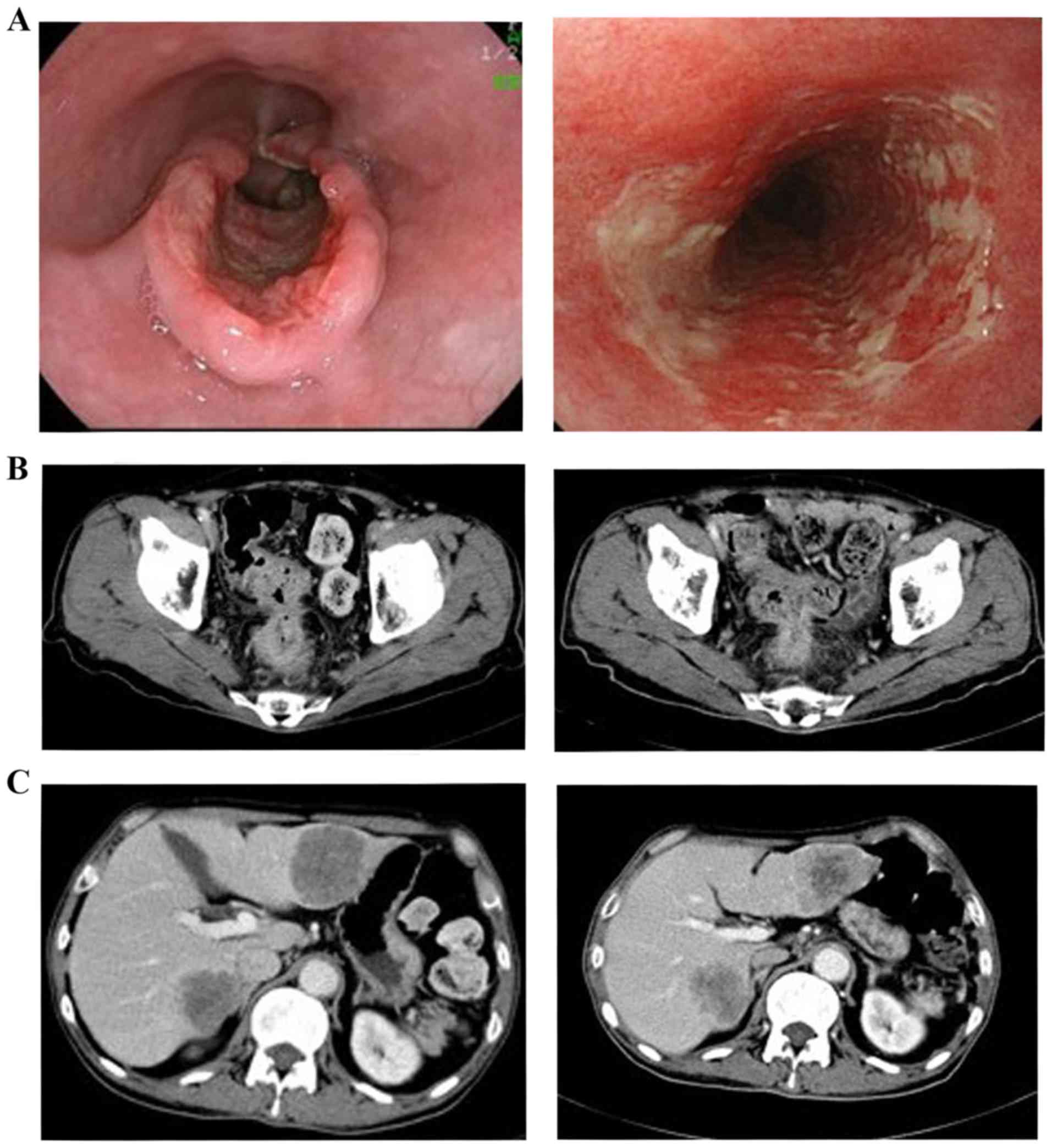
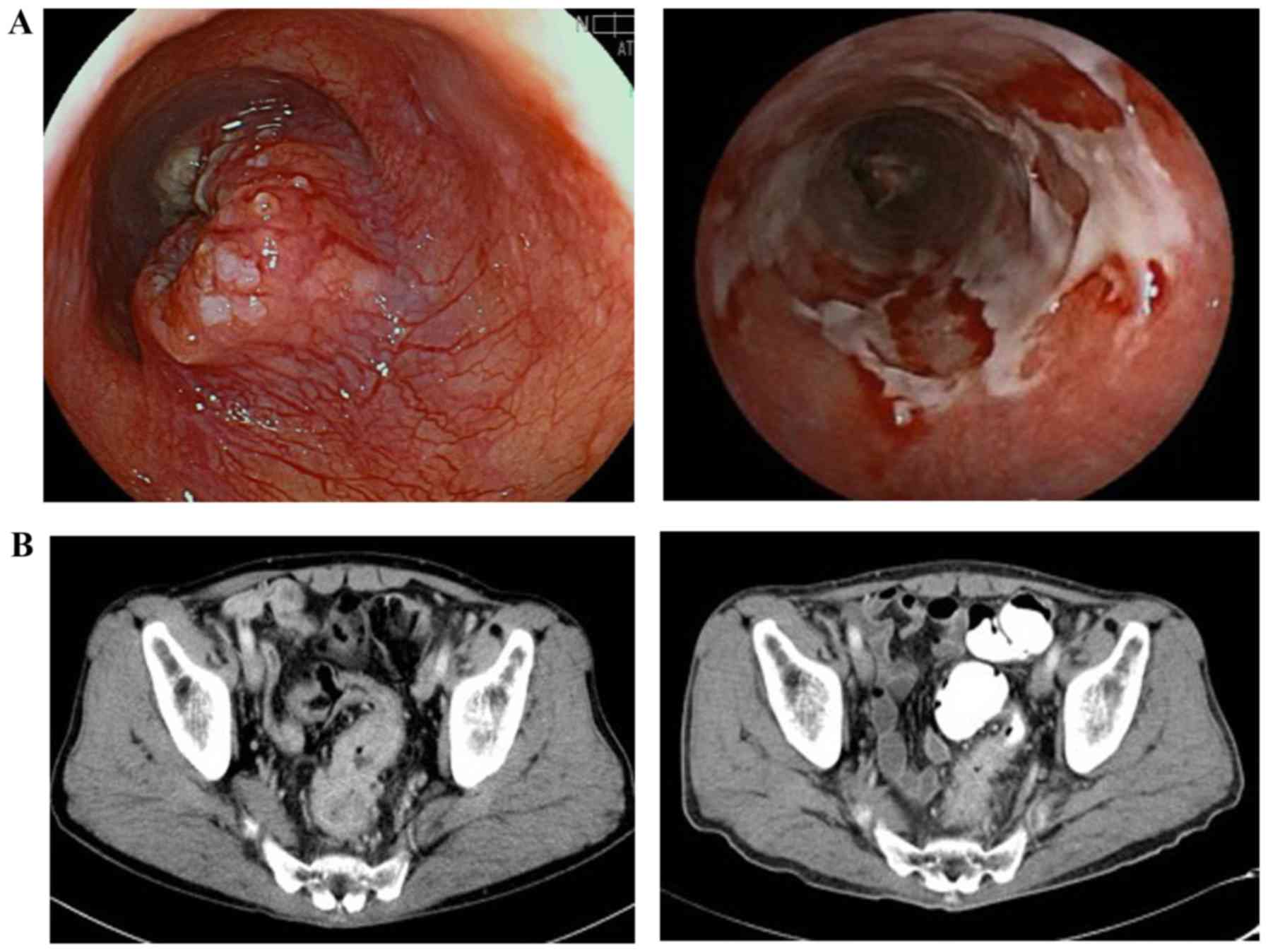
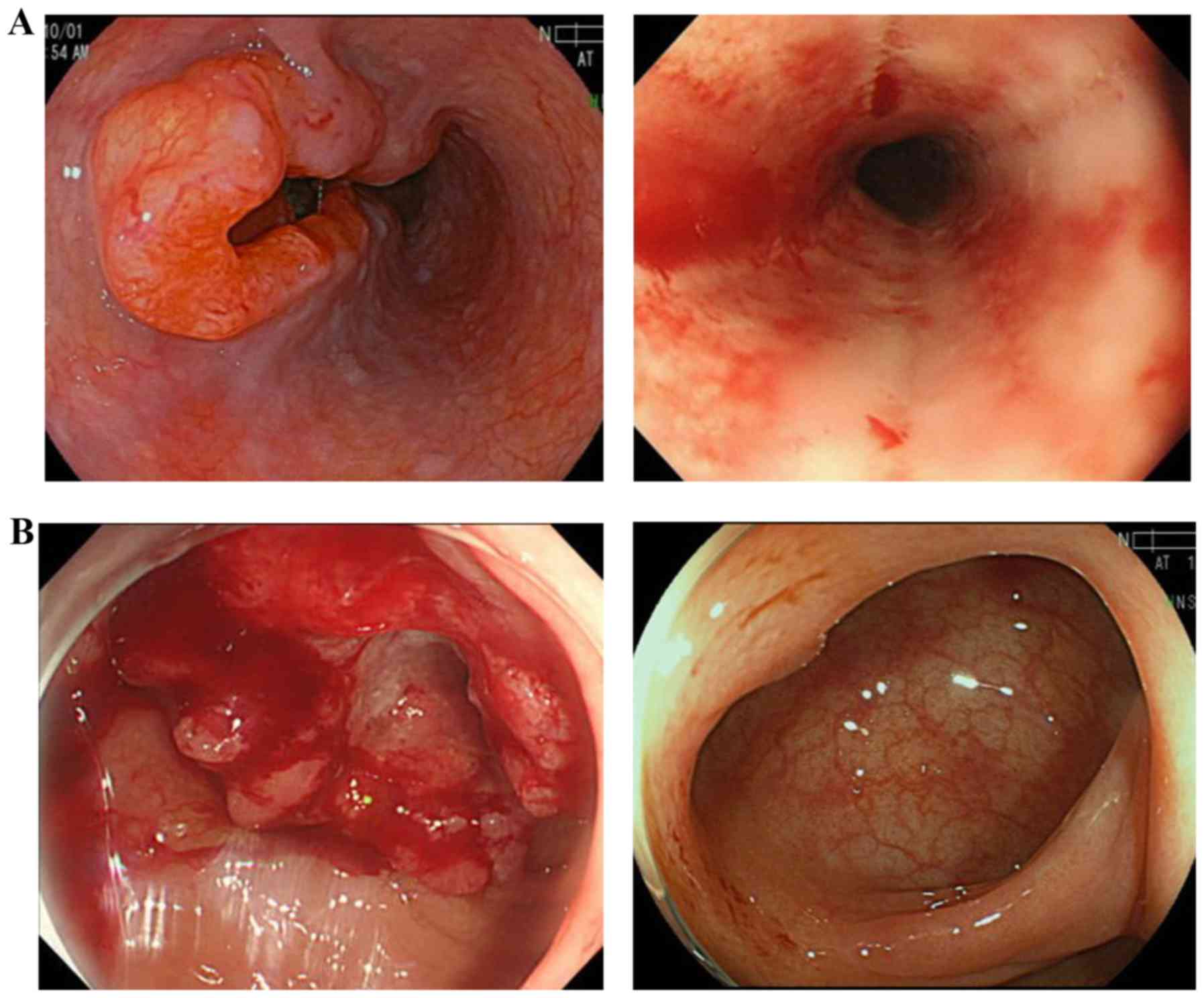
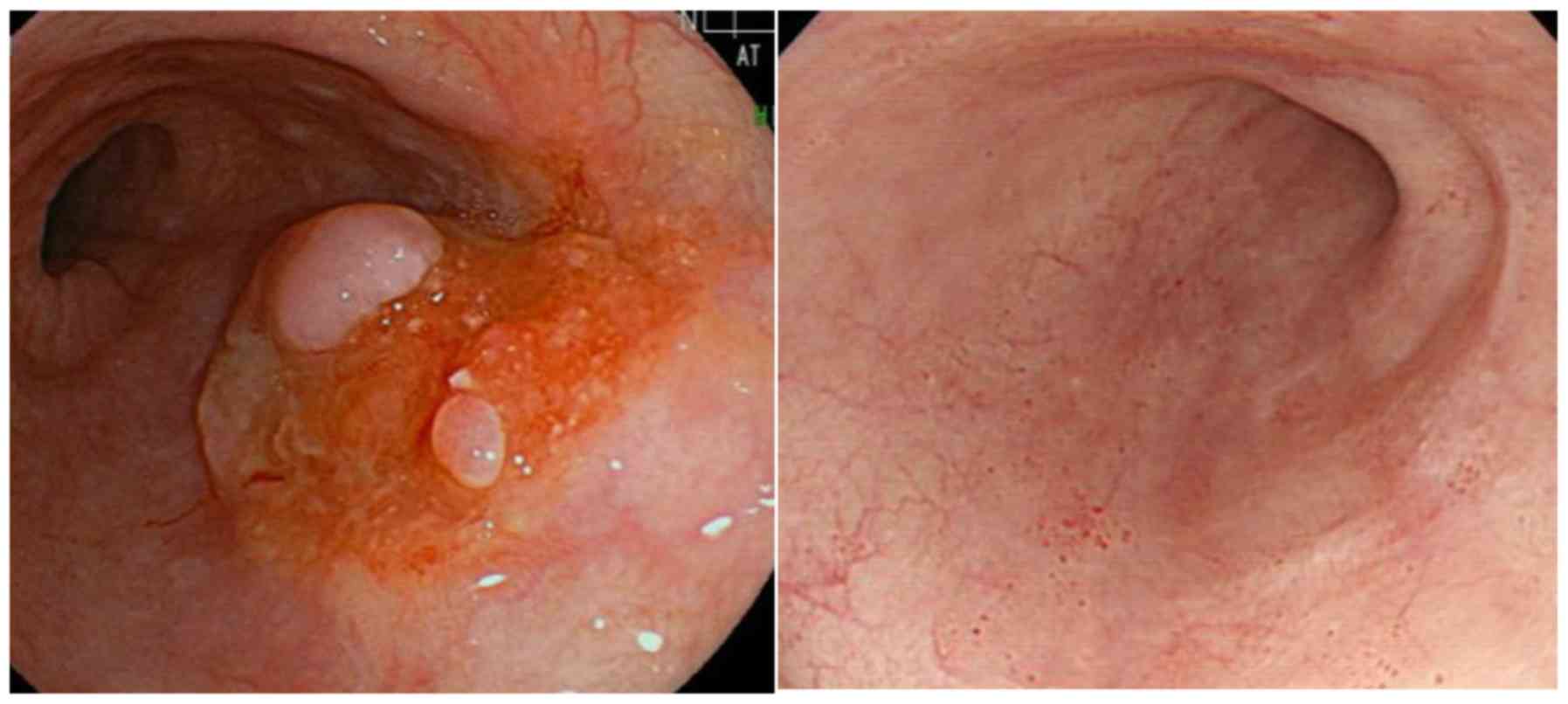
|
1
|
Global Burden of Disease Cancer
Collaboration, ; Fitzmaurice C, Dicker D, Pain A, Hamavid H,
Moradi-Lakeh M, MacIntyre MF, Allen C, Hansen G, Woodbrook R, et
al: The global burden of cancer 2013. JAMA Oncol. 1:505–527. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
National Comprehensive Cancer Network, .
Clinical practice oncology: Esophageal and esophagogastric junction
cancers (version 2. 2017).
|
|
3
|
Stahl M, Mariette C, Haustermans K,
Cervantes A and Arnold D; ESMO Guidelines Working Group, :
Oesophageal cancer: ESMO clinical practice guidelines for
diagnosis, treatment and follow-up. Ann Oncol. 24 (Suppl
6):vi51–vi56. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cooper JS, Guo MD, Herskovic A, Macdonald
JS, Martenson JA Jr, Al-Sarraf M, Byhardt R, Russell AH, Beitler
JJ, Spencer S, et al: Chemoradiotherapy of locally advanced
esophageal cancer: Long-term follow-up of a prospective randomized
trial (RTOG 85-01). Radiation therapy oncology group. JAMA.
281:1623–1627. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Minsky BD, Pajak TF, Ginsberg RJ, Pisansky
TM, Martenson J, Komaki R, Okawara G, Rosenthal SA and Kelsen DP:
INT 0123 (radiation therapy oncology group 94-05) phase III trial
of combined-modality therapy for esophageal cancer: High-dose
versus standard-dose radiation therapy. J Clin Oncol. 20:1167–1174.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
al-Sarraf M, Martz K, Herskovic A,
Leichman L, Brindle JS, Vaitkevicius VK, Cooper J, Byhardt R, Davis
L and Emami B: Progress report of combined chemoradiotherapy versus
radiotherapy alone in patients with esophageal cancer: An
intergroup study. J Clin Oncol. 15:277–284. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Conroy T, Yataghene Y, Etienne PL, Michel
P, Senellart H, Raoul JL, Mineur L, Rives M, Mirabel X, Lamezec B,
et al: Phase II randomised trial of chemoradiotherapy with FOLFOX4
or cisplatin plus fluorouracil in oesophageal cancer. Br J Cancer.
103:1349–1355. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Conroy T, Galais MP, Raoul JL, Bouche O,
Gourgou-Bourgade S, Douillard JY, Etienne PL, Boige V, Martel-Lafay
I, Michel P, et al: Definitive chemoradiotherapy with FOLFOX versus
fluorouracil and cisplatin in patients with oesophageal cancer
(PRODIGE5/ACCORD17): Final results of a randomised, phase 2/3
trial. Lancet Oncol. 15:305–314. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Messager M, Mirabel X, Tresch E, Paumier
A, Vendrely V, Dahan L, Glehen O, Vasseur F, Lacornerie T, Piessen
G, et al: Preoperative chemoradiation with paclitaxel-carboplatin
or with fluorouracil-oxaliplatin-folinic acid (FOLFOX) for
resectable esophageal and junctional cancer: The PROTECT-1402,
randomized phase 2 trial. BMC Cancer. 16:3182016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Shinoda M, Ando N, Kato K, Ishikura S,
Kato H, Tsubosa Y, Minashi K, Okabe H, Kimura Y, Kawano T, et al:
Randomized study of low-dose versus standard-dose chemoradiotherapy
for unresectable esophageal squamous cell carcinoma (JCOG0303).
Cancer Sci. 106:407–412. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kato K, Muro K, Minashi K, Ohtsu A,
Ishikura S, Boku N, Takiuchi H, Komatsu Y, Miyata Y, Fukuda H, et
al: Phase II study of chemoradiotherapy with 5-fluorouracil and
cisplatin for stage II–III esophageal squamous cell carcinoma: JCOG
trial (JCOG 9906). Int J Radiat Oncol Biol Phys. 81:684–690. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kato H, Sato A, Fukuda H, Kagami Y,
Udagawa H, Togo A, Ando N, Tanaka O, Shinoda M, Yamana H and
Ishikura S: A phase II trial of chemoradiotherapy for stage I
esophageal squamous cell carcinoma: Japan clinical oncology group
study (JCOG9708). Jpn J Clin Oncol. 39:638–643. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Japan Esophageal Society, . Japanase
classification of esophageal cancer, 11th edition: Part II and III.
Esophagus. 14:37–65. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Eisenhauera EA, Therasseb P, Bogaertsc J,
Schwartzd H, Sargente D, Ford R, Dancey J, Gwyther S, Mooney M,
Rubinstein L, et al: New response evaluation criteria in solid
tumours: Revised RECIST guideline (version 1.1). Eur J Cancer.
45:228–247. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
National Institutes of Health Common
Terminology Criteria for Adverse Events (CTCAE) Version 4.0US
Department of Health and Human Services. National Institutes of
Health, National Cancer Center, National Cancer Center; Washington
DC: 2009
|
|
16
|
de Gramont A, Figer A, Seymour M, Homerin
M, Hmissi A, Cassidy J, Boni C, Cortes-Funes H, Cervantes A, Freyer
G, et al: Leucovorin and fluorouracil with or without oxaliplatin
as first-line treatment in advanced colorectal cancer. J Clin
Oncol. 18:2938–2947. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Andre T, Boni C, Mounedji-Boudiaf L,
Navarro M, Tabernero J, Hickish T, Topham C, Zaninelli M, Clingan
P, Bridgewater J, et al: Oxaliplatin, fluorouracil, and leucovorin
as adjuvant treatment for colon cancer. N Engl J Med.
350:2343–2351. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Khushalani NI, Leichman CG, Proulx G, Nava
H, Bodnar L, Klippenstein D, Litwin A, Smith J, Nava E, Pendyala L,
et al: Oxaliplatin in combination with protracted-infusion
fluorouracil and radiation: Report of a clinical trial for patients
with esophageal cancer. J Clin Oncol. 20:2844–2850. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chiarion-Sileni V, Innocente R, Cavina R,
Ruol A, Corti L, Pigozzo J, Del Bianco P, Fumagalli U, Santoro A
and Ancona E: Multi-center phase II trial of chemo-radiotherapy
with 5-fluorouracil, leucovorin and oxaliplatin in locally advanced
esophageal cancer. Cancer Chemother Pharmacol. 63:1111–1119. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
O'Connor BM, Chadha MK, Pande A, Lombardo
JC, Nwogu CE, Nava HR, Yang G and Javle MM: Concurrent oxaliplatin,
5-fluorouracil, and radiotherapy in the treatment of locally
advanced esophageal carcinoma. Cancer J. 13:119–124. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cunningham D, Allum WH, Stenning SP,
Thompson JN, Van de Velde CJ, Nicolson M, Scarffe JH, Lofts FJ,
Falk SJ, Iveson TJ, et al: Perioperative chemotherapy versus
surgery alone for resectable gastroesophageal cancer. N Engl J Med.
355:11–20. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bang YJ, Kim YW, Yang HK, Chung HC, Park
YK, Lee KH, Lee KW, Kim YH, Noh SI, Cho JY, et al: Adjuvant
capecitabine and oxaliplatin for gastric cancer after D2
gastrectomy (CLASSIC): A phase 3 open-label, randomised controlled
trial. Lancet. 379:315–321. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yamada Y, Higuchi K, Nishikawa K, Gotoh M,
Fuse N, Sugimoto N, Nishina T, Amagai K, Chin K, Niwa Y, et al:
Phase III study comparing oxaliplatin plus S-1 with cisplatin plus
S-1 in chemotherapy-naive patients with advanced gastric cancer.
Ann Oncol. 26:141–148. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Masuishi T, Kadowaki S, Kondo M, Komori A,
Sugiyama K, Mitani S, Honda K, Narita Y, Taniguchi H, Ura T, et al:
FOLFOX as first-line therapy for gastric cancer with severe
peritoneal metastasis. Anticancer Res. 37:7037–7042.
2017.PubMed/NCBI
|
|
25
|
Watanabe Y, Tsutsui M, Takeda S, Yoshino S
and Oka M: A case of esophageal cancer associated with colon cancer
successfully treated with combination chemotherapy of FOLFOX and
concurrent radiotherapy. Gan To Kagaku Ryoho. 36:2439–2441.
2009.(In Japanese). PubMed/NCBI
|
|
26
|
Allegra CJ, Yothers G, O'Connell MJ,
Sharif S, Colangelo LH, Lopa SH, Petrelli NJ, Goldberg RM, Atkins
JN, Seay TE, et al: Initial safety report of NSABP C-08: A
randomized phase III study of modified FOLFOX6 with or without
bevacizumab for the adjuvant treatment of patients with stage II or
III colon cancer. J Clin Oncol. 27:3385–3390. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mitani S, Kadowaki S, Kato K, Masuishi T
and Muro K: Combination of oxaliplatin and
5-fluorouracil/leucovorin for advanced esophageal squamous cell
carcinoma refractory or intolerant to standard therapies. Case Rep
Oncol. 12:304–310. 2019. View Article : Google Scholar : PubMed/NCBI
|