Introduction
Pancreatic cancer usually metastasizes at the liver
and the peritoneal cavity. The spine represents an unusual site of
pancreatic metastatic disease, while the exact incidence of this
metastasis remains unclear (1).
These lesions most times are osteolytic. However, lesions of
osteoblastic nature, though rare, have also been reported (2). Usually, spine metastases occur in
patients with advanced disease, long after the initial diagnosis
(2).
In this report a patient with locally limited
pancreatic cancer and initial complain only upper back pain due to
a solitary bone blastic metastatic lesion of the fourth thoracic
vertebra (T4) is presented.
Case report
A 54-year-old male presented to the outpatient
clinic, complaining for severe upper back pain during the last 4
months. The pain was worse at night, causing severe sleep
disturbance, and did not respond to ‘over the counter’ painkillers.
He was stable (blood pressure 120/75 mmHg, heart rate 85 beats/min,
SpO2 99%) and afebrile (36.6˚C). His medical history
included heavy smoking, as well as severe anxiety and panic attacks
treated with perphenazine/amitriptyline.
Clinical examination was unremarkable except for
tenderness at palpation of the upper thoracic spine. There was no
neurological deficit. Laboratory findings included: Hemoglobin=12.7
g/dl, normal white blood cell (WBC=5.400) and platelet count
(PLT=210 k/µl). Hepatic (SCOT=25 IU/l, SGPT=30 IU/l, gGT=76 IU/l
and ALP=95/IU/l) and renal (Cr 1.06 mg/dl, UR=28 mg/dl) functions
were normal. C-reactive protein (CRP=0.43 mg/dl) and prostatic
specific antigen (PSA=2.96 ng/ml) were within normal range.
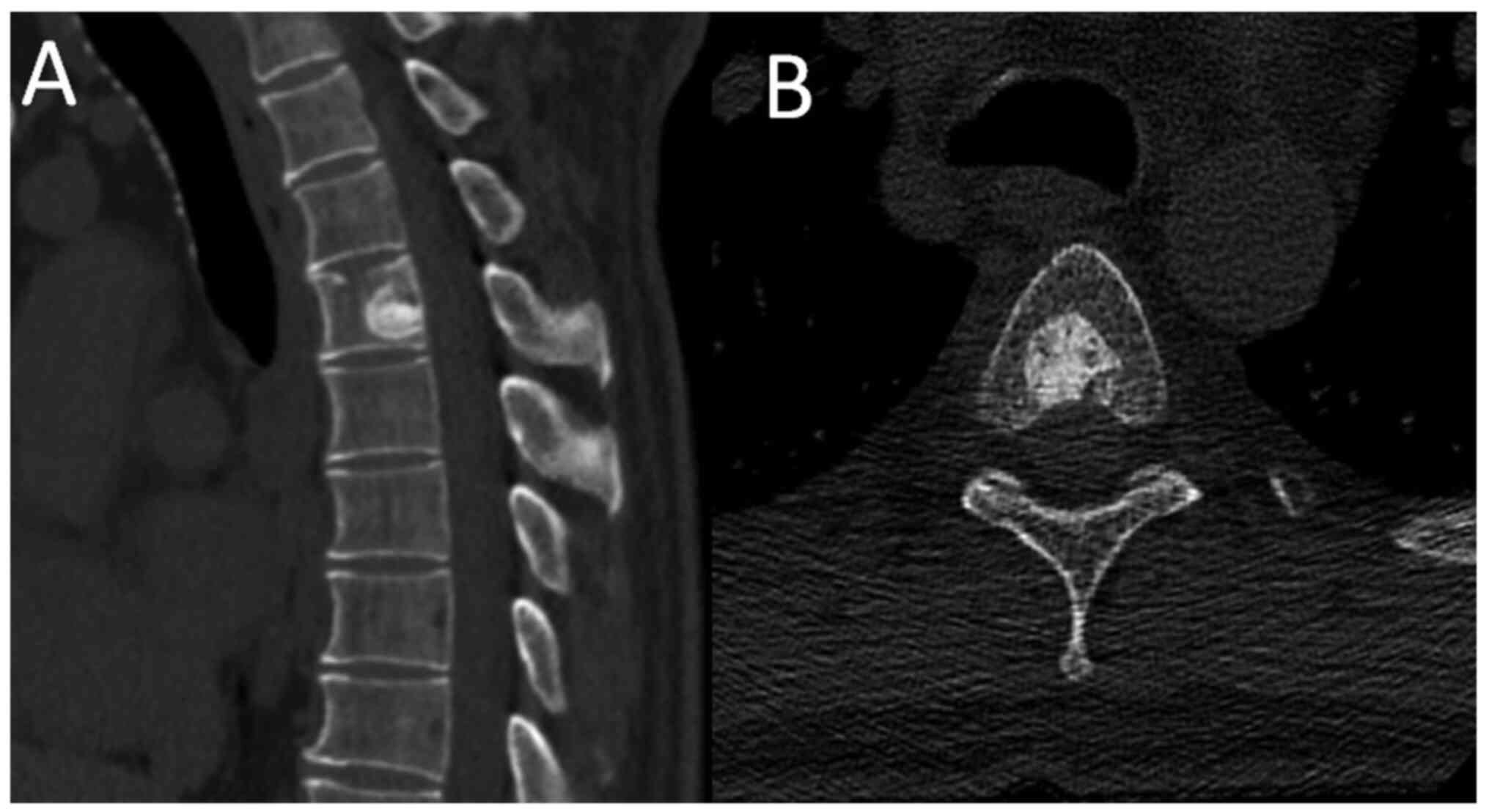
CT scan of the thoracic spine revealed a rounded,
mostly sclerotic bone lesion, with smooth transitional margins and
small inner lucent areas at the T4 vertebral body (Fig. 1). Thus, it was diagnosed by imaging
specialists as a benign lesion unrelated to his symptoms.
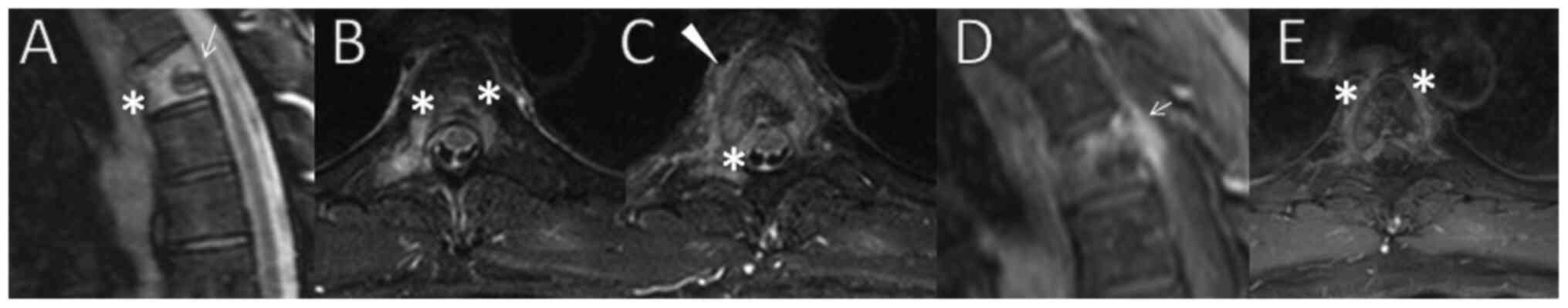
Three months later, due to persistence of the pain,
he underwent a thoracic spine MRI scan, revealing bone marrow edema
of part of the T4 vertebral body and presence of paraspinal and
epidural soft tissue mass, along with the already known sclerotic
lesion (Fig. 2). Since these
findings were highly indicative of malignant nature, bone
scintigraphy followed which was, surprisingly, normal.
The patient was further investigated with CTs of the
thorax and abdomen and a mass infiltrating part of the body and tail
of the pancreas indicating pancreatic tumor was revealed.
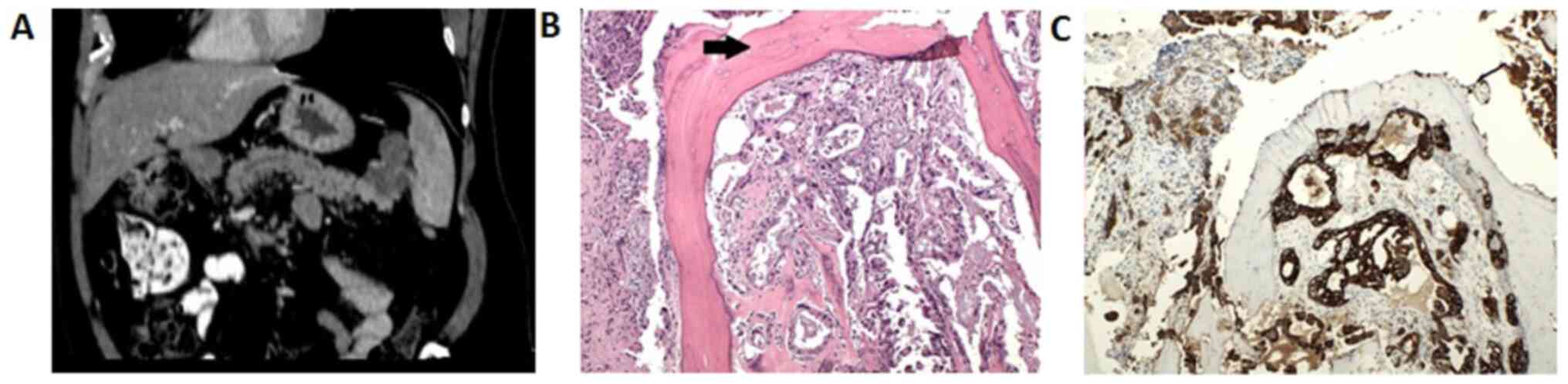
Consequently a C-arm guided biopsy of the T-4 vertebra revealed
histology compatible with pancreatic bone metastasis. In
particular, histology demonstrated tumor cells in cavities
in-between regenerated bone, consistent with a poor differentiated
adenocarcinoma with features indicating pancreatic cancer (Fig. 3).
Hence the diagnosis of pancreatic cancer was
confirmed and the patient was referred to the Department of Medical
Oncology for antineoplastic treatment. It is of note that, up to
that point in time, the patient remained in good condition without
weight loss or other usual signs and symptoms related to pancreatic
cancer.
Discussion
Pancreatic cancer, at early stages, is often
difficult to be diagnosed due to the absence of specific signs and
symptoms; therefore, at the time of diagnosis most patients present
with advanced disease (3).
Cardinal symptoms and signs include pain located at
the upper abdomen radiating at the back, malaise, weight loss
and/or jaundice. Less often patients present with recurrent
thrombophlebitis and in rare cases with mental and/or personality
disorders (4). Most symptoms are
related to the location of the primary tumor. Pancreatic body or
tail malignancies commonly appear with pain and weight loss, while
tumors of the pancreatic head usually present with obstructive
jaundice (4). However, the present
patient did not have any of these usual symptoms, with the thoracic
spinal pain being the only one.
Common metastatic sites include the regional lymph
nodes, the liver and less commonly the lungs. Osseous metastases
are unusual, while spinal metastatic lesions, as initial
presentation, are extremely rare (3). The exact prevalence of bone metastases
of this tumor remains unclear. Generally, it is quite rare for the
disease to spread outside the abdomen, in the absence of liver
disease. Hess et al reported on their tumor registry data
from 4,399 patients. In the group of pancreatic adenocarcinoma
patients, 85% of metastases occurred in the liver, 12% in the lung
and 3% in the skeleton (5). Borad
et al, in a retrospective study including 323 patients with
pancreatic cancer, reported 7 patients (2.2%) with skeletal disease
with all of them having spinal involvement (6). Iguchi et al reported 13
patients (7.3%) with secondary bone lesions in a retrospective
study of 178 patients (7). In the
same study, the authors underline that serum levels of the bone
resorption marker 1CTP, as well as IL-6 and VEGF were elevated in
most cases, while elevation of serum PTHrP levels was found in 3
out of 13 patients. Hence, the authors suggest periodical
measurement of serum 1CTP in addition to bone scintigraphy for
earlier diagnosis of metastatic bone disease (7).
Bone metastases are mainly lytic. Purely blastic
lesions have rarely been reported (2). Ray et al published a case of a
solitary osteblastic bone metastasis, in the absence of other
systemic disease, occurring 7 years after surgical resection and
adjuvant treatment for a locally advanced pancreatic adenocarcinoma
(8). The patient had good
performance status, with no clinical evidence of recurrence, until
an elevation of the CA 19-9 tumor marker initiated a systemic
workup. Therefore, an isolated sclerotic lesion in the right sacral
ala was identified on imaging and was biopsied, histologically
revealing adenocarcinoma, consistent with the primary tumor.
Spinal pain as presenting complaint of metastatic
pancreatic cancer is rare, with only few cases in the literature
reporting painful vertebral metastatic lesions as first clinical
manifestation (9). Pneumaticos
et al reported a case of cancer of the body of the pancreas
presented with severe back pain due to an osteoblastic lesion to
third lumbar vertebra (10). The
present patient suffered severe back pain and he had had a CT scan
of the upper thoracic spine. A sclerotic lesion had been found,
which was misdiagnosed as benign, irrelevant to the patient's
symptoms.
Although there are many reliable indicators in the
radiological evaluation of a potential osteolytic bone tumor (zone
of transition, periosteal reaction, cortical destruction), further
investigation should be recommended in cases of atypical
osteoblastic lesions, due to lack of similar helpful signs in such
lesions (with the exception of a pathognomonic sclerotic bone
tumor). Certainly, other clinical symptoms should be sought in the
presence of such spinal lesions.
Spinal metastases are rare in pancreatic cancer but
they can cause significant morbidity such as excruciating pain and
neurological dysfunction, even paralysis, due to neural tissue
compression (11). Although the
overall prognosis of metastatic pancreatic cancer is very poor,
early diagnosis and management of spinal metastases is crucial for
the patients' quality of life (10).
In conclusion, pancreatic cancer is not usually
included in the differential diagnosis of a bone metastasis of
unknown origin. Clinicians should increase their awareness and
index of suspicion regarding the types of the disease's
presentation and to include pancreatic cancer in the investigation
of bone metastases of unknown primary tumor.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published report.
Authors' contributions
KA, CK, GS and KR made substantial contribution to
the conception and design of the current study. KM, KS, GD acquired
and analyzed the data. KA, GS and CK drafted the manuscript. GS,
GD, KR, KS critically revised the manuscript. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Informed consent for publication was received from
the patient.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Puri A, Chang J, Dragovich T, Lucente P
and Kundranda MN: Skeletal metastases in advanced pancreatic ductal
adenocarcinoma (PDAC): A retrospective analysis. J Clin Oncol.
36(245)2018.
|
|
2
|
Joffe N and Antonioli DA: Osteoblastic
bone metastases secondary to adenocarcinoma of the pancreas. Clin
Radiol. 29:41–46. 1978.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Lin CT, Tang CT, Liu MY and Ma HI: Unusual
osteoblastic metastases in the spine secondary to adenocarcinoma of
the pancreas. Acta Chir Belg. 111:44–45. 2011.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Bakkevold KE, Arnesjø B and Kambestad B:
Carcinoma of the pancreas and papilla of Vater: Presenting
symptoms, signs, and diagnosis related to stage and tumour site. A
prospective multicentre trial in 472 patients. Norwegian pancreatic
cancer trial. Scand J Gastroenterol. 27:317–325. 1992.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Hess KR, Varadhachary GR, Taylor SH, Wei
W, Raber MN, Lenzi R and Abbruzzese JL: Metastatic patterns in
adenocarcinoma. Cancer. 106:1624–1633. 2006.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Borad MJ, Saadati H, Lakshmipathy A,
Campbell E, Hopper P, Jameson G, Von Hoff DD and Saif MW: Skeletal
metastases in pancreatic cancer: A retrospective study and review
of the literature. Yale J Biol Med. 82:1–6. 2009.PubMed/NCBI
|
|
7
|
Iguchi H, Yasuda M, Matsuo T, Sumii T and
Funakoshi A: Clinical features and management of pancreatic cancer
with bone metastases. Nihon Shokakibyo Gakkai Zasshi. 101:872–878.
2004.PubMed/NCBI(In Japanese).
|
|
8
|
Ray AE, Faltings L, Machnick S, Goenka A,
Opher E, Steinberg J, Ratzon F and Novoselac AV: Bone metastasis as
the only site of disease in a patient 7 years post treatment for a
locally advanced pancreatic adenocarcinoma. J Pancreas. 19:296–302.
2018.
|
|
9
|
Rosenberg E and Buchtel L: Cervical spine
pain as a presenting complaint in metastatic pancreatic cancer: A
case report. Postgrad Med. 128:331–333. 2016.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Pneumaticos SG, Savidou C, Korres DS and
Chatziioannou SN: Pancreatic cancer's initial presentation: Back
pain due to osteoblastic bone metastasis. Eur J Cancer Care (Engl).
19:137–140. 2010.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Rades D, Huttenlocher S, Schild SE and
Bartscht T: Metastatic spinal cord compression from pancreatic
cancer. Anticancer Res. 34:3727–3730. 2014.PubMed/NCBI
|