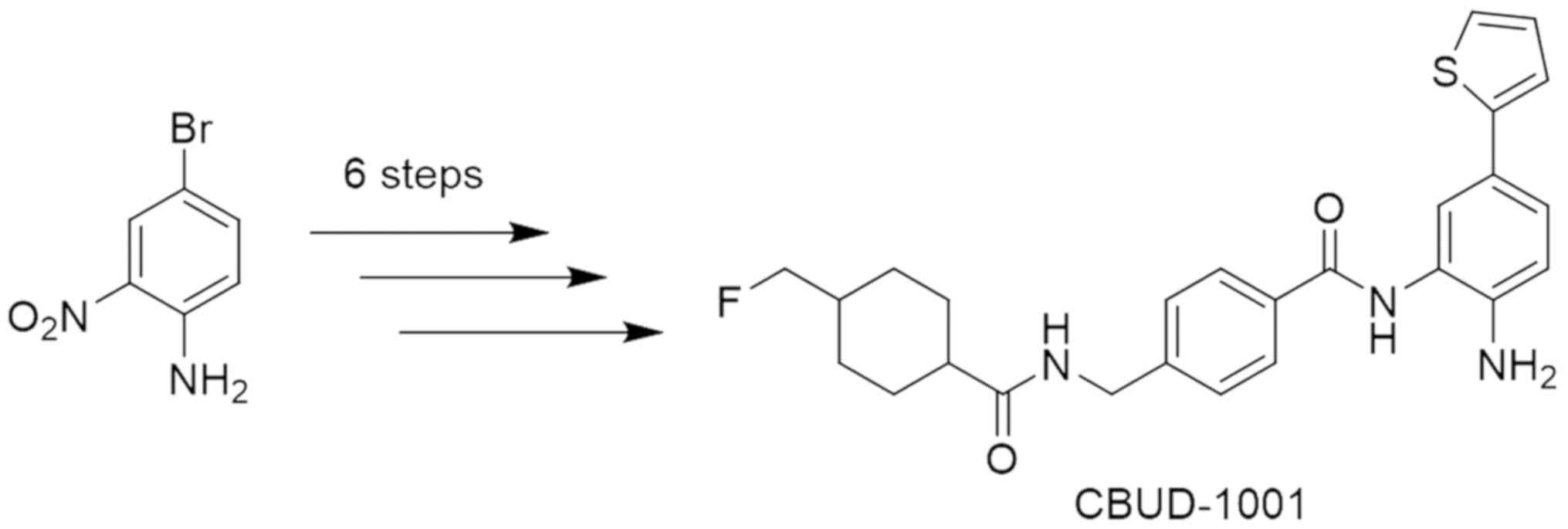
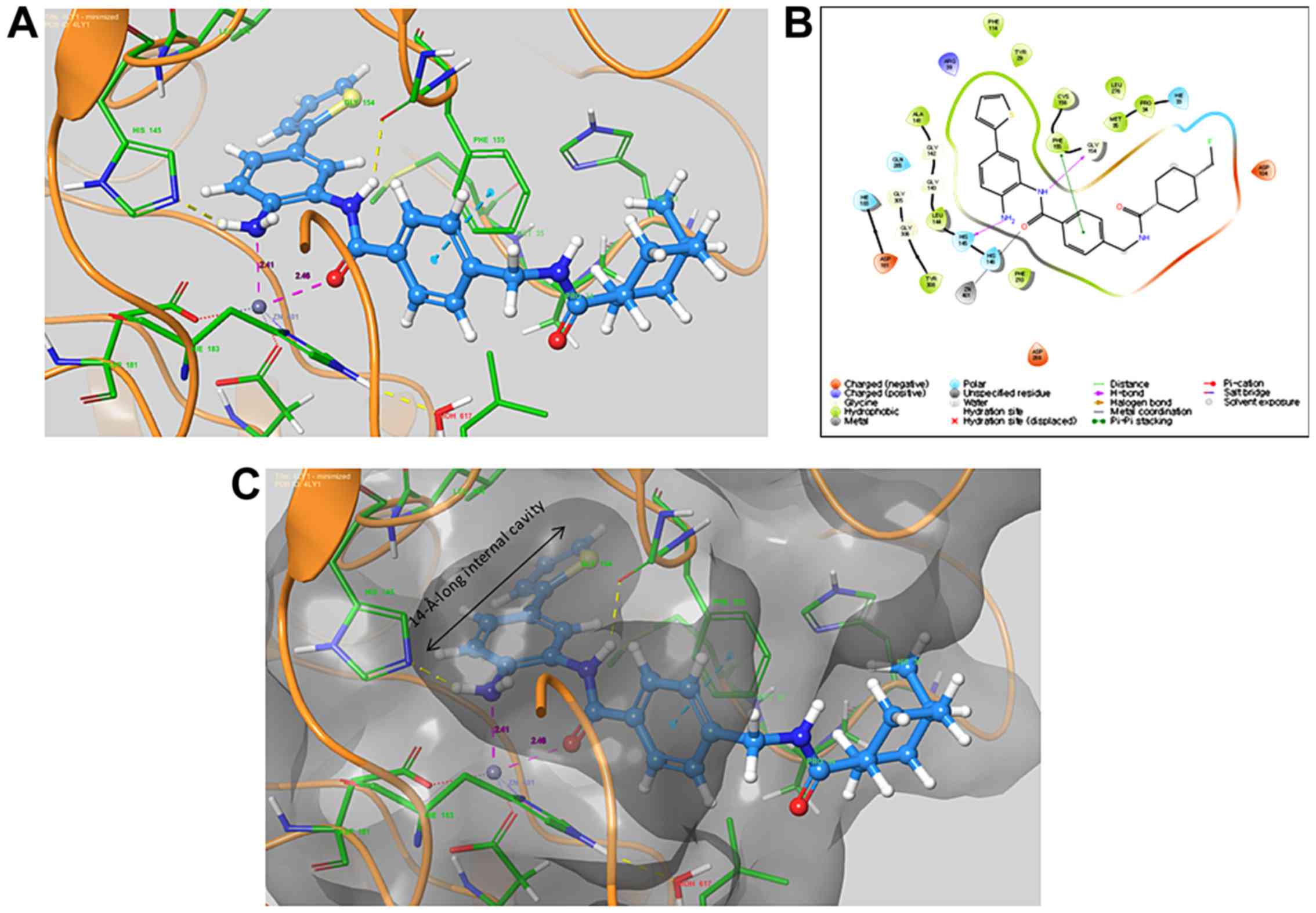
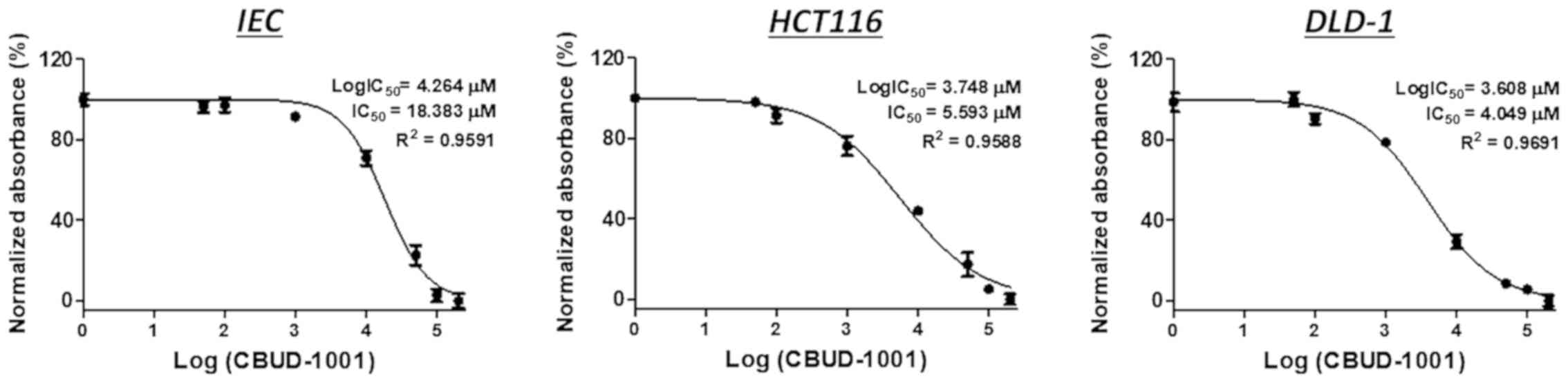
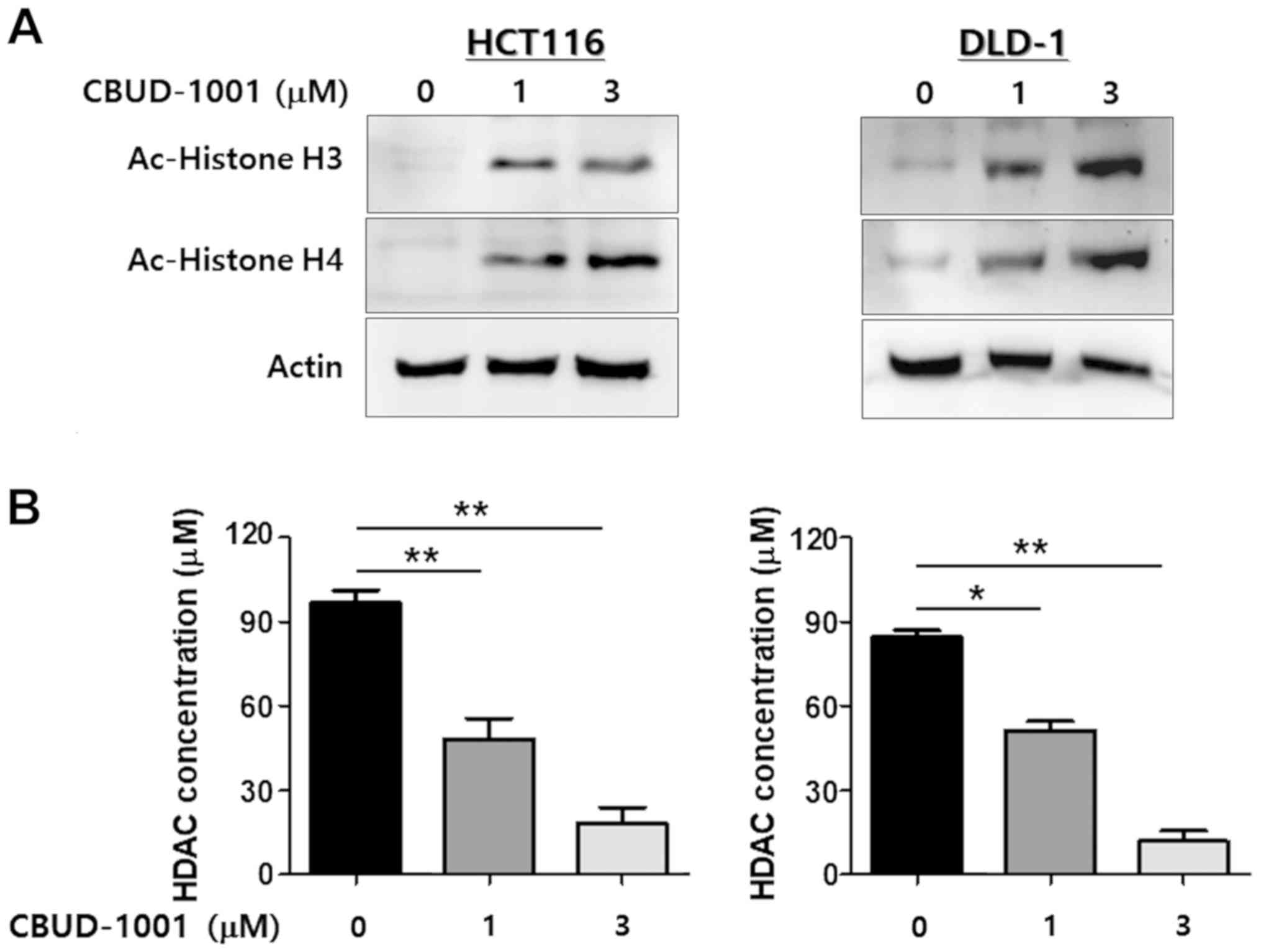
|
1
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar
|
|
2
|
Arnold M, Sierra MS, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global patterns and trends in
colorectal cancer incidence and mortality. Gut. 66:683–691. 2017.
View Article : Google Scholar
|
|
3
|
Haggar FA and Boushey RP: Colorectal
cancer epidemiology: Incidence, mortality, survival, and risk
factors. Clin Colon Rectal Surg. 22:191–197. 2009. View Article : Google Scholar
|
|
4
|
Lao VV and Grady WM: Epigenetics and
colorectal cancer. Nat Rev Gastroenterol Hepatol. 8:686–700. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bertrand P: Inside HDAC with HDAC
inhibitors. Eur J Med Chem. 45:2095–2116. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jackson MD and Denu JM: Structural
identification of 2′- and 3′-O-acetyl-ADP-ribose as novel
metabolites derived from the Sir2 family of beta-NAD+-dependent
histone/protein deacetylases. J Biol Chem. 277:18535–18544. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lombardi PM, Cole KE, Dowling DP and
Christianson DW: Structure, mechanism, and inhibition of histone
deacetylases and related metalloenzymes. Curr Opin Struct Biol.
21:735–743. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Haberland M, Montgomery RL and Olson EN:
The many roles of histone deacetylases in development and
physiology: Implications for disease and therapy. Nat Rev Genet.
10:32–42. 2009. View
Article : Google Scholar
|
|
9
|
Kelly RD and Cowley SM: The physiological
roles of histone deacetylase (HDAC) 1 and 2: Complex co-stars with
multiple leading parts. Biochem Soc Trans. 41:741–749. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lagger G, Doetzlhofer A, Schuettengruber
B, Haidweger E, Simboeck E, Tischler J, Chiocca S, Suske G,
Rotheneder H, Wintersberger E and Seiser C: The tumor suppressor
p53 and histone deacetylase 1 are antagonistic regulators of the
cyclin-dependent kinase inhibitor p21/WAF1/CIP1 gene. Mol Cell
Biol. 23:2669–2679. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ropero S and Esteller M: The role of
histone deacetylases (HDACs) in human cancer. Mol Oncol. 1:19–25.
20072007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hill R, Bodzak E, Blough MD and Lee PW:
p53 Binding to the p21 promoter is dependent on the nature of DNA
damage. Cell Cycle. 7:2535–2543. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li Y and Seto E: HDACs and HDAC inhibitors
in cancer development and therapy. Cold Spring Harb Perspect Med.
6:a0268312016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhu P, Martin E, Mengwasser J, Schlag P,
Janssen KP and Göttlicher M: Induction of HDAC2 expression upon
loss of APC in colorectal tumorigenesis. Cancer Cell. 5:455–463.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Stypula-Cyrus Y, Damania D, Kunte DP, Cruz
MD, Subramanian H, Roy HK and Backman V: HDAC up-regulation in
early colon field carcinogenesis is involved in cell tumorigenicity
through regulation of chromatin structure. PLoS One. 8:e646002013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Spaderna S, Schmalhofer O, Hlubek F, Berx
G, Eger A, Merkel S, Jung A, Kirchner T and Brabletz T: A
transient, EMT-linked loss of basement membranes indicates
metastasis and poor survival in colorectal cancer.
Gastroenterology. 131:830–840. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Barrallo-Gimeno A and Nieto MA: The Snail
genes as inducers of cell movement and survival: Implications in
development and cancer. Development. 132:3151–3161. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Peinado H, Ballestar E, Esteller M and
Cano A: Snail mediates E-cadherin repression by the recruitment of
the Sin3A/histone deacetylase 1 (HDAC1)/HDAC2 complex. Mol Cell
Biol. 24:306–319. 2004. View Article : Google Scholar
|
|
19
|
Jurkin J, Zupkovitz G, Lagger S,
Grausenburger R, Hagelkruys A, Kenner L and Seiser C: Distinct and
redundant functions of histone deacetylases HDAC1 and HDAC2 in
proliferation and tumorigenesis. Cell Cycle. 10:406–412. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mariadason JM: HDACs and HDAC inhibitors
in colon cancer. Epigenetics. 3:28–37. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sun PC, Tzao C, Chen BH, Liu CW, Yu CP and
Jin JS: Suberoylanilide hydroxamic acid induces apoptosis and
sub-G1 arrest of 320 HSR colon cancer cells. J Biomed Sci.
17:762010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang TY, Jia YL, Zhang X, Sun QL, Li YC,
Zhang JH, Zhao CP, Wang XY and Wang L: Treating colon cancer cells
with FK228 reveals a link between histone lysine acetylation and
extensive changes in the cellular proteome. Sci Rep. 5:184432015.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhijun H, Shusheng W, Han M, Jianping L,
Li-Sen Q and Dechun L: Pre-clinical characterization of 4SC-202, a
novel class I HDAC inhibitor, against colorectal cancer cells.
Tumour Biol. 37:10257–10267. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ji M, Lee EJ, Kim KB, Kim Y, Sung R, Lee
SJ, Kim DS and Park SM: HDAC inhibitors induce
epithelial-mesenchymal transition in colon carcinoma cells. Oncol
Rep. 33:2299–2308. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Patel MM and Patel BM: Repurposing of
sodium valproate in colon cancer associated with diabetes mellitus:
Role of HDAC inhibition. Eur J Pharm Sci. 121:188–199. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bracker TU, Sommer A, Fichtner I, Faus H,
Haendler B and Hess-Stumpp H: Efficacy of MS-275, a selective
inhibitor of class I histone deacetylases, in human colon cancer
models. Int J Oncol. 35:909–920. 2009.PubMed/NCBI
|
|
27
|
Jin JS, Tsao TY, Sun PC, Yu CP and Tzao C:
SAHA inhibits the growth of colon tumors by decreasing histone
deacetylase and the expression of cyclin D1 and survivin. Pathol
Oncol Res. 18:713–7120. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chou CW, Wu MS, Huang WC and Chen CC: HDAC
inhibition decreases the expression of EGFR in colorectal cancer
cells. PLoS One. 6:e180872011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Khan N, Jeffers M, Kumar S, Hackett C,
Boldog F, Khramtsov N, Qian X, Mills E, Berghs SC, Carey N, et al:
Determination of the class and isoform selectivity of
small-molecule histone deacetylase inhibitors. Biochem J.
409:581–589. 2008. View Article : Google Scholar
|
|
30
|
Kelly WK, O'Connor OA, Krug LM, Chiao JH,
Heaney M, Curley T, MacGregore-Cortelli B, Tong W, Secrist JP,
Schwartz L, et al: Phase I study of an oral histone deacetylase
inhibitor, suberoylanilide hydroxamic acid, in patients with
advanced cancer. J Clin Oncol. 23:3923–3931. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Flis S, Gnyszka A, Flis K and Spławiński
J: MS275 enhances cytotoxicity induced by 5-fluorouracil in the
colorectal cancer cells. Eur J Pharmacol. 627:26–32. 2010.
View Article : Google Scholar
|
|
32
|
Zhang L, Zhang J, Jiang Q, Zhang L and
Song W: Zinc binding groups for histone deacetylase inhibitors. J
Enzyme Inhib Med Chem. 33:714–721. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Lu H, Chen YD, Yang B and You QD: Design,
synthesis and biological evaluation of novel histone deacetylase
inhibitors based on virtual screening. Acta Pharmaceutica Sinica B.
1:240–247. 2011. View Article : Google Scholar
|
|
34
|
Vaisburg A, Paquin I, Bernstein N,
Frechette S, Gaudette F, Leit S, Moradei O, Raeppel S, Zhou N,
Bouchain G, et al:
N-(2-Amino-phenyl)-4-(heteroarylmethyl)-benzamides as new histone
deacetylase inhibitors. Bioorg Med Chem Lett. 17:6729–6733. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Methot JL, Chakravarty PK, Chenard M,
Close J, Cruz JC, Dahlberg WK, Fleming J, Hamblett CL, Hamill JE,
Harrington P, et al: Exploration of the internal cavity of histone
deacetylase (HDAC) with selective HDAC1/HDAC2 inhibitors (SHI-1:
2). Bioorg Med Chem Lett. 18:973–978. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lauffer BEL, Mintzer R, Fong RN, Mukund S,
Tam C, Zilberleyb I, Flicke B, Ritscher A, Fedorowicz G, Vallero R,
et al: Histone deacetylase (HDAC) inhibitor kinetic rate constants
correlate with cellular histone acetylation but not transcription
and cell viability. J Biol Chem. 288:26926–26943. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zamzami N, Marchetti P, Castedo M, Zanin
C, V Vayssière JL, Petit PX and Kroemer G: Reduction in
mitochondrial potential constitutes an early irreversible step of
programmed lymphocyte death in vivo. J Exp Med. 181:1661–1672.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Seto E and Yoshida M: Erasers of histone
acetylation: The histone deacetylase enzymes. Cold Spring Harb
Perspect Biol. 6:a0187132014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Spurling CC, Godman CA, Noonan EJ,
Rasmussen TP, Rosenberg DW and Giardina C: HDAC3 overexpression and
colon cancer cell proliferation and differentiation. Mol Carcinog.
47:137–147. 2008. View Article : Google Scholar
|
|
40
|
Chen HP, Zhao YT and Zhao TC: Histone
deacetylases and mechanisms of regulation of gene expression. Crit
Rev Oncog. 20:35–47. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Audia JE and Campbell RM: Histone
modifications and cancer. Cold Spring Harb Perspect Biol.
8:a0195212016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
De Souza C and Chatterji BP: HDAC
inhibitors as novel anti-cancer therapeutics. Recent Pat Anticancer
Drug Discov. 10:145–162. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Bozorgi AH, Bagheri M, Aslebagh R and
Rajabi MS: A structure-activity relationship survey of histone
deacetylase (HDAC) inhibitors. Chemometr Intell Lab. 125:132–138.
2013. View Article : Google Scholar
|
|
44
|
Huang BH, Laban M, Leung CH, Lee L, Lee
CK, Salto-Tellez M, Raju GC and Hooi SC: Inhibition of histone
deacetylase 2 increases apoptosis and p21Cip1/WAF1 expression,
independent of histone deacetylase 1. Cell Death Differ.
12:395–404. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Weichert W, Roske A, Niesporek S, Noske A,
Buckendahl AC, Dietel M, Gekeler V, Boehm M, Beckers T and Denkert
C: Class I histone deacetylase expression has independent
prognostic impact in human colorectal cancer: Specific role of
class I histone deacetylases in vitro and in vivo. Clin Cancer Res.
14:1669–1677. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Senese S, Zaragoza K, Minardi S, Muradore
I, Ronzoni S, Passafaro A, Bernard L, Draetta GF, Alcalay M, Seiser
C and Chiocca S: Role for histone deacetylase 1 in human tumor cell
proliferation. Mol Cell Biol. 27:4784–4795. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Glaser KB, Li J, Staver MJ, Wei RQ, Albert
DH and Davidsen SK: Role of class I and class II histone
deacetylases in carcinoma cells using siRNA. Biochem Biophys Res
Commun. 310:529–536. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Santoro F, Botrugno OA, Dal Zuffo R,
Pallavicini I, Matthews GM, Cluse L, Barozzi I, Senese S, Fornasari
L, Moretti S, et al: A dual role for Hdac1: Oncosuppressor in
tumorigenesis, oncogene in tumor maintenance. Blood. 121:3459–3468.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Huang L, Sowa Y, Sakai T and Pardee AB:
Activation of the p21WAF1/CIP1 promoter independent of p53 by the
histone deacetylase inhibitor suberoylanilide hydroxamic acid
(SAHA) through the Sp1 sites. Oncogene. 19:5712–5719. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Rikiishi H: Autophagic and apoptotic
effects of HDAC inhibitors on cancer cells. J Biomed Biotechnol.
2011:8302602011. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Green DR and Reed JC: Mitochondria and
apoptosis. Science. 281:1309–1312. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Yang E and Korsmeyer SJ: Molecular
thanatopsis: A discourse on the BCL2 family and cell death. Blood.
88:386–401. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Kroemer G and Reed JC: Mitochondrial
control of cell death. Nat Med. 6:513–519. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Adams JM and Cory S: The Bcl-2 protein
family: Arbiters of cell survival. Science. 281:1322–1326. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Gross A, McDonnell JM and Korsmeyer SJ:
BCL-2 family members and the mitochondria in apoptosis. Genes Dev.
13:1899–1911. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Lencz T, Guha S, Liu C, Rosenfeld J,
Mukherjee S, DeRosse P, John M, Cheng L, Zhang C, Badner JA, et al:
Genome-wide association study implicates NDST3 in schizophrenia and
bipolar disorder. Nat Commun. 4:27392013. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Eckelman BP, Salvesen GS and Scott FL:
Human inhibitor of apoptosis proteins: Why XIAP is the black sheep
of the family. EMBO Rep. 7:988–994. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Grutter MG: Caspases: Key players in
programmed cell death. Curr Opin Struct Biol. 10:649–655. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Lee JM, Dedhar S, Kalluri R and Thompson
EW: The epithelial-mesenchymal transition: New insights in
signaling, development, and disease. J Cell Biol. 172:973–981.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Zhang Y and Weinberg RA:
Epithelial-to-mesenchymal transition in cancer: Complexity and
opportunities. Front Med. 12:361–373. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Sakamoto T, Kobayashi S, Yamada D, Nagano
H, Tomokuni A, Tomimaru Y, Noda T, Gotoh K, Asaoka T, Wada H, et
al: A Histone deacetylase inhibitor suppresses
epithelial-mesenchymal transition and attenuates chemoresistance in
biliary tract cancer. PLoS One. 11:e01459852016. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Mateen S, Raina K, Agarwal C, Chan D and
Agarwal R: Silibinin synergizes with histone deacetylase and DNA
methyltransferase inhibitors in upregulating E-cadherin expression
together with inhibition of migration and invasion of human
non-small cell lung cancer cells. J Pharmacol Exp Ther.
345:206–214. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Feng J, Cen J, Li J, Zhao R, Zhu C, Wang
Z, Xie J and Tang W: Histone deacetylase inhibitor valproic acid
(VPA) promotes the epithelial mesenchymal transition of colorectal
cancer cells via up regulation of Snail. Cell Adh Migr. 9:495–501.
2015. View Article : Google Scholar : PubMed/NCBI
|