Introduction
Patients receiving chemotherapy are at high risk for
severe infections and complications such as acute respiratory
syndrome. The most commonly used adjuvant chemotherapy protocols
(docetaxel-cyclophosphamide every 3 weeks or the dose-dense
regimen, doxorubicin-cyclophosphamide every 2 weeks followed by
paclitaxel) incorporate granulocyte-colony stimulating factor
(G-CSF) (1-3).
G-CSF is routinely administered to prevent chemotherapy-associated
neutropenia but often results in significant neutrophilia.
We describe a patient with breast cancer who was
successfully treated for severe COVID-19 respiratory syndrome while
under treatment with adjuvant chemotherapy
(docetaxel-cyclophosphamide) and long-term G-CSF support. In
addition, we discuss the potential effect of G-CSF on the
respiratory deterioration of the patient given its cardinal role in
innate inflammation and, accordingly, the cytokine storm associated
with COVID-19. Our case shows how solutions to the immunity
challenge faced when treating a patient with chemotherapy may be
the source of a bigger problem in the coronavirus COVID-19
pandemic.
Case report
A 58-year-old woman with a history of obesity,
hypertension, and dyslipidemia was diagnosed with grade 2
Estrogen/Progesteron-positive, HER2-negative invasive ductal breast
carcinoma (7 mm) with axillary lymph node involvement (N=1/6) and
extracapsular invasion. The patient underwent right lumpectomy and
sentinel node biopsy. Systemic staging was negative for distant
metastases. The 21-gene Recurrence Score (Oncotype-DX™) was 19. She
was started on a regimen of intravenous (i.v.) docetaxel 75
mg/m2 and i.v. cyclophosphamide 600 mg/m2
every 3 weeks and subcutaneous (s.c.) pegfilgrastim (pegylated
G-CSF) 6 mg on day 3 of each cycle. On day 7 of cycle 2, the
patient presented with fever (38.2˚C) and neutropenia [absolute
neutrophil count (ANC) 700 cells/µl] and tested positive for
COVID-19. Two tests were performed. The first in the emergency
room, using geneXpert Xpress by Cepheid. The second test was
Allplex TM 2019-nCoV assay by Seegene. The second test was
performed while she was already in the department.
She reported a history of fatigue followed by fever
of 4 days' duration. She was hemodynamically stable, with oxygen
saturation 95% in room air with mild tachypnea (20 breaths/min).
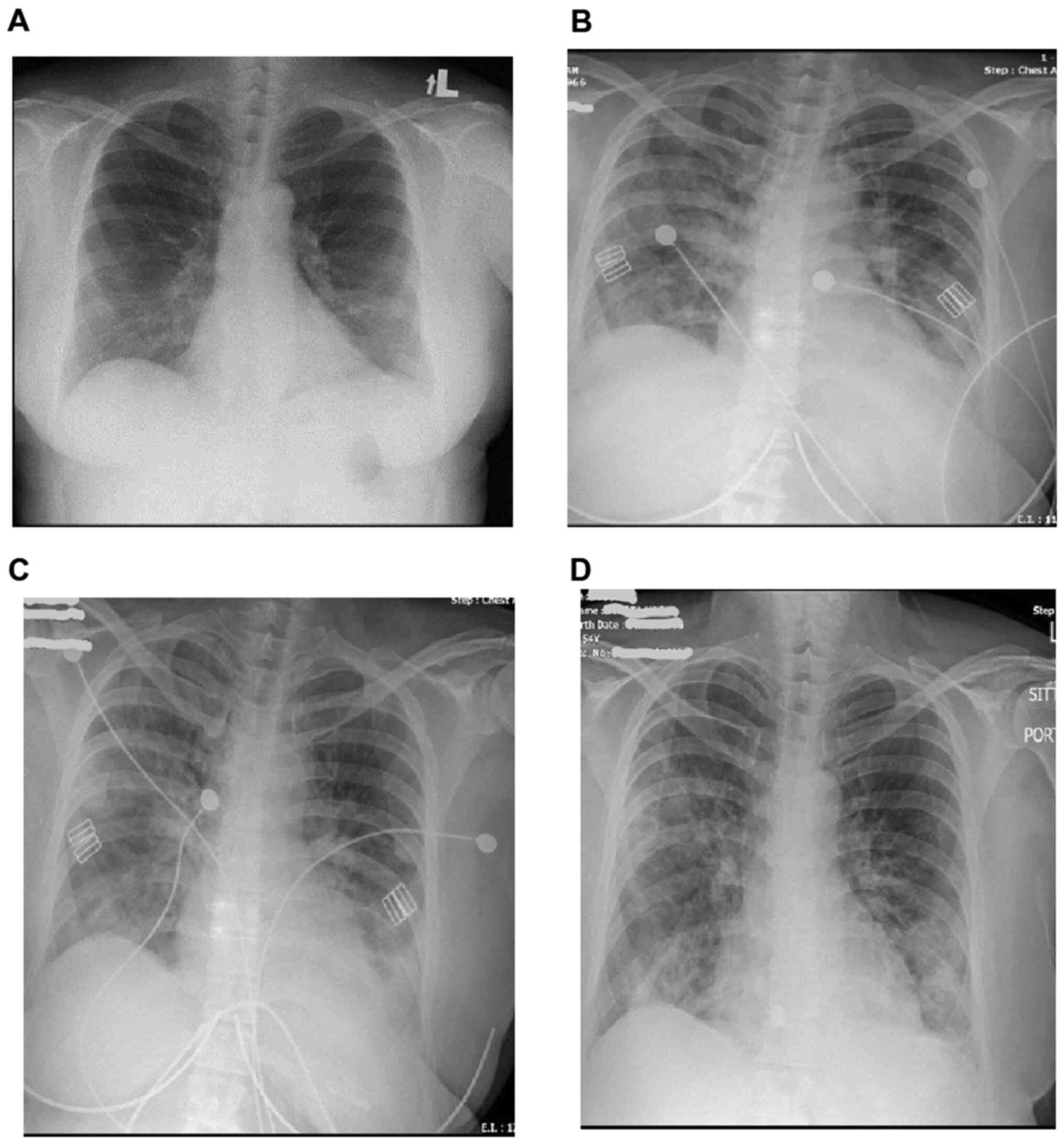
There was no cough, chest pain, or shortness of breath. Chest X-ray
showed mild bilateral infiltrates (Fig.
1). The patient also reported diarrhea grade 1.
After treatment with i.v. piperacillin/tazobactam
(4.5 g every 8 h), the neutropenic fever resolved. However, the
patient was increasingly tachypneic (respiratory rate 30
breaths/min, 88% desaturation) and was placed on 4L oxygen by nasal
cannula. Blood and stool cultures were negative; Clostridium
toxins were not detected. She also received s.c. enoxaparin sodium
(1 mg/kg bid), i.v. dexamethasone (6 mg qd for 10 days), and i.v.
remdesivir (loading dose 200 mg followed by 100 mg bid for 3
days).
The next day, the dyspnea and desaturation worsened.
Work-up revealed ANC 4,700 cells/µl sterile blood cultures, and
bilateral multifocal opacities on chest X-ray (Fig. 1). The patient was placed on a
high-flow nasal cannula (HFNC) therapy (flow 40 l/min,
FiO2 0.4%). On day 4, the ANC/absolute leukocyte count
(ALC) ratio peaked at 18.5, and HFNC parameters were increased
(flow 60 l/min, FiO2 0.8%). Acute atrial fibrillation
developed and was treated with amiodarone with conversion to sinus
rhythm.
On day 6 of hospitalization (day 10 of COVID-19
symptoms), gradual clinical improvement was noted along with a
decrease in ANC/ALC ratio. On day 14, the patient was discharged
home with no significant respiratory symptoms (Fig. 1).
Discussion
During the COVID-19 epidemic, to minimize the risk
of febrile neutropenia in patients receiving adjuvant chemotherapy
and to avoid overwhelming emergency rooms and hospitals, the NCCN
extended the prophylactic use of G-CSF from high-risk-only patients
(>20%) to intermediate-to-high-risk patients (10-20%). A
cautionary note was added that cases of respiratory infection,
respiratory symptoms, or confirmed or suspected COVID-19, G-CSF
posed a risk of increased pulmonary inflammation or an increase in
levels of inflammatory cytokines associated with an adverse outcome
(4).
Our patient had pre-existing comorbidities known to
be associated with respiratory and other systemic complications
(5-7)
in addition to two life-threatening conditions of febrile
neutropenia and active COVID-19. Although per-protocol G-CSF
administration may have prevented severe bacterial infection, it
could also have caused further deterioration in her
COVID-19-related lung status. Yang et al found that in
patients with COVID-19, a high ratio of neutrophil count to
lymphocyte count, high ratio was associated with a high rate of
rapid development of severe disease (8).
Neutrophils are short-lived closely controlled cells
that form an essential part of the host defense and inflammatory
response. G-CSF regulates neutrophil production by inducing the
proliferation and maturation of myeloid progenitors and promoting
neutrophil release from bone marrow (9). Like G-CSF, granulocyte-macrophage CSF
(GM-CSF) belongs to the hematopoietin/cytokine receptor superfamily
and increases neutrophil chemotaxis and migration. However, its
response kinetics differ and it is considered more pro-inflammatory
(10). It also contributes to the
development and maintenance of alveolar macrophages (11,12).
Cumulative data suggest that both factors may play a cardinal role
in innate inflammation and are potential mediators of the cytokine
storm (13-15)
as part of a positive feedback loop with inflammatory
cytokines/chemokines, T-helper cells, and neighbouring cell
populations (16).
Most of the reports on the involvement of
stimulating factors in COVID-19 have focused on GM-CSF (13,14)
and the incorporation of anti GM-CSF receptor to mitigate the
COVID-19 cytokine storm (15,17,18).
Using a prospective design, De Luca et al showed that
administration of mavrilimumab, an anti-GSF monoclonal antibody,
led to better clinical outcomes than standard treatment in
mechanically ventilated patients with severe COVID-19 pneumonia,
hypoxia, and systemic hyperinflammation (17).
There are no standardized treatments available for
COVID-19. It is clear that one protocol does not fit all, and
researchers are seeking accurate predictive markers to aid in the
selection and manner of incorporation of the available drugs,
including steroids (19).
Dexamethasone is part of the standard of care for respiratory
distress or severe pneumonia, and it seems that it may be of
benefit in modulating G-CSF associated respiratory syndrome. In our
patient, in view of the expected G-CSF-induced neutrophil
activation the clinical course, and the developing cytokine storm,
it was reasonable to add steroids.
We do not know which specific part(s) of the
treatment (antivirals, steroids, supportive care, or their
combination) had the greatest impact on the patient's outcome.
Nevertheless, it is encouraging that an immune-suppressed patient
with comorbidities and an apparently looming cytokine storm could
recover in a short time with current treatment protocols. Given the
potential involvement of cytokines in exacerbation of COVID-19, the
new risk/benefit balance of active treatments, especially those
including G-CSF injections, needs to be carefully considered
(20).
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
RY is the primary care-give in the Oncology
institute, conceptualized and co-wrote the manuscript and performed
the literature search. HG is part of the breast-unit
multidisciplinary group in the oncology instute which advised
regarding the patient's treatment, and in addition co-wrote to the
manuscript. MS was the main care-giver during her hospitalization,
in addition he co-wrote the manuscript. JD and RM treated the
patient at her initial Covid-19 diagnosis. IK acted as medical
supervisor during the patient's hospitalization with COVID-19
infection and co-wrote the manuscript. All authors had access to
the data and a role in writing the manuscript. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The Helsinki Committee of Rabin Medical Center
provided exemption for this case report since it does not contain
any studies with human participants performed by any of the
authors.
Patient consent for publication
The Helsinki Committee of Rabin Medical Center
provided exemption for this case report since it does not contain
any studies with human participants performed by any of the
authors.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Citron ML, Berry DA, Cirrincione C, Hudis
C, Winer EP, Gradishar WJ, Davidson NE, Martino S, Livingston R,
Ingle JN, et al: Randomized trial of dose-dense versus
conventionally scheduled and sequential versus concurrent
combination chemotherapy as postoperative adjuvant treatment of
node-positive primary breast cancer: First report of intergroup
trial C9741/cancer and leukemia group B trial 9741. J Clin Oncol.
21:1431–1439. 2003.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Jones SE, Savin MA, Holmes FA,
O'Shaughnessy JA, Blum JL, Vukelja S, McIntyre KJ, Pippen JE,
Bordelon JH, Kirby R, et al: Phase III trial comparing doxorubicin
plus cyclophosphamide with docetaxel plus cyclophosphamide as
adjuvant therapy for operable breast cancer. J Clin Oncol.
24:5381–5387. 2006.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Early Breast Cancer Trialists'
Collaborative Group (EBCTCG). Increasing the dose intensity of
chemotherapy by more frequent administration or sequential
scheduling: A patient-level meta-analysis of 37 298 women with
early breast cancer in 26 randomised trials. Lancet. 393:1440–1452.
2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
NCCN Hematopoietic Growth Factors:
Short-term recommendations specific to issues with COVID-19
(SARS-CoV-2). https://www.nccn.org/covid-19/pdf/HGF_COVID-19.pdf.
Accessed June 12, 2020.
|
|
5
|
Long L, Zeng X, Zhang X, Xiao W, Guo E,
Zhan W, Yang X, Li C, Wu C, Xu T, et al: Short-term outcomes of
COVID-19 and risk factors for progression. Eur Respir J.
55(2000990)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Wu C, Chen X, Cai Y, Xia J, Zhou X, Xu S,
Huang H, Zhang L, Zhou X, Du C, et al: Risk factors associated with
acute respiratory distress syndrome and death in patients with
coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern
Med. 180:934–943. 2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Yang J, Zheng Y, Gou X, Pu K, Chen Z, Guo
Q, Ji R, Wang H, Wang Y and Zhou Y: Prevalence of comorbidities and
its effects in patients infected with SARS-CoV-2: A systematic
review and meta-analysis. Int J Infect Dis. 94:91–95.
2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Yang AP, Liu JP, Tao WQ and Li HM: The
diagnostic and predictive role of NLR, d-NLR and PLR in COVID-19
patients. Int Immunopharmacol. 84(106504)2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Eyles JL, Hickey MJ, Norman MU, Croker BA,
Roberts AW, Drake SF, James WG, Metcalf D, Campben IK and Wicks IP:
A key role for G-CSF-induced neutrophil production and trafficking
during inflammatory arthritis. Blood. 112:5193–5201.
2008.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Hamilton JA and Achuthan A: Colony
stimulating factors and myeloid cell biology in health and disease.
Trends Immunol. 34:81–89. 2013.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Guilliams M, De Kleer I, Henri S, Post S,
Vanhoutte C, De Prijck S, Deswarte K, Malissen B, Hammad H and
Lambrecht BN: Alveolar macrophages develop from fetal monocytes
that differentiate into long-lived cells in the first week of life
via GM-CSF. J Exp Med. 210:1977–1992. 2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Trapnell BC, Nakata K, Bonella F, Campo I,
Griese M, Hamilton J, Wang T, Morgan C, Cottin V and McCarthy C:
Pulmonary alveolar proteinosis. Nat Rev Dis Primers.
5(16)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Favalli EG and Caporali R: GM-CSF in the
treatment of COVID-19: A new conductor in the pathogenesis of
cytokine storm? Lancet Rheumatol. 2(e448)2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Nawar T, Morjaria S, Kaltsas A, Patel D,
Perez-Johnston R, Daniyan AF, Mailankody S and Parameswaran R:
Granulocyte-colony stimulating factor in COVID-19: Is it
stimulating more than just the bone marrow? Am J Hematol.
95:E210–E213. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Lang FM, Lee KM, Teijaro JR, Becher B and
Hamilton JA: GM-CSF-based treatments in COVID-19: Reconciling
opposing therapeutic approaches. Nat Rev Immunol. 20:507–514.
2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Komuczki J, Tuzlak S, Friebel E, Hartwig
T, Spath S, Rosenstiel P, Waisman A, Opitz L, Oukka M, Schreiner B,
et al: Fate-mapping of GM-CSF expression identifies a discrete
subset of inflammation-driving T helper cells regulated by
cytokines IL-23 and IL-1β. Immunity. 50:1289–1304.e6.
2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
De Luca G, Cavalli G, Campochiaro C,
Della-Torre E, Angelillo P, Tomelleri A, Boffini N, Tentori S,
Mette F, Farina N, et al: GM-CSF blockade with mavrilimumab in
severe COVID-19 pneumonia and systemic hyperinflammation: A
single-centre, prospective cohort study. Lancet Rheumatol.
2:e465–e473. 2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Mehta P, Porter JC, Manson JJ, Isaacs JD,
Openshaw PJM, McInnes IB, Summers C and Chambers RC: Therapeutic
blockade of granulocyte macrophage colony-stimulating factor in
COVID-19-associated hyperinflammation: Challenges and
opportunities. Lancet Respir Med. 8:822–830. 2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Lev S, Gottesman T, Sahaf Levin G,
Lederfein D, Berkov E, Diker D, Zaidman A, Nutman A, Ber TI, Angel
A, et al: Real-time IP-10 measurements as a new tool for
inflammation regulation within a clinical decision support protocol
for managing severe COVID-19 patients. medRxiv:
2020.07.21.20158782, 2020. doi: https://doi.org/10.1101/2020.07.21.20158782.
|
|
20
|
Brunetti O, Derakhshani A, Baradaran B,
Galvano A, Russo A and Silvestris N: COVID-19 infection in cancer
patients: How can oncologists deal with these patients? Front
Oncol. 10(734)2020.PubMed/NCBI View Article : Google Scholar
|