FASEB J.
16:61–71. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Smith MA, Seibel NL, Altekruse SF, Ries
LA, Melbert DL, O'Leary M, Smith FO and Reaman GH: Outcomes for
children and adolescents with cancer: Challenges for the
twenty-first century. J Clin Oncol. 28:2625–2634. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Greaves M: Infection, immune responses and
the aetiology of childhood leukaemia. Nat Rev Cancer. 6:193–203.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Greaves MF and Wiemels J: Origins of
chromosome translocations in childhood leukaemia. Nat Rev Cancer.
3:639–649. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Krajinovic M, Sinnett H, Richer C, Labuda
D and Sinnett D: Role of NQO1, MPO and CYP2E1 genetic polymorphisms
in the susceptibility to childhood acute lymphoblastic leukemia.
Int J Cancer. 97:230–236. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Webster JC, Oakley RH, Jewell CM and
Cidlowski JA: Proinflammatory cytokines regulate human
glucocorticoid receptor gene expression and lead to the
accumulation of the dominant negative beta isoform: A mechanism for
the generation of glucocorticoid resistance. Proc Natl Acad Sci
USA. 98:6865–6870. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Alves NL, Derks IA, Berk E, Spijker R, van
Lier RA and Eldering E: The Noxa/Mcl-1 axis regulates
susceptibility to apoptosis under glucose limitation in dividing T
cells. Immunity. 24:703–716. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Berrou I, Demonacos C and Krstic-Demonacos
M: Molecular mechanisms conferring resistance/sensitivity to
glucocorticoid-induced apoptosis. Glucocorticoids - New Recognition
of Our Familiar Friend. Qian X: InTech; pp. 151–174. 2012
|
|
10
|
Rosen DB, Putta S, Covey T, Huang YW,
Nolan GP, Cesano A, Minden MD and Fantl WJ: Distinct patterns of
DNA damage response and apoptosis correlate with Jak/Stat and
PI3kinase response profiles in human acute myelogenous leukemia.
PLoS One. 5:e124052010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Healy J, Bélanger H, Beaulieu P, Larivière
M, Labuda D and Sinnett D: Promoter SNPs in G1/S checkpoint
regulators and their impact on the susceptibility to childhood
leukemia. Blood. 109:683–692. 2007. View Article : Google Scholar
|
|
12
|
Nicolaides NC, Galata Z, Kino T, Chrousos
GP and Charmandari E: The human glucocorticoid receptor: Molecular
basis of biologic function. Steroids. 75:1–12. 2010. View Article : Google Scholar :
|
|
13
|
Tome ME, Jaramillo MC and Briehl MM:
Hydrogen peroxide signaling is required for glucocorticoid-induced
apoptosis in lymphoma cells. Free Radic Biol Med. 51:2048–2059.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang J, Wang X, Vikash V, Ye Q, Wu D, Liu
Y and Dong W: ROS and ROS-mediated cellular signaling. Oxid Med
Cell Longev. 2016:43509652016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Burhans WC and Heintz NH: The cell cycle
is a redox cycle: Linking phase-specific targets to cell fate. Free
Radic Biol Med. 47:1282–1293. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liemburg-Apers DC, Willems PH, Koopman WJ
and Grefte S: Interactions between mitochondrial reactive oxygen
species and cellular glucose metabolism. Arch Toxicol.
89:1209–1226. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tsiotra PC and Tsigos C: Stress, the
endoplasmic reticulum, and insulin resistance. Ann NY Acad Sci.
1083:63–76. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kato H and Nishitoh H: Stress responses
from the endoplasmic reticulum in cancer. Front Oncol. 5:932015.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cao SS and Kaufman RJ: Endoplasmic
reticulum stress and oxidative stress in cell fate decision and
human disease. Antioxid Redox Signal. 21:396–413. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zeeshan HM, Lee GH, Kim HR and Chae HJ:
Endoplasmic reticulum stress and associated ROS. Int J Mol Sci.
17:3272016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Almanza A, Carlesso A, Chintha C,
Creedican S, Doultsinos D, Leuzzi B, Luís A, McCarthy N,
Montibeller L, More S, et al: Endoplasmic reticulum stress
signalling - from basic mechanisms to clinical applications. FEBS
J. 286:241–278. 2019. View Article : Google Scholar
|
|
22
|
Llanos-González E, Henares-Chavarino ÁA,
Pedrero-Prieto CM, García-Carpintero S, Frontiñán-Rubio J,
Sancho-Bielsa FJ, Alcain FJ, Peinado JR, Rabanal-Ruíz Y and
Durán-Prado M: Interplay between mitochondrial oxidative disorders
and proteostasis in Alzheimer's disease. Front Neurosci.
13:14442020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Delprat B, Crouzier L, Su TP and Maurice
T: At the crossing of ER stress and MAMs: A key role of Sigma-1
receptor. Calcium Signaling. Islam MS: Springer International
Publishing; Cham: pp. 699–718. 2020, View Article : Google Scholar
|
|
24
|
Ferro F, Servais S, Besson P, Roger S,
Dumas JF and Brisson L: Autophagy and mitophagy in cancer metabolic
remodelling. Semin Cell Dev Biol. 98:129–138. 2020. View Article : Google Scholar
|
|
25
|
Brown MK and Naidoo N: The endoplasmic
reticulum stress response in aging and age-related diseases. Front
Physiol. 3:2632012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Binder RJ: Functions of heat shock
proteins in pathways of the innate and adaptive immune system. J
Immunol. 193:5765–5771. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Merksamer PI and Papa FR: The UPR and cell
fate at a glance. J Cell Sci. 123:1003–1006. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Corazzari M, Gagliardi M, Fimia GM and
Piacentini M: Endoplasmic reticulum stress, unfolded protein
response, and cancer cell fate. Front Oncol. 7:782017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ogata M, Hino S, Saito A, Morikawa K,
Kondo S, Kanemoto S, Murakami T, Taniguchi M, Tanii I, Yoshinaga K,
et al: Autophagy is activated for cell survival after endoplasmic
reticulum stress. Mol Cell Biol. 26:9220–9231. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wei Y, Pattingre S, Sinha S, Bassik M and
Levine B: JNK1-mediated phosphorylation of Bcl-2 regulates
starvation-induced autophagy. Mol Cell. 30:678–688. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kouroku Y, Fujita E, Tanida I, Ueno T,
Isoai A, Kumagai H, Ogawa S, Kaufman RJ, Kominami E and Momoi T: ER
stress (PERK/eIF2alpha phosphorylation) mediates the
polyglutamine-induced LC3 conversion, an essential step for
autophagy formation. Cell Death Differ. 14:230–239. 2007.
View Article : Google Scholar
|
|
32
|
Kharabi Masouleh B, Chevet E, Panse J,
Jost E, O'Dwyer M, Bruemmendorf TH and Samali A: Drugging the
unfolded protein response in acute leukemias. J Hematol Oncol.
8:872015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sano R and Reed JC: ER stress-induced cell
death mechanisms. Biochim Biophys Acta. 1833:3460–3470. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Csordás G, Weaver D and Hajnóczky G:
Endoplasmic reticulum-mitochondrial contactology: Structure and
signaling functions. Trends Cell Biol. 28:523–540. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Akl H, Vervloessem T, Kiviluoto S,
Bittremieux M, Parys JB, De Smedt H and Bultynck G: A dual role for
the anti-apoptotic Bcl-2 protein in cancer: Mitochondria versus
endoplasmic reticulum. Biochim Biophys Acta. 1843:2240–2252. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hammadi M, Oulidi A, Gackière F,
Katsogiannou M, Slomianny C, Roudbaraki M, Dewailly E, Delcourt P,
Lepage G, Lotteau S, et al: Modulation of ER stress and apoptosis
by endoplasmic reticulum calcium leak via translocon during
unfolded protein response: Involvement of GRP78. FASEB J.
27:1600–1609. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Xia M, Zhang Y, Jin K, Lu Z, Zeng Z and
Xiong W: Communication between mitochondria and other organelles: A
brand-new perspective on mitochondria in cancer. Cell Biosci.
9:272019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kondratskyi A, Kondratska K, Skryma R,
Klionsky DJ and Prevarskaya N: Ion channels in the regulation of
autophagy. Autophagy. 14:3–21. 2018. View Article : Google Scholar :
|
|
39
|
Sun F, Xu X, Wang X and Zhang B:
Regulation of autophagy by Ca(2). Tumour Biol. 37:15467–15476.
2016. View Article : Google Scholar
|
|
40
|
Bootman MD, Chehab T, Bultynck G, Parys JB
and Rietdorf K: The regulation of autophagy by calcium signals: Do
we have a consensus? Cell Calcium. 70:32–46. 2018. View Article : Google Scholar
|
|
41
|
Kania E, Pająk B and Orzechowski A:
Calcium homeostasis and ER stress in control of autophagy in cancer
cells. BioMed Res Int. 2015:3527942015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Raetz EA and Teachey DT: T-cell acute
lymphoblastic leukemia. Hematology Am Soc Hematol Educ Program.
2016:580–588. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Terwilliger T and Abdul-Hay M: Acute
lymphoblastic leukemia: A comprehensive review and 2017 update.
Blood Cancer J. 7:e5772017. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Irwin ME, Rivera-Del Valle N and Chandra
J: Redox control of leukemia: From molecular mechanisms to
therapeutic opportunities. Antioxid Redox Signal. 18:1349–1383.
2013. View Article : Google Scholar :
|
|
45
|
Harmon JM and Thompson EB: Isolation and
characterization of dexamethasone-resistant mutants from human
lymphoid cell line CEM-C7. Mol Cell Biol. 1:512–521. 1981.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Foley GE, Lazarus H, Farber S, Uzman BG,
Boone BA and McCarthy RE: Continuous culture of human lymphoblasts
from peripheral blood of a child with acute leukemia. Cancer.
18:522–529. 1965. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Saenz GJ, Hovanessian R, Gisis AD and Medh
RD: Glucocorticoid-mediated co-regulation of RCAN1-1, E4BP4 and BIM
in human leukemia cells susceptible to apoptosis. Biochem Biophys
Res Commun. 463:1291–1296. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Jiang L, Xu L, Xie J, Li S, Guan Y, Zhang
Y, Hou Z, Guo T, Shu X, Wang C, et al: Inhibition of autophagy
overcomes gluco-corticoid resistance in lymphoid malignant cells.
Cancer Biol Ther. 16:466–476. 2015. View Article : Google Scholar :
|
|
49
|
Norman MR and Thompson EB:
Characterization of a gluco-corticoid-sensitive human lymphoid cell
line. Cancer Res. 37:3785–3791. 1977.PubMed/NCBI
|
|
50
|
Lynch JT, Rajendran R, Xenaki G, Berrou I,
Demonacos C and Krstic-Demonacos M: The role of glucocorticoid
receptor phosphorylation in Mcl-1 and NOXA gene expression. Mol
Cancer. 9:382010. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Qattan MY, Bakker EY, Rajendran R, Chen
DW, Saha V, Liu J, Zeef L, Schwartz JM, Mutti L, Demonacos C, et
al: Differential regulation of cell death pathways by the
microenvironment correlates with chemoresistance and survival in
leukaemia. PLoS One. 12:e01786062017. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2−ΔΔCT method. Methods. 25:402–408. 2001. View Article : Google Scholar
|
|
53
|
Mauthe M, Orhon I, Rocchi C, Zhou X, Luhr
M, Hijlkema KJ, Coppes RP, Engedal N, Mari M and Reggiori F:
Chloroquine inhibits autophagic flux by decreasing
autophagosome-lysosome fusion. Autophagy. 14:1435–1455. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Sehgal P, Szalai P, Olesen C, Praetorius
HA, Nissen P, Christensen SB, Engedal N and Møller JV: Inhibition
of the sarco/endoplasmic reticulum (ER) Ca2+-ATPase by
thapsigargin analogs induces cell death via ER Ca2+
depletion and the unfolded protein response. J Biol Chem.
292:19656–19673. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Chen Y, McMillan-Ward E, Kong J, Israels
SJ and Gibson SB: Mitochondrial electron-transport-chain inhibitors
of complexes I and II induce autophagic cell death mediated by
reactive oxygen species. J Cell Sci. 120:4155–4166. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Zorova LD, Popkov VA, Plotnikov EY,
Silachev DN, Pevzner IB, Jankauskas SS, Babenko VA, Zorov SD,
Balakireva AV, Juhaszova M, et al: Mitochondrial membrane
potential. Anal Biochem. 552:50–59. 2018. View Article : Google Scholar :
|
|
57
|
Wilkinson L, Verhoog NJ and Louw A:
Disease- and treatment-associated acquired glucocorticoid
resistance. Endocr Connect. 7:R328–R349. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Bakker E, Qattan M, Mutti L, Demonacos C
and Krstic-Demonacos M: The role of microenvironment and immunity
in drug response in leukemia. Biochim Biophys Acta. 1863:414–426.
2016. View Article : Google Scholar
|
|
59
|
Djavaheri-Mergny M, Giuriato S, Tschan MP
and Humbert M: Therapeutic modulation of autophagy in leukaemia and
lymphoma. Cells. 8:E1032019. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Pua HH and He YW: Autophagy and lymphocyte
homeostasis. Curr Top Microbiol Immunol. 335:85–105.
2009.PubMed/NCBI
|
|
61
|
Botbol Y, Guerrero-Ros I and Macian F: Key
roles of autophagy in regulating T-cell function. Eur J Immunol.
46:1326–1334. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Hotamisligil GS and Davis RJ: Cell
signaling and stress responses. Cold Spring Harb Perspect Biol.
8:a0060722016. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Davies L, Karthikeyan N, Lynch JT, Sial
EA, Gkourtsa A, Demonacos C and Krstic-Demonacos M: Cross talk of
signaling pathways in the regulation of the glucocorticoid receptor
function. Mol Endocrinol. 22:1331–1344. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Chen DW, Saha V, Liu JZ, Schwartz JM and
Krstic-Demonacos M: Erg and AP-1 as determinants of glucocorticoid
response in acute lymphoblastic leukemia. Oncogene. 32:3039–3048.
2013. View Article : Google Scholar
|
|
65
|
Medh RD, Webb MS, Miller AL, Johnson BH,
Fofanov Y, Li T, Wood TG, Luxon BA and Thompson EB: Gene expression
profile of human lymphoid CEM cells sensitive and resistant to
glucocorticoid-evoked apoptosis. Genomics. 81:543–555. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Evangelisti C, Evangelisti C, Chiarini F,
Lonetti A, Buontempo F, Neri LM, McCubrey JA and Martelli AM:
Autophagy in acute leukemias: A double-edged sword with important
therapeutic implications. Biochim Biophys Acta. 1853:14–26. 2015.
View Article : Google Scholar
|
|
67
|
Auberger P and Puissant A: Autophagy, a
key mechanism of oncogenesis and resistance in leukemia. Blood.
129:547–552. 2017. View Article : Google Scholar
|
|
68
|
Takahashi H, Inoue J, Sakaguchi K, Takagi
M, Mizutani S and Inazawa J: Autophagy is required for cell
survival under L-asparaginase-induced metabolic stress in acute
lymphoblastic leukemia cells. Oncogene. 36:4267–4276. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Rothe K, Porter V and Jiang X: Current
outlook on autophagy in human leukemia: Foe in cancer stem cells
and drug resistance, friend in new therapeutic interventions. Int J
Mol Sci. 20:E4612019. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Laane E, Tamm KP, Buentke E, Ito K,
Kharaziha P, Oscarsson J, Corcoran M, Björklund AC, Hultenby K,
Lundin J, et al: Cell death induced by dexamethasone in lymphoid
leukemia is mediated through initiation of autophagy. Cell Death
Differ. 16:1018–1029. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Simioni C, Neri LM, Tabellini G, Ricci F,
Bressanin D, Chiarini F, Evangelisti C, Cani A, Tazzari PL,
Melchionda F, et al: Cytotoxic activity of the novel Akt inhibitor,
MK-2206, in T-cell acute lymphoblastic leukemia. Leukemia.
26:2336–2342. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Verfaillie T, Salazar M, Velasco G,
Agostinis P and Linking ER: Linking ER stress to autophagy:
Potential implications for cancer therapy. Int J Cell Biol.
2010:9305092010. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Martinvalet D: The role of the
mitochondria and the endoplasmic reticulum contact sites in the
development of the immune responses. Cell Death Dis. 9:3362018.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Dengler MA, Staiger AM, Gutekunst M,
Hofmann U, Doszczak M, Scheurich P, Schwab M, Aulitzky WE and van
der Kuip H: Oncogenic stress induced by acute hyper-activation of
Bcr-Abl leads to cell death upon induction of excessive aerobic
glycolysis. PLoS One. 6:e251392011. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Lee AS: Glucose-regulated proteins in
cancer: Molecular mechanisms and therapeutic potential. Nat Rev
Cancer. 14:263–276. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Biswas C, Sriram U, Ciric B, Ostrovsky O,
Gallucci S and Argon Y: The N-terminal fragment of GRP94 is
sufficient for peptide presentation via professional
antigen-presenting cells. Int Immunol. 18:1147–1157. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Ansa-Addo EA, Thaxton J, Hong F, Wu BX,
Zhang Y, Fugle CW, Metelli A, Riesenberg B, Williams K, Gewirth DT,
et al: Clients and oncogenic roles of molecular chaperone
gp96/grp94. Curr Top Med Chem. 16:2765–2778. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Duan XF and Xin YW: Overexpression of
molecule GRP94 favors tumor progression in lung adenocarcinoma by
interaction with regulatory T cells. Thorac Cancer. 11:704–712.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Wiersma VR, Michalak M, Abdullah TM,
Bremer E and Eggleton P: Mechanisms of translocation of ER
chaperones to the cell surface and immunomodulatory roles in cancer
and autoimmunity. Front Oncol. 5:72015. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Binder RJ: Immunosurveillance of cancer
and the heat shock protein-CD91 pathway. Cell Immunol.
343:1038142019. View Article : Google Scholar
|
|
81
|
Sundman-Engberg B, Tidefelt U, Gruber A
and Paul C: Intracellular concentrations of mitoxantrone in
leukemic cells in vitro vs. in vivo Leuk Res. 17:347–52. 1993.
View Article : Google Scholar
|