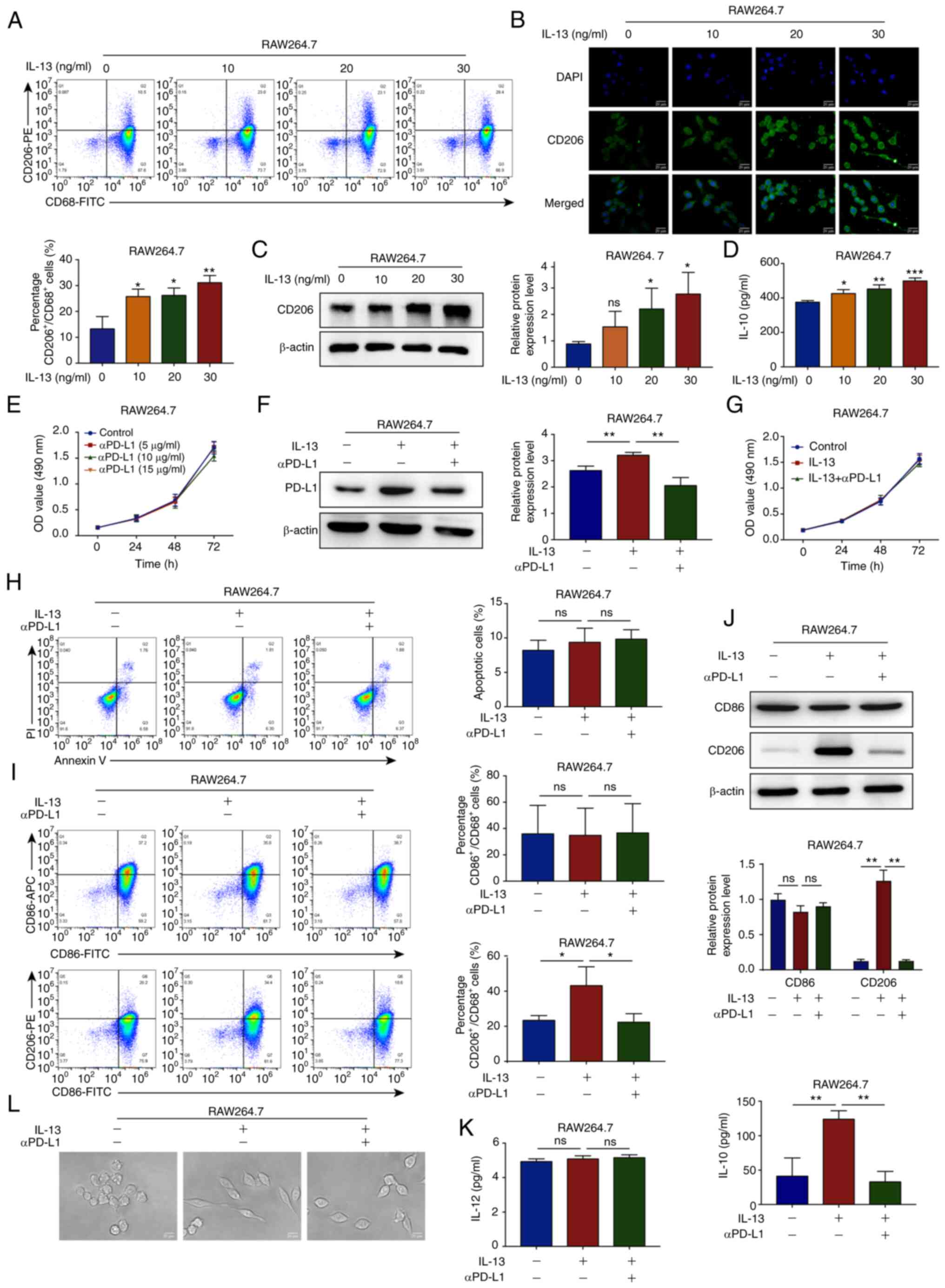
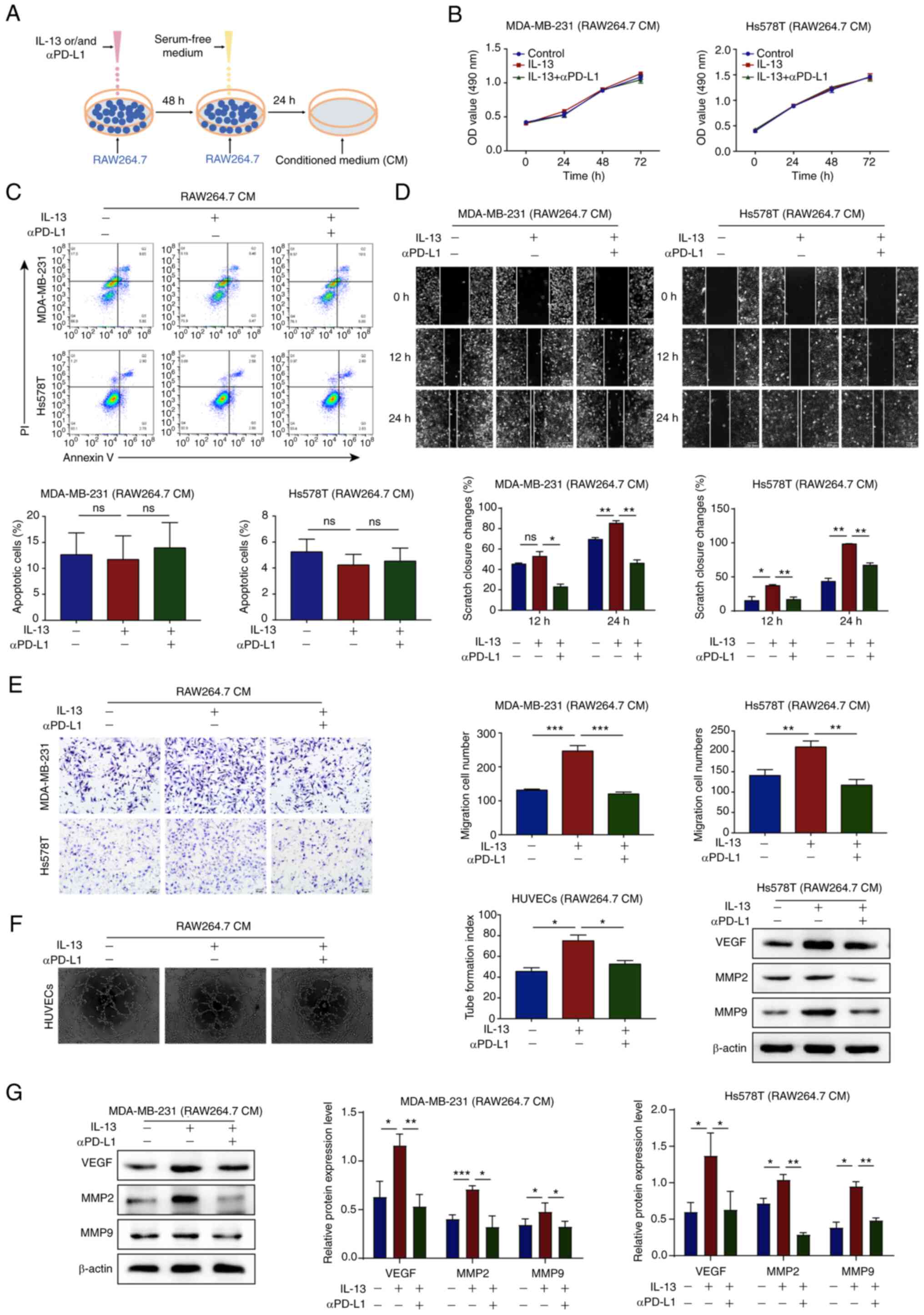
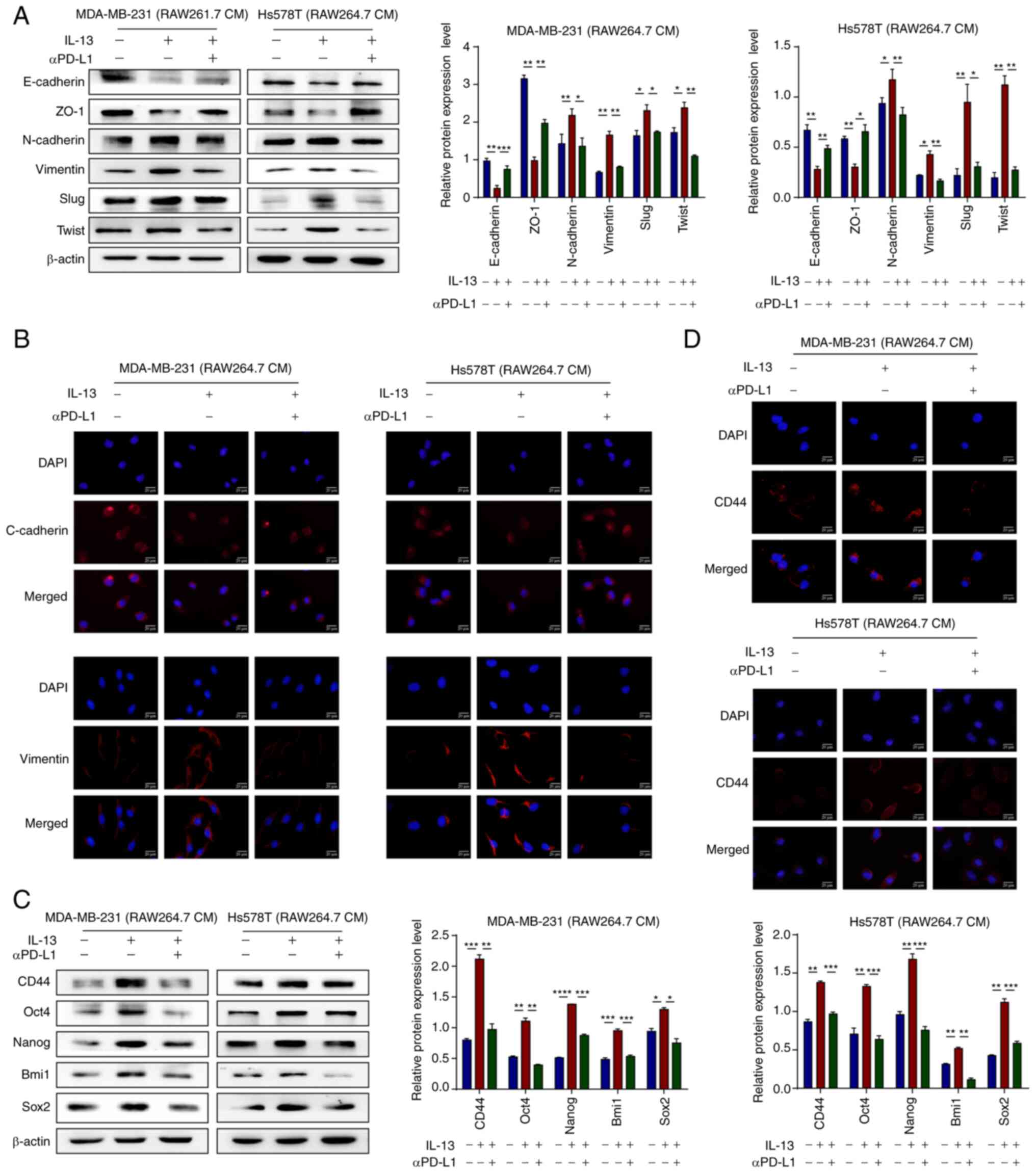
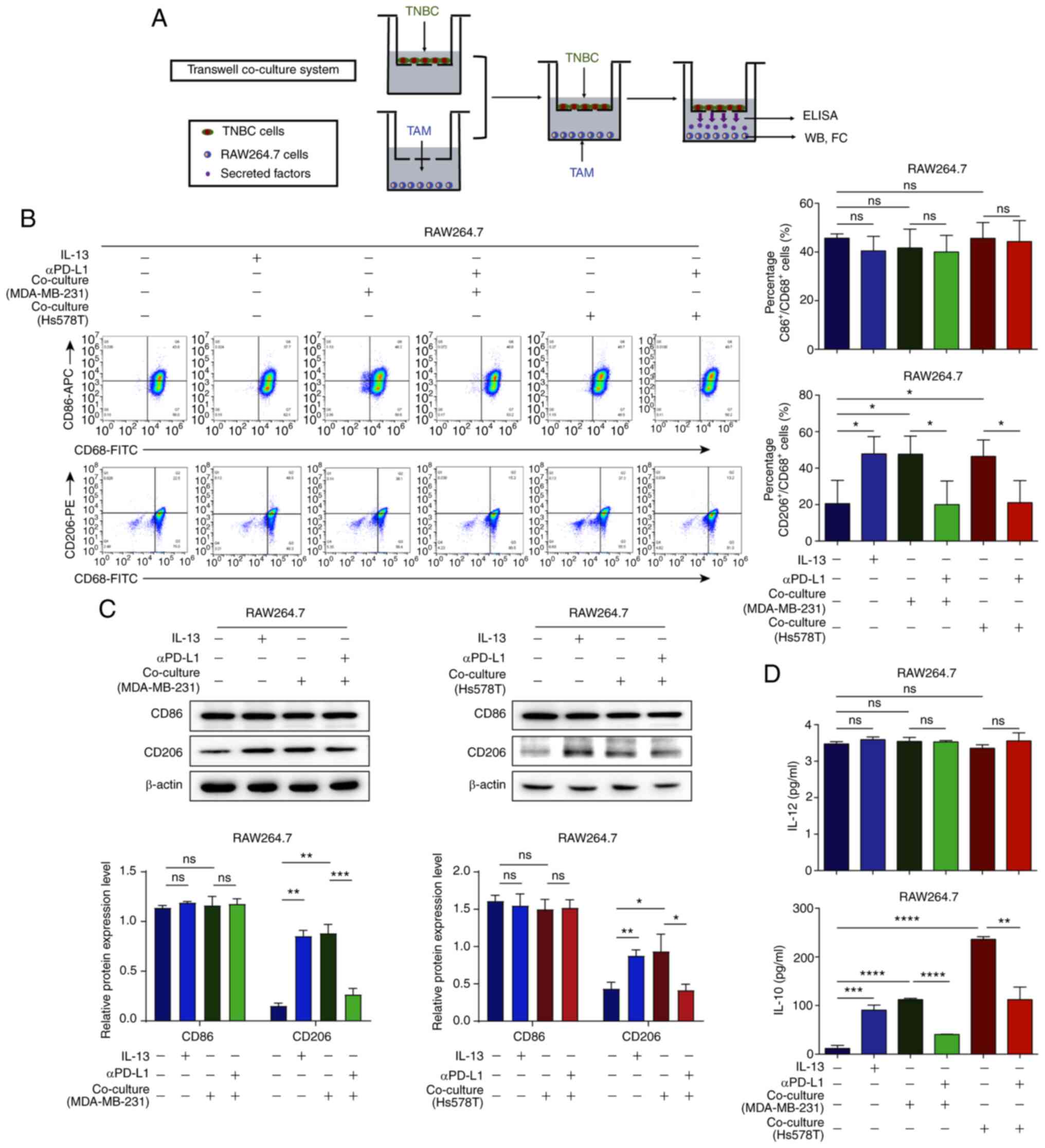
|
1
|
Zuo TT, Zheng RS, Zeng HM, Zhang SW and
Chen WQ: Female breast cancer incidence and mortality in China,
2013. Thorac Cancer. 8:214–218. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global Cancer Statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shaikh S and Rasheed A: Predicting
molecular subtypes of breast cancer with mammography and ultrasound
findings: Introduction of Sono-mammometry score. Radiol Res Pract.
2021:66919582021.PubMed/NCBI
|
|
4
|
Jiagge EM, Ulintz PJ, Wong S, McDermott
SP, Fossi SI, Suhan TK, Hoenerhoff MJ, Bensenhaver JM, Salem B,
Dziubinski M, et al: Multiethnic PDX models predict a possible
immune signature associated with TNBC of African ancestry. Breast
Cancer Res Treat. 186:391–401. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Manjunath M and Choudhary B:
Triple-negative breast cancer: A run-through of features,
classification and current therapies. Oncol Lett. 22:5122021.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li H, Fan X and Houghton J: Tumor
microenvironment: The role of the tumor stroma in cancer. J Cell
Biochem. 101:805–815. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Martinez-Reyes I and Chandel NS: Cancer
metabolism: Looking forward. Nat Rev Cancer. 21:669–680. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Emami F, Pathak S, Nguyen TT, Shrestha P,
Maharjan S, Kim JO, Jeong JH and Yook S: Photoimmunotherapy with
cetuximab-conjugated gold nanorods reduces drug resistance in
triple negative breast cancer spheroids with enhanced infiltration
of tumor-associated macrophages. J Control Release. 329:645–664.
2021. View Article : Google Scholar
|
|
9
|
Mantovani A, Marchesi F, Malesci A, Laghi
L and Allavena P: Tumour-associated macrophages as treatment
targets in oncology. Nat Rev Clin Oncol. 14:399–416. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Adorno-Cruz V, Hoffmann AD, Liu X,
Dashzeveg NK, Taftaf R, Wray B, Keri RA and Liu H: ITGA2 promotes
expression of ACLY and CCND1 in enhancing breast cancer stemness
and metastasis. Genes Dis. 8:493–508. 2021. View Article : Google Scholar :
|
|
11
|
Turajlic S and Swanton C: Metastasis as an
evolutionary process. Science. 352:169–175. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Deng Y, Hu JC, He SH, Lou B, Ding TB, Yang
JT, Mo MG, Ye DY, Zhou L, Jiang XC, et al: Sphingomyelin synthase 2
facilitates M2-like macrophage polarization and tumor progression
in a mouse model of triple-negative breast cancer. Acta Pharmacol
Sin. 42:149–159. 2021. View Article : Google Scholar :
|
|
13
|
Zhang XL, Hu LP, Yang Q, Qin WT, Wang X,
Xu CJ, Tian GA, Yang XM, Yao LL, Zhu L, et al: CTHRC1 promotes
liver metastasis by reshaping infiltrated macrophages through
physical interactions with TGF-β receptors in colorectal cancer.
Oncogene. 40:3959–3973. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tang C, Lei X, Xiong L, Hu Z and Tang B:
HMGA1B/2 transcriptionally activated-POU1F1 facilitates gastric
carcinoma metastasis via CXCL12/CXCR4 axis-mediated macrophage
polarization. Cell Death Dis. 12:4222021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pastushenko I and Blanpain C: EMT
transition states during tumor progression and metastasis. Trends
Cell Biol. 29:212–226. 2019. View Article : Google Scholar
|
|
16
|
Yang C, Dou R, Wei C, Liu K, Shi D, Zhang
C, Liu Q, Wang S and Xiong B: Tumor-derived exosomal
microRNA-106b-5p activates EMT-cancer cell and M2-subtype TAM
interaction to facilitate CRC metastasis. Mol Ther. 29:2088–2107.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Clevers H: The cancer stem cell: Premises,
promises and challenges. Nat Med. 17:313–319. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chen Y, Tan W and Wang C: Tumor-associated
macrophage-derived cytokines enhance cancer stem-like
characteristics through epithelial-mesenchymal transition. Onco
Targets Ther. 11:3817–3826. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Larionova I, Kazakova E, Gerashchenko T
and Kzhyshkowska J: New angiogenic regulators produced by TAMs:
Perspective for targeting tumor angiogenesis. Cancers (Basel).
13:32532021. View Article : Google Scholar
|
|
20
|
Long Y, Yu X, Chen R, Tong Y and Gong L:
Noncanonical PD-1/PD-L1 axis in relation to the efficacy of Anti-PD
therapy. Front Immunol. 13:9107042022. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gatalica Z, Snyder C, Maney T, Ghazalpour
A, Holterman DA, Xiao N, Overberg P, Rose I, Basu GD, Vranic S, et
al: Programmed cell death 1 (PD-1) and its ligand (PD-L1) in common
cancers and their correlation with molecular cancer type. Cancer
Epidemiol Biomarkers Prev. 23:2965–2970. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cimino-Mathews A, Foote JB and Emens LA:
Immune targeting in breast cancer. Oncology (Williston Park).
29:375–385. 2015.
|
|
23
|
Moser JC and Hu-Lieskovan S: Mechanisms of
resistance to PD-1 checkpoint blockade. Drugs. 80:459–465. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Noguchi T, Ward JP, Gubin MM, Arthur CD,
Lee SH, Hundal J, Selby MJ, Graziano RF, Mardis ER, Korman AJ and
Schreiber RD: Temporally Distinct PD-L1 expression by tumor and
host cells contributes to immune escape. Cancer Immunol Res.
5:106–117. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Juneja VR, McGuire KA, Manguso RT, LaFleur
MW, Collins N, Haining WN, Freeman GJ and Sharpe AH: PD-L1 on tumor
cells is sufficient for immune evasion in immunogenic tumors and
inhibits CD8 T cell cytotoxicity. J Exp Med. 214:895–904. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Deng L, Liang H, Burnette B, Beckett M,
Darga T, Weichselbaum RR and Fu YX: Irradiation and anti-PD-L1
treatment synergistically promote antitumor immunity in mice. J
Clin Invest. 124:687–695. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Fu LQ, Du WL, Cai MH, Yao JY, Zhao YY and
Mou XZ: The roles of tumor-associated macrophages in tumor
angiogenesis and metastasis. Cell Immunol. 353:1041192020.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang W, Zhao Y, Yao S, Cui X, Pan W, Huang
W, Gao J, Dong T and Zhang S: Nigericin inhibits epithelial ovarian
cancer metastasis by suppressing the cell cycle and
Epithelial-mesenchymal transition. Biochemistry (Mosc). 82:933–941.
2017. View Article : Google Scholar
|
|
29
|
Stankevicius V, Kunigenas L, Stankunas E,
Kuodyte K, Strainiene E, Cicenas J, Samalavicius NE and Suziedelis
K: The expression of cancer stem cell markers in human colorectal
carcinoma cells in a microenvironment dependent manner. Biochem
Biophys Res Commun. 484:726–733. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wylie PG and Bowen WP: Determination of
cell colony formation in a high-content screening assay. Clin Lab
Med. 27:193–199. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lawrence T and Natoli G: Transcriptional
regulation of macrophage polarization: Enabling diversity with
identity. Nat Rev Immunol. 11:750–761. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Neamatallah T: Mitogen-activated protein
kinase pathway: A critical regulator in Tumor-associated macrophage
polarization. J Microsc Ultrastruct. 7:53–56. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
LaPorte SL, Juo ZS, Vaclavikova J, Colf
LA, Qi X, Heller NM, Keegan AD and Garcia KC: Molecular and
structural basis of cytokine receptor pleiotropy in the
interleukin-4/13 system. Cell. 132:259–272. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Boutilier AJ and Elsawa SF: Macrophage
polarization states in the tumor microenvironment. Int J Mol Sci.
22:69952021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ma YY, He XJ, Wang HJ, Xia YJ, Wang SL, Ye
ZY and Tao HQ: Interaction of coagulation factors and
tumor-associated macrophages mediates migration and invasion of
gastric cancer. Cancer Sci. 102:336–342. 2011. View Article : Google Scholar
|
|
36
|
Sami E, Paul BT, Koziol JA and ElShamy WM:
The immunosuppressive microenvironment in BRCA1-IRIS-overexpressing
TNBC tumors is induced by bidirectional interaction with
tumor-associated macrophages. Cancer Res. 80:1102–1117. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Santoni M, Romagnoli E, Saladino T,
Foghini L, Guarino S, Capponi M, Giannini M, Cognigni PD, Ferrara G
and Battelli N: Triple negative breast cancer: Key role of
Tumor-associated macrophages in regulating the activity of
anti-PD-1/PD-L1 agents. Biochim Biophys Acta Rev Cancer.
1869:78–84. 2018. View Article : Google Scholar
|
|
38
|
Nasser MW, Wani NA, Ahirwar DK, Powell CA,
Ravi J, Elbaz M, Zhao H, Padilla L, Zhang X, Shilo K, et al: RAGE
mediates S100A7-induced breast cancer growth and metastasis by
modulating the tumor microenvironment. Cancer Res. 75:974–985.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Nieto MA, Huang RY, Jackson RA and Thiery
JP: EMT: 2016. Cell. 166:21–45. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Li B, Lu Y, Yu L, Han X, Wang H, Mao J,
Shen J, Wang B, Tang J, Li C and Song B: miR-221/222 Promote cancer
stem-like cell properties and tumor growth of breast cancer via
targeting PTEN and sustained Akt/NF-κB/COX-2 activation. Chem Biol
Interact. 277:33–42. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhou Y, Chen D, Qi Y, Liu R, Li S, Zou H,
Lan J, Ju X, Jiang J, Liang W, et al: Evaluation of expression of
cancer stem cell markers and fusion gene in synovial sarcoma:
Insights into histogenesis and pathogenesis. Oncol Rep.
37:3351–3360. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wu T, Tang C, Tao R, Yong X, Jiang Q and
Feng C: PD-L1-mediated immunosuppression in oral squamous cell
carcinoma: Relationship with macrophage infiltration and epithelial
to mesenchymal transition markers. Front Immunol. 12:6938812021.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Samples J, Willis M and Klauber-Demore N:
Targeting angiogenesis and the tumor microenvironment. Surg Oncol
Clin N Am. 22:629–639. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Barnett FH, Rosenfeld M, Wood M, Kiosses
WB, Usui Y, Marchetti V, Aguilar E and Friedlander M: Macrophages
form functional vascular mimicry channels in vivo. Sci Rep.
6:366592016. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Dong R, Gong Y, Meng W, Yuan M, Zhu H,
Ying M, He Q, Cao J and Yang B: The involvement of M2 macrophage
polarization inhibition in fenretinide-mediated chemopreventive
effects on colon cancer. Cancer Lett. 388:43–53. 2017. View Article : Google Scholar
|
|
46
|
Wang H, Yung MMH, Ngan HYS, Chan KKL and
Chan DW: The impact of the tumor microenvironment on macrophage
polarization in cancer metastatic progression. Int J Mol Sci.
22:65602021. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Watroba S, Wisniowski T, Bryda J and
Kurzepa J: The role of matrix metalloproteinases in pathogenesis of
human bladder cancer. Acta Biochim Pol. 68:547–555. 2021.PubMed/NCBI
|
|
48
|
Veremeyko T, Yung AWY, Anthony DC,
Strekalova T and Ponomarev ED: Early growth response Gene-2 is
essential for M1 and M2 macrophage activation and plasticity by
modulation of the transcription factor CEBPβ. Front Immunol.
9:25152018. View Article : Google Scholar
|
|
49
|
Xu J, Zhang J, Zhang Z, Gao Z, Qi Y, Qiu
W, Pan Z, Guo Q, Li B, Zhao S, et al: Hypoxic glioma-derived
exosomes promote M2-like macrophage polarization by enhancing
autophagy induction. Cell Death Dis. 12:3732021. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Qian M, Wang S, Guo X, Wang J, Zhang Z,
Qiu W, Gao X, Chen Z, Xu J, Zhao R, et al: Hypoxic glioma-derived
exosomes deliver microRNA-1246 to induce M2 macrophage polarization
by targeting TERF2IP via the STAT3 and NF-κB pathways. Oncogene.
39:428–442. 2020. View Article : Google Scholar
|