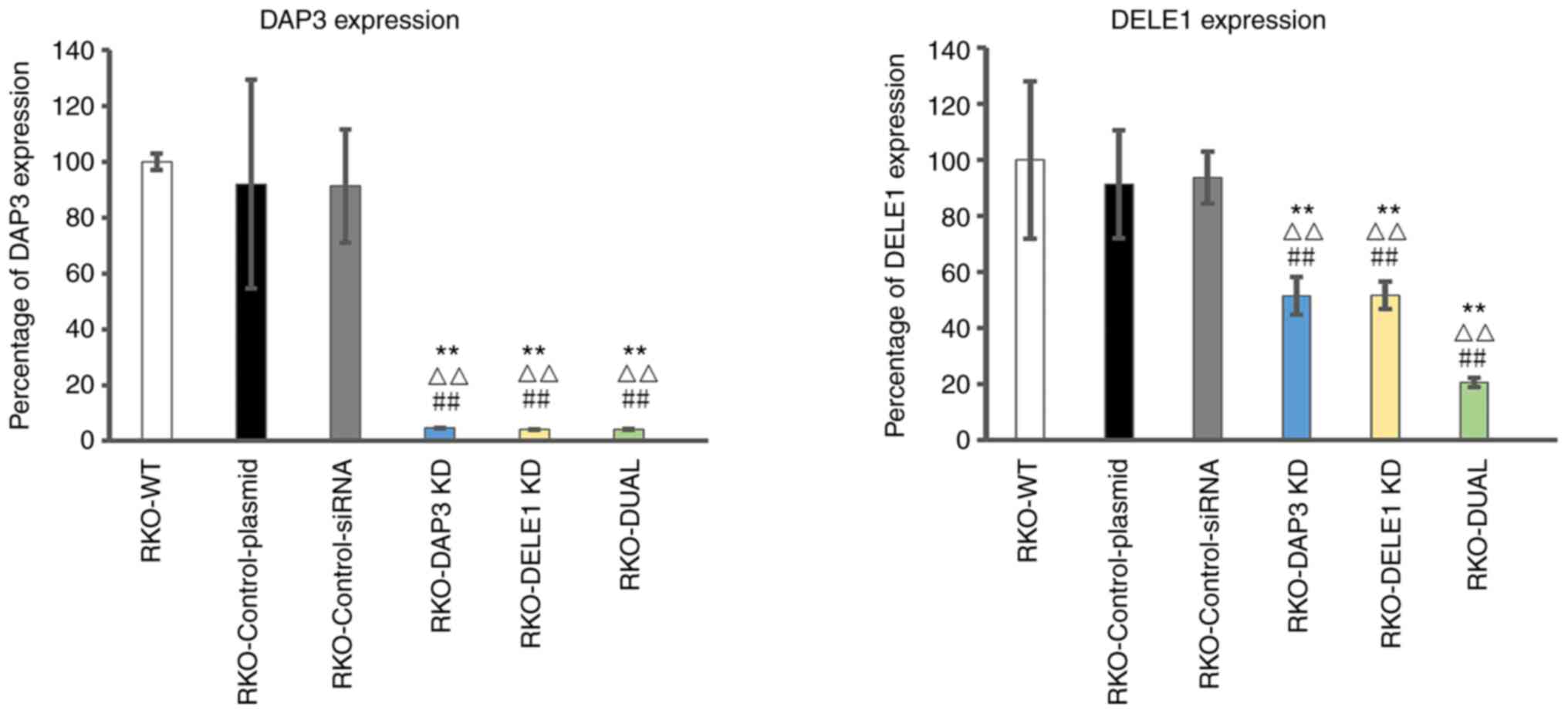
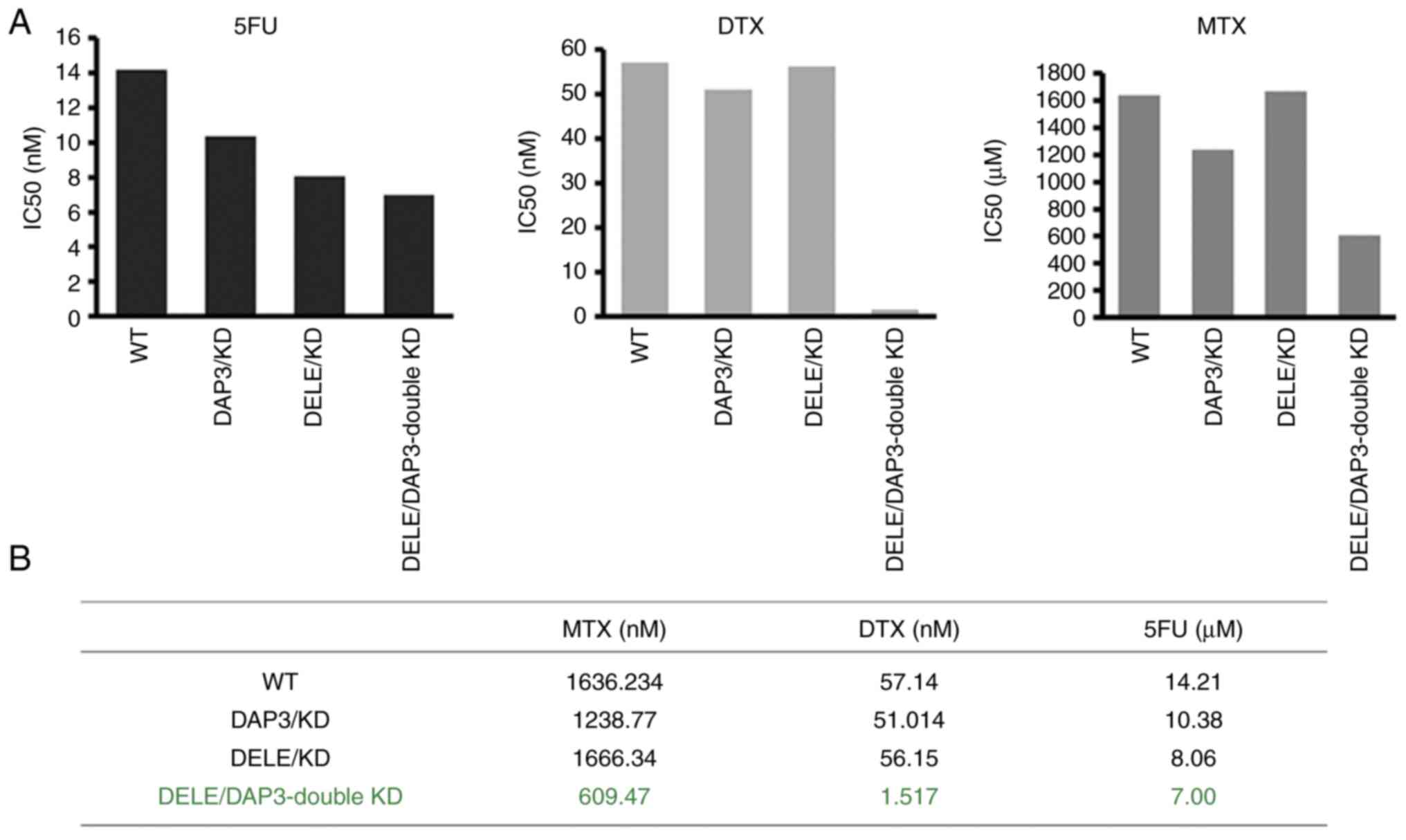
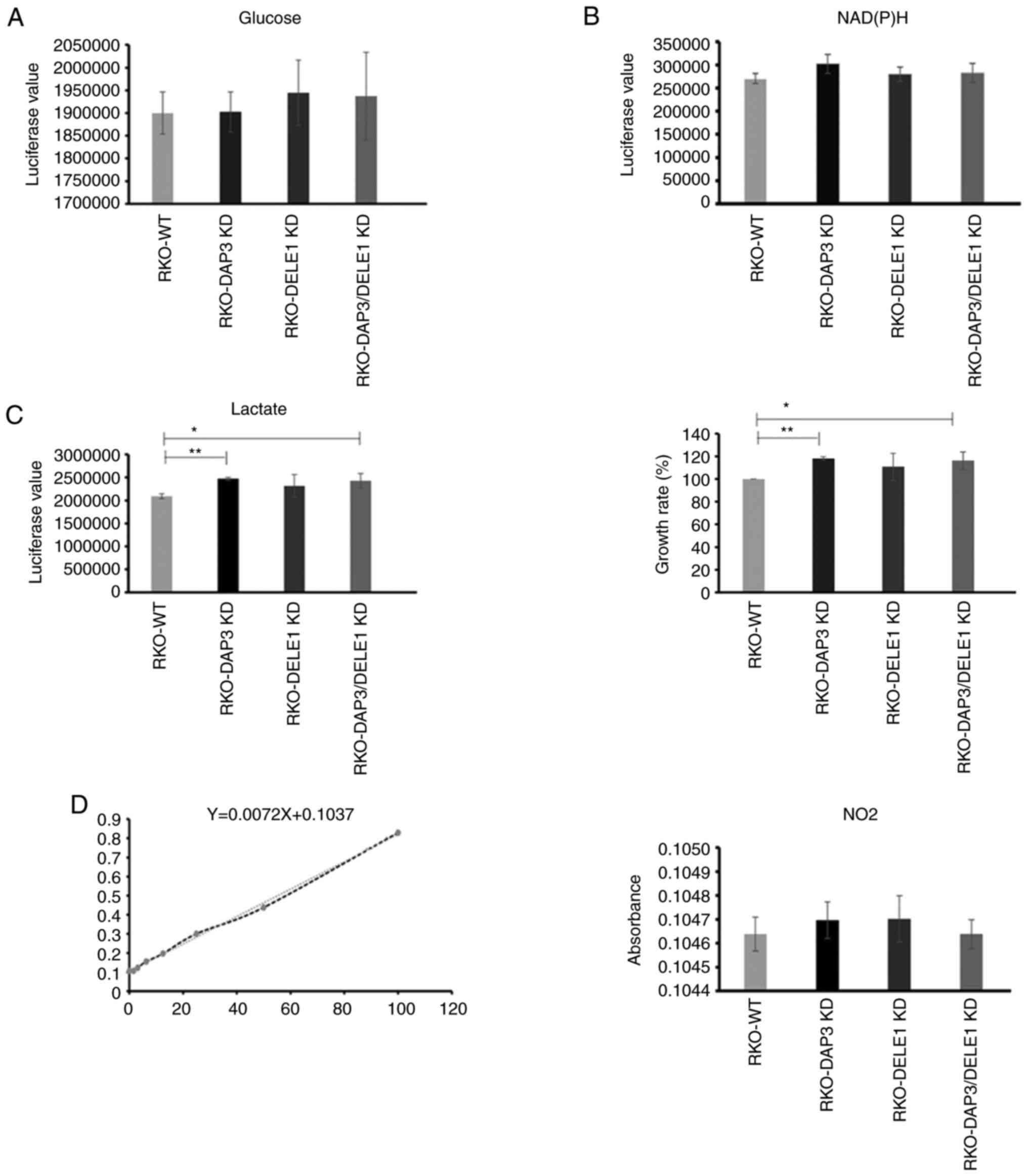
|
1
|
Deiss LP, Feinstein E, Berissi H, Cohen O
and Kimchi A: Identification of a novel serine/threonine kinase and
a novel 15-kD protein as potential mediators of the gamma
interferon-induced cell death. Genes Dev. 9:15–30. 1995.
|
|
2
|
Kissil JL, Deiss LP, Bayewitch M, Raveh T,
Khaspekov G and Kimchi A: Isolation of DAP3, a novel mediator of
interferon-gamma-induced cell death. J Biol Chem. 270:27932–27936.
1995.
|
|
3
|
Koren I, Reem E and Kimchi A: DAP1, a
novel substrate of mTOR, negatively regulates autophagy. Curr Biol.
20:1093–1098. 2010.
|
|
4
|
Wazir U, Jiang WG, Sharma AK and Mokbel K:
The mRNA expression of DAP1 in human breast cancer: Correlation
with clinicopathological parameters. Cancer Genomics Proteomics.
9:199–201. 2012.
|
|
5
|
Wybranska I, Polus A, Mikolajczyk M, Knapp
A, Sliwa A, Zapala B, Staszel T and Dembinska-Kiec A:
Apoptosis-related gene expression in glioblastoma (LN-18) and
medulloblastoma (Daoy) cell lines. Hum Cell. 26:137–148. 2013.
|
|
6
|
Sui L, Ye L, Sanders AJ, Yang Y, Hao C,
Hargest R and Jiang WG: Expression of death associated proteins
DAP1 and DAP3 in human pancreatic cancer. Anticancer Res.
41:2357–2362. 2021.
|
|
7
|
Miyazaki T, Shen M, Fujikura D, Tosa N,
Kim HR, Kon S, Uede T and Reed JC: Functional role of
death-associated protein 3 (DAP3) in anoikis. J Biol Chem.
279:44667–44672. 2004.
|
|
8
|
Takeda S, Iwai A, Nakashima M, Fujikura D,
Chiba S, Li HM, Uehara J, Kawaguchi S, Kaya M, Nagoya S, et al:
LKB1 is crucial for TRAIL-mediated apoptosis induction in
osteosarcoma. Anticancer Res. 27:761–768. 2007.
|
|
9
|
Mariani L, Beaudry C, McDonough WS,
Hoelzinger DB, Kaczmarek E, Ponce F, Coons SW, Giese A, Seiler RW
and Berens ME: Death-associated protein 3 (Dap-3) is overexpressed
in invasive glioblastoma cells in vivo and in glioma cell lines
with induced motility phenotype in vitro. Clin Cancer Res.
7:2480–2489. 2001.
|
|
10
|
Sasaki H, Ide N, Yukiue H, Kobayashi Y,
Fukai I, Yamakawa Y and Fujii Y: Arg and DAP3 expression was
correlated with human thymoma stage. Clin Exp Metastasis.
21:507–513. 2004.
|
|
11
|
Davidsson J, Andersson A, Paulsson K,
Heidenblad M, Isaksson M, Borg A, Heldrup J, Behrendtz M,
Panagopoulos I, Fioretos T and Johansson B: Tiling resolution array
comparative genomic hybridization, expression and methylation
analyses of dup(1q) in Burkitt lymphomas and pediatric high
hyperdiploid acute lymphoblastic leukemias reveal clustered
near-centromeric breakpoints and overexpression of genes in
1q22-32.3. Hum Mol Genet. 16:2215–2225. 2007.
|
|
12
|
Jia Y, Ye L, Ji K, Zhang L, Hargest R, Ji
J and Jiang WG: Death-associated protein-3, DAP-3, correlates with
preoperative chemotherapy effectiveness and prognosis of gastric
cancer patients following perioperative chemotherapy and radical
gastrectomy. Br J Cancer. 110:421–429. 2014.
|
|
13
|
Wazir U, Jiang WG, Sharma AK and Mokbel K:
The mRNA expression of DAP3 in human breast cancer: Correlation
with clinicopathological parameters. Anticancer Res. 32:671–674.
2012.
|
|
14
|
Sato Y, Yoshino H, Kashiwakura I and
Tsuruga E: DAP3 is involved in modulation of cellular radiation
response by RIG-I-Like receptor agonist in human lung
adenocarcinoma cells. Int J Mol Sci. 22:4202021.
|
|
15
|
Gressner O, Schilling T, Lorenz K, Schulze
Schleithoff E, Koch A, Schulze-Bergkamen H, Lena AM, Candi E,
Terrinoni A, Catani MV, et al: TAp63alpha induces apoptosis by
activating signaling via death receptors and mitochondria. EMBO J.
24:2458–2471. 2005.
|
|
16
|
Woo Lee J, Hwa Soung Y, Young Kim S, Woo
Nam S, Sang Park W, Young Lee J, Jin Yoo N and Hyung Lee S:
Mutational analysis of proapoptotic death associated protein 3
(DAP3) P-loop domain in common human carcinomas. Acta Oncol.
45:489–490. 2006.
|
|
17
|
Han J, An O, Ren X, Song Y, Tang SJ, Shen
H, Ke X, Ng VH, Tay DJ, Tan HQ, et al: Multilayered control of
splicing regulatory networks by DAP3 leads to widespread
alternative splicing changes in cancer. Nat Commun.
13:17932022.
|
|
18
|
Harada T, Iwai A and Miyazaki T:
Identification of DELE, a novel DAP3-binding protein which is
crucial for death receptor-mediated apoptosis induction. Apoptosis.
15:1247–1255. 2010.
|
|
19
|
Fessler E, Eckl EM, Schmitt S, Mancilla
IA, Meyer-Bender MF, Hanf M, Philippou-Massier J, Krebs S, Zischka
H and Jae LT: A pathway coordinated by DELE1 relays mitochondrial
stress to the cytosol. Nature. 579:433–437. 2020.
|
|
20
|
Alavi MV: OMA1-An integral membrane
protease? Biochim Biophys Acta Proteins Proteom.
1869:1405582021.
|
|
21
|
Guo X, Aviles G, Liu Y, Tian R, Unger BA,
Lin YT, Wiita AP, Xu K, Correia MA and Kampmann M: Mitochondrial
stress is relayed to the cytosol by an OMA1-DELE1-HRI pathway.
Nature. 579:427–432. 2020.
|
|
22
|
Xiao L, Xian H, Lee KY, Xiao B, Wang H, Yu
F, Shen HM and Liou YC: Death-associated protein 3 regulates
mitochondrial-encoded protein synthesis and mitochondrial dynamics.
J Biol Chem. 290:24961–24974. 2015.
|
|
23
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249. 2021.
|
|
24
|
Lee S, Zhang S, Ma C, Ou FS, Wolfe EG,
Ogino S, Niedzwiecki D, Saltz LB, Mayer RJ, Mowat RB, et al: Race,
income, and survival in stage III colon cancer: CALGB 89803
(Alliance). JNCI Cancer Spectr. 5:pkab0342021.
|
|
25
|
Nazarenko IA, Bhatnagar SK and Hohman RJ:
A closed tube format for amplification and detection of DNA based
on energy transfer. Nucleic Acids Res. 25:2516–2521. 1997.
|
|
26
|
Jiang WG, Watkins G, Lane L, Cunnick GH,
Douglas-Jones A, Mokbel K and Mansel RE: Prognostic value of rho
GTPases and rho guanine nucleotide dissociation inhibitors in human
breast cancers. Clin Cancer Res. 9:6432–6440. 2003.
|
|
27
|
Chae SY, Lee M, Kim SW and Bae YH:
Protection of insulin secreting cells from nitric oxide induced
cellular damage by crosslinked hemoglobin. Biomaterials.
25:843–850. 2004.
|
|
28
|
Zhou Y, Xu B, Zhou Y, Liu J, Zheng X, Liu
Y, Deng H, Liu M, Ren X, Xia J, et al: Identification of key genes
with differential correlations in lung adenocarcinoma. Front Cell
Dev Biol. 9:6754382021.
|
|
29
|
Wazir U, Sanders AJ, Wazir AM, Ye L, Jiang
WG, Ster IC, Sharma AK and Mokbel K: Effects of the knockdown of
death-associated protein 3 expression on cell adhesion, growth and
migration in breast cancer cells. Oncol Rep. 33:2575–2582.
2015.
|
|
30
|
Guerra F, Arbini AA and Moro L:
Mitochondria and cancer chemoresistance. Biochim Biophys Acta
Bioenerg. 1858:686–699. 2017.
|
|
31
|
Cheng M, Yu H, Kong Q, Wang B, Shen L,
Dong D and Sun L: The mitochondrial PHB2/OMA1/DELE1 pathway
cooperates with endoplasmic reticulum stress to facilitate the
response to chemotherapeutics in ovarian cancer. Int J Mol Sci.
23:13202022.
|
|
32
|
Porporato PE, Filigheddu N, Pedro JM,
Kroemer G and Galluzzi L: Mitochondrial metabolism and cancer. Cell
Res. 28:265–280. 2018.
|
|
33
|
Missiroli S, Perrone M, Genovese I, Pinton
P and Giorgi C: Cancer metabolism and mitochondria: Finding novel
mechanisms to fight tumours. EBioMedicine. 59:1029432020.
|
|
34
|
Shin YK, Yoo BC, Chang HJ, Jeon E, Hong
SH, Jung MS, Lim SJ and Park JG: Down-regulation of mitochondrial
F1F0-ATP synthase in human colon cancer cells with induced
5-fluorouracil resistance. Cancer Res. 65:3162–3170. 2005.
|
|
35
|
Vellinga TT, Borovski T, de Boer VC,
Fatrai S, van Schelven S, Trumpi K, Verheem A, Snoeren N, Emmink
BL, Koster J, et al: SIRT1/PGC1α-dependent increase in oxidative
phosphorylation supports chemotherapy resistance of colon cancer.
Clin Cancer Res. 21:2870–2879. 2015.
|
|
36
|
Kluza J, Jendoubi M, Ballot C, Dammak A,
Jonneaux A, Idziorek T, Joha S, Dauphin V, Malet-Martino M,
Balayssac S, et al: Exploiting mitochondrial dysfunction for
effective elimination of imatinib-resistant leukemic cells. PLoS
One. 6:e219242011.
|
|
37
|
Li J, Zhao S, Zhou X, Zhang T, Zhao L,
Miao P, Song S, Sun X, Liu J, Zhao X and Huang G: Inhibition of
lipolysis by mercaptoacetate and etomoxir specifically sensitize
drug-resistant lung adenocarcinoma cell to paclitaxel. PLoS One.
8:e746232013.
|
|
38
|
Chen AN, Luo Y, Yang YH, Fu JT, Geng XM,
Shi JP and Yang J: Lactylation, a novel metabolic reprogramming
code: Current status and prospects. Front Immunol.
12:6889102021.
|