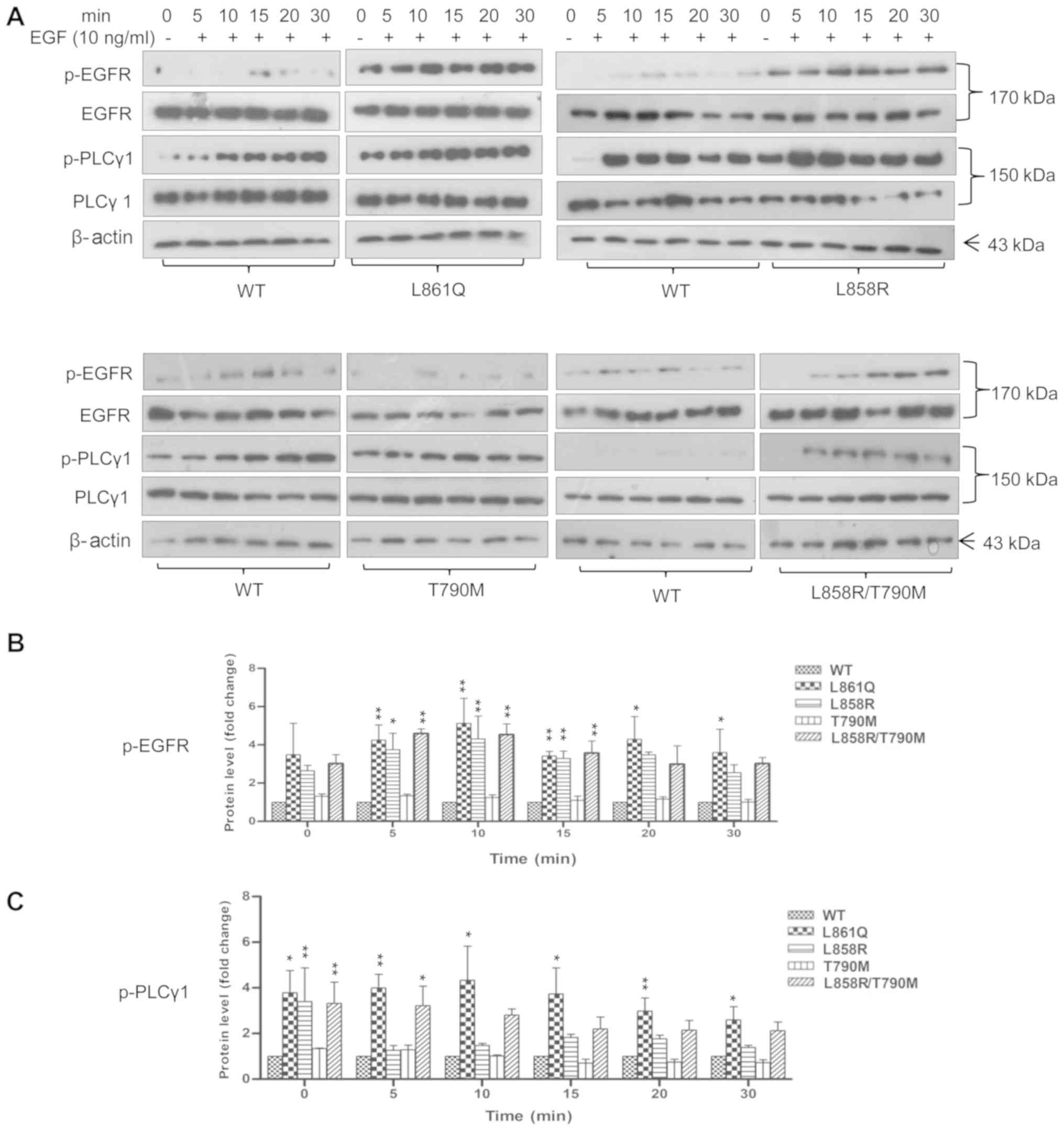
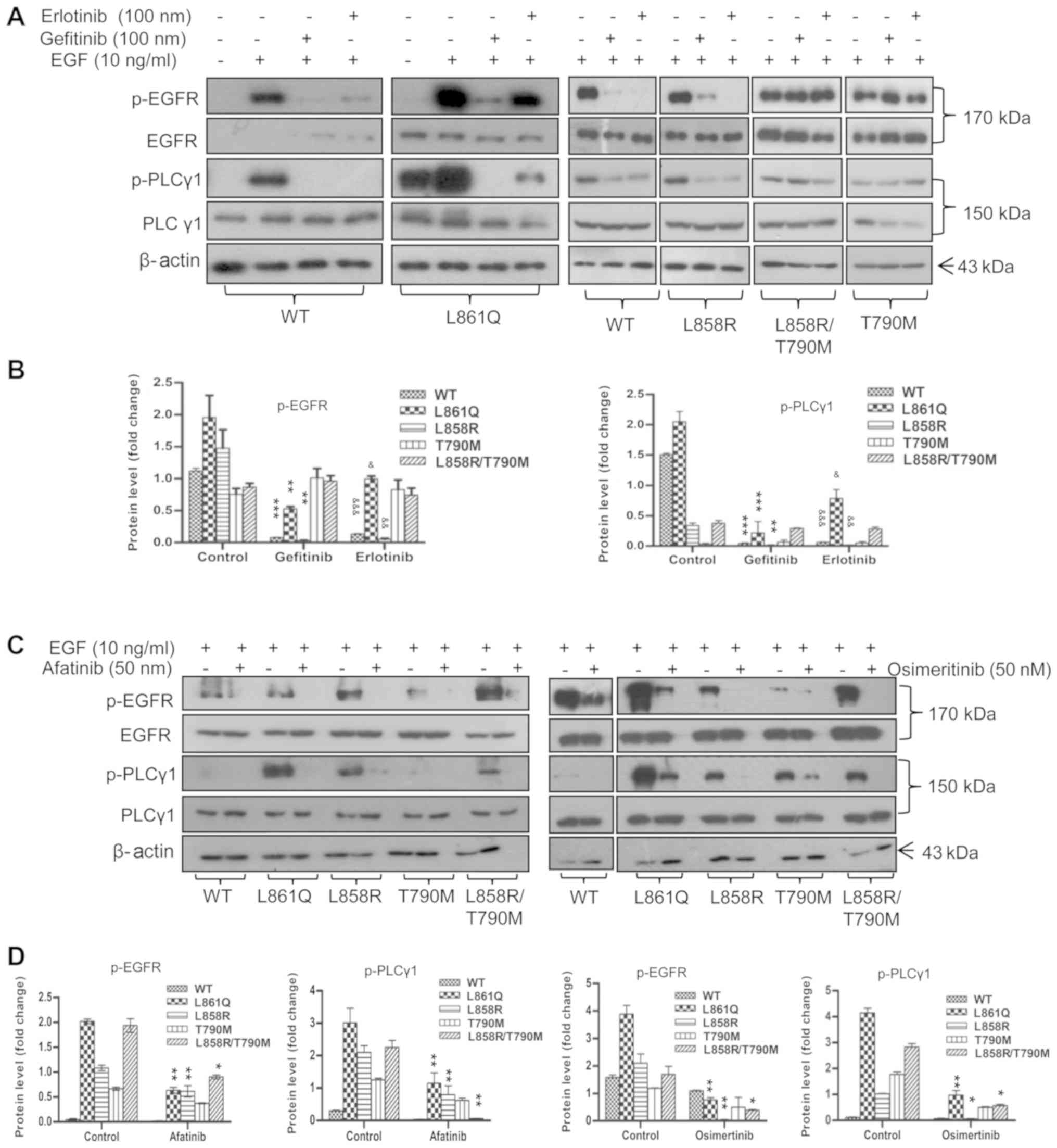
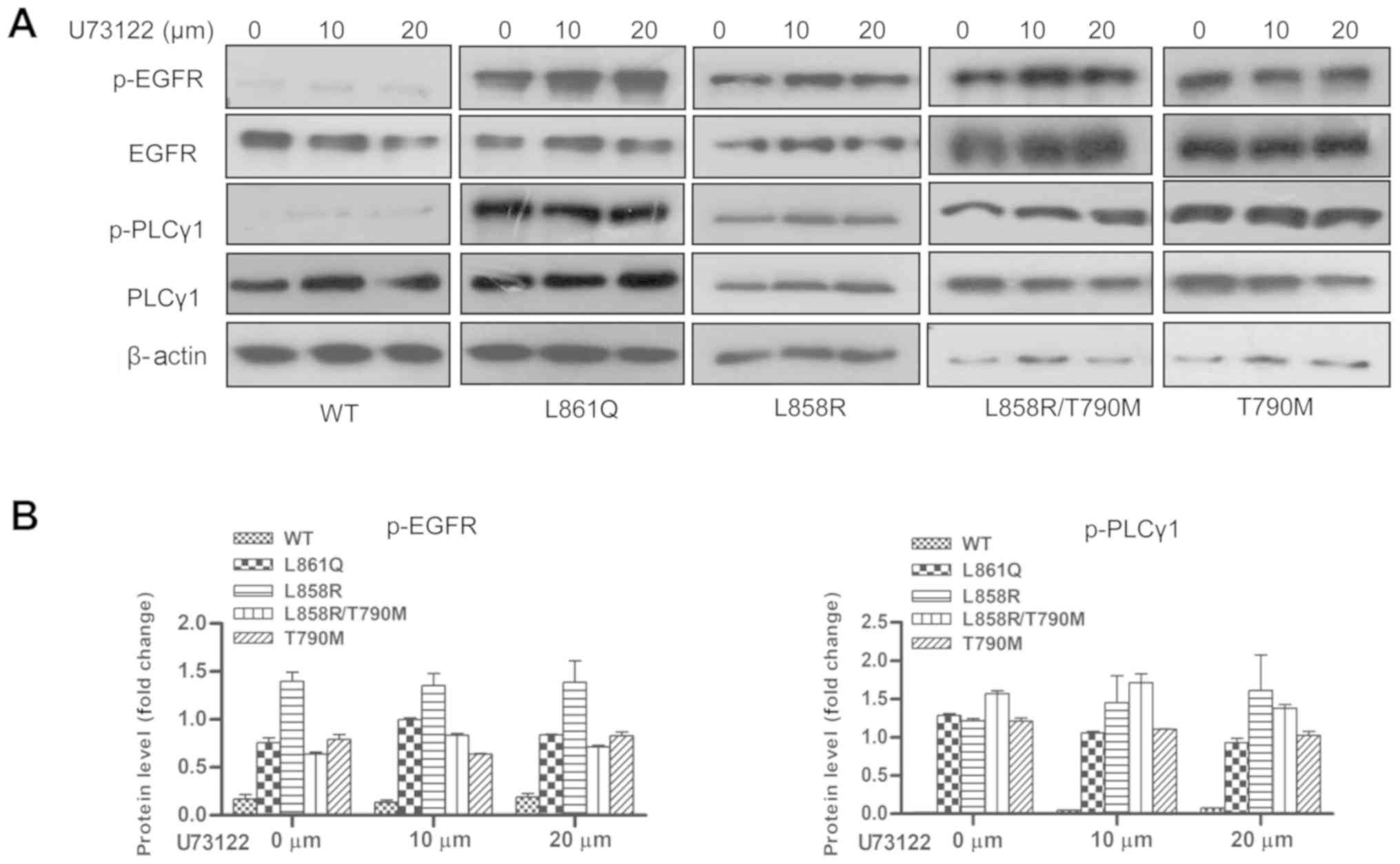
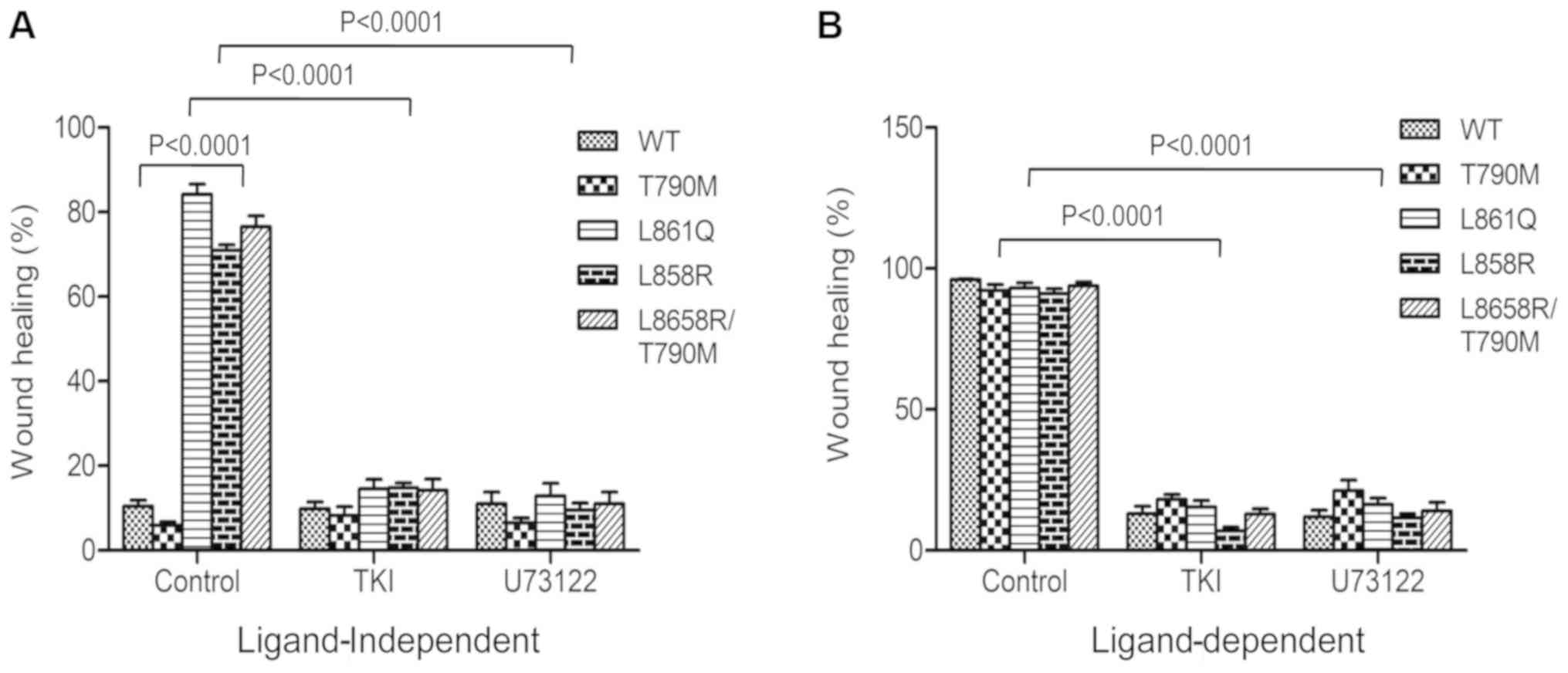
|
1
|
Chen Z, Fillmore CM, Hammerman PS, Kim CF
and Wong K-K: Non-small-cell lung cancers: A heterogeneous set of
diseases. Nat Rev Cancer. 14:535–546. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Paez JG, Jänne PA, Lee JC, Tracy S,
Greulich H, Gabriel S, Herman P, Kaye FJ, Lindeman N, Boggon TJ, et
al: EGFR mutations in lung cancer: correlation with clinical
response to gefitinib therapy. Science. 304:1497–1500. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lynch TJ, Bell DW, Sordella R,
Gurubhagavatula S, Okimoto RA, Brannigan BW, Harris PL, Haserlat
SM, Supko JG, Haluska FG, et al: Activating mutations in the
epidermal growth factor receptor underlying responsiveness of
non-small-cell lung cancer to gefitinib. N Engl J Med.
350:2129–2139. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Eberhard DA, Johnson BE, Amler LC, Goddard
AD, Heldens SL, Herbst RS, Ince WL, Jänne PA, Januario T, Johnson
DH, et al: Mutations in the epidermal growth factor receptor and in
KRAS are predictive and prognostic indicators in patients with
non-small-cell lung cancer treated with chemotherapy alone and in
combination with erlotinib. J Clin Oncol. 23:5900–5909. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lièvre A, Bachet J-B, Le Corre D, Boige V,
Landi B, Emile JF, Côté JF, Tomasic G, Penna C, Ducreux M, et al:
KRAS mutation status is predictive of response to cetuximab therapy
in colorectal cancer. Cancer Res. 66:3992–3995. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Raymond E, Faivre S and Armand JP:
Epidermal growth factor receptor tyrosine kinase as a target for
anticancer therapy. Drugs. 60(Suppl 1): 15–23; discussion 41–42.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nguyen K-SH, Kobayashi S and Costa DB:
Acquired resistance to epidermal growth factor receptor tyrosine
kinase inhibitors in non-small-cell lung cancers dependent on the
epidermal growth factor receptor pathway. Clin Lung Cancer.
10:281–289. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kobayashi S, Boggon TJ, Dayaram T, Jänne
PA, Kocher O, Meyerson M, Johnson BE, Eck MJ, Tenen DG and Halmos
B: EGFR mutation and resistance of non-small-cell lung cancer to
gefitinib. N Engl J Med. 352:786–792. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Oxnard GR, Arcila ME, Sima CS, Riely GJ,
Chmielecki J, Kris MG, Pao W, Ladanyi M and Miller VA: Acquired
resistance to EGFR tyrosine kinase inhibitors in EGFR-mutant lung
cancer: Distinct natural history of patients with tumors harboring
the T790M mutation. Clin Cancer Res. 17:1616–1622. 2011. View Article : Google Scholar
|
|
10
|
Liao B-C, Lin C-C and Yang JC-H: Second
and third-generation epidermal growth factor receptor tyrosine
kinase inhibitors in advanced nonsmall cell lung cancer. Curr Opin
Oncol. 27:94–101. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Jänne PA, Yang JC-H, Kim D-W, Planchard D,
Ohe Y, Ramalingam SS, Ahn MJ, Kim SW, Su WC, Horn L, et al: AZD9291
in EGFR inhibitor-resistant non-small-cell lung cancer. N Engl J
Med. 372:1689–1699. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cross DAE, Ashton SE, Ghiorghiu S,
Eberlein C, Nebhan CA, Spitzler PJ, Orme JP, Finlay MR, Ward RA,
Mellor MJ, et al: AZD9291, an irreversible EGFR TKI, overcomes
T790M-mediated resistance to EGFR inhibitors in lung cancer. Cancer
Discov. 4:1046–1061. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sordella R, Bell DW, Haber DA and
Settleman J: Gefitinib-sensitizing EGFR mutations in lung cancer
activate anti-apoptotic pathways. Science. 305:1163–1167. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim Y, Apetri M, Luo B, Settleman JE and
Anderson KS: Differential effects of tyrosine kinase inhibitors on
normal and oncogenic EGFR signaling and downstream effectors. Mol
Cancer Res. 13:765–774. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kamath A, Joseph AM, Gupta K, Behera D,
Jaiswal A, Dewan R and Rajala MS: Proteomic analysis of HEK293
cells expressing non small cell lung carcinoma associated epidermal
growth factor receptor variants reveals induction of heat shock
response. Exp Hematol Oncol. 4:162015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Rasband WS: US National Institutes of
Health, MD, USA. http://imagejnih.gov/ij/uri.
2011, Accessed August 28, 2008.
|
|
17
|
Piccolo E, Innominato PF, Mariggio MA,
Maffucci T, Iacobelli S and Falasca M: The mechanism involved in
the regulation of phospholipase Cgamma1 activity in cell migration.
Oncogene. 21:6520–6529. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wells A and Grandis JR: Phospholipase C-γ1
in tumor progression. Clin Exp Metastasis. 20:285–290. 2003.
View Article : Google Scholar
|
|
19
|
Li D, Ambrogio L, Shimamura T, Kubo S,
Takahashi M, Chirieac LR, Padera RF, Shapiro GI, Baum A,
Himmelsbach F, et al: BIBW2992, an irreversible EGFR/HER2 inhibitor
highly effective in preclinical lung cancer models. Oncogene.
27:4702–4711. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kobayashi Y and Mitsudomi T: Not all
epidermal growth factor receptor mutations in lung cancer are
created equal: Perspectives for individualized treatment strategy.
Cancer Sci. 107:1179–1186. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang F, Wang S, Yin L, Yang Y, Guan Y,
Wang W, Xu H and Tao N: Quantification of epidermal growth factor
receptor expression level and binding kinetics on cell surfaces by
surface plasmon resonance imaging. Anal Chem. 87:9960–9965. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yart A, Laffargue M, Mayeux P, Chretien S,
Peres C, Tonks N, Roche S, Payrastre B, Chap H and Raynal P: A
critical role for phosphoinositide 3-kinase upstream of Gab1 and
SHP2 in the activation of ras and mitogen-activated protein kinases
by epidermal growth factor. J Biol Chem. 276:8856–8864. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hashimoto A, Kurosaki M, Gotoh N, Shibuya
M and Kurosaki T: Shc regulates epidermal growth factor-induced
activation of the JNK signaling pathway. J Biol Chem.
274:20139–20143. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shtiegman K, Kochupurakkal BS, Zwang Y,
Pines G, Starr A, Vexler A, Citri A, Katz M, Lavi S, Ben-Basat Y,
et al: Defective ubiquitinylation of EGFR mutants of lung cancer
confers prolonged signaling. Oncogene. 26:6968–6978. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Nomoto K, Tomita N, Miyake M, Xhu DB,
LoGerfo PR and Weinstein IB: Expression of phospholipases γ1, β1,
and δ1 in primary human colon carcinomas and colon carcinoma cell
lines. Mol Carcinog. 12:146–152. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Arteaga CL, Johnson MD, Todderud G, Coffey
RJ, Carpenter G and Page DL: Elevated content of the tyrosine
kinase substrate phospholipase C-gamma 1 in primary human breast
carcinomas. Proc Natl Acad Sci USA. 88:10435–10439. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen P, Xie H, Sekar MC, Gupta K and Wells
A: Epidermal growth factor receptor-mediated cell motility:
Phospholipase C activity is required, but mitogen-activated protein
kinase activity is not sufficient for induced cell movement. J Cell
Biol. 127:847–857. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kundra V, Escobedo JA, Kazlauskas A, Kim
HK, Rhee SG, Williams LT and Zetter BR: Regulation of chemotaxis by
the platelet-derived growth factor receptor-β. Nature. 367:474–476.
1994. View
Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bornfeldt KE, Raines EW, Nakano T, Graves
LM, Krebs EG and Ross R: Insulin-like growth factor-I and
platelet-derived growth factor-BB induce directed migration of
human arterial smooth muscle cells via signaling pathways that are
distinct from those of proliferation. J Clin Invest. 93:1266–1274.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wells A, Kassis J, Solava J, Turner T and
Lauffenburger DA: Growth factor-induced cell motility in tumor
invasion. Acta Oncol. 41:124–130. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Polk DB: Epidermal growth factor
receptor-stimulated intestinal epithelial cell migration requires
phospholipase C activity. Gastroenterology. 114:493–502. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang Y, Wu J and Wang Z: Akt binds to and
phosphorylates phospholipase C-gamma1 in response to epidermal
growth factor. Mol Biol Cell. 17:2267–2277. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Appert-Collin A, Hubert P, Crémel G and
Bennasroune A: Role of ErbB receptors in cancer cell migration and
invasion. Front Pharmacol. 6:2832015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sos ML, Koker M, Weir BA, Heynck S,
Rabinovsky R, Zander T, Seeger JM, Weiss J, Fischer F, Frommolt P,
et al: PTEN loss contributes to erlotinib resistance in EGFR-mutant
lung cancer by activation of Akt and EGFR. Cancer Res.
69:3256–3261. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bidkhori G, Moeini A and Masoudi-Nejad A:
Modeling of tumor progression in NSCLC and intrinsic resistance to
TKI in loss of PTEN expression. PLoS One. 7:e480042012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kanda R, Kawahara A, Watari K, Murakami Y,
Sonoda K, Maeda M, Fujita H, Kage M, Uramoto H, Costa C, et al:
Erlotinib resistance in lung cancer cells mediated by integrin
β1/Src/Akt-driven bypass signaling. Cancer Res. 73:6243–6253. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Engelman JA, Zejnullahu K, Mitsudomi T,
Song Y, Hyland C, Park JO, Lindeman N, Gale CM, Zhao X, Christensen
J, et al: MET amplification leads to gefitinib resistance in lung
cancer by activating ERBB3 signaling. Science. 316:1039–1043. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Pao W, Wang TY, Riely GJ, Miller VA, Pan
Q, Ladanyi M, Zakowski MF, Heelan RT, Kris MG and Varmus HE: KRAS
mutations and primary resistance of lung adenocarcinomas to
gefitinib or erlotinib. PLoS Med. 2:e172005. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wahl M and Carpenter G: Selective
phospholipase C activation. BioEssays. 13:107–113. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Jang H-J, Suh P-G, Lee YJ, Shin KJ, Cocco
L and Chae YC: PLCγ1: Potential arbitrator of cancer progression.
Adv Biol Regul. 67:179–189. 2018. View Article : Google Scholar
|
|
41
|
Xie Z, Peng J, Pennypacker SD and Chen Y:
Critical role for the catalytic activity of phospholipase C-gamma1
in epidermal growth factor-induced cell migration. Biochem Biophys
Res Commun. 399:425–428. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Davies G, Martin TA, Ye L, Lewis-Russell
JM, Mason MD and Jiang WG: Phospholipase-C gamma-1 (PLCγ-1) is
critical in hepatocyte growth factor induced in vitro invasion and
migration without affecting the growth of prostate cancer cells.
Urol Oncol. 26:386–391. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Thomas SM, Coppelli FM, Wells A, Gooding
WE, Song J, Kassis J, Drenning SD and Grandis JR: Epidermal growth
factor receptor-stimulated activation of phospholipase Cgamma-1
promotes invasion of head and neck squamous cell carcinoma. Cancer
Res. 63:5629–5635. 2003.PubMed/NCBI
|
|
44
|
Dai L, Zhuang L and Zhang B, Wang F, Chen
X, Xia C and Zhang B: DAG/PKCδ and IP3/Ca2+/CaMK IIβ
operate in parallel to each other in PLCγ1-driven cell
proliferation and migration of human gastric adenocarcinoma cells,
through Akt/mTOR/S6 pathway. Int J Mol Sci. 16:28510–28522. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Tang W, Zhou Y, Sun D, Dong L, Xia J and
Yang B: Oncogenic role of phospholipase C-γ1 in progression of
hepatocellular carcinoma. Hepatol Res. 49:559–569. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhang P, Zhao Y, Zhu X, Sedwick D, Zhang X
and Wang Z: Cross-talk between phospho-STAT3 and PLCγ1 plays a
critical role in colorectal tumorigenesis. Mol Cancer Res.
9:1418–1428. 2011. View Article : Google Scholar : PubMed/NCBI
|