Cancer consists of more than 200 types with
incomplete knowledge of the origin, tumorigenesis, and progression
(1-3). According to the most recent
statistics, approximately 19.3 million cases of new patients have
been diagnosed with cancer, and a fatality of approximately 10
million (4). The majority of
these massive numbers of deaths are caused by cancer metastasis
(5). Cancer metastasis involves
the process of tumor cells spreading from a primary tumor mass to
different sites through blood and lymphatic vessels. It involves a
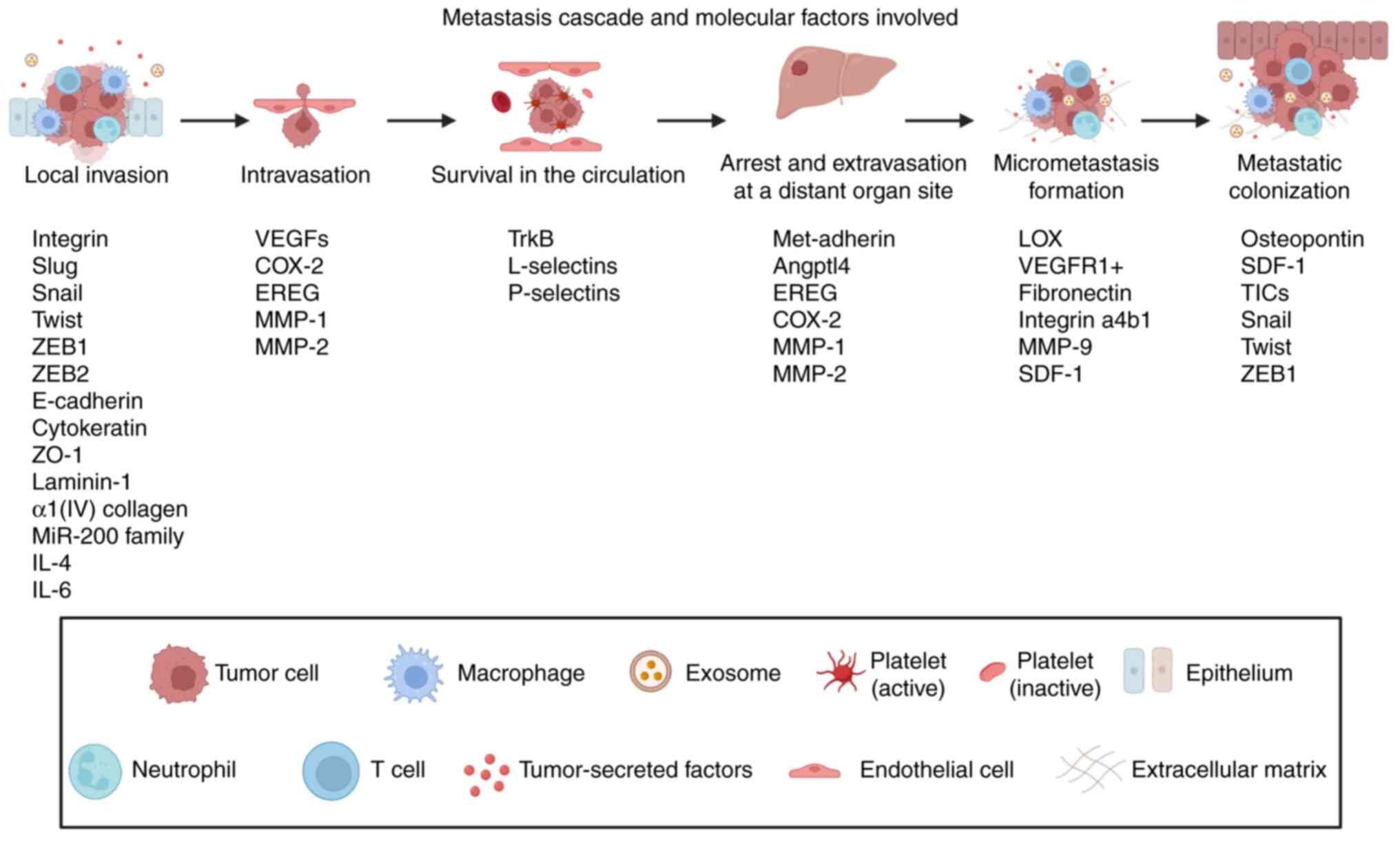
series of events known as the cancer metastasis cascade (Fig. 1). First, the epithelial cancer
cells in primary tumors invade the extracellular matrix (ECM) and
stromal cell layers within the site and enter into the lumina of
the blood vessels. After that, cancer cells deal with several
unfavorable conditions during their transport in the circulation
until they arrive at specific organs. Next, cancer cells
extravasate into the parenchyma of these organs, survive in these
new microenvironments and initiate micrometastases. Finally, cancer
cells restart their proliferative ability at the metastatic sites,
thereby generating neoplastic growths known as 'metastatic
colonization' (6).
During the last few decades, cumulative scientific
discoveries that have been made in the field of molecular and
cellular oncology have transformed clinical practice. For instance,
the precise detection of cancer at an earlier stage and tailoring a
therapeutic approach toward tumor type-specific intervention have
been improved significantly (7).
Molecular phenotypes associated with cancer metastasis have been
extensively studied which has led to identifying potential
metastasis-associated targets. The design of molecular-targeted
therapies that specifically interact with those targets, whether
they are components of a specific intracellular pathway or
cell-cell communication signaling, have demonstrated potential
benefits by overcoming systemic toxicities associated with
traditional treatments such as chemotherapy and radiation therapy
as well as improving the pharmacokinetics and pharmacodynamics of
these traditional treatments (8,9).
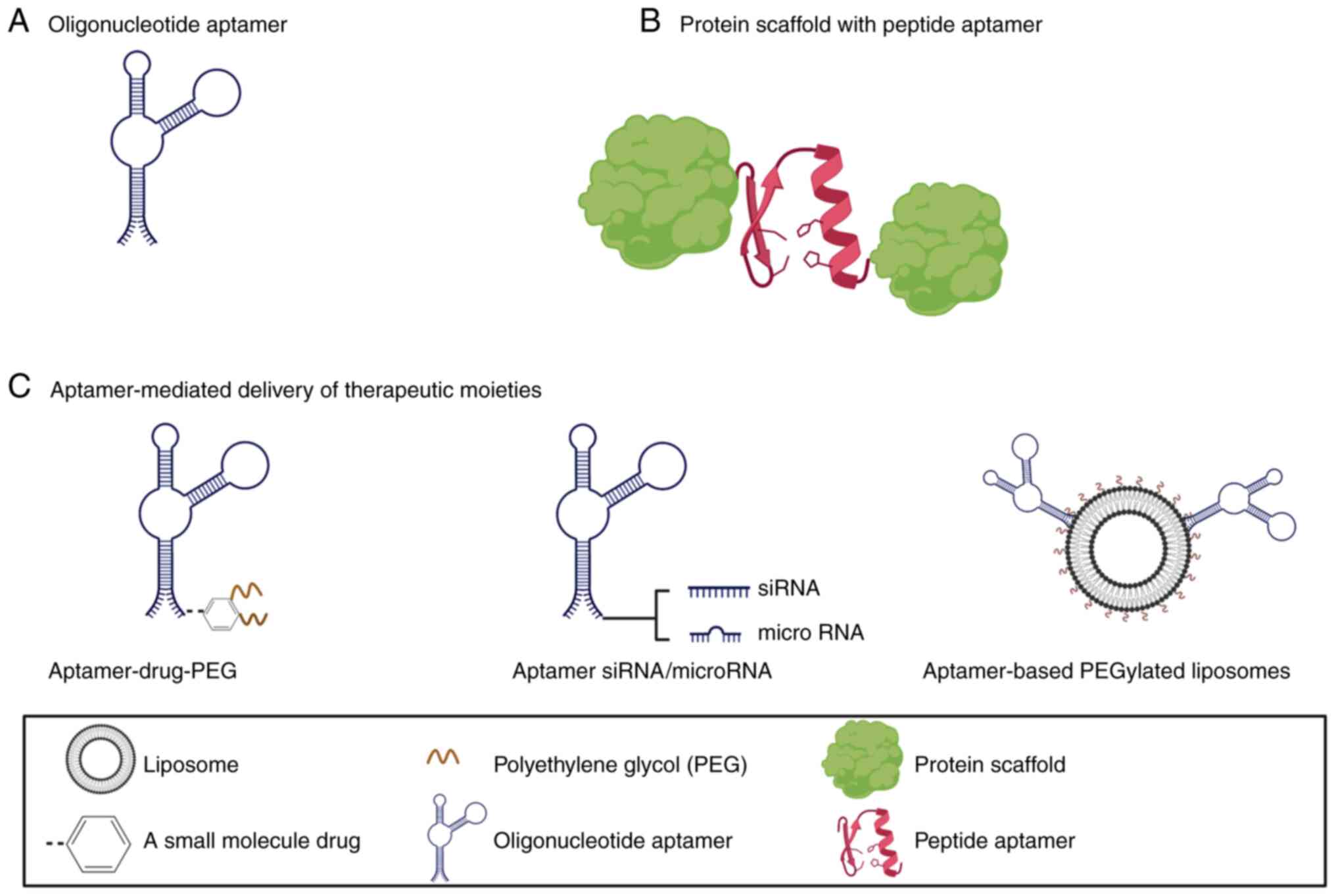
One of the most recently developed
molecular-targeted therapy is short sequences of single-stranded
nucleic acids (ssDNA or RNA) or amino acids which are known as
oligonucleotide or peptide aptamers, respectively (Fig. 2). Aptamers mimic antibodies in
which they possess high selectivity when they bind with the
selected targets. The first observation of the binding capability
of aptamers was reported when researchers found that a
subpopulation of isolated RNA molecules was able to bind specific
ligands. An in vitro technique called SELEX (Systemic
Evolution of Ligands by Exponential Enrichment) was introduced as a
procedure to generate these oligonucleotide aptamers (10,11). The three-dimensional structure of
a short sequence (20-100 bases long) RNA or ssDNA gives aptamers
the capability to interact specifically with particular ligands
with selectivity and affinity similar to those of antibodies.
However, aptamers are more favorable compared to antibodies due to
their cheap and rapid synthesis (12,13). A few years after oligonucleotide
aptamers had been introduced, a research study demonstrated that
designing a short peptide (5 to 20 amino acids long) then embedding
it in a protein scaffold generated a protein with high specificity
to a selected target. Therefore, a new concept launched for this
type of aptamer is called peptide aptamer (14-16). To date, peptide and
oligonucleotide aptamers have been developed and exploited for
different diagnostic and therapeutic purposes. Aptamers have been
employed for diagnostic applications in cancer detection and
imaging (17) and infectious
disease (18). For therapeutic
purposes, aptamers have been utilized to treat different diseases
such as cancer (17,19), infectious diseases (18,20), coagulation disorders (21), diabetic nephropathy (22) and ocular vascular diseases
(23). This review aims to
summarize recent advances in utilizing aptamers as a treatment
strategy against cancer metastasis specifically and highlight key
mediators and molecular factors involved during different
metastasis stages. Moreover, potential opportunities to improve
aptamer-based antimetastatic therapeutics are discussed. We used
Web of Science and PubMed databases to retrieve the most recent
information in this review.
Local invasion is the access of the cancer cells
that were contained within the primary tumor into the surrounding
stroma, and then into the adjoining normal parenchymal tissue. To
enter the stroma, the carcinoma cells modify the ECM, which has a
significant function in arranging epithelial tissues. The integrins
(transmembrane proteins) bind to the ECM and form the
integrin-mediated cell-matrix adhesions (24). This binding initiates several
pathways leading to signal transduction events within the carcinoma
cells that leads to disturbances in cell polarity, proliferation,
invasiveness, and survival (25).
A majority of the carcinomas have the ability to
invade as cohesive multicellular units known as a collective
invasion. However, a single cancer cell may attack through two
different mechanisms: integrin-dependent (mesenchymal invasion) and
integrin-independent, Rho/ROCK-dependent (amoeboid invasion)
pathways (26). It may be noted
that the cancer cells may interconvert between these mechanisms due
to the change in the microenvironment (27). While the patterns of cancer cell
invasion are classified as collective and individual cell migration
(28), the individual cell
invasion mechanism is not compatible with an important element of
epithelial tissue organization, specifically the
E-cadherin-mediated intercellular junctions that lead to the
development of the epithelial cell sheets and remain associated
with the surrounding epithelial cells. To attenuate tight junctions
and cellular polarity, carcinoma cells may undergo
epithelial-mesenchymal transition (EMT). EMT is vital for different
facets of normal embryonic morphogenesis which ultimately help to
liberate cancer cells from epithelial cell sheets (29). Several transcription factors,
namely Slug, Snail, Twist, zinc finger E-box-binding homeobox 1
(ZEB1), and ZEB2 play significant roles in EMT. They trigger
mesenchymal entry by downregulating the expression of E-cadherin
and other epithelial markers such as cytokeratin, zonula
occludens-1 (ZO-1), laminin-1, and α1(IV) collagen (29). In addition, other regulatory
non-coding genes, such as microRNAs (miRNAs/miRs), govern EMT. The
miR-200 family regulates EMT programs by post-transcriptionally
suppressing the expression of ZEB1 and ZEB2, while on the other
hand, ZEB1 and ZEB2 inhibit the transcription of the miR-200
family. Such a relationship establishes a double-negative-feedback
loop that operates as a bistable switch controlling the fate of
cancer cells to go in either the mesenchymal or epithelial state
(29). The debilitation of the
ECM is further aggravated by active proteolysis activated by the
matrix metalloproteinases (MMPs), which promotes the invasion of
carcinoma cells to the stromal compartment. As the stroma becomes
more chronically inflamed upon tumor progression, cancer cells are
challenged by fibroblasts, endothelial cells, adipocytes,
mesenchymal stem cells from the bone marrow, macrophages, and other
immune cells (30). These stromal
cells further influence the aggressiveness of carcinoma cells via
different types of heterotypic signaling. For example, the
invasiveness of breast cancer is stimulated by IL-6 secreted by the
adipocytes (31). Similarly, it
has also been demonstrated that the cathepsin protease activity in
tumor-associated macrophages is activated by the secretion of IL-4,
which fuels the invasiveness of carcinoma cells (32).
Intravasation occurs when the carcinoma cells enter
into the lumina of lymphatic or blood vessels. This process is
normally observed in human tumors and signifies a vital prognostic
marker for its progression; dissemination through blood vessels is
the key mechanism for the spread of metastatic carcinoma cells
(33). Intravasation is enhanced
by molecular variations that increase the capacity of carcinoma
cells to penetrate the microvessels that are composed of pericyte
and endothelial cells. Intravasation is influenced by the vascular
endothelial growth factors (VEGFs) secreted by the tumor cells
which enhance the generation of new blood vessels within their
local microenvironment through neoangiogenesis. In comparison to
the normal blood vessels, the neovasculature developed by carcinoma
cells is prone to leakiness and is subjected to continuous
reconfiguration (34).
Furthermore, it has been reported earlier that cyclooxygenase-2
(COX-2), epiregulin (EREG), MMP-1, and MMP-2 synergistically
promote breast carcinoma intravasation due to their capacity to
stimulate neoangiogenesis (35).
After the intravasation has been achieved, the
carcinoma cells are widely transported through the systemic
circulation, known as circulating tumor cells (CTCs). Before
reaching other organs, the CTCs deal with different types of
stresses for its survival, such as the absence of the
integrin-dependent adhesion to ECM components that is required for
cell survival. Consequently, the epithelial cells undergo anoikis,
which is a form of apoptosis that is activated by the loss of
attachment to the substratum (36). However, the tyrosine kinase TrkB
has been observed to suppress anoikis (37). Additionally, the tumor cells also
face potential damage due to the hemodynamic shear forces and the
innate immune system, specifically natural killer cells. These
challenges have been observed to be evaded simultaneously through
the formation of large emboli through the interactions with blood
platelets (regulated by the tissue factor and/or L- and P-selectins
by the carcinoma cells) (30). In
this way, the platelet-coated tumor cells escape immune detection
until they are arrested at distant organ sites.
Despite the capacity of the CTCs to spread to a wide
range of distant organ sites, it has been previously observed that
specific carcinoma types metastasize to specific organs which leads
to the proposal of the 'seed and soil hypothesis' (6). Seed and soil hypothesis suggests
that cancer cells (seed) disseminated from the primary tumor spread
to all organs, but only specific microenvironments (fertile soil)
that support metastatic tumor formation in specific organs. Once
the cancer cells reach the suitable organs, the organ
microenvironment supports the attachment and seeding of cancer
cells in the specific organ. This is because some carcinoma cells
depend on specific adhesive interactions in particular tissues that
assist their trapping. For example, the generation of metadherin in
breast cancer cells initiates the spread of carcinoma cells to the
lungs through increased binding to the pulmonary vasculature
(38). In addition, it has been
observed that a proinflammatory environment in the liver causes
Kupffer cells to secrete chemokines which upregulate vascular
adhesion receptors and ultimately enable adhesion of the
circulating colorectal and lung carcinoma cells to the liver
microvasculature (39). Then, the
trapped cancer cells grow inside the vasculature and develop a
microcolony that ultimately penetrates through the surrounding
vessels, resulting in the straight contact of the tumor cells with
the tissue parenchyma (40). In
another way, the carcinoma cells might rupture from vessel lumina
into the tissue parenchyma by going through the endothelial cell
and pericyte layers that separate the vessel lumina from the
stromal microenvironment. This process is known as extravasation.
Further, the physical barriers to extravasation may be breached due
to the ability of the primary tumors to secrete factors that cause
an imbalance in the microenvironments and induce vascular
hyperpermeability. For example, the factors protein
angiopoietin-like-4 (Angptl4), epiregulin (EREG), COX-2, MMP-1, and
MMP-2, disrupt pulmonary vascular endothelial cell-cell junctions
in order to facilitate the extravasation of breast carcinoma cells
in the lungs (35,41).
The formation of micrometastases is initiated
following the survival of the extravasated cancer cells in the
parenchyma of distant tissues. It is worth mentioning that several
factors contribute to this stage of cancer metastasis including the
type of stromal cells, ECM constituents, growth factors, and
cytokines. In the beginning, cancer cells establish a
'premetastatic niche' to ascertain the compatibility in the foreign
microenvironment (42). For this
to happen, the primary tumors liberate systemic signals consisting
of lysyl oxidase (LOX) (43),
that stimulates organ-specific upregulation of fibronectin from
fibroblasts, which in turn, activates VEGF receptor 1-positive
(VEGFR1+) hematopoietic progenitor cells from the bone
marrow to these prospective sites of metastasis through homing
interactions between the deposited fibronectin and its cognate
receptor, integrin a4b1that is expressed by the hematopoietic
progenitor cells. The hematopoietic progenitor cells secrete MMP-9
that changes the immediate microenvironments at these loci. The
MMP-9 activation triggers the release of various integrins and
discharge of molecules from the ECM, such as the carcinoma cell
chemoattractant stromal cell-derived factor 1 (SDF-1) (42). All of these changes convert the
distant microenvironments into growth sites for the disseminated
tumor cells.
Even after the successful survival of the tumor
cells in the new microenvironment, it is still not ensured that
they can grow and form metastases, the process known as metastatic
colonization. Instead, it has been observed that a large number of
tumor cells either slowly perish over a period of time or are
sustained as microcolonies during long-term dormancy, retaining the
overall cell number (44). These
dormant microcolonies may continue to remain dormant because of
incompatibilities with the foreign microenvironments that surround
them (44), such as in mammary
carcinoma cells, where the focal adhesion kinase (FAK), integrin
b1, and Src pathways are unable to engage within distant tissues
(45-47). However, they may escape dormancy
to initiate active proliferation cell-nonautonomous mechanisms that
are stimulated by osteopontin (OPN) or SDF-1 (48,49). Secondly, the dormant microcolonies
may proliferate continuously but the overall number may remain the
same due to the high apoptotic rate. For example, the prostate
tumor cell secretes prosaposin (Psap) that inhibits metastatic
colonization by upregulating the anti-angiogenic factor
thrombospondin-1 in stromal cells (50). Metastatic colonization is also
dependent on another attribute known as 'tumor-initiating cells'
(TICs), which possess such an extensive self-renewal capacity to
achieve malignant growth. The entry into the TIC state is promoted
by miRNAs and the EMT-promoting transcription factors, such as
Snail, Twist, and ZEB1, as already discussed (51).
Exploiting aptamers as a new generation of
therapeutics has attracted the attention of the scientific
community due to various advantages that are offered by aptamers.
An individual aptamer possesses high specificity since it depends
on its three-dimensional conformation to bind to its specific
target. The aptamer molecule binds to the target through a hydrogen
bond, electrostatic interaction, van der Waals, hydrophobic
interactions, or stacking interactions (14,52). Moreover, aptamers can be
chemically synthesized with flexible customization providing an
opportunity to improve pharmacokinetics and meet a wide range of
applications needed (53,54). They can bind diverse targets,
ranging from small molecules, proteins to viruses and cells
(55-58). From a therapeutic perspective,
aptamers can be used to exert pharmacological action by themselves
as agonists (target activation), antagonists (target inhibition),
or to act as ligands for targeted delivery of therapeutics
(12) (Fig. 2). Most of the developed aptamers
fall into the former category. Pegaptanib (Macugen®) is
the first FDA-approved aptamer and works by antagonizing the action
of vascular endothelial growth factor (VEGF) (23). For targeted delivery purposes,
aptamers can be constructed to deliver a wide range of diagnostic
or therapeutic moieties such as fluorescent materials,
radioisotopes, cytotoxic drugs, RNA oligonucleotides, and
nanoparticles (59-63). In addition, a multifunctional,
aptamer-based theranostic conjugate can be designed by coupling a
diagnostic marker and a therapeutic moiety with the aptamer for
simultaneous diagnosis and treatment (64-66).
Once an aptamer enters the body, it is susceptible
to various factors that minimize or prevent its therapeutic action.
It is vital to implement strategies to overcome obstacles and
produce aptamers suitable for clinical settings. Stability in the
circulation and tumor microenvironment, rapid excretion by the
kidney, and delivery to intracellular targets affect the
therapeutic actions of the aptamers. First, oligonucleotide
aptamers can undergo enzymatic degradation by nucleases present in
the circulation and tumor microenvironment. Strategies to produce
nuclease-resistant aptamers include chemical modification of the
structure by attaching a functional group to one of the following
positions: 2′position of monosaccharide and 3′ or 5′termini of the
aptamer (67,68). Pegaptanib aptamer, an approved
therapeutic aptamer against age-related macular degeneration, is an
example of such a modification. Another strategy is synthesizing an
RNA or DNA backbone composed of L-ribose or
L-deoxyribose for RNA and ssDNA aptamers, respectively.
Spiegelmer is the name used to refer to these types of aptamers.
This strategy relies on the fact that nucleases degrade
D-oligonucleotide while the L-oligonucleotide is
resistant to degradation (68,69). A second challenge facing an
aptamer inside the body is the rapid excretion of the aptamer by
the kidneys. The low molecular weight of the aptamer is responsible
for the short-time presence of an aptamer in the circulation. An
effective approach to overcome this obstacle is by conjugating the
aptamer with cholesterol or high molecular weight moieties, such as
proteins and polyethylene glycol (70-72). Aptamer-PEG conjugate possesses an
enhanced half-life in the circulation compared to the unconjugated
form of the aptamer (72).
Crossing the cell membrane is another obstacle that can prevent an
aptamer from exerting its pharmacological action. While the
majority of targeted aptamers can easily interact with targets that
are present in the circulation or on the surface of the cancer
cells, some aptamers face difficulties crossing the cell membrane
to reach their intercellular target. To overcome this obstacle,
several strategies have been explored. The first strategy is the
use of cell-internalization SELEX to develop a therapeutic
cell-internalizing aptamer which has the capability to bind its
target on the cell membrane and subsequently internalize to exert
its action (73-75). The second strategy relies on
coupling the therapeutic aptamer to protein transduction domains
(PTDs), also known as cell-penetrating peptide. PTDs can cause the
internalization of the therapeutic aptamer (76-78). The third strategy depends on the
cellular uptake of a vector containing the DNA sequence of the
therapeutic aptamer which is subsequently expressed
intracellularly. The expressed aptamer is called an intramer. After
cellular uptake of the vector by the targeted cell, intramer
expression takes place inside the cells which makes the intramer
available to exert its action (79,80).
A small population of cancer cells inside the tumor
mass, known as cancer stem cells (CSCs), have been found to possess
the capability of self-renewal and generation of cancer progeny
cells (81). These cells defeat
the process of anoikis, a type of programmed cell death that is
triggered when the cells are removed from the surrounding ECM. In
addition, research has found that these CSCs are resistant to
therapeutic drugs since there is an upregulation in the expression
of the ATP binding cassette transporter which results in drug
efflux. In vitro and in vivo studies show that CSCs
play a role in the formation of metastatic nodules (81-83). For example, it has been reported
that the pluripotent genes octamer-binding transcription factor 4
(OCT4) and NANOG force-expressed in lung
adenocarcinoma (LAC) increase the tumorigenic and metastatic
capability of the cells since they induce EMT through the Slug
protein. In addition to the increase in the number of
CD133+ cells, the new cells have increased
sphere-forming ability, drug resistance, and migration (84). In addition, CD24−/low
breast cancer stem cells were found to have an increased capability
for the formation of the tumor as compared to CD24+
breast cancer cells. Several important pathways are engaged in the
CSC self-renewal process, such as the Wnt/β-catenin and NOTCH
pathways. Further, the stemness of CSCs is maintained through the
hepatocyte growth factor (HGF), and the transcription factors,
OCT4, NANOG, SOX2, and BMI-1 (85-89). OCT4 expression has been reported
to induce dysplasia and expansion of progenitor cells in the
intestines (90). Furthermore,
SRY-box transcription factor 2 (SOX2) maintains the vital signaling
cascades for tumorigenesis. BMI1 proto-oncogene, polycomb ring
finger (BMI-1) has a vital role in the self-renewal of normal stem
cells. It has been observed that the knockdown of BMI-1 expression
in CD133+ laryngeal cancer cells led to the restriction
of cell growth, colonization, cell invasion in vitro, and
tumorigenesis in vivo (91). It has been earlier demonstrated
that the Y-box binding protein 1 could augment the stemness
characteristics of human hepatoma cell lines (92,93). Moreover, the KRAS signal c-MYC
axis supports the advancement of stem cell characteristics in
pancreatic cancer (94).
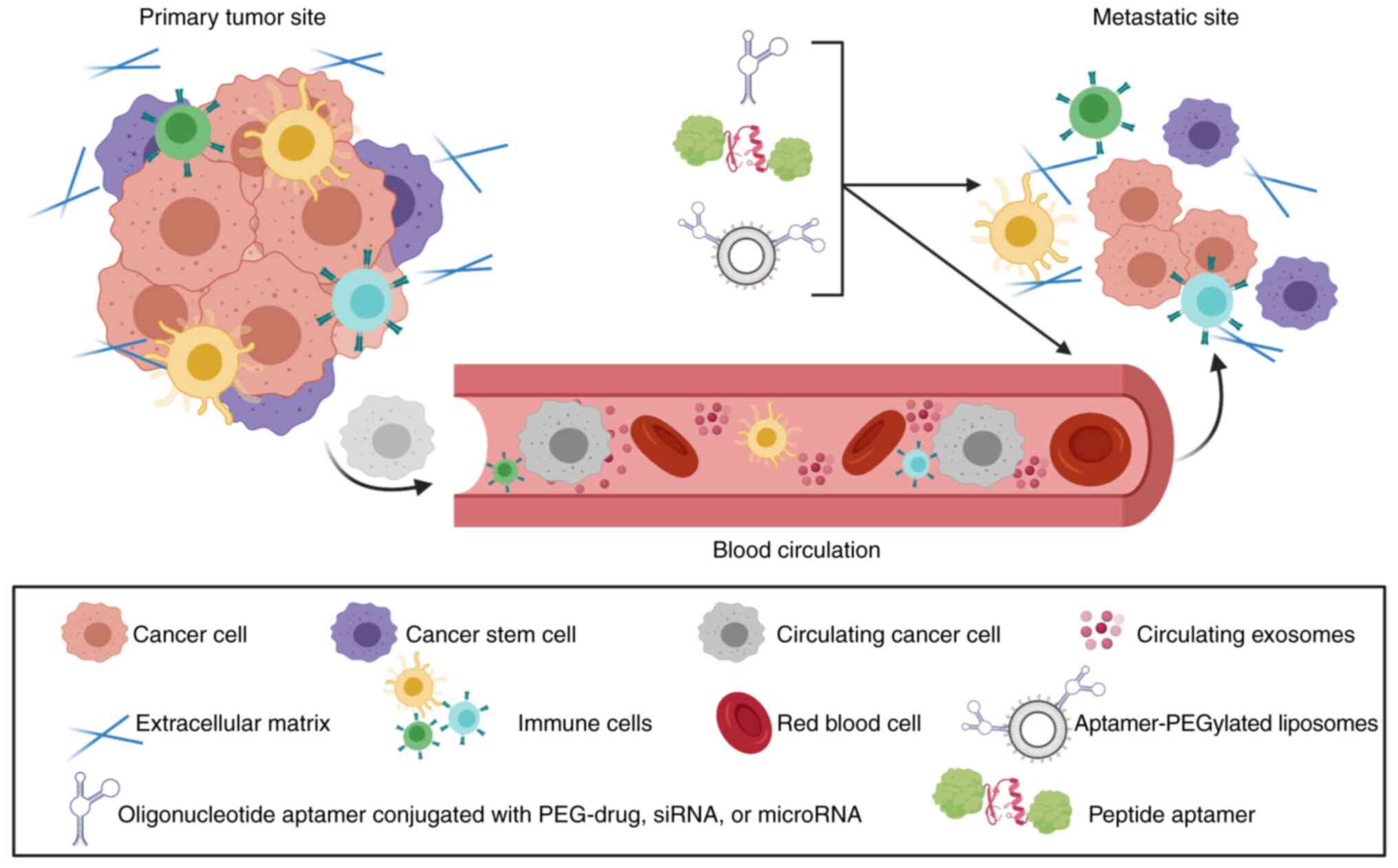
The selective targeting capability of aptamers has
been exploited to deliver cytotoxic drugs or nanoparticles carrying
different types of gene therapy or small-molecule drugs to CSCs
(Fig. 3). These therapeutic
aptamers selectively target proteins overexpressed on the surface
of the cell membrane of CSCs such as, but not limited to, human
epidermal growth factor receptor 2 (HER2), CD133, CD44, CD20, and
EpCAM receptors. In addition, CSC-targeted aptamers were developed
against different types of cancer (95-100). A multifunctional nanoparticle
decorated with RNA aptamer that can specifically target the HER2
receptor overexpressed on the surface of human breast cancer cells
was examined in vitro and in vivo. The HER2 aptamer
was able to deliver the nanoparticle carrying two different siRNAs
against mediator complex subunit 1 (MED1) to HER2-overexpressing
breast cancer cells and inhibit metastatic behavior of the cells
in vitro. An in vivo study demonstrated that the
targeted nanoparticle eliminated breast cancer metastatic nodules
in the lung and significantly downregulated the expression levels
of proteins involved in cancer metastasis such as c-Myc, MMP-9,
Trefoil factor 1 (TFF-1), and cyclin D1. Most importantly, the
examination of the stem cell population showed a significant
decline and complete depletion of
CD44+/CD24−/low stem cells after monotherapy
and combination therapy with tamoxifen, respectively (98). Moreover, RNA aptamers targeting
CD133 receptors that present on the surface of breast cancer cell
stem cells were used to deliver nanoparticles carrying
anti-microRNA to inhibit microRNA-21. An in vitro study
demonstrated selective nanoparticle uptake by breast cancer stem
cells compared to a control cell line with a loss of metastatic
capability of the stem cells (96). In addition, functional
nanoparticles decorated with two different aptamers against CD44
and transmembrane glycoprotein mucin 1 antigen (MUC1) were able to
simultaneously deliver doxorubicin to CSCs and cancer cells,
respectively. Dual-aptamer targeting nanoparticles inhibited breast
cancer metastasis to the lung in an animal model of breast cancer
metastasis (100).
Following the progression of the disease in the
primary organ, the cancer cells leave the organ, survive in the
blood circulation, and metastasize in an organ-specific manner.
This process was first reported by Stephen Paget (101). Research evidence has
demonstrated the vital role of CTCs in the development of
metastasis. Poor prognosis is associated with the increased numbers
of CTCs in the circulation. This makes targeting CTCs a novel
strategy to disrupt cancer metastasis cascade (102). Comprehensive knowledge
established concerning molecular changes such as overexpression of
adhesion molecules that support the attachment of CTCs to the
tissues of the distant organs led scientists to develop
therapeutics that block or prevent CTCs from attaching to the new
organ. A clinical study examining the impact of using anti-EpCAM
monoclonal antibodies, which target EpCAM, which is overexpressed
in CTCs, on patients with metastatic colorectal carcinoma was
conducted. The study demonstrated a significant prolongation of
patient survival (103). Another
treatment strategy that showed prolonged survival of an animal
model of metastatic ovarian cancer involved utilizing
nanotechnology and a magnetic field to isolate CTCs (104).
The development of aptamer-based therapeutics that
target receptors that are involved in CTC attachment has shown
effective prevention of cancer metastasis. DNA aptamer that
displayed the capability to selectively target carcinoembryonic
antigen (CEA), which plays a critical role in CTC adhesion and
implantation, has been developed. Researchers mixed the
anti-adhesive DNA aptamer with CEA-overexpressing colonic carcinoma
cells before they injected these pretreated cells intraperitoneally
in a mouse model. The in vivo study showed a significant
decrease in the numbers and volumes of metastatic nodules compared
to the control group (105,106). Another study developed a DNA
aptamer against sialyl Lewis X (sLex), which helps
cancer cells to bind endothelial-selectin (E-selectin) and
metastasize to distant organs. In vitro study demonstrated
that treating metastatic hepatocellular carcinoma cells (HepG2)
with the DNA aptamer against sLex significantly
inhibited adhesion and migration of HepG2 cells (107,108).
Extracellular vesicles (EVs) are cell-derived,
cytosol-containing vesicles that are involved in cell
communications with the surrounding environment. They have been
categorized based on the size into three types: exosomes (30-100 nm
diameter), microvesicles (MVs) (100-1000 nm diameter), and larger
vesicles known as oncosomes (1-10 µm diameter) (109-112). It has been found that EVs play a
critical role in supporting metastasis. Tumor-secreted EVs contain
thousands of active constituents such as lipids, proteins, and
genetic materials for intercellular communication (113-116), which act as messengers in local
and distant microenvironments (117-119). RNAs function as the chief
bioactive factor of tumor cell-derived EVs, together with other
non-coding RNAs namely microRNAs (miRNAs), long non-coding RNAs
(lncRNAs), and circular RNAs (120-122). These non-coding RNAs delivered
by exosomes to recipient cells can control the expression of many
genes to support oncogenic reprogramming of malignant cells, tumor
growth, local invasion, and premetastatic or metastatic niches
formation (123-126). For example, it has also been
reported that exosomes can destroy the blood-brain barrier by the
action of miR-181c that increases central nervous system (CNS)
metastasis (127). Furthermore,
the breast cancer cell-derived exosomes were found to utilize
miR-122 to create a metastatic niche in the brain and enhance
disease progression by downregulating glucose uptake in non-tumor
cells and rerouting the available nutrients for cancer cells
themselves (128). Moreover, the
exosomes also adversely affect the immune function leading to an
immunosuppressed phenotype and facilitating tumor progression
(129). It has been observed
that the c-Myc mRNA in the recipient microglia and
macrophages was decreased due to the uptake of glioblastoma-derived
exosomes by microglia/macrophages that led to an immunosuppressive
phenotype mediated by the transport of miR-21 and miR-451 (130). It is worth mentioning that EVs
can be released from stroma cells and support tumor metastasis. For
example, exosomes released by astrocytes were found to downregulate
phosphatase and tensin homolog (PTEN), a tumor-suppressor
gene, in brain tumor cells leading to elevated oncogenicity
(131).
The growing body of evidence regarding the crucial
role of EVs in the development of cancer metastasis has attracted
some researchers to identify aptamers that target EVs and seek to
develop aptamer-based therapeutics to treat cancer metastasis.
Recently, a novel modification of SELEX technology, which has been
called Exo-SELEX, has been established to identify aptamers that
specifically bind cell-derived exosomes. Using the most aggressive
subtypes of breast cancer, triple-negative and HER2+,
Exo-SELEX helped researchers to identify novel Ex-50.T aptamer. In
addition, in vitro assessment of the therapeutic activity of
EX-50.T was carried out using MDA-MB-231 and MCF-7 cell lines. The
study relied on the fact that treating MCF-7 cells with exosomes
derived from the highly metastatic MDA-MB-231 cells stimulates
MCF-7 migration. The results demonstrated that MCF-7 cell migration
was significantly inhibited when treating MCF-7 cells with exosomes
that were pre-incubated with Ex-50.T (132). Another approach for targeting
cancer cell-derived EVs is with the help of nanotechnology.
Researchers have exploited the well-known fact regarding the liver
uptake of mesoporous silica nanoparticles and the subsequent
elimination of the nanoparticles into the small intestine to
develop nanoparticles that specifically bind and eliminate
circulating EVs from the blood. Mesoporous silica nanoparticles
decorated with an aptamer that specifically target epidermal growth
factor receptor (EGFR-targeting aptamer) that presents on the
surface of EVs were developed. In vivo study to monitor the
biodistribution of injected, labeled exosomes derived from
metastatic lung cancer showed that the aptamer-targeted
nanoparticles were able to significantly increase the accumulation
of labeled exosomes to the liver and consecutively to the small
intestine. In addition, aptamer-targeted nanoparticles inhibited
the pulmonary metastasis formation in a subcutaneous murine tumor
model (133).
Immune cells can play dual opposite roles in cancer.
Immune cells can recognize and kill immunogenic cancer cells and
inhibit tumor growth. Conversely, they can promote tumor growth by
inducing the formation of new blood vessels, known as angiogenesis,
and facilitating cancer metastasis (134-136). Intercellular communication
between cancer cells, immune cells, and other stroma cells in the
tumor microenvironment is responsible for this observed plasticity
of immune cell function. Myeloid-derived suppressor cells (MDSCs)
and regulatory T cells (Tregs) recruited to the primary and
secondary tumor sites contribute to the immunosuppressive
environment. They are recruited in response to numerous growth
factors, chemokines, and cytokines that are secreted by the cancer
cells and other stromal cells (137-141). Through different mechanisms,
MDSCs and Tregs negatively affect the antitumor activity of NK
cells and prevent tumor infiltration of antitumor cytotoxic
CD8+ T cells (139,142). In addition, the types and levels
of bioactive molecules and growth factors secreted in the tumor
microenvironment and involved in signaling pathways can modulate
the function of immune cells. For example, pro-inflammatory
cytokines stimulate the antitumor activity of the immune cells
while anti-inflammatory cytokines create an immunosuppressive
environment that shifts the function of immune cells toward tissue
repair and regeneration. Interleukin-4 (IL-4) and interleukin-13
(IL-13) can shift tumor-associated macrophages toward the
alternatively activated M2 phenotype which promotes tumor growth
and stimulates the metastatic behavior of cancer cells. Similarly,
transforming growth factor-β (TGF-β) shifts tumor-associated
neutrophils toward a protumor phenotype (141,143,144). The communication between cancer
cells and the immune cells continues during different stages of the
cancer metastasis cascade. During cancer cell invasion and
intravasation, immune cells secrete enzymes, such as MMPs, that
contribute to ECM remodeling. In addition, the newly developed
blood vessels facilitate cancer cell spread to different organs.
Receptor activator of nuclear factor-B ligand (RANKL) which is
secreted by T regulator cells improves CTC survival during the
circulation and contributes to the development of the
pre-metastatic niche in the secondary site (145,146). More details regarding the role
of immune cells in cancer metastasis are reviewed elsewhere
(147,148).
The use of aptamer-based therapeutics to inhibit the
recruitment and function of immunosuppressive cells and modulate
the immune cell phenotype from a protumor to an antitumor phenotype
are promising strategies by which to inhibit cancer metastasis. The
development of aptamer-based therapeutics that prevent
immunosuppressive cells from exerting their action has been
exploited. The interleukin 4 receptor-α (IL4R-α) antagonist aptamer
that targets IL4R-α-expressing M2 tumor-associated macrophages and
myeloid-derived suppressor cells led to the elimination of
immunosuppressive cells in vivo. Subsequently, the numbers
of antitumor CD8+ T cells were increased which
suppressed the formation of pulmonary metastasis of 4T1 metastatic
breast cancer cells (149). The
utilization of aptamers to deliver therapeutics to tumor metastases
also has been developed. Doxorubicin-loaded liposomes decorated
with a T1 aptamer that target MDSCs showed significant depletion of
MDSCs and increased intratumoral accumulation of cytotoxic T cells
in animals with bone metastasis of MDA-MB-231 breast cancer cells
(150). In addition, the 4-1BB
aptamer was used to deliver a small non-coding antisense RNA
(sasRNA) to Treg cells for transcriptional gene silencing of a key
regulator of the immunosuppressive phenotype of Treg cells.
Aptamer-sasRNA conjugate inhibited the Treg immunosuppressive
phenotype in vitro and significantly improved coadministered
antitumor vaccine against B16F10 metastatic melanoma cells that
were grown subcutaneously in vivo (142,151). Similar findings have been
reported after using an aptamer that targets cytotoxic T
lymphocyte-associated antigen 4 (CTLA4) to deliver STAT3 siRNA to
Treg cells. The CTLA4-STAT3 siRNA conjugate depleted Treg cells in
the primary and metastatic sites and prevented pulmonary metastasis
of B16 melanoma cells in vivo (19). In addition, aptamers that target
and prevent the functions of anti-inflammatory cytokines or their
receptors can inhibit immune cell polarization toward a protumor
phenotype. Aptamers that target IL-6, IL-6R, IL-10R have been
developed (152-155), yet their therapeutic activities
against metastasis need to be examined.
The development of aptamer-based anticancer
therapeutics has been increasingly growing in the last few years.
Encouraging findings introduce aptamer-based therapeutics as a
potential class of agents in the treatment of primary tumors.
However, cancer metastasis is the main cause for cancer-related
deaths, thus preventing or treating cancer metastasis is pivotal to
improving survival rates. Recently, extensive efforts have been
directed toward developing and evaluating aptamer-based
therapeutics against cancer metastasis. Table I documents examples of
aptamer-based therapeutics that have been examined against cancer
metastasis in preclinical and clinical settings and reported
between 2019 and 2022 (133,156-161). Unfortunately, no aptamer-based
therapeutic has been approved by the FDA to treat cancer metastasis
thus far limiting the use of aptamer-based therapeutics in cancer
metastasis treatment. It has been mentioned earlier in this review
that some strategies had been introduced to overcome the challenges
concerning aptamer pharmacokinetics such as enzymatic degradation,
rapid excretion by the kidney, and poor delivery to intracellular
targets. Nevertheless, other challenges still exist, and overcoming
these challenges is crucial before aptamer-based technology can be
approved for use against cancer metastasis. For example,
immunological reactions resulting from aptamer administration have
been observed. A randomized clinical trial evaluating the effect of
REG1 anticoagulation system, which is an RNA aptamer-based
inhibitor of the coagulation factor IXa, was terminated due to a
severe allergic reaction (162).
Research has demonstrated that allergic reactions observed with
aptamer-based therapeutics result from factors such as the
CpG-containing sequence and PEGylation (163-165). Understanding the underlying
mechanisms by which the immunological reactions are triggered as
well as finding the design elements contributing to immunogenicity
is important for the successful development of safe aptamers.
Furthermore, it is extremely important to identify unwanted
immunological reactions when using aptamers that target and
modulate immune cells involved in cancer metastasis. In addition,
information concerning the long-term consequences of targeting
tumor-associated immune cells, and whether the impact can be
extended to affect other cells in the immune system network are
lacking. In addition, insufficient toxicity data and lack of
appropriate regulatory guidelines for preclinical toxicity
assessment studies are challenges that could restrain aptamers from
being approved for treatment against cancer metastasis. Toxicity
observed with other oligonucleotide-based therapeutics such as
antisense oligonucleotides have raised a red flag about possible
toxicity that could not be detected with current study designs when
assessing the toxicity of oligonucleotide aptamers (166,167). Pegaptanib is the only
FDA-approved aptamer-based therapeutic for the treatment of
neovascular age-related macular degeneration. However,
postmarketing surveillance of the adverse drug reactions of
pegaptanib suggests a critical need for a long-term assessment for
possible severe adverse drug reactions and toxicity (168). Moreover, a survey conducted by
the Oligonucleotide Working Group of the European Federation of
Pharmaceutical Industries and Associations (EFPIA) demonstrated a
discrepancy in preclinical safety assessment studies performed for
oligonucleotide-based therapeutics (169). Thus, establishing optimal
toxicity assessment guidelines is critical for fully characterizing
aptamer toxicity. Another limitation is the lengthy and complex
production process which is an obstacle for large-scale production
for clinical use. The SELEX process is carried out in multiple
rounds in which each round consists of multiple steps: incubation
of the target with a random library, removing the unbound sequences
from the bound sequence, elution of the desirable bound sequence,
and amplification of the desirable bound sequence (11). Moreover, a successful selection of
an aptamer necessitates optimizing the experimental conditions such
as temperature, pH, ionic strength, and ratio of the target to the
random library which creates a complex process. Therefore,
identification of strategies to improve the aptamer selection
process will help facilitate the large-scale production of
aptamer-based therapeutics.
Targeting key mediators of cancer metastasis using
aptamer-based therapeutics is a promising strategy against
metastasis; yet, researchers should solve concerns with the current
research. First, exploring new targets and assessing the efficacy
of aptamer-based therapy in relevant in vivo models are
still limited. Enormous molecular targets and factors that regulate
cancer metastasis have not been explored as potential targets for
aptamer-based antimetastatic therapy. For example, miRNAs are RNAs
with short, highly conserved, non-coding sequences. By binding to
the 3′untranslated region of target mRNAs, miRNAs control gene
expression at the posttranscriptional level. Recent findings
demonstrate that miRNAs control metastasis by regulating
metastasis-related genes in cancer stem cells and during the
processes of EMT and metastatic colonization (170,171). Using aptamers to bind oncogenic
precursor microRNAs to inhibit functional miRNA formation or
deliver tumor suppressor miRNAs in the form of aptamer-miRNA
conjugate may show antitumor activity (172-175). Furthermore, circular RNAs
(circRNAs), which range in length from a few hundred to thousands
of nucleotides, is a type of RNA that has been recently
re-recognized (176). Contrary
to linear RNAs that have 5′caps and 3′tails, circRNAs are
single-stranded with covalently closed circular transcripts
(177). They were earlier known
to be the products of mis-splicing or by-products of pre-mRNA
processing with poor abundance, but now they have been recognized
as a class of non-coding RNAs following the use of high-throughput
RNA sequencing (RNA-seq) technologies. circRNAs are observed to be
involved in the pathogenesis of cancer tumorigenesis, metastasis,
and therapy resistance (178,179). These potential strategies have
been examined in vitro or clinically irrelevant in
vivo models (142,173,175). Thus, assessment of the efficacy
of developed aptamer technology against newly identified targets in
appropriate animal models of cancer metastasis is crucial to
develop effective aptamer-based therapeutics.
In conclusion, this review summarizes recent
advances in the development of aptamer-based antimetastatic
therapeutics. Studies discussed here demonstrate that targeted
aptamers possess a promising future in fighting cancer metastasis.
Despite the potential findings in the field of molecular and
cellular oncology, there are still opportunities to explore aptamer
technology against potential targets. This could result in the
development of aptamer-based antimetastatic therapy to target
cancer metastasis. In addition, more research is needed to examine
the existing antimetastatic aptamers in situations that mimic
cancer metastasis inside the body.
All information provided in this review is
documented by relevant references.
YA and HA contributed in the idea and writing the
manuscript. SA and AA participated in writing and editing the
manuscript. All authors revised and approved the final version.
Not applicable.
Not applicable.
Author names and ORCID nos. are as follows: Yahya
Alhamhoom: 0000-0002-8368-9047; Homood M. As Sobeai:
0000-0003-3073-3072; Sary Alsanea: 0000-0002-5323-5781; Ali
Alhoshani: 0000-0002-3450-283X.
The authors declare no competing interests.
Not applicable.
The authors reported there is no funding associated with this
work.
|
1
|
Chiang CJ, Lo WC, Yang YW, You SL, Chen CJ
and Lai MS: Incidence and survival of adult cancer patients in
Taiwan, 2002-2012. J Formos Med Assoc. 115:1076–1088. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Liu CM, Peng CY, Liao YW, Lu MY, Tsai ML,
Yeh JC, Yu CH and Yu CC: Sulforaphane targets cancer stemness and
tumor initiating properties in oral squamous cell carcinomas via
miR-200c induction. J Formos Med Assoc. 116:41–48. 2017. View Article : Google Scholar
|
|
3
|
Tsai CE, Wu KL, Chiu YC, Chuah SK, Tai WC,
Hu ML and Liang CM: The incidence and clinical associated factors
of interval colorectal cancers in Southern Taiwan. J Formos Med
Assoc. 117:185–190. 2018. View Article : Google Scholar
|
|
4
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global Cancer Statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dillekas H, Rogers MS and Straume O: Are
90% of deaths from cancer caused by metastases? Cancer Med.
8:5574–5576. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fidler IJ: The pathogenesis of cancer
metastasis: The 'seed and soil' hypothesis revisited. Nat Rev
Cancer. 3:453–458. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Stewart CM and Tsui DWY: Circulating
cell-free DNA for non-invasive cancer management. Cancer Genet.
228-229:169–179. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hafeez U, Gan HK and Scott AM: Monoclonal
antibodies as immunomodulatory therapy against cancer and
autoimmune diseases. Curr Opin Pharmacol. 41:114–121. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Dominiak A, Chelstowska B, Olejarz W and
Nowicka G: Communication in the cancer microenvironment as a target
for therapeutic interventions. Cancers (Basel). 12:12322020.
View Article : Google Scholar
|
|
10
|
Tuerk C and Gold L: Systematic evolution
of ligands by exponential enrichment: RNA ligands to bacteriophage
T4 DNA polymerase. Science. 249:505–510. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ellington AD and Szostak JW: In vitro
selection of RNA molecules that bind specific ligands. Nature.
346:818–822. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhou J and Rossi J: Aptamers as targeted
therapeutics: Current potential and challenges. Nat Rev Drug
Discov. 16:181–202. 2017. View Article : Google Scholar
|
|
13
|
Toh SY, Citartan M, Gopinath SC and Tang
TH: Aptamers as a replacement for antibodies in enzyme-linked
immunosorbent assay. Biosens Bioelectron. 64:392–403. 2015.
View Article : Google Scholar
|
|
14
|
Mascini M, Palchetti I and Tombelli S:
Nucleic acid and peptide aptamers: Fundamentals and bioanalytical
aspects. Angew Chem Int Ed Engl. 51:1316–1332. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Colas P, Cohen B, Jessen T, Grishina I,
McCoy J and Brent R: Genetic selection of peptide aptamers that
recognize and inhibit cyclin-dependent kinase 2. Nature.
380:548–550. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Reverdatto S, Burz DS and Shekhtman A:
Peptide aptamers: Development and applications. Curr Top Med Chem.
15:1082–1101. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Han J, Gao L and Wang J and Wang J:
Application and development of aptamer in cancer: From clinical
diagnosis to cancer therapy. J Cancer. 11:6902–6915. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pan Q, Luo F, Liu M and Zhang XL:
Oligonucleotide aptamers: Promising and powerful diagnostic and
therapeutic tools for infectious diseases. J Infect. 77:83–98.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Herrmann A, Priceman SJ, Swiderski P,
Kujawski M, Xin H, Cherryholmes GA, Zhang W, Zhang C, Lahtz C,
Kowolik C, et al: CTLA4 aptamer delivers STAT3 siRNA to
tumor-associated and malignant T cells. J Clin Invest.
124:2977–2987. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ospina-Villa JD, Zamorano-Carrillo A,
Castanon-Sanchez CA, Ramirez-Moreno E and Marchat LA: Aptamers as a
promising approach for the control of parasitic diseases. Braz J
Infect Dis. 20:610–618. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Liu M, Zaman K and Fortenberry YM:
Overview of the therapeutic potential of aptamers targeting
coagulation factors. Int J Mol Sci. 22:38972021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ninichuk V, Clauss S, Kulkarni O, Schmid
H, Segerer S, Radomska E, Eulberg D, Buchner K, Selve N, Klussmann
S and Anders HJ: Late onset of Ccl2 blockade with the Spiegelmer
mNOX-E36-3′PEG prevents glomerulosclerosis and improves glomerular
filtration rate in db/db mice. Am J Pathol. 172:628–637. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ng EW, Shima DT, Calias P, Cunningham ET
Jr, Guyer DR and Adamis AP: Pegaptanib, a targeted anti-VEGF
aptamer for ocular vascular disease. Nat Rev Drug Discov.
5:123–132. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gauthier NC and Roca-Cusachs P:
Mechanosensing at integrin-mediated cell-matrix adhesions: From
molecular to integrated mechanisms. Curr Opin Cell Biol. 50:20–26.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bissell MJ and Hines WC: Why don't we get
more cancer? A proposed role of the microenvironment in restraining
cancer progression. Nat Med. 17:320–329. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Friedl P and Wolf K: Tumour-cell invasion
and migration: Diversity and escape mechanisms. Nat Rev Cancer.
3:362–374. 2003. View Article : Google Scholar
|
|
27
|
Zheng X, Yu C and Xu M: Linking tumor
microenvironment to plasticity of cancer stem cells: Mechanisms and
application in cancer therapy. Front Oncol. 11:6783332021.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Krakhmal NV, Zavyalova MV, Denisov EV,
Vtorushin SV and Perelmuter VM: Cancer invasion: Patterns and
mechanisms. Acta Naturae. 7:17–28. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial-mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Joyce JA and Pollard JW:
Microenvironmental regulation of metastasis. Nat Rev Cancer.
9:239–252. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Dirat B, Bochet L, Dabek M, Daviaud D,
Dauvillier S, Majed B, Wang YY, Meulle A, Salles B, Le Gonidec S,
et al: Cancer-associated adipocytes exhibit an activated phenotype
and contribute to breast cancer invasion. Cancer Res. 71:2455–2465.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gocheva V, Wang HW, Gadea BB, Shree T,
Hunter KE, Garfall AL, Berman T and Joyce JA: IL-4 induces
cathepsin protease activity in tumor-associated macrophages to
promote cancer growth and invasion. Genes Dev. 24:241–255. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gupta GP and Massague J: Cancer
metastasis: Building a framework. Cell. 127:679–695. 2006.
View Article : Google Scholar
|
|
34
|
Carmeliet P and Jain RK: Principles and
mechanisms of vessel normalization for cancer and other angiogenic
diseases. Nat Rev Drug Discov. 10:417–427. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gupta GP, Nguyen DX, Chiang AC, Bos PD,
Kim JY, Nadal C, Gomis RR, Manova-Todorova K and Massagué J:
Mediators of vascular remodelling co-opted for sequential steps in
lung metastasis. Nature. 446:765–770. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Guo W and Giancotti FG: Integrin
signalling during tumour progression. Nat Rev Mol Cell Biol.
5:816–826. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Douma S, Van Laar T, Zevenhoven J,
Meuwissen R, Van Garderen E and Peeper DS: Suppression of anoikis
and induction of metastasis by the neurotrophic receptor TrkB.
Nature. 430:1034–1039. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Brown DM and Ruoslahti E: Metadherin, a
cell surface protein in breast tumors that mediates lung
metastasis. Cancer Cell. 5:365–374. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Auguste P, Fallavollita L, Wang N, Burnier
J, Bikfalvi A and Brodt P: The host inflammatory response promotes
liver metastasis by increasing tumor cell arrest and extravasation.
Am J Pathol. 170:1781–1792. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Al-Mehdi AB, Tozawa K, Fisher AB, Shientag
L, Lee A and Muschel RJ: Intravascular origin of metastasis from
the proliferation of endothelium-attached tumor cells: A new model
for metastasis. Nat Med. 6:100–102. 2000. View Article : Google Scholar
|
|
41
|
Padua D, Zhang XH, Wang Q, Nadal C, Gerald
WL, Gomis RR and Massagué J: TGFbeta primes breast tumors for lung
metastasis seeding through angiopoietin-like 4. Cell. 133:66–77.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Psaila B and Lyden D: The metastatic
niche: Adapting the foreign soil. Nat Rev Cancer. 9:285–293. 2009.
View Article : Google Scholar
|
|
43
|
Erler JT, Bennewith KL, Cox TR, Lang G,
Bird D, Koong A, Le QT and Giaccia AJ: Hypoxia-induced lysyl
oxidase is a critical mediator of bone marrow cell recruitment to
form the premetastatic niche. Cancer Cell. 15:35–44. 2009.
View Article : Google Scholar
|
|
44
|
Chambers AF, Groom AC and MacDonald IC:
Dissemination and growth of cancer cells in metastatic sites. Nat
Rev Cancer. 2:563–572. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
45
|
Barkan D, El Touny LH, Michalowski AM,
Smith JA, Chu I, Davis AS, Webster JD, Hoover S, Simpson RM,
Gauldie J and Green JE: Metastatic growth from dormant cells
induced by a col-I-enriched fibrotic environment. Cancer Res.
70:5706–5716. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Barkan D, Kleinman H, Simmons JL, Asmussen
H, Kamaraju AK, Hoenorhoff MJ, Liu ZY, Costes SV, Cho EH, Lockett
S, et al: Inhibition of metastatic outgrowth from single dormant
tumor cells by targeting the cytoskeleton. Cancer Res.
68:6241–6250. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Shibue T and Weinberg RA: Integrin
beta1-focal adhesion kinase signaling directs the proliferation of
metastatic cancer cells disseminated in the lungs. Proc Natl Acad
Sci USA. 106:10290–10295. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Hiratsuka S, Duda DG, Huang Y, Goel S,
Sugiyama T, Nagasawa T, Fukumura D and Jain RK: C-X-C receptor type
4 promotes metastasis by activating p38 mitogen-activated protein
kinase in myeloid differentiation antigen (Gr-1)-positive cells.
Proc Natl Acad Sci USA. 108:302–307. 2011. View Article : Google Scholar :
|
|
49
|
McAllister SS, Gifford AM, Greiner AL,
Kelleher SP, Saelzler MP, Ince TA, Reinhardt F, Harris LN, Hylander
BL, Repasky EA and Weinberg RA: Systemic endocrine instigation of
indolent tumor growth requires osteopontin. Cell. 133:994–1005.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kang SY, Halvorsen OJ, Gravdal K,
Bhattacharya N, Lee JM, Liu NW, Johnston BT, Johnston AB, Haukaas
SA, Aamodt K, et al: Prosaposin inhibits tumor metastasis via
paracrine and endocrine stimulation of stromal p53 and Tsp-1. Proc
Natl Acad Sci USA. 106:12115–12120. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Shimono Y, Zabala M, Cho RW, Lobo N,
Dalerba P, Qian D, Diehn M, Liu H, Panula SP, Chiao E, et al:
Downregulation of miRNA-200c links breast cancer stem cells with
normal stem cells. Cell. 138:592–603. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Cai S, Yan J, Xiong H, Liu Y, Peng D and
Liu Z: Investigations on the interface of nucleic acid aptamers and
binding targets. Analyst. 143:5317–5338. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Ni S, Zhuo Z, Pan Y, Yu Y, Li F, Liu J,
Wang L, Wu X, Li D, Wan Y, et al: Recent progress in aptamer
discoveries and modifications for therapeutic applications. ACS
Appl Mater Interfaces. 13:9500–9519. 2021. View Article : Google Scholar
|
|
54
|
Zhang Y, Lai BS and Juhas M: Recent
advances in aptamer discovery and applications. Molecules.
24:9412019. View Article : Google Scholar :
|
|
55
|
Dong Y, Zhang T, Lin X, Feng J, Luo F, Gao
H, Wu Y, Deng R and He Q: Graphene/aptamer probes for small
molecule detection: from in vitro test to in situ imaging.
Mikrochim Acta. 187:1792020. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Cho SJ, Woo HM, Kim KS, Oh JW and Jeong
YJ: Novel system for detecting SARS coronavirus nucleocapsid
protein using an ssDNA aptamer. J Biosci Bioeng. 112:535–540. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Li HY, Jia WN, Li XY, Zhang L, Liu C and
Wu J: Advances in detection of infectious agents by aptamer-based
technologies. Emerg Microbes Infect. 9:1671–1681. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Hassan EM, Willmore WG and DeRosa MC:
Aptamers: Promising tools for the detection of circulating tumor
cells. Nucleic Acid Ther. 26:335–347. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Orava EW, Cicmil N and Gariepy J:
Delivering cargoes into cancer cells using DNA aptamers targeting
internalized surface portals. Biochim Biophys Acta. 1798:2190–2200.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Zhang GX, Liu YL, Yang M, Huang WS and Xu
JH: An aptamer-based, fluorescent and radionuclide dual-modality
probe. Biochimie. 171-172:55–62. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Hashemitabar S, Yazdian-Robati R, Hashemi
M, Ramezani M, Abnous K and Kalalinia F: ABCG2 aptamer selectively
delivers doxorubicin to drug-resistant breast cancer cells. J
Biosci. 44:392019. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Kruspe S and Giangrande PH: Aptamer-siRNA
chimeras: Discovery, progress and future prospects. Biomedicines.
5:452017. View Article : Google Scholar
|
|
63
|
Jiang L, Wang H and Chen S: Aptamer
(AS1411)-conjugated liposome for enhanced therapeutic efficacy of
miRNA-29b in ovarian cancer. J Nanosci Nanotechnol. 20:2025–2031.
2020. View Article : Google Scholar
|
|
64
|
Meng HM, Liu H, Kuai H, Peng R, Mo L and
Zhang XB: Aptamer-integrated DNA nanostructures for biosensing,
bioimaging and cancer therapy. Chem Soc Rev. 45:2583–2602. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Xiang D, Shigdar S, Qiao G, Wang T,
Kouzani AZ, Zhou SF, Kong L, Li Y, Pu C and Duan W: Nucleic acid
aptamer-guided cancer therapeutics and diagnostics: The next
generation of cancer medicine. Theranostics. 5:23–42. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Wu C, Han D, Chen T, Peng L, Zhu G, You M,
Qiu L, Sefah K, Zhang X and Tan W: Building a multifunctional
aptamer-based DNA nanoassembly for targeted cancer therapy. J Am
Chem Soc. 135:18644–18650. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Kuwahara M and Sugimoto N: Molecular
evolution of functional nucleic acids with chemical modifications.
Molecules. 15:5423–5444. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Mayer G: The chemical biology of aptamers.
Angew Chem Int Ed Engl. 48:2672–2689. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Turner JJ, Hoos JS, Vonhoff S and
Klussmann S: Methods for L-ribooligonucleotide sequence
determination using LCMS. Nucleic Acids Res. 39:e1472011.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Lee CH, Lee SH, Kim JH, Noh YH, Noh GJ and
Lee SW: Pharmacokinetics of a Cholesterol-conjugated aptamer
against the Hepatitis C Virus (HCV) NS5B protein. Mol Ther Nucleic
Acids. 4:e2542015. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Dougan H, Lyster DM, Vo CV, Stafford A,
Weitz JI and Hobbs JB: Extending the lifetime of anticoagulant
oligodeoxynucleotide aptamers in blood. Nucl Med Biol. 27:289–297.
2000. View Article : Google Scholar
|
|
72
|
Healy JM, Lewis SD, Kurz M, Boomer RM,
Thompson KM, Wilson C and McCauley TG: Pharmacokinetics and
biodistribution of novel aptamer compositions. Pharm Res.
21:2234–2246. 2004. View Article : Google Scholar
|
|
73
|
Marshall ML and Wagstaff KM: Internalized
functional DNA aptamers as alternative cancer therapies. Front
Pharmacol. 11:11152020. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Thiel WH, Thiel KW, Flenker KS, Bair T,
Dupuy AJ, McNamara JO II, Miller FJ and Giangrande PH:
Cell-internalization SELEX: Method for identifying
cell-internalizing RNA aptamers for delivering siRNAs to target
cells. Methods Mol Biol. 1218:187–199. 2015. View Article : Google Scholar :
|
|
75
|
Alamudi SH, Kimoto M and Hirao I: Uptake
mechanisms of cell-internalizing nucleic acid aptamers for
applications as pharmacological agents. RSC Med Chem. 12:1640–1649.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Nagel-Wolfrum K, Buerger C, Wittig I, Butz
K, Hoppe-Seyler F and Groner B: The interaction of specific peptide
aptamers with the DNA binding domain and the dimerization domain of
the transcription factor Stat3 inhibits transactivation and induces
apoptosis in tumor cells. Mol Cancer Res. 2:170–182.
2004.PubMed/NCBI
|
|
77
|
Taylor RE and Zahid M: Cell penetrating
peptides, novel vectors for gene therapy. Pharmaceutics.
12:2252020. View Article : Google Scholar :
|
|
78
|
Moutal A, Francois-Moutal L, Brittain JM,
Khanna M and Khanna R: Differential neuroprotective potential of
CRMP2 peptide aptamers conjugated to cationic, hydrophobic, and
amphipathic cell penetrating peptides. Front Cell Neurosci.
8:4712014.
|
|
79
|
Famulok M, Blind M and Mayer G: Intramers
as promising new tools in functional proteomics. Chem Biol.
8:931–939. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Kunz C, Borghouts C, Buerger C and Groner
B: Peptide aptamers with binding specificity for the intracellular
domain of the ErbB2 receptor interfere with AKT signaling and
sensitize breast cancer cells to Taxol. Mol Cancer Res. 4:983–998.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Shiozawa Y, Nie B, Pienta KJ, Morgan TM
and Taichman RS: Cancer stem cells and their role in metastasis.
Pharmacol Ther. 138:285–293. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Velasco-Velazquez MA, Popov VM, Lisanti MP
and Pestell RG: The role of breast cancer stem cells in metastasis
and therapeutic implications. Am J Pathol. 179:2–11. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Eyler CE and Rich JN: Survival of the
fittest: Cancer stem cells in therapeutic resistance and
angiogenesis. J Clin Oncol. 26:2839–2845. 2008. View Article : Google Scholar
|
|
84
|
Chiou SH, Wang ML, Chou YT, Chen CJ, Hong
CF, Hsieh WJ, Chang HT, Chen YS, Lin TW, Hsu HS and Wu CW:
Coexpression of Oct4 and Nanog enhances malignancy in lung
adenocarcinoma by inducing cancer stem cell-like properties and
epithelial-mesenchymal transdifferentiation. Cancer Res.
70:10433–10444. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Chiou SH, Yu CC, Huang CY, Lin SC, Liu CJ,
Tsai TH, Chou SH, Chien CS, Ku HH and Lo JF: Positive correlations
of Oct-4 and Nanog in oral cancer stem-like cells and high-grade
oral squamous cell carcinoma. Clin Cancer Res. 14:4085–4095. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Leis O, Eguiara A, Lopez-Arribillaga E,
Alberdi MJ, Hernandez-Garcia S, Elorriaga K, Pandiella A, Rezola R
and Martin AG: Sox2 expression in breast tumours and activation in
breast cancer stem cells. Oncogene. 31:1354–1365. 2012. View Article : Google Scholar
|
|
87
|
Lonardo E, Hermann PC, Mueller MT, Huber
S, Balic A, Miranda-Lorenzo I, Zagorac S, Alcala S,
Rodriguez-Arabaolaza I, Ramirez JC, et al: Nodal/Activin signaling
drives self-renewal and tumorigenicity of pancreatic cancer stem
cells and provides a target for combined drug therapy. Cell Stem
Cell. 9:433–446. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Paranjape AN, Balaji SA, Mandal T, Krushik
EV, Nagaraj P, Mukherjee G and Rangarajan A: Bmi1 regulates
self-renewal and epithelial to mesenchymal transition in breast
cancer cells through Nanog. BMC Cancer. 14:7852014. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Villodre ES, Kipper FC, Pereira MB and
Lenz G: Roles of OCT4 in tumorigenesis, cancer therapy resistance
and prognosis. Cancer Treat Rev. 51:1–9. 2016. View Article : Google Scholar
|
|
90
|
Hochedlinger K, Yamada Y, Beard C and
Jaenisch R: Ectopic expression of Oct-4 blocks progenitor-cell
differentiation and causes dysplasia in epithelial tissues. Cell.
121:465–477. 2005. View Article : Google Scholar
|
|
91
|
Wei X, He J, Wang J, Yang X and Ma B:
Bmi-1 is essential for the oncogenic potential in CD133(+) human
laryngeal cancer cells. Tumour Biol. 36:8931–8942. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Chao HM, Huang HX, Chang PH, Tseng KC,
Miyajima A and Chern E: Y-box binding protein-1 promotes
hepatocellular carcinoma-initiating cell progression and
tumorigenesis via Wnt/beta-catenin pathway. Oncotarget.
8:2604–2616. 2017. View Article : Google Scholar
|
|
93
|
Chen YR, Sekine K, Nakamura K, Yanai H,
Tanaka M and Miyajima A: Y-box binding protein-1 down-regulates
expression of carbamoyl phosphate synthetase-I by suppressing CCAAT
enhancer-binding protein-alpha function in mice. Gastroenterology.
137:330–340. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Guerra C, Schuhmacher AJ, Cañamero M,
Grippo PJ, Verdaguer L, Pérez-Gallego L, Dubus P, Sandgren EP and
Barbacid M: Chronic pancreatitis is essential for induction of
pancreatic ductal adenocarcinoma by K-Ras oncogenes in adult mice.
Cancer Cell. 11:291–302. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Macdonald J, Henri J, Goodman L, Xiang D,
Duan W and Shigdar S: Development of a bifunctional aptamer
targeting the transferrin receptor and epithelial cell adhesion
molecule (EpCAM) for the treatment of brain cancer metastases.
Chronic pancreatitis is essential for induction of pancreatic
ductal adenocarcinoma by K-Ras oncogenes in adult mice. ACS Chem
Neurosci. 8:777–784. 2017. View Article : Google Scholar
|
|
96
|
Yin H, Xiong G, Guo S, Xu C, Xu R, Guo P
and Shu D: Delivery of Anti-miRNA for Triple-negative breast cancer
therapy Using RNA nanoparticles targeting stem cell marker CD133.
Mol Ther. 27:1252–1261. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Zeng YB, Yu ZC, He YN, Zhang T, Du LB,
Dong YM, Chen HW, Zhang YY and Wang WQ: Salinomycin-loaded
lipid-polymer nanoparticles with anti-CD20 aptamers selectively
suppress human CD20+ melanoma stem cells. Acta Pharmacol Sin.
39:261–274. 2018. View Article : Google Scholar :
|
|
98
|
Zhang Y, Leonard M, Shu Y, Yang Y, Shu D,
Guo P and Zhang X: Overcoming tamoxifen resistance of human breast
cancer by targeted gene silencing using multifunctional pRNA
nanoparticles. ACS Nano. 11:335–346. 2017. View Article : Google Scholar
|
|
99
|
Zhou G, Da Won Bae S, Nguyen R, Huo X, Han
S, Zhang Z, Hebbard L, Duan W, Eslam M, Liddle C, et al: An
aptamer-based drug delivery agent (CD133-apt-Dox) selectively and
effectively kills liver cancer stem-like cells. Cancer Lett.
501:124–132. 2021. View Article : Google Scholar
|
|
100
|
Kim DM, Kim M, Park HB, Kim KS and Kim DE:
Anti-MUC1/CD44 dual-aptamer-conjugated liposomes for cotargeting
breast cancer cells and cancer stem cells. ACS Applied Bio
Materials. 2:4622–4633. 2019. View Article : Google Scholar
|
|
101
|
Talmadge JE and Fidler IJ: AACR centennial
series: The biology of cancer metastasis: Historical perspective.
Cancer Res. 70:5649–5669. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Sleeman JP, Nazarenko I and Thiele W: Do
all roads lead to Rome? Routes to metastasis development. Int J
Cancer. 128:2511–2526. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Liljefors M, Nilsson B, Fagerberg J,
Ragnhammar P, Mellstedt H and Frodin JE: Clinical effects of a
chimeric anti-EpCAM monoclonal antibody in combination with
granulocyte-macrophage colony-stimulating factor in patients with
metastatic colorectal carcinoma. Int J Oncol. 26:1581–1589.
2005.PubMed/NCBI
|
|
104
|
Scarberry KE, Mezencev R and McDonald JF:
Targeted removal of migratory tumor cells by functionalized
magnetic nanoparticles impedes metastasis and tumor progression.
Nanomedicine (Lond). 6:69–78. 2011. View Article : Google Scholar
|
|
105
|
Orava EW, Abdul-Wahid A, Huang EH, Mallick
AI and Gariepy J: Blocking the attachment of cancer cells in vivo
with DNA aptamers displaying anti-adhesive properties against the
carcinoembryonic antigen. Mol Oncol. 7:799–811. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Abdul-Wahid A, Huang EH, Cydzik M,
Bolewska-Pedyczak E and Gariepy J: The carcinoembryonic antigen
IgV-like N domain plays a critical role in the implantation of
metastatic tumor cells. Mol Oncol. 8:337–350. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Wang XK, Peng Y, Tao HR, Zhou FF, Zhang C,
Su F, Wang SP, Liu Q, Xu LH, Pan XK, et al: Inhibition of adhesion
and metastasis of HepG2 hepatocellular carcinoma cells in vitro by
DNA aptamer against sialyl Lewis X. J Huazhong Univ Sci Technolog
Med Sci. 37:343–347. 2017. View Article : Google Scholar
|
|
108
|
Brodt P, Fallavollita L, Bresalier RS,
Meterissian S, Norton CR and Wolitzky BA: Liver endothelial
E-selectin mediates carcinoma cell adhesion and promotes liver
metastasis. Int J Cancer. 71:612–619. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Kowal J, Arras G, Colombo M, Jouve M,
Morath JP, Primdal-Bengtson B, Dingli F, Loew D, Tkach M and Théry
C: Proteomic comparison defines novel markers to characterize
heterogeneous populations of extracellular vesicle subtypes. Proc
Natl Acad Sci USA. 113:E968–E977. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Minciacchi VR, Freeman MR and Di Vizio D:
Extracellular vesicles in cancer: Exosomes, microvesicles and the
emerging role of large oncosomes. Semin Cell Dev Biol. 40:41–51.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Raposo G and Stoorvogel W: Extracellular
vesicles: Exosomes, microvesicles, and friends. J Cell Biol.
200:373–383. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Yang F, Ning Z, Ma L, Liu W, Shao C, Shu Y
and Shen H: Exosomal miRNAs and miRNA dysregulation in
cancer-associated fibroblasts. Mol Cancer. 16:1482017. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Colombo M, Raposo G and Thery C:
Biogenesis, secretion, and intercellular interactions of exosomes
and other extracellular vesicles. Annu Rev Cell Dev Biol.
30:255–289. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Gould SJ and Raposo G: As we wait: Coping
with an imperfect nomenclature for extracellular vesicles. J
Extracell Vesicles. 2:203892013. View Article : Google Scholar
|
|
115
|
Li W, Ma H, Zhang J, Zhu L, Wang C and
Yang Y: Unraveling the roles of CD44/CD24 and ALDH1 as cancer stem
cell markers in tumorigenesis and metastasis. Sci Rep. 7:138562017.
View Article : Google Scholar : PubMed/NCBI
|
|
116
|
van Niel G, D'Angelo G and Raposo G:
Shedding light on the cell biology of extracellular vesicles. Nat
Rev Mol Cell Biol. 19:213–228. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Hannafon BN and Ding WQ: Intercellular
communication by exosome-derived microRNAs in cancer. Int J Mol
Sci. 14:14240–14269. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Milane L, Singh A, Mattheolabakis G,
Suresh M and Amiji MM: Exosome mediated communication within the
tumor microenvironment. J Control Release. 219:278–294. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Valadi H, Ekstrom K, Bossios A, Sjostrand
M, Lee JJ and Lotvall JO: Exosome-mediated transfer of mRNAs and
microRNAs is a novel mechanism of genetic exchange between cells.
Nat Cell Biol. 9:654–659. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Gezer U, Ozgur E, Cetinkaya M, Isin M and
Dalay N: Long non-coding RNAs with low expression levels in cells
are enriched in secreted exosomes. Cell Biol Int. 38:1076–1079.
2014.PubMed/NCBI
|
|
121
|
Li Z, Huang C, Bao C, Chen L, Lin M, Wang
X, Zhong G, Yu B, Hu W, Dai L, et al: Exon-intron circular RNAs
regulate transcription in the nucleus. Nat Struct Mol Biol.
22:256–264. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Sun T, Kalionis B, Lv G, Xia S and Gao W:
Role of exosomal noncoding RNAs in lung carcinogenesis. Biomed Res
Int. 2015:1258072015. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Hood JL, San RS and Wickline SA: Exosomes
released by melanoma cells prepare sentinel lymph nodes for tumor
metastasis. Cancer Res. 71:3792–3801. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Lobb RJ, Lima LG and Moller A: Exosomes:
Key mediators of metastasis and pre-metastatic niche formation.
Semin Cell Dev Biol. 67:3–10. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Luga V, Zhang L, Viloria-Petit AM,
Ogunjimi AA, Inanlou MR, Chiu E, Buchanan M, Hosein AN, Basik M and
Wrana JL: Exosomes mediate stromal mobilization of autocrine
Wnt-PCP signaling in breast cancer cell migration. Cell.
151:1542–1556. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Peinado H, Alečković M, Lavotshkin S,
Matei I, Costa-Silva B, Moreno-Bueno G, Hergueta-Redondo M,
Williams C, García-Santos G, Ghajar C, et al: Melanoma exosomes
educate bone marrow progenitor cells toward a pro-metastatic
phenotype through MET. Nat Med. 18:883–891. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Tominaga N, Kosaka N, Ono M, Katsuda T,
Yoshioka Y, Tamura K, Lötvall J, Nakagama H and Ochiya T: Brain
metastatic cancer cells release microRNA-181c-containing
extracellular vesicles capable of destructing blood-brain barrier.
Nat Commun. 6:67162015. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Fong MY, Zhou W, Liu L, Alontaga AY,
Chandra M, Ashby J, Chow A, O'Connor ST, Li S, Chin AR, et al:
Breast-cancer-secreted miR-122 reprograms glucose metabolism in
premetastatic niche to promote metastasis. Nat Cell Biol.
17:183–194. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Szajnik M, Czystowska M, Szczepanski MJ,
Mandapathil M and Whiteside TL: Tumor-derived microvesicles induce,
expand and up-regulate biological activities of human regulatory T
cells (Treg). PLoS One. 5:e114692010. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
van der Vos KE, Abels ER, Zhang X, Lai C,
Carrizosa E, Oakley D, Prabhakar S, Mardini O, Crommentuijn MH,
Skog J, et al: Directly visualized glioblastoma-derived
extracellular vesicles transfer RNA to microglia/macrophages in the
brain. Neuro Oncol. 18:58–69. 2016. View Article : Google Scholar
|
|
131
|
Zhang L, Zhang S, Yao J, Lowery FJ, Zhang
Q, Huang WC, Li P, Li M, Wang X, Zhang C, et al:
Microenvironment-induced PTEN loss by exosomal microRNA primes
brain metastasis outgrowth. Nature. 527:100–104. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Esposito CL, Quintavalle C, Ingenito F,
Rotoli D, Roscigno G, Nuzzo S, Thomas R, Catuogno S, de Franciscis
V and Condorelli G: Identification of a novel RNA aptamer that
selectively targets breast cancer exosomes. Mol Ther Nucleic Acids.
23:982–994. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Xie X, Nie H, Zhou Y, Lian S, Mei H, Lu Y,
Dong H, Li F, Li T, Li B, et al: Eliminating blood oncogenic
exosomes into the small intestine with aptamer-functionalized
nanoparticles. Nat Commun. 10:54762019. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Dunn GP, Old LJ and Schreiber RD: The
immunobiology of cancer immunosurveillance and immunoediting.
Immunity. 21:137–148. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Leek RD, Lewis CE, Whitehouse R, Greenall
M, Clarke J and Harris AL: Association of macrophage infiltration
with angiogenesis and prognosis in invasive breast carcinoma.
Cancer Res. 56:4625–4629. 1996.PubMed/NCBI
|
|
136
|
DeNardo DG, Barreto JB, Andreu P, Vasquez
L, Tawfik D, Kolhatkar N and Coussens LM: CD4(+) T cells regulate
pulmonary metastasis of mammary carcinomas by enhancing protumor
properties of macrophages. Cancer Cell. 16:91–102. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Moaaz M, Lotfy H, Elsherbini B, Motawea MA
and Fadali G: TGF-beta Enhances the Anti-inflammatory effect of
tumor-infiltrating CD33+11b+HLA-DR myeloid-derived suppressor cells
in gastric cancer: A possible relation to MicroRNA-494. Asian Pac J
Cancer Prev. 21:3393–3403. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Shitara K and Nishikawa H: Regulatory T
cells: A potential target in cancer immunotherapy. Ann N Y Acad
Sci. 1417:104–115. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Kumar V, Patel S, Tcyganov E and
Gabrilovich DI: The Nature of Myeloid-derived suppressor cells in
the tumor microenvironment. Trends Immunol. 37:208–220. 2016.
View Article : Google Scholar :
|
|
140
|
Sarvaiya PJ, Guo D, Ulasov I, Gabikian P
and Lesniak MS: Chemokines in tumor progression and metastasis.
Oncotarget. 4:2171–2185. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Briukhovetska D, Dorr J, Endres S, Libby
P, Dinarello CA and Kobold S: Interleukins in cancer: From biology
to therapy. Nat Rev Cancer. 21:481–499. 2021. View Article : Google Scholar :
|
|
142
|
Manrique-Rincón AJ, Ruas LP, Fogagnolo CT,
Brenneman RJ, Berezhnoy A, Castelucci B, Consonni SR, Gilboa E and
Bajgelman MC: Aptamer-mediated transcriptional gene silencing of
Fox 3 inhibits regulatory T cells and potentiates antitumor
response. Mol Ther Nucleic Acids. 25:143–151. 2021. View Article : Google Scholar
|
|
143
|
Borsig L, Wolf MJ, Roblek M, Lorentzen A
and Heikenwalder M: Inflammatory chemokines and metastasis-tracing
the accessory. Oncogene. 33:3217–3224. 2014. View Article : Google Scholar
|
|
144
|
Fridlender ZG, Sun J, Kim S, Kapoor V,
Cheng G, Ling L, Worthen GS and Albelda SM: Polarization of
tumor-associated neutrophil phenotype by TGF-beta: 'N1' versus 'N2'
TAN. Cancer Cell. 16:183–194. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Tan W, Zhang W, Strasner A, Grivennikov S,
Cheng JQ, Hoffman RM and Karin M: Tumour-infiltrating regulatory T
cells stimulate mammary cancer metastasis through RANKL-RANK
signalling. Nature. 470:548–553. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Monteiro AC, Leal AC, Goncalves-Silva T,
Mercadante AC, Kestelman F, Chaves SB, Azevedo RB, Monteiro JP and
Bonomo A: T cells induce pre-metastatic osteolytic disease and help
bone metastases establishment in a mouse model of metastatic breast
cancer. PLoS One. 8:e681712013. View Article : Google Scholar
|
|
147
|
Blomberg OS, Spagnuolo L and de Visser KE:
Immune regulation of metastasis: Mechanistic insights and
therapeutic opportunities. Dis Model Mech. 11:dmm0362362018.
View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Kitamura T, Qian BZ and Pollard JW: Immune
cell promotion of metastasis. Nat Rev Immunol. 15:73–86. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Roth F, De La Fuente AC, Vella JL, Zoso A,
Inverardi L and Serafini P: Aptamer-mediated blockade of IL4Rα
triggers apoptosis of MDSCs and limits tumor progression. Cancer
Res. 72:1373–1383. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Liu H, Mai J, Shen J, Wolfram J, Li Z,
Zhang G, Xu R, Li Y, Mu C, Zu Y, et al: A Novel DNA Aptamer for
dual targeting of polymorphonuclear myeloid-derived suppressor
cells and tumor cells. Theranostics. 8:31–44. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Eberting CL, Shrayer DP, Butmarc J and
Falanga V: Histologic progression of B16 F10 metastatic melanoma in
C57BL/6 mice over a six week time period: Distant metastases before
local growth. J Dermatol. 31:299–304. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Meyer C, Eydeler K, Magbanua E, Zivkovic
T, Piganeau N, Lorenzen I, Grötzinger J, Mayer G, Rose-John S and
Hahn U: Interleukin-6 receptor specific RNA aptamers for cargo
delivery into target cells. RNA Biol. 9:67–80. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Gupta S, Hirota M, Waugh SM, Murakami I,
Suzuki T, Muraguchi M, Shibamori M, Ishikawa Y, Jarvis TC, Carter
JD, et al: Chemically modified DNA aptamers bind interleukin-6 with
high affinity and inhibit signaling by blocking its interaction
with interleukin-6 receptor. J Biol Chem. 289:8706–8719. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Berezhnoy A, Stewart CA, McNamara JO II,
Thiel W, Giangrande P, Trinchieri G and Gilboa E: Isolation and
optimization of murine IL-10 receptor blocking oligonucleotide
aptamers using high-throughput sequencing. Mol Ther. 20:1242–1250.
2012. View Article : Google Scholar :
|
|
155
|
Levay A, Brenneman R, Hoinka J, Sant D,
Cardone M, Trinchieri G, Przytycka TM and Berezhnoy A: Identifying
high-affinity aptamer ligands with defined cross-reactivity using
high-throughput guided systematic evolution of ligands by
exponential enrichment. Nucleic Acids Res. 43:e822015. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Yoon S, Huang KW, Andrikakou P,
Vasconcelos D, Swiderski P, Reebye V, Sodergren M, Habib N and
Rossi JJ: Targeted Delivery of C/EBPalpha-saRNA by RNA aptamers
shows anti-tumor effects in a mouse model of advanced PDAC. Mol
Ther Nucleic Acids. 18:142–154. 2019. View Article : Google Scholar :
|
|
157
|
Camorani S, Passariello M, Agnello L,
Pedone E, Pirone L, Chesta CA, Palacios RE, Fedele M and Cerchia L:
Aptamer targeted therapy potentiates immune checkpoint blockade in
triple-negative breast cancer. J Exp Clin Cancer Res. 39:1802020.
View Article : Google Scholar :
|
|
158
|
Quirico L, Orso F, Esposito CL, Bertone S,
Coppo R, Conti L, Catuogno S, Cavallo F, de Franciscis V and
Taverna D: Axl-148b chimeric aptamers inhibit breast cancer and
melanoma progression. Int J Biol Sci. 16:1238–1251. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Shelley G, Dai J, Keller JM and Keller ET:
Pheno-SELEX: Engineering Anti-metastatic aptamers through targeting
the invasive phenotype using systemic evolution of ligands by
exponential enrichment. Bioengineering (Basel). 8:2122021.
View Article : Google Scholar
|
|
160
|
Li T, Li Y, Rehmani H, Guo J, Padia R,
Calbay O, Ding Z, Jiang Y, Jin L and Huang S: Attenuated
miR-203b-3p is critical for ovarian cancer progression and
aptamer/miR-203b-3p chimera can be explored as a therapeutic. Adv
Cancer Biol-Metastasis. 4:1000312022. View Article : Google Scholar
|
|
161
|
Liu K, Xie F, Zhao T, Zhang R, Gao A, Chen
Y, Li H, Zhang S, Xiao Z, Li J, et al: Targeting SOX2 protein with
peptide aptamers for therapeutic gains against esophageal squamous
cell carcinoma. Mol Ther. 28:901–913. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Lincoff AM, Mehran R, Povsic TJ,
Zelenkofske SL, Huang Z, Armstrong PW, Steg PG, Bode C, Cohen MG,
Buller C, et al: Effect of the REG1 anticoagulation system versus
bivalirudin on outcomes after percutaneous coronary intervention
(REGULATE-PCI): A randomised clinical trial. Lancet. 387:349–356.
2016. View Article : Google Scholar
|
|
163
|
Bruno JG: Potential inherent stimulation
of the innate immune system by nucleic acid aptamers and possible
corrective approaches. Pharmaceuticals (Basel). 11:622018.
View Article : Google Scholar
|
|
164
|
Verhoef JJ, Carpenter JF, Anchordoquy TJ
and Schellekens H: Potential induction of anti-PEG antibodies and
complement activation toward PEGylated therapeutics. Drug Discov
Today. 19:1945–1952. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Ganson NJ, Povsic TJ, Sullenger BA,
Alexander JH, Zelenkofske SL, Sailstad JM, Rusconi CP and
Hershfield MS: Pre-existing anti-polyethylene glycol antibody
linked to first-exposure allergic reactions to pegnivacogin, a
PEGylated RNA aptamer. J Allergy Clin Immunol. 137:1610–1613.e7.
2016. View Article : Google Scholar
|
|
166
|
Shen W, De Hoyos CL, Sun H, Vickers TA,
Liang XH and Crooke ST: Acute hepatotoxicity of 2′fluoro-modified
5-10-5 gapmer phosphorothioate oligonucleotides in mice correlates
with intracellular protein binding and the loss of DBHS proteins.
Nucleic Acids Res. 46:2204–2217. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Burdick AD, Sciabola S, Mantena SR,
Hollingshead BD, Stanton R, Warneke JA, Zeng M, Martsen E, Medvedev
A, Makarov SS, et al: Sequence motifs associated with
hepatotoxicity of locked nucleic acid-modified antisense
oligonucleotides. Nucleic Acids Res. 42:4882–4891. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Penedones A, Mendes D, Alves C and Batel
Marques F: Safety monitoring of ophthalmic biologics: A systematic
review of pre- and postmarketing safety data. J Ocul Pharmacol
Ther. 30:729–751. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Tessier Y, Achanzar W, Mihalcik L, Amuzie
C, Andersson P, Parry JD, Moggs J and Whiteley LO: Outcomes of the
European federation of pharmaceutical industries and associations
oligonucleotide Working Group Survey on Nonclinical Practices and
Regulatory Expectations for Therapeutic Oligonucleotide Safety
Assessment. Nucleic Acid Ther. 31:7–20. 2021. View Article : Google Scholar
|
|
170
|
Kim J, Yao F, Xiao Z, Sun Y and Ma L:
MicroRNAs and metastasis: Small RNAs play big roles. Cancer
Metastasis Rev. 37:5–15. 2018. View Article : Google Scholar :
|
|
171
|
Chan SH and Wang LH: Regulation of cancer
metastasis by microRNAs. J Biomed Sci. 22:92015. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Sczepanski JT and Joyce GF: Specific
inhibition of MicroRNA processing using L-RNA aptamers. J Am Chem
Soc. 137:16032–16037. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Daei P, Ramezanpour M, Khanaki K, Tabarzad
M, Nikokar I, Hedayati Ch M and Elmi A: Aptamer-based targeted
delivery of miRNA let-7d to gastric cancer cells as a novel
anti-tumor therapeutic agent. Iran J Pharm Res. 17:1537–1549.
2018.PubMed/NCBI
|
|
174
|
Esposito CL, Catuogno S and de Franciscis
V: Aptamer-MiRNA conjugates for cancer cell-targeted delivery.
Methods Mol Biol. 1364:197–208. 2016. View Article : Google Scholar
|
|
175
|
Wang H, Zhao X, Guo C, Ren D, Zhao Y, Xiao
W and Jiao W: Aptamer-dendrimer bioconjugates for targeted delivery
of miR-34a expressing plasmid and antitumor effects in non-small
cell lung cancer cells. PLoS One. 10:e01391362015. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Guo JU, Agarwal V, Guo H and Bartel DP:
Expanded identification and characterization of mammalian circular
RNAs. Genome Biol. 15:4092014. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Chen LL and Yang L: Regulation of circRNA
biogenesis. RNA Biol. 12:381–388. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Du WW, Yang W, Liu E, Yang Z, Dhaliwal P
and Yang BB: Foxo3 circular RNA retards cell cycle progression via
forming ternary complexes with p21 and CDK2. Nucleic Acids Res.
44:2846–2858. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Guarnerio J, Bezzi M, Jeong JC, Paffenholz
SV, Berry K, Naldini MM, Lo-Coco F, Tay Y, Beck AH and Pandolfi PP:
Oncogenic role of Fusion-circRNAs derived from cancer-associated
chromosomal translocations. Cell. 165:289–302. 2016. View Article : Google Scholar
|