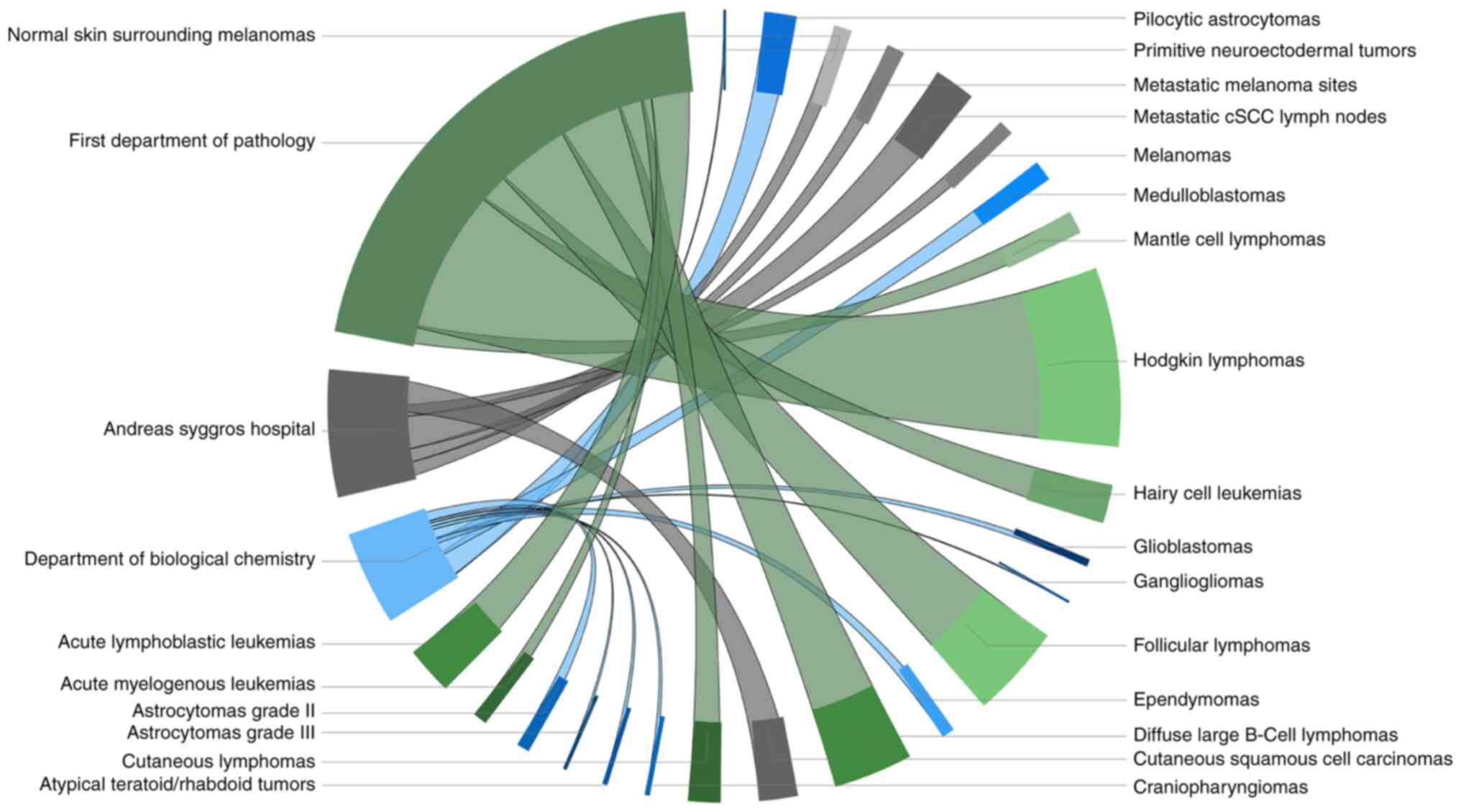
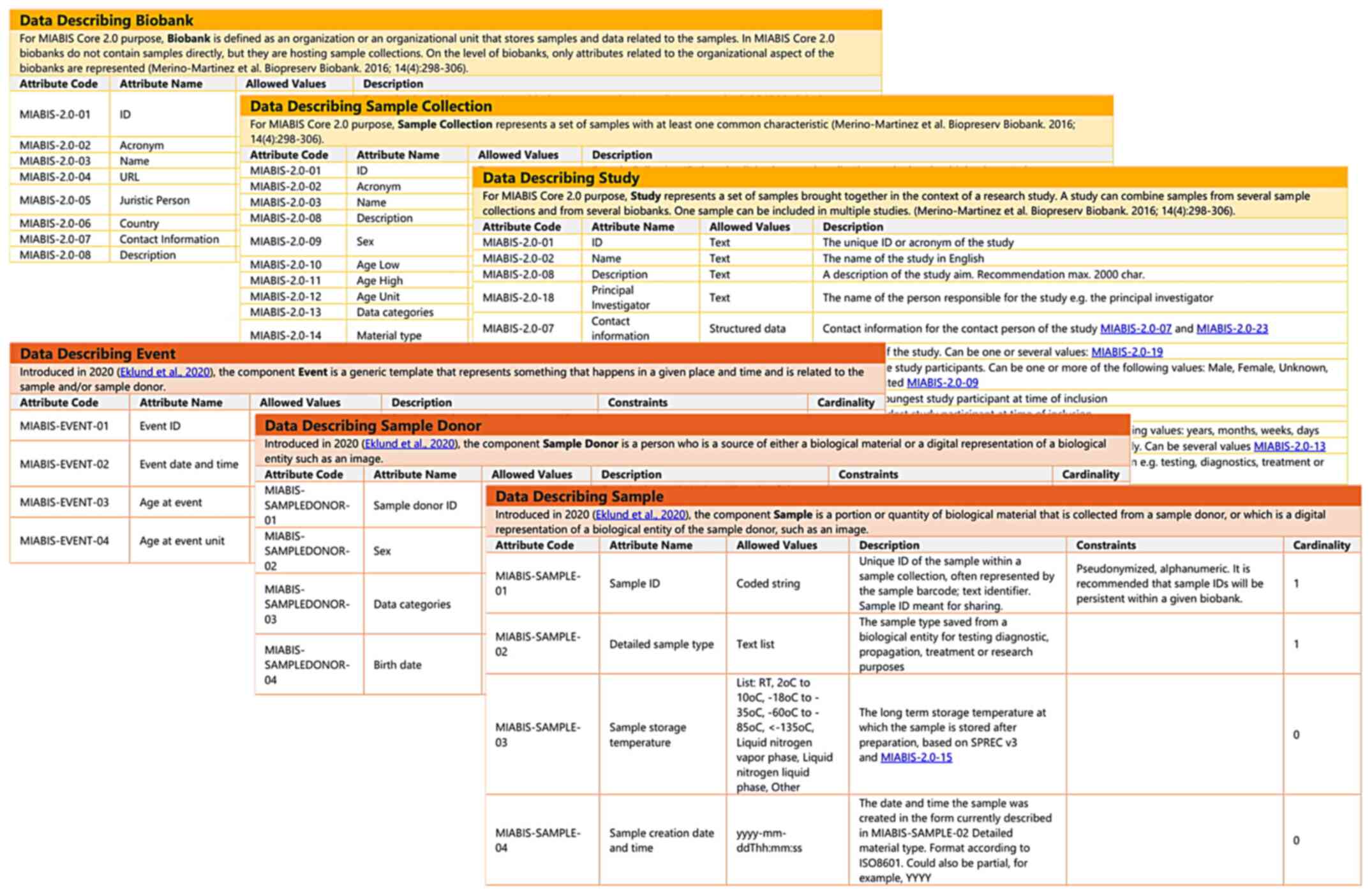
|
1
|
Kuhn TS: The structure of scientific
revolutions. 2nd edition. University of Chicago Press; Chicago, IL:
1970
|
|
2
|
Toward precision medicine: Building a
knowledge network for biomedical research and a new taxonomy of
disease. National Academies Press (US); Washington, DC: 2011
|
|
3
|
Hellenic Network of Precision Medicine on
Cancer: What does precision medicine mean? https://oncopmnet.gr/?page_id=2784&lang=en.
Accessed July 15, 2021.
|
|
4
|
Kohler S: Precision medicine-moving away
from one-size-fits-all. Quest Science for South Africa. 14. Academy
of Science of South Africa; Pretoria: pp. 12–15. 2018
|
|
5
|
Garraway LA and Lander ES: Lessons from
the cancer genome. Cell. 153:17–37. 2013. View Article : Google Scholar
|
|
6
|
Re A, Nardella C, Quattrone A and Lunardi
A: Editorial: Precision medicine in oncology. Front Oncol.
8:4792018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Garraway LA, Verweij J and Ballman KV:
Precision oncology: An overview. J Clin Oncol. 31:1803–1805. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Garraway LA: Genomics-driven oncology:
Framework for an emerging paradigm. J Clin Oncol. 31:1806–1814.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hyman DM, Taylor BS and Baselga J:
Implementing genome-driven oncology. Cell. 168:584–599. 2017.
View Article : Google Scholar :
|
|
10
|
Sakurai M and Sandberg AA: Chromosomes and
causation of human cancer and leukemia. XI. Correlation of
karyotypes with clinical features of acute myeloblastic leukemia.
Cancer. 37:285–299. 1976. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Flores M, Glusman G, Brogaard K, Price ND
and Hood L: P4 medicine: How systems medicine will transform the
healthcare sector and society. Per Med. 10:565–576. 2013.
View Article : Google Scholar
|
|
12
|
Hood L, Balling R and Auffray C:
Revolutionizing medicine in the 21st century through systems
approaches. Biotechnol J. 7:992–1001. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hood L, Heath JR, Phelps ME and Lin B:
Systems biology and new technologies enable predictive and
preventative medicine. Science. 306:640–643. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Weston AD and Hood L: Systems biology,
proteomics, and the future of health care: Toward predictive,
preventative, and personalized medicine. J Proteome Res. 3:179–196.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
International Human Genome and Sequencing
Consortium: Finishing the euchromatic sequence of the human genome.
Nature. 431:931–945. 2004. View Article : Google Scholar
|
|
16
|
Harmston N, Filsell W and Stumpf MP: What
the papers say: Text mining for genomics and systems biology. Hum
Genomics. 5:17–29. 2010. View Article : Google Scholar
|
|
17
|
Dong L, Wang W, Li A, Kansal R, Chen Y,
Chen H and Li X: Clinical next generation sequencing for precision
medicine in cancer. Curr Genomics. 16:253–263. 2015. View Article : Google Scholar
|
|
18
|
Behjati S and Tarpey PS: What is next
generation sequencing? Arch Dis Child Educ Pract Ed. 98:236–238.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
International Cancer Genome Consortium;
Hudson TJ, Anderson W, Artez A, Barker AD, Bell C, Bernabé RR, Bhan
MK, Calvo F, Eerola I, et al International network of cancer genome
projects. Nature. 464:993–998. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
De Souza YG and Greenspan JS: Biobanking
past, present and future: Responsibilities and benefits. AIDS.
27:303–312. 2013. View Article : Google Scholar
|
|
21
|
Loft S and Poulsen HE: Cancer risk and
oxidative DNA damage in man. J Mol Med (Berl). 74:297–312. 1996.
View Article : Google Scholar
|
|
22
|
Castillo-Pelayo T, Babinszky S, LeBlanc J
and Watson PH: The importance of biobanking in cancer research.
Biopreserv Biobank. 13:172–177. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kinkorová J: Biobanks in the era of
personalized medicine: Objectives, challenges, and innovation:
Overview. EPMA J. 7:42016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Patil S, Majumdar B, Awan KH, Sarode GS,
Sarode SC, Gadbail AR and Gondivkar S: Cancer oriented biobanks: A
comprehensive review. Oncol Rev. 12:3572018.PubMed/NCBI
|
|
25
|
Coppola L, Cianflone A, Grimaldi AM,
Incoronato M, Bevilacqua P, Messina F, Baselice S, Soricelli A,
Mirabelli P and Salvatore M: Biobanking in health care: Evolution
and future directions. J Transl Med. 17:1722019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Watson PH and Barnes RO: A proposed schema
for classifying human research biobanks. Biopreserv Biobank.
9:327–333. 2011. View Article : Google Scholar
|
|
27
|
European Commission: Biobanks for Europe:
A Challenge for Governance. Directorate-General for Research and
Innovation Science in society, EUR 25302 EN; 2012
|
|
28
|
Harris JR, Burton P, Knoppers BM,
Lindpaintner K, Bledsoe M, Brookes AJ, Budin-Ljøsne I, Chisholm R,
Cox D, Deschênes M, et al: Toward a roadmap in global biobanking
for health. Eur J Hum Genet. 20:1105–1111. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yuille M, van Ommen GJ, Bréchot C,
Cambon-Thomsen A, Dagher G, Landegren U, Litton JE, Pasterk M,
Peltonen L, Taussig M, et al: Biobanking for Europe. Brief
Bioinform. 9:14–24. 2008. View Article : Google Scholar
|
|
30
|
Catchpoole DR, Florindi F, Ahern C, Garcia
DL, Mullins P, Van Enckevort E, Zaayenga A, Mayrhofer MT and Holub
P: Expanding the BBMRI-ERIC directory into a global catalogue of
COVID-19-ready collections: A joint initiative of BBMRI-ERIC and
ISBER. Biopreserv Biobank. 18:479–480. 2020. View Article : Google Scholar
|
|
31
|
Brand AM and Probst-Hensch NM: Biobanking
for epidemiological research and public health. Pathobiology.
74:227–238. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Riegman PH, Dinjens WN and Oosterhuis JW:
Biobanking for interdisciplinary clinical research. Pathobiology.
74:239–244. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Mendy M, Caboux E, Lawlor RT, Wright J and
Wild CP: Common minimum technical standards and protocols for
biobanks dedicated to cancer research. (IARC Technical Publication
No. 44). International Agency for Research on Cancer; Lyon:
2017
|
|
34
|
Slokenberga S, Tzortzatou O and Reichel J:
GDPR and biobanking: Individual rights, public interest and
research regulation across Europe. Springer International
Publishing; Cham: pp. 291–307. 2021
|
|
35
|
Lee LW, Griffith J, Zenick H and Hulka BS:
Human tissue monitoring and specimen banking: Opportunities for
exposure assessment, risk assessment, and epidemiologic research.
Environ Health Perspect. 103(Suppl 3): S3–S8. 1995.
|
|
36
|
Palazzani L: Innovation in Scientific
Research and Emerging Technologies: A Challenge to Ethics and Law.
Springer Nature; 2019, View Article : Google Scholar
|
|
37
|
Pitt KE, Campbell L, Skubitz APN, Aamodt
RL, Anouna A, Baird P, Beck JC, Bledsoe M, De Souza Y, Grizzle W,
et al: Best practices for repositories I: Collection, storage, and
retrieval of human biological materials for research. Cell Preserv
Technol. 3:5–48. 2005. View Article : Google Scholar
|
|
38
|
ISBER: Best practices for repositories:
Collection storage, retrieval and distribution of biological
materials for research. Cell Preserv Technol. 6:3–58. 2008.
View Article : Google Scholar
|
|
39
|
Campbell LD, Betsou F, Garcia DL, Giri JG,
Pitt KE, Pugh RS, Sexton KC, Skubitz AP and Somiari SB: Development
of the ISBER best practices for repositories: Collection, storage,
retrieval and distribution of biological materials for research.
Biopreserv Biobank. 10:232–233. 2012. View Article : Google Scholar
|
|
40
|
Campbell LD, Astrin JJ, DeSouza Y, Giri J,
Patel AA, Rawley-Payne M, Rush A and Sieffert N: The 2018 revision
of the ISBER best practices: Summary of changes and the editorial
team's development process. Biopreserv Biobank. 16:3–6. 2018.
View Article : Google Scholar
|
|
41
|
Brochhausen M, Fransson MN, Kanaskar NV,
Eriksson M, Merino-Martinez R, Hall RA, Norlin L, Kjellqvist S,
Hortlund M, Topaloglu U, et al: Developing a semantically rich
ontology for the biobank-administration domain. J Biomed Semantics.
4:232013. View Article : Google Scholar
|
|
42
|
Eklund N, Andrianarisoa NH, van Enckevort
E, Anton G, Debucquoy A, Müller H, Zaharenko L, Engels C, Ebert L,
Neumann M, et al: Extending the minimum information about biobank
data sharing terminology to describe samples, sample donors, and
events. Biopreserv Biobank. 18:155–164. 2020. View Article : Google Scholar
|
|
43
|
Merino-Martinez R, Norlin L, van Enckevort
D, Anton G, Schuffenhauer S, Silander K, Mook L, Holub P, Bild R,
Swertz M and Litton JE: Toward global biobank integration by
implementation of the minimum information about biobank data
sharing (MIABIS 2.0 Core). Biopreserv Biobank. 14:298–306. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Norlin L, Fransson MN, Eriksson M,
Merino-Martinez R, Anderberg M, Kurtovic S and Litton JE: A minimum
data set for sharing biobank samples, information, and data:
MIABIS. Biopreserv Biobank. 10:343–348. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
National Cancer Institute (NCI): NCI Best
Practices for Biospecimen Resources. NCI; Bethesda, MD: 2021,
https://biospecimens.cancer.gov/.
Accessed January 13, 2021.
|
|
46
|
Betsou F, Lehmann S, Ashton G, Barnes M,
Benson EE, Coppola D, DeSouza Y, Eliason J, Glazer B, Guadagni F,
et al: Standard preanalytical coding for biospecimens: Defining the
sample PREanalytical code. Cancer Epidemiol Biomarkers Prev.
19:1004–1011. 2010. View Article : Google Scholar
|
|
47
|
Sándor J, Drakopoulou A and Bárd P: The
legal regulation of biobanks: National report: Greece. (CELEB Paper
Series, No 2). 2009. View Article : Google Scholar
|
|
48
|
Papadavid E, Korkolopoulou P, Levidou G,
Saetta AA, Papadaki T, Siakantaris M, Nikolaou V, Oikonomidi A,
Chatziandreou I, Marinos L, et al: In situ assessment of PI3K and
PTEN alterations in mycosis fungoides: Correlation with
clinicopathological features. 23:931–933. 2014.PubMed/NCBI
|
|
49
|
Argyropoulos KV, Pulitzer M, Perez S,
Korkolopoulou P, Angelopoulou M, Baxevanis C, Palomba ML and
Siakantaris M: Tumor-infiltrating and circulating granulocytic
myeloid-derived suppressor cells correlate with disease activity
and adverse clinical outcomes in mycosis fungoides. Clin Transl
Oncol. 22:1059–1066. 2020. View Article : Google Scholar
|
|
50
|
Mansouri L, Noerenberg D, Young E, Mylonas
E, Abdulla M, Frick M, Asmar F, Ljungström V, Schneider M, Yoshida
K, et al: Frequent NFKBIE deletions are associated with poor
outcome in primary mediastinal B-cell lymphoma. Blood.
128:2666–2670. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Lakiotaki E, Levidou G, Angelopoulou MK,
Adamopoulos C, Pangalis G, Rassidakis G, Vassilakopoulos T, Gainaru
G, Flevari P, Sachanas S, et al: Potential role of AKT/mTOR
signalling proteins in hairy cell leukaemia: Association with
BRAF/ERK activation and clinical outcome. Sci Rep. 6:212522016.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Sachanas S, Pangalis GA, Vassilakopoulos
TP, Korkolopoulou P, Kontopidou FN, Athanasoulia M, Yiakoumis X,
Kalpadakis C, Georgiou G, Masouridis S, et al: Combination of
rituximab with chlorambucil as first line treatment in patients
with mantle cell lymphoma: A highly effective regimen. Leuk
Lymphoma. 52:387–393. 2011. View Article : Google Scholar
|
|
53
|
Pouliou E, Xochelli A, Kanellis G, Stalika
E, Sutton LA, Nava r ro A, Agathangelidis A, Dimosthenous K,
Anagnostopoulos A, Patsouris E, et al: Numerous ontogenetic roads
to mantle cell lymphoma: Immunogenetic and immunohistochemical
evidence. Am J Pathol. 187:1454–1458. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Vassilakopoulos P: Levidou T, Milionis G,
Hartmann V, Lakiotaki S, Sepsa E, Thymara A, Ntailiani I, Spanou P,
Angelopoulou K K M, et al Thioredoxin-1, chemokine (C-X-C motif)
ligand-9 and interferon-γ expression in the neoplastic cells and
macrophages of Hodgkin lymphoma: Clinicopathologic correlations and
potential prognostic implications. Leuk Lymphoma. 58:1–13. 2017.
View Article : Google Scholar
|
|
55
|
Dimtsas GS, Georgiadi EC, Karakitsos P,
Vassilakopoulos TP, Thymara I, Korkolopoulou P, Patsouris E, Kittas
C and Doussis-Anagnostopoulou IA: Prognostic significance of
immunohistochemical expression of the angiogenic molecules vascular
endothelial growth factor-A, vascular endothelial growth factor
receptor-1 and vascular endothelial growth factor receptor-2 in
patients with classical Hodgkin lymphoma. Leuk Lymphoma.
55:558–564. 2014. View Article : Google Scholar
|
|
56
|
Lakiotaki E, Giaginis C, Tolia M,
Alexandrou P, Delladetsima I, Giannopoulou I, Kyrgias G, Patsouris
E and Theocharis S: Clinical significance of cannabinoid receptors
CB1 and CB2 expression in human malignant and benign thyroid
lesions. Biomed Res Int. 2015:8394032015. View Article : Google Scholar
|
|
57
|
Dimonitsas E, Liakea A, Sakellariou S,
Thymara I, Giannopoulos A, Stratigos A, Soura E, Saetta A and
Korkolopoulou P: An update on molecular alterations in melanocytic
tumors with emphasis on Spitzoid lesions. Ann Transl Med.
6:2492018. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Kostaki M, Manona AD, Stavraka I,
Korkolopoulou P, Levidou G, Trigka EA, Christofidou E, Champsas G,
Stratigos AJ, Katsambas A, et al: High-frequency p16(INK) (4A)
promoter methylation is associated with histone methyltransferase
SETDB1 expression in sporadic cutaneous melanoma. Exp Dermatol.
23:332–338. 2014. View Article : Google Scholar
|
|
59
|
Davies NP, Wilson M, Harris LM, Natarajan
K, Lateef S, Macpherson L, Sgouros S, Grundy RG, Arvanitis TN and
Peet AC: Identification and characterisation of childhood
cerebellar tumours by in vivo proton MRS. NMR Biomed. 21:908–918.
2008. View Article : Google Scholar
|
|
60
|
Levidou G, Siakantaris M, Papadaki T,
Papadavid E, Vassilakopoulos TP, Angelopoulou MK, Marinos L,
Nikolaou V, Economidi A, Antoniou C, et al: A comprehensive
immunohistochemical approach of AKT/mTOR pathway and p-STAT3 in
mycosis fungoides. J Am Acad Dermatol. 69:375–384. 2013. View Article : Google Scholar
|
|
61
|
Rassidakis GZ, Medeiros LJ,
Vassilakopoulos TP, Viviani S, Bonfante V, Nadali G, Herling M,
Angelopoulou MK, Giardini R, Chilosi M, et al: BCL-2 expression in
Hodgkin and Reed-Sternberg cells of classical Hodgkin disease
predicts a poorer prognosis in patients treated with ABVD or
equivalent regimens. Blood. 100:3935–3941. 2002. View Article : Google Scholar
|
|
62
|
Georgiadi EC, Sachinis N, Dimtsas G,
Vassilakopoulos TP, Kittas C and Doussis-Anagnostopoulou IA:
Evaluation of apoptosis in classical Hodgkin's lymphoma comparing
different methods. J BUON. 17:746–752. 2012.
|
|
63
|
Lippi G, Simundic AM, Rodriguez-Manas L,
Bossuyt P and Banfi G: Standardizing in vitro diagnostics tasks in
clinical trials: A call for action. Ann Transl Med. 4:1812016.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Mendy M, Caboux E, Lawlor RT, Wright J and
Wild CP: Common minimum technical standards and protocols for
biobanks dedicated to cancer research. World Health Organization,
International Agency for Research on Cancer; Lyon: 2017
|
|
65
|
Betsou F: Quality assurance and quality
control in biobanking. Biobanking of Human Biospecimens: Principles
and Practice. Hainaut P, Vaught J, Zatloukal K and Pasterk M:
Springer International Publishing; Cham: pp. 23–49. 2017,
View Article : Google Scholar
|
|
66
|
Betsou F, Bulla A, Cho SY, Clements J,
Chuaqui R, Coppola D, De Souza Y, De Wilde A, Grizzle W, Guadagni
F, et al: Assays for qualification and quality stratification of
clinical biospecimens used in research: A technical report from the
isber biospecimen science working group. Biopreserv Biobank.
14:398–409. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Spencer DH, Sehn JK, Abel HJ, Watson MA,
Pfeifer JD and Duncavage EJ: Comparison of clinical targeted
next-generation sequence data from formalin-fixed and fresh-frozen
tissue specimens. J Mol Diagn. 15:623–633. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Graw S, Meier R, Minn K, Bloomer C, Godwin
AK, Fridley B, Vlad A, Beyerlein P and Chien J: Robust gene
expression and mutation analyses of RNA-sequencing of
formalin-fixed diagnostic tumor samples. Sci Rep. 5:123352015.
View Article : Google Scholar
|
|
69
|
Norton N, Sun Z, Asmann YW, Serie DJ,
Necela BM, Bhagwate A, Jen J, Eckloff BW, Kalari KR, Thompson KJ,
et al: Gene expression, single nucleotide variant and fusion
transcript discovery in archival material from breast tumors. PLoS
One. 8:e819252013. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Kokkat TJ, Patel MS, McGarvey D, LiVolsi
VA and Baloch ZW: Archived formalin-fixed paraffin-embedded (FFPE)
blocks: A valuable underexploited resource for extraction of DNA,
RNA, and protein. Biopreserv Biobank. 11:101–106. 2013. View Article : Google Scholar
|
|
71
|
Hedegaard J, Thorsen K, Lund MK, Hein AM,
Hamilton-Dutoit SJ, Vang S, Nordentoft I, Birkenkamp-Demtröder K,
Kruhøffer M, Hager H, et al: Next-generation sequencing of RNA and
DNA isolated from paired fresh-frozen and formalin-fixed
paraffin-embedded samples of human cancer and normal tissue. PLoS
One. 9:e981872014. View Article : Google Scholar
|
|
72
|
Kerick M, Isau M, Timmermann B, Sültmann
H, Herwig R, Krobitsch S, Schaefer G, Verdorfer I, Bartsch G,
Klocker H, et al: Targeted high throughput sequencing in clinical
cancer settings: Formaldehyde fixed-paraffin embedded (FFPE) tumor
tissues, input amount and tumor heterogeneity. BMC Med Genomics.
4:682011. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
GitHub: MIABIS: Minimum information about
biobank data sharing repository. https://github.com/BBMRI-ERIC/miabis. Accessed
January 20, 2021.
|
|
74
|
BBMRI-ERIC: Common Services IT: What are
we aiming to achieve? BBMRI-ERIC, Graz. 2021, https://www.bbmri-eric.eu/bbmri-eric/common-service-it/.
Accessed February 29, 2021.
|
|
75
|
Yesley MS: What's ELSI got to do with it?
Bioethics and the human genome project. New Genet Soc. 27:1–6.
2008. View Article : Google Scholar
|
|
76
|
Council of Europe: Convention for the
protection of human rights and dignity of the human being with
regard to the application of biology and medicine: Convention on
Human Rights and Biomedicine (ETS No. 164). https://www.coe.int/en/web/conven-tions/full-list?module=treaty-detail&treatynum=164.
Accessed January 17, 2021.
|
|
77
|
European Commission: Clinical trials -
Regulation EU 536/2014. https://ec.europa.eu/health/human-use/clinical-trials/regulation_en.
Accessed January 17, 2021.
|
|
78
|
Council of Europe: Convention for the
Protection of Human Rights and Fundamental Freedoms (ETS No. 005).
https://www.coe.int/en/web/conventions/full-list/-/conventions/treaty/005.
Accessed January 16, 2021.
|
|
79
|
Council of Europe: Convention for the
Protection of Individuals with regard to Automatic Processing of
Personal Data (ETS No. 108). https://www.coe.int/en/web/conventions/full-list/-/conventions/treaty/108.
Accessed January 16, 2021.
|
|
80
|
Council of Europe: Convention for the
protection of human rights and dignity of the Human Being with
regard to the application of biology and medicine: Convention on
Human Rights and Biomedicine (ETS No. 164). https://www.coe.int/en/web/conventions/full-list/-/conventions/treaty/164.
Accessed January 15, 2021.
|
|
81
|
Council of Europe: Data Protection
Directive. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A31995L0046.
Accessed January 16, 2021.
|
|
82
|
Council of Europe: Recommendation CM/Rec
(2016) 6 of the Committee of Ministers to member States on research
on biological materials of human origin. https://search.coe.int/cm/Pages/result_details.aspx?ObjectID=090000168064e8ff#globalcontainer.
Accessed January 15, 2021.
|
|
83
|
United Nations: Universal declaration of
human rights. https://www.un.org/en/universal-declaration-human-rights/.
Accessed January 15, 2021.
|
|
84
|
Council of Europe: Setting standards of
quality and safety for the donation, procurement, testing,
processing, preservation, storage and distribution of human tissues
and cells. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32004L0023.
Accessed January 16, 2021.
|
|
85
|
World Medical Association: The declaration
of Helsinki - Ethical principles for medical research involving
human subjects (Last amended 2013). https://www.wma.net/policies-post/wma-decla-ration-of-helsinki-ethical-principles-for-medical-research-involving-human-subjects/.
Accessed June 13, 2021.
|
|
86
|
World Medical Association: The declaration
of Taipei on Ethical considerations regarding health databases and
biobanks (Last revised 2016). https://www.wma.net/policies-post/wma-declaration-of-taipei-on-ethical-consid-erations-regarding-health-databases-and-biobanks/.
Accessed June 13, 2021.
|
|
87
|
National Bioethics Commission: On banks of
biological material of human origin (biobanks) in biomedical
research. http://www.bioethics.gr/images/pdf/ENGLISH/OPINIONS_REPORTS/biobanks_recom_eng.pdf.
Accessed December 27, 2020.
|
|
88
|
Daemen J and Rijmen V: The block cipher
rijndael. Lecture Notes in Computer Science. 1820. Springer-Verlag;
Berlin: pp. 277–284. 2000, View Article : Google Scholar
|
|
89
|
Nechvatal J, Barker E, Bassham L, Burr W,
Dworkin M, Foti J and Roback E: Report on the development of the
advanced encryption standard (AES). J Res Natl Inst Stand Technol.
106:511–577. 2001. View Article : Google Scholar
|
|
90
|
Wendler D: Broad versus blanket consent
for research with human biological samples. Hastings Cent Rep.
43:3–4. 2013. View Article : Google Scholar
|
|
91
|
Grady C, Eckstein L, Berkman B, Brock D,
Cook-Deegan R, Fullerton SM, Greely H, Hansson MG, Hull S, Kim S,
et al: Broad consent for research with biological samples: Workshop
conclusions. Am J Bioeth. 15:34–42. 2015. View Article : Google Scholar
|
|
92
|
Gille F, Vayena E and Blasimme A:
Future-proofing biobanks' governance. Eur J Hum Genet. 28:989–996.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Juliusson G and Hough R: Leukemia. Prog
Tumor Res. 43:87–100. 2016. View Article : Google Scholar
|
|
94
|
Haferlach T and Schmidts I: The power and
potential of integrated diagnostics in acute myeloid leukaemia. Br
J Haematol. 188:36–48. 2020. View Article : Google Scholar :
|
|
95
|
Willemze R, Jaffe ES, Burg G, Cerroni L,
Berti E, Swerdlow SH, Ralfkiaer E, Chimenti S, Diaz-Perez JL,
Duncan LM, et al: WHO-EORTC classification for cutaneous lymphomas.
Blood. 105:3768–3785. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Kempf W, Zimmermann AK and Mitteldorf C:
Cutaneous lymphomas-an update 2019. Hematol Oncol. 37(Suppl 1):
S43–S47. 2019. View Article : Google Scholar
|
|
97
|
Willemze R, Cerroni L, Kempf W, Berti E,
Facchetti F, Swerdlow SH and Jaffe ES: The 2018 update of the
WHO-EORTC classification for primary cutaneous lymphomas. Blood.
133:1703–1714. 2019. View Article : Google Scholar :
|
|
98
|
Hernández JM, del Cañizo MC, Cuneo A,
García JL, Gutiérrez NC, González M, Castoldi G and San Miguel JF:
Clinical, hematological and cytogenetic characteristics of atypical
chronic myeloid leukemia. Ann Oncol. 11:441–444. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Hernández L, Beà S, Pinyol M, Ott G,
Katzenberger T, Rosenwald A, Bosch F, López-Guillermo A, Delabie J,
Colomer D, et al: CDK4 and MDM2 gene alterations mainly occur in
highly proliferative and aggressive mantle cell lymphomas with
wild-type INK4a/ARF locus. Cancer Res. 65:2199–2206. 2005.
View Article : Google Scholar
|
|
100
|
Li S, Young KH and Medeiros LJ: Diffuse
large B-cell lymphoma. Pathology. 50:74–87. 2018. View Article : Google Scholar
|
|
101
|
Horsman DE, Gascoyne RD, Coupland RW,
Coldman AJ and Adomat SA: Comparison of cytogenetic analysis,
southern analysis, and polymerase chain reaction for the detection
of t(14; 18) in follicular lymphoma. Am J Clin Pathol. 103:472–478.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Rowley JD: Chromosome studies in the
non-Hodgkin's lymphomas: The role of the 14;18 translocation. J
Clin Oncol. 6:919–925. 1988. View Article : Google Scholar
|
|
103
|
Freedman A and Jacobsen E: Follicular
lymphoma: 2020 Update on diagnosis and management. Am J Hematol.
95:316–327. 2020. View Article : Google Scholar
|
|
104
|
Maitre E, Wiber M, Cornet E and Troussard
X: Hairy cell leukemia. Presse Med. 48:842–849. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Mathas S, Hartmann S and Küppers R:
Hodgkin lymphoma: Pathology and biology. Semin Hematol. 53:139–147.
2016. View Article : Google Scholar
|
|
106
|
Wang HW, Balakrishna JP, Pittaluga S and
Jaffe ES: Diagnosis of Hodgkin lymphoma in the modern era. Br J
Haematol. 184:45–59. 2019. View Article : Google Scholar
|
|
107
|
Swerdlow SH, Campo E, Pileri SA, Harris
NL, Stein H, Siebert R, Advani R, Ghielmini M, Salles GA, Zelenetz
AD and Jaffe ES: The 2016 revision of the World Health Organization
classification of lymphoid neoplasms. Blood. 127:2375–2390. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Jain P and Wang M: Mantle cell lymphoma:
2019 Update on the diagnosis, pathogenesis, prognostication, and
management. Am J Hematol. 94:710–725. 2019. View Article : Google Scholar
|
|
109
|
Klonou A, Korkolopoulou P, Gargalionis AN,
Kanakoglou DS, Katifelis H, Gazouli M, Chlamydas S, Mitsios A,
Kalamatianos T, Stranjalis G, et al: Histone mark profiling in
pediatric astrocytomas reveals prognostic significance of H3K9
trimethylation and histone methyltransferase SUV39H1.
Neurotherapeutics. 18:2073–2090. 2021. View Article : Google Scholar
|
|
110
|
Treisman J and Garlie N: Systemic therapy
for cutaneous melanoma. Clin Plast Surg. 37:127–146. 2010.
View Article : Google Scholar
|
|
111
|
Garbe C, Amaral T, Peris K, Hauschild A,
Arenberger P, Bastholt L, Bataille V, Del Marmol V, Dréno B,
Fargnoli MC, et al: European consensus-based interdisciplinary
guideline for melanoma. Part 2: Treatment-update 2019. Eur J
Cancer. 126:159–177. 2020. View Article : Google Scholar
|
|
112
|
Nikolaou VA, Stratigos AJ, Flaherty KT and
Tsao H: Melanoma: New insights and new therapies. J Invest
Dermatol. 132:854–863. 2012. View Article : Google Scholar
|
|
113
|
Lugowska I, Teterycz P and Rutkowski P:
Immunotherapy of melanoma. Contemp Oncol (Pozn). 22:61–67.
2018.
|
|
114
|
Karia PS, Han J and Schmults CD: Cutaneous
squamous cell carcinoma: Estimated incidence of disease, nodal
metastasis, and deaths from disease in the United States. 2012.J Am
Acad Dermatol. 68:957–966. 2013. View Article : Google Scholar
|
|
115
|
Stratigos AJ, Garbe C, Dessinioti C, Lebbe
C, Bataille V, Bastholt L, Dreno B, Fargnoli MC, Forsea AM, Frenard
C, et al: European interdisciplinary guideline on invasive squamous
cell carcinoma of the skin: Part 1. Epidemiology, diagnostics and
prevention. Eur J Cancer. 128:60–82. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Parikh SA, Patel VA and Ratner D: Advances
in the management of cutaneous squamous cell carcinoma. F1000Prime
Rep. 6:702014. View
Article : Google Scholar : PubMed/NCBI
|
|
117
|
Cranmer LD, Engelhardt C and Morgan SS:
Treatment of unresectable and metastatic cutaneous squamous cell
carcinoma. Oncologist. 15:1320–1328. 2010. View Article : Google Scholar
|
|
118
|
Soura E, Gagari E and Stratigos A:
Advanced cutaneous squamous cell carcinoma: How is it defined and
what new therapeutic approaches are available? Curr Opin Oncol.
31:461–468. 2019. View Article : Google Scholar
|