Introduction
Gastric cancer (GC) is the third most common cause
of cancer-related deaths worldwide (1). Differentiated-type early-stage GC,
with a low risk of lymph node metastasis, can be successfully
treated using local endoscopic procedures, such as endoscopic
submucosal dissection (ESD) (2) or
PDT (3). ESD has already been
suggested as a treatment for intramucosal and superficial
submucosal GC, whereas PDT is the preferred strategy to treat
submucosal GC without lymph (and other distant) metastases.
Alternatively, PDT can be used as a palliative therapy for advanced
and localized GC in order to reduce the tumor burden (4).
PDT elicits localized cell death in tumors, using a
combination of light, a photosensitizer, and tissue oxygen
(5). In theory, because
photosensitizers specifically accumulate in tumors, there should
not be any off-target effects of PDT, allowing for the selective
treatment of tumor lesions. When a photosensitizer is irradiated
with light of a specific wavelength, it is excited from a ground
state (S0) to an S1 state. Subsequently, due to intersystem
crossing, the photosensitizer reaches a triplet state (S3) then
returns to S0 while transferring energy to oxygen. The oxygen
molecule that receives the energy subsequently reaches S1 (singlet
oxygen), thus destroying cancer cells (5).
PDT has several advantages over more invasive
procedures such as ESD, including a reduced risk of bleeding, and
can be safely performed in conjunction with the administration of
antiplatelet medication (6). In
Japan, the first-generation photosensitizer (porfimer sodium, PS)
and second-generation photosensitizers [talaporfin sodium (TS) and
verteporfin (VP)] have already obtained regulatory approval for use
with PDT. Using PDT to treat early-stage GC is approved only when
combining PS with excimer dye laser (EDL) or Yttrium Aluminum
Garnet (YAG) optical parameter oscillator (OPO) lasers (YAG-OPO:
Ishikawajima-Harima Heavy Industries Co., Ltd.: IHI). However,
because EDL and YAG OPO devices are very expensive, their
production has been stopped (3).
Furthermore, in order to avoid sunlight sensitivity after
treatment, a light-blocking period of four weeks or more is
required with this approach. Even the second-generation
photosensitizer TS requires a continuous light-blocking period of
at least two weeks. By contrast, VP-based PDT requires a
light-blocking period of no more than two days, which is
considerably shorter than the clinically available
photosensitizers. As the effectiveness and safety of VP are well
established, it is widely used in PDT to treat age-related macular
degeneration (7,8). Furthermore, the absorption peak of VP
is 689 nm, whereas the absorption peaks of PS and TS are 630 and
664 nm, respectively. The longer wavelength light (689 nm)
penetrates deeper and can pass through the submucosal layer into
the mucosal lamina propria, resulting in effectiveness even for
invasive GC. In 2014, a phase I/II study to treat locally advanced
pancreatic cancer with VP-PDT was conducted, and the safety of the
approach was confirmed (8). The
notable efficacy of VP-PDT, in combination with paclitaxel, on GC
cells (NCI-N87 derived from liver metastases) was demonstrated
previously (9); however, the
particular effect of VP-PDT on GC cells has yet to be adequately
elucidated.
The aim of the present study was to evaluate the
effect of VP-PDT on two different superficial submucosal GC cell
lines, MKN45 and MKN74, which were derived from undifferentiated
and well-differentiated adenocarcinoma, respectively. The two cell
types were exposed individually or in combination to VP-PDT. VP-PDT
was effective on these cells using a combination of light and a
photosensitizer. Through this study, we pave the way for future
clinical applications of VP-PDT for GC (with nominal risks of lymph
node and distant metastasis), such as intramucosal GC and GC with
superficial invasion into the muscular propriae (common in aged
individuals).
Materials and methods
Cell lines
Human gastric cancer cell lines and cultures,
MKN45-Luc and MKN74/CMV-Luc cells, were obtained from the JCRB cell
bank. Cells were cultured in a humidified incubator (5%
CO2, 37˚C) in RPMI-1640 medium without antibiotics,
supplemented with 10% fetal bovine serum and 1% L-glutamine
solution. Immortalized human pancreatic duct epithelial cell
(T0005; Applied Biological Materials Inc.) and was cultured in
Prigrow I medium and D-MEM/Ham's F-12 medium, respectively. Both
media were supplemented with 20% fetal bovine serum without
antibiotics. A normal rat gastric epithelial cell line; RGM-1 (a
gift from Dr Matsui; University of Tsukuba) (10) was cultured in D-MEM/Ham's F-12
medium supplemented with 10% fetal bovine serum without
antibiotics.
Reagents
VP (SML0534) was purchased from Merck KGaA. For
microscopy, MitoBright Green (MT06) and Hoechst-33342 solution
(cat. no. 346-07951) were purchased from Dojindo Laboratories, Co.,
Ltd. Singlet Oxygen Sensor Green (cat. no. S36002) was purchased
from Thermo Fisher Scientific, Inc.
Microscopic imaging
Cells were visualized with a fluorescence microscope
(BZ-X710; Keyence Co.), using BZ-X filter GFP (OP-87763; Keyence
Co.) and BZ-X filter DAPI (OP-87762; Keyence Co.). In addition, to
visualize VP, a filter cube (OP-87767; Keyence Co.) was used with
relevant excitation (405BP20) and fluorescence (RPE630LP) filters.
Magnification of the objective lens was x10. For merging, noise
reduction and signal intensity enhancement, BZ-analyzer (Keyence
Co.) was used.
Photodynamic therapy protocol and
proliferation assay
Cells were exposed to VP in serum-free medium,
avoiding sunlight and room lights. VP treatment times were from 15
min to 1 h. The cultures were irradiated with 660 nm light
(LEDR-660DL; Optocode Co., Ltd.) at 2.5 J/cm2 (11), and after 24 h. Cell viability was
measured using the MTS
(3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium)
assay (12). The MTS assay was
performed by adding 20 µl proliferation assay solution (G3580,
CellTiter 96® Aqueous One Solution Cell Proliferation
Assay; Promega Co.) to 100 µl culture medium, and after 1 h, the
absorbance at 490 nm was measured with a microplate reader
(Vientonano; DS Pharma Biochemical Co., Ltd.) and the viability of
the treated cells was compared to that of the untreated control
cells.
Fluorescent staining of intracellular
organelles
Cells were incubated with 0.1 µM VP for 1 h at 37˚C,
with avoiding sunlight and room lights, were washed twice with
phosphate-buffered saline (PBS), and exposed to 0.1 µM MitoBright
Green for 10 min at 20-25˚C. The degree of staining was dependent
on the membrane potential of the mitochondria, as the compound
accumulated specifically in normal mitochondria after cell membrane
permeation. Cells were washed again with PBS and imaged using a
fluorescence microscope.
Singlet oxygen staining
Cells were incubated with 0.1 µM VP (1 h at 37˚C
with avoiding sunlight and room lights), washed twice with PBS, and
exposed to 1 µM Singlet Oxygen Sensor Green. This reagent emits a
green fluorescent signal in the presence of S1 oxygen. Irradiation
was performed at a wavelength of 660 nm at 2.5 J/cm2 and
after 2 h, the cells were visualized using a fluorescent
microscope.
Mitochondrial ATP analysis
Necrosis and mitochondrial ATP were evaluated using
the Mitochondrial ToxGlo™ Assay (G8000; Promega Co.). Briefly, 20
µl cytotoxicity reagent was added to 100 µl medium including cells,
the cells were incubated for 30 min at 37˚C, and the fluorescence
was measured at 485 nmEx/535 nmEm.
Subsequently, 100 µl ATP detection reagent was added and the cells
were incubated for 5 min at 20-25˚C. Fluorescence and luminescence
were measured by multi detection mode plate reader (Infinite F500;
Tecan Japan Co., Ltd.).
Detection of apoptosis
To detect apoptosis, Hoechst staining was performed.
Cells were incubated with 0.5 µM VP for 1 h at 37˚C, with avoiding
sunlight and room lights. Subsequently, the cells were irradiated
with LED with the power shown in each figure. After LED
irradiation, the cells were incubated for 12 h at 37˚C, washed, and
incubated with 1 µg/ml Hoechst-33342 solution for 15 min at
20-25˚C, with avoiding sunlight and room lights. The cells were
then visualized using a fluorescence microscope.
Statistical analysis
All experiments were repeated at least 3 times. The
Kolmogorov-Smirnov test was used to assess normal distribution.
Differences between groups were analyzed using one-way analysis of
variance test for normally distributed variables with post hoc
Dunnet's test. The Kruskal-Wallis test with post hoc Dunn's test
was used for non-normally distributed variables. Data are expressed
as mean ± standard error and differences were considered
statistically significant at P<0.05.
Results
Induction of singlet oxygen and
apoptosis
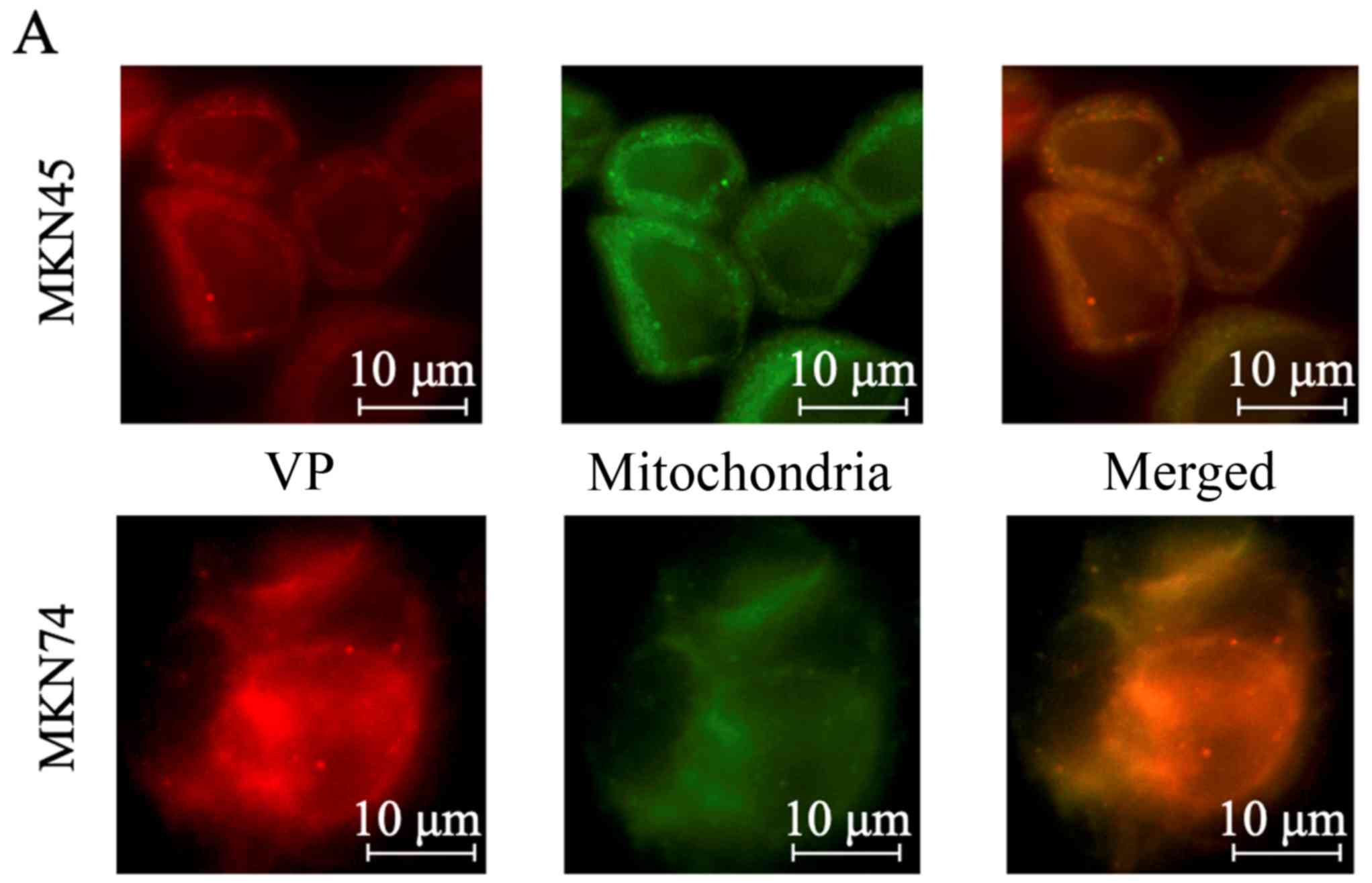
VP was localized to the mitochondria after uptake
into GC cells (Fig. 1A) and
irradiation generated S1 oxygen molecules (Fig. 1B). Furthermore, chromatin
condensation and fragmentation, which are characteristic of
apoptosis, were observed in GC cells (Fig. 1C), whereas the control cells
(without VP-PDT) did not show apoptotic cell death after
irradiation.
VP-PDT cytotoxicity on MKN45 and MKN74
cells
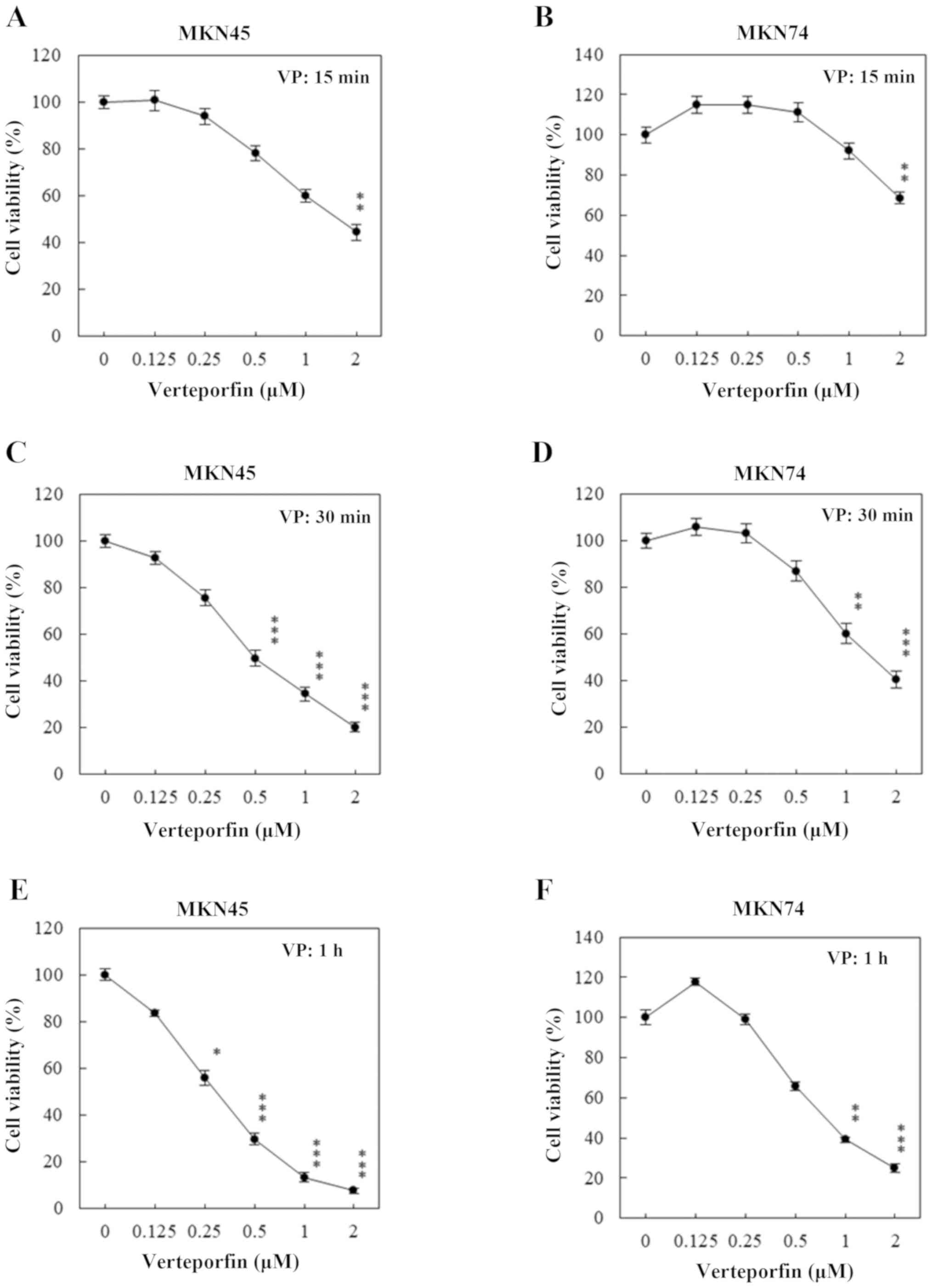
As the results of the proliferation assay reveal,
the effect of VP-PDT (2.5 J/cm2) on MKN45 and MKN74
cells was already apparent after 15 min of VP treatment (Fig. 2A and B). Treatment for 30 min and 1 h increased
the effectiveness of the treatment (Fig. 2C-F, respectively). The
EC50 values for VP are shown in Table I. Cell viability did not reach below
50% after 15 min of treatment (Fig.
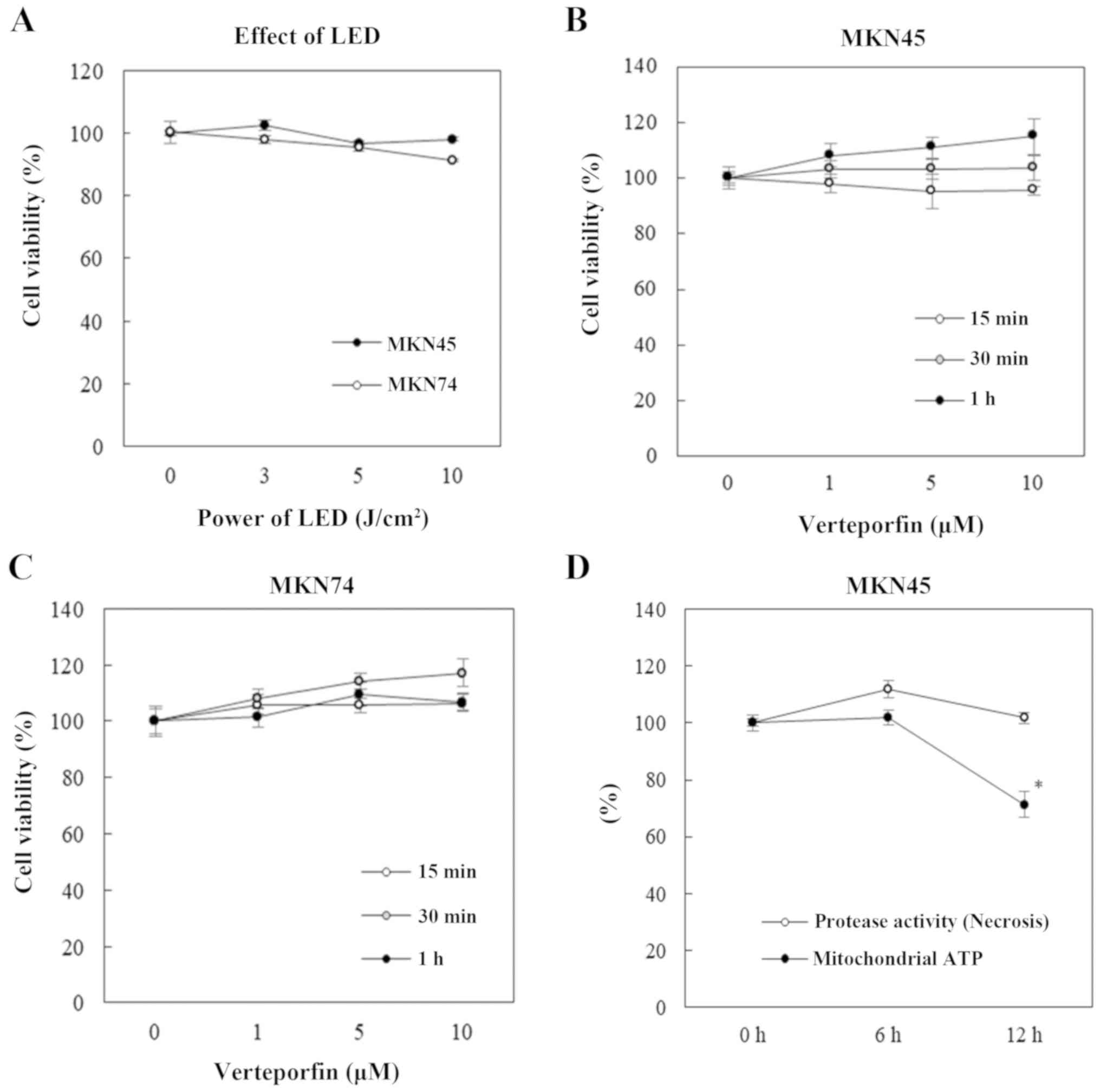
2B). Furthermore, cell damage was not observed with LED
irradiation (3, 5 and 10 J/cm2; Fig. 3A) or VP treatment (1, 5 and 10 µM;
Fig. 3B and C) alone. VP-PDT reduced mitochondrial ATP,
but not protease activity, indicating that VP-PDT exertded the
effect through apoptosis (Fig.
3D).
 | Table IEC50 values of VP. |
Table I
EC50 values of VP.
| | EC50 of VP
(µM) |
|---|
| Treatment time
(min) | MKN45 | MKN74 |
|---|
| 30 | 0.61 | 1.21 |
| 60 | 0.32 | 0.80 |
Discussion
In the present study, VP-PDT induced apoptosis via
the production of S1 oxygen molecules, reactive oxygen species, and
by mitochondrial damage, as previously reported (13). Furthermore, although VP has been
reported to inhibit yes-associated protein (YAP)-transcriptional
enhanced associate domain (TEAD) and human retinoblastoma cells
growth in vitro without light activation (14), VP alone did not show an antitumor
effect in this study. As VP alone showed YAP inhibition at higher
concentrations and longer times (data not shown), it is considered
that only mitochondrial damage via reactive oxygen species was
induced in this condition, not the effect of VP alone. Furthermore,
comparing our study results with those of clinical (14) or in vitro (15) studies, the VP concentration used in
PDT was approximately 1/10 of that when used alone as a YAP-TEAD
inhibitor. Therefore, VP-PDT is expected to have fewer side effects
such as photosensitivity and less burden on patients. The
possibility that VP-PDT, which is approved only for age-related
macular degeneration, can be extended to GC treatment has also been
shown (7). In addition, since VP is
effective at lower concentration than TS concentration for local
failure after chemoradiotherapy for esophageal cancer, high safety
is expected in the treatment of GC.
As such, we demonstrated that VP-PDT is effective
for inhibition of proliferation on MKN45 and MKN74 GC cell lines.
Of note, it has been reported that the intracellular uptake of VP
is related to the LDL receptor (16). In a previous study, we reported that
the LDL receptor has a role in the mode-of-action of TS-PDT. More
specifically, the expression of the LDL receptor was different
between MKN45 and MKN74 GC cells; the latter demonstrated a lower
expression of the LDL receptor, and was thus resistant to TS-PDT
(17). However, MKN74 cells showed
no resistance to VP-PDT. A plausible reason for this difference may
be the involvement of non-LDL receptors in the uptake of VP.
Alternatively, the uptake of very small amounts of VP via the
low-abundance LDL receptor may explain the effects that we
observed. Nonetheless, testing this hypothesis was beyond the scope
of the current study, which focused on evaluating the efficacy of
VP-PDT in an in vitro model for its potential future
clinical applications. In addition, because a non-liposomal reagent
was used in this study, we expect that a liposomal VP (Visudyne;
Novartis Pharma, Co.) would show stronger anti-tumor effects on GC
cells with a low expression of the LDL receptors. More
specifically, here the uptake might be primarily via endocytosis,
rather than via the LDL receptor.
Human pancreatic duct epithelial cells and normal
rat gastric epithelial cells were used as control cells. However,
the effect of VP-PDT on these cells was similar to that on MKN45
and MKN74 cells. Additionally, the EC50 value of VP-PDT
(690 nm and 10 J/cm2) for human vascular smooth cell was
0.01785 µM (18). The power of LED
of that study was reduced. Therefore, we presume that the results
are similar between the current study and that study. However, the
control cells we used in the present study were modified for cell
culture; thus, the results of our data may not be directly applied
to healthy body. This is a limitation of this study using these
control cells.
The EC50 calculated for VP-PDT in this
study was approximately 2/3th to 1/6th of the maximun blood
concentration (1.84 µM) used to treat aging macular degeneration
(Visudyne). This EC50 was lower than that of TS-PDT, as
previously reported (17). In
detail, the EC50 of TS-PDT was over 20 µM and treatment
time of TS required at least 4 h. Furthermore, VP-PDT was effective
on MKN74 cells that demonstrated resistance against TS-PDT
(17). Additionally, the
EC50 of VP in this study was approximately identical to
the effective VP-PDT concentration that was used against
gemcitabine-resistant pancreatic cancer cells (13). Clinical trials using VP-PDT to treat
pancreatic cancer have already been conducted, and the efficacy of
the method has already been demonstrated (8). As such, we predict that the same dose
of VP-PDT could be used safely against GC in the clinical setting.
However, we used LED light with a wavelength of 660 nm, which is
covered by the broad absorption band of VP; it has been reported
that the peak absorption coefficient of VP is 689 nm (19). Therefore, a more pronounced effect
of VP-PDT on GC cell lines is expected when LEDs with a wavelength
closer to 689 nm are used. However, we used 660 nm LED light
because of lack of necessary equipment. Further research currently
in progress has focused on the development of an experimental LED
device, capable of illumination at 689 nm, for use in combination
with VP, which may lead to further improvements with regard to the
depth of invasion. It is also necessary to consider the optimal LED
wavelength. Furthermore, it has not been confirmed whether similar
effects can be obtained in animals and humans.
Limitations of this study should also be considered.
First, although the peak absorption coefficient of VP was 689 nm,
use of LED at 689 nm for irradiation could not be carried out owing
to absence of equipment. However, we observed that irradiation with
LED at even 660 nm inhibited GC cell proliferation. Second, we
revealed inhibition of proliferation only in vitro.
Therefore, in future, detailed clinical or in vivo
experiments should be performed to support the arguments presented
in this study. Furthermore, the cell lines used in the evaluation
of VP-PDT on normal cells are derived from different organs or
different species, constituting another limitation of this
study.
In conclusion, we have demonstrated that VP-PDT is
an effective strategy for inhibition of proliferation on GC cells,
and anticipate that in the future, VP will be implemented in the
clinical treatment of GC with PDT.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
YM and TK acquired data, analyzed and interpreted
the data, and drafted the manuscript. TSu and TT also analyzed and
interpreted the data. HKi, TSa, TH, RT, ME and HKu assisted in
acquiring the data. YI and KK made contributions to the conception
and design of the study. HI made substantial contributions to the
conception and design of the study and drafted the manuscript. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2017. CA Cancer J Clin. 67:7–30. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Isomoto H, Shikuwa S, Yamaguchi N, Fukuda
E, Ikeda K, Nishiyama H, Ohnita K, Mizuta Y, Shiozawa J and Kohno
S: Endoscopic submucosal dissection for early gastric cancer: A
large-scale feasibility study. Gut. 58:331–336. 2009.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Oinuma T, Nakamura T and Nishiwaki Y:
Report on the national survey of photodynamic therapy (PDT) for
gastric cancer in Japan (a secondary publication). Laser Ther.
25:87–98. 2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Nakamura T and Oinuma T: Usefulness of
photodynamic diagnosis and therapy using talaporfin sodium for an
advanced-aged patient with inoperable gastric cancer (a secondary
publication). Laser Ther. 23:201–210. 2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Ethirajan M, Chen Y, Joshi P and Pandey
RK: The role of porphyrin chemistry in tumor imaging and
photodynamic therapy. Chem Soc Rev. 40:340–362. 2011.PubMed/NCBI View
Article : Google Scholar
|
|
6
|
Brown SB and Melish KJ: Verteporfin: A
milestone in opthalmology and photodynamic therapy. Expert Opin
Pharmacother. 2:351–361. 2001.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Messmer KJ and Abel SR: Verteporfin for
age-related macular degeneration. Ann Pharmacother. 35:1593–1598.
2001.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Huggett MT, Jermyn M, Gillams A, Illing R,
Mosse S, Novelli M, Kent E, Bown SG, Hasan T, Pogue BW and Pereira
SP: Phase I/II study of verteporfin photodynamic therapy in locally
advanced pancreatic cancer. Br J Cancer. 110:1698–1704.
2014.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Park S, Hong SP, Oh TY, Bang S, Chung JB
and Song SY: Paclitaxel augments cytotoxic effect of photodynamic
therapy using verteporfin in gastric and bile duct cancer cells.
Photochem Photobiol Sci. 7:769–74. 2008.PubMed/NCBI View
Article : Google Scholar
|
|
10
|
Shimokawa O, Matsui H, Nagano Y, Kaneko T,
Shibahara T, Nakahara A, Hyodo I, Yanaka A, Majima HJ, Nakamura Y
and Matsuzaki Y: Neoplastic transformation and induction of H+,K+
-adenosine triphosphatase by N-methyl-N'-nitro-N-nitrosoguanidine
in the gastric epithelial RGM-1 cell line. In Vitro Cell Dev Biol
Anim. 44:26–30. 2008.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Nanashima A, Isomoto H, Abo T, Nonaka T,
Morisaki T, Arai J, Takagi K, Ohnita K, Shoji H, Urabe S, et al:
How to access photodynamic therapy for bile duct carcinoma. Ann
Transl Med. 2(23)2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Yang MY, Chang CJ and Chen LY: Blue light
induced reactive oxygen species from flavin mononucleotide and
flavin adenine dinucleotide on lethality of HeLa cells. J Photochem
Photobiol. 173:325–332. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Celli JP, Solban N, Liang A, Pereira SP
and Hasan T: Verteporfin-based photodynamic therapy overcomes
gemcitabine insensitivity in a panel of pancreatic cancer cell
lines. Lasers Surg Med. 43:565–574. 2011.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Katarzyna B, Moujahed A, Marmalidou A,
Horste MM zu, Cichy J, Miller JW, Gragoudas E and Vavvas DG: The
clinically used photosensitizer verteporfin (VP) inhibits YAP-TEAD
and human retinoblastoma cell growth in vitro without light
activation. Exp Eye Res. 124:67–73. 2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kang MH, Jeong GS, Smoot DT, Ashktorab H,
Hwang CM, Kim BS, Kim HS and Park YY: Verteporfin inhibits gastric
cancer cell growth by suppressing adhesion molecule FAT1.
Oncotarget. 8:98887–98897. 2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Newman DK: Photodynamic therapy: Current
role in the treatment of chorioretinal conditions. Eye. 30:202–210.
2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kanda T, Sugihara T, Takata T, Mae Y,
Kinoshita H, Sakaguchi T, Hasegawa T, Kurumi H, Ikebuchi Y,
Murakami T and Isomoto H: Low-density lipoprotein receptor
expression is involved in the beneficial effect of photodynamic
therapy using talaporfin sodium on gastric cancer cells. Oncol
Lett. 17:3261–3266. 2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Labow RS, Higginson LA, Irvine J, Keaney
M, Masters RG, Marquis JF, Meek E, Mussivand T, Walley VM, Logan P,
et al: Assessment of the cytotoxicity of the photosensitizing drug
BPD verteporfin using human vascular smooth muscle cells in
culture. J Cardiovasc Pharmacol. 26:729–736. 1995.PubMed/NCBI
|
|
19
|
Houle JM and Strong HA: Clinical
pharmacokinetics of verteporfin. J Clin Pharmacol. 42:547–557.
2002.PubMed/NCBI View Article : Google Scholar
|