Introduction
Hybrid endoscopic full-thickness resection (EFTR)
for the treatment of gastric gastrointestinal stromal tumors
(GISTs) has become an endoscopic and laparoscopic surgery
cross-technique. An increasing number of studies have focused on
hybrid EFTR, which is performed in combination with endoscopy and
laparoscopy (1–3). The indications for hybrid EFTR
include GISTs that do not require lymphadenectomy and can be cured
by radical tumor enucleation. Although a number of animal
experiments using hybrid natural orifice translumenal endoscopic
surgery (NOTES) have been reported, studies conducted on humans are
required (4–6).
Patients and methods
Patients and identification of
GISTs
The present study included 16 patients who underwent
hybrid EFTR between September 2009 and February 2013 for the
treatment of gastric GIST. The study was approved by the Ethics
Committee of Kagawa University (Kagawa, Japan), and enrollment was
conducted using the University Hospital Medical Information Network
(no. 000004722 and 000008691). Written informed consent was
obtained from each patient.
The GIST risk factors were evaluated using the
Fletcher classification (?) as follows: Tumor size; histological
findings, including mitosis; and immunohistochemical staining. The
tumor size was measured with an endoscopic ultrasound (EUS) (20-MHz
radial scan). Patients with a tumor size <40 mm were selected
for the study as GISTs that are >40 mm cannot be removed through
the esophagus. However, in the present study, four patients were
included with tumors >40 mm (42, 54, 46 and 51 mm). An
EUS-fine-needle aspiration (FNA) was performed, as well as an en
bloc biopsy using endoscopic submucosal dissection (ESD) for
histopathology, as described previously (7). For small GISTs, ~10 mm, an en bloc
biopsy was performed using ESD techniques as EUS-FNA is difficult
to perform with small targets. Patients were selected using the
following inclusion criteria for histological findings: Mitotic
counts <5/high-power field (HPF) and immunohistochemical stains
positive for KIT or cluster of differentiation 34 (CD34). The
patients with tumors >50 mm, mitotic counts >10/50 HPF or
immunohistochemical stains negative for KIT or CD34 were excluded
from the study due to the high-associated risk for GISTs. The study
examined the EFTR success rate, surgical time (duration of the
surgical procedure), presence or absence of complications, and
length of hospital stay.
Statistical analysis
Data are presented as the mean ± standard deviation
values. Patient baseline statistics were analyzed using the Kruskal
Wallis H-test, unpaired t-test and Mann-Whitney U test. The mitosis
count and positive number of KIT/CD34 were analyzed using the
unpaired t-test. The histological risk was analyzed using the
Kruskal Wallis H-test and Mann-Whitney U test. P<0.05 was
considered to indicate a statistically significant difference. The
statistical analysis was performed using Graph Pad Prism version 5
for Windows (GraphPad Software, San Diego, CA, USA).
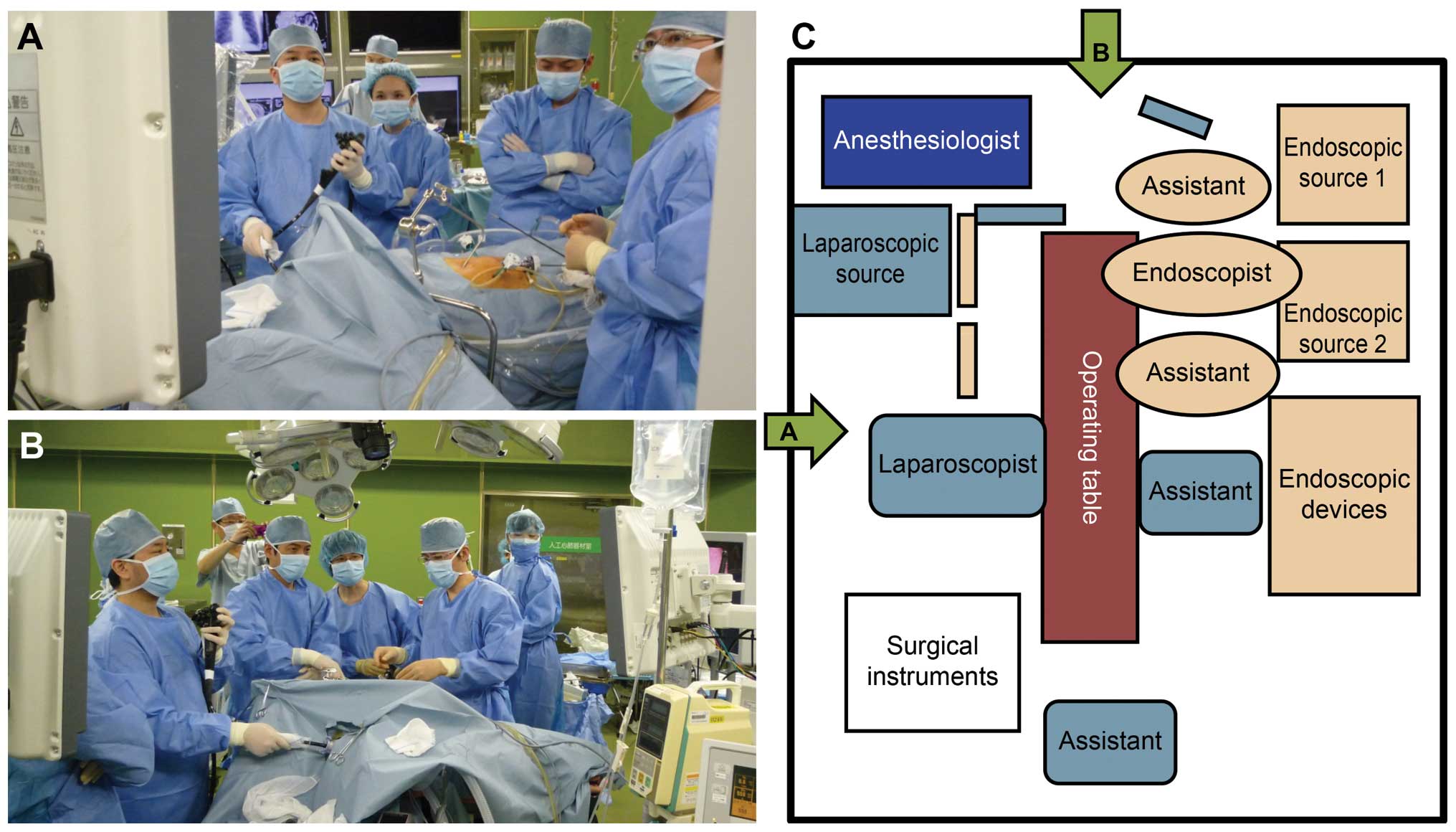
Arrangement of the operating room
Two sources of light for the flexible endoscope and
a stand for the endoscopic device were placed on the left side of
the patient. The laparoscope operator was positioned in a manner
similar to that of the conventional arrangement in laparoscopic
surgery (Fig. 1A and B).
Operative devices
The following operative devices were used: i)
Endoscopes: GIF type Q260J, GIF type H260Z and GIF type XP260NS
(sterilized with EtO gas; Olympus Co., Tokyo, Japan); ii)
incisional knives: Dual knife (KD-650L; Olympus Co.) and
insulation-tipped (IT)-knife-2 (KD-611L; Olympus Co.); iii)
hemostatic forceps: Coagrasper (FD-410LR; Olympus Co.); iv) tip
attachment: Elastic Touch Attachment (TOP Co., Tokyo, Japan); v)
incisional device: ERBE VIO300D (ERBE Elektromedizin GmbH,
Tübingen, Germany); and vi) CO2 insufflation device: UCR
(Olympus Co.); vii) Endo Catch (Covidien Japan, Tokyo, Japan).
Surgical procedure
The surgery was performed under general anesthesia
with the supervision of an anesthesiologist. A longitudinal 20-mm
incision was made in the umbilical region, and the following
laparoscopic ports were created: A 12-mm camera port, two 12-mm
ports at the upper right and left umbilical regions and a 5-mm port
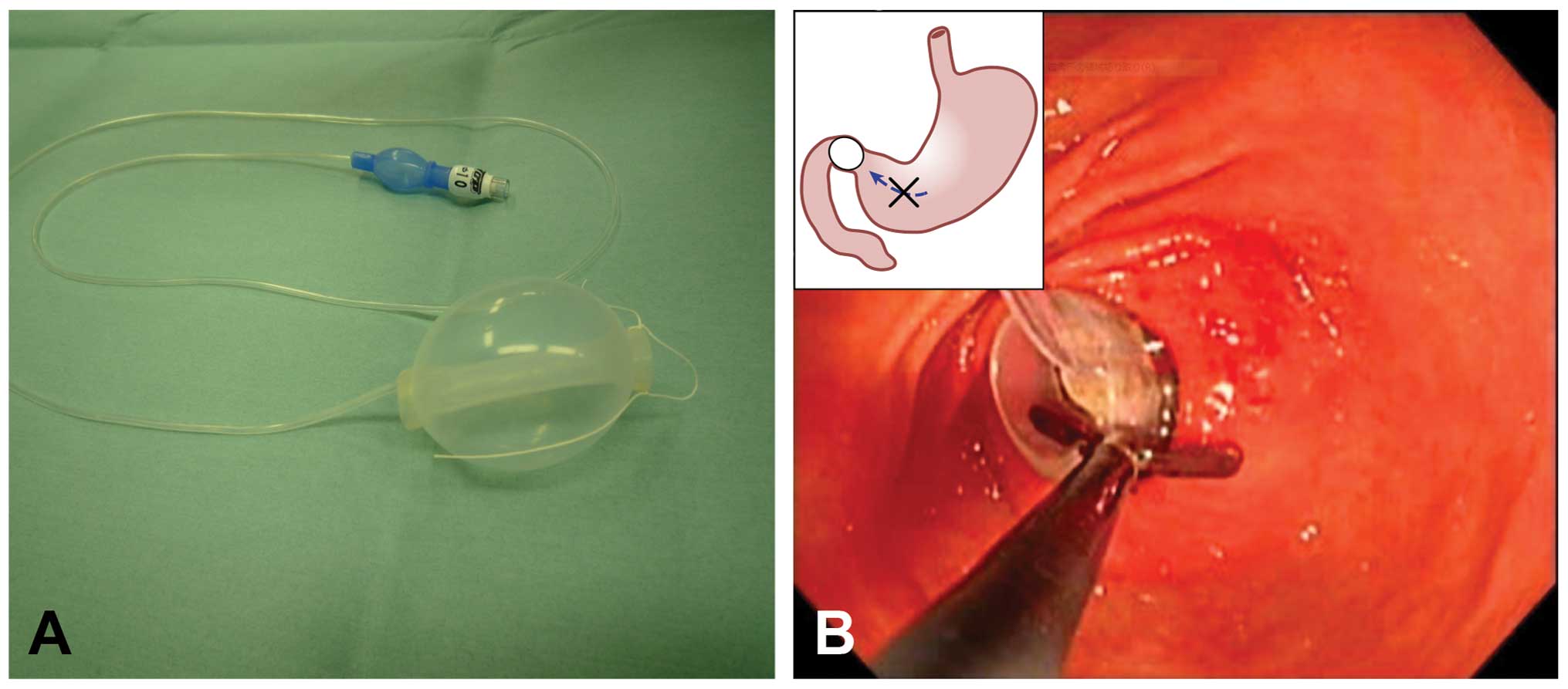
in the left upper quadrant of the abdomen. Using a nylon thread, a
loop was created at the tip of a balloon designed for use in
esophageal variceal sclerotherapy (length, 52 mm; inner diameter,
10 mm) (TOP Co.) (Fig. 2A). The
balloon was inserted into the duodenal bulb and inflated with 60–70
ml of air (using the duodenal balloon occlusion method) (Fig. 2B) (8). The esophagus and stomach were
carefully lavaged with 2,000 ml of normal saline to disinfect the
area in preparation for the full-thickness resection of the gastric
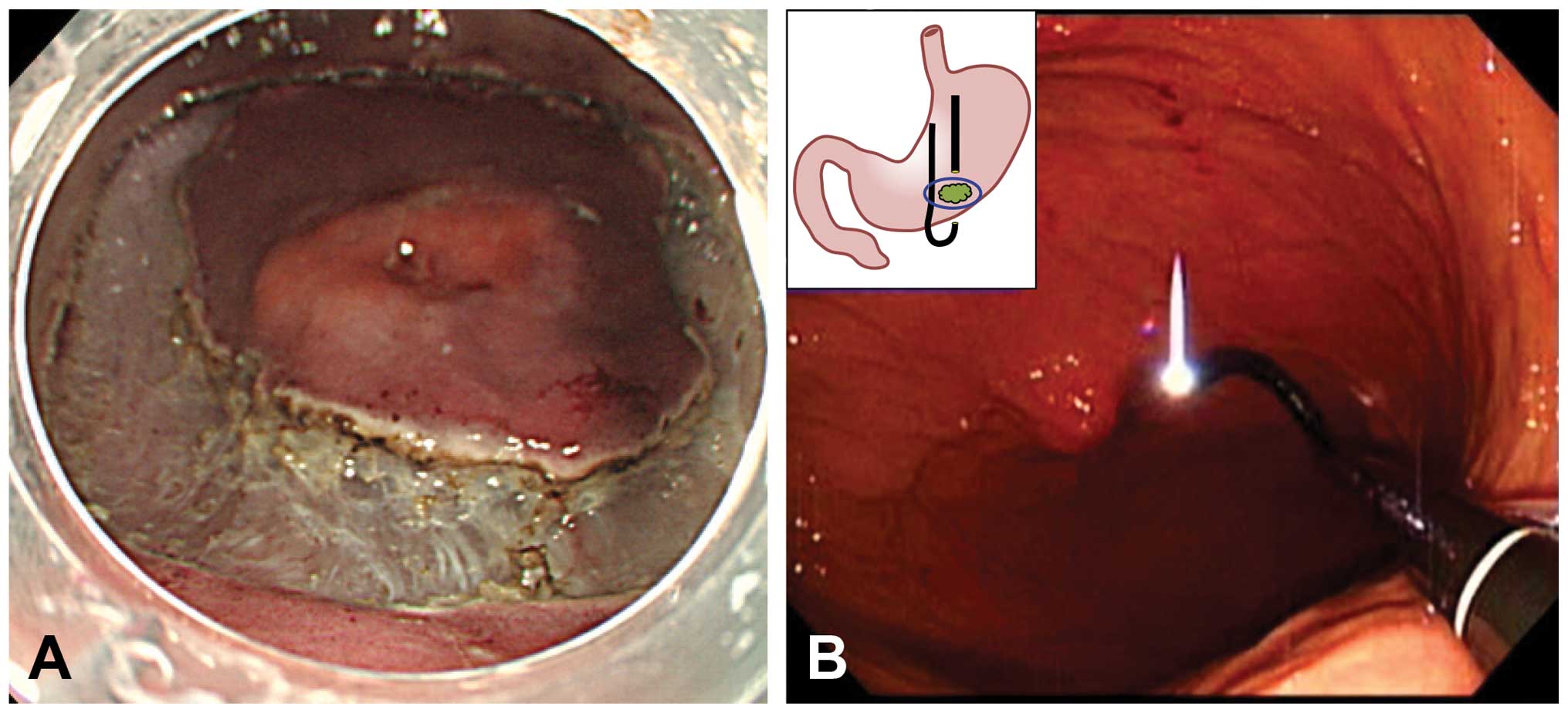
wall (9). With an 8-mm safety
margin around the tumor, preparations were made for a full incision
into the submucosal layer and for an incision into the muscle layer
(the incisions were made in accordance with the ESD method)
(Fig. 3A) (4). The incision was widened to
accommodate the 5-mm diameter transnasal endoscope. The transnasal
endoscope (GIF N260) was inserted into the abdominal cavity, and
the full-thickness resection was performed with the transoral
endoscope (GIF Q260J), while the procedure was observed through the
transnasal endoscope (the double-scope technique) (Fig. 3B) (4). The ERBE VIO300D was used as the
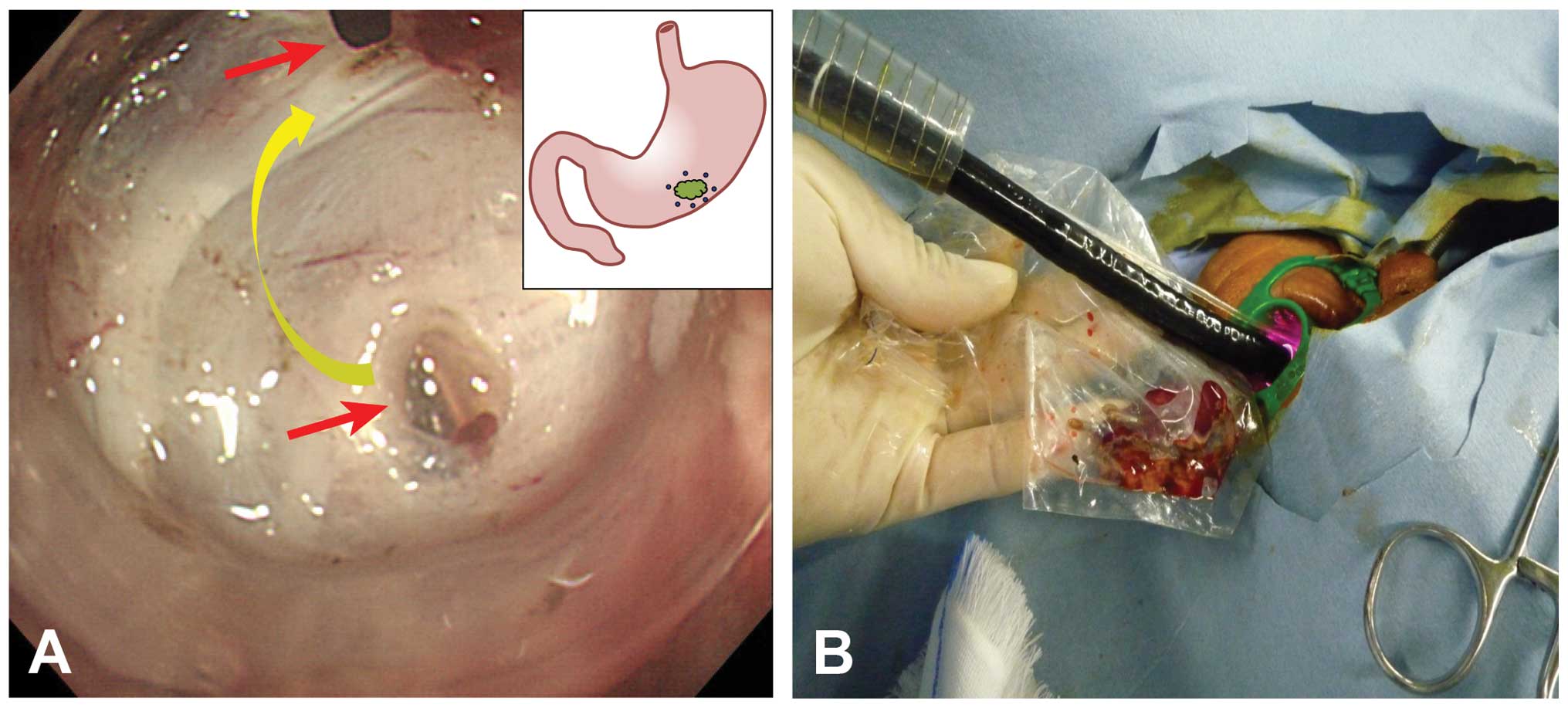
incisional generator device. Perforation holes were placed at 5-mm
intervals to secure a reliable incision line at the time of stomach
collapse. The full-thickness resection was performed using the
IT-knife-2 and by connecting two small holes, the openings of which
were of equal distance from the gastric wall (Fig. 4A) (4). Following the tumor excision, a
full-thickness suture was placed under laparoscopy. The tumor was
collected through the mouth using an Endo Catch (Covidien Japan)
(Fig. 4B).
Results
Patient characteristics
The patient characteristics were as follows: Mean
age of 68.2 years (range, 44–81 years); male-to-female ratio of
6:10, with no significant difference (P=0.627); lesion sites at
upper (U), middle (M) and lower (L) regions of the stomach (U/M/L)
of 9/6/1, with a significant difference between tumor locations.
Almost all GISTs were located in the U to M regions (P=0.005); and
the average tumor diameter was 28.3 mm (range, 8–54 mm) (Table I). A total of nine hybrid EFTR
procedures were performed in the U region, including three cases in
which the proximal margins were within 1.5 cm of the
esophagogastric junction. In the latter cases, an 8-mm safety
margin was created from the proximal side of the tumor margin. In
all three cases, the surgical margins were negative. EFTR was
successfully performed in all the patients and there were no
complications requiring conversion to open surgery, even in cases
with tumors >40 mm. The mean surgical time was 271 min (range,
100–480 min), and the surgical time became progressively faster
with each successive surgery. In the first seven cases, the
surgical times were 360–480 min, whereas the surgical times for the
last nine cases were 100–150 min. The surgical time appeared to be
longer when the tumors were located in the U region of the stomach.
As the sample size was small, a correlation between the tumor
location and surgical time could not definitively be
established.
 | Table IPatient characteristics. |
Table I
Patient characteristics.
| Variables | Patients (n=16) | P-value |
|---|
| Age, mean (range),
year | 68.2 (44–81) | |
| Gender
(male/female) | 6/10 | 0.627 NSa |
| Tumor location
(U/M/L) | 9/6/1
(56.3%/37.5%/6.2%) | 0.005c (H=10.5b) |
| Tumor size mean
(range), mm | 28.3 (8–54) | |
Results obtained
The pathological safety margin from the cut end to
the tumor was an average of 8 mm (range, 6–11 mm). All resected
tumors were within the resected masses. There were 12 and four
patients with mitotic counts of <5 and 5–10, respectively, with
a significant difference (P=0.01). Immunohistochemical stains
showed that tumors from 13 and 10 patients (81.2 and 62.5%) were
positive for KIT and CD34, respectively (P=0.328). The resected
surgical margins were negative. According to Fletcher's risk
classification, there were 5/8/3 patients at ‘extremely low’,
‘low’, and ‘intermediate’ risk (31.2/50/18.8%), respectively
(P=0.003). High-risk patients were not included. The mean
postoperative hospital stay was 12.3 days (range, 10–15 days)
(Table II). Patient safety was of
primary importance, and therefore, the mean hospital stay was
longer than the hospital stays for laparoscopic surgery. There were
no complications, including bleeding, peritonitis, anastomotic
leakage or stenosis.
 | Table IIResults of patients. |
Table II
Results of patients.
| Variables | Patients (n=16) | P-value |
|---|
| EFTR completion/shift
to laparotomy | 16/0 | |
| EFTR operation time,
mean (range), min | 271 (100–480) | |
| Mitosis (/50 high
power field) <5/5–10 | 12/4 (75%/25%) | 0.01a |
| Immunohistochemical
stain KIT/CD34 | 13/10
(81.2%/62.5%) | 0.328 NSa |
| Histological
risk | | |
| Extremely
low/low/intermediate/high (Fletcher classification) | 5/8/3/0
(31.2%/50%/18.8%) | c0.003 (bH=12.36) |
| Hospitalization, mean
(range), days | 12.3 (10–15) | |
Discussion
Several techniques have been suggested for the local
resection of GIST, mainly laparoscopic local gastric resection
(10, 11), laparoscopic and endoscopic
cooperative surgery (LECS) (12),
intragastric surgery (13) and
hybrid EFTR (NOTES) (14). Of
these procedures, hybrid EFTR has the advantage of enabling the use
of electric scalpels (including the IT-knife-2 and dual knife).
These scalpels are designed for flexible endoscopes and enable
detailed and minimal tumor resection, even when a curved incision
line or complex incision line along the tumor contour is made. As
the sample size was small, the surgical time was shortened to
approximately the same length as that of laparoscopic local gastric
resection, indicating that surgical time is not an issue in hybrid
EFTR. However, this experimental procedure has certain limitations.
One limitation is that there have not been any randomized trials to
compare hybrid EFTR with conventional techniques. Multi-center
trials prior to the clinical acceptance of hybrid EFTR should
therefore be conducted. The surgical procedure was designed to
ensure incisional accuracy and excisional safety using the
following steps: Setting the incision line according to the ESD
method, using the double-scope technique to make and visualize the
incision, creating the holes at equal distances from the abdominal
wall (4) and using the duodenal
balloon occlusion method (8). If
hybrid EFTR is performed in accordance with these surgical
procedures, LECS and hybrid EFTR may be established as less
invasive, tailored surgical options.
In conclusion, hybrid EFTR, which is an
ultra-minimally invasive surgery, is a safe and established
surgical endoscopy procedure.
Acknowledgements
The authors acknowledge the Kagawa NOTES project for
supporting the present study.
References
|
1
|
Kaouk JH, Haber GP, Goel RK, et al: Pure
natural orifice translumenal endoscopic surgery (NOTES)
transvaginal nephrectomy. Eur Urol. 57:723–726. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Isariyawongse JP, McGee MF, Rosen MJ,
Cherullo EE and Ponsky LE: Pure natural orifice transluminal
endoscopic surgery (NOTES) nephrectomy using standard laparoscopic
instruments in the porcine model. J Endourol. 22:1087–1091. 2008.
View Article : Google Scholar
|
|
3
|
Isayama H, Kogure H and Koike K:
Endoscopic transgastric pure NOTES cholecystectomy with
naso-gallbladder drainage tube placement and injection of a
hyaluronic acid mixture (with Video). J Hepatobiliary Pancreat Sci.
18:106–111. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mori H, Kobara H, Kobayashi M, et al:
Establishment of pure NOTES procedure using a conventional flexible
endoscope: review of six cases of gastric gastrointestinal stromal
tumors. Endoscopy. 43:631–634. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Martínek J, Ryska O, Filípková T, et al:
Natural orifice transluminal endoscopic surgery vs laparoscopic
ovariectomy: complications and inflammatory response. World J
Gastroenterol. 18:3558–3564. 2012.PubMed/NCBI
|
|
6
|
Vieira JP, Linhares MM, Caetano EM Jr, et
al: Evaluation of the clinical and inflammatory responses in
exclusively NOTES transvaginal cholecystectomy versus laparoscopic
routes: an experimental study in swine. Surg Endosc. 26:3232–3244.
2012. View Article : Google Scholar
|
|
7
|
Kobara H, Mori H, Fujiwara S, Nishiyama N,
Kobayashi M and Masaki T: Bloc biopsy by tunneling method using the
endoscopic submucosal dissection for upper gastrointestinal
submucosal tumor. Endoscopy. 44 (Suppl 2):E197–E198. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mori H, Kobara H, Inoue H, et al: New
technique for safer endoscopic submucosal dissection using the
duodenal balloon occlusion method. J Gastroenterol Hepatol.
27:81–85. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mori H, Kobara H, Rafiq K, et al: Effects
of gastric irrigation on bacterial counts before endoscopic
submucosal dissection: a randomized case control prospective study.
PLoS One. 8:e653772013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Choi SM, Kim MC, Jung GJ, et al:
Laparoscopic wedge resection for gastric GIST: long-term follow-up
results. Eur J Surg Oncol. 33:444–447. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Warsi AA and Peyser PM: Laparoscopic
resection of gastric GIST and benign gastric tumours: evolution of
a new technique. Surg Endosc. 24:72–78. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hiki N, Yamamoto Y, Fukunaga T, et al:
Laparoscopic and endoscopic cooperative surgery for
gastrointestinal stromal tumor dissection. Surg Endosc.
22:1729–1735. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hara J, Nakajima K, Takahashi T, et al:
Laparoscopic intragastric surgery revisited: its role for
submucosal tumors adjacent to the esophagogastric junction. Surg
Laparosc Endosc Percutan Tech. 22:251–254. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Abe N, Takeuchi H, Ooki A, et al: Recent
developments in gastric endoscopic submucosal dissection: towards
the era of endoscopic resection of layers deeper than the
submucosa. Dig Endosc. 25 (Suppl 1):64–70. 2013. View Article : Google Scholar : PubMed/NCBI
|