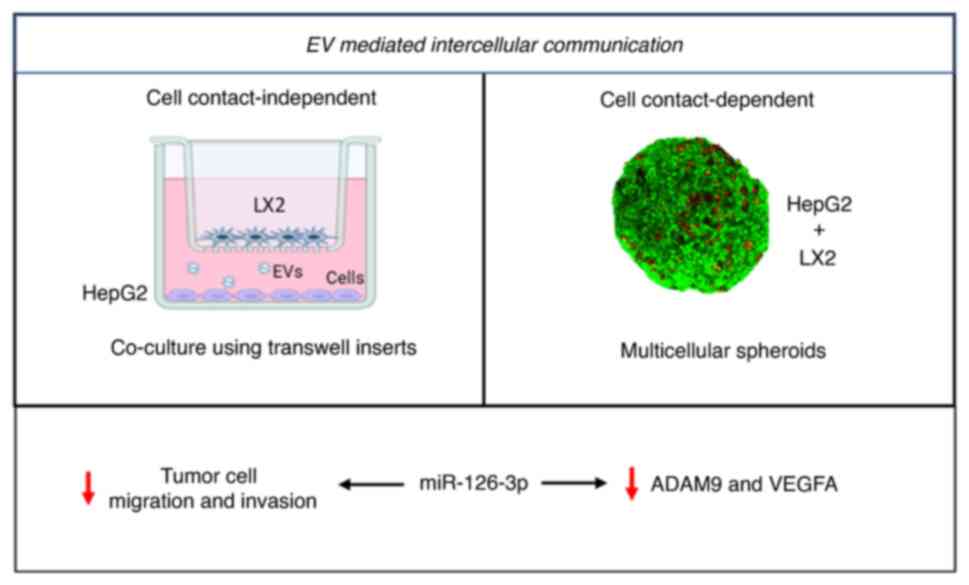
Introduction
The contribution of the local microenvironment to
tumor cell behavior and tumor progression is well recognized. The
tumor microenvironment (TME) is comprised of diverse cells,
including stromal cells such as fibroblasts, myofibroblasts,
endothelial cells, pericytes, adipose cells, immune and
inflammatory cells, and extracellular matrix (ECM) (1,2).
Stromal components within the local microenvironment may constrain
tumor growth during early stages of tumor formation or may promote
tumor growth in later stages. Stromal cells such as fibroblasts and
mesenchymal stem cells are critical contributors to tumor growth.
Interactions between tumor and stromal cells are mediated by
soluble factors with growth-promoting or -inhibitory signals,
cytokines or growth factors (3-5).
Liver cancer (LC) is the most common tumor type of
the liver and is a leading cause of cancer-related death worldwide
(6,7). LC frequently develops in the setting
of chronic liver injury accompanied by a fibrotic milieu that
arises from injury or inflammation-related activation of hepatic
stellate cells (HSCs) (3). After
activation, HSCs may trans-differentiate into myofibroblast-like
cells and may proliferate, acquire contractile properties, secrete
inflammatory cytokines, increase α-smooth muscle actin (α-SMA)
expression and produce ECM proteins, including collagen (3,8). An
active participation of stromal cells within the TME may contribute
to the initiation and maintenance of carcinogenesis (5). For instance, interaction between LC
cells and activated HSCs creates a permissive pro-angiogenic
microenvironment with enhanced vascular endothelial growth factor A
(VEGF-A) and matrix metalloprotease 9 (MMP9) production that
increases migration and growth of the cancer cells (8,9).
Understanding the mechanisms by which tumor and
stromal cells interact may provide valuable information regarding
the role of tumor-stromal interactions in LC growth. The release of
extracellular vesicles (EVs) has been recognized as a potential
mechanism for intercellular communication (10,11).
EVs are a heterogeneous group of cell-derived vesicles, including
exosomes (50-150 nm in size and of endosomal origin) (12) and microvesicles (100-1,000 nm in
size and directly budding from the plasma membrane) (12,13),
containing diverse biomolecules such as lipids, RNA, DNA and
proteins, some of which may be capable of eliciting functional
biological effects following their uptake by recipient cells
(10,11,13).
Thus, EV-mediated transfer may contribute to cell-to-cell
communication in the local environment or at distant sites
(14,15). EVs may be released and taken up by
tumor cells as well as by stromal cells (12). However, the contribution and role
of EVs as a mediator of intercellular signaling in the TME remains
to be fully elucidated.
EVs have been implicated in suppressing immune
responses, supporting endothelial angiogenesis and transforming
benign cells, thus helping to establish a pre-metastatic niche
(14). Furthermore, the RNAs
encapsulated inside the EVs are protected from RNase-A, providing a
consistent source that may regulate gene expression in the
recipient cells and thus altering the TME (16). Even though several EV-RNAs have
been identified to be present or enriched within EVs from LC cells,
or implicated in LC, e.g. microRNA (miRNA/miR)-192, miR-122 and
long non-coding RNA-regulator of reprogramming their contributions
to intercellular interactions within the TME remain to be
elucidated (13). Thus, the aims
of the present study were to evaluate the role of EVs and the
functional contribution of EV RNA to intercellular communications
between tumor and stromal cells in the liver TME. In addition, the
present study sought to identify EV-RNAs that may serve as
biomarkers of TME interactions and potential targets for liver
cancer.
Materials and methods
Cells and cell culture
HepG2 human LC cells were obtained from the American
Type Culture collection and cells were authenticated by short
tandem repeat profiling. The human HSC line LX2 was generated by Xu
et al (17) and kindly
provided by Dr Gregory J Gores (Division of Gastroenterology and
Hepatology, Mayo Clinic, Rochester, Minnesota, USA). The cell lines
were maintained in DMEM (Hyclone; Cytiva) supplemented with 10% FBS
(Gemini Bio Products) and 1% penicillin-streptomycin (Gemini Bio
Products) for individual cell culture. For co-culture studies,
HepG2 (4×105) and LX2 (1×105) were seeded on
cell-culture inserts (Falcon 0.4 µm pore size cell culture
insert; BD Biosciences) in EV-depleted complete media in a
humidified incubator at 37°C with 5% CO2.
Reagents
Precursor hsa-miR-126-3p (mature miRNA sequence of
miR-126-3p: 5′-UCG UAC CGU GAG UAA UAA UGC G-3′; cat. no. AM17100;
assay ID: PM12841), negative control miRNA precursor (cat. no.
AM17110), antimiR-126-3p (cat. no. AM17000; assay ID: AM12841) and
negative control antimiRNA inhibitor (cat. no. AM17010) were
purchased from Ambion (Thermo Fisher Scientific, Inc.).
Lipofectamine® 3000 was purchased from Thermo Fisher
Scientific, Inc. Regorafenib and Sorafenib were obtained from
Selleck Chemicals.
3D co-culture spheroid model
3D co-culture spheroids were generated from
combining HepG2 and LX2 cells at a 4:1 ratio and using
methylcellulose (18). After
reaching 70% confluency, cells were trypsinized. The concentration
of cells in suspension was determined and they were diluted with
complete media to a concentration of 1×104
cells/µl. Six-well tissue culture plates were coated with
poly-2-hydroxyethyl methacrylate (pHEMA) to prepare a non-adherent
surface. 2 µl of cell suspension was injected in a 3% w/v
methylcellulose (MilliporeSigma; Merck KGaA) matrix to generate
spheroids with 2×104 cells each. After 3 days, the
spheroids were manually retrieved from the methylcellulose matrix
and cultured in fresh DMEM-high glucose (HG) media for further
study.
Live/dead cell imaging in 3D co-culture
spheroid model
At day 3 of 3D co-culture in methylcellulose, the
spheroids were retrieved in complete DMEM-HG media for live/dead
cell imaging. The harvested spheroids were then stained in a 2
µM of calcein AM and 2 µM of ethidium bromide
homodimer mixture for 5 min at room temperature, followed by a wash
in PBS. The stained spheroids were observed under an LSM880
confocal laser scanning microscope (Carl Zeiss AG) (18).
Histological and immunohistochemical
analysis
The histological analysis was performed as per a
previously described protocol. In brief, harvested spheroids were
fixed in 4% paraformaldehyde for 15 min at room temperature,
followed by sectioning. For morphological analysis, spheroids
sections were stained with hematoxylin and eosin (H&E)
(18). Furthermore, α-SMA staining
in spheroids was performed as per a previous protocol (19). Immunohistochemical staining was
performed using anti α-SMA rabbit polyclonal antibody (cat no.
Ab5694; 1:200 dilution; Abcam) and horseradish peroxidase labeled
goat anti-Rabbit antibody (cat. no. AB_2630375; Dako; Agilent
Technologies, Inc.) along with 3,3-diaminobenzidine (Dako; Agilent
Technologies, Inc.).
miRNA transfection of 2D cells and 3D
co-culture spheroids
For 2D culture, HepG2 cells were seeded in 6-well
plates (5×105/well) in 2 ml complete media and
transfected with 100 nM of precursor or antimiR-126-3p using
Lipofectamine® 3000 in OptiMEM (Thermo Fisher
Scientific, Inc.) at 70-80% confluency. After 24 h, the OptiMEM was
replaced with complete media. For co-culture of HepG2+LX2 cells as
3D spheroids, 25 spheroids per group were transferred with 500
µl OptiMEM media into a 24-well plate precoated with pHEMA.
Spheroids were transfected with 200 nM of miR-126-3p precursor and
antimiR-126-3p or respective negative control. After 24 h, the
media was replaced with 500 ml of complete media. The spheroids
were collected for further experiments 48 h after transfection.
EV isolation and characterization
HepG2 EVs were isolated by sequential
ultracentrifugation and the pellets were re-suspended in PBS, as
described in a previous study by our group (20). The EV concentration was determined
by nanoparticle tracking analysis (NTA) using a Nanosight LM10
(Nanosight Ltd.). In brief, the solution of diluted EVs was loaded
into the LM10 instrument and a series of three one-minute videos
were captured of different fields of view. The average particle
concentration and size parameters were calculated.
RNA isolation and sequencing
Total cellular RNA was isolated using TRIzol (Thermo
Fisher Scientific, Inc.). For isolation of EV-RNA, cells were
cultured in monoculture or 2D co-culture. After 48 h, the media was
removed and centrifuged at 3,000 × g for 15 min, EVs were isolated
using ExoQuick-TC (System Biosciences, LLC) and EV-RNA was isolated
using SeraMir (Systems Biosciences, LLC). The final concentration
of the cellular RNA or EV-RNA was measured using a NanoDrop 2000c
Spectrophotometer (Thermo Fisher Scientific, Inc.). RNA sequencing
was performed by Exiqon (Qiagen Sciences, Inc.).
Generation of cDNA
Isolated RNAs were first treated with DNase. Reverse
transcription (RT) was performed using a TaqMan MicroRNA RT kit
(Applied Biosystems; Thermo Fisher Scientific, Inc.) using 100 ng
total RNA in a reaction mixture containing 0.15 µl 100 mM
dNTPs, 1 µl reverse transcriptase, 1.5 µl 10X RT
buffer, 0.19 µl RNase inhibitor and 3 µl of 5X RT
primer specific for miR-126-3p (cat. no. 4427975; assay ID: 002228;
Ambion; Thermo Fisher Scientific, Inc.). U6snRNA (cat. no. 4427975;
assay ID: 001973; Ambion; Thermo Fisher Scientific, Inc.) was used
as an internal control for the normalization of miR-126-3p
expression in the experimental groups. The reactions were performed
using a MyCycler™ Thermal Cycler (Bio-Rad Laboratories, Inc.) for
30 min at 16°C, 30 min at 42°C and 5 min at 85°C, followed by a
hold at 4°C.
Droplet digital PCR (ddPCR)
Synthesized cDNA (4µl) was used for
amplification in a 22-µl reaction volume containing 10
µl 2X ddPCR Supermix (Bio-Rad Laboratories, Inc.), 1
µl of 20X TaqMan miRNA PCR primer probe set of miR-126-3p
(cat. no. 4427975; assay ID: 002228; Ambion; Thermo Fisher
Scientific, Inc.), U6snRNA (cat. no. 4427975; assay ID: 001973;
Ambion; Thermo Fisher Scientific, Inc.) and 7 µl
nuclease-free water. Of this PCR mixture, 20 µl were loaded
onto the middle wells of the Droplet Generator cartridge (Bio-Rad
Laboratories, Inc.) with 70 µl of droplet generation oil
(Bio-Rad Laboratories, Inc.) in the lower wells. For all assays,
duplicate samples and a no template control were included. Droplets
were generated using a QX200 Droplet Generator (Bio-Rad
Laboratories, Inc.) and then transferred to a 96-well PCR plate.
PCR amplification was performed using a C1000 Touch Thermal Cycler
(Bio-Rad Laboratories, Inc.) at 95°C for 10 min, followed by 40
cycles of 94°C for 30 sec and 60°C for 60 sec (ramping rate reduced
to 2%), and a final elongation step at 98°C for 10 min. The plate
was then loaded into the QX200 Droplet Reader (Bio-Rad
Laboratories, Inc.) and the copies of miRNA were quantitated by
counting the number of positive droplets with analysis performed
using the QuantaSoft software™ (version 1.7.4.0917; Bio-Rad
Laboratories, Inc.).
Real-time qPCR
Real-time PCR for miR-126-3p was performed using
TaqMan miRNA assays (miR-126-3p: Cat. no. 4427975; assay ID:
002228; Ambion; Thermo Fisher Scientific, Inc.; and U6snRNA: Cat.
no. 4427975; assay ID: 001973; Ambion; Thermo Fisher Scientific,
Inc.) as per the manufacturer's protocol. PCR amplification was
performed using a Light cycler 96 thermal cycler (Roche
Diagnostics) at 95°C for 10 min, followed by 40 cycles of 95°C for
10 sec and 60°C for 60 sec. The threshold Cq was normalized with
U6snRNA used as an endogenous control. The relative amount of
miR-126-3p expression was calculated using the comparative Cq
method (21). Real time-qPCR for
disintegrin and metalloproteinase domain-containing protein 9
(ADAM9), CRK like proto-oncogene, adaptor protein (CRKL), CRK and
GAPDH expression were performed using the following primers: ADAM9
forward, 5′-AGA CAG CCG GGG AGTG TTC CTC-3′ and reverse, 5′-AGG TGG
CCT TGA TGG GAA CTG CT-3′; CRK forward, 5′-AGG GTT ATC CAG AAG CGA
GTC-3′ and reverse, 5′-CTT CCCA CTG ACC ACT CAC AT-3′, CRKL
forward, 5′-GTG CTT ATG ACA AGA CTG CCT-3′ and reverse, 5′-CAC TCG
TTT TCA TCT GGG TTT-3′; collagen-1A1 forward, 5′-CAG GTC TCG GTC
ATG GTA CCT-3′ and reverse, 5′-GTC GAG GGC CAA GAC GAA-3′; GAPDH
forward, 5′-GAG TCA ACG GAT TTG GTC GT-3′ and reverse, 5′-TTG ATT
TTG GAG GGA TCT CG-3′; 18S forward, 5′-GTA ACC CGT TGA ACC CCA
TT-3′ and reverse, 5′-CCA TC CAA TCG GTA GTA GCG-3′ using SYBR
green (22). PCR amplification was
performed using the light cycler 96 with pre-incubation at 95°C for
120 sec, amplification at 40 cycles of 95°C for 30 sec, 60°C for 30
sec and 72°C for 30 sec and then a final extension at 72°C for 5
min.
Western blot analysis
Protein lysates were prepared using RIPA buffer and
the total protein concentration was determined by using a BCA
protein kit (Thermo Fisher Scientific, Inc.) (23). Subsequently, 30 µg of total
protein was separated on NuPAGE-Tris Mini Gels (Novex; Thermo
Fisher Scientific, Inc.). The separated proteins were then
transferred onto nitro-cellulose membrane (Invitrogen; Thermo
Fisher Scientific, Inc.) using the iBlot-2 system (Invitrogen;
Thermo Fisher Scientific, Inc.). After transfer, the membrane was
blocked using intercept® blocking buffer (LI-COR
Biosciences) for 1 h at room temperature, followed by overnight
incubation at 4°C with the following primary antibodies: Rabbit
monoclonal antibody (mAb) to ADAM9 (cat. no. 4151S; 1:1,000
dilution; Cell Signaling Technology, Inc.), goat polyclonal Ab to
β-actin (cat. no. sc-1616; 1:1,000 dilution; Santa Cruz
Biotechnology, Inc.), rabbit mAb to CD9 (cat. no. 13174S; 1:1,000
dilution; Cell Signaling Technology, Inc.) and mouse mAb to CD81
(cat. no. sc-166029; 1:1,000 dilution; Santa Cruz Biotechnology,
Inc.). Target proteins were detected using secondary antibody
1:15,000 dilution (IRDye 680RD donkey anti-goat secondary antibody;
cat. no. 926-6807, IRDye 800CW goat anti-rabbit IgG secondary
antibody; cat. no. 926-32211 IRDye® 800CW goat
anti-mouse IgG secondary antibody; cat. no. 926-32210, LI-COR
Biosciences) for 1 h at room temperature. Visualization was
performed using the Odyssey Imaging system (LI-COR Biosciences).
β-actin expression was determined concomitantly and used to
normalize the protein expression.
VEGF ELISA
The VEGF expression was assessed by ELISA as per the
manufacturer's protocol (cat. no. ab100662; Abcam) (24). In brief, the samples were added
into the wells and incubated at room temperature for 2.5 h.
Subsequently, the wells were washed and incubated with biotinylated
antibody, followed by incubation with horseradish
peroxidase-conjugated streptavidin. The wells were washed,
incubated with substrate solution and the reaction was terminated
using stop solution. Finally, the absorbance of the wells was read
at 450 nm using the FLUOstar Omega multimode microplate reader (BMG
Labtech).
Proliferation assay
Cell proliferation was assessed at various
time-points after transfection with precursor or antimiR using the
MTS assay (25). In brief,
miR-126-3p precursor or antimiR-126-3p or their respective
NC-transfected cells were seeded in a 96 well plate
(1×104 cells/well). For assessing proliferation, cells
were incubated with MTS reagent for 2-3 h and absorbance was
measured at 490 nm using the FLUOstar Omega multimode microplate
reader (BMG LABTECH).
Migration and invasion assay
HepG2 cells were transfected with precursor
miR-126-3p and then seeded (1×105 cells/well) in the
upper chambers of Transwell inserts (8-µm pore size Falcon
cell culture inserts; BD Biosciences) in serum-free media, while
the lower chamber was filled with complete media. For invasion
studies, wells were coated with Matrigel® (Corning,
Inc.). After 24 h, non-migrated cells in the upper chamber were
removed using a cotton swab, migrated cells on the lower surface of
the chamber were carefully washed with PBS and fixed with 4%
paraformaldehyde for 10 min at room temperature. The fixed cells
were then again carefully washed with PBS and stained with 0.1%
crystal violet for 15 min at room temperature. Images were captured
from five different fields and the total number of migrated cells
was quantitated using Wimasis image analysis (https://www.wimasis.com/en/; Wimasis GmbH).
Cell viability assay
HepG2 cells were transfected with precursor or
antimiR-126-3p. After 24 h, cells were treated with 350 nM
Sorafenib (Selleckchem) or 1 µM Regorafenib (Selleckchem) in
DMSO (final concentration of DMSO was kept at <1%) and cell
viability was assessed at different time-points using the MTS assay
as described above.
In-vitro tumorigenic assay
AntimiR-126-3p-transfected LX2 cells were used for
this assay. In brief, HepG2 and LX2 cells were mixed at a 1:1 ratio
with 3,000 cells each and seeded in ultra low-attachment plates
(cat. no. 07200603; Corning; Thermo Fisher Scientific, Inc.) to
generate tumor spheres. Images were captured at various
time-points. The number and size of tumor spheres formed were
determined by image analysis using Image Pro Plus software version
6.0 (Media Cybernetics).
Statistical analysis
All analyses were performed using GraphPad Prism 8
(GraphPad Software, Inc.). Differences between the
Pre-miR-NC/AntimiR-NC and pre-miR-126/AntimiR-126 groups were
analyzed by Student's t-test (two-tailed, unpaired), while one-way
analysis of variance (Tukey's multiple-comparisons test) was used
to compare three or more groups. All experiments were performed in
triplicate with three independent replicates. P<0.05 was
considered to indicate statistical significance. Values are
expressed as the mean ± standard deviation.
Results
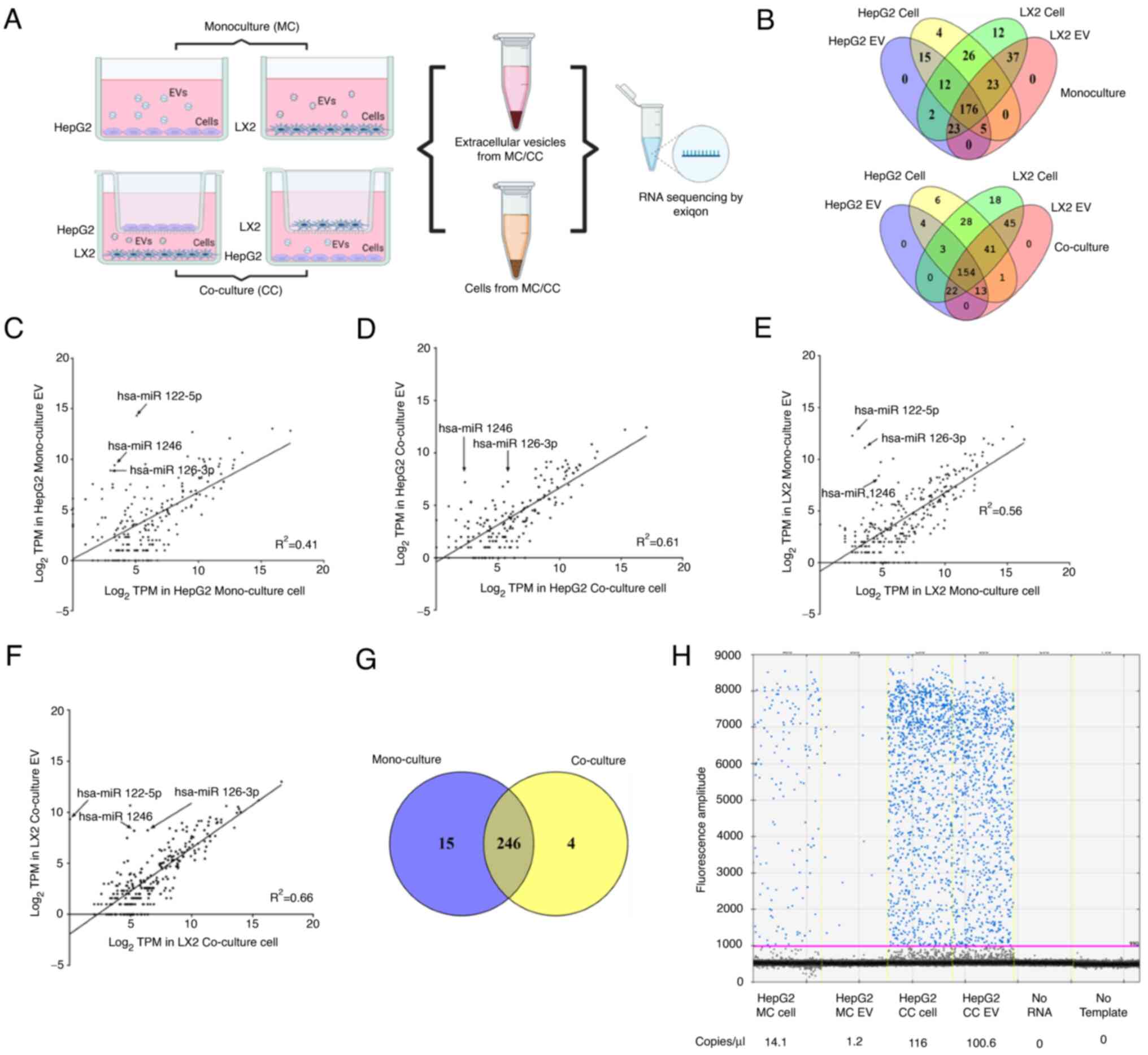
Cellular and EV miRNA content in HepG2
and LX2 cells
First, the levels of characteristic extracellular
RNAs were determined within EVs released from LC cells (HepG2) or
HSCs (LX2) in culture, either alone in monoculture or in co-culture
with each other (Fig. 1A). The
size distribution was analyzed using NTA (Fig. S1A), and CD9 and CD81 surface
protein expression was determined by western blot to characterize
the isolated vesicles (Fig. S1B).
In order to identify the miRNA, RNA sequencing was performed using
RNA obtained from cells or from the EV released from the cells. The
miRNA expression profiles in cells and EVs released from these
cells in monoculture and co-culture settings were depicted in
heatmaps in Fig. S1C.
A total of 261 distinct cellular miRNAs and 233 EV
miRNAs were detected from HepG2 cells, whereas 311 cellular miRNAs
and 264 EV miRNAs were identified from LX2 cells in monoculture. Of
these, 176 miRNAs were found in both cells and EVs from either
HepG2 or LX2 cells (Fig. 1B). In
co-culture settings, 250 cellular and 196 EV miRNAs were identified
from HepG2 cells during co-culture with LX2 cells. Similarly, 311
cellular and 276 EV miRNAs were identified from LX2 cells
co-cultured with HepG2 cells. 154 miRNAs were detected in both the
cells and EVs of either HepG2 or LX2 (Fig. 1B).
MiRNAs that were expressed in HepG2 cells in
mono-culture and in co-culture with LX2 cells were then identified
(Fig. 1C-F). A total of 246 miRNAs
were common to both cell types (Fig.
1G). Amongst these, 22 miRNAs were increased by >2 fold in
HepG2 during co-culture of these cells with LX2 cells (Table SI). Among the top 5 miRNAs that
were increased in HepG2 cells, miR-126-3p was also highly expressed
in LX2, cells as well as in the EVs released from LX2 cells
(Table SII). Therefore, the
expression of miR-126-3p was further validated by PCR with
quantitative analysis by ddPCR, revealing 116 copies/µl of
miR-126-3p in HepG2 cells co-cultured with LX2 compared with 14
copies/µl in HepG2 cells in mono-culture (Fig. 1H). These results indicated that
co-culture of HepG2 cells resulted in an increase in miR-126-3p in
HepG2 cells, and that this may involve the transfer of miR-126-3p
in EV from LX-2 cells.
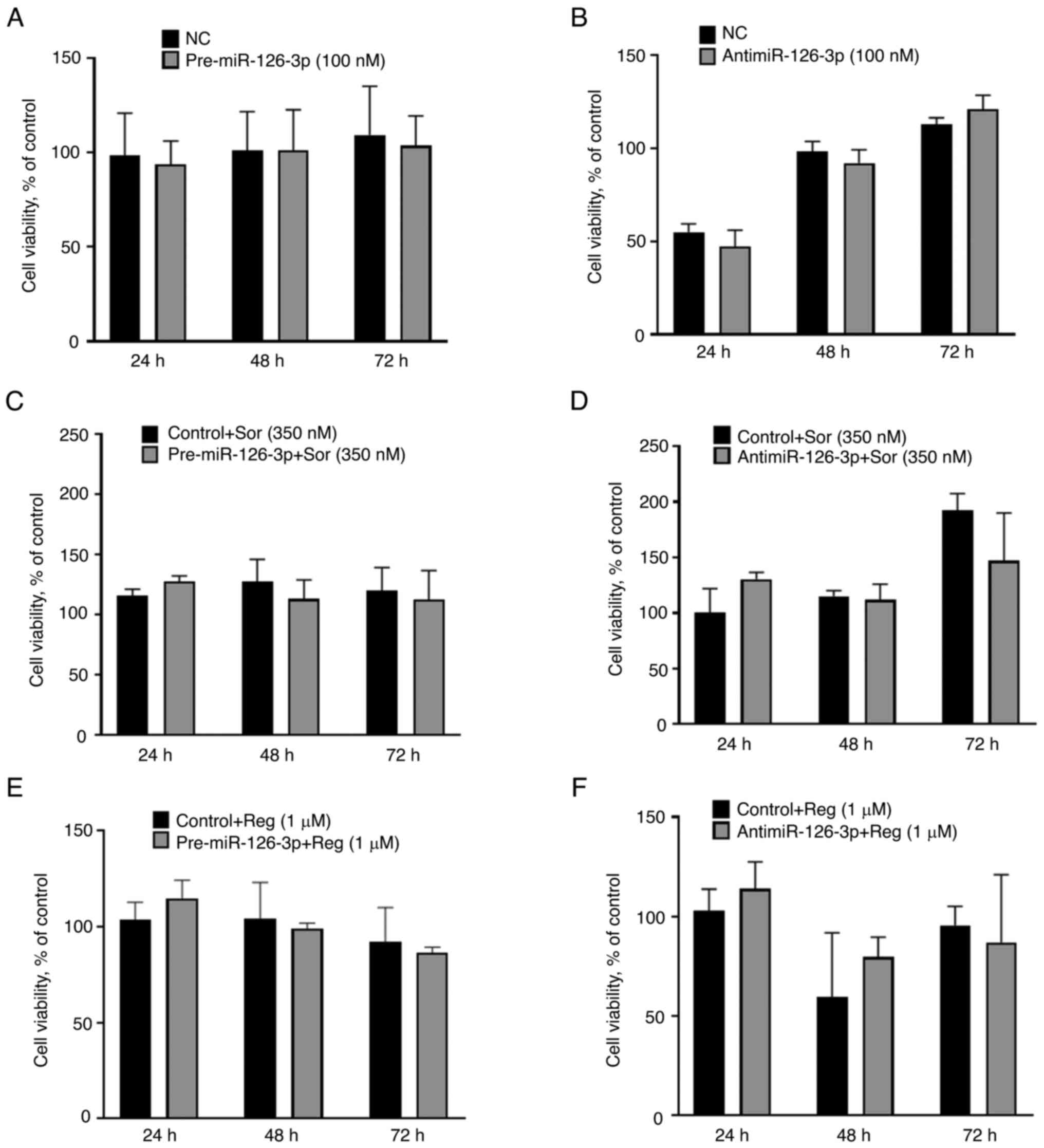
Alteration of tumor cell phenotype by
miR-126-3p
To determine the cellular effects of alterations in
cellular miR-126-3p expression that occur during co-culture, the
effect of modulation of miR-126-3p expression on cell viability was
assessed. HepG2 cells were transiently transfected with either
pre-miR-126-3p to enhance expression or antimiR-126-3p to reduce
expression (Fig. S1D and E).
Target gene expression studies verified the effects of transfection
of these constructs compared with their respective controls. Cell
proliferation was assessed over a 72-h period in cells following
transient transfection with pre-miR-126-3p or antimiR-126-3p, but
no difference was observed with either construct (Fig. 2A and B). The effect of modulation
of miR-126-3p on the cellular response to therapeutic drugs was
then further evaluated. HepG2 cells were transfected with either
pre-126-3p or antimiR-126-3p or their respective controls prior to
exposure for 72 h to either sorafenib or regorafenib. However,
modulation of miR-126-3p did not alter the sensitivity of HepG2
cells to either drug (Fig. 2C-F).
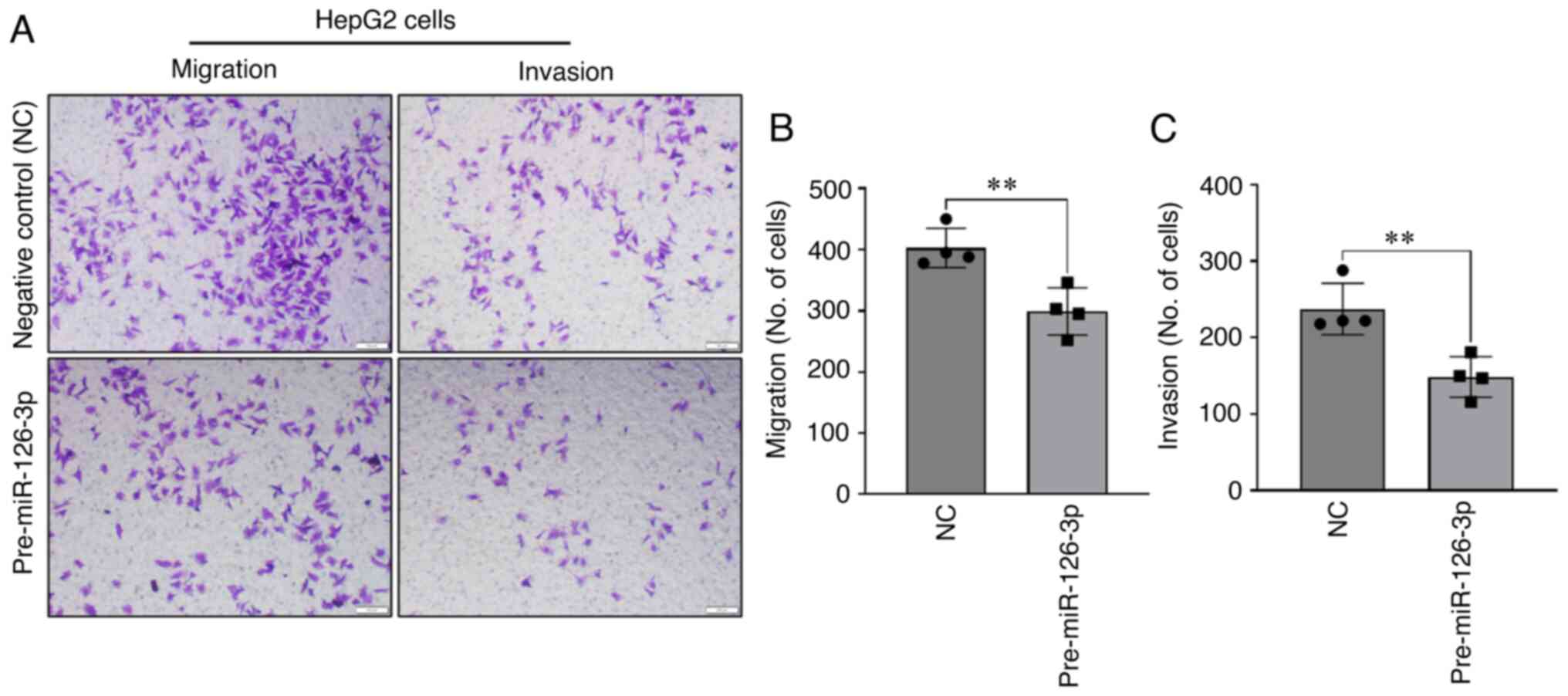
Next, the effects of modulation of miR-126-3p on migration and
invasion, malignant cellular phenotypes associated with tumor
behavior, were examined. Enforced expression of miR-126-3p by
transient transfection with precursor miR-126-3p decreased cell
migration compared to that of cells transfected with NC precursor
(Fig. 3A and B). Similarly, a
decrease in HepG2 cell invasion across Matrigel was observed with
miR-126-3p overexpression (Fig. 3A and
C). To further evaluate the phenotypic effects of altered
miR-126-3p on tumorigenesis, miR-126-3p was downregulated in LX2
cells using antimiR-126-3p (Fig.
4A) and HepG2/LX2 cell spheroids were generated using these
cells. Of note, these spheres were more compact (Fig. 4B) but similar in size (pixel size
in diameter) compared with HepG2/LX2 spheroids formed using LX2
cells transfected with negative inhibitor of miR-126-3p (Fig. 4C). However, there was no
considerable change in the number of spheroids formed between the
two groups (Fig. 4D). Consistent
with this, an increase in collagen-1A1 was observed following
knockdown of miR-126-3p in LX2, indicating ECM production (Fig. 4E).
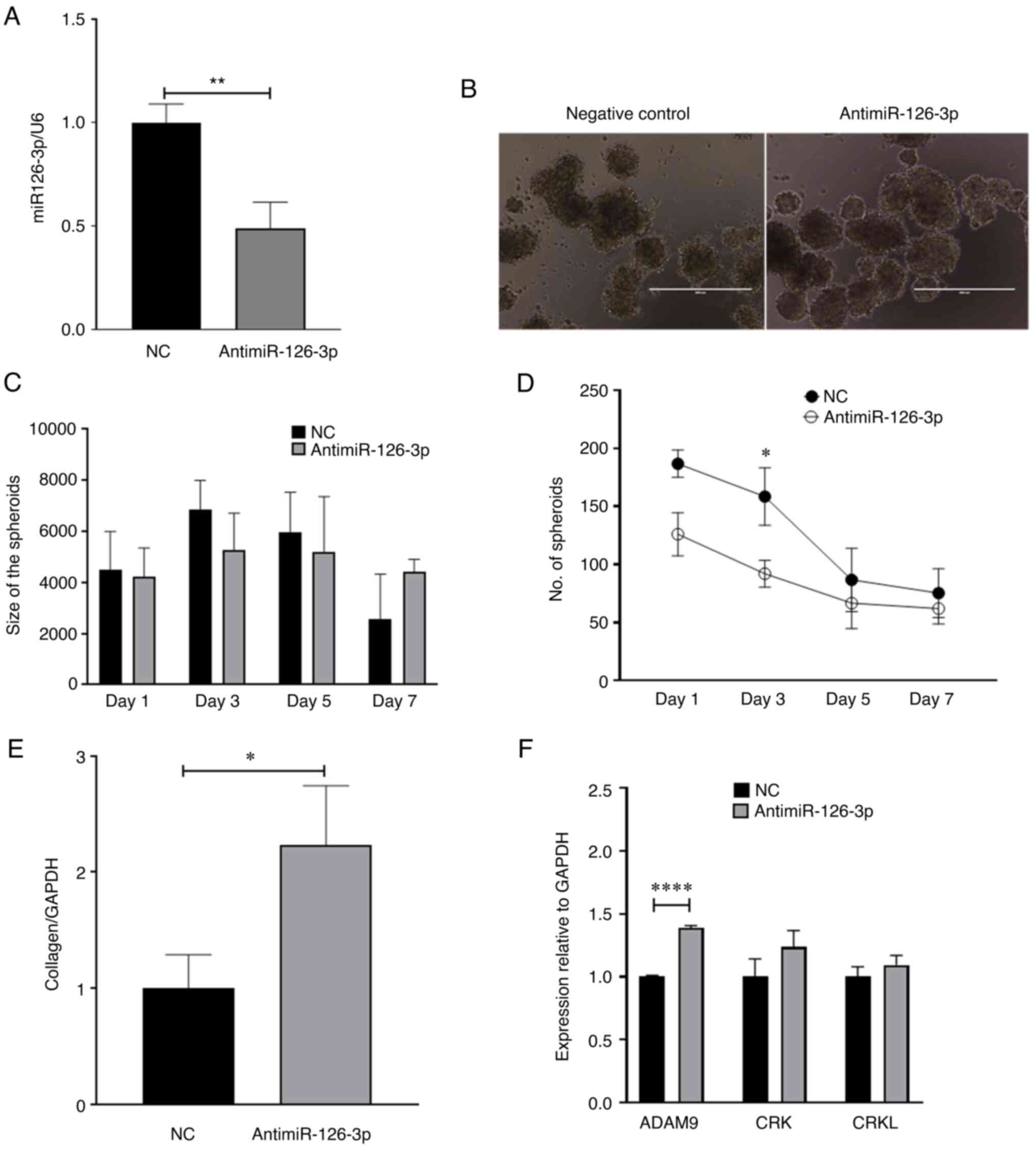 | Figure 4Effect of miR-126-3p knockdown in LX2
cells and HepG2 cells. LX2 cells were transfected with
antimiR-126-3p or a non-targeting control antimiR for 48 h.
Transfected LX2 cells were then cultured with HepG2 cells in
low-attachment conditions and spheroid growth was assessed. (A)
Real time-qPCR analysis of miR-126-3p expression in LX2 cells
transfected with antimiR-126-3p. Data represent the fold change
relative to the control after normalization to U6snRNA as an
internal control. (B) Microscopy of spheroids indicated increased
compactness in the antimiR-126-3p group compared with the negative
control (scale bars, 400 µm). (C) Spheroid size (pixel size
in diameter) and (D) number of spheroids were determined at the
indicated times after cultures were initiated. (E) Real time-qPCR
analysis of collagen-1A1 expression in LX2 cells transfected with
antimiR-126-3p. Data represent the fold-change relative to control
after normalization to GAPDH as an internal control. (F) Real
time-qPCR analysis of ADAM9, CRK and CRKL expression. Data
represent the fold change relative to control after normalization
to GAPDH as an internal control. Values are expressed as the mean ±
standard deviation (n=3 replicates). *P<0.05,
**P<0.01, ****P<0.0001. ADAM9,
disintegrin and metalloproteinase domain-containing protein 9;
CRKL, CRK like proto-oncogene, adaptor protein; miR, microRNA; NC,
negative control; qPCR, quantitative PCR. |
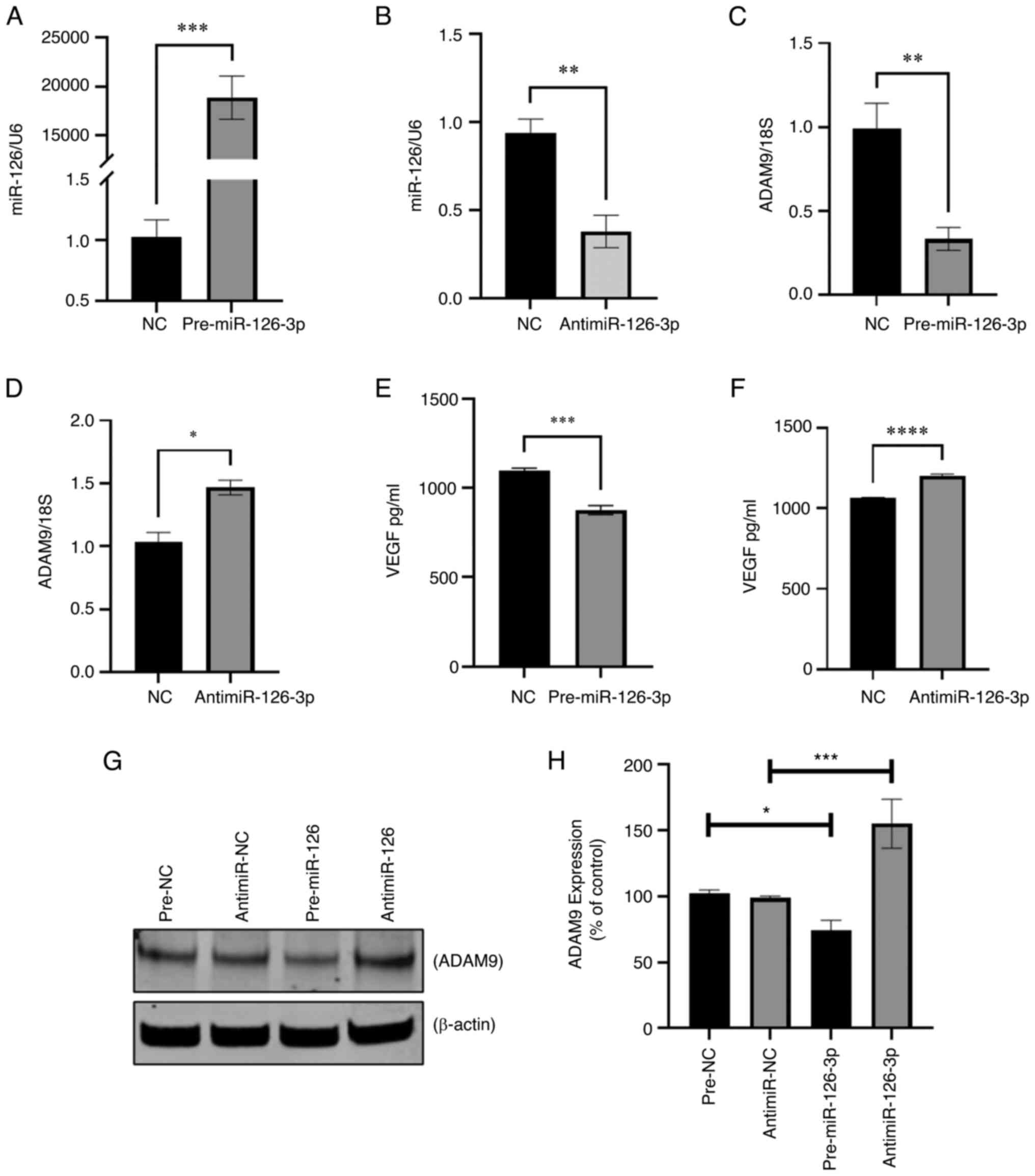
Downstream targets of miR-126-3p in
cancer cells
In order to identify potential intercellular targets
of miR-126-3p that may contribute to phenotypic changes, including
cell migration, a TargetScan search (https://www.targetscan.org/vert_80/) was performed,
which identified 28 predicted targets. Amongst these, ADAM9, CRK
and CRKL have been reported to be involved in inducing cancer cell
migration (26-28). Therefore, the effects of
antisense-mediated inhibition of miR-126-3p on the expression of
these genes were experimentally determined. Transient transfection
with antimiR-126-3p significantly decreased miR-126-3p expression
in HepG2 cells (Fig. S1E).
Concomitantly, the expression of ADAM9 mRNA, but not CRK and CRKL,
was increased in cells transfected with antimiR-126-3p compared to
negative control transfectants (Fig.
4F). These findings suggest that miR-126 downregulation is
linked to ADAM9 expression in LC to regulate cancer metastasis,
invasion and chemoresistance.
Modulation of tumor and stromal cell
spheroid growth by miR-126-3p
Cells in 2D cultures may not fully recapitulate
in vivo situations due to lack of cell-cell interaction. To
further assess the effect of tumor-stromal interactions in
vitro, a multicellular spheroid model comprised of LC cells and
HSCs was generated. A previously reported methyl cellulose-based 3D
co-culture model was used (Fig.
5A) (18). Spheroids were
first generated using a 1:1, 4:1 and 24:1 ratio of HepG2 to LX2
cells (Fig. S2). After 3 days,
the 4:1 ratio resulted in a more compact spheroid, which is more
physiologically relevant (Fig.
5B). Confocal microscopy after live/dead staining revealed
non-viable cells in a central necrotic core (Fig. 5C). The cell distribution in the
spheroid was assessed by H&E staining (Fig. 5D). Activation of HSC is known to
increase the expression of α-SMA within the LC TME (29), which was confirmed in the present
study by immunohistochemical staining of 3D-co-culture spheroids
(Fig. 5D). These results
corroborated the previous findings that, compared with cell
spheroids generated from LC cells alone, multicellular spheroids
generated from co-cultures of LC cells and HSCs were more compact
than spheroids comprised of LC cells alone (9). Next, the expression of miR-126 in 3D
HepG2/LX2 spheroids was determined using real-time qPCR. Of note,
miR-126 was downregulated in the multicellular spheroids compared
with spheroids generated only from HepG2 cells in contrast to the
results from 2D culture (Fig.
5E).
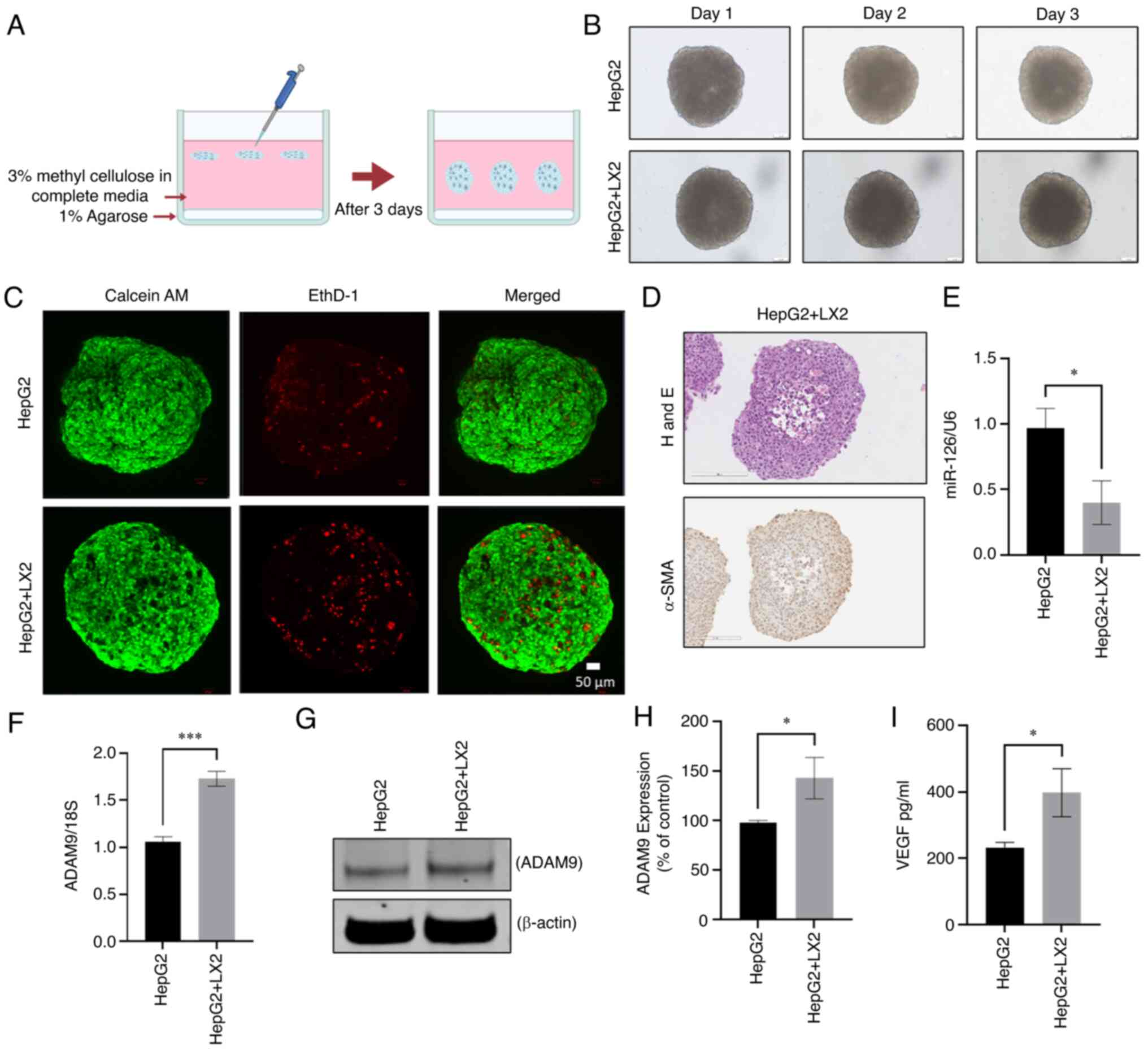 | Figure 5Generation and characterization of 3D
co-culture spheroid model of HepG2 and LX2 cells. (A) Schematic
presentation of 3D co-culture spheroid model by using methyl
cellulose as a matrix. (B) Microscopic images of 3D MC of HepG2 and
3D CC of HepG2 and LX2 at days 1, 2 and 3 (scale bars, 50
µm). (C) Confocal imaging of 3D MC/CC spheroids with
live/dead staining (green calcein AM staining indicated live cells,
while red EtBr homodimer staining indicated dead cells; scale bars,
50 µm). (D) Microscopic images of hematoxylin and eosin
staining of 3D CC spheroids represent cell arrangement and
immunohistochemical staining for α-SMA (scale bars, 200 µm).
(E) The expression of miR-126-3p in 3D MC and 3D CC spheroids was
determined by Real time-qPCR. Data represent the fold change
relative to the control after normalization to 18s rRNA as an
internal control. (F and G) ADAM9 in 3D MC and CC was determined
(F) by real time-qPCR at the RNA level and (G) western blot at the
protein level. (H) Densitometric quantification of the relative
expression of ADAM9 from the western blots normalized to β-actin.
(I) The level of VEGF secretion from the 3D MC and CC spheroids was
measured by ELISA. Values are expressed as the mean ± standard
deviation (n=3 replicates). *P<0.05,
***P<0.001. ADAM9, disintegrin and metalloproteinase
domain-containing protein 9; α-SMA, α-smooth muscle actin; CC,
co-culture; MC, monoculture; EtBr, ethidium bromide; VEGF, vascular
endothelial growth factor. |
Modulation of expression of downstream
targets in LC/HSC spheroids by miR-126
In the current 2D culture settings, miR-126-3p
modulation resulted in regulation of ADAM9 in HepG2 cells. Hence,
the expression level of the downstream candidate target of
miR-126-3p, ADAM9, was evaluated in 3D spheroids. In multicellular
spheroids, the mRNA level of ADAM9 was increased compared with that
observed in HepG2-only unicellular spheroids (Fig. 5F). Increased ADAM9 protein
expression was likewise confirmed by western blot analysis
(Fig. 5G and H). Previously,
miR-126-3p has also been reported to target VEGFA to modulate
angiogenesis (30). Therefore, the
present study investigated the expression of VEGFA protein in the
co-culture system. ELISA confirmed that VEGFA protein was
upregulated in multicellular spheroids compared to monocellular
ones (Fig. 5I). Next, to determine
the effect of alterations in expression of miR-126-3p on ADAM9 and
VEGFA, HepG2/LX2 spheroids were transfected with either
pre-miR-126-3p or antimiR-126-3p. Overexpression and knockdown of
miR-126 was confirmed by real time-qPCR (Fig. 6A and B, respectively). The target
gene effects were verified by real time-qPCR: ADAM9 mRNA and
protein were reduced, and VEGF expression was also reduced after
transfection with pre-miR-126. On the other hand, antimiR-126
increased ADAM9 expression as well as VEGFA protein expression
(Fig. 6C-H). These results
validated the findings from the in vitro 2D experiments.
Discussion
Tumor cells frequently arise in the setting of a
fibrotic milieu or may be accompanied by HSCs. The presence of
activated HSCs contributes to hepatic fibrosis and comprises an
essential element of the tumor stroma (31). Activation of focal adhesion
kinase-MMP9 signaling by HSC in the TME promotes LC cell migration
and invasion (32) and the
presence of HSCs in the microenvironment contributes to LC
chemoresistance by secretion of hepatocyte growth factor (33). Secretion of laminin 5 in the tumor
milieu by activated HSCs may stimulate the migratory ability of LC
(34). In a co-culture study of LC
and HSC, CD147 was identified as a key molecule in inflammation and
cancer (35). Furthermore,
glypican 3 has been identified to regulate HSC viability and
fibrinogenesis (36). Thus,
elucidating the mechanisms of LC interactions is important in order
to understand the contributions of the stromal cells or fibrotic
milieu on tumor growth and progression.
Extracellular vesicles may serve as vehicles for
mediators of intercellular crosstalk between the cells within the
TME. To mimic the interaction between LC cells and their
microenvironment, the potential contribution of EV non-coding RNA
crosstalk between cell types was evaluated using in vitro
cell co-culture models and cell spheroid models. Co-culture studies
have been widely used to study inter-cellular crosstalk between LC
cells and HSCs to understand the microenvironmental impact on
cancer progression. To study cargo within EVs released from either
cell, the cell supernatant and the EVs released within the
supernatant were collected. In the present study, alterations in
EV-RNA and phenotypic alterations in co-culture compared with
monoculture conditions were identified. While an analysis of
cellular effects and associated phenotypes related to vesicles
released from cells in monoculture is certainly of interest, and
should be conducted, the systematic characterization and analysis
of the associated phenotypes is beyond the scope of the present
study.
Recent studies on EVs as a carrier of information
between cells in the microenvironment have garnered much attention.
Indirect co-culture of tumor-endothelial cells has indicated that
EVs laden with stimulating factors are released by tumor cells and
may be taken up by endothelial cells, inducing proliferation and
reduction of apoptosis (37).
LC-derived EVs deliver 14-3-3ζ to tumor-infiltrating T lymphocytes
(TILs), inhibiting their antitumor functions (38). EVs from macrophages exposed to
apoptotic cancer cells have increased IL-6 that leads to
phosphorylation of STAT3 and increased transcription of target
genes inducing proliferation and migration in breast cancer cells
(39).
In the present study, a systematic assessment of EV
miRNA was performed using next-generation sequencing. Of note,
changes in a select number of miRNAs were observed when HepG2 cells
were co-cultured with LX-2 cells. Communication between tumor cells
and stromal elements may have a key role in cancer progression.
EV-mediated transfer of miRNAs, such as miR-122 (40), miR-21 and miR-192 (41), has been reported. The EV cargo
comprises numerous diverse types of biomolecules, including small
RNA, such as miRNAs, which may have other downstream effects in
target cells following their transfer. EV-mediated transfer of
miRNA has been implicated in numerous different settings (19), including Twist1 (42), k-ras (43) miR-122 (44) miR-200 (45). HSC EVs were reported to transfer
miR-335-5p to inhibit proliferation and invasion in target LC cells
(46), while LC-derived EV miR-21
was indicated to transform HSCs into cancer-associated fibroblasts
(CAFs) to promote cancer progression (47). Similarly, miR-1247-3p in EVs from
tumor cells was able to convert normal fibroblasts to CAFs
(48).
An increase in miR-126-3p in EVs occurs when LC
cells are co-cultured with HSCs. Of note, miR-126-3p has been
indicated to have a tumor-suppressive role in LC and other cancers.
Indeed, low intratumoral expression of miR-126-3p is associated
with recurrence and poor survival of patients with HCC, whereas
forced overexpression markedly impairs HCC tumor cell
proliferation, invasion and development of metastatic lung nodules
(49,50). Therefore, the present study aimed
to further explore the role of miR-126-3p in the liver TME. While
modulation of miR-126-3p altered migration and invasion of human LC
cells in the present study, there was no appreciable difference in
sensitivity to drugs, in the proliferative capacity in 2D cultures
or in size or number of tumor-cell spheroids in 3D culture.
However, other studies reported that miR-126-3p is able to inhibit
not only the migration and invasion, but also the proliferation of
tumor cells and tumor angiogenesis in vitro and in
vivo in HCC tumor-bearing mice (50,51).
These effects are mediated by modulation of downstream targets such
as low-density lipoprotein related protein 6 (50) and ADAM9 (52) to suppress tumor metastasis,
phosphoinositide-3-kinase regulatory subunit 2 to impair tumor cell
migration/invasion and epidermal growth factor like domain 7 to
inhibit tumor angiogenesis (50).
Validation of miR-126-mediated regulation of ADAM9 expression has
been extensively reported, including an in vitro 3′UTR
luciferase assay (53) and by
target seed sequence prediction software (52,54);
therefore, it was not further validated in the present study. The
role of miR-126 in modulating sensitivity to therapy has been noted
with altered sensitivity to cisplatin in HCC through the repression
of insulin receptor substrate 1 expression (55), or to sorafenib through the
downregulation of sprout-related EVH1 domain-containing protein 1
(56). MiR-126-3p has also been
indicated to elicit tumor suppressor effects in other
gastrointestinal and other solid tumor types, such as lung cancer
(57,58).
Manipulation of extracellular RNA signaling may have
therapeutic implications. A single miRNA may regulate the
expression of multiple genes, thus making them worthwhile targets
for therapeutic approaches (59,60).
Specific miRNAs, such as miR-126-3p, have a growing body of
evidence for multiple tumor-suppressive roles that may be mediated
through multiple targets. While diverse EV-RNAs may be present in
the local TME and may theoretically participate in EV-RNA signaling
across cells, studies focused on a single candidate HSC-derived
EV-RNA demonstrate the functional contribution of EV-RNA signaling
across cells in the local microenvironment to modulate LC cell
behavior. With this observation, determining the contributions of
other candidates or their combinatorial effects on local
microenvironmental cell-to-cell signaling is justified. These
studies enable us to understand the contributions of EV-based
miR-126-3p-mediated intercellular communication on tumor behavior.
Based on these findings, enrichment of miR-126-3p in LC cells may
be used as a therapeutic approach to inhibit tumor growth through
manipulated expression of key mediators, such as ADAM9 and VEGFA,
to modulate critical processes involved in tumor growth, such as
metastasis or angiogenesis (Fig.
7). However, further studies should be performed to explore
other downstream targets of miR-126-3p in the liver TME.
Supplementary Data
Availability of data and materials
The dataset for RNA-sequencing presented in this
study may be obtained from the online repository Gene Expression
Omnibus (GEO) under accession no. GSE196131 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE196131).
The data may be accessed using the password 'mxsnqoawhnchnet' on
the above link. The datasets used and/or analyzed during the
current study are available from the corresponding author upon
reasonable request.
Authors' contributions
Conceptualization, TP; methodology, TP, AM, PG, AAS
and JD; formal analysis, AM, PG and AAS; investigation, AM, PG,
AAS, JD, IY and TP; resources, TP; data curation, AM, PG and AAS;
writing-original draft, TP, AM and PG; writing-review &
editing, AM, PG, AAS, IY, TP and JD; visualization, AM and PG;
supervision, TP; funding acquisition, TP. The authenticity of the
raw data has been verified by AM, PG, AAS and TP. All authors have
read and approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interest
The authors declare that they have no competing
interests.
Acknowledgments
The authors would like to acknowledge Dr. Gregory j
Gores (Division of Gastroenterology and Hepatology, Mayo Clinic,
Rochester, Minnesota, USA) for providing the LX2 cell line.
Funding
The present study was supported by the National Cancer Institute
(grant no. CA217833), the James C. and Sarah K. Kennedy Deanship
and the Alfred D. and Audrey M. Petersen Professorship to TP.
Abbreviations:
|
ADAM9
|
disintegrin and metalloproteinase
domain-containing protein 9
|
|
CAFs
|
cancer-associated fibroblasts
|
|
CC
|
co-culture
|
|
ECM
|
extracellular matrix
|
|
EVs
|
extracellular vesicles
|
|
HSC
|
hepatic stellate cells
|
|
HG
|
high glucose
|
|
MC
|
monoculture
|
|
TME
|
tumor microenvironment
|
|
LC
|
liver cancer
|
|
TPM
|
transcripts per million
|
|
VEGF
|
vascular endothelial growth factor
|
|
RT
|
reverse transcription
|
References
|
1
|
Mbeunkui F and Johann DJ Jr: Cancer and
the tumor microenvironment: A review of an essential relationship.
Cancer Chemother Pharmacol. 63:571–582. 2009. View Article : Google Scholar
|
|
2
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang X, Hassan W, Jabeen Q, Khan GJ and
Iqbal F: Interdependent and independent multidimensional role of
tumor microenvironment on hepatocellular carcinoma. Cytokine.
103:150–159. 2018. View Article : Google Scholar
|
|
4
|
Tahmasebi Birgani M and Carloni V: Tumor
microenvironment, a paradigm in hepatocellular carcinoma
progression and therapy. Int J Mol Sci. 18:4052017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kamil F and Rowe JH: How does the tumor
microenvironment play a role in hepatobiliary tumors? J
Gastrointest Oncol. 9:180–195. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Balogh J, Victor D III, Asham EH,
Burroughs SG, Boktour M, Saharia A, Li X, Ghobrial RM and Monsour
HP Jr: Hepatocellular carcinoma: A review. J Hepatocell Carcinoma.
3:41–53. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yang S, Yang L, Li X, Li B, Li Y, Zhang X,
Ma Y, Peng X, Jin H and Li H: New insights into autophagy in
hepatocellular carcinoma: Mechanisms and therapeutic strategies. Am
J Cancer Res. 9:1329–1353. 2019.PubMed/NCBI
|
|
8
|
Baglieri J, Brenner DA and Kisseleva T:
The role of fibrosis and liver-associated fibroblasts in the
pathogenesis of hepatocellular carcinoma. Int J Mol Sci.
20:17232019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Song Y, Kim SH, Kim KM, Choi EK, Kim J and
Seo HR: Activated hepatic stellate cells play pivotal roles in
hepatocellular carcinoma cell chemoresistance and migration in
multicellular tumor spheroids. Sci Rep. 6:367502016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Laurenzana I, Lamorte D, Trino S, De Luca
L, Ambrosino C, Zoppoli P, Ruggieri V, Del Vecchio L, Musto P,
Caivano A and Falco G: Extracellular vesicles: A new prospective in
crosstalk between microenvironment and stem cells in hematological
malignancies. Stem Cells Int. 2018:98631942018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bebelman MP, Smit MJ, Pegtel DM and Baglio
SR: Biogenesis and function of extracellular vesicles in cancer.
Pharmacol Ther. 188:1–11. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jabalee J, Towle R and Garnis C: The role
of extracellular vesicles in cancer: Cargo, function, and
therapeutic implications. Cells. 7:932018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yang J, Li C, Zhang L and Wang X:
Extracellular vesicles as carriers of non-coding RNAs in liver
diseases. Front Pharmacol. 9:4152018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Vu LT, Peng B, Zhang DX, Ma V,
Mathey-Andrews CA, Lam CK, Kiomourtzis T, Jin J, McReynolds L,
Huang L, et al: Tumor-secreted extracellular vesicles promote the
activation of cancer-associated fibroblasts via the transfer of
microRNA-125b. J Extracell Vesicles. 8:15996802019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Au Yeung CL: Co NN, Tsuruga T, Yeung TL,
Kwan SY, Leung CS, Li Y, Lu ES, Kwan K, Wong KK, et al Exosomal
transfer of stroma-derived miR21 confers paclitaxel resistance in
ovarian cancer cells through targeting APAF1. Nat Commun.
7:111502016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lee H, Abston E, Zhang D, Rai A and Jin Y:
Extracellular vesicle: An emerging mediator of intercellular
crosstalk in lung inflammation and injury. Front Immunol.
9:9242018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xu L, Hui A, Albanis E, Arthur MJ, O'Byrne
SM, Blaner WS, Mukherjee P, Friedman SL and Eng FJ: Human hepatic
stellate cell lines, LX-1 and LX-2: New tools for analysis of
hepatic fibrosis. Gut. 54:142–151. 2005. View Article : Google Scholar
|
|
18
|
Sayyed AA, Gondaliya P, Mali M, Pawar A,
Bhat P, Khairnar A, Arya N and Kalia K: MiR-155 inhibitor-laden
exosomes reverse resistance to cisplatin in a 3D tumor spheroid and
xenograft model of oral cancer. Mol Pharm. 18:3010–3025. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Matsuda A, Ishiguro K, Yan IK and Patel T:
Extracellular vesicle-based therapeutic targeting of β-catenin to
modulate anticancer immune responses in hepatocellular cancer.
Hepatol Commun. 3:525–541. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kogure T and Patel T: Isolation of
extracellular nanovesicle microRNA from liver cancer cells in
culture. Kosaka N: Circulating MicroRNAs. Methods in Molecular
Biology. 1024. Humana Press; Totowa, NJ: pp. 11–18. 2013,
View Article : Google Scholar
|
|
21
|
Schmittgen TD and Livak KJ: Analyzing
real-time PCR data by the comparative C(T) method. Nat Protoc.
3:1101–1108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sharma N, Soni R, Sharma M, Chatterjee S,
Parihar N, Mukarram M, Kale R, Sayyed AA, Behera SK and Khairnar A:
Chlorogenic acid: A polyphenol from coffee rendered neuroprotection
against rotenone-induced parkinson's disease by GLP-1 secretion.
Mol Neurobiol. 59:6834–6856. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Parekh P, Sharma N, Sharma M, Gadepalli A,
Sayyed AA, Chatterjee S, Kate A and Khairnar A: AMPK-dependent
autophagy activation and alpha-Synuclein clearance: A putative
mechanism behind alpha-mangostin's neuroprotection in a
rotenone-induced mouse model of Parkinson's disease. Metab Brain
Dis. Sep 30–2022.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gondaliya P, Sayyed AA, Bhat P, Mali M,
Arya N, Khairnar A and Kalia K: Mesenchymal stem cell-derived
exosomes loaded with miR-155 inhibitor ameliorate diabetic wound
healing. Mol Pharm. 19:1294–1308. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Takahashi K, Yan IK, Kogure T, Haga H and
Patel T: Extracellular vesicle-mediated transfer of long non-coding
RNA ROR modulates chemosensitivity in human hepatocellular cancer.
FEBS Open Bio. 4:458–467. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hsieh MH, Tsai JP, Yang SF, Chiou HL, Lin
CL, Hsieh YH and Chang HR: Fisetin suppresses the proliferation and
metastasis of renal cell carcinoma through upregulation of
MEK/ERK-targeting CTSS and ADAM9. Cells. 8:9482019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kumar S, Davra V, Obr AE, Geng K, Wood TL,
De Lorenzo MS and Birge RB: Crk adaptor protein promotes PD-L1
expression, EMT and immune evasion in a murine model of
triple-negative breast cancer. Oncoimmunology. 7:e13761552017.
View Article : Google Scholar
|
|
28
|
Guo C, Zhao D, Zhang Q, Liu S and Sun MZ:
miR-429 suppresses tumor migration and invasion by targeting CRKL
in hepatocellular carcinoma via inhibiting Raf/MEK/ERK pathway and
epithelial-mesenchymal transition. Sci Rep. 8:23752018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Carpino G, Morini S, Ginanni Corradini S,
Franchitto A, Merli M, Siciliano M, Gentili F, Onetti Muda A,
Berloco P, Rossi M, et al: Alpha-SMA expression in hepatic stellate
cells and quantitative analysis of hepatic fibrosis in cirrhosis
and in recurrent chronic hepatitis after liver transplantation. Dig
Liver Dis. 37:349–356. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Alhasan L: MiR-126 modulates angiogenesis
in breast cancer by targeting VEGF-A-mRNA. Asian Pac J Cancer Prev.
20:193–197. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Amann T, Bataille F, Spruss T, Mühlbauer
M, Gäbele E, Schölmerich J, Kiefer P, Bosserhoff AK and Hellerbrand
C: Activated hepatic stellate cells promote tumorigenicity of
hepatocellular carcinoma. Cancer Sci. 100:646–653. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Han S, Han L, Yao Y, Sun H, Zan X and Liu
Q: Activated hepatic stellate cells promote hepatocellular
carcinoma cell migration and invasion via the activation of
FAK-MMP9 signaling. Oncol Rep. 31:641–648. 2014. View Article : Google Scholar
|
|
33
|
Yu G, Jing Y, Kou X, Ye F, Gao L, Fan Q,
Yang Y, Zhao Q, Li R, Wu M and Wei L: Hepatic stellate cells
secreted hepatocyte growth factor contributes to the
chemoresistance of hepatocellular carcinoma. PLoS One.
8:e733122013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Santamato A, Fransvea E, Dituri F,
Caligiuri A, Quaranta M, Niimi T, Pinzani M, Antonaci S and
Giannelli G: Hepatic stellate cells stimulate HCC cell migration
via laminin-5 production. Clin Sci (Lond). 121:159–168. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ma T, Wang Z, Yang Z and Chen J: Cluster
of differentiation 147 is a key molecule during hepatocellular
carcinoma cell-hepatic stellate cell cross-talk in the rat liver.
Mol Med Rep. 12:111–118. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Magistri P, Leonard SY, Tang CM, Chan JC,
Lee TE and Sicklick JK: The glypican 3 hepatocellular carcinoma
marker regulates human hepatic stellate cells via Hedgehog
signaling. J Surg Res. 187:377–385. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Jamshidi-Parsian A, Griffin RJ, Kore RA,
Todorova VK and Makhoul I: Tumor-endothelial cell interaction in an
experimental model of human hepatocellular carcinoma. Exp Cell Res.
372:16–24. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wang X, Shen H, Zhangyuan G, Huang R,
Zhang W, He Q, Jin K, Zhuo H, Zhang Z, Wang J, et al: 14-3-3ζ
delivered by hepatocellular carcinoma-derived exosomes impaired
anti-tumor function of tumor-infiltrating T lymphocytes. Cell Death
Dis. 9:1592018. View Article : Google Scholar
|
|
39
|
Yu X, Zhang Q, Zhang X, Han Q, Li H, Mao
Y, Wang X, Guo H, Irwin DM, Niu G and Tan H: Exosomes from
macrophages exposed to apoptotic breast cancer cells promote breast
cancer proliferation and metastasis. J Cancer. 10:2892–2906. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Basu S and Bhattacharyya SN: Insulin-like
growth factor-1 prevents miR-122 production in neighbouring cells
to curtail its intercellular transfer to ensure proliferation of
human hepatoma cells. Nucleic Acids Res. 42:7170–7185. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Chiba M, Kimura M and Asari S: Exosomes
secreted from human colorectal cancer cell lines contain mRNAs,
microRNAs and natural antisense RNAs, that can transfer into the
human hepatoma HepG2 and lung cancer A549 cell lines. Oncol Rep.
28:1551–1558. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Sun T, Zhao N, Zhao XL, Gu Q, Zhang SW,
Che N, Wang XH, Du J, Liu YX and Sun BC: Expression and functional
significance of Twist1 in hepatocellular carcinoma: Its role in
vasculogenic mimicry. Hepatology. 51:545–556. 2010. View Article : Google Scholar
|
|
43
|
Tao J, Zhang R, Singh S, Poddar M, Xu E,
Oertel M, Chen X, Ganesh S, Abrams M and Monga SP: Targeting
β-catenin in hepatocellular cancers induced by coexpression of
mutant β-catenin and K-Ras in mice. Hepatology. 65:1581–1599. 2017.
View Article : Google Scholar
|
|
44
|
Hsu SH, Wang B, Kota J, Yu J, Costinean S,
Kutay H, Yu L, Bai S, La Perle K, Chivukula RR, et al: Essential
metabolic, anti-inflammatory, and anti-tumorigenic functions of
miR-122 in liver. J Clin Invest. 122:2871–2883. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Xue X, Zhang Y, Zhi Q, Tu M, Xu Y, Sun J,
Wei J, Lu Z, Miao Y and Gao W: MiR200-upregulated Vasohibin 2
promotes the malignant transformation of tumors by inducing
epithelial-mesenchymal transition in hepatocellular carcinoma. Cell
Commun Signal. 12:622014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wang F, Li L, Piontek K, Sakaguchi M and
Selaru FM: Exosome miR-335 as a novel therapeutic strategy in
hepatocellular carcinoma. Hepatology. 67:940–954. 2018. View Article : Google Scholar
|
|
47
|
Zhou Y, Ren H, Dai B, Li J, Shang L, Huang
J and Shi X: Hepatocellular carcinoma-derived exosomal miRNA-21
contributes to tumor progression by converting hepatocyte stellate
cells to cancer-associated fibroblasts. J Exp Clin Cancer Res.
37:3242018. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Fang T, Lv H, Lv G, Li T, Wang C, Han Q,
Yu L, Su B, Guo L, Huang S, et al: Tumor-derived exosomal
miR-1247-3p induces cancer-associated fibroblast activation to
foster lung metastasis of liver cancer. Nat Commun. 9:1912018.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chen H, Miao R, Fan J, Han Z, Wu J, Qiu G,
Tang H and Peng Z: Decreased expression of miR-126 correlates with
metastatic recurrence of hepatocellular carcinoma. Clin Exp
Metastasis. 30:651–658. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Du C, Lv Z, Cao L, Ding C, Gyabaah OA, Xie
H, Zhou L, Wu J and Zheng S: MiR-126-3p suppresses tumor metastasis
and angiogenesis of hepatocellular carcinoma by targeting LRP6 and
PIK3R2. J Transl Med. 12:2592014. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Gong C, Fang J, Li G, Liu HH and Liu ZS:
Effects of microRNA-126 on cell proliferation, apoptosis and tumor
angiogenesis via the down-regulating ERK signaling pathway by
targeting EGFL7 in hepatocellular carcinoma. Oncotarget.
8:52527–52542. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Xiang LY, Ou HH, Liu XC, Chen ZJ, Li XH,
Huang Y and Yang DH: Loss of tumor suppressor miR-126 contributes
to the development of hepatitis B virus-related hepatocellular
carcinoma metastasis through the upregulation of ADAM9. Tumour
Biol. 39:10104283177091282017. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Hamada S, Satoh K, Fujibuchi W, Hirota M,
Kanno A, Unno J, Masamune A, Kikuta K, Kume K and Shimosegawa T:
MiR-126 acts as a tumor suppressor in pancreatic cancer cells via
the regulation of ADAM9miR-126/ADAM9 regulates invasive growth of
pancreatic cancer. Mol Cancer Res. 10:3–10. 2012. View Article : Google Scholar
|
|
54
|
Jia AY, Castillo-Martin M, Bonal DM,
Sánchez-Carbayo M, Silva JM and Cordon-Cardo C: MicroRNA-126
inhibits invasion in bladder cancer via regulation of ADAM9. Br J
Cancer. 110:2945–2954. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zuo J, Luo R, Huang C, Lou X and Li L:
MiR-126 enhances cisplatin chemosensitivity in hepatocellular
carcinoma cells by targeting IRS1. Trop J Pharm Res. 18:25–30.
2019. View Article : Google Scholar
|
|
56
|
Tan W, Lin Z, Chen X, Li W, Zhu S, Wei Y,
Huo L, Chen Y and Shang C: miR-126-3p contributes to sorafenib
resistance in hepatocellular carcinoma via downregulating SPRED1.
Ann Transl Med. 9:382021. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Hu M, Xiong S, Chen Q, Zhu S and Zhou X:
Novel role of microRNA-126 in digestive system cancers: From bench
to bedside. Oncol Lett. 17:31–41. 2019.PubMed/NCBI
|
|
58
|
Miko E, Margitai Z, Czimmerer Z, Várkonyi
I, Dezso B, Lányi A, Bacsó Z and Scholtz B: miR-126 inhibits
proliferation of small cell lung cancer cells by targeting SLC7A5.
FEBS Lett. 585:1191–1196. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Kitano M and Bloomston PM: Hepatic
stellate cells and microRNAs in pathogenesis of liver fibrosis. J
Clin Med. 5:382016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Sayyed AA, Gondaliya P, Bhat P, Mali M,
Arya N, Khairnar A and Kalia K: Role of miRNAs in cancer
diagnostics and therapy: A recent update. Curr Pharm Des.
28:471–487. 2022. View Article : Google Scholar
|