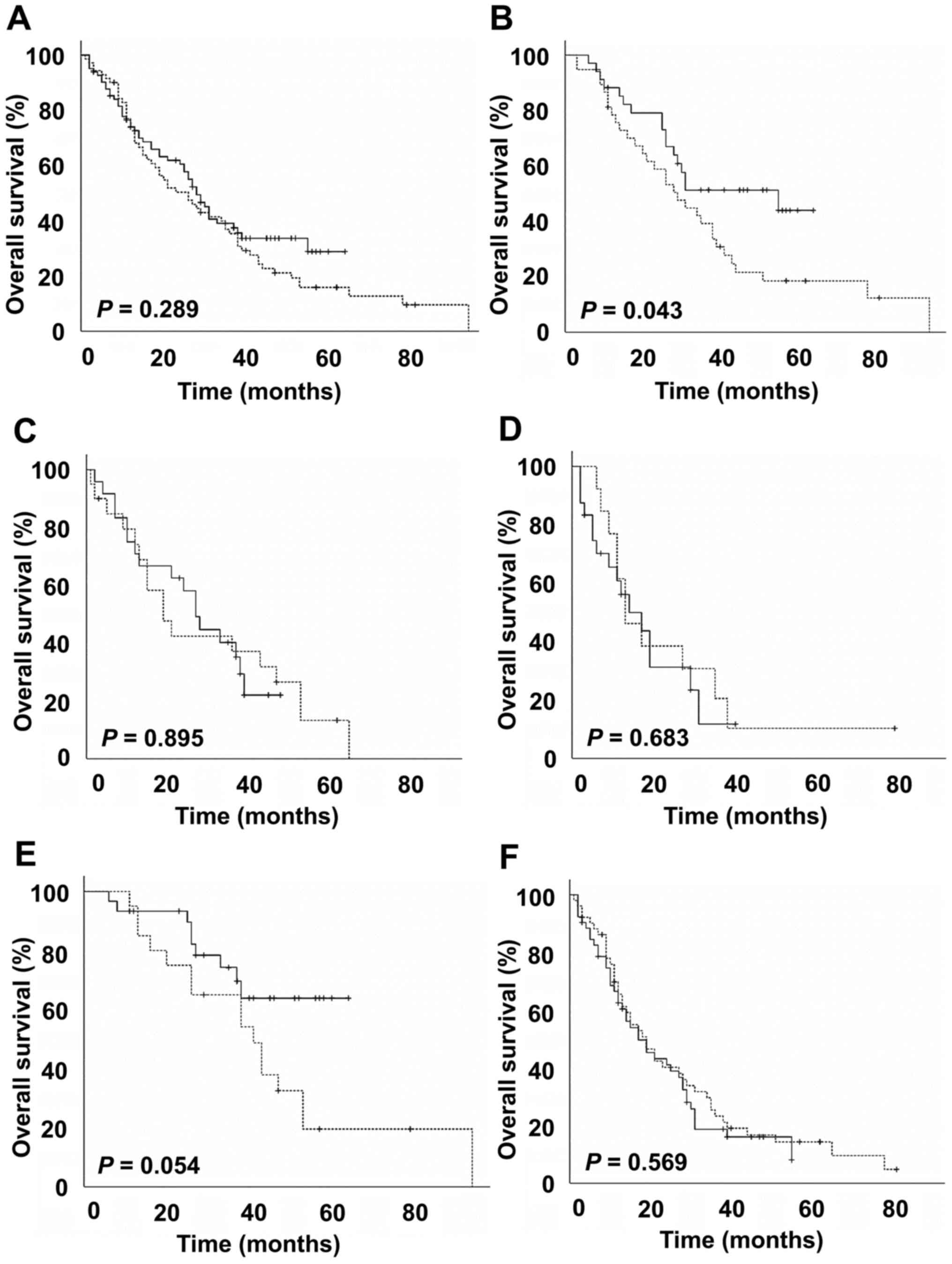
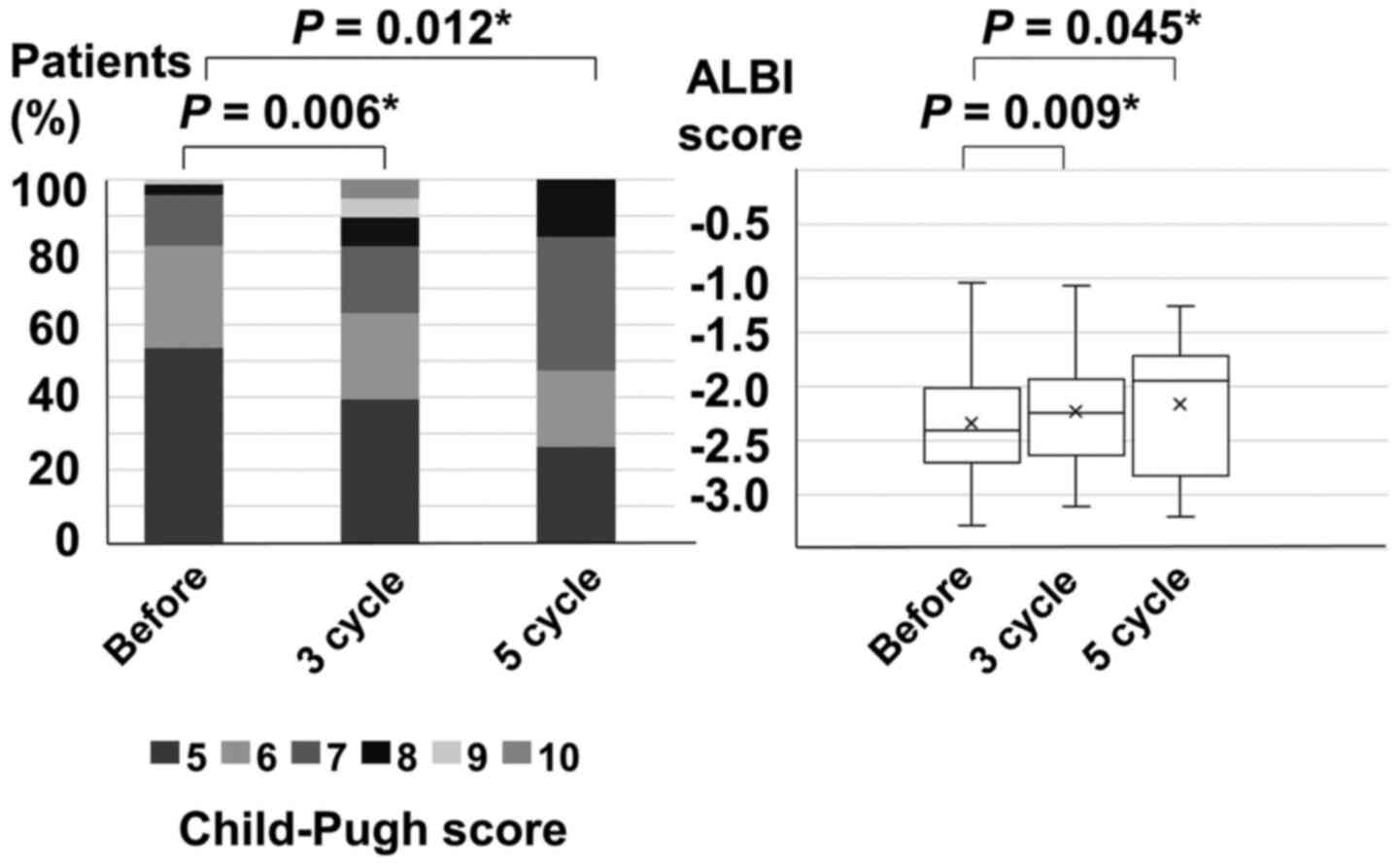
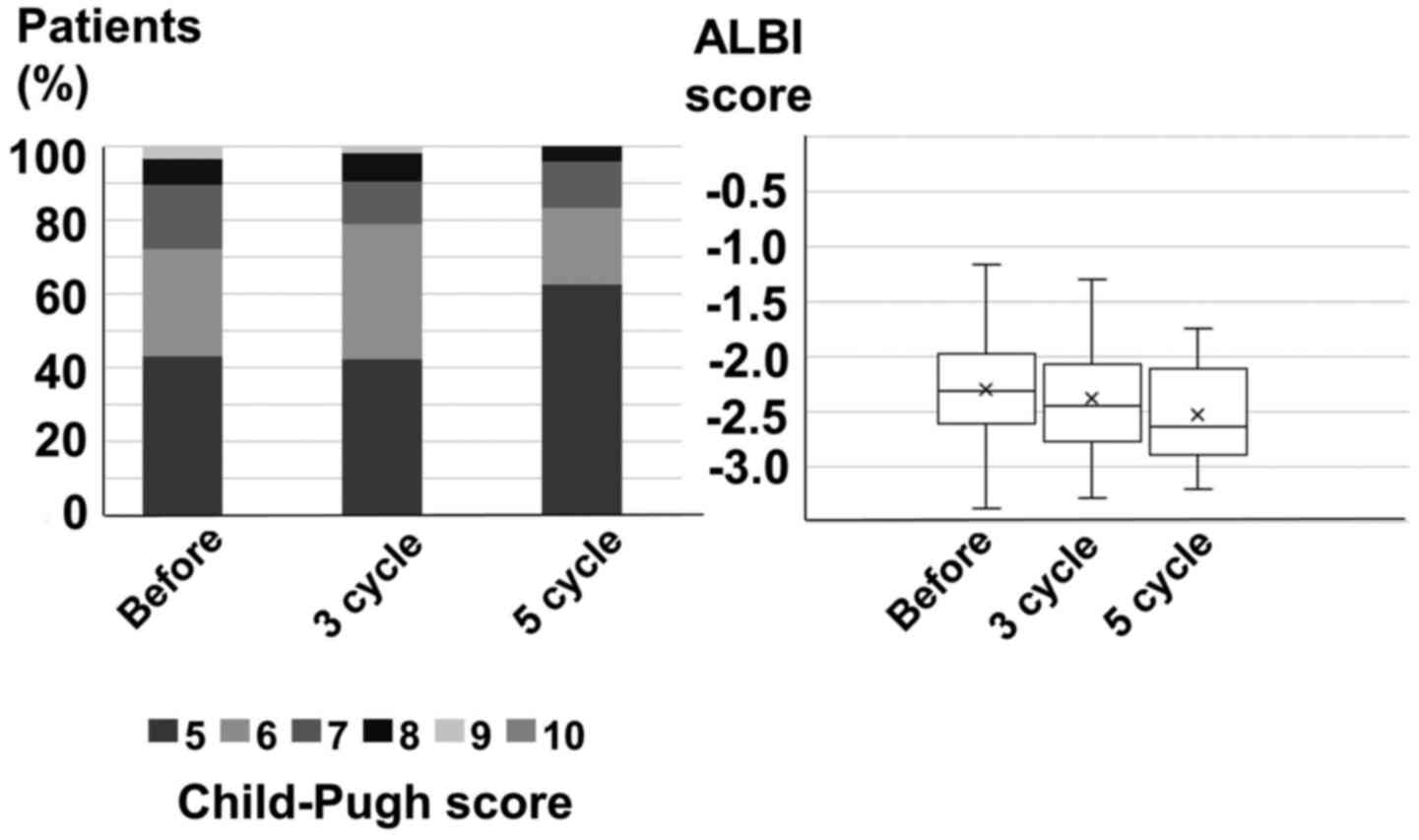
|
1
|
Forner A, Reig M and Bruix J:
Hepatocellular carcinoma. Lancet. 391:1301–1314. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Vauthey JN, Dixon E, Abdalla EK, Helton
WS, Pawlik TM, Taouli B, Brouquet A and Adams RB: American
Hepato-Pancreato-Biliary Association; Society of Surgical Oncology;
Society for Surgery of the Alimentary Tract. Pretreatment
assessment of hepatocellular carcinoma: Expert consensus statement.
HPB (Oxford). 12:289–299. 2010.PubMed/NCBI View Article : Google Scholar
|
|
3
|
European Association for the Study of the
Liver. Electronic address: simpleeasloffice@easloffice.eu;
European Association for the Study of the Liver. EASL clinical
practice guidelines: Management of hepatocellular carcinoma. J
Hepatol. 69:182–236. 2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Heimbach JK, Kulik LM, Finn RS, Sirlin CB,
Abecassis MM, Roberts LR, Zhu AX, Murad MH and Marrero JA: AASLD
guidelines for the treatment of hepatocellular carcinoma.
Hepatology. 67:358–380. 2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Omata M, Cheng AL, Kokudo N, Kudo M, Lee
JM, Jia J, Tateishi R, Han KH, Chawla YK, Shiina S, et al:
Asia-Pacific clinical practice guidelines on the management of
hepatocellular carcinoma: A 2017 update. Hepatol Int. 11:317–370.
2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Facciorusso A, Serviddio G and Muscatiello
N: Transarterial radioembolization vs. chemoembolization for
hepatocarcinoma patients: A systematic review and meta-analysis.
World J Hepatol. 8:770–778. 2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Facciorusso A, Di Maso M and Muscatiello
N: Drug-eluting beads versus conventional chemoembolization for the
treatment of unresectable hepatocellular carcinoma: A
meta-analysis. Dig Liver Dis. 48:571–577. 2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Facciorusso A, Mariani L, Sposito C,
Spreafico C, Bongini M, Morosi C, Cascella T, Marchianò A, Camerini
T, Bhoori S, et al: Drug-eluting beads versus conventional
chemoembolization for the treatment of unresectable hepatocellular
carcinoma. J Gastroenterol Hepatol. 31:645–653. 2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Golfieri R, Giampalma E, Renzulli M, Cioni
R, Bargellini I, Bartolozzi C, Breatta AD, Gandini G, Nani R,
Gasparini D, et al: Randomised controlled trial of
doxorubicin-eluting beads vs. conventional chemoembolisation for
hepatocellular carcinoma. Br J Cancer. 111:255–264. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Osuga K, Khankan AA, Hori S, Okada A,
Sugiura T, Maeda M, Nagano H, Yamada A, Murakami T and Nakamura H:
Transarterial embolization for large hepatocellular carcinoma with
use of superabsorbent polymer microspheres: Initial experience. J
Vasc Interv Radiol. 13:929–934. 2002.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Grosso M, Vignali C, Quaretti P, Nicolini
A, Melchiorre F, Gallarato G, Bargellini I, Petruzzi P, Massa
Saluzzo C, Crespi S and Sarti I: Transarterial chemoembolization
for hepatocellular carcinoma with drug-eluting microspheres:
Preliminary results from an Italian multicentre study. Cardiovasc
Intervent Radiol. 31:1141–1149. 2008.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Seki A, Hori S, Kobayashi K and Narumiya
S: Transcatheter arterial chemoembolization with epirubicin-loaded
superabsorbent polymer microspheres for 135 hepatocellular
carcinoma patients: Single-center experience. Cardiovasc Intervent
Radiol. 34:557–565. 2011.PubMed/NCBI View Article : Google Scholar
|
|
13
|
van Malenstein H, Maleux G, Vandecaveye V,
Heye S, Laleman W, van Pelt J, Vaninbroukx J, Nevens F and Verslype
C: A randomized phase II study of drug-eluting beads versus
transarterial chemoembolization for unresectable hepatocellular
carcinoma. Onkologie. 34:368–376. 2011.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Duan F, Wang EQ, Lam MG, Abdelmaksoud MH,
Louie JD, Hwang GL, Kothary N, Kuo WT, Hofmann LV and Sze DY:
Superselective chemoembolization of HCC: Comparison of short-term
safety and efficacy between drug-eluting LC beads, quadraspheres,
and conventional ethiodized oil emulsion. Radiology. 278:612–621.
2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kucukay F, Badem S, Karan A, Ozdemir M,
Okten RS, Ozbulbul NI, Kucukay MB, Unlu I, Bostanci EB and Akdogan
M: A single-center retrospective comparison of doxorubicin-loaded
hepasphere transarterial chemoembolization with conventional
transarterial chemoembolization for patients with unresectable
hepatocellular carcinoma. J Vasc Interv Radiol. 26:1622–1629.
2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Morimoto M, Kobayashi S, Moriya S, Ueno M,
Tezuka S, Irie K, Goda Y and Ohkawa S: Short-term efficacy of
transarterial chemoembolization with epirubicin-loaded
superabsorbent polymer microspheres for hepatocellular carcinoma:
Comparison with conventional transarterial chemoembolization. Abdom
Radiol (NY). 42:612–619. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Facciorusso A: Drug-eluting beads
transarterial chemoembolization for hepatocellular carcinoma:
Current state of the art. World J Gastroenterol. 24:161–169.
2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Bruix J, Qin S, Merle P, Granito A, Huang
YH, Bodoky G, Pracht M, Yokosuka O, Rosmorduc O, Breder V, et al:
Regorafenib for patients with hepatocellular carcinoma who
progressed on sorafenib treatment (RESORCE): A randomised,
double-blind, placebo-controlled, phase 3 trial. Lancet. 389:56–66.
2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Kudo M, Finn RS, Qin S, Han KH, Ikeda K,
Piscaglia F, Baron A, Park JW, Han G, Jassem J, et al: Lenvatinib
versus sorafenib in first-line treatment of patients with
unresectable hepatocellular carcinoma: A randomised phase 3
non-inferiority trial. Lancet. 391:1163–1173. 2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Llovet JM, Ricci S, Mazzaferro V, Hilgard
P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A,
et al: Sorafenib in advanced hepatocellular carcinoma. N Engl J
Med. 359:378–390. 2008.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Hiraoka A, Kumada T, Kudo M, Hirooka M,
Koizumi Y, Hiasa Y, Tajiri K, Toyoda H, Tada T, Ochi H, et al:
Hepatic function during repeated TACE procedures and prognosis
after introducing sorafenib in patients with unresectable
hepatocellular carcinoma: Multicenter analysis. Dig Dis.
35:602–610. 2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Miksad RA, Ogasawara S, Xia F, Fellous M
and Piscaglia F: Liver function changes after transarterial
chemoembolization in US hepatocellular carcinoma patients: The
LiverT study. BMC Cancer. 19(795)2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Lencioni R, de Baere T, Burrel M, Caridi
JG, Lammer J, Malagari K, Martin RC, O'Grady E, Real MI, Vogl TJ,
et al: Transcatheter treatment of hepatocellular carcinoma with
doxorubicin-loaded DC Bead (DEBDOX): Technical recommendations.
Cardiovasc Intervent Radiol. 35:980–985. 2012.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Johnson PJ, Berhane S, Kagebayashi C,
Satomura S, Teng M, Reeves HL, O'Beirne J, Fox R, Skowronska A,
Palmer D, et al: Assessment of liver function in patients with
hepatocellular carcinoma: A new evidence-based approach-the ALBI
grade. J Clin Oncol. 33:550–558. 2015.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Idee JM and Guiu B: Use of Lipiodol as a
drug-delivery system for transcatheter arterial chemoembolization
of hepatocellular carcinoma: A review. Crit Rev Oncol Hematol.
88:530–549. 2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Pinato DJ, Yen C, Bettinger D, Ramaswami
R, Arizumi T, Ward C, Pirisi M, Burlone ME, Thimme R, Kudo M and
Sharma R: The albumin-bilirubin grade improves hepatic reserve
estimation post-sorafenib failure: Implications for drug
development. Aliment Pharmacol Ther. 45:714–722. 2017.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Ueshima K, Nishida N, Hagiwara S, Aoki T,
Minami T, Chishina H, Takita M, Minami Y, Ida H, Takenaka M, et al:
Impact of baseline ALBI grade on the outcomes of hepatocellular
carcinoma patients treated with lenvatinib: A multicenter study.
Cancers (Basel). 11(952)2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
El-Khoueiry AB, Sangro B, Yau T, Crocenzi
TS, Kudo M, Hsu C, Kim TY, Choo SP, Trojan J, Welling TH Rd, et al:
Nivolumab in patients with advanced hepatocellular carcinoma
(CheckMate 040): An open-label, non-comparative, phase 1/2 dose
escalation and expansion trial. Lancet. 389:2492–2502.
2017.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Abou-Alfa GK, Meyer T, Cheng AL,
El-Khoueiry AB, Rimassa L, Ryoo BY, Cicin I, Merle P, Chen Y, Park
JW, et al: Cabozantinib in patients with advanced and progressing
hepatocellular carcinoma. N Engl J Med. 379:54–63. 2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Zhu AX, Finn RS, Edeline J, Cattan S,
Ogasawara S, Palmer D, Verslype C, Zagonel V, Fartoux L, Vogel A,
et al: Pembrolizumab in patients with advanced hepatocellular
carcinoma previously treated with sorafenib (KEYNOTE-224): A
non-randomised, open-label phase 2 trial. Lancet Oncol. 19:940–952.
2018.PubMed/NCBI View Article : Google Scholar
|