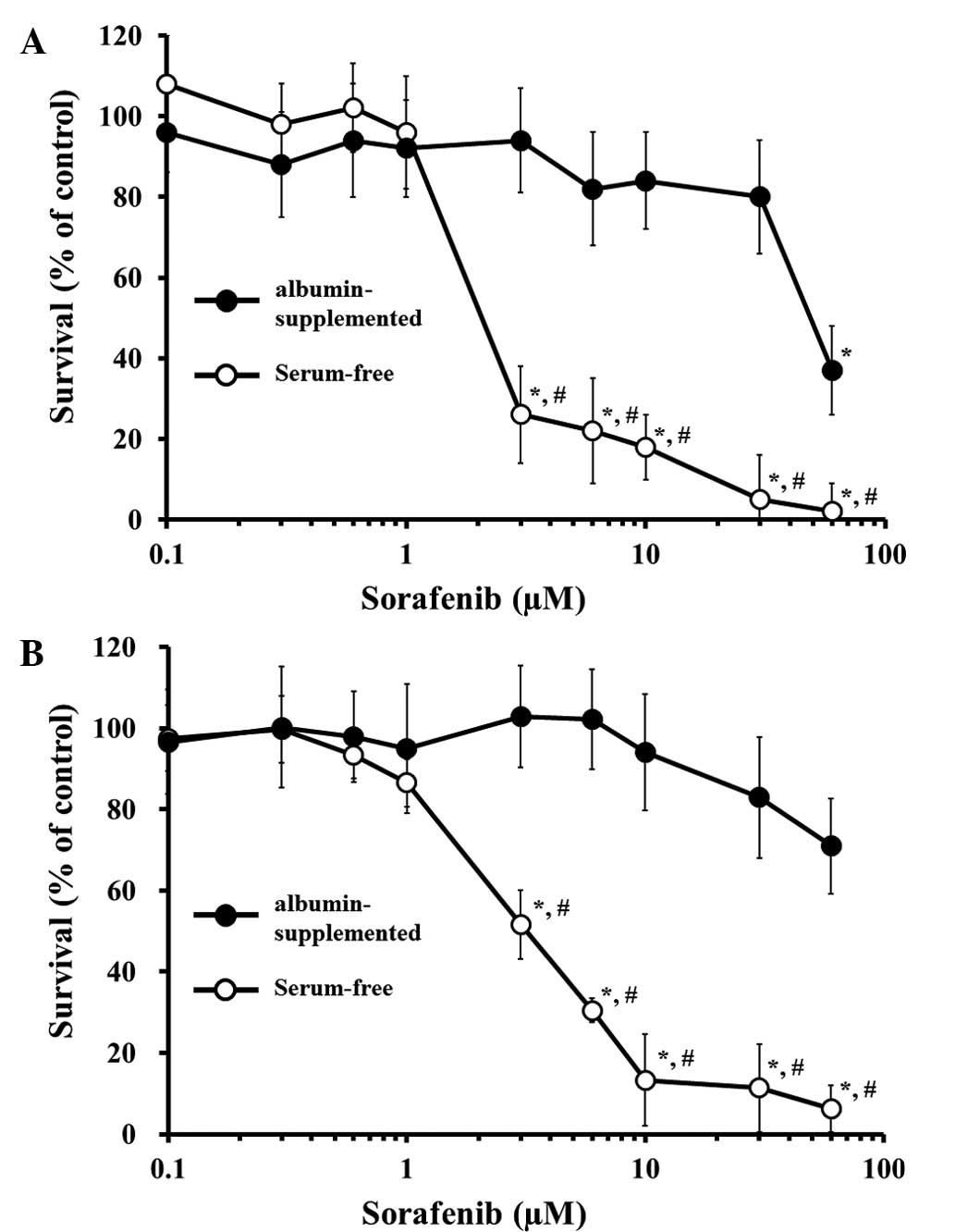
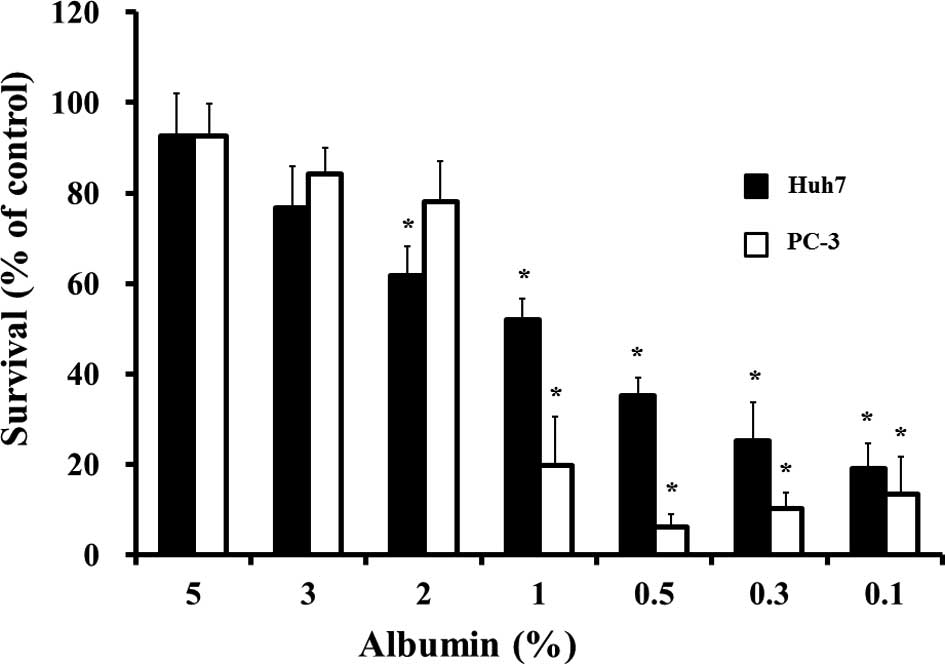
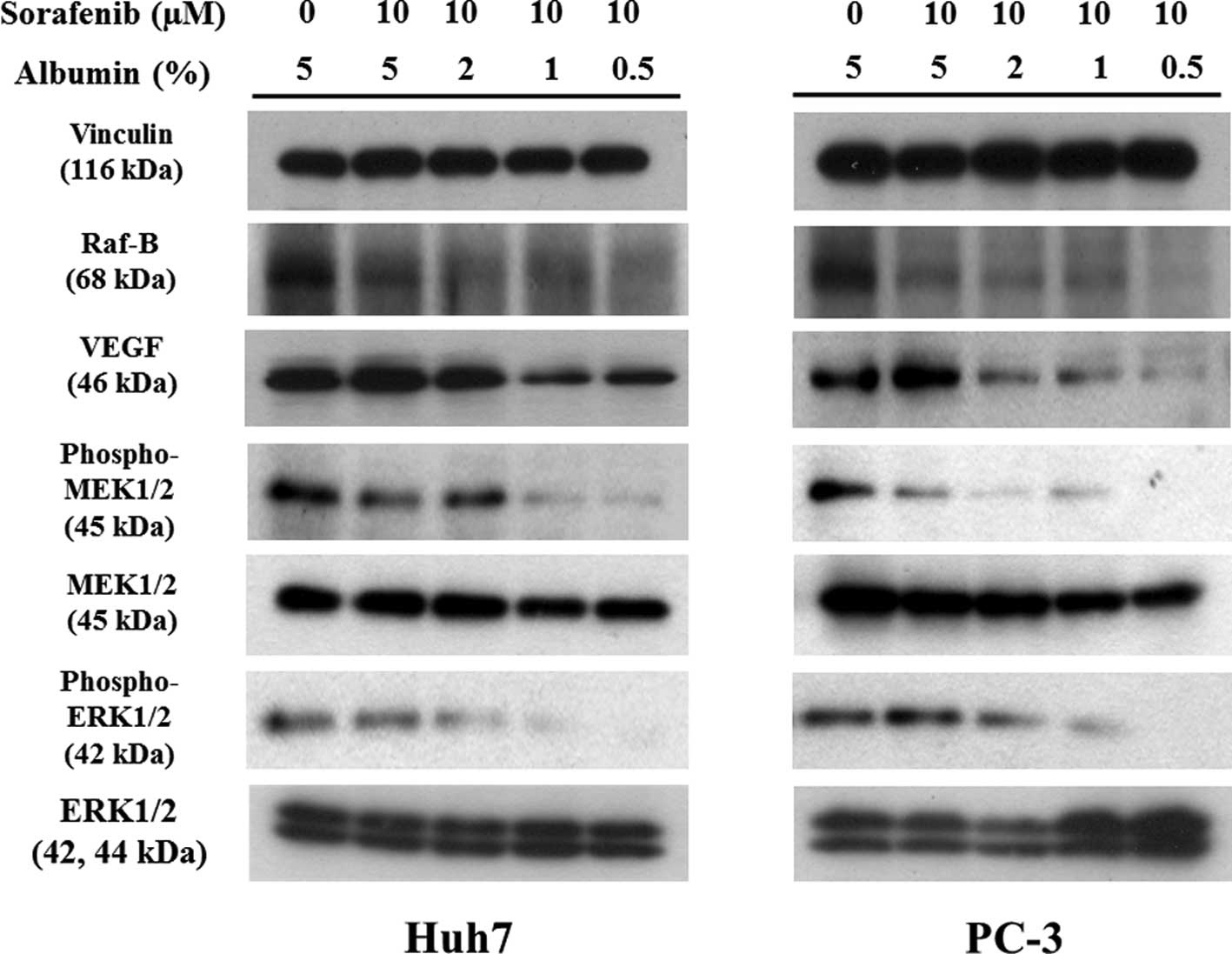
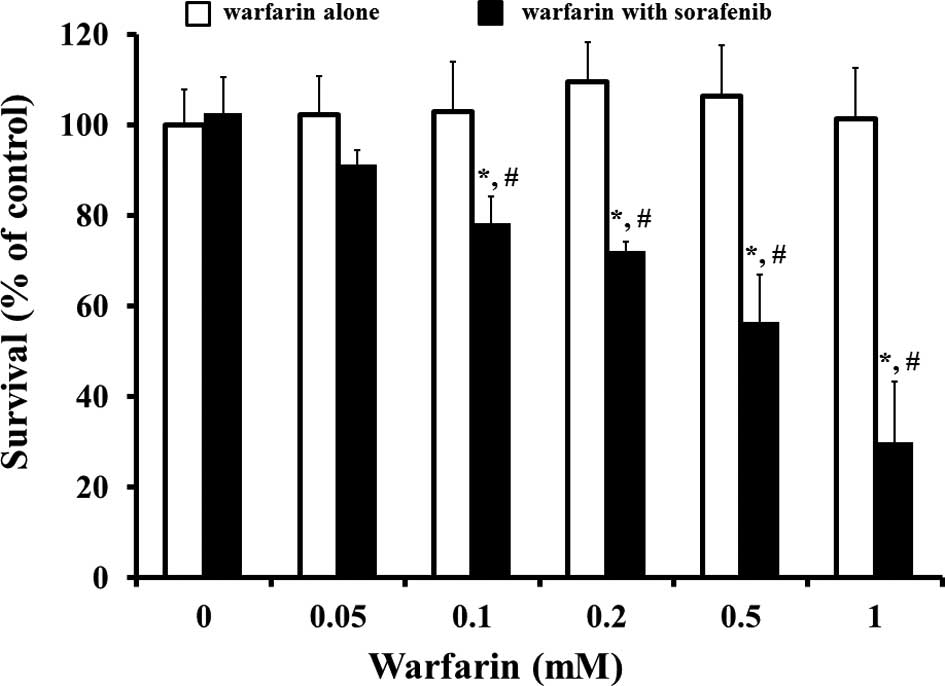
|
1.
|
Peters T: All About Albumin: Biochemistry,
Genetics, and Medical Applications. Academic Press; San Diego:
1996
|
|
2.
|
Ballmer PE, McNurlan MA, Milne E, Heys SD,
Buchan V, Calder AG and Garlick PJ: Measurement of albumin
synthesis in humans: a new approach employing stable isotopes. Am J
Physiol. 259:E797–E803. 1990.
|
|
3.
|
Waldmann TA: Protein-losing enteropathy
and kinetic studies of plasma protein metabolism. Semin Nucl Med.
2:251–263. 1972.
|
|
4.
|
Ballmer PE, Walshe D, McNurlan MA, Watson
H, Brunt PW and Garlick PJ: Albumin synthesis rates in cirrhosis:
correlation with Child-Turcotte classification. Hepatology.
18:292–297. 1993.
|
|
5.
|
Pasanisi F, Orban A, Scalfi L, Alfonsi L,
Santarpia L, Zurlo E, Celona A, Potenza A and Contaldo F:
Predictors of survival in terminal-cancer patients with
irreversible bowel obstruction receiving home parenteral nutrition.
Nutrition. 17:581–584. 2001.
|
|
6.
|
Ruot B, Breuillé D, Rambourdin F, Bayle G,
Capitan P and Obled C: Synthesis rate of plasma albumin is a good
indicator of liver albumin synthesis in sepsis. Am J Physiol
Endocrinol Metab. 279:E244–E251. 2000.
|
|
7.
|
Ballmer PE: Causes and mechanisms of
hypoalbuminaemia. Clin Nutr. 20:271–273. 2001.
|
|
8.
|
Wilhelm S, Carter C, Lynch M, Lowinger T,
Dumas J, Smith RA, Schwartz B, Simantov R and Kelley S: Discovery
and development of sorafenib: a multikinase inhibitor for treating
cancer. Nat Rev Drug Discov. 5:835–844. 2006.
|
|
9.
|
Wilhelm SM, Carter C, Tang L, et al: BAY
43-9006 exhibits broad spectrum oral antitumor activity and targets
the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in
tumor progression and angiogenesis. Cancer Res. 64:7099–7109.
2004.
|
|
10.
|
Liu L, Cao Y, Chen C, Zhang X, McNabola A,
Wilkie D, Wilhelm S, Lynch M and Carter C: Sorafenib blocks the
RAF/MEK/ERK pathway, inhibits tumor angiogenesis, and induces tumor
cell apoptosis in hepatocellular carcinoma model PLC/PRF/5. Cancer
Res. 66:11851–11858. 2006.
|
|
11.
|
Escudier B, Eisen T, Stadler WM, et al:
Sorafenib for treatment of renal cell carcinoma: final efficacy and
safety results of the phase III treatment approaches in renal
cancer global evaluation trial. J Clin Oncol. 27:3312–3318.
2009.
|
|
12.
|
Llovet JM, Ricci S, Mazzaferro V, et al:
Sorafenib in advanced hepatocellular carcinoma. N Engl J Med.
359:378–390. 2008.
|
|
13.
|
Wilhelm SM, Adnane L, Newell P, Villanueva
A, Llovet JM and Lynch M: Preclinical overview of sorafenib, a
multikinase inhibitor that targets both Raf and VEGF and PDGF
receptor tyrosine kinase signaling. Mol Cancer Ther. 7:3129–3140.
2008.
|
|
14.
|
Kloos RT, Ringel MD, Knopp MV, et al:
Phase II trial of sorafenib in metastatic thyroid cancer. J Clin
Oncol. 27:1675–1684. 2009.
|
|
15.
|
Dahut WL, Scripture C, Posadas E, et al: A
phase II clinical trial of sorafenib in androgen-independent
prostate cancer. Clin Cancer Res. 14:209–214. 2008.
|
|
16.
|
Jain L, Woo S, Gardner ER, Dahut WL, Kohn
EC, Kummar S, Mould DR, Giaccone G, Yarchoan R, Venitz J and Figg
WD: Population pharmacokinetic analysis of sorafenib in patients
with solid tumours. Br J Clin Pharmacol. 72:294–305. 2011.
|
|
17.
|
Tod M, Mir O, Bancelin N, et al:
Functional and clinical evidence of the influence of sorafenib
binding to albumin on sorafenib disposition in adult cancer
patients. Pharm Res. 28:3199–3207. 2011.
|
|
18.
|
Villarroel MC, Pratz KW, Xu L, Wright JJ,
Smith BD and Rudek MA: Plasma protein binding of sorafenib, a multi
kinase inhibitor: in vitro and in cancer patients. Invest New
Drugs. Nov 17–2011, (E-pub ahead of print).
|
|
19.
|
Kanno S, Kurauchi K, Tomizawa A, Yomogida
S and Ishikawa M: Albumin modulates docosahexaenoic acid-induced
cytotoxicity in human hepatocellular carcinoma cell lines. Toxicol
Lett. 200:154–161. 2011.
|
|
20.
|
Kanno S, Higurashi A, Watanabe Y, Shouji
A, Asou K and Ishikawa M: Susceptibility to cytosine arabinoside
(Ara-C)-induced cytotoxicity in human leukemia cell lines. Toxicol
Lett. 152:149–158. 2004.
|
|
21.
|
Kanno SI, Maeda N, Tomizawa A, Yomogida S,
Katoh T and Ishikawa M: Involvement of p21waf1/cip1
expression in the cytotoxicity of the potent histone deacetylase
inhibitor spiruchostatin B towards susceptible NALM-6 human B cell
leukemia cells. Int J Oncol. 40:1391–1396. 2012.
|
|
22.
|
Tesseromatis C and Alevizou A: The role of
the protein-binding on the mode of drug action as well the
interactions with other drugs. Eur J Drug Metab Pharmacokinet.
33:225–230. 2008.
|
|
23.
|
Mather JP: Making informed choices:
medium, serum, and serum-free medium. How to choose the appropriate
medium and culture system for the model you wish to create. Methods
Cell Biol. 57:19–30. 1998.
|
|
24.
|
Fanali G, di Masi A, Trezza V, Marino M,
Fasano M and Ascenzi P: Human serum albumin: from bench to bedside.
Mol Aspects Med. 33:209–290. 2012.
|
|
25.
|
Blanchet B, Billemont B, Barete S,
Garrigue H, Cabanes L, Coriat R, Francès C, Knebelmann B and
Goldwasser F: Toxicity of sorafenib: clinical and molecular
aspects. Expert Opin Drug Saf. 9:275–287. 2010.
|
|
26.
|
Ullén A, Farnebo M, Thyrell L, Mahmoudi S,
Kharaziha P, Lennartsson L, Grandér D, Panaretakis T and Nilsson S:
Sorafenib induces apoptosis and autophagy in prostate cancer cells
in vitro. Int J Oncol. 37:15–20. 2010.
|
|
27.
|
Sun S, Wang M, Su L, Li J, Li H and Gu D:
Study on warfarin plasma concentration and its correlation with
international normalized ratio. J Pharm Biomed Anal. 42:218–222.
2006.
|