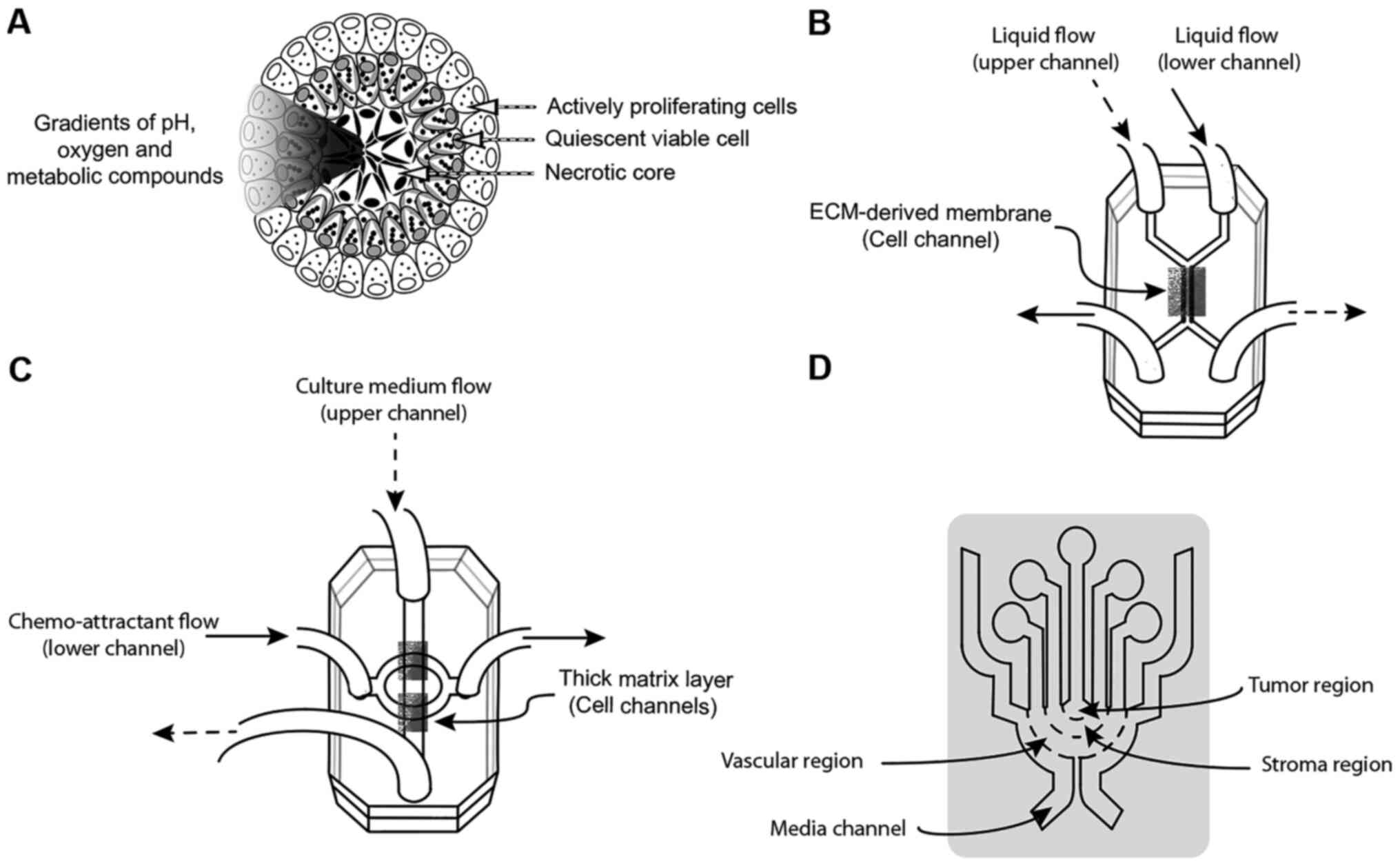
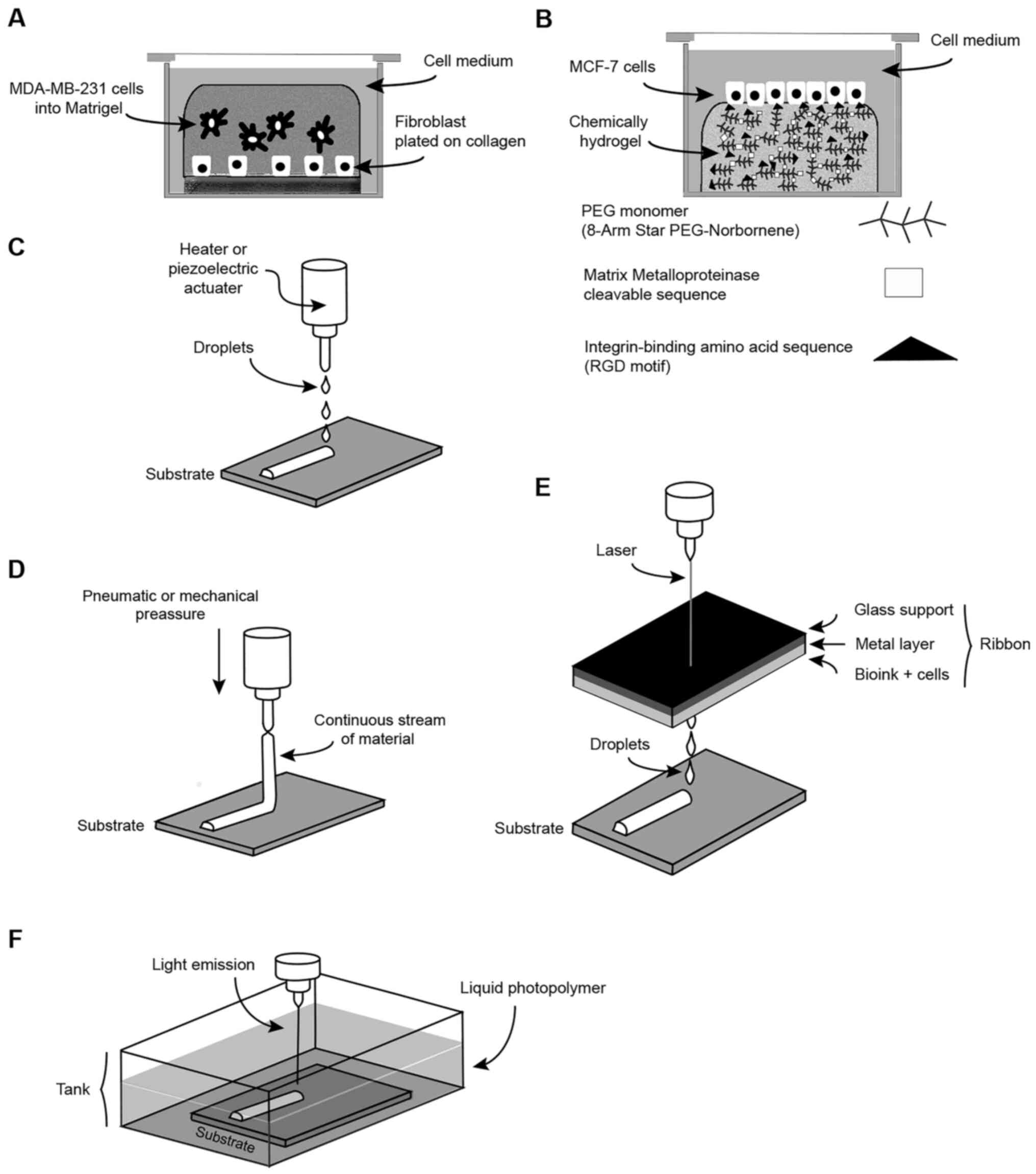
|
1
|
Ahmad A: Breast cancer statistics: Recent
trends. Adv Exp Med Biol. 1152:1–7. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rivera-Franco MM and Leon-Rodriguez E:
Delays in breast cancer detection and treatment in developing
countries. Breast Cancer (Auckl). 12:11782234177526772018.
|
|
3
|
Global Burden of Disease Cancer
Collaboration; Fitzmaurice C, Dicker D, Pain A, Hamavid H,
Moradi-Lakeh M, MacIntyre MF, Allen C, Hansen G, Woodbrook R, et
al: The global burden of cancer 2013. JAMA Oncol. 1:505–527. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hanahan D and Coussens LM: Accessories to
the crime: Functions of cells recruited to the tumor
microenvironment. Cancer Cell. 21:309–322. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mittal S, Brown NJ and Holen I: The breast
tumor microenvironment: Role in cancer development, progression and
response to therapy. Expert Rev Mol Diagn. 18:227–243. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Conklin MW and Keely PJ: Why the stroma
matters in breast cancer: Insights into breast cancer patient
outcomes through the examination of stromal biomarkers. Cell Adh
Migr. 6:249–260. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Insua-Rodríguez J and Oskarsson T: The
extracellular matrix in breast cancer. Adv Drug Deliv Rev.
97:41–55. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Friedl P and Alexander S: Cancer invasion
and the microenvironment: Plasticity and reciprocity. Cell.
147:992–1009. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kaushik S, Pickup MW and Weaver VM: From
transformation to metastasis: Deconstructing the extracellular
matrix in breast cancer. Cancer Metastasis Rev. 35:655–667. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Natal RA, Paiva GR, Pelegati VB, Marenco
L, Alvarenga CA, Vargas RF, Derchain SF, Sarian LO, Franchet C,
Cesar CL, et al: Exploring collagen parameters in pure special
types of invasive breast cancer. Sci Rep. 9:77152019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vuong D, Simpson PT, Green B, Cummings MC
and Lakhani SR: Molecular classification of breast cancer. Virchows
Arch. 465:1–14. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Comşa Ş, Cimpean AM and Raica M: The story
of MCF-7 breast cancer cell line: 40 years of experience in
research. Anticancer Res. 35:3147–3154. 2015.PubMed/NCBI
|
|
13
|
Ravi M, Sneka MK and Joshipura A: The
culture conditions and outputs from breast cancer cell line in
vitro experiments. Exp Cell Res. 383:1115482019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim JB, O'Hare MJ and Stein R: Models of
breast cancer: Is merging human and animal models the future?
Breast Cancer Res. 6:22–30. 2004. View
Article : Google Scholar :
|
|
15
|
Bersini S, Jeon JS, Dubini G, Arrigoni C,
Chung S, Charest JL, Moretti M and Kamm RD: A microfluidic 3D in
vitro model for specificity of breast cancer metastasis to bone.
Biomaterials. 35:2454–2461. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
dit Faute MA, Laurent L, Ploton D, Poupon
MF, Jardillier JC and Bobichon H: Distinctive alterations of
invasiveness, drug resistance and cell-cell organization in
3D-cultures of MCF-7, a human breast cancer cell line, and its
multidrug resistant variant. Clin Exp Metastasis. 19:161–168. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lovitt CJ, Shelper TB and Avery VM:
Evaluation of chemotherapeutics in a three-dimensional breast
cancer model. J Cancer Res Clin Oncol. 141:951–959. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mehta G, Hsiao AY, Ingram M, Luker GD and
Takayama S: Opportunities and challenges for use of tumor spheroids
as models to test drug delivery and efficacy. J Control Release.
164:192–204. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Froehlich K, Haeger JD, Heger J,
Pastuschek J, Photini SM, Yan Y, Lupp A, Pfarrer C, Mrowka R,
Schleußner E, et al: Generation of multicellular breast cancer
tumor spheroids: Comparison of different protocols. J Mammary Gland
Biol Neoplasia. 21:89–98. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bahcecioglu G, Basara G, Ellis BW, Ren X
and Zorlutuna P: Breast cancer models: Engineering the tumor
microenvironment. Acta Biomater. 106:1–21. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nagelkerke A, Bussink J, Sweep FC and Span
PN: Generation of multicellular tumor spheroids of breast cancer
cells: How to go three-dimensional. Anal Biochem. 437:17–19. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Singh M, Mukundan S, Jaramillo M,
Oesterreich S and Sant S: Three-dimensional breast cancer models
mimic hallmarks of size-induced tumor progression. Cancer Res.
76:3732–3743. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang W, Li C, Baguley BC, Zhou F, Zhou W,
Shaw JP, Wang Z, Wu Z and Liu J: Optimization of the formation of
embedded multicellular spheroids of MCF-7 cells: How to reliably
produce a biomimetic 3D model. Anal Biochem. 515:47–54. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Asghar W, El Assal R, Shafiee H, Pitteri
S, Paulmurugan R and Demirci U: Engineering cancer
microenvironments for in vitro 3-D tumor models. Mater Today
(Kidlington). 18:539–553. 2015. View Article : Google Scholar
|
|
25
|
Rodríguez CE, Berardi DE, Abrigo M, Todaro
LB, Bal de Kier Joffé ED and Fiszman GL: Breast cancer stem cells
are involved in trastuzumab resistance through the HER2 modulation
in 3D culture. J Cell Biochem. 119:1381–1391. 2018. View Article : Google Scholar
|
|
26
|
Zhao L, Xiu J, Liu Y, Zhang T, Pan W,
Zheng X and Zhang X: A 3D printed hanging drop dripper for tumor
spheroids analysis without recovery. Sci Rep. 9:197172019.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Metzger W, Sossong D, Bächle A, Pütz N,
Wennemuth G, Pohlemann T and Oberringer M: The liquid overlay
technique is the key to formation of co-culture spheroids
consisting of primary osteoblasts, fibroblasts and endothelial
cells. Cytotherapy. 13:1000–1012. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Huang S, Yuan N, Wang G, Wu F, Feng L, Luo
M, Li M, Luo A, Zhao X and Zhang L: Cellular communication promotes
mammosphere growth and collective invasion through microtubule-like
structures and angiogenesis. Oncol Rep. 40:3297–3312.
2018.PubMed/NCBI
|
|
29
|
Berger AJ, Renner CM, Hale I, Yang X,
Ponik SM, Weisman PS, Masters KS and Kreeger PK: Scaffold stiffness
influences breast cancer cell invasion via EGFR-linked Mena
upregulation and matrix remodeling. Matrix Biol. 85:80–93. 2020.
View Article : Google Scholar
|
|
30
|
Guzman A, Sánchez Alemany V, Nguyen Y,
Zhang CR and Kaufman LJ: A novel 3D in vitro metastasis model
elucidates differential invasive strategies during and after
breaching basement membrane. Biomaterials. 115:19–29. 2017.
View Article : Google Scholar
|
|
31
|
Dubois C, Dufour R, Daumar P, Aubel C,
Szczepaniak C, Blavignac C, Mounetou E, Penault-Llorca F and Bamdad
M: Development and cytotoxic response of two proliferative
MDA-MB-231 and non-proliferative SUM1315 three-dimensional cell
culture models of triple-negative basal-like breast cancer cell
lines. Oncotarget. 8:953162017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Smalley KS, Lioni M, Noma K, Haass NK and
Herlyn M: In vitro three-dimensional tumor microenvironment models
for anti-cancer drug discovery. Expert Opin Drug Discov. 3:1–10.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Costa EC, Gaspar VM, Coutinho P and
Correia IJ: Optimization of liquid overlay technique to formulate
heterogenic 3D co-cultures models. Biotechnol Bioeng.
111:1672–1685. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Costa EC, de Melo-Diogo D, Moreira AF,
Carvalho MP and Correia IJ: Spheroids formation on non-adhesive
surfaces by liquid overlay technique: Considerations and practical
approaches. Biotechnol J. 13:2018. View Article : Google Scholar
|
|
35
|
Kosaka T, Tsuboi S, Fukaya K, Pu H, Ohno
T, Tsuji T, Miyazaki M and Namba M: Spheroid cultures of human
hepatoblastoma cells (HuH-6 line) and their application for
cytotoxicity assay of alcohols. Acta Medica Okayama. 50:61–66.
1996.PubMed/NCBI
|
|
36
|
Kelm JM, Timmins NE, Brown CJ, Fussenegger
M and Nielsen LK: Method for generation of homogeneous
multicel-lular tumor spheroids applicable to a wide variety of cell
types. Biotechnol Bioeng. 83:173–180. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Robertson FM, Ogasawara MA, Ye Z, Chu K,
Pickei R, Debeb BG, Woodward WA, Hittelman WN, Cristofanilli M and
Barsky SH: Imaging and analysis of 3D tumor spheroids enriched for
a cancer stem cell phenotype. J Biomol Screen. 15:820–829. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lorenzo C, Frongia C, Jorand R, Fehrenbach
J, Weiss P, Maandhui A, Gay G, Ducommun B and Lobjois V: Live cell
division dynamics monitoring in 3D large spheroid tumor models
using light sheet microscopy. Cell Div. 6:222011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Leonard F and Godin B: 3D in vitro model
for breast cancer research using magnetic levitation and
bioprinting method. Methods Mol Biol. 1406:239–251. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Jaganathan H, Gage J, Leonard F,
Srinivasan S, Souza GR, Dave B and Godin B: Three-dimensional in
vitro co-culture model of breast tumor using magnetic levitation.
Sci Rep. 4:64682014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Eckhardt BL, Gagliardi M, Iles L, Evans K,
Ivan C, Liu X, Liu CG, Souza G, Rao A, Meric-Bernstam F, et al:
Clinically relevant inflammatory breast cancer patient-derived
xenograft-derived ex vivo model for evaluation of tumor-specific
therapies. PLoS One. 13:e01959322018. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lin RZ and Chang HY: Recent advances in
three-dimensional multicellular spheroid culture for biomedical
research. Biotechnol J. 3:1172–1184. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Swaminathan S, Hamid Q, Sun W and Clyne
AM: Bioprinting of 3D breast epithelial spheroids for human cancer
models. Biofabrication. 11:0250032019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ong CS, Yesantharao P, Huang CY, Mattson
G, Boktor J, Fukunishi T, Zhang H and Hibino N: 3D bioprinting
using stem cells. Pediatr Res. 83:223–231. 2018. View Article : Google Scholar
|
|
45
|
Kingsley DM, Roberge CL, Rudkouskaya A,
Faulkner DE, Barroso M, Intes X and Corr DT: Laser-based 3D
bioprinting for spatial and size control of tumor spheroids and
embryoid bodies. Acta Biomater. 95:357–370. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ham SL, Joshi R, Luker GD and Tavana H:
Engineered breast cancer cell spheroids reproduce biologic
properties of solid tumors. Adv Healthc Mater. 5:2788–2798. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Ham SL, Thakuri PS, Plaster M, Li J, Luker
KE, Luker GD and Tavana H: Three-dimensional tumor model mimics
stromal-breast cancer cells signaling. Oncotarget. 9:249–267. 2017.
View Article : Google Scholar
|
|
48
|
Atefi E, Lemmo S, Fyffe D, Luker GD and
Tavana H: High throughput, polymeric aqueous two-phase printing of
tumor spheroids. Adv Funct Mater. 24:6509–6515. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Lemmo S, Atefi E, Luker GD and Tavana H:
Optimization of aqueous biphasic tumor spheroid microtechnology for
anti-cancer drug testing in 3D culture. Cell Mol Bioeng. 7:344–354.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Shahi Thakuri P, Ham SL, Luker GD and
Tavana H: Multiparametric analysis of oncology drug screening with
aqueous two-phase tumor spheroids. Mol Pharm. 13:3724–3735. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Sackmann EK, Fulton AL and Beebe DJ: The
present and future role of microfluidics in biomedical research.
Nature. 507:181–189. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Sen AK, Raj A, Banerjee U and Iqbal SR:
Soft lithography, molding, and micromachining techniques for
polymer micro devices. Microfluidic Electrophoresis. Methods in
Molecular Biology. Dutta D: 1906. Humana Press Springer; New York,
NY: pp. 13–54. 2019, View Article : Google Scholar
|
|
53
|
Halldorsson S, Lucumi E, Gómez-Sjöberg R
and Fleming RMT: Advantages and challenges of microfluidic cell
culture in polydimethylsiloxane devices. Biosens Bioelectron.
63:218–231. 2015. View Article : Google Scholar
|
|
54
|
Sontheimer-Phelps A, Hassell BA and Ingber
DE: Modelling cancer in microfluidic human organs-on-chips. Nat Rev
Cancer. 19:65–81. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Yum K, Hong SG, Healy KE and Lee LP:
Physiologically relevant organs on chips. Biotechnol J. 9:16–27.
2014. View Article : Google Scholar
|
|
56
|
Wang JD, Douville NJ, Takayama S and
ElSayed M: Quantitative analysis of molecular absorption into PDMS
microfluidic channels. Ann Biomed Eng. 40:1862–1873. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Choi Y, Hyun E, Seo J, Blundell C, Kim HC,
Lee E, Lee SH, Moon A, Moon WK and Huh D: A microengineered
patho-physiological model of early-stage breast cancer. Lab Chip.
15:3350–3357. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Montanez-Sauri SI, Sung KE, Berthier E and
Beebe DJ: Enabling screening in 3D microenvironments: Probing
matrix and stromal effects on the morphology and proliferation of
T47D breast carcinoma cells. Integr Biol (Camb). 5:631–640. 2013.
View Article : Google Scholar
|
|
59
|
Lang JD, Berry SM, Powers GL, Beebe DJ and
Alarid ET: Hormonally responsive breast cancer cells in a
microfluidic co-culture model as a sensor of microenvironmental
activity. Integr Biol (Camb). 5:807–816. 2013. View Article : Google Scholar
|
|
60
|
Regier MC, Maccoux LJ, Weinberger EM,
Regehr KJ, Berry SM, Beebe DJ and Alarid ET: Transitions from
mono-to co-to tri-culture uniquely affect gene expression in breast
cancer, stromal, and immune compartments. Biomed Microdevices.
18:702016. View Article : Google Scholar
|
|
61
|
Dongre A and Weinberg RA: New insights
into the mechanisms of epithelial-mesenchymal transition and
implications for cancer. Nat Rev Mol Cell Biol. 20:69–84. 2019.
View Article : Google Scholar
|
|
62
|
Eslami Amirabadi H, SahebAli S, Frimat JP,
Luttge R and den Toonder JMJ: A novel method to understand tumor
cell invasion: Integrating extracellular matrix mimicking layers in
microfluidic chips by 'selective curing'. Biomed Microdevices.
19:922017. View Article : Google Scholar
|
|
63
|
Eslami Amirabadi H, Tuerlings M,
Hollestelle A, SahebAli S, Luttge R, van Donkelaar CC, Martens JWM
and den Toonder JMJ: Characterizing the invasion of different
breast cancer cell lines with distinct E-cadherin status in 3D
using a microfluidic system. Biomed Microdevices. 21:1012019.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Toh YC, Raja A, Yu H and van Noort D: A 3D
microfluidic model to recapitulate cancer cell migration and
invasion. Bioengineering (Basel). 5:292018. View Article : Google Scholar
|
|
65
|
Blaha L, Zhang C, Cabodi M and Wong JY: A
microfluidic platform for modeling metastatic cancer cell matrix
invasion. Biofabrication. 9:0450012017. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Devadas D, Moore TA, Walji N and Young
EWK: A microfluidic mammary gland coculture model using parallel 3D
lumens for studying epithelial-endothelial migration in breast
cancer. Biomicrofluidics. 13:0641222019. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Li R, Hebert JD, Lee TA, Xing H,
Boussommier-Calleja A, Hynes RO, Lauffenburger DA and Kamm RD:
Macrophage-secreted TNFα and TGFβ1 influence migration speed and
persistence of cancer cells in 3D tissue culture via independent
pathways. Cancer Res. 77:279–290. 2017. View Article : Google Scholar
|
|
68
|
Nagaraju S, Truong D, Mouneimne G and
Nikkhah M: Microfluidic tumor-vascular model to study breast cancer
cell invasion and intravasation. Adv Healthc Mater. 7:17012572018.
View Article : Google Scholar
|
|
69
|
Tang Y, Soroush F, Sheffield JB, Wang B,
Prabhakarpandian B and Kiani MF: A biomimetic microfluidic tumor
microenvironment platform mimicking the EPR effect for rapid
screening of drug delivery systems. Sci Rep. 7:93592017. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Lanz HL, Saleh A, Kramer B, Cairns J, Ng
CP, Yu J, Trietsch SJ, Hankemeier T, Joore J, Vulto P, et al:
Therapy response testing of breast cancer in a 3D high-throughput
perfused microfluidic platform. BMC Cancer. 17:7092017. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Bahlmann LC, Smith LJ and Shoichet MS:
Designer biomaterials to model cancer cell invasion in vitro:
Predictive tools or just pretty pictures. Adv Funct Mater.
30:19090322020. View Article : Google Scholar
|
|
72
|
Chen L, Xiao Z, Meng Y, Zhao Y, Han J, Su
G, Chen B and Dai J: The enhancement of cancer stem cell properties
of MCF-7 cells in 3D collagen scaffolds for modeling of cancer and
anti-cancer drugs. Biomaterials. 33:1437–1444. 2012. View Article : Google Scholar
|
|
73
|
Antoine EE, Vlachos PP and Rylander MN:
Review of collagen I hydrogels for bioengineered tissue
microenvironments: Characterization of mechanics, structure, and
transport. Tissue Eng Part B Rev. 20:683–696. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Liu J, Tan Y, Zhang H, Zhang Y, Xu P, Chen
J, Poh YC, Tang K, Wang N and Huang B: Soft fibrin gels promote
selection and growth of tumorigenic cells. Nat Mater. 11:734–741.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Kleinman HK and Martin GR: Matrigel:
Basement membrane matrix with biological activity. Semin Cancer
Biol. 15:378–386. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Krause S, Maffini MV, Soto AM and
Sonnenschein C: The microenvironment determines the breast cancer
cells' phenotype: Organization of MCF7 cells in 3D cultures. BMC
Cancer. 10:2632010. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Whitman NA, Lin ZW, Kenney RM, Albertini L
and Lockett MR: Hypoxia differentially regulates estrogen receptor
alpha in 2D and 3D culture formats. Arch Biochem Biophys. 671:8–17.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Palomeras S, Rabionet M, Ferrer I, Sarrats
A, Garcia-Romeu ML, Puig T and Ciurana J: Breast cancer stem cell
culture and enrichment using poly(ε-Caprolactone) scaffolds.
Molecules. 21:5372016. View Article : Google Scholar
|
|
79
|
Wessels DJ, Pradhan N, Park YN, Klepitsch
MA, Lusche DF, Daniels KJ, Conway KD, Voss ER, Hegde SV, Conway TP
and Soll DR: Reciprocal signaling and direct physical interactions
between fibroblasts and breast cancer cells in a 3D environment.
PLoS One. 14:e02188542019. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
James-Bhasin M, Siegel PM and Nazhat SN: A
three-dimensional dense collagen hydrogel to model cancer
cell/osteoblast interactions. J Funct Biomater. 9:722018.
View Article : Google Scholar
|
|
81
|
Cox RF, Jenkinson A, Pohl K, O'Brien FJ
and Morgan MP: Osteomimicry of mammary adenocarcinoma cells in
vitro; increased expression of bone matrix proteins and
proliferation within a 3D collagen environment. PLoS One.
7:e416792012. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Berger AJ, Linsmeier KM, Kreeger PK and
Masters KS: Decoupling the effects of stiffness and fiber density
on cellular behaviors via an interpenetrating network of
gelatin-methacrylate and collagen. Biomaterials. 141:125–135. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Conklin MW, Eickhoff JC, Riching KM,
Pehlke CA, Eliceiri KW, Provenzano PP, Friedl A and Keely PJ:
Aligned collagen is a prognostic signature for survival in human
breast carcinoma. Am J Pathol. 178:1221–1232. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Bredfeldt JS, Liu Y, Conklin MW, Keely PJ,
Mackie TR and Eliceiri KW: Automated quantification of aligned
collagen for human breast carcinoma prognosis. J Pathol Inform.
5:282014. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Wang Y, Mirza S, Wu S, Zeng J, Shi W, Band
H, Band V and Duan B: 3D hydrogel breast cancer models for studying
the effects of hypoxia on epithelial to mesenchymal transition.
Oncotarget. 9:321912018. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Peppas NA, Hilt JZ, Khademhosseini A and
Langer R: Hydrogels in biology and medicine: From molecular
principles to bionanotechnology. Adv Mater. 18:1345–1360. 2006.
View Article : Google Scholar
|
|
87
|
Livingston MK, Morgan MM, Daly WT, Murphy
WL, Johnson BP, Beebe DJ and Virumbrales-Muñoz M: Evaluation of
PEG-based hydrogel influence on estrogen receptor driven responses
in MCF7 breast cancer cells. ACS Biomater Sci Eng. 5:6089–6098.
2019. View Article : Google Scholar
|
|
88
|
Bellis SL: Advantages of RGD peptides for
directing cell association with biomaterials. Biomaterials.
32:4205–4210. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Wang F, Li Y, Shen Y, Wang A, Wang S and
Xie T: The functions and applications of RGD in tumor therapy and
tissue engineering. Int J Mol Sci. 14:13447–13462. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Sawicki LA, Ovadia EM, Pradhan L, Cowart
JE, Ross KE, Wu CH and Kloxin AM: Tunable synthetic extracellular
matrices to investigate breast cancer response to biophysical and
biochemical cues. APL Bioeng. 3:0161012019. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Schmid R, Schmidt SK, Hazur J, Detsch R,
Maurer E, Boccaccini AR, Hauptstein J, Teßmar J, Blunk T, Schrüfer
S, et al: Comparison of hydrogels for the development of
well-defined 3D cancer models of breast cancer and melanoma.
Cancers (Basel). 12:23202020. View Article : Google Scholar :
|
|
92
|
Feng S, Duan X, Lo PK, Liu S, Liu X, Chen
H and Wang Q: Expansion of breast cancer stem cells with fibrous
scaffolds. Integr Biol (Camb). 5:768–777. 2013. View Article : Google Scholar
|
|
93
|
Zhang YS, Duchamp M, Oklu R, Ellisen LW,
Langer R and Khademhosseini A: Bioprinting the cancer
microenvironment. ACS Biomater Sci Eng. 2:1710–1721. 2016.
View Article : Google Scholar
|
|
94
|
Balachander GM, Balaji SA, Rangarajan A
and Chatterjee K: Enhanced metastatic potential in a 3D tissue
scaffold toward a comprehensive in vitro model for breast cancer
metastasis. ACS Appl Mater Interfaces. 7:27810–27822. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Guiro K, Patel SA, Greco SJ, Rameshwar P
and Arinzeh TL: Investigating breast cancer cell behavior using
tissue engineering scaffolds. PLoS One. 10:e01187242015. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Rijal G, Bathula C and Li W: Application
of synthetic polymeric scaffolds in breast cancer 3D tissue
cultures and animal tumor models. Int J Biomater. 2017:80748902017.
View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Kačarević ŽP, Rider PM, Alkildani S,
Retnasingh S, Smeets R, Jung O, Ivanišević Z and Barbeck M: An
introduction to 3D bioprinting: Possibilities, challenges and
future aspects. Materials (Basel). 11:21992018. View Article : Google Scholar
|
|
98
|
Cha rbe N, McCa r ron PA and Tambuwala MM:
Three-dimensional bio-printing: A new frontier in oncology
research. World J Clin Oncol. 8:21–36. 2017. View Article : Google Scholar
|
|
99
|
Derby B: Bioprinting: Inkjet printing
proteins and hybrid cell-containing materials and structures. J
Mater Chem. 18:5717–5721. 2008. View Article : Google Scholar
|
|
100
|
Negro A, Cherbuin T and Lutolf MP: 3D
inkjet printing of complex, cell-laden hydrogel structures. Sci
Rep. 8:170992018. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Panwar A and Tan LP: Current status of
bioinks for micro-extrusion-based 3D bioprinting. Molecules.
21:6852016. View Article : Google Scholar
|
|
102
|
Koch L, Gruene M, Unger C and Chichkov B:
Laser assisted cell printing. Curr Pharm Biotechnol. 14:91–97.
2013.PubMed/NCBI
|
|
103
|
Mondschein RJ, Kanitkar A, Williams CB,
Verbridge SS and Long TE: Polymer structure-property requirements
for stereo-lithographic 3D printing of soft tissue engineering
scaffolds. Biomaterials. 140:170–188. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Cui X, Boland T, D'Lima DD and Lotz MK:
Thermal inkjet printing in tissue engineering and regenerative
medicine. Recent Pat Drug Deliv Formul. 6:149–155. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Cheng E, Yu H, Ahmadi A and Cheung KC:
Investigation of the hydrodynamic response of cells in drop on
demand piezoelectric inkjet nozzles. Biofabrication. 8:0150082016.
View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Yi HG, Lee H and Cho DW: 3D printing of
organs-on-chips. Bioengineering (Basel). 4:102017. View Article : Google Scholar
|
|
107
|
Bishop ES, Mostafa S, Pakvasa M, Luu HH,
Lee MJ, Wolf JM, Ameer GA, He TC and Reid RR: 3-D bioprinting
technologies in tissue engineering and regenerative medicine:
Current and future trends. Genes Dis. 4:185–195. 2017. View Article : Google Scholar
|
|
108
|
Chang R, Nam J and Sun W: Effects of
dispensing pressure and nozzle diameter on cell survival from solid
freeform fabrication-based direct cell writing. Tissue Eng Part A.
14:41–48. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Murphy SV and Atala A: 3D bioprinting of
tissues and organs. Nat Biotechnol. 32:773–785. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Park JH, Jang J, Lee JS and Cho DW:
Three-dimensional printing of tissue/organ analogues containing
living cells. Ann Biomed Eng. 45:180–194. 2017. View Article : Google Scholar
|
|
111
|
Sears NA and Seshadri DR: A review of
three-dimensional printing in tissue engineering. Tissue Eng Part B
Rev. 22:298–310. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Raman R, Bhaduri B, Mir M, Shkumatov A,
Lee MK, Popescu G, Kong H and Bashir R: High-resolution projection
microstereolithography for patterning of neovasculature. Adv
Healthc Mater. 5:610–619. 2016. View Article : Google Scholar
|
|
113
|
Lin H, Zhang D, Alexander PG, Yang G, Tan
J, Cheng AW and Tuan RS: Application of visible light-based
projection stereo-lithography for live cell-scaffold fabrication
with designed architecture. Biomaterials. 34:331–339. 2013.
View Article : Google Scholar
|
|
114
|
Axpe E and Oyen ML: Applications of
alginate-based bioinks in 3D bioprinting. Int J Mol Sci.
17:19762016. View Article : Google Scholar
|
|
115
|
Nerger BA, Brun PT and Nelson CM:
Microextrusion printing cell-laden networks of type I collagen with
patterned fiber alignment and geometry. Soft Matter. 15:5728–5738.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Włodarczyk-Biegun MK and del Campo A: 3D
bioprinting of structural proteins. Biomaterials. 134:180–201.
2017. View Article : Google Scholar
|
|
117
|
Albritton JL and Miller JS: 3D
bioprinting: Improving in vitro models of metastasis with
heterogeneous tumor microenvironments. Dis Model Mech. 10:3–14.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Mancha Sánchez E, Gómez-Blanco JC, López
Nieto E, Casado JG, Macías-García A, Díaz Díez MA, Carrasco-Amador
JP, Torrejón Martín D, Sánchez-Margallo FM and Pagador JB:
Hydrogels for bioprinting: A systematic review of hydrogels
synthesis, bioprinting parameters, and bioprinted structures
behavior. Front Bioeng Biotechnol. 8:7762020. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Moore CA, Shah NN, Smith CP and Rameshwar
P: 3D bioprinting and stem cells. Methods Mol Biol. 1842:93–103.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Mollica PA, Booth-Creech EN, Reid JA,
Zamponi M, Sullivan SM, Palmer XL, Sachs PC and Bruno RD: 3D
bioprinted mammary organoids and tumoroids in human mammary derived
ECM hydrogels. Acta Biomater. 95:201–213. 2019. View Article : Google Scholar : PubMed/NCBI
|