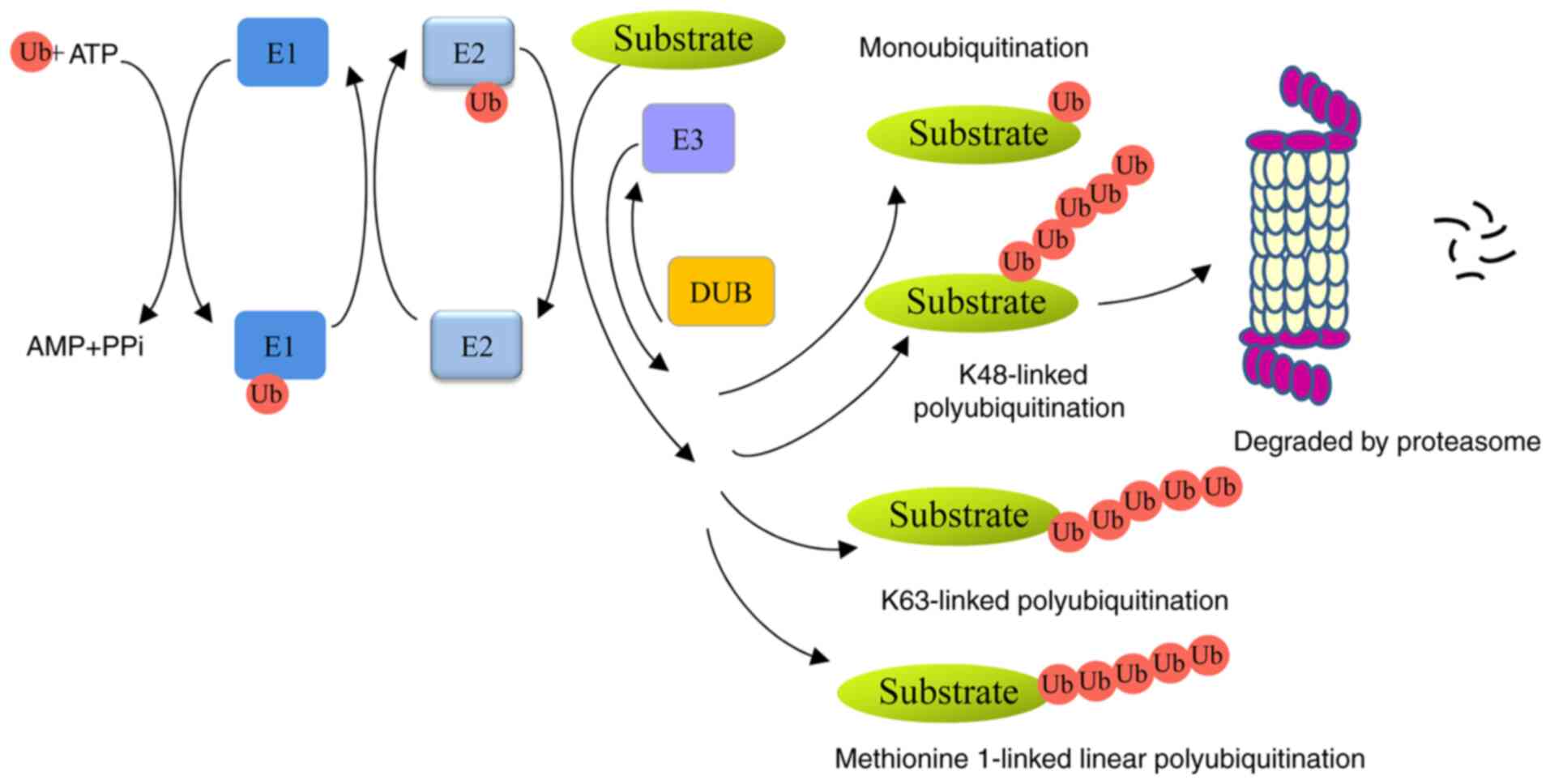
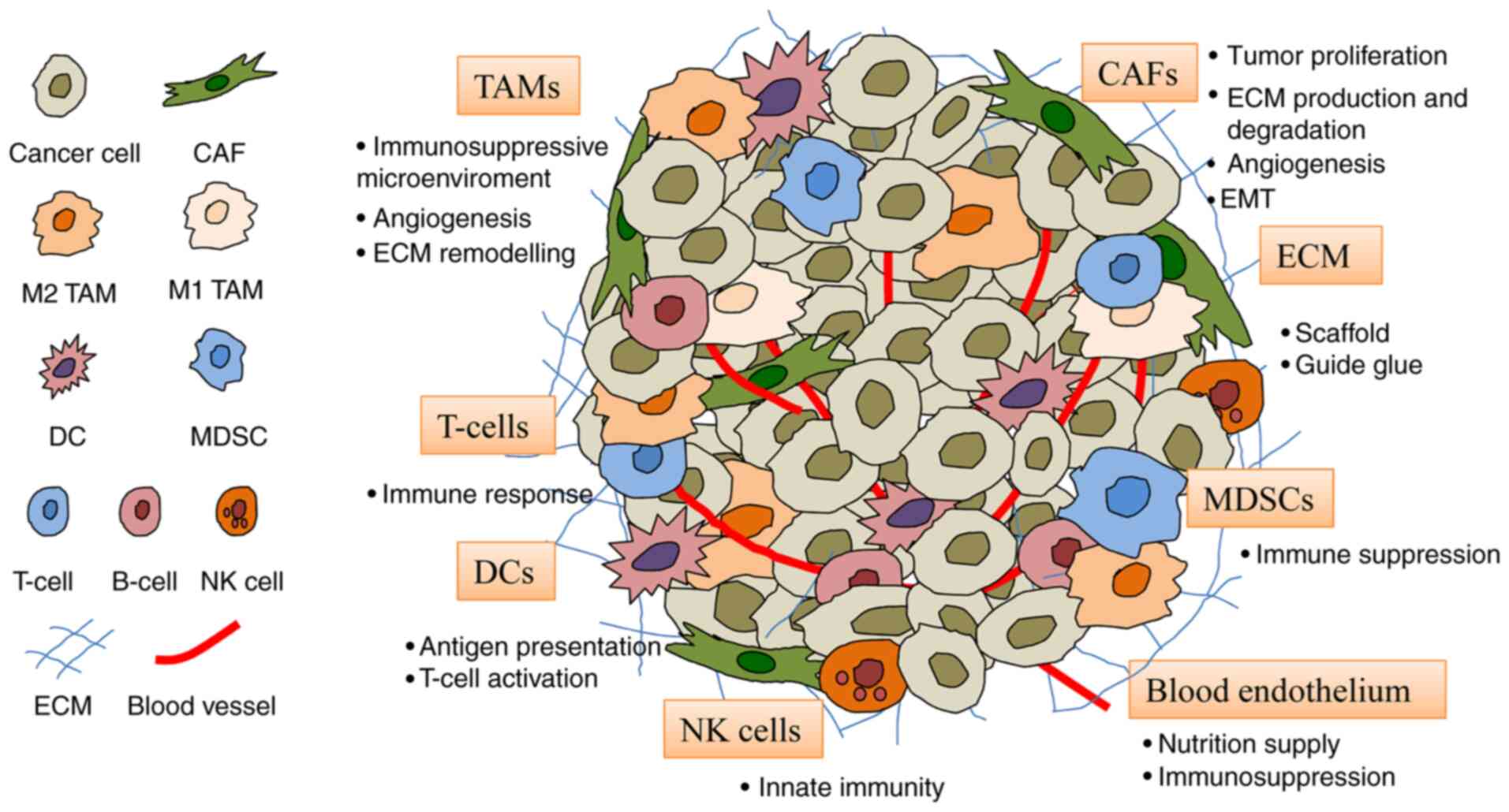
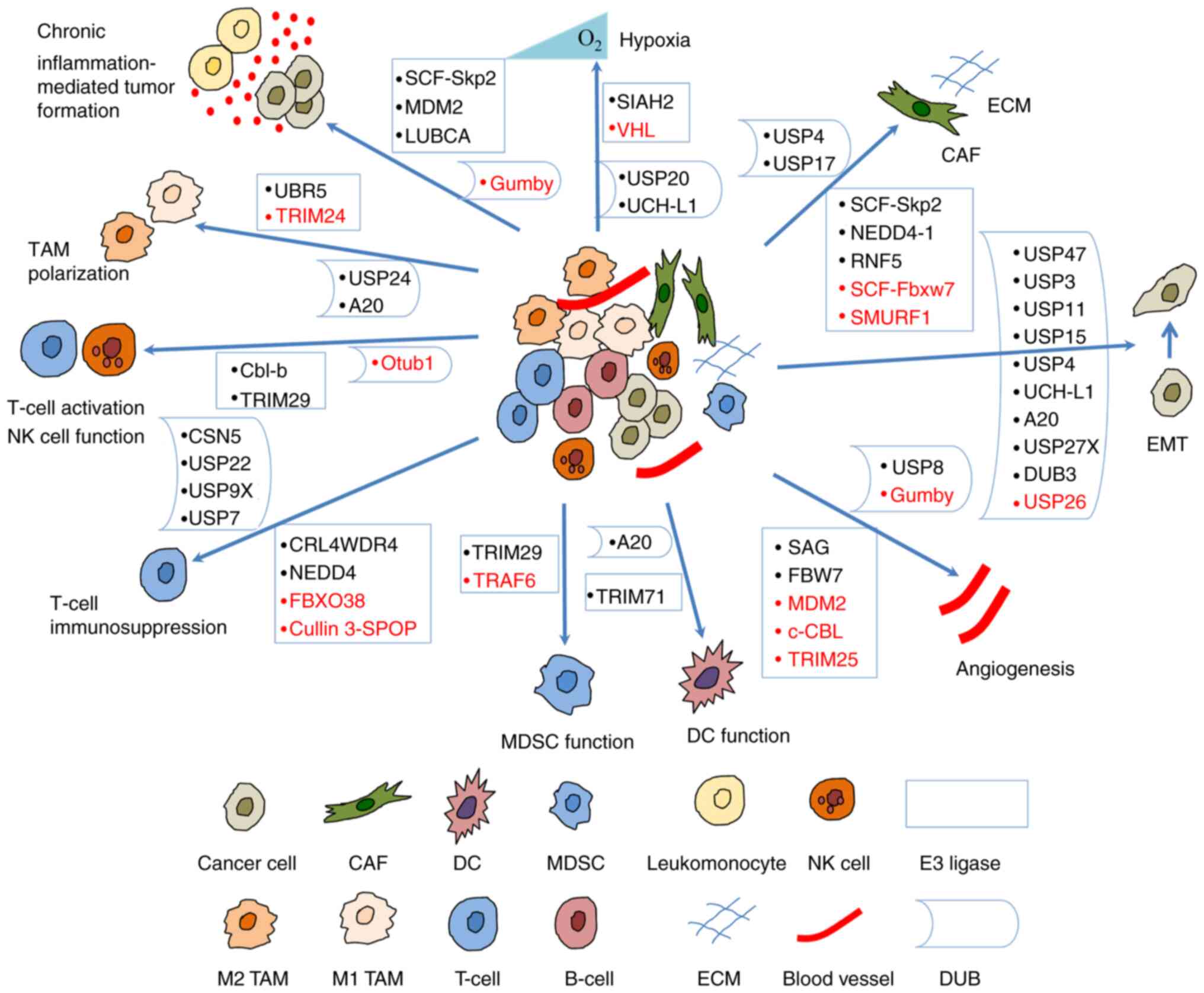
|
1
|
Casey SC, Amedei A, Aquilano K, Azmi AS,
Benencia F, Bhakta D, Bilsland AE, Boosani CS, Chen S, Ciriolo MR,
et al: Cancer prevention and therapy through the modulation of the
tumor microenvironment. Semin Cancer Biol. 35(Suppl): S199–S223.
2015. View Article : Google Scholar
|
|
2
|
Ribas A and Wolchok JD: Cancer
immunotherapy using checkpoint blockade. Science. 359:1350–1355.
2018. View Article : Google Scholar
|
|
3
|
Meng T, Huang R, Jin J, Gao J, Liu F, Wei
Z, Xu X, Chang Z, Lin J, Ta N, et al: A comparative integrated
multi-omics analysis identifies CA2 as a novel target for chordoma.
Neuro Oncol. 23:1709–1722. 2021. View Article : Google Scholar
|
|
4
|
Suryadinata R, Roesley SN, Yang G and
Sarcevic B: Mechanisms of generating polyubiquitin chains of
different topology. Cells. 3:674–689. 2014. View Article : Google Scholar
|
|
5
|
Haglund K, Di Fiore PP and Dikic I:
Distinct monoubiquitin signals in receptor endocytosis. Trends
Biochem Sci. 28:598–603. 2003. View Article : Google Scholar
|
|
6
|
Erpapazoglou Z, Walker O and
Haguenauer-Tsapis R: Versatile roles of k63-linked ubiquitin chains
in trafficking. Cells. 3:1027–1088. 2014. View Article : Google Scholar
|
|
7
|
Gerlach B, Cordier SM, Schmukle AC,
Emmerich CH, Rieser E, Haas TL, Webb AI, Rickard JA, Anderton H,
Wong WW, et al: Linear ubiquitination prevents inflammation and
regulates immune signalling. Nature. 471:591–596. 2011. View Article : Google Scholar
|
|
8
|
Jahan AS, Elbæk CR and Damgaard RB:
Met1-linked ubiquitin signalling in health and disease:
Inflammation, immunity, cancer, and beyond. Cell Death Differ.
28:473–492. 2021. View Article : Google Scholar
|
|
9
|
Bhattacharya S and Ghosh MK: Cell death
and deubiquitinases: Perspectives in cancer. Biomed Res Int.
2014:4351972014. View Article : Google Scholar
|
|
10
|
Senft D, Qi J and Ronai ZA: Ubiquitin
ligases in oncogenic transformation and cancer therapy. Nat Rev
Cancer. 18:69–88. 2018. View Article : Google Scholar
|
|
11
|
Morrow JK, Lin HK, Sun SC and Zhang S:
Targeting ubiquitination for cancer therapies. Future Med Chem.
7:2333–2350. 2015. View Article : Google Scholar
|
|
12
|
Wang D, Ma L, Wang B, Liu J and Wei W: E3
ubiquitin ligases in cancer and implications for therapies. Cancer
Metastasis Rev. 36:683–702. 2017. View Article : Google Scholar
|
|
13
|
Wei R, Liu X, Yu W, Yang T, Cai W, Liu J,
Huang X, Xu GT, Zhao S, Yang J and Liu S: Deubiquitinases in
cancer. Oncotarget. 6:12872–12889. 2015. View Article : Google Scholar
|
|
14
|
Ishii G, Ochiai A and Neri S: Phenotypic
and functional heterogeneity of cancer-associated fibroblast within
the tumor microenvironment. Adv Drug Deliv Rev. 99(Pt B): 186–196.
2016. View Article : Google Scholar
|
|
15
|
Vosseler S, Lederle W, Airola K,
Obermueller E, Fusenig NE and Mueller MM: Distinct
progression-associated expression of tumor and stromal MMPs in
HaCaT skin SCCs correlates with onset of invasion. Int J Cancer.
125:2296–2306. 2009. View Article : Google Scholar
|
|
16
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar
|
|
17
|
Griffioen AW, Damen CA, Blijham GH and
Groenewegen G: Tumor angiogenesis is accompanied by a decreased
inflammatory response of tumor-associated endothelium. Blood.
88:667–673. 1996. View Article : Google Scholar
|
|
18
|
Rodig N, Ryan T, Allen JA, Pang H, Grabie
N, Chernova T, Greenfield EA, Liang SC, Sharpe AH, Lichtman AH and
Freeman GJ: Endothelial expression of PD-L1 and PD-L2
down-regulates CD8+ T cell activation and cytolysis. Eur
J Immunol. 33:3117–3126. 2003. View Article : Google Scholar
|
|
19
|
Mulligan JK, Day TA, Gillespie MB,
Rosenzweig SA and Young MRI: Secretion of vascular endothelial
growth factor by oral squamous cell carcinoma cells skews
endothelial cells to suppress T-cell functions. Hum Immunol.
70:375–382. 2009. View Article : Google Scholar
|
|
20
|
Mulligan JK and Young MR: Tumors induce
the formation of suppressor endothelial cells in vivo. Cancer
Immunol Immunother. 59:267–277. 2010. View Article : Google Scholar
|
|
21
|
Talmadge JE and Gabrilovich DI: History of
myeloid-derived suppressor cells. Nat Rev Cancer. 13:739–752. 2013.
View Article : Google Scholar
|
|
22
|
Wang J, Li D, Cang H and Guo B: Crosstalk
between cancer and immune cells: Role of tumor-associated
macrophages in the tumor microenvironment. Cancer Med. 8:4709–4721.
2019. View Article : Google Scholar
|
|
23
|
Jayasingam SD, Citartan M, Thang TH, Mat
Zin AA, Ang KC and Ch'ng ES: Evaluating the polarization of
tumor-associated macrophages into M1 and M2 phenotypes in human
cancer tissue: Technicalities and challenges in routine clinical
practice. Front Oncol. 9:15122020. View Article : Google Scholar
|
|
24
|
Laviron M and Boissonnas A: Ontogeny of
tumor-associated macrophages. Front Immunol. 10:17992019.
View Article : Google Scholar
|
|
25
|
Jiang S and Yan W: T-cell immunometabolism
against cancer. Cancer Lett. 382:255–258. 2016. View Article : Google Scholar
|
|
26
|
Gabrilovich DI: Myeloid-derived suppressor
cells. Cancer Immunol Res. 5:3–8. 2017. View Article : Google Scholar
|
|
27
|
Oya Y, Hayakawa Y and Koike K: Tumor
microenvironment in gastric cancers. Cancer Sci. 111:2696–2707.
2020. View Article : Google Scholar
|
|
28
|
Tran Janco JM, Lamichhane P, Karyampudi L
and Knutson KL: Tumor-infiltrating dendritic cells in cancer
pathogenesis. J Immunol. 194:2985–2991. 2015. View Article : Google Scholar
|
|
29
|
Wu SY, Fu T, Jiang YZ and Shao ZM: Natural
killer cells in cancer biology and therapy. Mol Cancer. 19:1202020.
View Article : Google Scholar
|
|
30
|
Wu T and Dai Y: Tumor microenvironment and
therapeutic response. Cancer Lett. 387:61–68. 2017. View Article : Google Scholar
|
|
31
|
Jaakkola P, Mole DR, Tian YM, Wilson MI,
Gielbert J, Gaskell SJ, von Kriegsheim A, Hebestreit HF, Mukherji
M, Schofield CJ, et al: Targeting of HIF-alpha to the von
Hippel-Lindau ubiquitylation complex by O2-regulated prolyl
hydroxylation. Science. 292:468–472. 2001. View Article : Google Scholar
|
|
32
|
Mennerich D, Kubaichuk K and Kietzmann T:
DUBs, hypoxia, and cancer. Trends Cancer. 5:632–653. 2019.
View Article : Google Scholar
|
|
33
|
Li Z, Wang D, Messing EM and Wu G: VHL
protein-interacting deubiquitinating enzyme 2 deubiquitinates and
stabilizes HIF-1alpha. EMBO Rep. 6:373–378. 2005. View Article : Google Scholar
|
|
34
|
Goto Y, Zeng L, Yeom CJ, Zhu Y, Morinibu
A, Shinomiya K, Kobayashi M, Hirota K, Itasaka S, Yoshimura M, et
al: UCHL1 provides diagnostic and antimetastatic strategies due to
its deubiquitinating effect on HIF-1α. Nat Commun. 6:61532015.
View Article : Google Scholar
|
|
35
|
Choi BJ, Park SA, Lee SY, Cha YN and Surh
YJ: Hypoxia induces epithelial-mesenchymal transition in colorectal
cancer cells through ubiquitin-specific protease 47-mediated
stabilization of Snail: A potential role of Sox9. Sci Rep.
7:159182017. View Article : Google Scholar
|
|
36
|
Ma B, Cheng H, Mu C, Geng G, Zhao T, Luo
Q, Ma K, Chang R, Liu Q, Gao R, et al: The SIAH2-NRF1 axis
spatially regulates tumor microenvironment remodeling for tumor
progression. Nat Commun. 10:10342019. View Article : Google Scholar
|
|
37
|
Smith GA, Fearnley GW, Abdul-Zani I,
Wheatcroft SB, Tomlinson DC, Harrison MA and Ponnambalam S: VEGFR2
trafficking, signaling and proteolysis is regulated by the
ubiquitin isopeptidase USP8. Traffic. 17:53–65. 2016. View Article : Google Scholar
|
|
38
|
Lee JY, Park JH, Choi HJ, Won HY, Joo HS,
Shin DH, Park MK, Han B, Kim KP, Lee TJ, et al: LSD1 demethylates
HIF1α to inhibit hydroxylation and ubiquitin-mediated degradation
in tumor angiogenesis. Oncogene. 36:5512–5521. 2017. View Article : Google Scholar
|
|
39
|
Amelio I, Inoue S, Markert EK, Levine AJ,
Knight RA, Mak TW and Melino G: TAp73 opposes tumor angiogenesis by
promoting hypoxia-inducible factor 1α degradation. Proc Natl Acad
Sci USA. 112:226–231. 2015. View Article : Google Scholar
|
|
40
|
Pan T, Zhou D, Shi Z, Qiu Y, Zhou G, Liu
J, Yang Q, Cao L and Zhang J: Centromere protein U (CENPU) enhances
angiogenesis in triple-negative breast cancer by inhibiting
ubiquitin-proteasomal degradation of COX-2. Cancer Lett.
482:102–111. 2020. View Article : Google Scholar
|
|
41
|
Sun Y and Li H: Functional
characterization of SAG/RBX2/ROC2/RNF7, an antioxidant protein and
an E3 ubiquitin ligase. Protein Cell. 4:103–116. 2013. View Article : Google Scholar
|
|
42
|
Tan M, Li H and Sun Y: Endothelial
deletion of Sag/Rbx2/Roc2 E3 ubiquitin ligase causes embryonic
lethality and blocks tumor angiogenesis. Oncogene. 33:5211–5220.
2014. View Article : Google Scholar
|
|
43
|
Hasan SS, Tsaryk R, Lange M, Wisniewski L,
Moore JC, Lawson ND, Wojciechowska K, Schnittler H and Siekmann AF:
Endothelial Notch signalling limits angiogenesis via control of
artery formation. Nat Cell Biol. 19:928–940. 2017. View Article : Google Scholar
|
|
44
|
Rabellino A, Andreani C and Scaglioni PP:
Roles of ubiquitination and SUMOylation in the regulation of
angiogenesis. Curr Issues Mol Biol. 35:109–126. 2020. View Article : Google Scholar
|
|
45
|
Tsunematsu R, Nakayama K, Oike Y,
Nishiyama M, Ishida N, Hatakeyama S, Bessho Y, Kageyama R, Suda T
and Nakayama KI: Mouse Fbw7/Sel-10/Cdc4 is required for notch
degradation during vascular development. J Biol Chem.
279:9417–9423. 2004. View Article : Google Scholar
|
|
46
|
Zerlin M, Julius MA and Kitajewski J:
Wnt/Frizzled signaling in angiogenesis. Angiogenesis. 11:63–69.
2008. View Article : Google Scholar
|
|
47
|
MacDonald BT, Tamai K and He X:
Wnt/beta-catenin signaling: Components, mechanisms, and diseases.
Dev Cell. 17:9–26. 2009. View Article : Google Scholar
|
|
48
|
Shivanna S, Harrold I, Shashar M, Meyer R,
Kiang C, Francis J, Zhao Q, Feng H, Edelman ER, Rahimi N and
Chitalia VC: The c-Cbl ubiquitin ligase regulates nuclear β-catenin
and angiogenesis by its tyrosine phosphorylation mediated through
the Wnt signaling pathway. J Biol Chem. 290:12537–12546. 2015.
View Article : Google Scholar
|
|
49
|
Rivkin E, Almeida SM, Ceccarelli DF, Juang
YC, MacLean TA, Srikumar T, Huang H, Dunham WH, Fukumura R, Xie G,
et al: The linear ubiquitin-specific deubiquitinase gumby regulates
angiogenesis. Nature. 498:318–324. 2013. View Article : Google Scholar
|
|
50
|
Wang W, Li M, Ponnusamy S, Chi Y, Xue J,
Fahmy B, Fan M, Miranda-Carboni GA, Narayanan R, Wu J and Wu ZH:
ABL1-dependent OTULIN phosphorylation promotes genotoxic
Wnt/β-catenin activation to enhance drug resistance in breast
cancers. Nat Commun. 11:39652020. View Article : Google Scholar
|
|
51
|
Quintero-Fabián S, Arreola R,
Becerril-Villanueva E, Torres-Romero JC, Arana-Argáez V,
Lara-Riegos J, Ramírez-Camacho MA and Alvarez-Sánchez ME: Role of
matrix metalloproteinases in angiogenesis and cancer. Front Oncol.
9:13702019. View Article : Google Scholar
|
|
52
|
Murray GI, Duncan ME, Arbuckle E, Melvin
WT and Fothergill JE: Matrix metalloproteinases and their
inhibitors in gastric cancer. Gut. 43:791–797. 1998. View Article : Google Scholar
|
|
53
|
Pikarsky E, Porat RM, Stein I, Abramovitch
R, Amit S, Kasem S, Gutkovich-Pyest E, Urieli-Shoval S, Galun E and
Ben-Neriah Y: NF-kappaB functions as a tumour promoter in
inflammation-associated cancer. Nature. 431:461–466. 2004.
View Article : Google Scholar
|
|
54
|
Chang SC and Ding JL: Ubiquitination and
SUMOylation in the chronic inflammatory tumor microenvironment.
Biochim Biophys Acta Rev Cancer. 1870:165–175. 2018. View Article : Google Scholar
|
|
55
|
Peng SL: Forkhead transcription factors in
chronic inflammation. Int J Biochem Cell Biol. 42:482–485. 2010.
View Article : Google Scholar
|
|
56
|
Huang H and Tindall DJ: Dynamic FoxO
transcription factors. J Cell Sci. 120(Pt 15): 2479–2487. 2007.
View Article : Google Scholar
|
|
57
|
Ramezani A, Nikravesh H and Faghihloo E:
The roles of FOX proteins in virus-associated cancers. J Cell
Physiol. 234:3347–3361. 2019. View Article : Google Scholar
|
|
58
|
Huang H, Regan KM, Wang F, Wang D, Smith
DI, van Deursen JM and Tindall DJ: Skp2 inhibits FOXO1 in tumor
suppression through ubiquitin-mediated degradation. Proc Natl Acad
Sci USA. 102:1649–1654. 2005. View Article : Google Scholar
|
|
59
|
Fu W, Ma Q, Chen L, Li P, Zhang M,
Ramamoorthy S, Nawaz Z, Shimojima T, Wang H, Yang Y, et al: MDM2
acts downstream of p53 as an E3 ligase to promote FOXO
ubiquitination and degradation. J Biol Chem. 284:13987–14000. 2009.
View Article : Google Scholar
|
|
60
|
Chang SC and Ding JL: Ubiquitination by
SAG regulates macrophage survival/death and immune response during
infection. Cell Death Differ. 21:1388–1398. 2014. View Article : Google Scholar
|
|
61
|
Chang SC, Choo WQ, Toh HC and Ding JL:
SAG-UPS attenuates proapoptotic SARM and Noxa to confer survival
advantage to early hepatocellular carcinoma. Cell Death Discov.
1:150322015. View Article : Google Scholar
|
|
62
|
Chada S, Sutton RB, Ekmekcioglu S,
Ellerhorst J, Mumm JB, Leitner WW, Yang HY, Sahin AA, Hunt KK,
Fuson KL, et al: MDA-7/IL-24 is a unique cytokine-tumor suppressor
in the IL-10 family. Int Immunopharmacol. 4:649–667. 2004.
View Article : Google Scholar
|
|
63
|
Gopalan B, Shanker M, Scott A, Branch CD,
Chada S and Ramesh R: MDA-7/IL-24, a novel tumor
suppressor/cytokine is ubiquitinated and regulated by the
ubiquitin-proteasome system, and inhibition of MDA-7/IL-24
degradation enhances the anti-tumor activity. Cancer Gene Ther.
15:1–8. 2008. View Article : Google Scholar
|
|
64
|
Liu P, Zhang X, Li Z, Wei L, Peng Q, Liu
C, Wu Y, Yan Q and Ma J: A significant role of transcription
factors E2F in inflammation and tumorigenesis of nasopharyngeal
carcinoma. Biochem Biophys Res Commun. 524:816–824. 2020.
View Article : Google Scholar
|
|
65
|
Swarnalatha M, Singh AK and Kumar V:
Promoter occupancy of MLL1 histone methyltransferase seems to
specify the proliferative and apoptotic functions of E2F1 in a
tumour microenvironment. Cell Sci. 126(Pt 20): 4636–4646. 2013.
|
|
66
|
Murata M: Inflammation and cancer. Environ
Health Prev Med. 23:502018. View Article : Google Scholar
|
|
67
|
Sun L, Wu J, Du F, Chen X and Chen ZJ:
Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates
the type I interferon pathway. Science. 339:786–791. 2013.
View Article : Google Scholar
|
|
68
|
Bakhoum SF, Ngo B, Laughney AM, Cavallo
JA, Murphy CJ, Ly P, Shah P, Sriram RK, Watkins TBK, Taunk NK, et
al: Chromosomal instability drives metastasis through a cytosolic
DNA response. Nature. 553:467–472. 2018. View Article : Google Scholar
|
|
69
|
Wu S, Zhang Q, Zhang F, Meng F, Liu S,
Zhou R, Wu Q, Li X, Shen L, Huang J, et al: HER2 recruits AKT1 to
disrupt STING signalling and suppress antiviral defence and
antitumour immunity. Nat Cell Biol. 21:1027–1040. 2019. View Article : Google Scholar
|
|
70
|
Kensche T, Tokunaga F, Ikeda F, Goto E,
Iwai K and Dikic I: Analysis of nuclear factor-κB (NF-κB) essential
modulator (NEMO) binding to linear and lysine-linked ubiquitin
chains and its role in the activation of NF-κB. J Biol Chem.
287:23626–23634. 2012. View Article : Google Scholar
|
|
71
|
Jo T, Nishikori M, Kogure Y, Arima H,
Sasaki K, Sasaki Y, Nakagawa T, Iwai F, Momose S, Shiraishi A, et
al: LUBAC accelerates B-cell lymphomagenesis by conferring
resistance to genotoxic stress on B cells. Blood. 136:684–697.
2020. View Article : Google Scholar
|
|
72
|
Song K, Cai X, Dong Y, Wu H, Wei Y,
Shankavaram UT, Cui K, Lee Y, Zhu B, Bhattacharjee S, et al: Epsins
1 and 2 promote NEMO linear ubiquitination via LUBAC to drive
breast cancer development. J Clin Invest. 131:e1293742021.
View Article : Google Scholar
|
|
73
|
Damgaard RB, Jolin HE, Allison MED, Davies
SE, Titheradge HL, McKenzie ANJ and Komander D: OTULIN protects the
liver against cell death, inflammation, fibrosis, and cancer. Cell
Death Differ. 27:1457–1474. 2020. View Article : Google Scholar
|
|
74
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View Article : Google Scholar
|
|
75
|
Lambies G, Miceli M, Martínez-Guillamon C,
Olivera-Salguero R, Peña R, Frías CP, Calderó I, Atanassov BS, Dent
SYR, Arribas J, et al: TGFβ-Activated USP27X deubiquitinase
regulates cell migration and chemoresistance via stabilization of
snail1. Cancer Res. 79:33–46. 2019. View Article : Google Scholar
|
|
76
|
Wu X, Liu M, Zhu H, Wang J, Dai W, Li J,
Zhu D, Tang W, Xiao Y, Lin J, et al: Ubiquitin-specific protease 3
promotes cell migration and invasion by interacting with and
deubiquitinating SUZ12 in gastric cancer. J Exp Clin Cancer Res.
38:2772019. View Article : Google Scholar
|
|
77
|
Wang W, Wang J, Yan H, Zhang K and Liu Y:
Upregulation of USP11 promotes epithelial-to-mesenchymal transition
by deubiquitinating Snail in ovarian cancer. Oncol Rep.
41:1739–1748. 2019.
|
|
78
|
Garcia DA, Baek C, Estrada MV, Tysl T,
Bennett EJ, Yang J and Chang JT: USP11 enhances TGFβ-Induced
epithelial-mesenchymal plasticity and human breast cancer
metastasis. Mol Cancer Res. 16:1172–1184. 2018. View Article : Google Scholar
|
|
79
|
Kit Leng Lui S, Iyengar PV, Jaynes P, Isa
ZFBA, Pang B, Tan TZ and Eichhorn PJA: USP26 regulates TGF-β
signaling by deubiquitinating and stabilizing SMAD7. EMBO Rep.
18:797–808. 2017. View Article : Google Scholar
|
|
80
|
Eichhorn PJ, Rodó L, Gonzàlez-Juncà A,
Dirac A, Gili M, Martínez-Sáez E, Aura C, Barba I, Peg V, Prat A,
et al: USP15 stabilizes TGF-β receptor I and promotes oncogenesis
through the activation of TGF-β signaling in glioblastoma. Nat Med.
18:429–435. 2012. View Article : Google Scholar
|
|
81
|
Zhang L, Zhou F, Drabsch Y, Gao R,
Snaar-Jagalska BE, Mickanin C, Huang H, Sheppard KA, Porter JA, Lu
CX and ten Dijke P: USP4 is regulated by AKT phosphorylation and
directly deubiquitylates TGF-β type I receptor. Nat Cell Biol.
14:717–726. 2012. View Article : Google Scholar
|
|
82
|
Jang MJ, Baek SH and Kim JH: UCH-L1
promotes cancer metastasis in prostate cancer cells through EMT
induction. Cancer Lett. 302:128–135. 2011. View Article : Google Scholar
|
|
83
|
Lee JH, Jung SM, Yang KM, Bae E, Ahn SG,
Park JS, Seo D, Kim M, Ha J, Lee J, et al: A20 promotes metastasis
of aggressive basal-like breast cancers through
multi-monoubiquitylation of Snail1. Nat Cell Biol. 19:1260–1273.
2017. View Article : Google Scholar
|
|
84
|
Pitarresi JR, Liu X, Avendano A, Thies KA,
Sizemore GM, Hammer AM, Hildreth BE III, Wang DJ, Steck SA, Donohue
S, et al: Disruption of stromal hedgehog signaling initiates
RNF5-mediated proteasomal degradation of PTEN and accelerates
pancreatic tumor growth. Life Sci Alliance. 1:e2018001902018.
View Article : Google Scholar
|
|
85
|
Karakasheva TA, Lin EW, Tang Q, Qiao E,
Waldron TJ, Soni M, Klein-Szanto AJ, Sahu V, Basu D, Ohashi S, et
al: IL-6 mediates cross-talk between tumor cells and activated
fibroblasts in the tumor microenvironment. Cancer Res.
78:4957–4970. 2018. View Article : Google Scholar
|
|
86
|
Wu X, Tao P, Zhou Q, Li J, Yu Z, Wang X,
Li J, Li C, Yan M, Zhu Z, et al: IL-6 secreted by cancer-associated
fibroblasts promotes epithelial-mesenchymal transition and
metastasis of gastric cancer via JAK2/STAT3 signaling pathway.
Oncotarget. 8:20741–20750. 2017. View Article : Google Scholar
|
|
87
|
Wu Y, Wang Y, Lin Y, Liu Y, Wang Y, Jia J,
Singh P, Chi YI, Wang C, Dong C, et al: Dub3 inhibition suppresses
breast cancer invasion and metastasis by promoting Snail1
degradation. Nat Commun. 8:142282017. View Article : Google Scholar
|
|
88
|
Borsig L, Wolf MJ, Roblek M, Lorentzen A
and Heikenwalder M: Inflammatory chemokines and metastasis-tracing
the accessory. Oncogene. 33:3217–3224. 2014. View Article : Google Scholar
|
|
89
|
Liu J, Chen S, Wang W, Ning BF, Chen F,
Shen W, Ding J, Chen W, Xie WF and Zhang X: Cancer-associated
fibroblasts promote hepatocellular carcinoma metastasis through
chemokine-activated hedgehog and TGF-β pathways. Cancer Lett.
379:49–59. 2016. View Article : Google Scholar
|
|
90
|
Yumimoto K and Nakayama KI: Fbxw7
suppresses cancer metastasis by inhibiting niche formation.
Oncoimmunology. 4:e10223082015. View Article : Google Scholar
|
|
91
|
Mehić M, de Sa VK, Hebestreit S, Heldin CH
and Heldin P: The deubiquitinating enzymes USP4 and USP17 target
hyaluronan synthase 2 and differentially affect its function.
Oncogenesis. 6:e3482017. View Article : Google Scholar
|
|
92
|
Stetler-Stevenson WG and Yu AE: Proteases
in invasion: Matrix metalloproteinases. Semin Cancer Biol.
11:143–152. 2001. View Article : Google Scholar
|
|
93
|
Stefanidakis M and Koivunen E:
Cell-surface association between matrix metalloproteinases and
integrins: Role of the complexes in leukocyte migration and cancer
progression. Blood. 108:1441–1450. 2006. View Article : Google Scholar
|
|
94
|
Gontero P, Banisadr S, Frea B and Brausi
M: Metastasis markers in bladder cancer: A review of the literature
and clinical considerations. Eur Urol. 46:296–311. 2004. View Article : Google Scholar
|
|
95
|
Li S and Luo W: Matrix metalloproteinase 2
contributes to aggressive phenotype, epithelial-mesenchymal
transition and poor outcome in nasopharyngeal carcinoma. Onco
Targets Ther. 12:5701–5711. 2019. View Article : Google Scholar
|
|
96
|
Yamada S, Yanamoto S, Naruse T, Matsushita
Y, Takahashi H, Umeda M, Nemoto TK and Kurita H: Skp2 regulates the
expression of MMP-2 and MMP-9, and enhances the invasion potential
of oral squamous cell carcinoma. Pathol Oncol Res. 22:625–632.
2016. View Article : Google Scholar
|
|
97
|
Hung WC, Tseng WL, Shiea J and Chang HC:
Skp2 overexpression increases the expression of MMP-2 and MMP-9 and
invasion of lung cancer cells. Cancer Lett. 288:156–161. 2010.
View Article : Google Scholar
|
|
98
|
Chernov AV, Sounni NE, Remacle AG and
Strongin AY: Epigenetic control of the invasion-promoting
MT1-MMP/MMP-2/TIMP-2 axis in cancer cells. J Biol Chem.
284:12727–12734. 2009. View Article : Google Scholar
|
|
99
|
Sakai K, Nishiuchi T, Tange S, Suzuki Y,
Yano S, Terashima M, Suzuki T and Matsumoto K: Proteasomal
degradation of polycomb-group protein CBX6 confers MMP-2 expression
essential for mesothelioma invasion. Sci Rep. 10:166782020.
View Article : Google Scholar
|
|
100
|
Priolo C, Tang D, Brahamandan M, Benassi
B, Sicinska E, Ogino S, Farsetti A, Porrello A, Finn S, Zimmermann
J, et al: The isopeptidase USP2a protects human prostate cancer
from apoptosis. Cancer Res. 66:8625–8632. 2006. View Article : Google Scholar
|
|
101
|
Qu Q, Mao Y, Xiao G, Fei X, Wang J, Zhang
Y, Liu J, Cheng G, Chen X, Wang J and Shen K: USP2 promotes cell
migration and invasion in triple negative breast cancer cell lines.
Tumour Biol. 36:5415–5423. 2015. View Article : Google Scholar
|
|
102
|
Nguyen HL, Kadam P, Helkin A, Cao K, Wu S,
Samara GJ, Zhang Q, Zucker S and Cao J: MT1-MMP Activation of TGF-β
signaling enables intercellular activation of an
epithelial-mesenchymal transition program in cancer. Curr Cancer
Drug Targets. 16:618–630. 2016. View Article : Google Scholar
|
|
103
|
Eisenach PA, de Sampaio PC, Murphy G and
Roghi C: Membrane type 1 matrix metalloproteinase (MT1-MMP)
ubiquitination at Lys581 increases cellular invasion through type I
collagen. J Biol Chem. 287:11533–11545. 2012. View Article : Google Scholar
|
|
104
|
Noy R and Pollard JW: Tumor-associated
macrophages: From mechanisms to therapy. Immunity. 41:49–61. 2014.
View Article : Google Scholar
|
|
105
|
Yu T, Gan S, Zhu Q, Dai D, Li N, Wang H,
Chen X, Hou D, Wang Y, Pan Q, et al: Modulation of M2 macrophage
polarization by the crosstalk between Stat6 and Trim24. Nat Commun.
10:43532019. View Article : Google Scholar
|
|
106
|
Rőszer T: Understanding the Mysterious M2
Macrophage through activation markers and effector mechanisms.
Mediators Inflamm. 2015:8164602015. View Article : Google Scholar
|
|
107
|
Wang YC, Wu YS, Hung CY, Wang SA, Young
MJ, Hsu TI and Hung JJ: USP24 induces IL-6 in tumor-associated
microenvironment by stabilizing p300 and β-TrCP and promotes cancer
malignancy. Nat Commun. 9:39962018. View Article : Google Scholar
|
|
108
|
Ning C, Xie B, Zhang L, Li C, Shan W, Yang
B, Luo X, Gu C, He Q, Jin H, et al: Infiltrating Macrophages Induce
ERα Expression through an IL17A-mediated epigenetic mechanism to
sensitize endometrial cancer cells to estrogen. Cancer Res.
76:1354–1366. 2016. View Article : Google Scholar
|
|
109
|
Lv Q, Xie L, Cheng Y, Shi Y, Shan W, Ning
C, Xie B, Yang B, Luo X, He Q, et al: A20-mediated deubiquitination
of ERα in the microenvironment of CD163+ macrophages sensitizes
endometrial cancer cells to estrogen. Cancer Lett. 442:137–147.
2019. View Article : Google Scholar
|
|
110
|
Clancy JL, Henderson MJ, Russell AJ,
Anderson DW, Bova RJ, Campbell IG, Choong DY, Macdonald GA, Mann
GJ, Nolan T, et al: EDD, the human orthologue of the hyperplastic
discs tumour suppressor gene, is amplified and overexpressed in
cancer. Oncogene. 22:5070–5081. 2003. View Article : Google Scholar
|
|
111
|
Song M, Yeku OO, Rafiq S, Purdon T, Dong
X, Zhu L, Zhang T, Wang H, Yu Z, Mai J, et al: Tumor derived UBR5
promotes ovarian cancer growth and metastasis through inducing
immunosuppressive macrophages. Nat Commun. 11:62982020. View Article : Google Scholar
|
|
112
|
Surh CD and Sprent J: Homeostasis of naive
and memory T cells. Immunity. 29:848–862. 2008. View Article : Google Scholar
|
|
113
|
Zhou X, Yu J, Cheng X, Zhao B, Manyam GC,
Zhang L, Schluns K, Li P, Wang J and Sun SC: The deubiquitinase
Otub1 controls the activation of CD8+ T cells and NK cells by
regulating IL-15-mediated priming. Nat Immunol. 20:879–889. 2019.
View Article : Google Scholar
|
|
114
|
Zhang W, Sloan-Lancaster J, Kitchen J,
Trible RP and Samelson LE: LAT: The ZAP-70 tyrosine kinase
substrate that links T cell receptor to cellular activation. Cell.
92:83–92. 1998. View Article : Google Scholar
|
|
115
|
Kunii N, Zhao Y, Jiang S, Liu X, Scholler
J, Balagopalan L, Samelson LE, Milone MC and June CH: Enhanced
function of redirected human T cells expressing linker for
activation of T cells that is resistant to ubiquitylation. Hum Gene
Ther. 24:27–37. 2013. View Article : Google Scholar
|
|
116
|
Chen RH, Lee YR and Yuan WC: The role of
PML ubiquitination in human malignancies. J Biomed Sci. 19:812012.
View Article : Google Scholar
|
|
117
|
Wang YT, Chen J, Chang CW, Jen J, Huang
TY, Chen CM, Shen R, Liang SY, Cheng IC, Yang SC, et al:
Ubiquitination of tumor suppressor PML regulates prometastatic and
immunosuppressive tumor microenvironment. J Clin Invest.
127:2982–2997. 2017. View Article : Google Scholar
|
|
118
|
Meng X, Liu X, Guo X, Jiang S, Chen T, Hu
Z, Liu H, Bai Y, Xue M, Hu R, et al: FBXO38 mediates PD-1
ubiquitination and regulates anti-tumour immunity of T cells.
Nature. 564:130–135. 2018. View Article : Google Scholar
|
|
119
|
Li CW, Lim SO, Xia W, Lee HH, Chan LC, Kuo
CW, Khoo KH, Chang SS, Cha JH, Kim T, et al: Glycosylation and
stabilization of programmed death ligand-1 suppresses T-cell
activity. Nat Commun. 7:126322016. View Article : Google Scholar
|
|
120
|
Zhang J, Bu X, Wang H, Zhu Y, Geng Y,
Nihira NT, Tan Y, Ci Y, Wu F, Dai X, et al: Cyclin D-CDK4 kinase
destabilizes PD-L1 via cullin 3-SPOP to control cancer immune
surveillance. Nature. 553:91–95. 2018. View Article : Google Scholar
|
|
121
|
Song Y, Xu Y, Pan C, Yan L, Wang ZW and
Zhu X: The emerging role of SPOP protein in tumorigenesis and
cancer therapy. Mol Cancer. 19:22020. View Article : Google Scholar
|
|
122
|
Lim SO, Li CW, Xia W, Cha JH, Chan LC, Wu
Y, Chang SS, Lin WC, Hsu JM, Hsu YH, et al: Deubiquitination and
Stabilization of PD-L1 by CSN5. Cancer Cell. 30:925–939. 2016.
View Article : Google Scholar
|
|
123
|
Wang Y, Sun Q, Mu N, Sun X, Wang Y, Fan S,
Su L and Liu X: The deubiquitinase USP22 regulates PD-L1
degradation in human cancer cells. Cell Commun Signal. 18:1122020.
View Article : Google Scholar
|
|
124
|
Huang X, Zhang Q, Lou Y, Wang J, Zhao X,
Wang L, Zhang X, Li S, Zhao Y, Chen Q, et al: USP22 Deubiquitinates
CD274 to Suppress Anticancer Immunity. Cancer Immunol Res.
7:1580–1590. 2019. View Article : Google Scholar
|
|
125
|
Li J, Yuan S, Norgard RJ, Yan F, Yamazoe
T, Blanco A and Stanger BZ: Tumor Cell-Intrinsic USP22 suppresses
antitumor immunity in pancreatic cancer. Cancer Immunol Res.
8:282–291. 2020. View Article : Google Scholar
|
|
126
|
Jingjing W, Wenzheng G, Donghua W, Guangyu
H, Aiping Z and Wenjuan W: Deubiquitination and stabilization of
programmed cell death ligand 1 by ubiquitin-specific peptidase 9,
X-linked in oral squamous cell carcinoma. Cancer Med. 7:4004–4011.
2018. View Article : Google Scholar
|
|
127
|
Naik E, Webster JD, DeVoss J, Liu J,
Suriben R and Dixit VM: Regulation of proximal T cell receptor
signaling and tolerance induction by deubiquitinase Usp9X. J Exp
Med. 211:1947–1955. 2014. View Article : Google Scholar
|
|
128
|
Sakaguchi S, Yamaguchi T, Nomura T and Ono
M: Regulatory T cells and immune tolerance. Cell. 133:775–787.
2008. View Article : Google Scholar
|
|
129
|
Wang F, Wang L, Wu J, Sokirniy I, Nguyen
P, Bregnard T, Weinstock J, Mattern M, Bezsonova I, Hancock WW and
Kumar S: Active site-targeted covalent irreversible inhibitors of
USP7 impair the functions of Foxp3+ T-regulatory cells by promoting
ubiquitination of Tip60. PLoS One. 12:e01897442017. View Article : Google Scholar
|
|
130
|
McHugh RS, Whitters MJ, Piccirillo CA,
Young DA, Shevach EM, Collins M and Byrne MC:
CD4(+)CD25(+) immunoregulatory T cells: Gene
expression analysis reveals a functional role for the
glucocorticoid-induced TNF receptor. Immunity. 16:311–323. 2002.
View Article : Google Scholar
|
|
131
|
Knee DA, Hewes B and Brogdon JL: Rationale
for anti-GITR cancer immunotherapy. Eur J Cancer. 67:1–10. 2016.
View Article : Google Scholar
|
|
132
|
Guo Y, Yang L, Lei S, Tan W and Long J:
NEDD4 Negatively Regulates GITR via ubiquitination in immune
microenvironment of melanoma. Onco Targets Ther. 12:10629–10637.
2019. View Article : Google Scholar
|
|
133
|
Trovato R, Fiore A, Sartori S, Canè S,
Giugno R, Cascione L, Paiella S, Salvia R, De Sanctis F, Poffe O,
et al: Immunosuppression by monocytic myeloid-derived suppressor
cells in patients with pancreatic ductal carcinoma is orchestrated
by STAT3. J Immunother Cancer. 7:2552019. View Article : Google Scholar
|
|
134
|
Song G, Zhang Y, Tian J, Ma J, Yin K, Xu H
and Wang S: TRAF6 regulates the immunosuppressive effects of
myeloid-derived suppressor cells in tumor-bearing host. Front
Immunol. 12:6490202021. View Article : Google Scholar
|
|
135
|
Zhang CX, Ye SB, Ni JJ, Cai TT, Liu YN,
Huang DJ, Mai HQ, Chen QY, He J, Zhang XS, et al: STING signaling
remodels the tumor microenvironment by antagonizing myeloid-derived
suppressor cell expansion. Cell Death Differ. 26:2314–2328. 2019.
View Article : Google Scholar
|
|
136
|
Chou FC, Chen HY, Kuo CC and Sytwu HK:
Role of galectins in tumors and in clinical immunotherapy. Int J
Mol Sci. 19:4302018. View Article : Google Scholar
|
|
137
|
Zhang CX, Huang DJ, Baloche V, Zhang L, Xu
JX, Li BW, Zhao XR, He J, Mai HQ, Chen QY, et al: Galectin-9
promotes a suppressive microenvironment in human cancer by
enhancing STING degradation. Oncogenesis. 9:652020. View Article : Google Scholar
|
|
138
|
Fang P, Li X, Dai J, Cole L, Camacho JA,
Zhang Y, Ji Y, Wang J, Yang XF and Wang H: Immune cell subset
differentiation and tissue inflammation. J Hematol Oncol.
11:972018. View Article : Google Scholar
|
|
139
|
Wang Y, Xiang Y, Xin VW, Wang XW, Peng XC,
Liu XQ, Wang D, Li N, Cheng JT, Lyv YN, et al: Dendritic cell
biology and its role in tumor immunotherapy. J Hematol Oncol.
13:1072020. View Article : Google Scholar
|
|
140
|
Bi E, Li R, Bover LC, Li H, Su P, Ma X,
Huang C, Wang Q, Liu L, Yang M, et al: E-cadherin expression on
multiple myeloma cells activates tumor-promoting properties in
plasmacytoid DCs. J Clin Invest. 128:4821–4831. 2018. View Article : Google Scholar
|
|
141
|
Hu Q, Ye Y, Chan LC, Li Y, Liang K, Lin A,
Egranov SD, Zhang Y, Xia W, Gong J, et al: Oncogenic lncRNA
down-regulates cancer cell antigen presentation and intrinsic tumor
suppression. Nat Immunol. 20:835–851. 2019. View Article : Google Scholar
|
|
142
|
Caraux A, Lu Q, Fernandez N, Riou S, Di
Santo JP, Raulet DH, Lemke G and Roth C: Natural killer cell
differentiation driven by Tyro3 receptor tyrosine kinases. Nat
Immunol. 7:747–754. 2006. View
Article : Google Scholar
|
|
143
|
Paolino M, Choidas A, Wallner S, Pranjic
B, Uribesalgo I, Loeser S, Jamieson AM, Langdon WY, Ikeda F, Fededa
JP, et al: The E3 ligase Cbl-b and TAM receptors regulate cancer
metastasis via natural killer cells. Nature. 507:508–512. 2014.
View Article : Google Scholar
|
|
144
|
Haglund K and Dikic I: The role of
ubiquitylation in receptor endocytosis and endosomal sorting. J
Cell Sci. 125(Pt 2): 265–275. 2012. View Article : Google Scholar
|
|
145
|
Wang Y, Zhang Y, Yi P, Dong W, Nalin AP,
Zhang J, Zhu Z, Chen L, Benson DM, Mundy-Bosse BL, et al: The
IL-15-AKT-XBP1s signaling pathway contributes to effector functions
and survival in human NK cells. Nat Immunol. 20:10–17. 2019.
View Article : Google Scholar
|
|
146
|
Dou Y, Xing J, Kong G, Wang G, Lou X, Xiao
X, Vivier E, Li XC and Zhang Z: Identification of the E3 Ligase
TRIM29 as a critical checkpoint regulator of NK cell functions. J
Immunol. 203:873–880. 2019. View Article : Google Scholar
|
|
147
|
Deng L, Meng T, Chen L, Wei W and Wang P:
The role of ubiquitination in tumorigenesis and targeted drug
discovery. Signal Transduct Target Ther. 5:112020. View Article : Google Scholar
|
|
148
|
Richardson PG, Hideshima T and Anderson
KC: Bortezomib (PS-341): A novel, first-in-class proteasome
inhibitor for the treatment of multiple myeloma and other cancers.
Cancer Control. 10:361–369. 2003. View Article : Google Scholar
|
|
149
|
Piva R, Ruggeri B, Williams M, Costa G,
Tamagno I, Ferrero D, Giai V, Coscia M, Peola S, Massaia M, et al:
CEP-18770: A novel, orally active proteasome inhibitor with a
tumor-selective pharmacologic profile competitive with bortezomib.
Blood. 111:2765–2775. 2008. View Article : Google Scholar
|
|
150
|
Anchoori RK, Karanam B, Peng S, Wang JW,
Jiang R, Tanno T, Orlowski RZ, Matsui W, Zhao M, Rudek MA, et al: A
bis-benzylidine piperidone targeting proteasome ubiquitin receptor
RPN13/ADRM1 as a therapy for cancer. Cancer Cell. 24:791–805. 2013.
View Article : Google Scholar
|
|
151
|
Song Y, Ray A, Li S, Das DS, Tai YT,
Carrasco RD, Chauhan D and Anderson KC: Targeting proteasome
ubiquitin receptor Rpn13 in multiple myeloma. Leukemia.
30:1877–1886. 2016. View Article : Google Scholar
|
|
152
|
Kimura K, Yamada T, Matsumoto M, Kido Y,
Hosooka T, Asahara S, Matsuda T, Ota T, Watanabe H, Sai Y, et al:
Endoplasmic reticulum stress inhibits STAT3-dependent suppression
of hepatic gluconeogenesis via dephosphorylation and deacetylation.
Diabetes. 61:61–73. 2012. View Article : Google Scholar
|
|
153
|
Soong RS, Anchoori RK, Yang B, Yang A,
Tseng SH, He L, Tsai YC, Roden RB and Hung CF: RPN13/ADRM1
inhibitor reverses immunosuppression by myeloid-derived suppressor
cells. Oncotarget. 7:68489–68502. 2016. View Article : Google Scholar
|
|
154
|
Yu GY, Wang X, Zheng SS, Gao XM, Jia QA,
Zhu WW, Lu L, Jia HL, Chen JH, Dong QZ, et al: RA190, a proteasome
subunit ADRM1 inhibitor, suppresses intrahepatic cholangiocarcinoma
by inducing NF-KB-Mediated cell apoptosis. Cell Physiol Biochem.
47:1152–1166. 2018. View Article : Google Scholar
|
|
155
|
Soong RS, Anchoori RK, Roden RBS, Cho RL,
Chen YC, Tseng SC, Huang YL, Liao PC and Shyu YC: Bis-benzylidine
Piperidone RA190 treatment of hepatocellular carcinoma via binding
RPN13 and inhibiting NF-κB signaling. BMC Cancer. 20:3862020.
View Article : Google Scholar
|
|
156
|
Powis G and Kirkpatrick L: Hypoxia
inducible factor-1alpha as a cancer drug target. Mol Cancer Ther.
3:647–654. 2004.
|
|
157
|
Lee YM, Kim GH, Park EJ, Oh TI, Lee S, Kan
SY, Kang H, Kim BM, Kim JH and Lim JH: Thymoquinone selectively
kills hypoxic renal cancer cells by suppressing HIF-1α-mediated
glycolysis. Int J Mol Sci. 20:10922019. View Article : Google Scholar
|
|
158
|
Ge Y, Yoon SH, Jang H, Jeong JH and Lee
YM: Decursin promotes HIF-1α proteasomal degradation and immune
responses in hypoxic tumour microenvironment. Phytomedicine.
78:1533182020. View Article : Google Scholar
|
|
159
|
Chen JJ, Ren YL, Shu CJ, Zhang Y, Chen MJ,
Xu J, Li J, Li AP, Chen DY, He JD, et al: JP3, an antiangiogenic
peptide, inhibits growth and metastasis of gastric cancer through
TRIM25/SP1/MMP2 axis. J Exp Clin Cancer Res. 39:1182020. View Article : Google Scholar
|
|
160
|
Chen Y, Huang Y, Huang Y, Xia X, Zhang J,
Zhou Y, Tan Y, He S, Qiang F, Li A, et al: JWA suppresses tumor
angiogenesis via Sp1-activated matrix metalloproteinase-2 and its
prognostic significance in human gastric cancer. Carcinogenesis.
35:442–451. 2014. View Article : Google Scholar
|
|
161
|
Godbersen JC, Humphries LA, Danilova OV,
Kebbekus PE, Brown JR, Eastman A and Danilov AV: The
Nedd8-activating enzyme inhibitor MLN4924 thwarts
microenvironment-driven NF-κB activation and induces apoptosis in
chronic lymphocytic leukemia B cells. Clin Cancer Res.
20:1576–1589. 2014. View Article : Google Scholar
|
|
162
|
Katsuya K, Hori Y, Oikawa D, Yamamoto T,
Umetani K, Urashima T, Kinoshita T, Ayukawa K, Tokunaga F and
Tamaru M: High-Throughput screening for linear ubiquitin chain
assembly complex (LUBAC) selective inhibitors using homogenous
time-resolved fluorescence (HTRF)-based assay system. SLAS Discov.
23:1018–1029. 2018. View Article : Google Scholar
|
|
163
|
Katsuya K, Oikawa D, Iio K, Obika S, Hori
Y, Urashima T, Ayukawa K and Tokunaga F: Small-molecule inhibitors
of linear ubiquitin chain assembly complex (LUBAC), HOIPINs,
suppress NF-κB signaling. Biochem Biophys Res Commun. 509:700–706.
2019. View Article : Google Scholar
|
|
164
|
Liu C, Billadeau DD, Abdelhakim H, Leof E,
Kaibuchi K, Bernabeu C, Bloom GS, Yang L, Boardman L, Shah VH and
Kang N: IQGAP1 suppresses TβRII-mediated myofibroblastic activation
and metastatic growth in liver. J Clin Invest. 123:1138–1156. 2013.
View Article : Google Scholar
|
|
165
|
Liu JL, Wang XY, Huang BX, Zhu F, Zhang RG
and Wu G: Expression of CDK5/p35 in resected patients with
non-small cell lung cancer: Relation to prognosis. Med Oncol.
28:673–678. 2011. View Article : Google Scholar
|
|
166
|
Gao L, Xia L, Ji W, Zhang Y, Xia W and Lu
S: Knockdown of CDK5 down-regulates PD-L1 via the
ubiquitination-proteasome pathway and improves antitumor immunity
in lung adenocarcinoma. Transl Oncol. 14:1011482021. View Article : Google Scholar
|
|
167
|
Liu Y, Liu X, Zhang N, Yin M, Dong J, Zeng
Q, Mao G, Song D, Liu L and Deng H: Berberine diminishes cancer
cell PD-L1 expression and facilitates antitumor immunity inhibiting
the deubiquitination activity of CSN5. Acta Pharm Sin B.
10:2299–2312. 2020. View Article : Google Scholar
|
|
168
|
Chauhan D, Tian Z, Nicholson B, Kumar KG,
Zhou B, Carrasco R, McDermott JL, Leach CA, Fulcinniti M, Kodrasov
MP, et al: A small molecule inhibitor of ubiquitin-specific
protease-7 induces apoptosis in multiple myeloma cells and
overcomes bortezomib resistance. Cancer Cell. 22:345–358. 2012.
View Article : Google Scholar
|
|
169
|
Nicholson B and Suresh Kumar KG: The
multifaceted roles of USP7: New therapeutic opportunities. Cell
Biochem Biophys. 60:61–68. 2011. View Article : Google Scholar
|
|
170
|
Shi L, Lin H, Li G, Sun Y, Shen J, Xu J,
Lin C, Yeh S, Cai X and Chang C: Cisplatin enhances NK cells
immunotherapy efficacy to suppress HCC progression via altering the
androgen receptor (AR)-ULBP2 signals. Cancer Lett. 373:45–56. 2016.
View Article : Google Scholar
|
|
171
|
Zhang X, Meng T, Cui S, Feng L, Liu D,
Pang Q and Wang P: Ubiquitination of nonhistone proteins in cancer
development and treatment. Front Oncol. 10:6212942021. View Article : Google Scholar
|
|
172
|
Ning B, Zhao W, Qian C, Liu P, Li Q, Li W
and Wang RF: USP26 functions as a negative regulator of cellular
reprogramming by stabilising PRC1 complex components. Nat Commun.
8:3492017. View Article : Google Scholar
|