Introduction
Euphorbia tirucalli (E. tirucalli),
commonly referred to as ‘aveloz’, is a tropical and subtropical
ornamental plant, traditionally used in folk medicine for the
treatment of syphilis, asthma, rheumatism, arthritis (1) and cancer (2–7). This
plant most likely originated from tropical East Africa, and is
reportedly endemic in a number of African countries; it may also be
found in southern Europe, Asia and the Americas, where it is used
for decorative as well as medicinal purposes (8). E. tirucalli is widely cultivated
in Brazil, particularly in the north and northeast regions
(9).
E. tirucalli produces a latex that is a
vesicant and is commonly used as a remedy against several diseases.
However, most of its medicinal properties are reported informally
and there appears to be little scientific evidence to validate them
(8). The main substances present in
E. tirucalli latex are cyclotirucanenol (triterpene),
diterpene ester, steroids and tirucalicine (diterpene) (8). Its active components include euphol,
euphorbol, euphorone, isoeuphoral, taraxasterol, tirucalol, citric
acid, glucose, kamepferol, malic acid, sapogenin acetate and
succinic acid (10,11). Recently, Palharini et al
(12) investigated eutirucallin, a
lectin with antitumor and antimicrobial properties. Some of these
active components have been reported to have biological activities,
such as preventive anticancer activity, antitumor, antimutagenic,
antibacterial, laxative, antiseptic, disinfectant,
anti-inflammatory, antistreptococcal, antiparasitic, antiulcer,
analgesic, antiasthmatic and expectorant properties, as well as
anticancer effects against specific types of cancer of the breast,
lung, cervix, esophagus and mouth (13).
Valadares et al (5) first reported that the extract of E.
tirucalli modulated myelopoiesis and reduced the growth of
mouse Ehrlich ascites tumor. Wang et al (14) demonstrated that euphol inhibited the
growth of T74D human breast cancer cells and reduced the levels of
cyclin A and B1 expression, which corresponded to the decreased
distribution of cells in the S and G2/M phases, respectively. These
results indicated that euphol is an active ingredient of E.
tirucalli that exerts anticancer effects, possibly by arresting
cell cycle progression of cancer cells. Santos et al
(13) also demonstrated that E.
tirucalli extract inhibited the growth of Ehrlich ascites
tumor. Furthermore, euphol inhibited the growth of human gastric
cancer cells by modulating apoptosis mediated by ERK1/2 and cyclin
D1 (7).
The aim of the present study was to investigate the
in vitro and in vivo effects of diluted E.
tirucalli latex on B16/F10 melanoma cells.
Materials and methods
E. tirucalli latex
Euphorbia tirucalli Lineu (Euphorbiaceae) was
obtained from the plant resource center Sabor da Fazenda (São
Paulo, Brazil) and was certified by the ECOCERT body (http://www.brazil.ecocert.com/index/).
In addition, a sample of pressed plant was obtained, dried in an
oven, fixed on a standard-size paperboard accompanied by a label
containing information on the plant and the collection site, and
stored in the herbarium of the Department of Botany of the
Institute of Biosciences of the University of São Paulo, Brazil
(http://www.ib.usp.br/en/botany-welcome.html).
All the E. tirucalli latex required for the
experiment was collected directly from the stems; latex drops were
collected from the plant stem in a sterilized glass beaker.
Dilution of E. tirucalli latex in saline
solution was performed according to its popular use, namely 9 drops
(0.0934 g) in 1 liter of saline solution. A total of 5 µl of this
solution, containing 0.467 µg of E. tirucalli latex (for an
animal weighing 25 g) was transferred to 200 µl (q.s.) and this
volume was administered to each mouse. This quantity of latex is
equivalent to that recommended in popular medicine for a person
weighing 70 kg (1.3 g).
Immediately after the preparation, the diluted latex
saline solution of E. tirucalli and the control saline
solution were stored at 4°C.
Culture of B16/F10 cells
B16/F10 murine melanoma cells were cultured with
Dulbecco's modified Eagle's medium (DMEM) supplemented with 10%
fetal bovine serum, penicillin (50 IU/ml), streptomycin (50 mg/ml)
and L-glutamine (2 mmol/l). The bottles were kept in a humidified
incubator at 37°C in an atmosphere of 5% CO2 and 95% air under
temperature-controlled conditions.
In vitro experiment
Evaluation of cell proliferation with
the MTT assay
On day 1, B16/F10 murine melanoma cells were plated
on three flat-bottomed 96-well plates at a density of
3×103 cells/well (150 µl of DMEM/well). DMEM was
supplemented with fetal bovine serum, penicillin (50 IU/ml),
streptomycin (50 mg/ml) and L-glutamine (2 mmol/l). A stock
solution of latex in DMEM at a concentration of 0.1037 µg/µl was
prepared. Serial dilutions were prepared using DMEM (1/2, 1/4, 1/8,
1/16, 1/32, 1/64, 1/128, 1/256, 1/512, 1/1,024, 1/2,048, 1/5,096
and 1/11,192). On day 2, 100 µl/well of each dilution of the stock
solution was added to each well. The amount of latex per dilution
is shown in Table I. The control
group was treated with 100 µl/well DMEM.
 | Table I.Amount of latex corresponding to each
dilution present in each well of the 250-µl plate. |
Table I.
Amount of latex corresponding to each
dilution present in each well of the 250-µl plate.
| Dilution | Latex amount (µg)
per well | Latex concentration
(ng/µl) per well |
|---|
| 1/2 |
5.185 |
82.96 |
| 1/4 |
2.5925 |
41.48 |
| 1/8 |
1.2962 |
20.74 |
| 1/16 |
0.6481 |
10.37 |
| 1/32 |
0.3240 |
5.185 |
| 1/64 |
0.1620 |
2.5925 |
| 1/128 |
0.0810 |
1.29625 |
| 1/256 |
0.0405 |
0.648125 |
| 1/512 |
0.0202 |
0.324063 |
| 1/1,024 |
0.0101 |
0.162031 |
| 1/2,048 |
0.0051 |
0.081016 |
| 1/5,096 |
0.0025 |
0.040508 |
| 1/11,192 |
0.0013 |
0.020254 |
After 24, 48 and 72 h, 10 µl tetrazolium
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT;
Amresco, LLC, Solon, OH, USA) was added to both the control and
experimental wells. The salt was added to each well of the plates
to be metabolized by viable cells, and incubated for 3 h in a
humidified chamber at 37°C in an atmosphere of 5% CO2
and 95% air under temperature-controlled conditions.
Subsequently, the cells were centrifuged at 2,755 ×
g for 10 min, and the supernatant was discarded. The formazan
crystals were dissolved with 100 µl/well DMSO. The reading was
performed in a Multiskan EX (Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) ELISA reader at 570 nm. The experiments were
carried out in six replicates.
In vivo experiment
Experimental animals
A total of 14 C57BL/6 male mice, aged 2 months and
weighing 25–30 g, were used in the experiments. The mice were
obtained from the Animal Facility of the Department of Pathology of
the School of Veterinary Medicine and Animal Science of the
University of São Paulo and were kept under the following
environmental conditions: 12-h light/dark cycle, temperature 22±2°C
and relative humidity 45–65%. During the experimental period, the
animals remained in the animal facility, and had access to water
and balanced feed ad libitum. The experimental protocols were
approved by the Committee on Ethics in Animal Use (CEUA) of the
School of Veterinary Medicine and animal Science of the University
of São Paulo (SVMAS-USP, process no. 3410250216) and the School of
Medicine of the University of São Paulo (FM-USP, process no.
043/16).
Inoculation of B16/F10 melanoma cells
into the tail vein for the development of lung metastasis in
mice
Cell suspensions containing murine melanoma B16/F10
cells (100 µl containing 5×105 cells/animal) were
inoculated into the tail vein of 6-month-old C57BL/6 male mice
(15–17). The animals were treated with E.
tirucalli latex daily by gavage (0.467 µg/25 g in 200 µl),
diluted as 9 drops of latex in 1 liter of saline solution, or
saline solution for 14 days (control), starting 1 week after
inoculation. At the end of the experiment, the animals were
euthanized with an overdose of intraperitoneal ketamine and
xylazine solution (doses >100 mg/kg ketamine and 10 mg/kg
xylazine were used), and death was confirmed by verifying the lack
of respiratory, cardiac and nervous functions. The lungs were
removed and fixed in buffered formalin solution (10%) for
histopathological and morphometric examination. Representative
samples of the lungs, liver, kidneys and spleen were fixed in 10%
formalin, embedded in paraffin wax and the 5-µm histological
sections were routinely processed and stained with hematoxylin and
eosin (H&E) for histopathological analysis.
Quantification of the volumetric
fraction of lungs occupied by metastatic melanoma
The volumetric fraction occupied by the lung
colonies of the melanoma group and the E. tirucalli
latex-treated melanoma group was calculated by the dot-counting
method (18). The lungs were fixed
in 10% formalin and embedded in paraffin. A total of 15
non-consecutive, randomly selected histological sections of the
lungs of each animal (7 animals from the melanoma group and 7
animals from the E. tirucalli latex-treated melanoma group)
were prepared.
The histological slides were photographed under the
Leica M165C stereo microscope (Leica Microsystems, Inc.), using the
image capture system composed by the Leica DFC 290 camera and the
LAS V4.1 program in the Laboratory of Stochastic Stereology and
Chemical Anatomy (LSSCA-USP). Next, the images covering the whole
area of the histological section were superimposed by a grid of
points where all the points that covered the lung section were
counted, and the points that covered only the pulmonary melanoma
metastases were registered. The process was repeated 5 times for
each image by rotating the grid. The percentage of points that
coincided with the metastases in all sections was calculated in
relation to the total number of points that coincided with the lung
section, and it corresponded to the proportion of volume occupied
by the nodules in relation to the total lung tissue.
Statistical analysis
To compare cell viability in cell cultures treated
with E. tirucalli latex, the Kruskall-Wallis statistical
test was used. Subsequent comparisons were performed by the Dunn's
post hoc test. To evaluate the volumetric fraction of lungs
occupied by metastatic melanoma, the Student's t-test was applied,
and data are expressed as mean ± standard deviation. Both the
statistical tests and the assembly of the graphs and tables were
performed with Prism 6 software (GraphPad Software, Inc., La Jolla,
CA, USA) and GraphPad InStat version 3.10. P-values <0.05 were
considered to indicate statistically significant differences.
Results
Evaluation of cell viability following
treatment of murine B16/F10 melanoma cells with E. tirucalli
latex
To investigate the effect of E. tirucalli
latex treatment on murine B16/F10 melanoma cells, different
dilutions (1/2, 1/4, 1/8, 1/16, 1/32, 1/64, 1/128, 1/256, 1/512,
1/1,024, 1/2,048, 1/5,096 and 1/11,192) were used. The cell
viability was evaluated by the MTT assay at 24, 48 and 72 h.
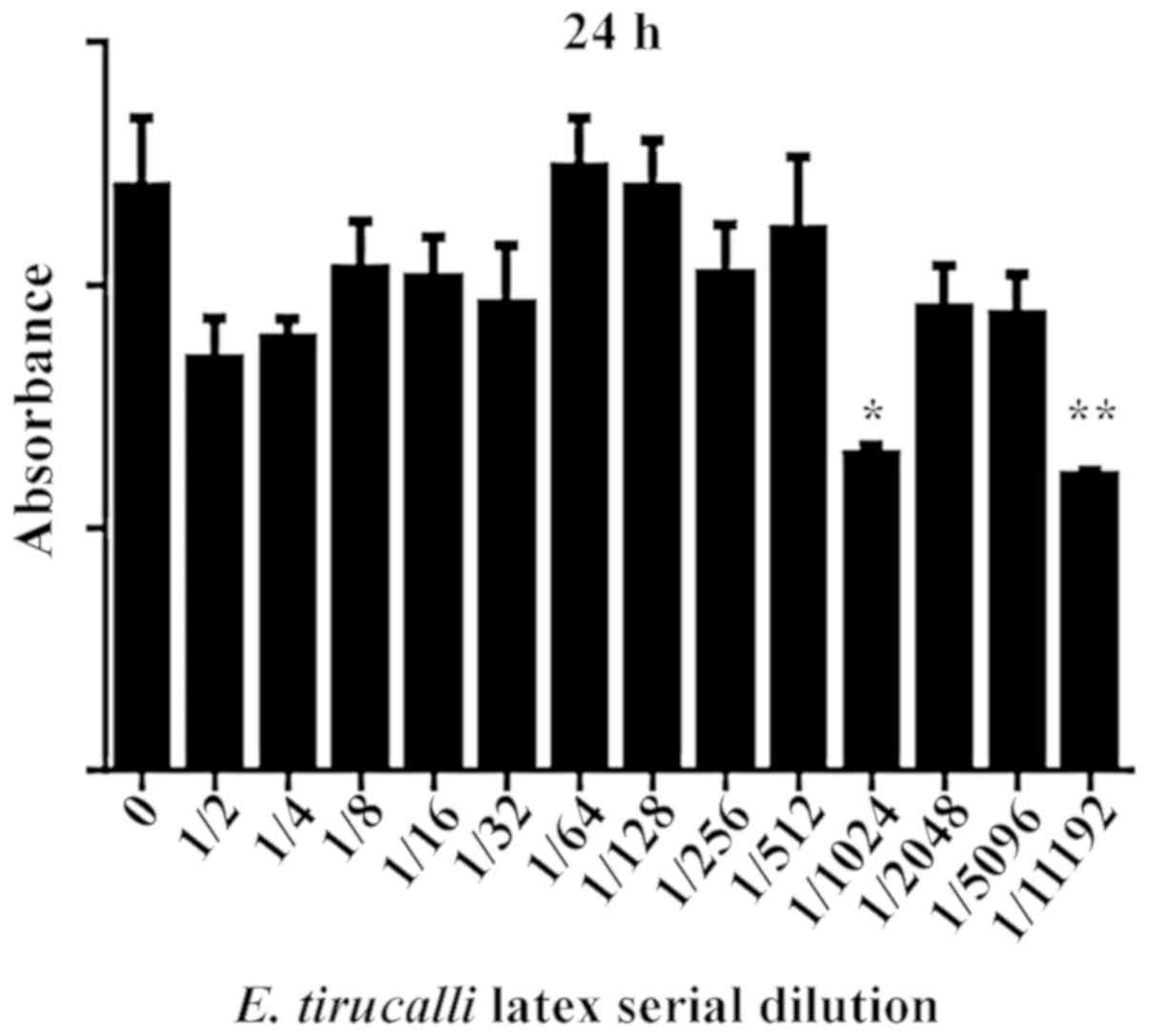
In this series of experiments, cell incubation at
different dilutions of E. tirucalli for 24 h reduced cell
viability at the dilutions of 1/1,024 (46.58%) and 1/11,192
(52.67%) (P<0.05; Fig. 1).
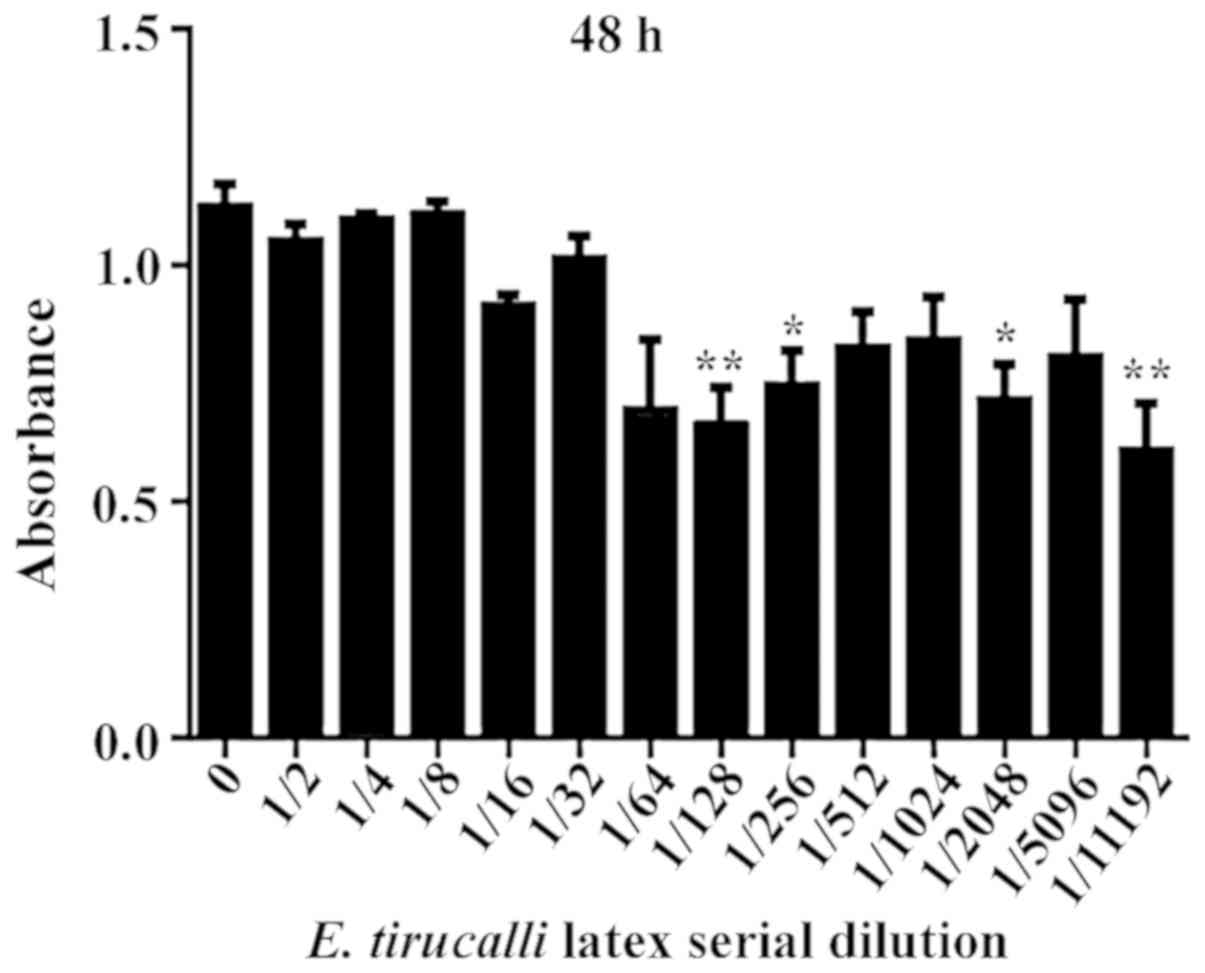
Incubation of the cells with different dilutions of
E. tirucalli for 48 h reduced cell viability at the
dilutions of 1/128 (47.67%), 1/256 (41.83%), 1/2,048 (43.17%) and
at 1/11,192 (49.83%) (P<0.0001; Fig.
2).
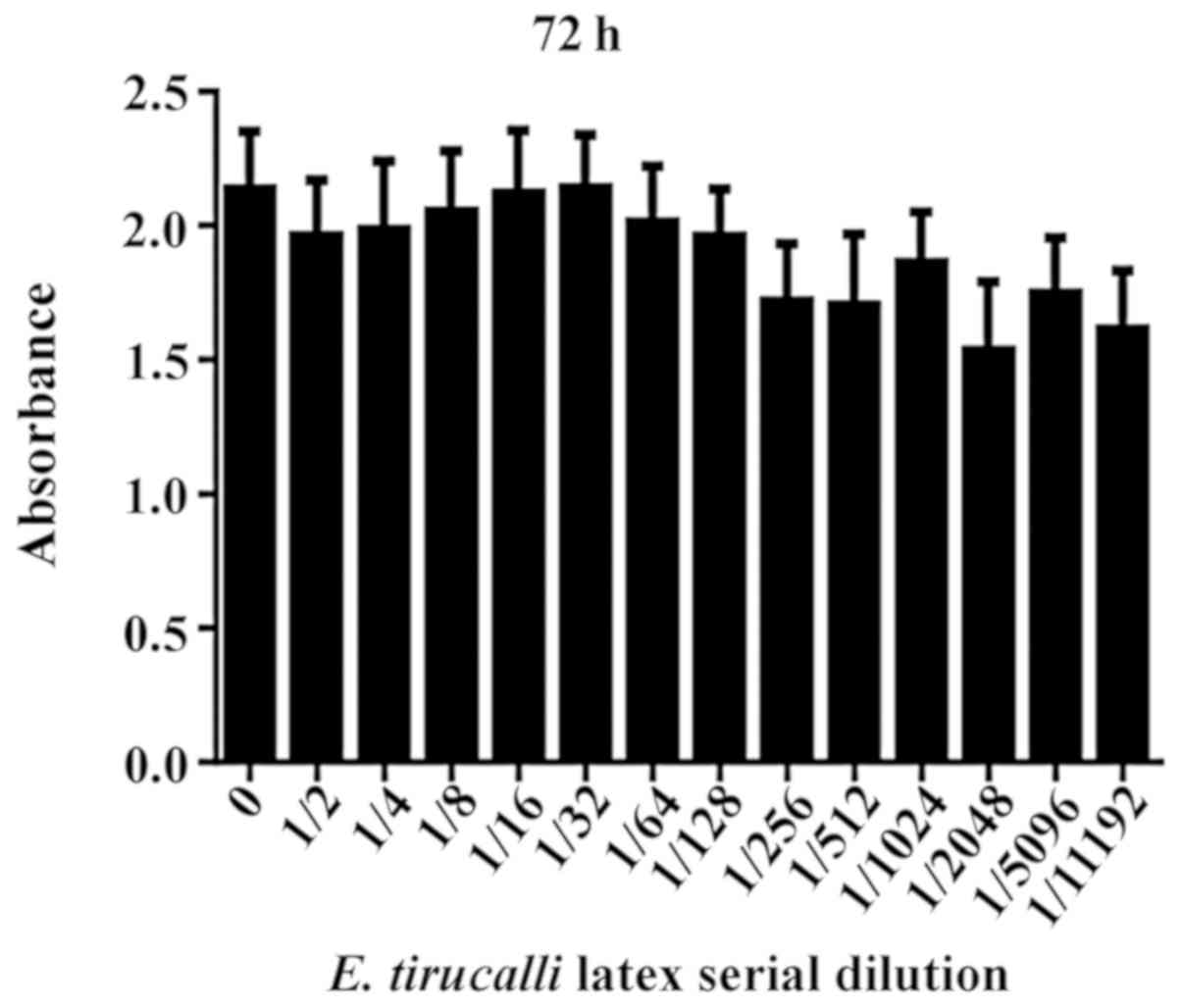
However, incubation with different dilutions of
E. tirucalli latex for 72 h did not significantly reduce
cell viability any further (P>0.05; Fig. 3).
Evaluation of the effects of E.
tirucalli treatment on the development of B16/F10 melanoma
metastasis
For this experiment, it was examined whether
treatment with E. tirucalli could also reduce the number of
lung metastases. A total of 100 µl of B16/F10 murine melanoma cell
suspension were inoculated into the tail vein of C57BL/6 mice
(5×105 cells/animal).
After 14 days of treatment with E. tirucalli,
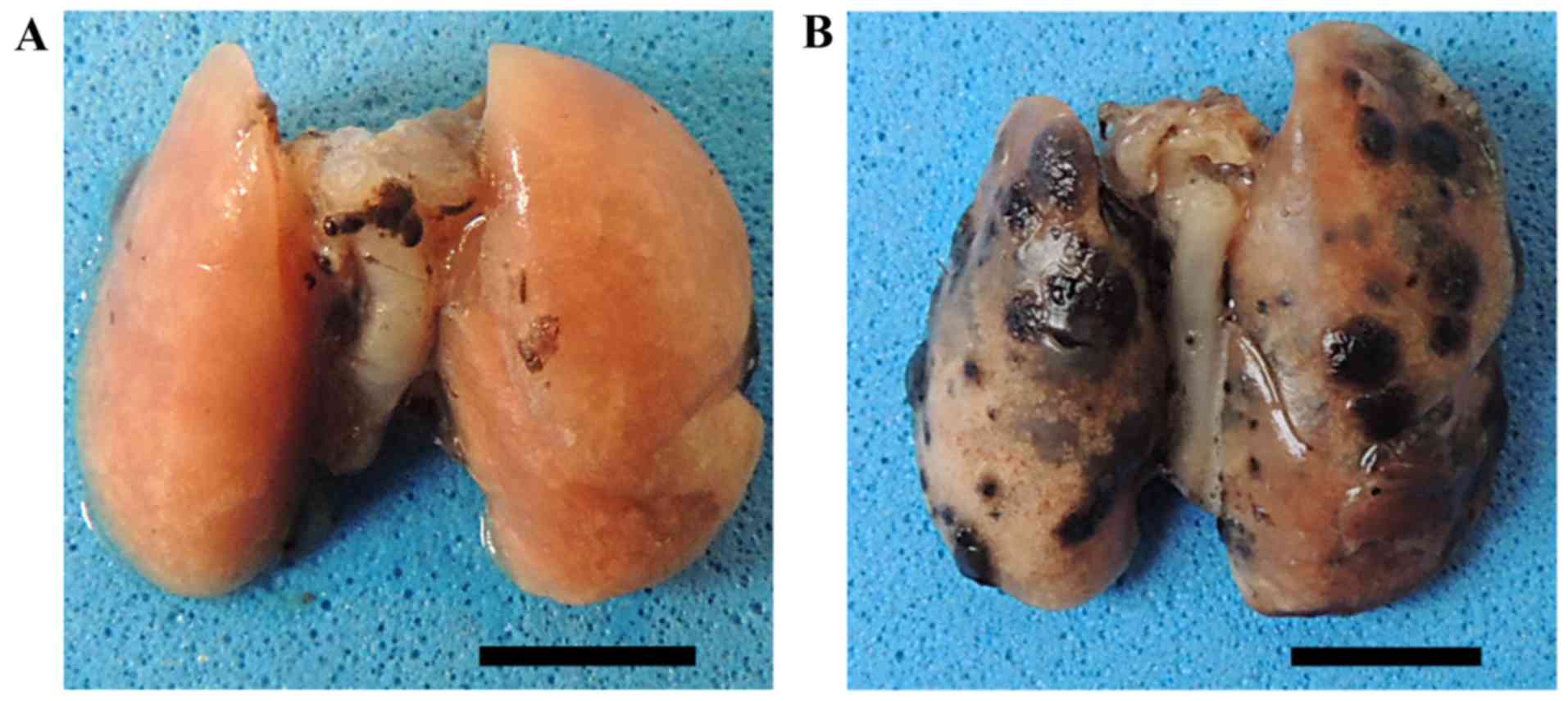
all mice were euthanized and necropsies were performed.
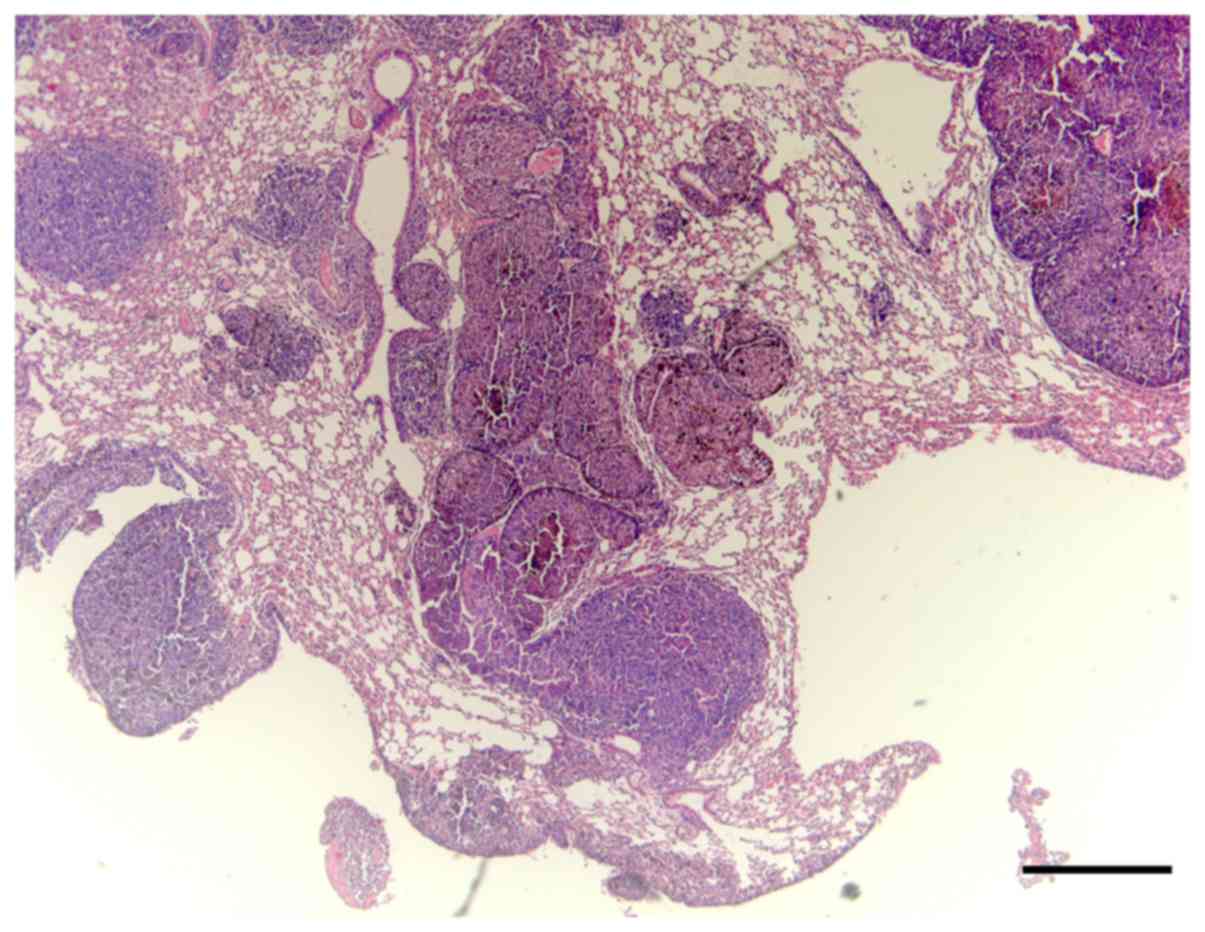
Macroscopically, the lungs of the control group had numerous
melanoma nodules, whereas the lungs of the mice treated with E.
tirucalli exhibited markedly smaller numbers of metastatic
nodules (Fig. 4).
Quantification of the volumetric
fraction occupied by metastatic melanoma
The results of the quantification of the volumetric
fraction occupied by metastatic melanoma foci in E.
tirucalli latex-treated (MT) or -untreated (M) C57Bl6 mouse
lungs are summarized in Table II.
While in control mice 35±18% of the lungs were occupied by
melanoma, only 10.5±7.7% of the lung area was occupied by melanoma
in E. tirucalli-treated mice. The difference was
statistically significant (Student's t-test, P=0.024; Figs. 5 and 6).
 | Table II.Volume fraction of lung occupied by
B16/F10 melanoma metastatic colonies in E. tirucalli
latex-treated or untreated C57Bl6 mice. |
Table II.
Volume fraction of lung occupied by
B16/F10 melanoma metastatic colonies in E. tirucalli
latex-treated or untreated C57Bl6 mice.
| Groups | Number of
animals | Mean (%) ± SD |
|---|
| Untreated mice | 7 | 65.7±15.4 |
| E.
tirucalli-treated mice | 7 | 13.8±7.5 |
|
P-valuea |
| 0.0001 |
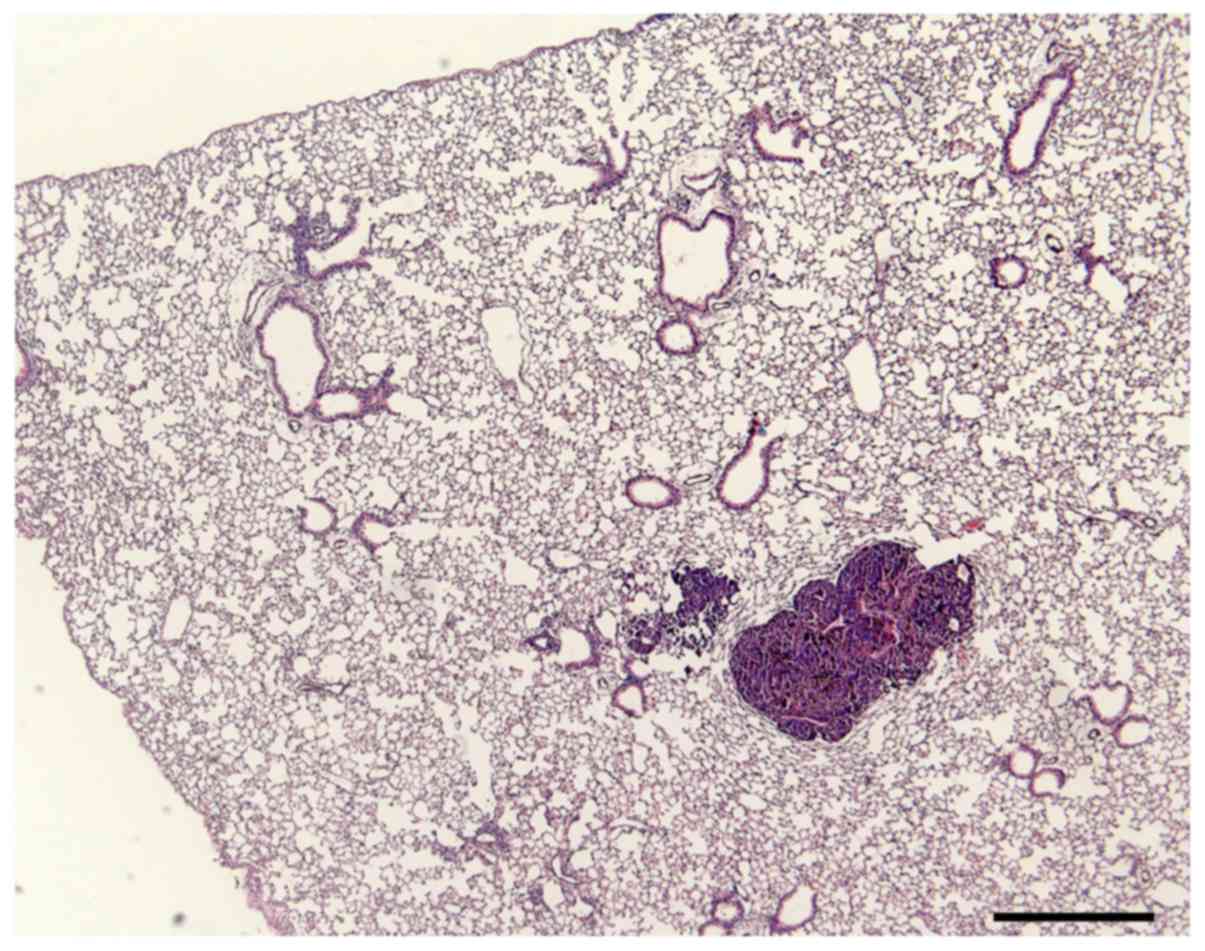
Histopathological analysis
No microscopic alterations were observed in the
histological sections of the liver, kidneys and spleen of mice
treated with diluted E. tirucalli latex; in the lungs, small
areas of congestion and hemorrhage were observed (control group).
These results indicate that the E. tirucalli latex does not
cause toxicity to mice at the abovementioned dilutions.
Discussion
Treatment modalities popular in folk medicine,
mostly medicinal plants, may be of therapeutic value in a number of
diseases. However, they must be approached with caution and
thoroughly investigated in order to objectively evaluate their
efficacy and safety (19).
The objective of the present study was to
scientifically evaluate the effects of E. tirucalli latex on
B16/F10 murine melanoma cells in vitro and in vivo.
Only little information is currently available in the literature on
E. tirucalli latex and, to the best of our knowledge, this
is the first such study on melanoma, a highly lethal cancer
affecting humans and animals.
In the in vitro experiment, it was observed
that E. tirucalli latex significantly reduced the viability
of B16/F10 murine melanoma cells at high dilutions. Silva et
al (20) demonstrated that
euphol, a major component of E. tirucalli latex, exerted
cytotoxic effects on several human cell lines.
In fact, dilutions of E. tirucalli latex were
effective in reducing melanoma cell viability (as detected by MTT
assay) after 24 and 48 h of treatment, but not after 72 h. In
addition, the higher dilutions of E. tirucalli were
consistently more effective in reducing the viability of tumor
cells. These findings indicate that the effectiveness of E.
tirucalli latex against melanoma cells was time-dependent
(therefore, it must be administered daily or every other day in
order to be effective), and its action apparently occurs at low
doses, or possibly at doses that effectively reach the tumor cells
inside the tumor mass when the diluted plant latex is administered
orally. However, this hormetic effect must be further
investigated.
Although in vitro studies are informative,
when isolated, they cannot predict the in vivo efficacy of
new therapies. These must be tested either in laboratory animals,
or, optimally, in well-conducted clinical trials. In the present
study, an in vivo experiment was conducted to confirm the
efficacy of the in vitro results.
Metastasis is characteristic of malignant tumors,
and is considered as the main cause of death among cancer patients.
Therefore, we investigated the effectiveness of E. tirucalli
latex in controlling metastasis in mice. Spontaneous metastasis
mouse models are rare; therefore, in the present study, mice were
inoculated with tumor cells through the tail vein to generate lung
metastases (15–17) in order to evaluate the possible
effect of E. tirucalli latex on metastatic growth. It was
observed that the 14-day treatment with diluted E. tirucalli
latex significantly reduced the volume fraction of the mouse lungs
occupied by metastatic melanoma nodules, indicating that it is
important to further evaluate the effect of this popular dilution
of E. tirucalli on other tumors and in the context of other
treatment protocols.
Furthermore, when the vital organs of the mice were
microscopically examined, it was verified that E. tirucalli
latex, at the popularly used dilution of 9 drops in 1 liter of
saline solution, was not associated with adverse histopathological
changes in the liver, kidneys or spleen. The absence of
histopathological alterations indicates lack of toxicity of E.
tirucalli at the commonly used dilution.
The experiments performed using B16/F10 melanoma
cells (in vitro and metastasis assays) revealed consistent
antineoplastic effects, confirming the efficacy of E.
tirucalli latex against B16/F10 murine melanoma. These
anticancer effects are most likely associated with euphol (7,14) and/or
eutirucallin (12). However, the
exact mechanisms underlying the effects of diluted E.
tirucalli latex require further investigation.
In conclusion, the goal of any experimental study is
to test hypotheses which, if scientifically proven, may be used to
improve the quality of life and the survival of patients. In the
present study, E. tirucalli latex has shown promising
antineoplastic properties that warrant further validation in
clinical trials.
Acknowledgements
This study is part of the Master's Dissertation of
Rafael Lanciani Brunetti at the Graduate Program on Experimental
Physiopathology of the School of Medicine of the University of São
Paulo, Brazil. Rafael Lanciani Brunetti was the recipient of a
fellowship from Capes, Ministry of Education, Brazil.
Funding
The present study was supported by grants from the
National Council for the Scientific and Technologic Development,
CNPq, Ministry of Science, Technology, Innovation and
Communications of Brazil, and the São Paulo Research Foundation,
FAPESP.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
RLB performed the in vitro and animal
studies. DPADP obtained the E. tirucalli plant, standardized
the harvesting and dilution of the latex, and helped in the in
vitro experiments. IIMDF, MKN, MPDG and COMSG helped in the
in vitro studies with melanoma cells, including the
statistical analysis. CMCM and NQH helped in the in vivo
experiments. MLZD and FJHB mentored the study, supervised the
students and reviewed the manuscript. All the authors have read and
approved the final version of this manuscript.
Ethics approval and consent to
participate
The experimental protocols were approved by the
Committee on Ethics in Animal Use (CEUA) of the School of
Veterinary Medicine and animal Science of the University of São
Paulo (SVMAS-USP, process no. 3410250216) and the School of
Medicine of the University of São Paulo (FM-USP, process no.
043/16).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bani S, Kaul A, Khan B, Gupta VK, Satti
NK, Suri KA and Qazi GN: Anti-arthritic activity of a biopolymeric
fraction from Euphorbia tirucalli. J Ethnopharmacol.
110:92–98. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Imai S, Sugiura M, Mizuno F, Ohigashi H,
Koshimizu K, Chiba S and Osato T: African Burkitt's lymphoma: A
plant, Euphorbia tirucalli, reduces Epstein-Barr
virus-specific cellular immunity. Anticancer Res. 14:933–936.
1994.PubMed/NCBI
|
|
3
|
Cataluña P and SMK Rates: The traditional
use of the latex from Euphorbia tirucalli Linnaeus
(Euphorbiaceae) in the treatment of cancer in South Brazil. Acta
Hortic. 501:289–296. 1999. View Article : Google Scholar
|
|
4
|
Betancur-Galvis LA, Morales GE, Forero JE
and Roldan J: Cytotoxic and antiviral activities of Colombian
medicinal plant extracts of the Euphorbia genus. Mem Inst Oswaldo
Cruz. 97:541–546. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Valadares MC, Carrucha SG, Accorsi W and
Queiroz ML: Euphorbia tirucalli L. modulates myelopoiesis and
enhances the resistance of tumour-bearing mice. Int
Immunopharmacol. 6:294–299. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Agra MF, Silva KN, Basílio IJ, Freitas PF
and Barbosa-Filho JM: Survey of medicinal plants used in the region
Northeast of Brazil. Rev Bras Farmacogn. 18:472–508. 2008.
View Article : Google Scholar
|
|
7
|
Lin MW, Lin AS, Wu DC, Wang SSW, Chang FR,
Wu YC and Huang YB and Huang YB: Euphol from Euphorbia
tirucalli selectively inhibits human gastric cancer cell growth
through the induction of ERK1/2-mediated apoptosis. Food Chem
Toxicol. 50:4333–4339. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mwine J, Van Damme P, Hastilestari BR and
Papenbrock J: Euphorbia tirucalli L. (Euphorbiaceae) - The
Miracle Tree: Current Status of KnowledgeAfrican Natural Plant
Products Volume II: Discoveries and Challenges in Chemistry,
Health, and Nutrition ACS Symposium Series. American Chemical
Society; Washington, DC: 2013
|
|
9
|
Lorenzi H and Matos FJ: Euphorbia
tirucalli Plantas Medicinais do Brasil: Nativas e
ExóticasInstituto Plantarum; São Paulo: pp. 211–212. 2002
|
|
10
|
Newbold GT and Spring FS: The Euphorbia
resins. Part I. Euphol. The isolation of Euphol and alfa-Euphorbol
from Euphorbium. J Chem Soc. 249–252. 1944. View Article : Google Scholar
|
|
11
|
Fürstenberger G and Hecker E: On the
active principles of the Euphorbiaceae, XII. Highly unsaturated
irritant diterpene esters from Euphorbia tirucalli
originating from Madagascar. J Nat Prod. 49:386–397. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Palharini JG, Richter AC, Silva MF,
Ferreira FB, Pirovani CP, Naves KS, Goulart VA, Mineo TW, Silva MJ
and Santiago FM: Eutirucallin: A lectin with antitumor and
antimicrobial properties. Front Cell Infect Microbiol. 7:1362017.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Santos OJ, Sauaia Filho EN, Nascimento FR,
Júnior FC, Fialho EM, Santos RH, Santos RA and Serra IC: Use of raw
Euphorbia tirucalli extract for inhibition of ascitic
Ehrlich tumor. Rev Col Bras Cir. 43:18–21. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang L, Wang G, Yang D, Guo X, Xu Y, Feng
B and Kang J: Euphol arrests breast cancer cells at the G1 phase
through the modulation of cyclin D1, p21 and p27 expression. Mol
Med Rep. 8:1279–1285. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fidler IJ, Gersten DM and Riggs CW:
Relationship of host immune status to tumor cell arrest,
distribution, and survival in experimental metastasis. Cancer.
40:46–55. 1977. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fukumasu H, Avanzo JL, Nagamine MK,
Barbuto JA, Rao KV and Dagli ML: Paullinia cupana Mart var.
sorbilis, guaraná, reduces cell proliferation and increases
apoptosis of B16/F10 melanoma lung metastases in mice. Braz J Med
Biol Res. 41:305–310. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Aherne WA and Dunnil MS: Morphometry.
Edward Arnold Ltd.; London: 1982
|
|
18
|
Weibel ER: Stereological Methods:
Practical Methods for Biological MorphometryAcademic Press; London:
1979
|
|
19
|
Zips D, Thames HD and Baumann M: New
anticancer agents: In vitro and in vivo evaluation. In Vivo.
19:1–7. 2005.PubMed/NCBI
|
|
20
|
Silva VA, Rosa MN, Tansini A, Oliveira RJ,
Martinho O, Lima JP, Pianowski LF and Reis RM: In vitro screening
of cytotoxic activity of euphol from Euphorbia tirucalli on
a large panel of human cancer-derived cell lines. Exp Ther Med.
16:557–566. 2018.PubMed/NCBI
|