Introduction
Treatment options for pancreatic cancer have
improved in past decades with the development of molecular targeted
therapies and chemotherapy (1,2);
however, most pancreatic cancers remain unresectable when detected,
and the 5-year survival rate is <10% due to difficulties in
early detection (3), rapid
progression of the primary tumor and metastatic lesions, resistance
to and/or the requirement for discontinuation of antitumor drugs
such as 5 fluorouracil (5-FU), cisplatin and molecular targeted
therapy due to insufficient efficacy (4), and strong side effects, including
myelosuppression and gastrointestinal disorders (5). It is important to develop novel
antitumor drugs that possess sufficient efficacy and safety,
particularly for elderly patients and patients with
comorbidities.
Recent studies have proposed that intestinal and
oral microbiomes are potential factors that influence the outcome
of patients with pancreatic cancer, despite most intestinal
bacteria not making direct contact with pancreatic tissues under
normal conditions (6,7); how the intestinal bacteria influence
the pathogenesis of pancreatic cancer is unclear. Probiotics are
live microorganisms that confer a health benefit on consumers when
they are administered in adequate quantities (8). Probiotics are associated with various
health benefits, including the conditioning of the intestinal
microflora, suppression of excess allergic responses and
tumor-suppressive effects (9-11).
Previous reports have suggested that certain probiotics exhibit
tumor-suppressive effects in colorectal cancer (12), breast cancer (13) and pancreatic cancer (14) in vitro and in vivo in
animal models, indicating that probiotic bacteria may be used
safely and effectively for cancer therapy. However, the
tumor-suppressive effects of probiotics are influenced by the
bacterial culture condition (15)
and the various individual intestinal conditions shaped by food
particles and medicines (16),
resulting in difficulties in achieving stable treatment
effects.
Conversely, certain reports have indicated that the
molecules derived from probiotics have tumor-suppressive effects.
Antimicrobial peptides m2163 and m2386, identified from
Lactobacillus casei (L. casei), induced apoptosis in
colorectal cancer cells (17). Our
previous study reported that inorganic polyphosphate exhibited
tumor-suppressive effects via the activation of ERK (18). Our previous studies also revealed
that ferrichrome, identified from L. casei ATCC334,
exhibited tumor-suppressive effects in colorectal cancer cells and
gastric cancer cells through the induction of DNA damage inducible
transcript 3 (DDIT3)-mediated apoptosis (19,20).
Ferrichrome was identified as a bacterial iron-chelating agent, as
bacteria acquire Fe3+ from the external environment
(21). These studies demonstrated
the antitumor functions of ferrichrome for the first time in host
mammal cells. Notably, the antitumor activity of ferrichrome was
demonstrated to be stronger than that of conventional antitumor
drugs, including 5-FU and cisplatin, in colorectal cancer cells.
Thus, probiotic-derived ferrichrome may exhibit antitumor effects
in refractory gastrointestinal cancers, including pancreatic
cancer.
In the present study, it was revealed that
ferrichrome exhibited an antitumor effect in pancreatic cancer
in vitro and in a mouse xenograft model in vivo,
through modulation of the cell cycle and apoptosis, even in
5-FU-resistant (FUR) cells, with no significant adverse events,
indicating that probiotic-derived ferrichrome is an attractive
candidate antitumor agent that may be applied in the treatment of
refractory pancreatic cancer.
Materials and methods
Cell culture
Human cancer cell lines were cultured in RPMI-1640
(FUJIFILM Wako Pure Chemical Corporation) [PANC-1 (American Type
Culture Collection), PK-1 (Cell Resource Center for Biomedical
Research) and PCI-43 (provided by Dr Hiroshi Ishikura at Hokkaido
University) (22)] or high-glucose
Dulbecco's modified Eagle's medium (DMEM; FUJIFILM Wako Pure
Chemical Corporation) [SUIT-2 (Health Science Research Resources
Bank) and MIA PaCa-II (JCRB Cell Bank)] supplemented with 10%
(vol/vol) FBS (Biosera), 2 mM L-glutamine, 50 U/ml penicillin and
50 µg/ml streptomycin in a humidified atmosphere containing
5% CO2 at 37°C.
Animal experiments
The animal experimental procedures performed were
approved by the Animal Experiments Committee of Asahikawa Medical
University based on guidelines for the protection of animals
published by The Japanese Association of Laboratory Animal
Facilities of National University Corporations.
Reagents
Ferrichrome (Sigma-Aldrich; Merck KGaA) was
dissolved in distilled water to a concentration of 5 mg/ml, which
was used as a stock solution. It was stored at 4°C and used for
assays within 6 months. 5-FU (Sigma-Aldrich; Merck KGaA) was
dissolved in DMSO to a concentration of 10 mg/ml, which was used as
a stock solution. It was stored at -20°C and = used for assays
within 6 months. Pifithrin-µ (Tokyo Chemical Industry Co.,
Ltd.) was dissolved by DMSO to a concentration of 10 mg/ml, which
was used as a stock solution. It was stored at 4°C and used for
assays within 6 months. Each reagent was diluted in DMEM and the
cells were treated.
Sulforhodamine B (SRB) assay
The cells were seeded on 96-well microplates at
0.25-1.0×104 cells/well at 24 h prior treatment with the
test reagents. An equivalent volume of solvent (distilled water or
DMSO) was used to treat control cells. The growth inhibition
effects of ferrichrome and 5-FU were evaluated in the range of
1-1,000 µg/ml and 1-10 µg/ml, respectively. Cells
were treated with ferrichrome (0.2 µg/ml) and 5-FU (0.2
µg/ml) in a combination study. Then, the cells were fixed in
5% trichloroacetic acid for 1 h at 4°C and washed 4 times in
distilled water. The microplates were then dehydrated at room
temperature, stained with 0.057% (wt/vol) SRB powder/distilled
water (100 µl/well) at room temperature for 30 min, washed 4
times in 0.1% acetic acid and re-dehydrated at room temperature.
The stained cells were lysed in 10 mM Tris-buffer and the optical
density was measured at 510 nm.
Flow cytometry
The cells were seeded in 60-mm dishes at
0.5×106 cells/dish. After incubation in ferrichrome (1
µg/ml)-containing medium for 2 days at 37°C, the cells were
trypsinized, washed twice with PBS, and fixed in a mixture of 0.5
ml PBS and 2 ml ethanol (100%) at 4°C (the final concentration of
ethanol was 80%) overnight. The fixed cells were incubated with 25
U/ml RNase (FUJIFILM Wako Pure Chemical Corporation) at room
temperature for 20 min, and propidium iodide solution was added at
a final concentration of 50 µg/ml at room temperature for 10
min. The cell cycle was assessed via flow cytometry using a BD
FACSCalibur™ (BD Biosciences); 20,000 events were obtained from
each sample. The acquired data were analyzed using CELLQuest Pro™
(v5.2.1; BS Biosciences) and ModFitLT™ (v3.0; Verity Software
House, Inc.) software.
TUNEL staining
The SUIT-2 cells were plated on 3.5-cm dishes
(2×105 cells/plate) and incubated in 1 µg/ml of
ferri-chrome or 3 µg/ml of 5-FU-containing medium for 3 days
at 37°C. Control cells were treated with an equal volume of solvent
(distilled water or DMSO). The dishes were fixed in 4%
paraformaldehyde at room temperature for 1 h and washed extensively
with PBS. The dishes were stained using an in situ Cell
Death Detection kit with TMR red (Roche Diagnostics) according to
the manufacturer's instructions. The DNA end labelling reaction was
performed at 37°C for 1 h. The cells were mounted with an anti-fade
mounting medium (Vector Laboratories, Inc.), and the TUNEL-positive
cells were visualized via fluorescence microscopy (Keyence
Corporation). The TUNEL-positive cells were counted in 6-8 random
fields (magnification, ×200).
Western blotting
Total protein (TP) was extracted from samples using
a mammalian cell extraction kit (BioVision, Inc.). The protein
concentration was determined using a Bradford protein assay
according to the manufacturer's instructions (Bio-Rad Laboratories,
Inc.). Equal quantities of protein (10-30 µg/lane) were
resolved via 12.5% SDS-PAGE, blotted onto a nitrocellulose membrane
and then blocked in SuperBlock™ T20 (PBS or TBS) Blocking Buffer
(Thermo Fisher Scientific, Inc.) at room temperature for 1 h. The
blots were incubated overnight at 4°C with the following primary
antibodies: Phosphorylated (p)-p53 (1:1,000; cat. no. ab1431;
Abcam), p53 (1:500; cat. no. 506135; Calbiochem; Merck KGaA),
cyclin B1 (1:1,000; cat. no. ab32053; Abcam), securin (1:1,000;
cat. no. ab79546; Abcam), cyclin D1 (1:500; cat. no. CC12;
Calbiochem; Merck KGaA), cyclin-dependent kinase inhibitor 1B
(CDKN1B; 1:1,000; cat. no. PAB10300; Abnova) and cleaved poly
(ADP-ribose) polymerase (PARP; 1:1,000; cat. no. 5625; Cell
Signaling Technology, Inc.). The blots were washed in PBS-0.5%
Tween 20 (T-PBS) or TBS-0.5% Tween 20 (T-TBS), incubated with
horseradish peroxidase-conjugated secondary antibodies (1:1,000;
cat. nos. HAF007 and HAF008; R&D Systems, Inc.) at room
temperature for 1 h, washed in T-PBS or T-TBS, and then developed
using a SuperSignal™ West Pico enhanced chemiluminescence system
(Thermo Fisher Scientific, Inc.). Densitometry was performed using
ImageJ v1.8.0 (National Institutes of Health). The averaged protein
expression was normalized to actin expression (1:5,000; cat. no.
612656; BD Biosciences).
ELISA
Whole blood was collected from the inferior vena
cava of ferrichrome-treated mice. After collection, 3.2% EDTA was
added to the blood, and plasma was retrieved via centrifugation at
2,500 × g for 10 min at room temperature. The plasma ferritin
levels were determined using a Mouse Ferritin ELISA kit (cat. no.
41-FERMS-E01; ALPCO) according to manufacturer's instructions.
Immunocytochemistry
Cells were plated on 4-well plastic chamber slides
(2×105/well), which were fixed in 4% paraformaldehyde at
room temperature for 1 h, washed extensively with PBS,
permeabilized with 0.1% Triton X-100 and blocked in SuperBlock T20
(PBS) Blocking Buffer at room temperature for 1 h. The slides were
then sequentially incubated with anti-α-tubulin antibody (1:100;
cat. no. NB100-690; Novus Biologicals, LLC) at 4°C overnight,
washed with PBS and incubated with Alexa Fluor®
594-conjugated secondary antibodies (1:100; cat. no. A11032;
Invitrogen; Thermo Fisher Scientific, Inc.) at room temperature for
1 h. The nuclei were counter-stained with Hoechst 33342 (1:2,000;
Invitrogen; Thermo Fisher Scientific, Inc.) at room temperature for
5 min. The cells were mounted with an anti-fade mounting medium,
and the immunofluorescence was visualized using a fluorescence
microscope (magnification, ×1,200; Keyence Corporation) in 11
(control) and 10 (ferrichrome) fields.
Animal experiments
The protocols of the animal experiments were
approved by the Asahikawa Medical University Institutional Animal
Care and Use Committee. All aspects of animal welfare were
considered, including efforts to minimize suffering and distress,
use of analgesics or anesthetics, and special housing conditions.
The sacrifice of animals maintained for scientific research was
determined according to the completion or discontinuation of the
experimental plan. All animals were treated with the test reagents
for 10-14 days and then euthanized. A total of 48 BALB/c nude mice
and 15 BALB/c mice (male; age, 6-10 weeks; weight, 20-25 g) housed
at 20-25°C with 30-60% humidity under a 12:12 h light/dark
cyclewith ad libitum access to food and water were used for
the xenograft experiment and safety test, respectively. Animal
health and behavior were monitored on the drug treatment day. For
sacrifice, 4-5% isoflurane was administered via inhalation to mice,
and then cervical dislocation was performed under anesthesia. The
death of mice was confirmed by monitoring respiratory and cardiac
arrest. The maximum loss of body weight of mice observed during the
study was 5.6%.
Xenografts
Pancreatic cancer cells (SUIT-2 cells,
1×106 cells; FUR SUIT-2 cells, 2×106 cells)
were injected subcutaneously into the back of male BALB/c nude mice
(6 weeks old). PBS (n=5, 6 and 8 for the studies evaluating the
effects of ferrichrome compared with PBS, the effects of
ferrichrome compared with 5-FU and the effects of ferrichrome on
FUR SUIT-2 cells, respectively), ferrichrome (10 mg/kg; n=5, 6 and
6, respectively) or 5-FU (10 mg/kg; n=6) treatments were
intraperitoneally administered after the injection of SUIT-2 cells.
The administration of each drug was started on the day after
transplantation, and the durations of treatment with each drug were
12 days (every 2 days), 8 days (daily) and 10 days (daily) for the
studies evaluating the effects of ferrichrome compared with PBS,
the effects of ferrichrome compared with 5-FU and the effects of
ferrichrome on FUR SUIT-2 cells, respectively. The tumor volume was
calculated by the following formula: Tumor volume (mm3)
= 0.5 × (major diameter) x (minor diameter)2.
Transcriptome analysis
Total RNA from SUIT-2 cells was extracted using an
RNeasy mini kit (Qiagen, Inc.) according to the manufacturer's
protocols. RNA libraries were generated using an Ion Total RNA-Seq
kit v2 (Thermo Fisher Scientific, Inc.) according to the
manufacturer's instructions. The RNA libraries were then processed
for emulsion PCR using an Ion OneTouch™ system and an Ion OneTouch
200 Template kit v3 (Thermo Fisher Scientific, Inc.).
Template-positive Ion Sphere™ particles were enriched and purified
for the sequencing reaction with an Ion OneTouch ES system (Thermo
Fisher Scientific, Inc.). The template-positive Ion Sphere
Particles were then applied on Ion PI™ Chips (Thermo Fisher
Scientific, Inc.), and a high throughput sequencing reaction was
performed using an Ion Proton™ Semiconductor sequencer (Thermo
Fisher Scientific, Inc.). All of the sequencing data were mapped on
a human reference genome sequence (GRCh38/hg38), the expression
analysis and gene functional annotation analysis for each sample
was imported into CLC Genomics Workbench software v9.0.1 (CLC bio;
Qiagen Digital Insights), and significant differences between the
samples were determined using unpaired Student's t-tests.
Construction of FUR SUIT-2 cells
FUR SUIT-2 cells were obtained by the repeated
treatment of non-FUR SUIT-2 cells with 5-FU as previously described
(23). The acquisition of 5-FU
resistance was confirmed by an SRB assay.
Histopathology
BALB/c mice (PBS, n=5; ferrichrome, n=5) were
treated with PBS or ferrichrome (10 mg/kg) administered daily via
tail vein injection for 14 days. The mice were sacrificed after the
administration of ferrichrome, their organs were fixed in 4%
paraformaldehyde at room temperature overnight. The fixed tissue
was embedded in paraffin and cut into 4-µm thick sections,
stained with hematoxylin at room temperature for 15 min and eosin
at room temperature for 30 sec, and then assessed under a light
microscope (magnification, ×200 or ×400). The histological changes
were assessed in 3 fields of view.
Complete blood count (CBC)
BALB/c mice (PBS, n=5; ferri-chrome, n=5; 5-FU, n=5)
were treated with PBS, ferrichrome (10 mg/kg) or 5-FU (10 mg/kg)
administered daily via tail vein injection for 14 days. The whole
blood of mice was collected from the inferior vena cava. After
collection, 3.2% EDTA was mixed with the whole blood to prevent
coagulation. CBC was performed by the New Drug Research Center,
Inc.
Serum biochemistry
Whole blood was collected from the inferior vena
cava, subjected to centrifugation at 2,500 × g for 10 min at room
temperature, and then the serum of PBS, ferrichrome or 5-FU-treated
mice was obtained. The serum samples were kept at -80°C and
biochemistry [creatinine (CRE), TP, albumin (ALB), alanine
aminotransferase (ALT), aspartate aminotransferase (AST), iron,
sodium, potassium, chlorine, calcium] was performed by Oriental
Yeast Co., Ltd.
Statistical analysis
The assay data were analyzed using Student's
unpaired t-test in two-group comparisons (control and ferrichrome)
and Williams test (dose-dependent effects of ferrichrome). To
analyze the effects of treatments (ferrichrome or 5-FU), one-way
analysis of variance (ANOVA) followed by Bonferroni's post hoc test
was performed. The datasets where measurements were collected at
multiple time points, and the effects of single and combination
treatment of ferrichrome and 5-FU were assessed using mixed or
between-subjects two-way ANOVA followed by Bonferroni's test and
two-way ANOVA followed by Tukey's post hoc test, respectively.
P<0.05 was considered to indicate a statistically significant
difference.
Results
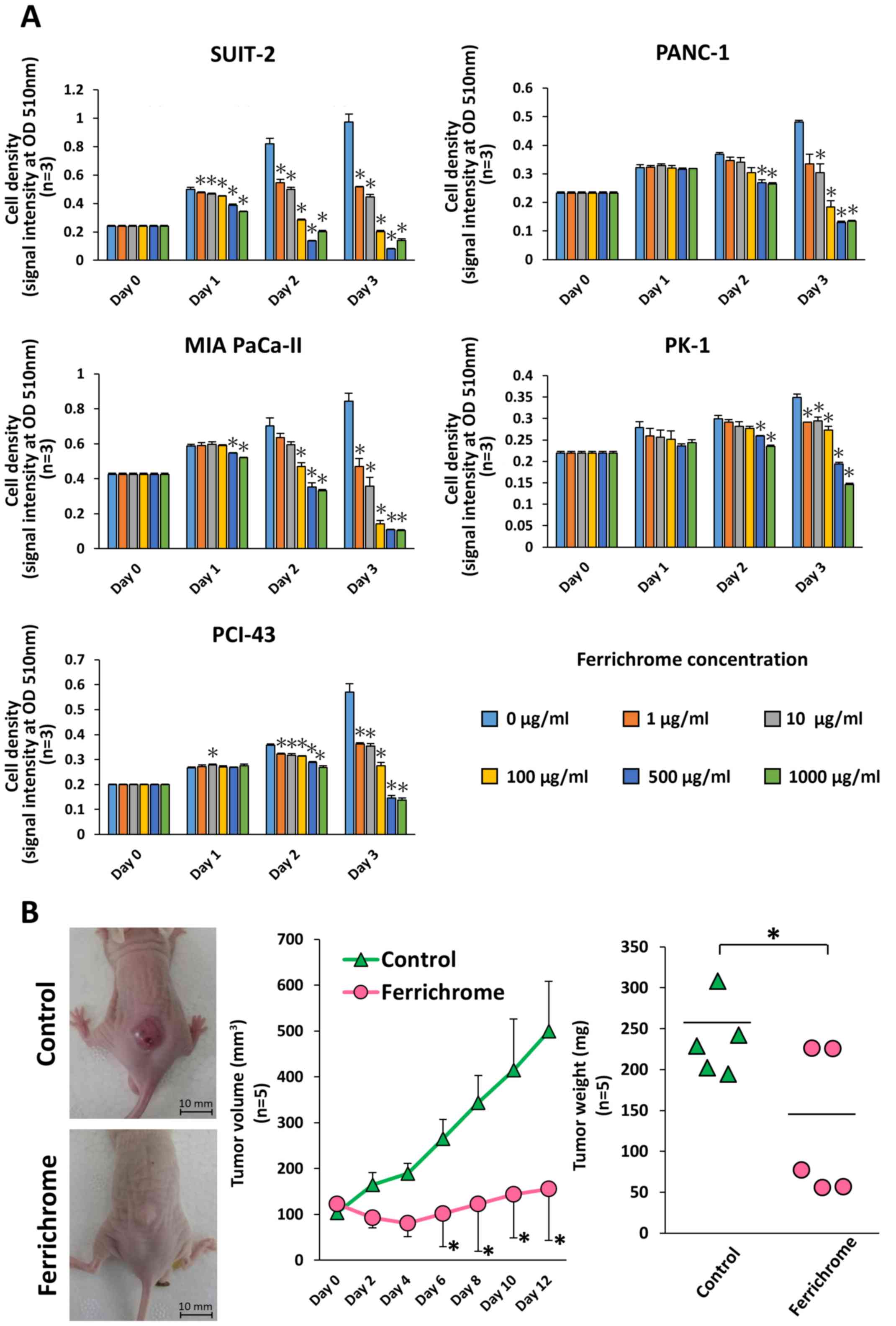
Ferrichrome inhibits pancreatic cancer
progression
To investigate the tumor-suppressive effects of
ferrichrome in pancreatic cancer cells, SUIT-2, PANC-1, MIA
PaCa-II, PK-1 and PCI-43 cells were treated with ferrichrome
(Fig. 1A). An SRB assay revealed
that ferrichrome significantly inhibited cell growth in a
dose-dependent manner in these pancreatic cancer cell lines, most
notably in SUIT-2, MIA PaCa-II and PCI-43 cells. To assess the
tumor-suppressive effects of ferrichrome in vivo, SUIT-2
cells were transplanted into nude mice, and ferrichrome was
intraperitoneally administered. Tumor volume and weight were
significantly decreased in animals treated with ferrichrome
(Fig. 1B). These data indicated
that ferrichrome exerted a tumor-suppressive effect in pancreatic
cancer cells.
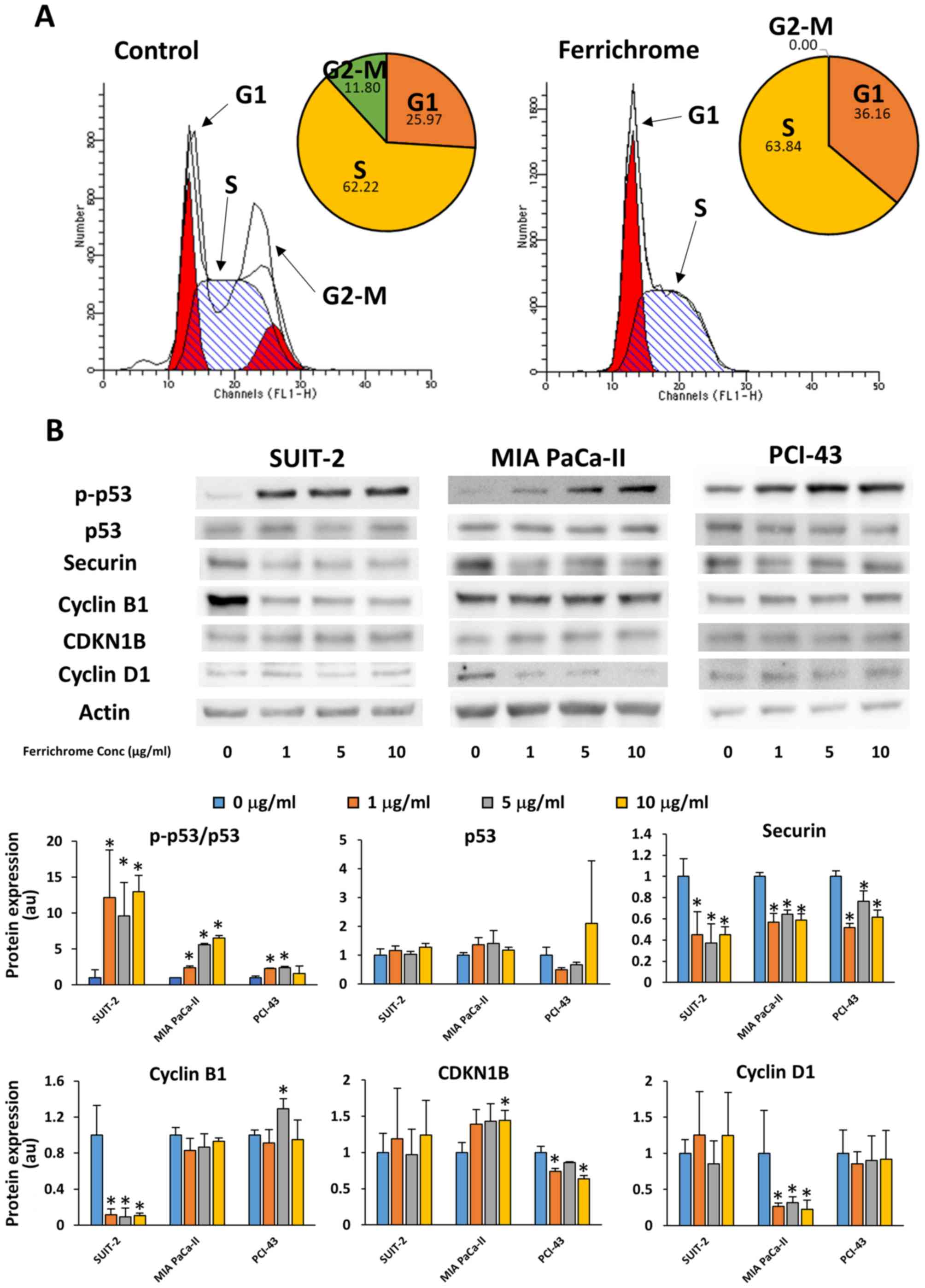
Ferrichrome inhibits the progression of
pancreatic cancer cells via p53-mediated cell cycle regulation
Flow cytometry and immunostaining were performed to
analyze the effects of ferrichrome on cell cycle progression. These
assays showed that the cell cycle was arrested in the S phase,
without chromosome misalignment in 1 µg/ml
ferrichrome-treated SUIT-2 cells (Figs. 2A and S1). Western blotting revealed that 1, 5
and 10 µg/ml ferrichrome significantly increased the
phosphorylation of p53, and decreased the expression of securin and
cyclin B1 in SUIT-2 cells (Fig.
2B). However, CDKN1B and cyclin D1, which are associated with
the progression of cells from the G1 phase to the S phase (24), were not affected by ferrichrome
treatment, suggesting that ferrichrome inhibited the progression of
the cells to the G2-M phase but not DNA synthesis in SUIT-2 cells.
To further determine whether the antitumor effects of ferrichrome
were mediated by p53 activation in other ferrichrome-sensitive
pancreatic cancer cells, phosphorylation of p53 was assessed in MIA
PaCa-II and PCI-43 cells. Western blotting analysis indicated that
p53 was activated in a dose-dependent manner without increasing
total p53 levels, whereas the downregulation of cyclin D1 and
CDKN1B was detected in MIA PaCa-II and PCI-43 cells, respectively
(Fig. 2B). These findings
indicated that p53 activation was required for ferrichrome to exert
its antitumor effects, and the roles of other cell cycle-associated
molecules were dependent upon the characteristics of the pancreatic
cancer cells.
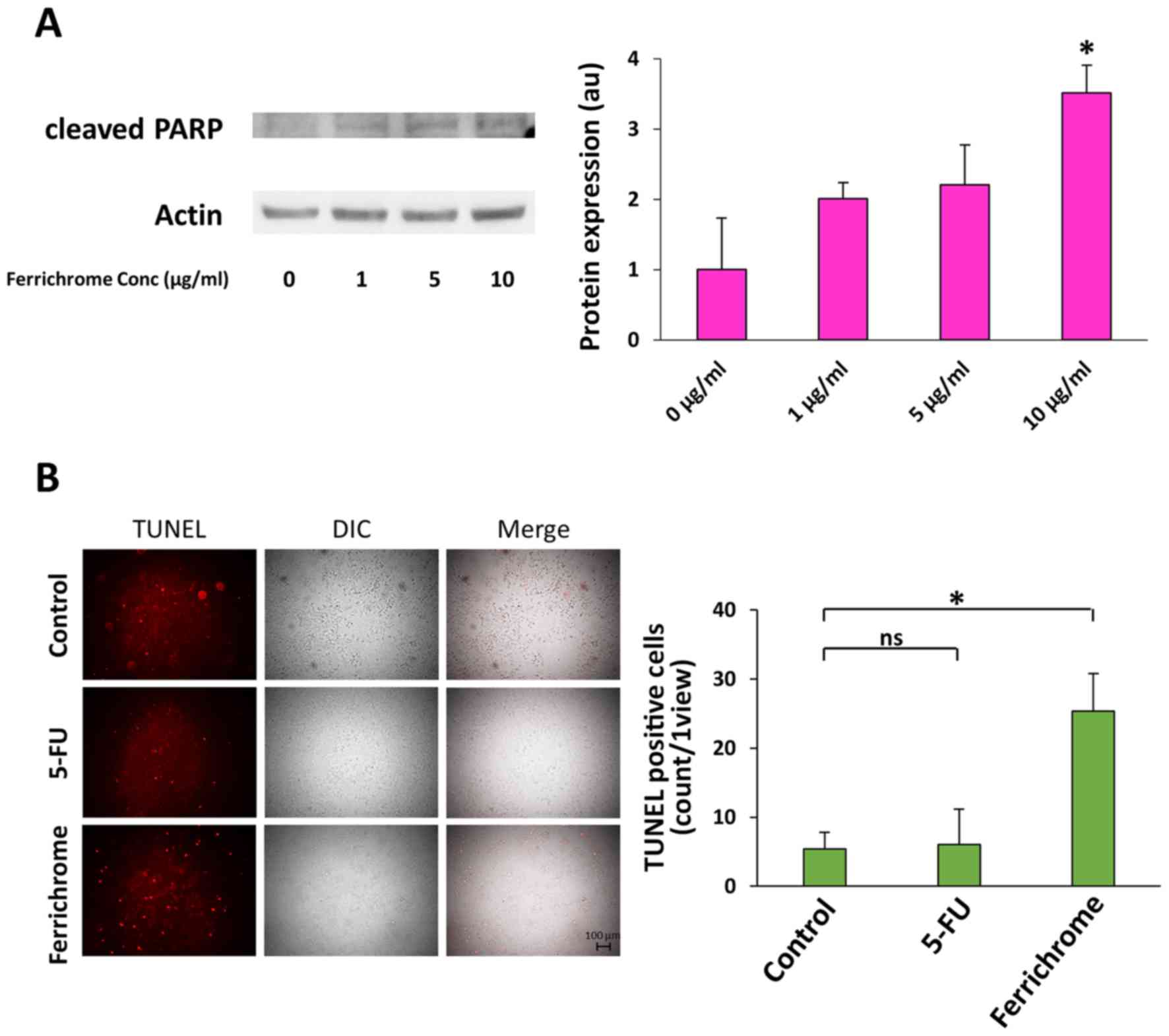
Ferrichrome induces cancer cell apoptosis
via upregulation of the p53 pathway
Western blotting showed that cleaved PARP levels in
10 µg/ml ferrichrome-treated SUIT-2 cells were significantly
increased compared with control cells (Fig. 3A). TUNEL staining indicated that
DNA fragmentation was induced by 1 µg/ml ferrichrome to a
much greater extent than by 3 µg/ml 5-FU treatment (Fig. 3B). To clarify whether the growth
suppression induced by ferrichrome treatment was mediated by p53
activation, SUIT-2 cells were treated with pifithrin-µ,
which inhibits p53 function by directly binding to the DNA-binding
domain of p53 (25). The growth
suppression induced by 5 µg/ml ferrichrome treatment was
reduced from 72 to 35% by treatment with 1 µg/ml
pifithrin-µ (Table I),
suggesting that ferrichrome suppressed pancreatic cancer cell
progression via the upregulation of p53-mediated transcription of
mRNAs. A transcriptome analysis was performed to further
investigate the induction of p53-related mRNAs by ferrichrome
treatment in pancreatic cancer cells. A total of 30 mRNAs that are
directly regulated by activated p53 were significantly (>2-fold)
induced in 10 µg/ml ferrichrome-treated SUIT-2 cells in
comparison to control cells (Table
II), as well as 48 apoptosis-inducible factors (Table SI) and 10 iron-related genes
(Table III). These findings
suggested that ferrichrome exhibited antitumor effects via the
upregulation of p53-mediated mRNA transcription.
 | Table IGrowth-inhibiting effect of 5
µg/ml ferrichrome. |
Table I
Growth-inhibiting effect of 5
µg/ml ferrichrome.
| Ferrichrome
(µg/ml) | Pifithrin-µ
(µg/ml) | Growth inhibition
effect (%) |
|---|
| 0 | 0 | 100±3.2 |
| 5 | 0 | 72±3.4 |
| 5 | 0.1 | 60±4.2a |
| 5 | 0.5 | 57±1.4a |
| 5 | 1 | 35±9.4a |
 | Table IIList of mRNAs with significantly
altered expression in ferrichrome-treated SUIT−2
cells. |
Table II
List of mRNAs with significantly
altered expression in ferrichrome-treated SUIT−2
cells.
| Gene | Fold change | P-value |
|---|
| ITGAM | 2.29 |
1.99×10−2 |
| ISG20 | 2.21 |
6.10×10−3 |
| DUSP5 | 2.04 |
8.34×10−4 |
| KLF7 | 2.03 |
4.51×10−3 |
| TMEM29 | 2.31 |
2.88×10−2 |
| JDP2 | 3.69 |
4.32×10−3 |
| PDGF-R-β | 2.41 |
1.12×10−3 |
| G6PE | 2.03 |
2.62×10−2 |
| ARTN | 2.09 |
4.00×10−2 |
| LIPIN1 | 2.04 |
8.54×10−5 |
| TEL2 | 3.24 |
8.59×10−4 |
| Syntaxin 11 | 3.51 |
6.04×10−3 |
| SLC7A11 | 2.75 |
1.59×10−4 |
| Syk | 2.05 |
2.90×10−2 |
| ABCC11 | 2.56 |
2.82×10−2 |
| DENND2C | 2.20 |
5.02×10−4 |
| FGF18 | 2.58 |
4.50×10−2 |
| DAB1 | 2.21 |
5.14×10−3 |
| CYP3A7 | 6.20 |
3.17×10−2 |
| iASPP | 2.06 |
6.93×10−3 |
| BNIPL | 2.07 |
4.71×10−2 |
| FLJ11259 | 2.94 |
6.66×10−4 |
| Gdap1 | 2.48 |
9.30×10−3 |
| NF-κB2 (p100) | 2.05 |
2.55×10−3 |
|
TRUNDD(TNFRSF10D) | 3.10 |
1.47×10−4 |
| VEGF-A | 2.57 |
4.17×10−3 |
| SORBS1 | 2.07 |
3.71×10−6 |
| Keratin 15 | 3.93 |
2.98×10−4 |
| ATF-3 | 2.36 |
9.41×10−3 |
| REDD1 | 5.10 |
6.19×10−4 |
 | Table IIIAltered expression of iron-related
genes in ferrichrome-treated SUIT-2 cells. |
Table III
Altered expression of iron-related
genes in ferrichrome-treated SUIT-2 cells.
| Gene | Fold change | P-value |
|---|
| CYP7A1 | 14.78 |
6.40×10−3 |
| MIOX | 7.97 |
1.11×10−2 |
|
CYP3A7-CYP3A51P | 6.20 |
3.17×10−2 |
| RHAG | 2.67 |
1.54×10−2 |
| RFESD | 2.16 |
3.53×10−2 |
| CYP2C18 | -2.04 |
4.86×10−3 |
| MUTYH | -2.12 |
4.50×10−3 |
| NOS3 | -2.53 |
4.29×10−2 |
| SLC40A1 | -3.61 |
7.75×10−3 |
| EXO5 | -3.78 |
1.91×10−4 |
Ferrichrome inhibits tumor progression of
5-FU-resistant SUIT-2 cells as well as 5-FU-non-resistant SUIT-2
cells
To compare the tumor-suppressive effects of
ferrichrome and 5-FU, SUIT-2 cells were treated with ferrichrome or
5-FU. Growth inhibition was detected following 2 days treatment
with ferrichrome (IC50: 0.23 µg/ml) or 5-FU
(IC50: 2.96 µg/ml), and the tumor-suppressive
effects of the molecules were not significantly different (Fig. 4A). The therapeutic effects of the
combined treatment of 0.2 µg/ml of ferrichrome and 0.2
µg/ml of 5-FU were also assessed. Low-dose ferrichrome or
5-FU mildly suppressed tumor cell growth, whereas the combination
of these agents synergistically exerted an antitumor effect in
SUIT-2 cells (Fig. 4B). To
determine the tumor-suppressive effects in vivo, a mouse
xenograft model was generated via the transplantation of
1×106 of SUIT-2 cells, and ferrichrome (10 mg/kg) or
5-FU (10 mg/kg) was intraperitoneally administered. The tumor
volumes of the ferrichrome- and 5-FU-treated groups were
significantly reduced in comparison to the PBS-treated group from 4
days after treatment onwards (Fig.
4C).
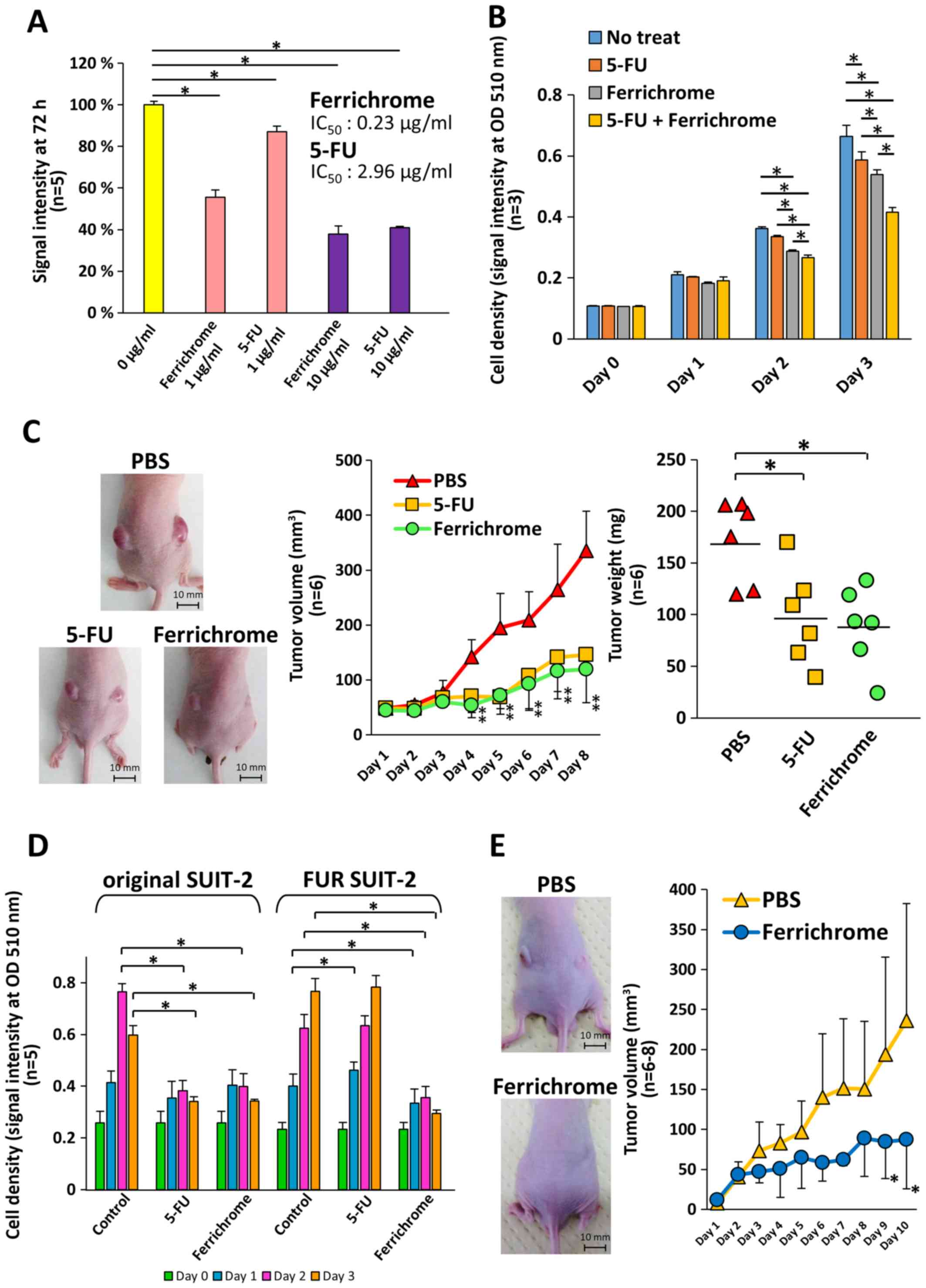 | Figure 4Ferrichrome exhibits antitumor
effects in FUR SUIT-2 cells. (A) SRB assay showing dose-dependent
effects of ferrichrome (IC50: 0.23 µg/ml) and 5-FU (IC50:
2.96 µg/ml) on SUIT-2 cells (n=5). (B) Combination effect of
ferrichrome and 5-FU in SUIT-2 cells (n=3). (C) Ferrichrome exerted
anti-tumor effects equal to or stronger than 5-FU in a mouse
xenograft model (n=6). Ferrichrome and 5-FU were dissolved in PBS,
and 10 mg/kg of ferrichrome, PBS or 10 mg/kg of 5-FU was
administered intraperitoneally every day for 8 days to BALB/c nude
mice transplanted with 1×106 SUIT-2 cells. The maximum
tumor volume and diameter of the transplanted tumor were 684
mm3 and 11.7 mm (PBS), 303 mm3 and 9.1 mm
(5-FU), and 194 mm3 and 8.4 mm (ferrichrome),
respectively. The maximum combined tumor diameters were 22.8 mm
(PBS), 18.1 mm (5-FU) and 16.3 mm (ferrichrome). (D) SRB assay
demonstrating that 1 µg/ml ferrichrome significantly reduced
tumor cell growth in both 5-FU-sensitive and FUR SUIT-2 cells after
2 days of treatment (n=5). (E) Ferrichrome suppressed tumor
progression in FUR SUIT-2 cell-transplanted mice (n=6-8). The
maximum tumor volume and diameter of the transplanted tumors were
468 mm3 and 11.3 mm (PBS) and 213 mm3 and 8.2
mm (ferrichrome), respectively. The maximum combined tumor
diameters were 17.6 mm (PBS) and 14.9 mm (ferrichrome). Data are
presented as the mean ± SD. Data were analyzed via one-way, two-way
or mixed ANOVA followed by Tukey's or Bonferroni's post hoc tests.
*P<0.05 vs. PBS or as indicated. 5-FU,
5-fluorouracil; FUR, 5-FU-resistant; SRB, sulforhodamine B; OD,
optical density. |
Next, FUR SUIT-2 cells were constructed to determine
the antitumor effects of ferrichrome in antitumor agent-resistant
cells. An SRB assay showed that the growth of FUR SUIT-2 cells was
not inhibited by treatment with 3 µg/ml 5-FU, while growth
was significantly inhibited by treatment with 1 µg/ml
ferrichrome, to the same extent as in the original SUIT-2 cells
(Fig. 4D). To assess the
tumor-suppressive effects of ferrichrome in FUR cells in
vivo, 2×106 FUR SUIT-2 cells were transplanted into
nude mice, and PBS or ferrichrome (10 mg/kg) was intraperitoneally
administered daily. The tumor volume in the ferrichrome-treated
mice was significantly reduced compared with the PBS-treated mice
at 9 days after the start of treatment (Fig. 4E). These data indicated that
ferrichrome exhibited tumor-suppressive effects in vitro and
in vivo, even in FUR cells, and suggested that the mechanism
of action differed from that of 5-FU.
Influence of ferrichrome on the
biochemistry and pathological findings of organs in normal
mice
To investigated hepatotoxicity, nephrotoxicity and
electrolyte imbalance induced by to ferrichrome treatment, PBS or
ferrichrome (10 mg/kg) was administered daily via tail vein
injection for 14 days. There were no changes in the test values of
CRE, TP, ALB, AST, ALT, iron or electrolytes between the control
and ferrichrome groups (Fig. 5A and
B). Ferrichrome treatment was not associated with pathological
changes of the organs, including the heart, kidney, small and large
intestines, skin, liver, brain, and bone marrow (Fig. S2).
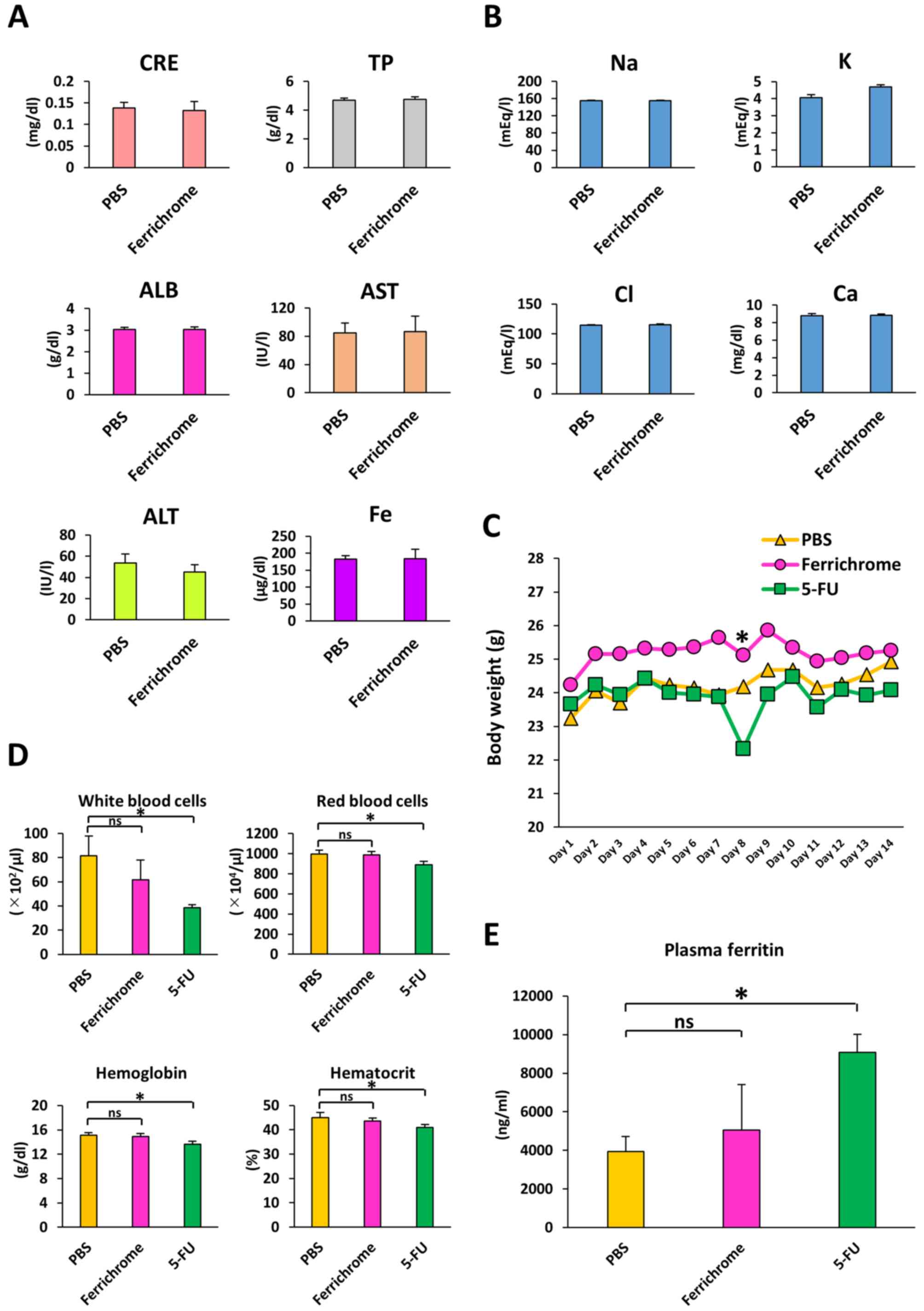 | Figure 5Ferrichrome does not affect serum
biochemistry. (A) Biochemical analysis of CRE, TP, ALB, AST, ALT
and iron. (B) Biochemical analysis of serum electrolytes. (C) Body
weights of ferrichrome (10 mg/kg)- or 5-FU (10 mg/kg)-treated mice
were not significantly altered following an administration period
of 14 days. (D) Complete blood count was not significantly altered
in the ferrichrome group, but the white and red blood cell,
hemoglobin and hematocrit values were significantly reduced in the
5-FU group. (E) Plasma ferritin levels were significantly increased
in the 5-FU group but not in the ferrichrome group. Data are
presented as the mean ± SD. Data were analyzed using Student's
t-test, or one-way or mixed ANOVA followed by Bonferroni's post hoc
test. *P<0.05 vs. PBS or as indicated. CRE,
creatinine; TP, total protein; ALB, albumin; AST, aspartate
aminotransferase; ALT, alanine aminotransferase; 5-FU,
5-fluorouracil; ns, not significant. |
To compare the safety of ferrichrome to 5-FU in
vivo, body weight, survival rates and CBC were assessed in mice
treated with PBS, ferrichrome (10 mg/kg) or 5-FU (10 mg/kg) via
daily tail vein injection for 14 days. No cases of animal mortality
were observed, and body weight was not markedly changed by
ferrichrome or 5-FU treatment (Fig.
5C). The CBC was not significantly altered in the ferrichrome
group compared with the PBS group, whereas the leukocyte,
erythrocyte, hemoglobin and hematocrit counts were significantly
reduced in the 5-FU group compared with the PBS group (Fig. 5D). To assess the presence of tissue
injury and ferric abnormality, plasma ferritin was evaluated.
Plasma ferritin levels were significantly increased in the 5-FU
group compared with the PBS group, but not in the ferrichrome group
(Fig. 5E). These data indicated
that ferrichrome did not induce anemia, myelosuppression or tissue
injury in the therapeutic range in mice. These findings suggested
that ferrichrome exerted tumor-suppressive effects in pancreatic
cancer cells with no notable adverse events.
Discussion
The present study revealed for the first time, to
the best of the authors' knowledge, that probiotic-derived
ferrichrome exhibited antitumor effects in pancreatic cancer cells
in vitro and in vivo. Notably, the tumor-suppressive
effects of ferrichrome were equal to those of 5-FU in SUIT-2 cells
and superior to those of 5-FU in FUR cells, indicating the strong
efficacy of ferrichrome in the treatment of chemotherapy-naive as
well as FUR pancreatic cancer. Furthermore, daily intravenous
injection of ferrichrome was not associated with adverse events in
the organs of mice. The results of the present study suggested that
probiotic-derived ferrichrome may a useful and safe agent for the
treatment of pancreatic cancer, which is frequently resistant to
existing antitumor drugs.
A previous study indicated that microbiome diversity
is strongly associated with the progression of gastrointestinal
cancers, including pancreatic cancer (6,7).
Michaud et al (26) and Fan
et al (27), reported high
levels of Porphyromonas gingi- valis in patients with
pancreatic cancer. Pushalkar et al (28) found that Proteobacteria was
the major genus of gut bacteria in patients with pancreatic cancer.
Certain bacteria have recently been detected in pancreatic cyst
fluids in intraductal papillary mucosal neoplasms, as well as
non-neoplastic cysts (29). These
microbial modifications are proposed to induce dysbiosis, thereby
promoting tumor progression due to immunological abnormalities
(30). In contrast, the present
study using pancreatic cancer cells reported that probiotic-derived
ferrichrome directly suppressed tumor progression, suggesting that
probiotics stably exert an antitumor effect regardless of
intestinal conditions or immunological status. Further analyses are
required to identify other probiotic antitumor molecules and
clarify the role of these molecules, thereby uncovering novel
mechanisms underlying the antitumor functions of probiotics in
pancreatic cancer, as well as other organ cancers.
Flow cytometry revealed that ferrichrome inhibited
the entry of pancreatic cancer cells into the G2-M phase. Likewise,
TUNEL staining and western blotting of cleaved PARP showed that
ferrichrome induced apoptosis in SUIT-2 cells. Subsequently, the
status of cell cycle-associated molecules, including p53, securin,
CDKN1B, and cyclins B1 and D1, were examined, and ferrichrome
treatment was determined to significantly induce the
phosphorylation of p53, and downregulation of securin and cyclin
B1, but not CDKN1B or cyclin D1, which are associated with
progression to the G1 phase (24).
p53 activation, but not reductions in cyclin B1, was observed in
other pancreatic cancer cells (MIA PaCa-II and PCI-43), indicating
that activation of p53 was a key mechanism via which ferrichrome
exerted its antitumor effects, and that downstream events of p53
activation depended on the characteristics of pancreatic cancer
cells. Notably, the p53 inhibitor pifithrin-µ repressed the
antitumor effects of ferrichrome. Furthermore, transcriptome
analysis showed that the expression of p53-associated mRNAs was
altered by ferrichrome treatment. These data clearly indicated that
p53 phosphorylation mediated the antitumor effects of ferrichrome
in pancreatic cancer cells. It is widely reported that p53 is a
pivotal gatekeeper molecule of the cell cycle in various types of
mammalian cells, including pancreatic cells (31,32).
It has also been reported that an ER-associated molecule, scotin,
induced p53 activation under excessive endoplasmic reticulum (ER)
stress, thereby inducing cell apoptosis (33). Our previous studies have reported
that ferrichrome upregulated ER stress, leading to the activation
of the JNK-DDIT3 pathway, and subsequent induction of apoptosis in
colorectal and gastric cancer cells (19,20).
Collectively, probiotic-derived ferrichrome is hypothesized to
expose cancer cells to ER stress, and thereby induce apoptosis via
the activation of ER stress-associated pathways, including
JNK-DDIT3 and/or p53, which is a novel mechanism of the antitumor
function of probiotic bacteria.
An SRB assay indicated that the antitumor effects of
ferri-chrome were not completely abolished by the p53 functional
inhibitor pifithrin-µ. Pifithrin-µ was administered
at 1 µg/ml as higher concentrations completely inhibited the
growth of SUIT-2 cells. The p53-inhibitory effect of
pifithrin-µ was observed to be partial at 1 µg/ml,
and p53 with its original properties as a tumor suppressor gene was
activated by ferrichrome treatment, resulting in the p53 pathway
being partially functional during ferrichrome treatment. It was
hypothesized that other pathways associated with cell growth may
also continue to function under ferrichrome treatment.
Transcriptome analysis also indicated that ferrichrome induced the
expression of apoptosis-inducible factors, including CHAC1 and
DDIT4, suggesting that other pathways in addition to the p53
pathway were activated and may induce tumor-suppressive effects
following ferrichrome treatment of pancreatic cancer cells.
Transcriptome and gene functional annotation
analysis of ferrichrome-treated SUIT-2 cells showed that 319 genes
were iron-related, and that the expression of 10 iron-related genes
was significantly altered by ferrichrome treatment. As ferrichrome
interacts with iron ions, it is important to assess the
relationship between the antitumor effects of ferrichrome and
expression changes in these 10 genes. These molecules are
classified by CLC Genomics Workbench software as 'drug-degrading
enzymes containing iron ions' (CYP7A1, CYP3A7-CYP3A51P, CYP2C18,
MIOX and RFESD), 'indicators of Rh blood type' (RHAG), 'iron
transporters' (SLC40A1), 'nitric oxide synthases' (NOS3) and 'DNA
damage repair molecules' (MUTYH and EXO5). Previous studies
reported that the downregulation of SLC40A1, NOS3 and MUTYH
(34-37) promoted tumor cell progression,
suggesting that these genes worked as tumor-suppressor genes. In
the present study, it was shown that ferrichrome induced antitumor
effects in pancreatic cancer cells, whereas these tumor-suppressive
genes were downregulated. Therefore, changes in these
iron-associated genes are not essential for ferrichrome to
function, suggesting that the contributions of iron-related genes
are secondary in ferrichrome-treated cells.
It was shown in the present study that the antitumor
effects of ferrichrome were equal to or greater than those of 5-FU
via intraperitoneal injection. To assess the safety of ferrichrome
in pancreatic cancer treatment, the CBC, plasma ferritin and serum
biochemical test values (CRE, TP, ALB, AST, ALT, iron,
electrolytes) of mice treated by ferrichrome or 5-FU via
intravenous injection for 14 days were checked. The test values of
ferrichrome-treated mice did not show any abnormal changes. Of
note, the CBC was not markedly altered by the intravenous injection
of ferrichrome; however, the leukocyte, erythrocyte, hemoglobin and
hematocrit values of 5-FU-treated mice were significantly
decreased. Plasma ferritin levels were significantly increased in
the 5-FU group but not in the ferrichrome group, suggesting that
ferrichrome did not influence ferritin production. Likewise,
abnormal histological changes were not detected in the
ferrichrome-treated mice. These data indicated that the safety and
therapeutic efficacy of ferrichrome were superior to those of
classical antitumor drugs, such as 5-FU.
In FUR SUIT-2 cells, ferrichrome inhibited growth to
the same degree as was observed in 5-FU-sensitive SUIT-2 cells. In
cancer cells, 5-FU is known to bind to thymidylate synthase,
inhibit DNA synthesis and thereby inhibit progression to the S
phase (38). The present study
showed that ferrichrome inhibited the progression of cancer cells
to the G2-M phase via the upregulation of p53, and downregulation
of securin and cyclin B1. These data indicated that the mechanisms
underlying the antitumor effects of ferrichrome differ from those
of 5-FU, suggesting that ferrichrome could be used clinically as an
anti-tumor drug for the treatment of pancreatic cancer that shows
resistance to existing drugs.
In conclusion, it was revealed the antitumor effects
of probiotic-derived ferrichrome in pancreatic cancer cells,
including FUR cells. The mechanism via which ferrichrome suppressed
cancer cells appeared to involve the induction of cancer cell
apoptosis via p53 upregulation, which differs from the mechanisms
of existing drugs. These findings indicated that probiotics are
associated with pancreatic tumor progression, and probiotic-derived
ferrichrome is expected to be a novel attractive ant-tumor agent
for the treatment of refractory pancreatic cancer.
Supplementary Data
Abbreviations:
|
5-FU
|
5-fluorouracil
|
|
PARP
|
poly (ADP-ribose) polymerase
|
|
DDIT3
|
DNA damage inducible transcript 3
|
|
DMEM
|
Dulbecco's modified Eagle's medium
|
|
SRB
|
sulforhodamine B
|
|
CDKN1B
|
cyclin-dependent kinase inhibitor
1B
|
|
FUR
|
5-FU-resistant
|
|
CRE
|
creatinine
|
|
TP
|
total protein
|
|
ALB
|
albumin
|
|
AST
|
aspartate aminotransferase
|
|
ALT
|
alanine aminotransferase
|
|
ER
|
endoplasmic reticulum
|
Acknowledgments
We thank Ms. Kotoe Shibusa (Division of
Gastroenterology and Hematology/Oncology, Department of Medicine,
Asahikawa Medical University) for technical assistance, and Mr.
Hiroaki Akutsu and Mr. Shinichi Chiba (Center for Advanced Research
and Education Asahikawa Medical University) for assistance with
flow cytometric analysis and genetic analysis. CBC and serum
biochemistry analyses were performed by New Drug Research Center,
Inc. and Oriental Yeast Co., Ltd., respectively.
Funding
The present study was supported by Grants-in-Aid for
Scientific Research supported by Japan Society for the Promotion of
Science (grant nos. 18K07927, 19K16484, 18K15770, 18K08906,
17K15913, 19K17419 and 19K08410) and the Takeda Science Foundation.
This study was also supported by research funds from Kamui Pharma,
Inc., Mochida Pharmaceutical Co., Ltd., Nippon Kayaku Co., Ltd.,
and EA Pharma Co., Ltd used to purchase experimental agents and
mice.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
AK, MF and HK were substantially involved in the
conception and design of the study, drafted the manuscript and
supervised all experiments. AK and HK performed the biochemical
experiments. HT performed histopathological assessments. SK, TI,
MI, YM, ST, TG, AS, KA NU, NO and TO were involved in the design of
the study, the interpretation of the data, and preparation and
review of the manuscript. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The animal experiments were approved by the
Asahikawa Medical University Institutional Animal Care and Use
Committee.
Patient consent for publication
Not applicable.
Competing interests
Mikihiro Fujiya received research funds from Kamui
Pharma, Inc., Mochida Pharmaceutical Co., Ltd., Nippon Kayaku Co.,
Ltd., and EA Pharma Co., Ltd., to purchase experimental agents and
mice.
References
|
1
|
Conroy T, Desseigne F, Ychou M, Bouché O,
Guimbaud R, Bécouarn Y, Adenis A, Raoul JL, Gourgou-Bourgade S, de
la Fouchardière C, et al Groupe Tumeurs Digestives of Unicancer;
PRODIGE Intergroup: FOLFIRINOX versus gemcitabine for metastatic
pancreatic cancer. N Engl J Med. 364:1817–1825. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Heinemann V, Boeck S, Hinke A, Labianca R
and Louvet C: Meta-analysis of randomized trials: Evaluation of
benefit from gemcitabine-based combination chemotherapy applied in
advanced pancreatic cancer. BMC Cancer. 8:822008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chand S, O'Hayer K, Blanco FF, Winter JM
and Brody JR: The landscape of pancreatic cancer therapeutic
resistance mechanisms. Int J Biol Sci. 12:273–282. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Loehrer PJ Sr, Einhorn LH, Williams SD,
Hui SL, Estes NC and Pennington K: Cisplatin plus 5-FU for the
treatment of adeno-carcinoma of the colon. Cancer Treat Rep.
69:1359–1363. 1985.PubMed/NCBI
|
|
6
|
Gaiser RA, Halimi A, Alkharaan H, Lu L,
Davanian H, Healy K, Hugerth LW, Ateeb Z, Valente R, Fernández Moro
C, et al: Enrichment of oral microbiota in early cystic precursors
to invasive pancreatic cancer. Gut. 68:2186–2194. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Half E, Keren N, Reshef L, Dorfman T,
Lachter I, Kluger Y, Reshef N, Knobler H, Maor Y, Stein A, et al:
Fecal microbiome signatures of pancreatic cancer patients. Sci Rep.
9:168012019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Food and Agriculture Organization of the
United Nations; World Health Organization: Guidelines for the
Evaluation of Probiotics in Food. Ontario, Canada: 2002
|
|
9
|
Gerritsen J, Smidt H, Rijkers GT and de
Vos WM: Intestinal microbiota in human health and disease: The
impact of probiotics. Genes Nutr. 6:209–240. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kalliomäki M, Salminen S, Arvilommi H,
Kero P, Koskinen P and Isolauri E: Probiotics in primary prevention
of atopic disease: A randomised placebo-controlled trial. Lancet.
357:1076–1079. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gu Y-H, Choi H, Yamashita T, Kang KM,
Iwasa M, Lee MJ, Lee KH and Kim CH: Pharmaceutical production of
anti-tumor and immune-potentiating Enterococcus faecalis-2001
β-glucans: Enhanced activity of macrophage and lymphocytes in
tumor-implanted mice. Curr Pharm Biotechnol. 18:653–661. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rowland IR, Rumney CJ, Coutts JT and
Lievense LC: Effect of Bifidobacterium longum and inulin on gut
bacterial metabolism and carcinogen-induced aberrant crypt foci in
rats. Carcinogenesis. 19:281–285. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Maroof H, Hassan ZM, Mobarez AM and
Mohamadabadi MA: Lactobacillus acidophilus could modulate the
immune response against breast cancer in murine model. J Clin
Immunol. 32:1353–1359. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Singhal B, Mukherjee A and Srivastav S:
Role of probiotics in pancreatic cancer prevention: The prospects
and challenges. Adv Biosci Biotechnol. 07:468–500. 2016. View Article : Google Scholar
|
|
15
|
Segawa S, Fujiya M, Konishi H, Ueno N,
Kobayashi N, Shigyo T and Kohgo Y: Probiotic-derived polyphosphate
enhances the epithelial barrier function and maintains intestinal
homeostasis through integrin-p38 MAPK pathway. PLoS One.
6:e232782011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hemarajata P and Versalovic J: Effects of
probiotics on gut microbiota: Mechanisms of intestinal
immunomodulation and neuromodulation. Therap Adv Gastroenterol.
6:39–51. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tsai TL, Li AC, Chen YC, Liao YS and Lin
TH: Antimicrobial peptide m2163 or m2386 identified from
Lactobacillus casei ATCC 334 can trigger apoptosis in the human
colorectal cancer cell line SW480. Tumour Biol. 36:3775–3789. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sakatani A, Fujiya M, Ueno N, Kashima S,
Sasajima J, Moriichi K, Ikuta K, Tanabe H and Kohgo Y:
Polyphosphate derived from lactobacillus brevis inhibits colon
cancer progression through induction of cell apoptosis. Anticancer
Res. 36:591–598. 2016.PubMed/NCBI
|
|
19
|
Konishi H, Fujiya M, Tanaka H, Ueno N,
Moriichi K, Sasajima J, Ikuta K, Akutsu H, Tanabe H and Kohgo Y:
Probiotic-derived ferrichrome inhibits colon cancer progression via
JNK-mediated apoptosis. Nat Commun. 7:123652016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ijiri M, Fujiya M, Konishi H, Tanaka H,
Ueno N, Kashima S, Moriichi K, Sasajima J, Ikuta K and Okumura T:
Ferrichrome identified from Lactobacillus casei ATCC334 induces
apoptosis through its iron-binding site in gastric cancer cells.
Tumour Biol. 39:10104283177113112017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ecker DJ, Passavant CW and Emery T: Role
of two siderophores in Ustilago sphaerogena. Regulation of
biosynthesis and uptake mechanisms. Biochim Biophys Acta.
720:242–249. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kato H, Ishikura H, Kawarada Y, Furuya M,
Kondo S, Kato H and Yoshiki T: Anti-angiogenic treatment for
peritoneal dissemination of pancreas adenocarcinoma: A study using
TNP-470. Jpn J Cancer Res. 92:67–73. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Li Z, Wang N, Huang C, Bao Y, Jiang Y and
Zhu G: Downregulation of caveolin-1 increases the sensitivity of
drug-resistant colorectal cancer HCT116 cells to 5-fluorouracil.
Oncol Lett. 13:483–487. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Moore JD: In the wrong place at the wrong
time: Does cyclin mislocalization drive oncogenic transformation?
Nat Rev Cancer. 13:201–208. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Leu JIJ, Pimkina J, Frank A, Murphy ME and
George DL: A small molecule inhibitor of inducible heat shock
protein 70. Mol Cell. 36:15–27. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Michaud DS, Izard J, Wilhelm-Benartzi CS,
You DH, Grote VA, Tjønneland A, Dahm CC, Overvad K, Jenab M,
Fedirko V, et al: Plasma antibodies to oral bacteria and risk of
pancreatic cancer in a large European prospective cohort study.
Gut. 62:1764–1770. 2013. View Article : Google Scholar
|
|
27
|
Fan X, Alekseyenko AV, Wu J, Peters BA,
Jacobs EJ, Gapstur SM, Purdue MP, Abnet CC, Stolzenberg-Solomon R,
Miller G, et al: Human oral microbiome and prospective risk for
pancreatic cancer: A population-based nested case-control study.
Gut. 67:120–127. 2018. View Article : Google Scholar
|
|
28
|
Pushalkar S, Hundeyin M, Daley D,
Zambirinis CP, Kurz E, Mishra A, Mohan N, Aykut B, Usyk M, Torres
LE, et al: The pancreatic cancer microbiome promotes oncogenesis by
induction of innate and adaptive immune suppression. Cancer Discov.
8:403–416. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li S, Fuhler GM, Bn N, Jose T, Bruno MJ,
Peppelenbosch MP and Konstantinov SR: Pancreatic cyst fluid harbors
a unique microbiome. Microbiome. 5:1472017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sethi V, Kurtom S, Tarique M, Lavania S,
Malchiodi Z, Hellmund L, Zhang L, Sharma U, Giri B, Garg B, et al:
Gut microbiota promotes tumor growth in mice by modulating immune
response. Gastroenterology. 155:33–37.e6. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kastan MB, Zhan Q, el-Deiry WS, Carrier F,
Jacks T, Walsh WV, Plunkett BS, Vogelstein B and Fornace AJ Jr: A
mammalian cell cycle checkpoint pathway utilizing p53 and GADD45 is
defective in ataxia-telangiectasia. Cell. 71:587–597. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kuerbitz SJ, Plunkett BS, Walsh WV and
Kastan MB: Wild-type p53 is a cell cycle checkpoint determinant
following irradiation. Proc Natl Acad Sci USA. 89:7491–7495. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bourdon JC, Renzing J, Robertson PL,
Fernandes KN and Lane DP: Scotin, a novel p53-inducible
proapoptotic protein located in the ER and the nuclear membrane. J
Cell Biol. 158:235–246. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Gu Z, Wang H, Xia J, Yang Y, Jin Z, Xu H,
Shi J, De Domenico I, Tricot G and Zhan F: Decreased ferroportin
promotes myeloma cell growth and osteoclast differentiation. Cancer
Res. 75:2211–2221. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Markkanen E, Dorn J and Hübscher U: MUTYH
DNA glyco-sylase: The rationale for removing undamaged bases from
the DNA. Front Genet. 4:182013. View Article : Google Scholar
|
|
36
|
Oka S, Leon J, Tsuchimoto D, Sakumi K and
Nakabeppu Y: MUTYH, an adenine DNA glycosylase, mediates p53 tumor
suppression via PARP-dependent cell death. Oncogenesis. 3:e121.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ali S, Zhang Y, Zhou M, Li H, Jin W, Zheng
L, Yu X, Stark JM, Weitzel JN and Shen B: Functional deficiency of
DNA repair gene EXO5 results in androgen-induced genomic
instability and prostate tumorigenesis. Oncogene. 39:1246–1259.
2020. View Article : Google Scholar
|
|
38
|
Longley DB, Harkin DP and Johnston PG:
5-fluorouracil: Mechanisms of action and clinical strategies. Nat
Rev Cancer. 3:330–338. 2003. View Article : Google Scholar : PubMed/NCBI
|