1. Introduction
Advanced-stage non-small cell lung cancer
(NSCLC)-related mortality occurs <18 months of diagnosis mainly
due to metastatic spread (1). A
total of ~50% of patients with NSCLC develop brain metastasis (BM)
(2-4), mostly in the cerebral hemisphere
(80%), the cerebellum (15%) and the brainstem (5%) (5). The management of BM includes: i)
multimodal surgery, the gold standard when there are neurological
symptoms or <3 BMs (6); ii)
radiotherapy (RT); iii) immunotherapy; and iv) tyrosine kinase
inhibitors (TKIs) (4). RT is
inevitably associated with radioresistance (RR). A search for
prognostic factors in NSCLC has shown that high expression of Hippo
pathway effectors is associated with decreased overall survival
(7). Hippo pathway dysregulation
(Fig. 1) mediates cancer cell
motility and subsequent metastasis formation as a drug or RT
resistance in several human cancers (Fig. 2), including NSCLC (8-13).
In NSCLC, the following events occur: deregulation of the Hippo
pathway occurring in all NSCLC due to epigenetic dysregulation
(14) and/or hypoxia (13), leads to the transformation of
healthy bronchial epithelial cells into tumour cells (8,9),
and subsequently promotes cell motility and subsequent metastasis
of bronchial origin by hyperactivating the nuclear dbf2-related
(NDR) 2 kinase and the transcription cofactor yes-associated
protein-1 (YAP1), and inhibiting the antimigratory small GTPase
RhoB (9). BMs of bronchial origin
are treated with surgery and/or RT, but invariably, RR is
established, and it is hypothesized that the deregulation of the
Hippo pathway, which is the origin of bronchial cancer and its
dissemination, is also involved in the RR of these BMs of bronchial
origin (4,5). This finding suggested that the Hippo
pathway is involved in the unsatisfactory treatment of NSCLC. Due
to the large scope of Hippo-dependent biological implications,
elucidating the mechanisms of Hippo-dependent treatment resistance,
notably RR, in BM from NSCLC is essential. The aim of the present
review is to emphasize the importance of Hippo in RR, providing a
comprehensive summary of the literature and identifying the
underlying mechanism involved in the treatment of BM from
NSCLC.
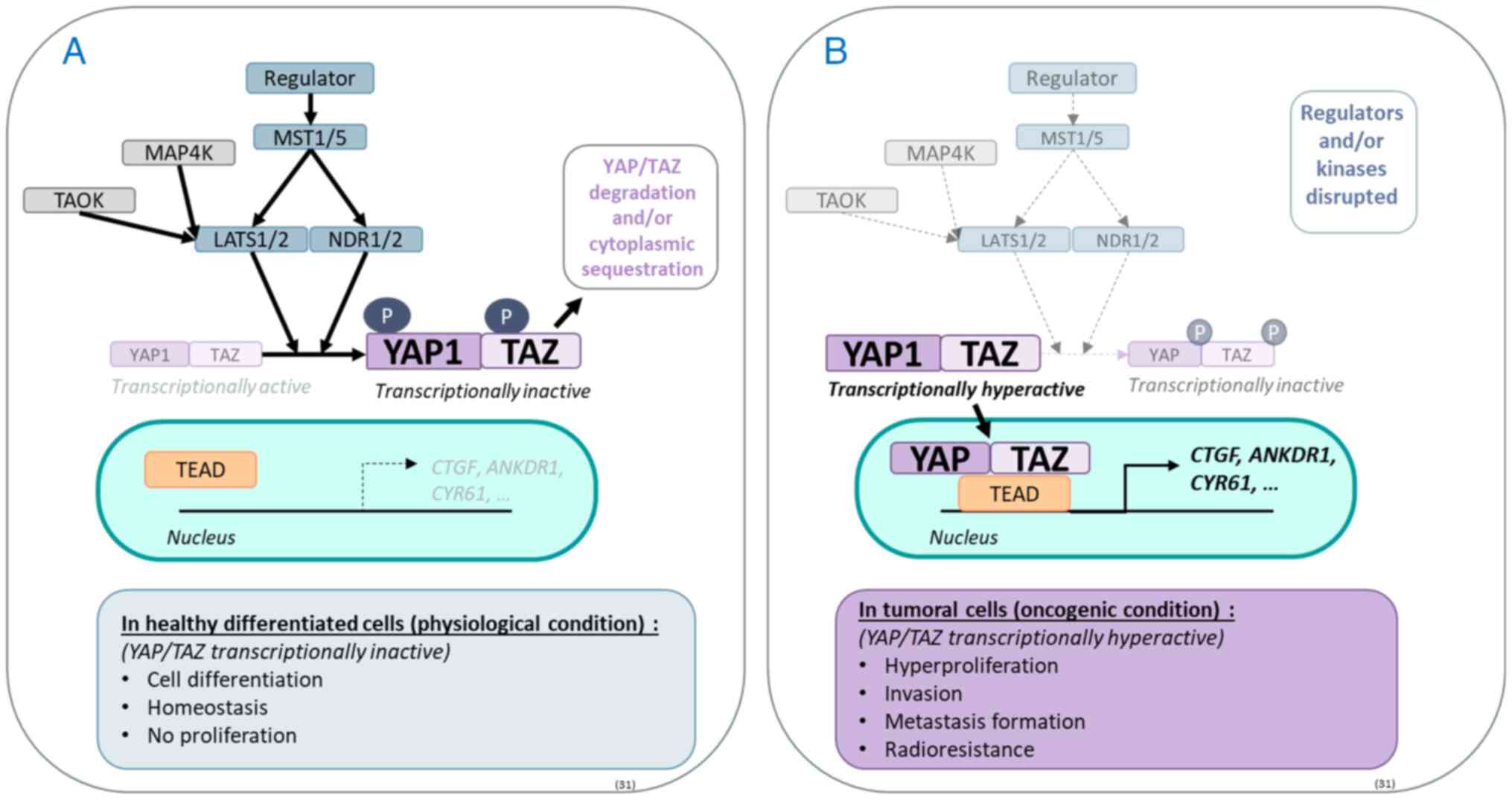 | Figure 1Hippo pathway and its physiological
and oncogenic roles. The Hippo pathway is comprised of
serine/threonine kinases activated by a phosphorylation cascade,
the core of which starts with MST1/5, which activates LATS1/2 and
NDR1/2. The latter two regulate by phosphorylation the activation
of the effector proteins YAP1 and TAZ. When phosphorylated, YAP1
and TAZ are degraded by the proteasome and/or sequestered in the
cytoplasm. (A) In a physiological context, YAP1 and TAZ are found
in an equilibrium of active and inactive forms that regulate the
transcription of various target genes such as CTFG, ANKDR1 and
CYR61. (B) In cancer, YAP1 and/or TAZ are often found in
hyperactive states due to reduced regulation of Hippo kinases,
thereby playing an oncogenic role. MST1/5, mammalian sterile
20-like kinase; LATS1/2, large tumour suppressor 1/2; NDR1/2,
nuclear dbf2-related kinase; YAP1, yes-associated protein 1; TAZ,
transcriptional coactivator with a PDZ-binding domain; TEAD, TEA
DNA-binding protein; TAOK, Thousand and one amino-acid kinase. |
2. RR of BM from NSCLC
Biological effects of X-rays (XRs)
Ionizing radiation (IR), notably XR, induces
single-strand breaks (SSBs) and double-strand breaks (DSBs) in DNA,
ultimately leading to cellular death by necrosis, apoptosis or
mitotic death (15). One gray
(Gy) of XR results in >2,000 base damages, 30 DNA-DNA
crosslinks, 1,000 SSBs and 40 DSBs per cell (16), although only the latter are
considered lethal (15) and
carcinogenic (16). Moreover,
there are indirect effects due to the radiolysis of water, which
creates reactive oxygen species (ROS), called the bystander effect,
that potentiate the effect of IR by increasing the number of DSBs
and propagating its effects on neighboring cells (17). After irradiation, the majority of
lesions are repaired by processes such as homologous recombination
repair in the synthesis (S)/growth (G)2 phase of the cell cycle
(16) and non-homologous end
joining (NHEJ) (15) in the G1
phase (16). The first is limited
to the S and G phases of the cell cycle, and the second is used to
repair DSBs independently of the cell cycle (15). IR activates the kinase activity of
ataxia-telangiectasia mutated (ATM), which interacts with a large
panel of proteins such P53, Brac2 and checkpoint kinase 2 (CHK2) to
pause the cell cycle, allowing DNA repair (15). Despite these reparations, a number
of these defects are not repaired or repaired with defects, leading
to cellular apoptosis or necrosis (15).
Mechanisms of RR
RR is defined by a reduced efficiency of RT emerging
through complex and interconnected mechanisms. An intrinsic RR
results from genetic or phenotypic alterations in response to XR
(15), whereas an extrinsic RR
can be due to the tumor environment (18). Hypoxic cancers, cancer stem cells
(CSCs) and altered metabolism favor RR (18-20), and modified cell cycle control and
DNA repair directly impact the efficiency of XR (21-24).
Intrinsic RR
Radiation induces the activation of a number of
genes, including early genes such as C-JUN, and epidermal
growth factor-1 (EGF-1) serves as regulator of other protective
genes (15), while later genes
are mostly associated with trophic factors such as platelet-derived
growth factor (PDGF), transforming growth factor-β (TGF-β) and
basic fibroblast growth factor expressed to modulate
radio-sensitivity (15).
Hypoxic microenvironment
Hypoxia reduces the production of ROS, which are
essential for radiation-induced DNA damage (19,20), activates cellular autophagy and
accelerates ROS clearance to further protect cancer cells (20). Hypoxia-inducible factor (HIF)
regulates vascular endothelial growth factor (VEGF) and PDGF gene
expression, protecting endothelial cells from radiation damage
while stimulating tumour blood vessel growth (19,20). HIF inhibits apoptosis by: i)
Stimulating NDRG2 and inhibiting BAX, a proapoptotic
gene; and ii) inhibiting p53-mediated apoptosis via direct
interaction with p53 (20).
Furthermore, HIFs are implicated in the activation of RR signaling
pathways such as CXCL8, AKT/mTOR/STAT3, MERK/ERK and DNA-protein
kinase (DNA-PK) (19,20).
CSCs
The presence of CSCs renders RT inefficient
(20). These cells express CD133
and CD44, with the latter being strongly upregulated post
radiation, promoting the stem phenotype and therefore RR (20). CSCs continuously modify their
environment to potentiate their survival (18); they are also implicated in
tumours, increasing the complexity of tumour treatment (18). The ability of stem cell-like cells
to replicate and differentiate provides more protection from RT;
however, a fraction of stem cells remain quiescent and are thus
unaltered (18). This process
facilitates CSC survival post-irradiation, followed by
proliferation and tumor invasion (18).
Altered cellular metabolism
Cellular metabolism adaptations are often observed
in RR cancer cells, notably through the Warburg effect, which
favors anaerobic glycolysis (20). This effect translates to an
increase in the glucose consumption rate, active glycolysis and a
high concentration of lactic acid (20). Lactic acid is found in large
quantities in the BM of patients with NSCLC and can stimulate the
release of hyaluronic acid by tumor-associated fibroblasts, which
promotes VEGF secretion and cell migration (20). A reduction in oxidative
phosphorylation in mitochondria reduces the rate of ROS generation
and renders the cell dependent on anaerobic glycolysis (20). Glucose transporter 1 (Glut1) is
stimulated under hypoxic conditions, during which its
overexpression is associated with RR (20). Moreover, Glut1 expression is
modulated by PI3K/AKT signaling, which is known for its oncogenic
functions (20). Additionally,
manganese superoxide dismutase (MnSOD) is an antioxidant enzyme
that is upregulated in certain cells after radiation and reduces
mortality by reducing ROS levels and preventing subsequent
apoptosis (20).
Cell cycle
The cell cycle has checkpoints that regulate the
passage of different phases to perform mitosis (21). The sensitivity of cells to
radiation varies during these phases; RT stops the cell cycle at a
checkpoint before the S phase (21). This checkpoint is used to repair
DNA before any errors are copied; if irreparable, the cell dies
(21). Dysregulation of the cell
cycle is observed in cancer cells (21). Proteins such as ATM, P53 and CHK2
regulate this process and are thus altered in cancer cells,
increasing RR (21). Activated
ATM can either activate CHK2, which, in turn, phosphorylates P53
stabilizing the protein, or sequester P53 in the nucleus (21). These proteins lead to powerful
cell cycle arrest in the S and G1 phases, allowing additional time
for DNA repair and increasing the survival rate of cancer cells
(21).
DNA repair enzymes
DNA repair enzymes are upregulated in RR cells to
reverse radiation-induced DNA damage and thus allow cells to
survive despite exposure to radiation (20). For instance, DNA-PKs and ATM are
implicated in NHEJ DNA repair, the major repair mechanism activated
post-irradiation (22). DNA-PK
recognizes DSBs in DNA and relinks the broken strands in a
non-homologous way, but this could favor the acquisition of new
oncogenic mutations (21).
Irradiation of the adenocarcinoma cell line A549 with inhibition of
DNA-PK results in a higher rate of DNA damage and apoptosis,
suggesting the involvement of DNA-PK in these mechanisms (22). Similarly, the inhibition of ATM in
irradiated cells reduces cellular survival and increases apoptosis
(22). Furthermore, in
EGFR-mutated NSCLC cells, the condensation of chromatin seems to
have radioprotective effects (23). Chromatin modification is a
survival mechanism used by cancer cells to protect their DNA from
radiation damage. The type of radiation (XR or carbon ion) does not
seem to modify cellular responses (24).
Signaling pathways involved in RR
Janus kinase (JAK)/signal transducer
and activator of transcription (STAT)
The JAK/STAT pathway involves fast-acting signaling
from the membrane to the nucleus activated in response to cytokines
and growth factors, leading to the activation of JAKs and
subsequent phosphorylation and activation of STAT proteins.
Activated STAT translocates to the nucleus to regulate the
transcription of genes involved in various cellular functions such
as differentiation, lipid metabolism, cell-cycle inhibition,
cell-cycle progression, apoptosis inhibition, etc (25). RT induces the production of IFN,
which binds to its receptor to activate the JAK/STAT pathway; this
also activates other pathways, such as the mTOR, NF-κB and MAPK
pathways (25). IFN activation of
the JAK/STAT pathway upregulates genes that control cell survival
and metabolism, DNA repair systems and immune protection (25). This pathway has been implicated in
RR lung cancer via an increase in STAT3 activation (25).
ERK/MAPK
The ERK/MAPK pathway is altered in a large portion
of NSCLCs, and confers invasive and metastatic properties to BM
(26). RT leads to the
phosphorylation of MEK1 and 2, which are activated following HER
activation (26). ERK modulates
NHEJ-mediated DNA repair by controlling ATM and ATM Rad3-related
but also homologous repair via DNA-PK, promoting cell survival
(26). Moreover, ERK promotes
G2/M cell cycle arrest, rendering cells more RR (26). Another possible mechanism is that
the proliferation signals induced by the ERK pathway stimulate
cells less exposed to irradiation or are already somewhat RR such
as CSCs, and these signals promote their proliferation to replace
dead cells (26).
PI3K/AKT/mTOR pathway
The PI3K/AKT/mTOR pathway is another pathway
upregulated and implicated in RR (27) in NSCLC and their BM (28). When this pathway is inhibited, a
reduction in colony formation post-irradiation has been observed in
prostate cancer cells, and these cells present more DSBs due to a
reduction in NHEJ DNA repair and autophagy (27). In NSCLC, the combination of PI3K
and extracellular matric (ECM) inhibitors prevents or delays RR
(28).
Wnt/β-catenin
Dysregulation of the Wnt/β-catenin pathway
contributes to RR by affecting the cell cycle, proliferation, DNA
repair, apoptosis and invasion (29). Indeed, Wnt is expressed at high
levels in CSCs known for their RR (29).
Hedgehog
The Hedgehog pathway regulates proliferation and
differentiation (30); one of its
members, Sonic hedgehog (Shh), exerts an inhibitory signal when
associated with its transmembrane receptor patched-1 (PTC-1)
(30). In addition, PTC-1 leads
to the expression of a transcription factor named glioma-associated
oncogene 1 (Gli1) (30). Gli1
regulates proliferation, differentiation, ECM interactions and stem
cell activation (30). Inhibition
of Gli1 in NSCLC slows the proliferation of RR CSCs and increases
radiosensitivity (30).
Hippo pathway
The Hippo pathway is a signaling pathway that
controls various biological functions, including cell growth,
survival, differentiation, determination of cellular fate, organ
size and tissue homeostasis (31); it is found at the crosstalk site
with all the others signaling pathways involved in RR, where it
confers metastatic properties in NSCLC (8,9).
The role of the Hippo pathway in RR is still unclear even if a
recent study of resistance to treatment induced by
YAP1/transcriptional coactivator with a PDZ-binding domain (TAZ)
revealed that 'whether YAP1/TAZ confers resistance to RT is an
important open question' (32).
The aim of the present review was to explore the potential
implications of Hippo in RR.
3. Hippo pathway in cancer and RR
Hippo pathway
The well-preserved center of the Hippo pathway is
primarily composed of a cascade of serine/threonine kinases,
including mammalian sterile 20-like kinase (MST1/5), NDR1/2 and
large tumour suppressor 1/2 (LATS1/2; Fig. 1) (31). Activated by phosphorylation, MST
kinases phosphorylate and activate NDR kinases (31). Active NDR kinases phosphorylate
and inhibit the downstream effectors YAP1 and TAZ by sequestering
them in the cytoplasm and directing them towards the proteasome
(31). In the absence of
phosphorylation, YAP1 and TAZ translocate to the nucleus and bind
to different transcription factors, such as TEA DNA-binding
proteins (TEAD 1-4) (31),
thereby regulating a variety of genes controlling proliferation,
stem cell renewal and survival through genes such as CTGF,
ANKDR1, CYR61, AXXL and BIRC5 (31). The panel of genes induced by
YAP-1/TAZ varies during organogenesis (Fig. 1), depends on the cell fate and
evolves between the non-oncogenic situation and the oncogenic
situation; during malignant transformation, the transcription of
the panel of genes promoted by YAP-1/TAZ is often exacerbated,
reflecting the hyperactivity of YAP-1/TAZ in cancer (31,32). Notably, the Hippo pathway is also
regulated by multiple pathways, resulting in the control of the
transcriptional activity of YAP/TAZ (31). For example, the mitogen-activated
protein kinase 4 and the thousand and one amino-acid kinase
directly phosphorylate LATS1/2, thereby working in tandem with
MST1/5 (31).
Hippo pathway in human cancers including
metastatic lung cancer
In most human cancers, including metastatic lung
cancer, the Hippo pathway is highly disrupted (Fig. 2) (31,32). In NSCLC, the upregulation of YAP1
sustains cancer cell invasion, drug resistance and metastasis
(8-11). The hyperactivation of YAP1 in
NSCLC results from various factors: Epigenetic dysregulation that
silences Ras association domain-containing protein 1
(RASSF1) expression (8,14),
hypoxia induced by tumour growth (33) or the presence of oncogenic drivers
such as EGFR-activating mutations (10). Hippo pathway deregulation leads to
BM formation in lung cancer cells (8-11).
Hypermethylation of the RASSF1 promoter blocks the negative
control of RASSF1A, a regulator of the Hippo pathway, on YAP1,
leading to its nuclear accumulation (8). RASSF1A prevents the
epithelial-to-mesenchymal transition (EMT) of human NSCLC cells via
the inhibition of YAP1 by NDR2/Rho guanine nucleotide exchange
factor H1/RhoB signaling (8).
Similarly, the inhibition of YAP1 by the RASSF1A gene blocks
metastasis formation in a murine model (8). In cellular models of BM from NSCLC,
H2030-BrM3 and PC9-BrM3 cells, YAP1 is overexpressed compared with
that in the parental cell lines H2030 and PC9 (34), and direct inhibition of YAP1 by
short-hairpin RNA (sh-RNA) blocks BM formation from H2030-BrM3 in a
murine model (10), which
supports that YAP1 is involved in metastasis formation.
Finally, YAP1 is implicated in MAPK/ERK signaling, a
pathway involved in RR, following EGFR mutation (10): Forced expression of YAP1 confers
EGFR-TKI resistance in NSCLC cells, while YAP1 inhibition
potentiates this effect (35).
Activated YAP1 increases the expression of certain EGFR ligands,
such as AREG and ERBB3/4, and thus promotes MAPK signaling to
induce tumour progression and drug resistance (10). Correspondingly, EGFR/MAPK
signaling regulates YAP1 via the inhibition of Hippo kinases
(35). YAP1 controls the
transcriptional regulation of programmed cell death-ligand 1 via
its interaction with TEAD, leading to the suppression of the
immune-related antitumoral response (36). These discoveries highlight the
importance of different drug development approaches targeting YAP1
(10). Indeed, inhibitors of the
interactions between YAP1/TAZ and TEADs have been tested using
in vitro and in vivo methods (31,32), with select candidates such as
VT3989 proceeding to clinical trials (37).
Role of the Hippo pathway in RR
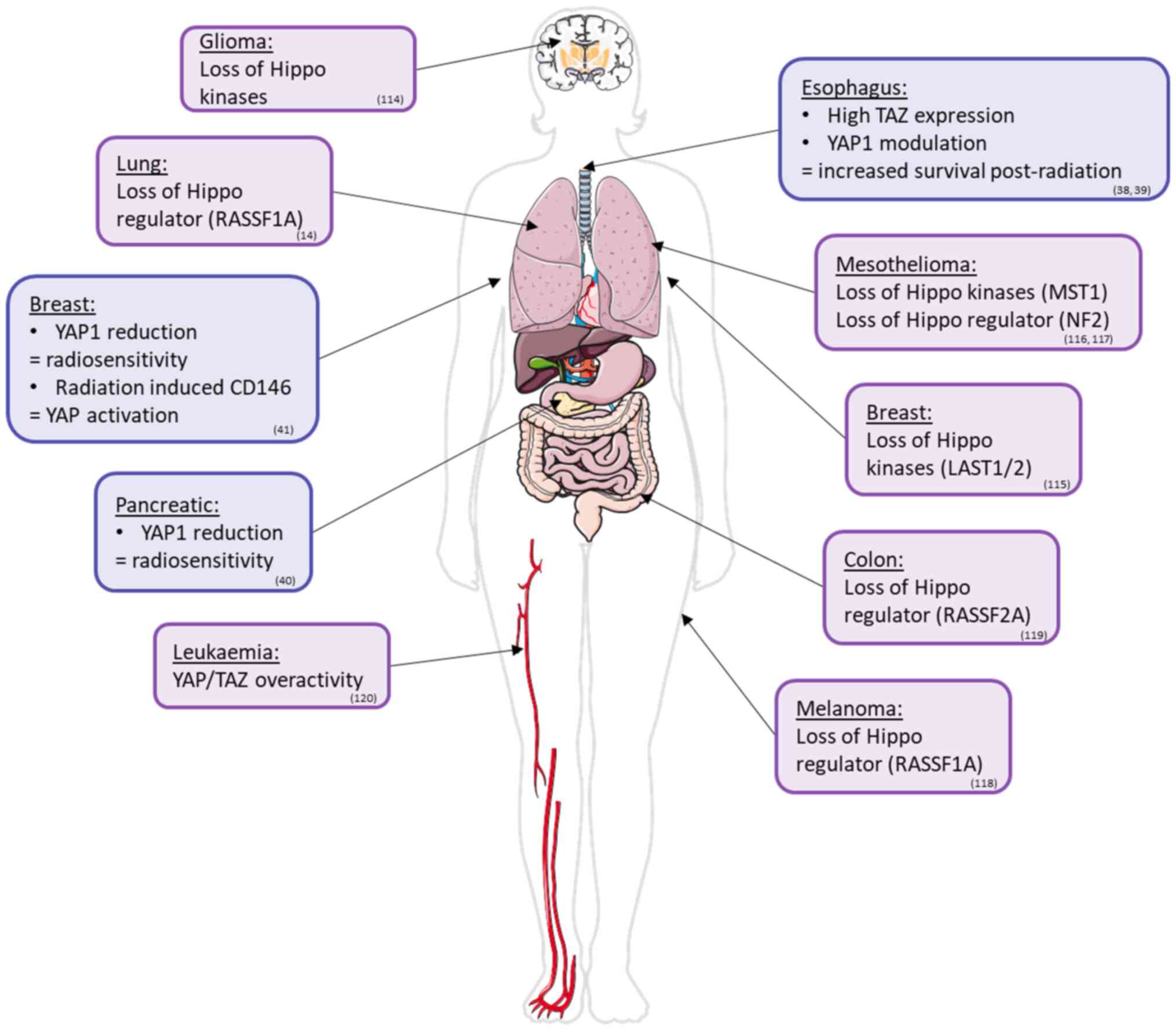
There is previous evidence of a link between the
Hippo pathway and RR in certain cancers (Table I). For instance, esophageal cancer
cells strongly expressing TAZ survive longer than cells with
reduced TAZ expression post-radiation (38). CD155 stimulates RR via modulation
of YAP1 phosphorylation (39).
Indeed, the overexpression of CD155 increases the quantity of
nuclear YAP1, whereas its inhibition favors cytoplasmic
localization (39). A reduction
in the expression of YAP1 and its target genes following catechol
treatment sensitizes pancreatic cancer cells to irradiation
(40). The inhibition of YAP1 by
shRNA in breast cancer cells triple negative for hormone receptors
increased the sensitivity to irradiation compared to that in shRNA
control cells (41). Finally,
YAP1 is translocated to the nucleus, where its activity is
essential for maintaining survival and proliferation signaling
after irradiation (41). In the
same breast cancer model, irradiation appeared to stimulate CD146,
which inhibits LATS1, in turn favoring YAP1 activation (42). This is associated with RR due to
DNA repair, cell cycle arrest and stem characteristics (42).
 | Table IEffects of Hippo modulation
post-radiation. |
Table I
Effects of Hippo modulation
post-radiation.
| First author(s),
year | Cancer type | In
vitro/In vivo | Model | IR dose, Gy | Hippo
modulation | Result | (Refs.) |
|---|
| Moon et al,
2021 | Pancreatic | In
vitro | Panc-1 cells | 2, 4 | Catechol
treatmenta | Decreased survival
fraction | (40) |
| Zhou et al,
2020 | Esophageal | In
vitro | Eca109, Kyse150 and
TE1 cells | 2, 4, 6, 10 |
SiTAZa | Decreased survival
fraction
Increased DNA damage | (38) |
| TAZ overexpression
via plasmidsb | Increased surviving
fraction
Reduced DNA damage |
| Xin et al,
2022 | | In
vitro | Eca109 and Kyse510
cells | 3, 6, 9 |
ShCD155a | Decreased nuclear
YAP
Decreased proliferation and migration | (39) |
| Andrade et
al, 2017 | Breast | In
vitro | MDA-MB231 and MDA-
MB468 cells | 2, 4, 6 |
ShYAP1a | Decreased survival
fraction | (41) |
| Liang et al,
2022 | | In vivo | Xenomorphic 4 (x3
radiation injection of MDA-MB-231 cells in mice | cycles) | YAP
overexpressionb | Increased tumour
growth | (42) |
| Zhang et al,
2021 | Glioma | In
vitro | U87 and U251
cells | 4, 6, 8, 10 | YAP overexpression
via vectorsb | Increased surviving
(108) fraction
Increased DNA repair | |
| In vivo | Xenomorphic
injection of U251 and GBM1 cells in mice | 10 | YAP overexpression
via vectorsb | Decreased tumour
size
Decreased survival | |
| Zeng et al,
2020 | Non-small cell lung
cancer | In
vitro | A549 and H1299
cells | 2, 4, 6, 8 | SiCDK5 (Hippo
modulator)a | Decreased DNA
damage | (12) |
| | In vivo | Xenomorphic
injection of H1299 cells in mice | 10 | ShCDK5 (Hippo
modulator)a | Decreased size of
tumour
Decreased DNA damage | |
| Li et al,
2022 | | In
vitro | A549, H1299 and
H460 cells | 2, 4, 6, 8 | p130cas (YAP
modulator) overexpression via vectorsb ShP130cas (YAP
modulator)a | Increased survival
fraction
Decreased DNA damage
Increased DNA damage
Increased tumour size | (44) |
| In vivo | Xenomorphic
injection of H1299 cells in mice | 8 (x3) | p130cas (YAP
modulator) overexpression via vectorsb | |
In SCLC, YAP1 overexpression is associated with an
unfavorable prognosis in patients following an irradiation protocol
because RR is modulated by CD133 expression associated with YAP1
expression (43). The Hippo
pathway is also implicated in NSCLC RR via an increase in TAZ
transcription (12). CDK5 is an
upstream regulator of the Hippo pathway; when CDK5 is silenced by
shRNA, the expression of TAZ decreases (12). This inhibition leads to an
increase in DSBs (γH2AX) and a decrease in DNA repair (RAD51) after
radiation in A549 cells (12).
Breast cancer anti-estrogen resistance protein 1 (p130cas)
interacts with and promotes via focal adhesion kinase the
stabilization of YAP1 when overexpressed, resulting in RR in NSCLC
cells (44). Inhibition of YAP1
with verteporfin restores the number of DSBs back to a normal level
after p130cas is overexpressed, thereby restoring radiation
efficiency (44). This evidence
shows an implication of the Hippo pathway in RR in different types
of cancers, yet its role in BM formation from NSCLC has yet to be
studied.
4. Key factors in RR and the Hippo
pathway
Mechanisms of RR and Hippo signaling
Numerous aspects of the cellular environment alter
the effectiveness of irradiation (Fig. 3). Cells can switch to different
states; for example, cells can dedifferentiate into a stem
phenotype or increase control of the cell cycle (45-51). These changes favor resistance to
RT (45-51). Finally, the mechanisms of DNA
repair (12,38,52), apoptosis regulation (53,54) and metabolic dysregulation
(55-57) also greatly alter the success of
RT. All of these mechanisms interact with and alter members of the
Hippo pathway in a large variety of cell types (Fig. 3).
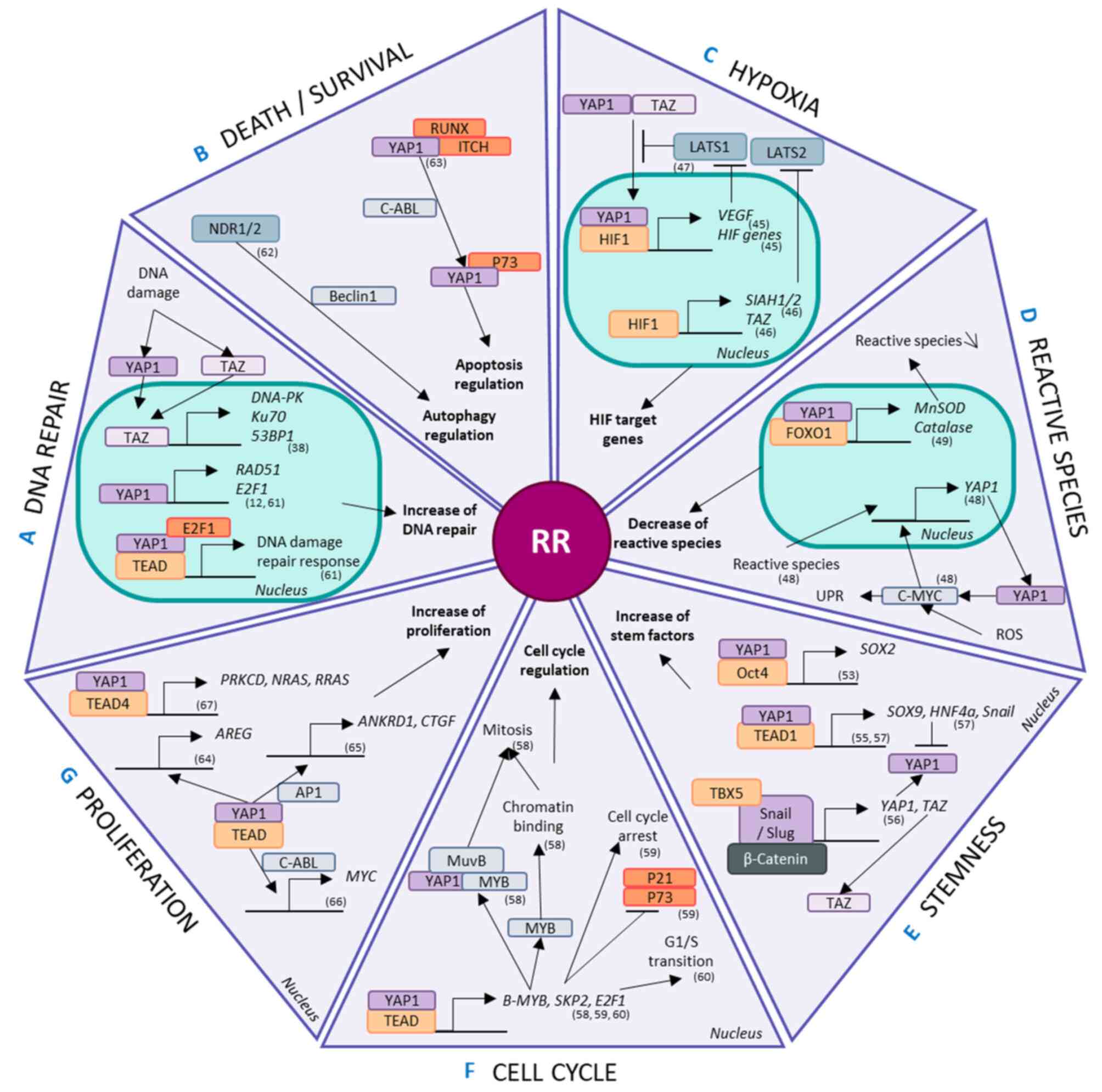 | Figure 3Involvement of the Hippo pathway in
radioresistance. Hippo effectors are modulated and implicated in
radioresistant phenomena, such as (A) DNA repair, (B) cell death
and survival regulation, (C) hypoxia, (D) reactive species
modulation, (E) stem properties, (F) cell cycle regulation and (G)
proliferation. YAP1 and/or TAZ regulate the transcription,
activation and regulation of mechanisms related to these phenomena.
MST1/5, mammalian sterile 20-like kinase; LATS1/2, large tumour
suppressor 1/2; NDR1/2, nuclear dbf2-related kinase; YAP1,
yes-associated protein 1; TAZ, transcriptional coactivator with a
PDZ-binding domain; TEAD, TEA DNA-binding protein; ROS, reactive
oxygen species; DNA-PK, DNA-protein kinase; MnSOD, manganese
superoxide dismutase; UPR, unfolded protein response; HIF,
hypoxia-inducible factor; VEGF, vascular endothelial growth factor;
RUNX, Runt-related transcription factor; ITCH, Itchy E3 ubiquitin
protein ligase; E2F1, E2F transcription factor 1; RAD51, RAD51
recombinase; PRKCD, protein kinase C δ; AREG, amphiregulin; ANKRD1,
ankyrin repeat domain-containing protein 1; CTGF, connective tissue
growth factor; AP1, activator protein 1; MYB, myeloblastosis viral
oncogene homolog; SKP2, S-phase kinase-associated protein-2; SIAH,
Siah E3 ubiquitin protein ligase. |
The Hippo pathway interferes with hypoxia through
the interaction of HIF1 with YAP1, inducing the expression of HIF
target genes (58). Furthermore,
in hypoxic conditions involving breast cancer stem cells, HIF1
stimulates the expression of the E2 ubiquitin ligases Siah E3
ubiquitin protein ligase (SIAH) 1 and 2, promoting the proteasomal
degradation of LATS2 (59). This
in turn favors the nuclear localization of YAP1 and TAZ. HIF1
increases TAZ expression by binding to the WWTR1 gene
(59). Moreover, using a
biosensor that monitors LATS kinase activity, VEGFR activation by
VEGF has been shown to inhibit LATS and activate the Hippo
effectors YAP1 and TAZ, highlighting the implication of the Hippo
pathway in neoangiogenesis (60).
There is evidence showing that reactive species
increase the mRNA and protein levels of YAP1, controlling the
proliferation of hepatocellular carcinoma cells (55). ROS-exposed cells act via the c-myc
pathway to stimulate YAP1 expression and lead to an increase in the
unfolded protein response (55).
Then, YAP1 coactivates the transcription of FOXO1, an essential
protein for antioxidant gene expression, in myoblast cells
(56). The binding of YAP1 and
FOXO1 to the promotor regions of antioxidant genes such as MnSOD
and catalase activates their transcription (56). These genes are also implicated in
cellular metabolism and are modified in a number of cancers such as
glioma, lung, breast, pancreatic, esophagus, mesothelioma, colon,
melanoma and leukemia because they adapt to their hostile new
microenvironment in part via the aid of YAP1 and TAZ (61). YAP1 upregulates Glut3 via binding
with TEAD in its promoter region in a kidney cell line (62), prioritizing glucose uptake in
these cancer cells and allowing greater energy usage. Consequently,
YAP1 favors the clearance of reactive species, decreasing the
efficiency of secondary IR effects and altering cellular
metabolism.
CSCs are maintained by a number of factors that
control stemness and dedifferentiation, such as Sox2, Sox9,
Snail/Slug and HNF4a (61). In
osteosarcomas, the Hippo pathway is a downstream effector of
Sox2-mediated stem maintenance, and this relationship is
antagonized by Nf2/WWC1 (63).
This regulatory effect has also been found in other cell types,
including an immortalized murine fibroblast line and primary
cultures of human glioblastoma cells (63). The loss of YAP1 decreases the
self-renewal potential of NSCLC stem cells (45). The interaction between the YAP1
and Oct4 transcription factors regulates Sox2 expression in these
NSCLC stem cells (45). However,
TEAD4 and YAP1 repress the expression of Sox2 in the early stages
in murine blastocysts through LATS1 control (64), suggesting that the functions of
YAP1 are altered depending on the stage of life. YAP1 and its
co-factor TEAD1 have been shown to regulate the transcription of
other stem factors, such as Sox9, promoting CSC-like properties in
esophageal cancer cells (46).
However, Hippo signaling pathways are regulated by several stem
factors. For example, in mesenchymal stem/stromal cells, Snail/Slug
mediate YAP1 and TAZ expression in association with β-catenin and
TBX5 (47). However, YAP1 also
directly controls the transcription of Snail and HNF4a via TEAD in
epithelial and hepatocyte cells (48). Despite this, HNF4a negatively
regulates the expression of YAP1 in hepatocytes (48). YAP1 represses the differentiation
of epithelial cells and hepatocytes by regulating MET genes
(48).
In combination, myeloblastosis viral oncogene
homolog (Myb)-MuvB and YAP1 regulate genes implicated in the cell
cycle and, notably, mitosis via direct action of YAP1 (49). B-MyB expression is under the
control of YAP1 and TEAD via a distal enhancer, favoring the
chromatin-binding activities of B-MyB required for mitosis
(49). Moreover, the physical
interaction of MyB-MuvB with YAP1 seems necessary for YAP1-induced
cell cycle progression (49).
YAP1 and TEAD respond to mechanical stress signals to control the
transcription of S-phase kinase-associated protein-2 (SKP2)
(50). SKP2 overexpression
inhibits its substrates, p21 and p73, and mediates cell cycle
arrest induced by YAP1 depletion (50). Furthermore, E2F1 is a downstream
target of YAP1 that regulates the G1/S transition through TEAD
(51). YAP1 and TEAD therefore
regulate the cell cycle.
The ability of Hippo pathway effectors to increase
DNA repair has already been explored in a number of different
models, including NSCLC (12).
YAP1 forms a complex with TEAD2 and E2F1 to regulate the cellular
response to DNA damage via the expression of Fanconi anemia
components (52). Similarly, TAZ
overexpression increases the expression of TP53BP1,
PRKCD and XRCC6 which are associated with the 53BP1,
DNA-PK and Ku70 proteins, respectively, which are implicated in the
NHEJ DNA repair mechanism in esophageal cancer (38).
NDR1/2, upstream regulators of YAP1 and TAZ,
contribute to the autophagic response to stress through a process
thought to involve Beclin1, a major player in autophagy (53). C-ABL favors the formation of a
YAP1/p73 complex that dissociates both RUNX and ITCH from YAP1
(51). The p73/YAP1 complex
controls apoptosis, particularly after DNA damage via C-ABLs
(51). In hepatocellular
carcinoma, YAP1 inhibition induces apoptosis under hypoxic
conditions compared normoxic conditions, which appears to be
associated with HIF1 (54). Thus,
the Hippo pathway is intertwined with autophagy and apoptosis
regulation, which controls cell survival.
Despite all these factors participating in RR, a
simple increase in proliferation also plays a part, and controlling
proliferation is a well-known aspect of the Hippo pathway. YAP1 and
TEAD regulate the expression of AREG (65), ANKRD1 and CTGF with
the aid of AP1 (66), SKP2
stabilized by p300 (50) and Myc
via the interaction of YAP1 with C-ABL (57), all of which play a role in
proliferation. Additionally, proliferation is controlled by Hippo
signaling through the interaction of YAP1 with TEAD4, which
specifically regulates the expression of PRLCD, NRAS
and RRAS (67).
Signaling pathways implicated in RR and
Hippo signaling
The driving pathways of RR involve varying levels of
interaction and crosstalk with the Hippo pathway and its
components, which could lead to RT failure (Fig. 4).
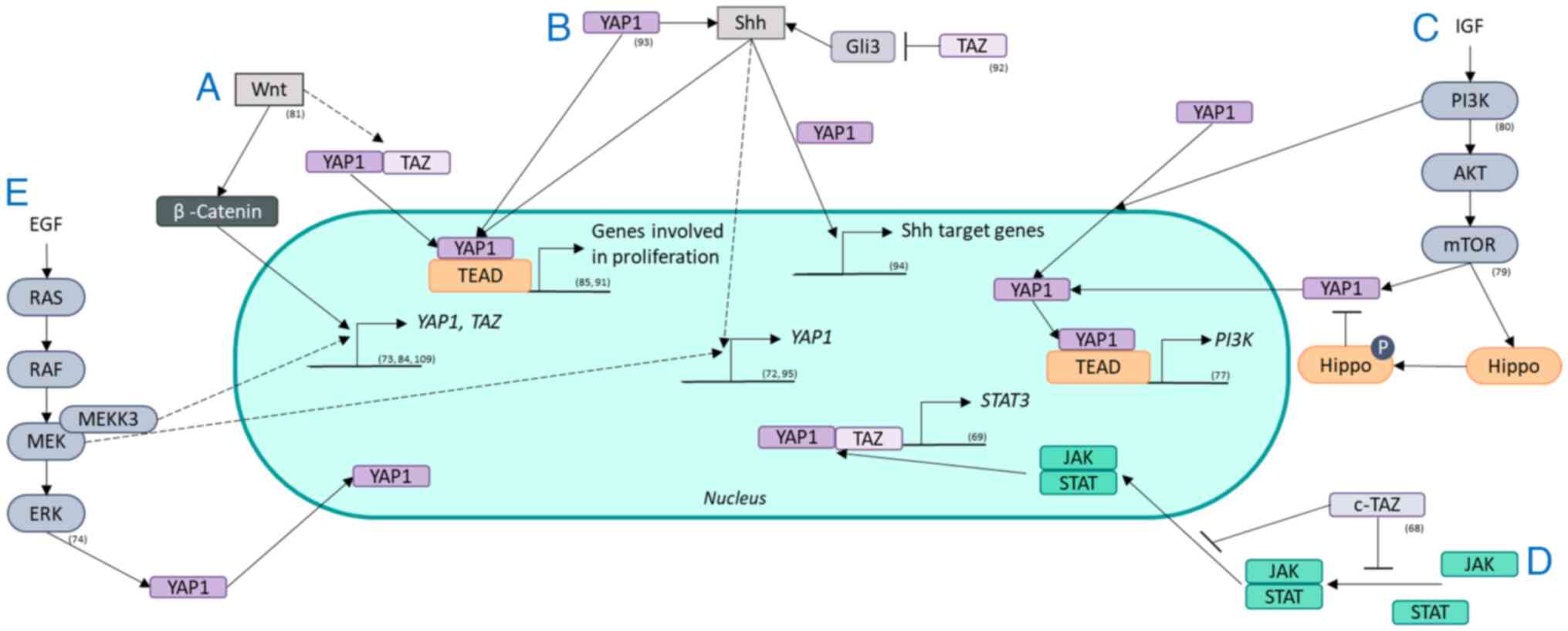 | Figure 4Crosstalk between radioresistance
signaling pathways and the Hippo pathway. The Hippo pathway
interacts with pathways known for radioresistance, such as (A) the
Wnt pathway, (B) the Shh pathway, (C) the PI3K/AKT/mTOR pathway,
(D) the JAK/STAT axis and (E) the RAS/RAF/MEK/ERK pathway. The
Hippo pathway and its effectors YAP1 and/or TAZ are regulated by
these pathways through transcription, localization and activity.
YAP1 and/or TAZ also regulate the activity of these pathways
through transcriptional regulation. YAP1, yes-associated protein 1;
TAZ, transcriptional coactivator with a PDZ-binding domain; EGF,
epidermal growth factor; Shh, Sonic hedgehog; P, phosphorylated;
JAK, Janus kinase; STAT, signal transducer and activation of
transcription; Gli, glioma-associated oncogene; IGF, insulin-like
growth factor. |
cTAZ is an isoform of TAZ that is not regulated by
TEAD which suppresses the JAK/STAT pathway by blocking dimerization
and nuclear transport of STAT factors that control antiviral
responses (68). YAP1 and TAZ
increase the transcription of STAT3 components capable of reacting
to oncogenic RAS and inflammation in pancreatitis (69) revealing the possible interplay of
these factors in radiation-stressed cells. The JAK/STAT pathway is
constitutively active in NSCLC (70). In NSCLC cells, an RR effect is
potentiated by the microenvironment of the cells via JAK/STAT
signaling (71).
EGF and insulin stimulate YAP in different human and
Drosophila cells via MAPK signaling; however, this interaction has
not been systematically investigated in another review (58). Specifically, MEK1 inhibition
reduces the expression and activity of YAP1, revealing that YAP1 is
regulated via MEK1, which is independent of the core Hippo pathway
and promotes tumorigenesis in liver cancer cells (72). MEKK3 regulates YAP1 and TAZ at the
transcriptional level in pancreatic cancer cells, promoting EMT and
stemness (73). Moreover, ERK1/2
regulate YAP1 protein expression to increase the viability and
invasion of NSCLC cells (74).
The regulation of YAP1 and TAZ by MEK in NSCLC cells was found
(74). YAP1 is implicated in
MAPK/ERK signaling via EGFR mutations in NSCLC through an increase
in the expression of EGFR ligands, such as AREG and
ERBB3/4, subsequently increasing MAPK signaling (10). Furthermore, EGFR/MAPK signaling
inhibits the phosphorylation and degradation of YAP1 by the Hippo
kinase (35). These interactions
between ERK/MAPK and the Hippo pathway play a part in stimulating
proliferation signals after stress, limiting the effects of RT.
Along with Src and PDK1, PI3K regulates the nuclear
localization of YAP1, favoring its activity (75). PI3K regulates both YAP1 and TAZ in
breast cancer via PDK1 and AKT signaling which play a role in
tumorigenesis (76). YAP1, in
turn, activates PI3K/AKT/mTOR signaling in human bronchial
epithelial cells via TEAD, leading to increased proliferation
(77). In medulloblastoma cells,
YAP1 increases the RR via IGF2/AKT signaling (78). An effector of this pathway, mTOR,
phosphorylates the Hippo pathway and favors YAP1 activity,
stimulating the proliferation and invasion of glioblastoma cells
(79). In colorectal cancer, the
PI3K/AKT pathway activates YAP1, leading to increased invasion and
migration (80).
The Wnt pathway regulates YAP1 and TAZ, similar to
β-catenin (58); ligands from
this pathway activate YAP1 and TAZ through the frizzled receptor
LATS1/2 and Rho-GTPases instead of the typical β-catenin pathway
(81). This TEAD-mediated
signaling leads to the expression of various genes: Osteogenic
differentiation and cell migration (81). In liver cancer, Tribbles
pseudokinase 2, a direct target of the Wnt pathway, stabilizes the
coactivation factor of YAP1 transcription (82). YAP1 and TAZ are activated by
oncogenic pathways such as the Wnt pathway (83). YAP1 transcription is elevated by
Wnt/β-catenin signaling in colorectal carcinoma cells (84). TAZ is regulated by the Wnt pathway
and increases the proliferation of colorectal cancer cells but also
controls mesenchymal stem cell differentiation (85). YAP1 stimulates Wnt/β-catenin
signaling in epithelial cells experiencing inflammation and
regeneration through the targeting of CDK5 (86). Specifically, YAP1 regulates Wnt
pathway activity differently depending on its localization;
cytoplasmic YAP1 inhibits Wnt pathway activity, whereas nucleic
YAP1 activates Wnt pathway activity (87). Inhibition of the Wnt pathway
increases sensitivity to radiation in NSCL (88). The interplay of inhibition or
activation of one another by the Hippo and Wnt pathways indicates
that there is a complex relationship between these pathways
possibly causing RR.
YAP1 blocks Shh-induced differentiation in smooth
muscle cells (89). However, when
YAP1 is inhibited in embryonic lung cells, the expression of Shh
and its target genes decreases (90). YAP1 and TAZ also regulate the Shh
pathway in epithelial lung cells (90). Human medulloblastoma cells develop
from cerebellar granule neuron precursors activated by the Shh
pathway and from high levels of YAP1 (91). In mouse Shh-induced
medulloblastomas, YAP1 is also upregulated (91). In both of these cell types, YAP1
interacts with TEAD1, leading to Shh-driven proliferation (91), confirming the regulation of Shh by
YAP1. On the other hand, TAZ suppresses the Shh pathway in in
vitro and in in vivo models, potentially through Gli3
repression (92). The cell
density regulation of Shh is regulated by the Hippo pathway
effector YAP1 (93). YAP1
controls proliferation and inhibits differentiation in a mouse
embryonic carcinoma cell line and increases the expression of Shh
signaling and patched 1, a downstream effector of Shh (94). However, the relationship between
these two pathways is not unilateral. Shh also regulates YAP1
activity via the hedgehog protein in regenerating murine liver
cells (95), revealing a feedback
loop. Since Shh activation increases the RR in NSCLC cells, these
interactions are noteworthy possible mechanisms (30).
Influence of radiation on the regulation
or dysregulation of the Hippo pathway
Although glycosylation, methylation and
hypermethylation of Hippo members can influence their function and
activity, to our knowledge, the roles they play in response to
radiation exposure have not been reported.
Ubiquitination is an alternative modification
permitting the control of various signaling pathways, including the
Hippo pathway (96). β-transducin
repeat containing E3 ubiquitin protein ligase is an E3 ligase that
targets YAP1 and TAZ, leading to a reduction in their activity
(96). Contrary to the regulation
of LATS1/2 by other upstream E3 ligases, ITCH promotes growth and
survival, and a hypoxia-activated E3 ligase, SIAH, promotes
oncogenic YAP1 activation (97).
Ubiquitin is overexpressed in a number of different NSCLC cell
lines and implicated in increased growth (98). Silencing UBB and
UBC, two key genes involved in the ubiquitination process,
decreases cellular growth and increases radiosensitivity, as shown
through pH2AX staining (98).
Furthermore, ubiquitination of the Hippo pathway is regulated at
both the transcriptional and post-translational levels and is
implicated in the maintenance of CSC stemness (99).
5. Radiopotentiation and the Hippo
pathway
Known methods of radiopotentiation
For advanced solid cancers, chemoRT refers to the
use of irradiation combined with molecular targeting to render the
tumour more radiosensitive (100). These molecular targets fall into
four major categories: i) Growth factor receptor signaling
inhibition; ii) targets of the DNA damage response and cell cycle
checkpoints; iii) cell adhesion molecules; and iv) heat shock
proteins (100). The most
commonly used drugs for the treatment of NSCLC or BM from NSCLC
fall into the first and second categories. For clinical treatment
of BM from NSCLC in patients with EGFR mutations, third-generation
TKIs, such as osimertinib, are used; these drugs fall into the
first category of targeted drugs because of their greater ability
to penetrate the central nervous system compared with previous
generations (6). In the case of
ALK rearrangements, lorlatinib, another third-generation TKI, is
used to target the BM of patients with NSCLC after the failure of
second generation TKIs such as alectinib and clertinib (6). The TGF-β1 inhibitor, SB431542, also
induces radiopotentiation in NSCLC cell lines depending on the p53
status of the cells (101).
Inhibition of P1K1 in p53 wild-type NSCLC cells induced
radiosensitivity, but this effect was not found in mutated p53
cells (102). In the second
category, the effects of various combinations of DNA damage
response inhibitors on NSCLC cell lines have been investigated
through the profiling of biomarkers and different genetic
alterations (103).
Eurycomalactone induces G2/M cell cycle arrest, a known
radiosensitive phase of the cell cycle, and delays the repair of
DSBs in NSCLC cells (104). A
poly (ADP-ribose) polymerase inhibitor increases the
radiosensitivity of the NSCLC cell line A549 (105). DNA damage response inhibitors
are also being studied for their potential radiopotentiating
effects on glioblastomas (106),
demonstrating their ability to potentially cross the blood brain
barrier which is the critical step in treating brain cancers such
as BM from NSCLC.
Radiopotentiation using the Hippo
pathway
Targeting the Hippo pathway to sensitize cancer
cells to irradiation is a promising idea. As it was aforementioned,
high levels of YAP1 and/or TAZ are associated with a poor RT
response in most cancers, including NSCLC (43). Post-radiation activation of these
factors has also been found in breast cancer (42) and metastatic breast cancer
(107), and their expression is
stable in NSCLC cells (44). YAP1
inhibition has radiopotentiating effects on pancreatic cancer
(40), gliomas (108) and NSCLC (12).
The Hippo pathway is implicated in a number of the
processes and pathways sustaining RR that are already targeted by
specific drugs. YAP1 and/or TAZ have been shown to respond to
hypoxia (58-60) and be induced by and decrease
reactive species (55,56,61,62). These factors also play important
roles in a feedback loop to maintain stem cell properties (45-48,63,64) and increase DNA repair (12,38,42,52,78,108). YAP1 and TAZ also regulate the
cell cycle (22,49,51), autophagy (53) and apoptosis (54,96). The Hippo pathway effectors
regulate certain factors in the JAK/STAT pathway (68,69) and the PI3K/AKT/mTOR pathway,
stimulating and activating YAP1 (75-80). YAP1 is increased and activated by
the ERK/MAPK pathway (72-74),
the Wnt pathway (58,81-87) and the Hedgehog pathway, which also
uses YAP1 as a transcription factor (89-95).
Inhibition of YAP1/TAZ
The use of drugs to inhibit YAP1 and TAZ is a
common technique for studying their implications. For example,
catechol treatment reduces the protein levels of YAP1 and its
target genes through AMPK phosphorylation, sensitizing pancreatic
cancer cells to irradiation (40). Verteporfin, a small inhibitor of
the interaction of YAP1 with TEADs, decreases the number of DSBs
back to a normal level after p130cas is overexpressed, restoring
radiation efficiency in NSCLC (44). This drug is safe when administered
via intraperitoneal injection at a dose of 100 mg/kg in mice
(44). Furthermore, verteporfin
is approved by the Food and Drug Administration (FDA), and is known
to decrease the proliferation and migration of glioma cell lines
(109). As a lipophile,
verteporfin can penetrate the brain at nontoxic doses and is
capable of inhibiting nuclear YAP1 in mouse models in vivo
(109).
Anti-YAP1/TAZ treatments are not yet available, but
IK-930, an oral TEAD inhibitor, is currently in phase 1
(NCT05228015) clinical trial for treating solid tumours. IK-930
blocks autopalmitoylation of TEAD by inhibiting TEAD-dependent
transcription of YAP1 and TAZ (110). TEAD palmitoylation inhibitors
stop mesothelioma cell line proliferation and block xenograft
growth (111). However, TEAD
palmitoylation inhibition increases VGLL3-mediated transcription of
PIK3C2B and Sox4, which activate AKT signaling, contributing to
cancer survival (112).
6. Summary and perspectives
NSCLC is a harsh disease in which 50% of patients
develop BM (2-4), resulting in a mere 19% 5-year
survival rate (113) despite the
use of a treatment plan that includes both surgery and RT (4). RR remains a major hurdle in the
treatment of BM from NSCLC. Notably, targeting the Hippo pathway to
provoke radiopotentiation of BM from NSCLC due to its a number of
potential implications for RR phenomena and the existence of
inhibitors, such as IK-930, in a phase I clinical trial (110) or with FDA approval, such as
verteporfin (109) is promising.
However, a better understanding of the role of Hippo in RR and thus
the potentially unforeseen side effects of targeting this pathway
in cancer treatment of healthy cells would also be beneficial.
Availability of data and materials
Not applicable.
Authors' contributions
JT and GL conceptualized the study; EB and GL
acquired funding, provided project administration and supervised
the study. Data were validated by JT, GL, FD and EB. JT and GL
wrote the original draft which was reviewed and edited by JT, GL,
FD and EB. All authors have read and approved the final version of
the manuscript. Data authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Abbreviations:
|
ATM
|
ataxia-telangiectasia mutated
|
|
BM
|
brain metastasis
|
|
CHK2
|
checkpoint kinase 2
|
|
CSC
|
cancer stem cell
|
|
DNA-PK
|
DNA-protein kinase
|
|
DSB
|
double-strand break
|
|
EGF
|
epidermal growth factor
|
|
EMT
|
epithelial-to-mesenchymal
transition
|
|
GF
|
growth factor
|
|
Gy
|
gray
|
|
GLI
|
glioma-associated oncogene
|
|
HIF
|
hypoxia-inducible factor
|
|
IR
|
ionizing radiation
|
|
JAK
|
Janus kinase
|
|
LATS
|
large tumour suppressor
|
|
MnSOD
|
manganese superoxide dismutase
|
|
MST
|
mammalian sterile 20-like kinase
|
|
NDR
|
nuclear dbf2-related
|
|
NHEJ
|
non-homologous end joining
|
|
NSCLC
|
non-small cell lung cancer
|
|
p130cas
|
breast cancer anti-estrogen
resistance protein 1
|
|
PDGF
|
platelet-derived growth factor
|
|
PTC1
|
patched 1
|
|
ROS
|
reactive oxygen species
|
|
RR
|
radioresistance/radioresistant
|
|
RT
|
radiotherapy
|
|
S
|
synthesis
|
|
Shh
|
Sonic hedgehog
|
|
SSB
|
single-strand break
|
|
STAT
|
signal transducer and activator of
transcription
|
|
TAZ
|
transcriptional coactivator with a
PDZ-binding domain
|
|
TEAD
|
TEA DNA-binding protein
|
|
TGF-β
|
transforming growth factor-β
|
|
TKI
|
tyrosine kinase inhibitor
|
|
VEGF
|
vascular endothelial growth
factor
|
|
XR
|
x-ray
|
|
YAP1
|
yes-associated protein-1
|
Acknowledgements
The authors thank the University Hospital of Caen
for its financial and institutional support. Figures were partly
created using Servier Medical Art, provided by Servier, licenced
under a Creative Commons Attribution 3.0 Unported Licence
(https://creativecommons.org/licenses/by/3.0/).
Funding
The present study was funded by the French Foundation for
Medical Research (grant no. FRM2022).
References
|
1
|
Wood SL, Pernemalm M, Crosbie PA and
Whetton AD: The role of the tumor-microenvironment in lung
cancer-metastasis and its relationship to potential therapeutic
targets. Cancer Treat Rev. 40:558–566. 2014. View Article : Google Scholar
|
|
2
|
Dawe DE, Greenspoon JN and Ellis PM: Brain
metastases in non-small-cell lung cancer. Clin Lung Cancer.
15:249–257. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fenske DC, Price GL, Hess LM, John WJ and
Kim ES: Systematic review of brain metastases in patients with
non-small-cell lung cancer in the United States, European Union,
and Japan. Clin Lung Cancer. 18:607–614. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Myall NJ, Yu H, Soltys SG, Wakelee HA and
Pollom E: Management of brain metastases in lung cancer: Evolving
roles for radiation and systemic treatment in the era of targeted
and immune therapies. Neurooncol Adv. 3(Suppl 5): v52–v62.
2021.PubMed/NCBI
|
|
5
|
Dempke WCM, Edvardsen K, Lu S, Reinmuth N,
Reck M and Inoue A: Brain metastases in NSCLC-are TKIs changing the
treatment strategy? Anticancer Res. 35:57972015.PubMed/NCBI
|
|
6
|
Ernani V and Stinchcombe TE: Management of
brain metastases in non-small-cell lung cancer. J Oncol Pract.
15:563–570. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jiang Y, Xie WJ, Chen RW, You WW, Ye WL,
Chen H, Chen WX and Xu JP: The Hippo signaling core components YAP
and TAZ as new prognostic factors in lung cancer. Front Surg.
9:8131232022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Dubois F, Keller M, Calvayrac O, Soncin F,
Hoa L, Hergovich A, Parrini MC, Mazières J, Vaisse-Lesteven M,
Camonis J, et al: RASSF1A suppresses the invasion and metastatic
potential of human non-small cell lung cancer cells by inhibiting
YAP activation through the GEF-H1/RhoB pathway. Cancer Res.
76:1627–1640. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Keller M, Dubois F, Teulier S, Martin APJ,
Levallet J, Maille E, Brosseau S, Elie N, Hergovich A, Bergot E, et
al: NDR2 kinase contributes to cell invasion and cytokinesis
defects induced by the inactivation of RASSF1A tumor-suppressor
gene in lung cancer cells. J Exp Clin Cancer Res. 38:1582019.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hsu PC, Jablons DM, Yang CT and You L:
Epidermal growth factor receptor (EGFR) pathway, yes-associated
protein (YAP) and the regulation of programmed death-ligand 1
(PD-L1) in non-small cell lung cancer (NSCLC). Int J Mol Sci.
20:38212019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dubois F, Bergot E and Levallet G: Cancer
and RASSF1A/RASSF1C, the two faces of Janus. Trends Cancer.
5:662–665. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zeng Y, Liu Q, Wang Y, Tian C, Yang Q,
Zhao Y, Liu L, Wu G and Xu S: CDK5 activates hippo signaling to
confer resistance to radiation therapy via upregulating TAZ in lung
cancer. Int J Radiat Oncol Biol Phys. 108:758–769. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Levallet J, Biojout T, Bazille C, Douyère
M, Dubois F, Ferreira DL, Taylor J, Teulier S, Toutain J, Elie N,
et al: Hypoxia-induced activation of NDR2 underlies brain
metastases from non-small cell lung cancer. Cell Death Dis.
14:8232023. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
de Fraipont F, Levallet G, Creveuil C,
Bergot E, Beau-Faller M, Mounawar M, Richard N, Antoine M,
Rouquette I, Favrot MC, et al: An apoptosis methylation prognostic
signature for early lung cancer in the IFCT-0002 trial. Clin Cancer
Res. 18:2976–2986. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Minniti G, Goldsmith C and Brada M:
Chapter 16-radiotherapy. Handb Clin Neurol. 104:215–228. 2012.
View Article : Google Scholar
|
|
16
|
Hall EJ and Giaccia AJ: Radiobiology for
the radiologist. 8th. Philadelphia Baltimore New York London Buenos
Aires: LWW; pp. 6242018
|
|
17
|
Loh ZH, Doumy G, Arnold C, Kjellsson L,
Southworth SH, Al Haddad A, Kumagai Y, Tu MF, Ho PJ, March AM, et
al: Observation of the fastest chemical processes in the radiolysis
of water. Science. 367:179–182. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Huang T, Song X, Xu D, Tiek D, Goenka A,
Wu B, Sastry N, Hu B and Cheng SY: Stem cell programs in cancer
initiation, progression, and therapy resistance. Theranostics.
10:8721–8743. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen Z, Han F, Du Y, Shi H and Zhou W:
Hypoxic microenvironment in cancer: Molecular mechanisms and
therapeutic interventions. Signal Transduct Target Ther. 8:702023.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tang L, Wei F, Wu Y, He Y, Shi L, Xiong F,
Gong Z, Guo C, Li X, Deng H, et al: Role of metabolism in cancer
cell radioresistance and radiosensitization methods. J Exp Clin
Cancer Res. 37:872018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kelley K, Knisely J, Symons M and Ruggieri
R: Radioresistance of brain tumors. Cancers (Basel). 8:422016.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yang Y, Gao Y, Mutter-Rottmayer L,
Zlatanou A, Durando M, Ding W, Wyatt D, Ramsden D, Tanoue Y,
Tateishi S and Vaziri C: DNA repair factor RAD18 and DNA polymerase
Polκ confer tolerance of oncogenic DNA replication stress. J Cell
Biol. 216:3097–3115. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang M, Kern AM, Hülskötter M, Greninger
P, Singh A, Pan Y, Chowdhury D, Krause M, Baumann M, Benes CH, et
al: EGFR-mediated chromatin condensation protects KRAS-mutant
cancer cells against ionizing radiation. Cancer Res. 74:2825–2834.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Carlos-Reyes A, Muñiz-Lino MA,
Romero-Garcia S, López-Camarillo C and Hernández-de la Cruz ON:
Biological adaptations of tumor cells to radiation therapy. Front
Oncol. 11:7186362021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shi LZ and Bonner JA: Bridging
radiotherapy to immunotherapy: The IFN-JAK-STAT axis. Int J Mol
Sci. 22:122952021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Marampon F, Ciccarelli C and Zani BM:
Biological rationale for targeting MEK/ERK pathways in anti-cancer
therapy and to potentiate tumour responses to radiation. Int J Mol
Sci. 20:25302019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chang L, Graham PH, Hao J, Ni J, Bucci J,
Cozzi PJ, Kearsley JH and Li Y: PI3K/Akt/mTOR pathway inhibitors
enhance radiosensitivity in radioresistant prostate cancer cells
through inducing apoptosis, reducing autophagy, suppressing NHEJ
and HR repair pathways. Cell Death Dis. 5:e14372014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chen K, Shang Z, Dai AL and Dai PL: Novel
PI3K/Akt/mTOR pathway inhibitors plus radiotherapy: Strategy for
non-small cell lung cancer with mutant RAS gene. Life Sci.
255:1178162020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang Y, Zhou H, Zhang G and Xue X:
Targeting the canonical Wnt/β-catenin pathway in cancer
radioresistance: Updates on the molecular mechanisms. J Cancer Res
Ther. 15:272–277. 2019. View Article : Google Scholar
|
|
30
|
Xie SY, Li G, Han C, Yu YY and Li N: RKIP
reduction enhances radioresistance by activating the Shh signaling
pathway in non-small-cell lung cancer. OncoTargets Ther.
10:5605–5619. 2017. View Article : Google Scholar
|
|
31
|
Calses PC, Crawford JJ, Lill JR and Dey A:
Hippo pathway in cancer: Aberrant regulation and therapeutic
opportunities. Trends Cancer. 5:297–307. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Thompson BJ: YAP/TAZ: Drivers of tumor
growth, metastasis, and resistance to therapy. Bioessays.
42:e19001622020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Salem A, Asselin MC, Reymen B, Jackson A,
Lambin P, West CML, O'Connor JPB and Faivre-Finn C: Targeting
hypoxia to improve non-small cell lung cancer outcome. J Natl
Cancer Inst. 110:14–30. 2018. View Article : Google Scholar
|
|
34
|
Nguyen DX, Chiang AC, Zhang XHF, Kim JY,
Kris MG, Ladanyi M, Gerald WL and Massagué J: WNT/TCF signaling
through LEF1 and HOXB9 mediates lung adenocarcinoma metastasis.
Cell. 138:51–62. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hsu PC, You B, Yang YL, Zhang WQ, Wang YC,
Xu Z, Dai Y, Liu S, Yang CT, Li H, et al: YAP promotes erlotinib
resistance in human non-small cell lung cancer cells. Oncotarget.
7:51922–51933. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Miao J, Hsu PC, Yang YL, Xu Z, Dai Y, Wang
Y, Chan G, Huang Z, Hu B, Li H, et al: YAP regulates PD-L1
expression in human NSCLC cells. Oncotarget. 8:114576–114587. 2017.
View Article : Google Scholar
|
|
37
|
Xiao Y and Dong J: The Hippo signaling
pathway in cancer: A cell cycle perspective. Cancers (Basel).
13:62142021. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhou W, Zhang L, Chen P, Li S and Cheng Y:
Thymine DNA glycosylase-regulated TAZ promotes radioresistance by
targeting nonhomologous end joining and tumor progression in
esophageal cancer. Cancer Sci. 111:3613–3625. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Xin H, Liu Y, Chen P, Yin T, Wang M, Liu
T, Wen Z and Cheng Y: CD155 promotes radioresistance and malignancy
of esophageal cancer by regulating Hippo-YAP pathway. Discov Oncol.
13:532022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Moon JY, Ediriweera MK, Ryu JY, Kim HY and
Cho SK: Catechol enhances chemo- and radio-sensitivity by targeting
AMPK/Hippo signaling in pancreatic cancer cells. Oncol Rep.
45:1133–1141. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Andrade D, Mehta M, Griffith J,
Panneerselvam J, Srivastava A, Kim TD, Janknecht R, Herman T,
Ramesh R and Munshi A: YAP1 inhibition radiosensitizes triple
negative breast cancer cells by targeting the DNA damage response
and cell survival pathways. Oncotarget. 8:98495–98508. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Liang Y, Zhou X, Xie Q, Sun H, Huang K,
Chen H, Wang W, Zhou B, Wei X, Zeng D and Lin H: CD146 interaction
with integrin β1 activates LATS1-YAP signaling and induces
radiation-resistance in breast cancer cells. Cancer Lett.
546:2158562022. View Article : Google Scholar
|
|
43
|
Yang K, Zhao Y, Du Y and Tang R:
Evaluation of Hippo pathway and CD133 in radiation resistance in
small-cell lung cancer. J Oncol. 2021:88425542021. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Li J, Zhang X, Hou Z, Cai S, Guo Y, Sun L,
Li A, Li Q, Wang E and Miao Y: P130cas-FAK interaction is essential
for YAP-mediated radioresistance of non-small cell lung cancer.
Cell Death Dis. 13:7832022. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Bora-Singhal N, Nguyen J, Schaal C,
Perumal D, Singh S, Coppola D and Chellappan S: YAP1 regulates Oct4
activity and Sox2 expression to facilitate self-renewal and
vascular mimicry of stem-like cells. Stem Cells. 33:1705–1718.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wang L, Zhang Z, Yu X, Huang X, Liu Z,
Chai Y, Yang L, Wang Q, Li M, Zhao J, et al: Unbalanced YAP-SOX9
circuit drives stemness and malignant progression in esophageal
squamous cell carcinoma. Oncogene. 38:2042–2055. 2019. View Article : Google Scholar
|
|
47
|
Tang Y and Weiss SJ: Snail/Slug-YAP/TAZ
complexes cooperatively regulate mesenchymal stem cell function and
bone formation. Cell Cycle. 16:399–405. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Noce V, Battistelli C, Cozzolino AM,
Consalvi V, Cicchini C, Strippoli R, Tripodi M, Marchetti A and
Amicone L: YAP integrates the regulatory Snail/HNF4α circuitry
controlling epithelial/hepatocyte differentiation. Cell Death Dis.
10:7682019. View Article : Google Scholar
|
|
49
|
Pattschull G, Walz S, Gründl M, Schwab M,
Rühl E, Baluapuri A, Cindric-Vranesic A, Kneitz S, Wolf E, Ade CP,
et al: The Myb-MuvB complex is required for YAP-dependent
transcription of mitotic genes. Cell Rep. 27:3533–3546.e7. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Jang W, Kim T, Koo JS, Kim S and Lim D:
Mechanical cue-induced YAP instructs Skp2-dependent cell cycle exit
and oncogenic signaling. EMBO J. 36:2510–2528. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Kim W, Cho YS, Wang X, Park O, Ma X, Kim
H, Gan W, Jho EH, Cha B, Jeung YJ, et al: Hippo signaling is
intrinsically regulated during cell cycle progression by
APC/CCdh1. Proc Natl Acad Sci USA. 116:9423–9432. 2019.
View Article : Google Scholar
|
|
52
|
Oku Y, Nishiya N, Tazawa T, Kobayashi T,
Umezawa N, Sugawara Y and Uehara Y: Augmentation of the therapeutic
efficacy of WEE1 kinase inhibitor AZD1775 by inhibiting the
YAP-E2F1-DNA damage response pathway axis. FEBS Open Bio.
8:1001–1012. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Hergovich A: The roles of NDR protein
kinases in Hippo signalling. Genes (Basel). 7:212016. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Zhu H, Wang DD, Yuan T, Yan FJ, Zeng CM,
Dai XY, Chen ZB, Chen Y, Zhou T, Fan GH, et al: Multikinase
inhibitor CT-707 targets liver cancer by interrupting the
hypoxia-activated IGF-1R-YAP axis. Cancer Res. 78:3995–4006. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Cho Y, Park MJ, Kim K, Kim SW, Kim W, Oh S
and Lee JH: Reactive oxygen species-induced activation of
yes-associated protein-1 through the c-Myc pathway is a therapeutic
target in hepatocellular carcinoma. World J Gastroenterol.
26:6599–6613. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Shao D, Zhai P, Del Re DP, Sciarretta S,
Yabuta N, Nojima H, Lim DS, Pan D and Sadoshima J: A functional
interaction between Hippo-YAP signalling and FoxO1 mediates the
oxidative stress response. Nat Commun. 5:33152014. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Xiao W, Wang J, Ou C, Zhang Y, Ma L, Weng
W, Pan Q and Sun F: Mutual interaction between YAP and c-Myc is
critical for carcinogenesis in liver cancer. Biochem Biophys Res
Commun. 439:167–172. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Yu FX, Zhao B and Guan KL: Hippo pathway
in organ size control, tissue homeostasis, and cancer. Cell.
163:811–828. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Xiang L, Gilkes DM, Hu H, Takano N, Luo W,
Lu H, Bullen JW, Samanta D, Liang H and Semenza GL:
Hypoxia-inducible factor 1 mediates TAZ expression and nuclear
localization to induce the breast cancer stem cell phenotype.
Oncotarget. 5:12509–12527. 2014. View Article : Google Scholar
|
|
60
|
Azad T, Janse van Rensburg HJ, Lightbody
ED, Neveu B, Champagne A, Ghaffari A, Kay VR, Hao Y, Shen H, Yeung
B, et al: A LATS biosensor screen identifies VEGFR as a regulator
of the Hippo pathway in angiogenesis. Nat Commun. 9:10612018.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Lopez-Hernandez A, Sberna S and Campaner
S: Emerging principles in the transcriptional control by YAP and
TAZ. Cancers (Basel). 13:42422021. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Wang W, Xiao ZD, Li X, Aziz KE, Gan B,
Johnson RL and Chen J: AMPK modulates Hippo pathway activity to
regulate energy homeostasis. Nat Cell Biol. 17:490–499. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Basu-Roy U, Bayin NS, Rattanakorn K, Han
E, Placantonakis DG, Mansukhani A and Basilico C: Sox2 antagonizes
the Hippo pathway to maintain stemness in cancer cells. Nat Commun.
6:64112015. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Frum T, Watts JL and Ralston A: TEAD4,
YAP1 and WWTR1 prevent the premature onset of pluripotency prior to
the 16-cell stage. Development. 146:dev1798612019. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Zhang J, Ji JY, Yu M, Overholtzer M,
Smolen GA, Wang R, Brugge JS, Dyson NJ and Haber DA: YAP-dependent
induction of amphiregulin identifies a non-cell-autonomous
component of the Hippo pathway. Nat Cell Biol. 11:1444–1450. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Koo JH, Plouffe SW, Meng Z, Lee DH, Yang
D, Lim DS, Wang CY and Guan KL: Induction of AP-1 by YAP/TAZ
contributes to cell proliferation and organ growth. Genes Dev.
34:72–86. 2020. View Article : Google Scholar :
|
|
67
|
Li H, Li Q, Dang K, Ma S, Cotton JL, Yang
S, Zhu LJ, Deng AC, Ip YT, Johnson RL, et al: YAP/TAZ activation
drives uveal melanoma initiation and progression. Cell Rep.
29:3200–3211.e4. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Fang C, Li J, Qi S, Lei Y, Zeng Y, Yu P,
Hu Z, Zhou Y, Wang Y, Dai R, et al: An alternatively transcribed
TAZ variant negatively regulates JAK-STAT signaling. EMBO Rep.
20:e472272019. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Gruber R, Panayiotou R, Nye E,
Spencer-Dene B, Stamp G and Behrens A: YAP1 and TAZ control
pancreatic cancer initiation in mice by direct up-regulation of
JAK-STAT3 signaling. Gastroenterology. 151:526–539. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Prabhu KS, Bhat AA, Siveen KS,
Kuttikrishnan S, Raza SS, Raheed T, Jochebeth A, Khan AQ, Chawdhery
MZ, Haris M, et al: Sanguinarine mediated apoptosis in non-small
cell lung cancer via generation of reactive oxygen species and
suppression of JAK/STAT pathway. Biomed Pharmacother.
144:1123582021. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Meng J, Li Y, Wan C, Sun Y, Dai X, Huang
J, Hu Y, Gao Y, Wu B, Zhang Z, et al: Targeting senescence-like
fibroblasts radiosensitizes non-small cell lung cancer and reduces
radiation-induced pulmonary fibrosis. JCI Insight. 6:e1463342021.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Li L, Wang J, Zhang Y, Zhang Y, Ma L, Weng
W, Qiao Y, Xiao W, Wang H, Yu W, et al: MEK1 promotes YAP and their
interaction is critical for tumorigenesis in liver cancer. FEBS
Lett. 587:3921–3927. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Santoro R, Zanotto M, Carbone C, Piro G,
Tortora G and Melisi D: MEKK3 sustains EMT and stemness in
pancreatic cancer by regulating YAP and TAZ transcriptional
activity. Anticancer Res. 38:1937–1946. 2018.PubMed/NCBI
|
|
74
|
You B, Yang YL, Xu Z, Dai Y, Liu S, Mao
JH, Tetsu O, Li H, Jablons DM and You L: Inhibition of ERK1/2
down-regulates the Hippo/YAP signaling pathway in human NSCLC
cells. Oncotarget. 6:4357–4368. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Kim NG and Gumbiner BM: Adhesion to
fibronectin regulates Hippo signaling via the FAK-Src-PI3K pathway.
J Cell Biol. 210:503–515. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Zhao Y, Montminy T, Azad T, Lightbody E,
Hao Y, SenGupta S, Asselin E, Nicol C and Yang X: PI3K positively
regulates YAP and TAZ in mammary tumorigenesis through multiple
signaling pathways. Mol Cancer Res. 16:1046–1058. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Gokey JJ, Sridharan A, Xu Y, Green J,
Carraro G, Stripp BR, Perl AT and Whitsett JA: Active epithelial
Hippo signaling in idiopathic pulmonary fibrosis. JCI Insight.
3:e987382018. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Fernandez-L A, Squatrito M, Northcott P,
Awan A, Holland EC, Taylor MD, Nahlé Z and Kenney AM: Oncogenic YAP
promotes radioresistance and genomic instability in medulloblastoma
through IGF2-mediated Akt activation. Oncogene. 31:1923–1937. 2012.
View Article : Google Scholar
|
|
79
|
Artinian N, Cloninger C, Holmes B,
Benavides-Serrato A, Bashir T and Gera J: Phosphorylation of the
Hippo pathway component AMOTL2 by the mTORC2 kinase promotes YAP
signaling, resulting in enhanced glioblastoma growth and
invasiveness. J Biol Chem. 290:19387–19401. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Takeda T, Yamamoto Y, Tsubaki M, Matsuda
T, Kimura A, Shimo N and Nishida S: PI3K/Akt/YAP signaling promotes
migration and invasion of DLD-1 colorectal cancer cells. Oncol
Lett. 23:1062022. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Park HW, Kim YC, Yu B, Moroishi T, Mo JS,
Plouffe SW, Meng Z, Lin KC, Yu FX, Alexander CM, et al: Alternative
Wnt signaling activates YAP/TAZ. Cell. 162:780–794. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Wang J, Park JS, Wei Y, Rajurkar M, Cotton
JL, Fan Q, Lewis BC, Ji H and Mao J: TRIB2 acts downstream of
Wnt/TCF in liver cancer cells to regulate YAP and C/EBPα function.
Mol Cell. 51:211–225. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Simula L, Alifano M and Icard P: How
phosphofructokinase-1 promotes PI3K and YAP/TAZ in cancer:
Therapeutic perspectives. Cancers (Basel). 14:24782022. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Konsavage WM Jr, Kyler SL, Rennoll SA, Jin
G and Yochum GS: Wnt/β-catenin signaling regulates Yes-associated
protein (YAP) gene expression in colorectal carcinoma cells. J Biol
Chem. 287:11730–11739. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Azzolin L, Zanconato F, Bresolin S,
Forcato M, Basso G, Bicciato S, Cordenonsi M and Piccolo S: Role of
TAZ as Mediator of Wnt signaling. Cell. 151:1443–1456. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Deng F, Peng L, Li Z, Tan G, Liang E, Chen
S, Zhao X and Zhi F: YAP triggers the Wnt/β-catenin signalling
pathway and promotes enterocyte self-renewal, regeneration and
tumorigenesis after DSS-induced injury. Cell Death Dis. 9:1532018.
View Article : Google Scholar
|
|
87
|
Jiang L, Li J, Zhang C, Shang Y and Lin J:
YAP-mediated crosstalk between the Wnt and Hippo signaling pathways
(review). Mol Med Rep. 22:4101–4106. 2020.PubMed/NCBI
|
|
88
|
Chen Y, Jin Y, Ying H, Zhang P, Chen M and
Hu X: Synergistic effect of PAF inhibition and X-ray irradiation in
non-small cell lung cancer cells. Strahlenther Onkol. 197:343–352.
2021. View Article : Google Scholar
|
|
89
|
Cotton JL, Li Q, Ma L, Park JS, Wang J, Ou
J, Zhu LJ, Ip YT, Johnson RL and Mao J: YAP/TAZ and hedgehog
coordinate growth and patterning in gastrointestinal mesenchyme.
Dev Cell. 43:35–47.e4. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Isago H, Mitani A, Mikami Y, Horie M,
Urushiyama H, Hamamoto R, Terasaki Y and Nagase T: Epithelial
expression of YAP and TAZ is sequentially required in lung
development. Am J Respir Cell Mol Biol. 62:256–266. 2020.
View Article : Google Scholar
|
|
91
|
Fernandez-L A, Northcott PA, Dalton J,
Fraga C, Ellison D, Angers S, Taylor MD and Kenney AM: YAP1 is
amplified and up-regulated in hedgehog-associated medulloblastomas
and mediates Sonic hedgehog-driven neural precursor proliferation.
Genes Dev. 23:2729–2741. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Tang C, Wang J, Yao M, Ji X, Shi W, Xu C,
Zeng LH and Wu X: Hippo signaling activates hedgehog signaling by
Taz-driven Gli3 processing. Cell Regen. 12:32023. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Tariki M, Dhanyamraju PK, Fendrich V,
Borggrefe T, Feldmann G and Lauth M: The yes-associated protein
controls the cell density regulation of Hedgehog signaling.
Oncogenesis. 3:e1122014. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Lin YT, Ding JY, Li MY, Yeh TS, Wang TW
and Yu JY: YAP regulates neuronal differentiation through Sonic
hedgehog signaling pathway. Exp Cell Res. 318:1877–1888. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Swiderska-Syn M, Xie G, Michelotti GA,
Jewell ML, Premont RT, Syn WK and Diehl AM: Hedgehog regulates
yes-associated protein 1 in regenerating mouse liver. Hepatology.
64:232–244. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Kim Y and Jho EH: Regulation of the Hippo
signaling pathway by ubiquitin modification. BMB Rep. 51:143–150.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Meng Z, Moroishi T and Guan KL: Mechanisms
of Hippo pathway regulation. Genes Dev. 30:1–17. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Tang Y, Geng Y, Luo J, Shen W, Zhu W, Meng
C, Li M, Zhou X, Zhang S and Cao J: Downregulation of ubiquitin
inhibits the proliferation and radioresistance of non-small cell
lung cancer cells in vitro and in vivo. Sci Rep. 5:94762015.
View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Deng L, Meng T, Chen L, Wei W and Wang P:
The role of ubiquitination in tumorigenesis and targeted drug
discovery. Signal Transduct Target Ther. 5:112020. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Hintelmann K, Kriegs M, Rothkamm K and
Rieckmann T: Improving the efficacy of tumor radiosensitization
through combined molecular targeting. Front Oncol. 10:12602020.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Zhao Y, Wang L, Huang Q, Jiang Y, Wang J,
Zhang L, Tian Y and Yang H: Radiosensitization of non-small cell
lung cancer cells by inhibition of TGF-β1 signaling with SB431542
is dependent on p53 status. Oncol Res. 24:1–7. 2016. View Article : Google Scholar
|
|
102
|
Van den Bossche J, Domen A, Peeters M,
Deben C, De Pauw I, Jacobs J, De Bruycker S, Specenier P, Pauwels
P, Vermorken JB, et al: Radiosensitization of non-small cell lung
cancer cells by the Plk1 inhibitor volasertib is dependent on the
p53 status. Cancers (Basel). 11:18932019. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Gill SJ, Wijnhoven PWG, Fok JHL, Lloyd RL,
Cairns J, Armenia J, Nikkilä J, Lau A, Bakkenist CJ, Galbraith SM,
et al: Radiopotentiation profiling of multiple inhibitors of the
DNA damage response for early clinical development. Mol Cancer
Ther. 20:1614–1626. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Dukaew N, Konishi T, Chairatvit K,
Autsavapromporn N, Soonthornchareonnon N and Wongnoppavich A:
Enhancement of radiosensitivity by eurycomalactone in human NSCLC
cells through G2/M Cell cycle arrest and delayed DNA
double-strand break repair. Oncol Res. 28:161–175. 2020. View Article : Google Scholar
|
|
105
|
Ryu H, Kim HJ, Song JY, Hwang SG, Kim JS,
Kim J, Bui THN, Choi HK and Ahn J: A small compound KJ-28d enhances
the sensitivity of non-small cell lung cancer to radio- and
chemotherapy. Int J Mol Sci. 20:60262019. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Majd NK, Yap TA, Koul D, Balasubramaniyan
V, Li X, Khan S, Gandy KS, Yung WKA and de Groot JF: The promise of
DNA damage response inhibitors for the treatment of glioblastoma.
Neurooncol Adv. 3:vdab0152021.PubMed/NCBI
|
|
107
|
La Verde G, Artiola V, Pugliese M, La
Commara M, Arrichiello C, Muto P, Netti PA, Fusco S and Panzetta V:
Radiation therapy affects YAP expression and intracellular
localization by modulating lamin A/C levels in breast cancer. Front
Bioeng Biotechnol. 10:9690042022. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Zhang Y, Wang Y, Zhou D, Wang K, Wang X,
Wang X, Jiang Y, Zhao M, Yu R and Zhou X: Radiation-induced YAP
activation confers glioma radioresistance via promoting FGF2
transcription and DNA damage repair. Oncogene. 40:4580–4591. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Barrette AM, Ronk H, Joshi T, Mussa Z,
Mehrotra M, Bouras A, Nudelman G, Jesu Raj JG, Bozec D, Lam W, et
al: Anti-invasive efficacy and survival benefit of the YAP-TEAD
inhibitor verteporfin in preclinical glioblastoma models. Neuro
Oncol. 24:694–707. 2022. View Article : Google Scholar :
|
|
110
|
Amidon BS, Sanchez-Martin M, Bartolini W,
Syed S, McGovern K, Xu L, Ecsedy J, Zhang XM, Constan A and Castro
AC: Abstract 2156: IK-930 is a novel TEAD inhibitor for the
treatment of cancers harboring mutations in the Hippo signal
transduction pathway. Cancer Res. 82(12 Suppl): S21562022.
View Article : Google Scholar
|
|
111
|
Tang TT, Konradi AW, Feng Y, Peng X, Ma M,
Li J, Yu FX, Guan KL and Post L: Small molecule inhibitors of TEAD
auto-palmitoylation selectively inhibit proliferation and tumor
growth of NF2-deficient mesothelioma. Mol Cancer Ther. 20:986–998.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Sun Y, Hu L, Tao Z, Jarugumilli GK, Erb H,
Singh A, Li Q, Cotton JL, Greninger P, Egan RK, et al:
Pharmacological blockade of TEAD-YAP reveals its therapeutic
limitation in cancer cells. Nat Commun. 13:67442022. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2020. CA Cancer J Clin. 70:7–30. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Levallet G, Creveuil C, Bekaert L, Péres
E, Planchard G, Lecot-Cotigny S, Guillamo JS, Emery E, Zalcman G
and Lechapt-Zalcman E: Promoter hypermethylation of genes encoding
for RASSF/Hippo pathway members reveals specific alteration pattern
in diffuse gliomas. J Mol Diagn. 21:695–704. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Wei C, Wang Y and Li X: The role of Hippo
signal pathway in breast cancer metastasis. Onco Targets Ther.
11:2185–2193. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Maille E, Brosseau S, Hanoux V, Creveuil
C, Danel C, Bergot E, Scherpereel A, Mazières J, Margery J,
Greillier L, et al: MST1/Hippo promoter gene methylation predicts
poor survival in patients with malignant pleural mesothelioma in
the IFCT-GFPC-0701 MAPS phase 3 trial. Br J Cancer. 120:387–397.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Spugnardi M, Tommasi S, Dammann R, Pfeifer
GP and Hoon DSB: Epigenetic inactivation of RAS association domain
family protein 1 (RASSF1A) in malignant cutaneous melanoma. Cancer
Res. 63:1639–1643. 2003.PubMed/NCBI
|
|
118
|
Riffet M, Eid Y, Faisant M, Fohlen A,
Menahem B, Alves A, Dubois F, Levallet G and Bazille C: Deciphering
promoter hypermethylation of genes encoding for RASSF/Hippo pathway
reveals the poor prognostic factor of RASSF2 gene silencing in
colon cancers. Cancers (Basel). 13:59572021. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Thurneysen C, Opitz I, Kurtz S, Weder W,
Stahel RA and Felley-Bosco E: Functional inactivation of NF2/merlin
in human mesothelioma. Lung Cancer. 64:140–147. 2009. View Article : Google Scholar
|
|
120
|
Noorbakhsh N, Hayatmoghadam B, Jamali M,
Golmohammadi M and Kavianpour M: The Hippo signaling pathway in
leukemia: function, interaction, and carcinogenesis. Cancer cell
international. 21:7052021. View Article : Google Scholar : PubMed/NCBI
|