Introduction
Acute malignant colorectal obstruction is a
complication of colorectal cancer (CRC) occurring in 7-29% of
patients (1-4)
and is associated with rapid colonic decompression, requiring
interventions, such as colostomy, resection, and ileal tube
placement. However, these emergency procedures are associated with
high rates of mortality and morbidity (5-9).
In 1990, Dohmoto et al first reported on colon stent
placement [self-expandable metallic stents (SEMSs)] for palliation
of malignant colorectal obstruction (10). Indications for the placement of SEMS
also include pre-operative colonic decompression (11). Recently, SEMS has become widely
available and is now considered an alternative therapeutic option
for the management of colorectal obstruction secondary to CRC
(5,12,13).
However, malignant colorectal obstruction is caused by CRC and by
infiltration and dissemination of extracolonic malignancy (ECM),
such as gastric, pancreatic, gynaecologic, and urinary system
cancers (5,14-19).
Obstruction in CRC results from intraluminal growth; ECM leads to
the development of intestinal obstruction owing to external
invasion or compression, and the axis of the colon may change
(14,20). Some studies have reported the safety
and efficacy of SEMS placement for malignant colorectal obstruction
in patients with CRC; however, only a few studies have reported the
use of SEMS placement for diseases caused by ECM (5,11,14-23).
Ahn et al (23) performed
SEMS placement in 72 patients with colorectal obstruction by ECM,
with 90.3% technical success rate and 87.7% clinical success rate.
However, Ahn et al did not report any comparison with the
CRC group; thus, it is unclear whether SEMS is equally effective
for CRC as well as ECM patients. Therefore, the safety and efficacy
of SEMS placement for ECM currently remain unknown. In this study,
we aimed to compare the clinical and technical success between CRC
and ECM when an SEMS is placed for malignant colorectal
obstruction. In addition, the incidence of perforation,
re-obstruction, and migration as complications was examined and
compared between the ECM and CRC groups. The time elapsed before
the onset of complications was also compared between the two groups
using the Kaplan-Meier method. In addition, risk factors for
re-obstruction were extracted through a multivariate analysis.
Materials and methods
Study subjects
We retrospectively evaluated the endoscopy and
clinical records of 68 patients who underwent procedures for SEMS
placement at our institution due to malignant colorectal
obstruction between January 2012 and September 2019. All patients
provided a written approval agreement. Patients who had SEMS
placement for benign diseases, such as diverticulosis and
postoperative stenosis, were excluded from this study. The study
was approved by the Standards of Official Conduct Committee at
Saiseikai Niigata Hospital (IRB no. E17-28). This study adhered to
the principles of the Declaration of Helsinki of 1964.
SEMS placement was performed under
endoscopic/fluoroscopic guidance (24). Patients used analgesics
(pentazocine; 7.7-15 mg) and sedatives (midazolam; 5-10 mg) in
response to their distress during the SEMS placement procedure. A
wide working channel endoscope (CF-H260AI, CF-H290I, CF-HQ290I,
PCF-Q260JI; Olympus) was introduced into the stenosed portion. The
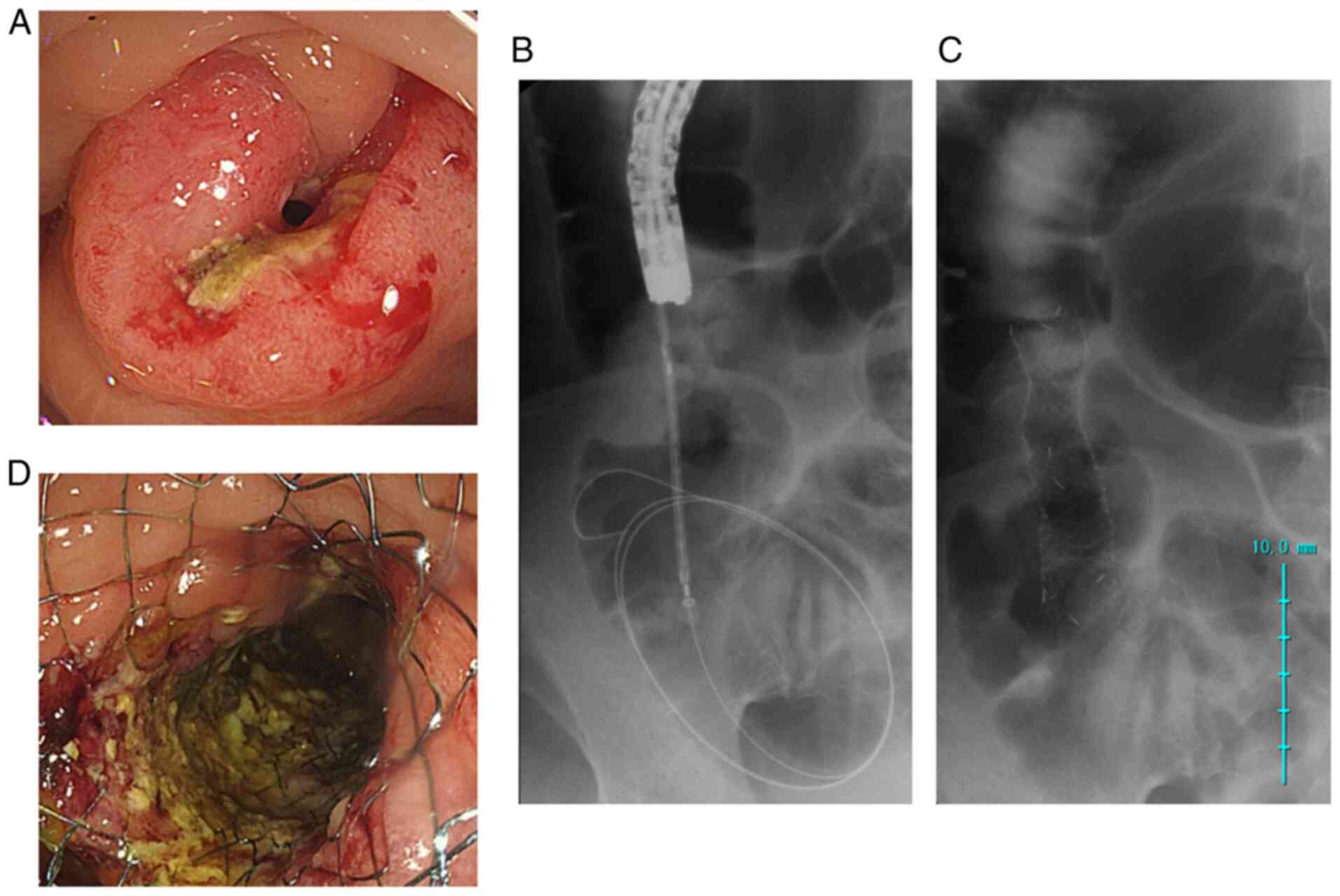
site of stenosis was detected on endoscopic imaging or was
visualized using a contrast medium under fluoroscopic and
endoscopic guidance (Fig. 1A). A
biliary guidewire (0.035 inches) was passed to traverse the
obstruction until a safety loop of the guidewire could be created.
A biliary catheter was then advanced to follow it. A water-soluble
contrast agent (amidotrizoic acid) was injected into the proximal
side of the stricture in order to assess it; we evaluated the
stricture and measured its exact length. The suitable length of the
stent was determined by adding 2-5 cm to the length of the
stricture. A longer stent was used for the stricture of the flexion
or ECM (20,25). The delivery system was inserted
through the guidewire facilitating entry into the site of
obstruction (Fig. 1B). While the
outer sheath was retracted under fluoroscopy with endoscopic
guidance, the centre of the stent was adjusted at the narrowest
point (21) (Fig. 1C and D).
Statistical analysis
Statistical analysis was conducted using the EZR
software (Saitama Medical Center, Jichi Medical University,
Saitama, Japan) (26). Patients'
information and clinical characteristics were presented as means ±
standard deviations, and median and range. The means of continuous
variables were compared between the CRC and ECM groups using the
Student's t-test or the Mann-Whitney U test, as appropriate.
Differences in outcomes between the two groups were examined using
χ2 or Fisher's exact tests. In addition, the time
elapsed before the onset of complications after stent placement was
analysed using the Kaplan-Meier method in both groups, and their
curves were compared using the log-rank method. In addition,
multivariate analysis (logistic regression) was performed on risk
factors for re-obstruction.
The results are expressed as mean ± standard
deviation or as percentages. P-values of <0.05 were considered
statistically significant.
Definitions
Technical success was defined as successful
deployment of the stent across the entire length of the stricture,
without any adverse events e.g., perforation, stent migration, and
major bleeding. Clinical success was defined as colonic
decompression and relief of obstructive symptoms within 48 h of
stent placement, with no need for reintervention (14,27).
The Colorectal Obstruction Scoring System (CROSS) is
a scoring system proposed by the Colonic Stent Safe Procedure
Research Group of Japan. To make scoring intestinal obstruction as
simple as possible, the CROSS asks about the patient's oral intake
and whether the patient is symptomatic, despite being able to eat.
It is described as follows: Score 0: Requiring a continuous
decompressive procedure; score 1: No oral intake; score 2: Liquid
or enteral nutrients; score 3: Soft solids, low-residue, and full
diet with symptoms of stricture; and score 4: Soft solids,
low-residue, and full diet without symptoms of stricture (28). CROSS scores before and after stent
placement in both groups were compared and analysed using
nonparametric tests.
Results
SEMS placement was performed for CRC in 59/68
patients (86.8%) at our institution. Of the 9/68 ECM patients
(13.2%), indications for SEMS placement were pancreatic (n=4),
gastric (n=4), and oesophageal (n=1) cancers. Patients'
demographics and stent details are summarised in Table I. No significant differences were
noted in age, male-to-female ratio, site of obstruction, and
clinical stage between the CRC and ECM groups. The length and width
of stents were chosen depending on the length and degree of flexion
of the stenotic portion of the colon. Uncovered stents were used in
all patients, and there were no significant differences in the
length, width, or type of stent between the two groups. In the CRC
group, 30 patients (50.8%) underwent surgery (28 patients underwent
colectomy and two underwent colostomy) after stent placement, and
29 patients (49.2%) underwent palliative placement. All patients in
the ECM group had a palliative placement, and there was a
significant difference between the two groups with respect to the
purpose of stent placement (P=0.003). There was no significant
difference in the combination of chemotherapy in the overall course
between the two groups: 27 patients (45.8%) in the CRC group and
seven patients (77.8%) in the ECM group. However, six patients
(10.2%) in the CRC group (XELOX + Bevacizumab, XELOX, mFOLFOX6 +
Cetuximab) and 5 patients (55.6%) in the ECM group (GEM, CDDP +
5-FU, TS-1 + CDDP) received chemotherapy prior to stenting, showing
a significant difference (P=0.004; Table I).
 | Table IBaseline characteristics of patients
in the CRC and ECM groups. |
Table I
Baseline characteristics of patients
in the CRC and ECM groups.
| Characteristic
Cases | CRC (n) 59 | % | ECM (n) 9 | % | P-value |
|---|
| Age | 73.17±13.15 | (46-95) | 69.11±9.83 | (55-88) | 0.379 |
| Male/female | 32/27 | | 5/4 | | NS |
| Site of
obstruction | | | | | |
|
Right side
colon | 19 | 32.2 | 4 | 44.4 | 0.710 |
|
Left side
colon | 40 | 67.8 | 5 | 55.5 | |
| cStage | | | | | |
|
II | 16 | 27.1 | 0 | 0.0 | 0.217 |
|
IIIa | 11 | 18.6 | 0 | 0.0 | |
|
IIIb | 2 | 3.4 | 0 | 0.0 | |
|
IV | 30 | 50.8 | 9 | 100.0 | |
| Primary site | | | | | |
|
Pancreas | | | 4 | 0.4 | |
|
Stomach | | | 4 | 0.4 | |
|
Esophagus | | | 1 | 0.1 | |
| Stent | | | | | |
|
Uncovered | 59 | 100.0 | 9 | 100.0 | NS |
|
Length
(cm) | 9.47±2.42 | (6-12) | 10.67±1.32 | (9-12) | 0.154 |
|
Width
(mm) | 20.44±1.97 | (18-22) | 20.22±2.11 | (18-22) | 0.759 |
| Product name of the
stent | | | | | |
|
Niti-S | 42 | 71.2 | 5 | 55.6 | 0.415 |
|
Wall
Flex | 6 | 10.2 | 2 | 22.2 | |
|
JENTLLY | 8 | 13.6 | 1 | 11.1 | |
|
HANARO | 3 | 5.1 | 1 | 11.1 | |
| Purpose | | | | | |
|
BTS | 30 | 50.8 | 0 | 0.0 | 0.004 |
|
PAL | 29 | 49.2 | 9 | 100.0 | |
| Chemotherapy | | | | | |
|
All | 27 | 45.8 | 7 | 77.8 | 0.150 |
|
Prior
stenting | 6 | 10.2 | 5 | 55.6 | 0.004 |
Clinical outcomes
The technical success rate of stent placement was
98.3% in the CRC group and 100% in the ECM group, with no
significant difference. In the CRC group, there was one case of
perforation of the guidewire at the time of placement, which
resulted in emergency surgery. The clinical success rate was
significantly different between the CRC (96.6%) and ECM (66.7%)
groups (P=0.01). The CROSS score before stent placement did not
significant differ between the CRC (0.508±1.006) and ECM
(0.222±0.441) groups. However, there was a significant difference
in the CROSS score after stenting between the CRC (3.881±0.59) and
ECM (2.778±1.856) groups (P=0.0006), indicating a poorer
improvement in the colorectal obstruction score in the ECM group.
Complications were observed in 15 patients (25.4%) in the CRC group
and in six patients (66.7%) in the ECM group, showing significant
difference (P=0.02). Complications were further investigated
according to types, such as perforation, migration, and
re-obstruction. There was no significant difference in the number
of perforations between the CRC (four patients, 6.8%) and ECM (two
patients, 22.2%) groups. Two patients in the ECM group who
underwent stent-in-stent placement with stent re-obstruction
developed perforation. One patient underwent emergency surgery, and
one patient was treated conservatively. In the CRC group, three of
the four patients with perforation underwent emergency surgery, and
one patient was treated conservatively. One patient received
chemotherapy with ramucirumab post-stent placement, another had
pancreatic cancer with extensive peritoneal dissemination, and the
other had re-obstruction of the stent and received a stent-in-stent
placement. There were four (6.8%) cases of migration in the CRC
group and none in the ECM group. The migration occurred in patients
whose tumours had shrunk with chemotherapy. Re-obstruction occurred
in 10 patients in the CRC group (16.9%) and six patients in the ECM
group (66.7%), showing statistically significant difference
(P=0.004). At the time of experiencing complications, there were
4/6 (66.7%) patients in the ECM group and 4/15 (26.7%) in the CRC
group who required emergency surgery (colostomy or ileostomy),
although the difference was not significant. There was no
significant difference in the number of patients who underwent
endoscopic procedures (addition of stents, stent cleaning, or
dilation) between the ECM and CRC groups [1/6 (16.7%) vs. 5/15
(33.3%)] (Table II).
 | Table IIClinical outcomes of patients in the
CRC and ECM groups. |
Table II
Clinical outcomes of patients in the
CRC and ECM groups.
| Outcome | CRC (n=59) | % | ECM (n=9) | % | P-value |
|---|
| Technical
success | 58 | 98.3 | 9 | 100.0 | NS |
| Clinical
success | 57 | 96.6 | 6 | 66.7 | 0.015 |
| CROSS | | | | | |
|
Before stent
placement | 0.508±1.006 | | 0.222±0.441 | | 0.406 |
|
After stent
placement | 3.881±0.590 | | 2.778±1.856 | | <0.001 |
| Complication | | | | | |
|
All | 15 | 25.4 | 6 | 66.7 | 0.021 |
|
Perforation | 4 | 6.8 | 2 | 22.2 | 0.177 |
|
Migration | 4 | 6.8 | 0 | 0.0 | NS |
|
Reobstruction | 10 | 16.9 | 6 | 66.7 | 0.004 |
|
Emergency
surgerya | 4 | 26.7 | 4 | 66.7 | 0.146 |
| | | (4/15) | | 0.146 | |
|
Endoscopic
procedureb | 5 | 33.3 | 1 | 16.7 | 0.623 |
| | | (5/15) | | (1/6) | |
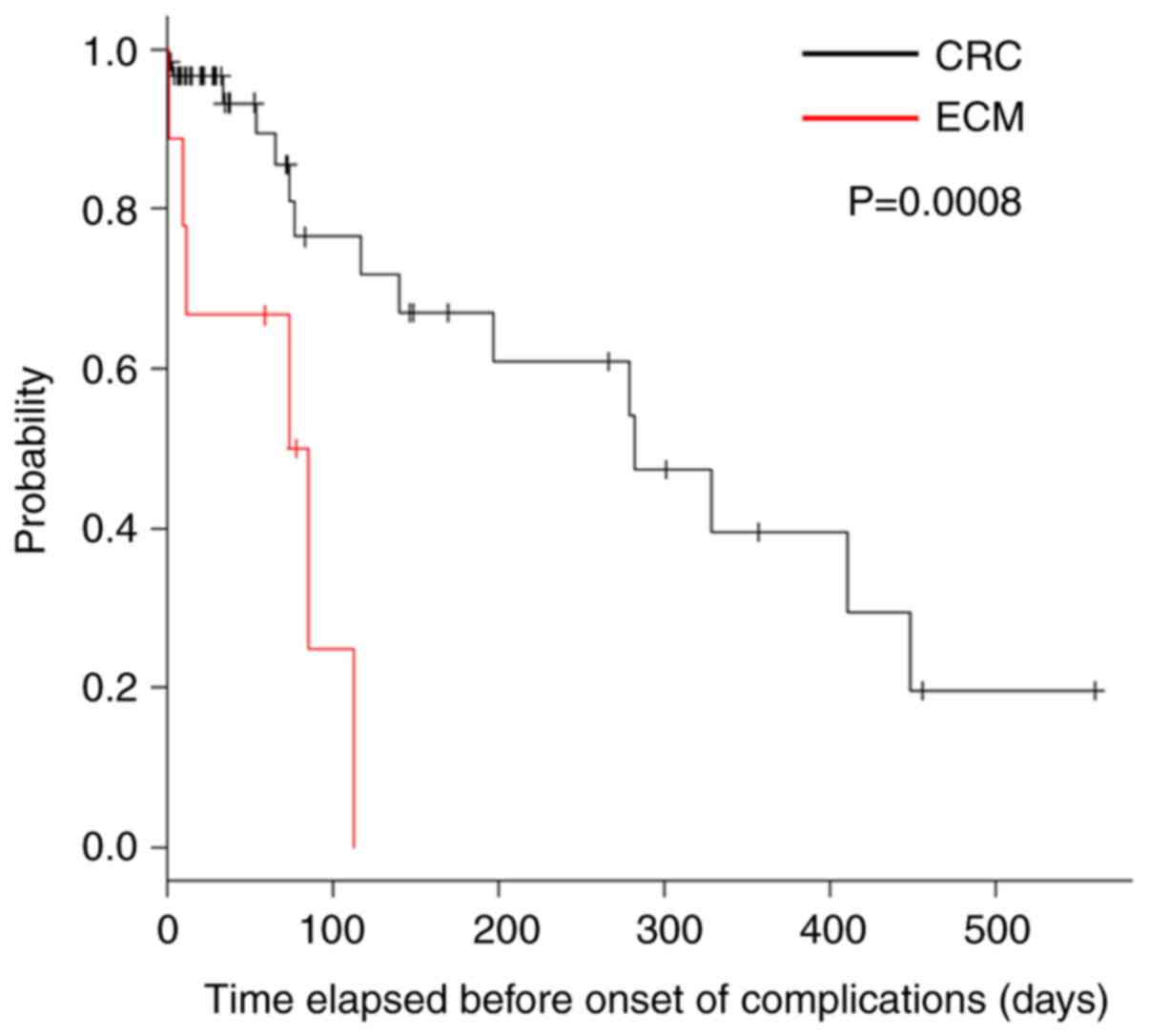
When the time elapsed before the onset of
complications was compared between the two groups, the time was
shorter in the ECM group (P=0.0008; Fig. 2). In addition, a multivariate
analysis of risk factors for re-obstruction after SEMS placement
was performed. Logistic regression analysis was performed using
ECM, palliative purpose, CROSS score post-stenting, and prior
chemotherapy as the independent variables. Risk factors for
re-obstruction were higher for ECM [odds ratio (OR): 7.76
(1.02-57.2), P=0.04] than for palliative stent placement (OR: 5.45
(1.01-29.5), P=0.05; Table
III).
 | Table IIIMultivariate analysis of risk factors
for re-obstruction. |
Table III
Multivariate analysis of risk factors
for re-obstruction.
| Risk factor | Odds ratio (95%
CI) | P-value |
|---|
| ECM | 7.76
(1.02-57.20) | 0.044 |
| Prior
chemotherapy | 1.02
(0.20-5.27) | 0.984 |
| PAL | 5.45
(1.01-29.50) | 0.049 |
| CROSS after stent
placement | 1.38
(0.69-2.78) | 0.367 |
Discussion
A systematic review reported a technical success
rate of 96.2% (range 66.6-100%) and a clinical success rate of 92%
(range 46-100%) in patients with CRC (20,29-31).
In contrast, among patients with ECM, the technical success rate
was 88.5% (range 67-100%) and the clinical success rate was 72.2%
(range 20-100%) in the 12 reviewed articles (14,15,19-21,25,32-35).
In our study, there was no difference in the technical success rate
between the CRC and ECM groups, but the clinical success rate was
significantly lower in the ECM group, which was similar to previous
reports. In addition, the incidence of complications was
significantly higher in the ECM group, and the incidence of
occlusion was particularly high. Although SEMS is preferred over
emergency surgery for colorectal obstruction, the efficacy of SEMS
in patients with malignant colorectal obstruction by ECM with
peritoneal carcinomatosis has not been demonstrated to date
(22). Compared to obstructions
associated with CRC, those caused by ECM tend to be complicated at
one or more potential locations (21). Kim et al reported no
significant difference in clinical success, complication, and stent
patency rates between the CRC and ECM groups (22). Ahn et al reported that 72
patients with colorectal obstruction caused by ECM were treated
with uncovered SEMS. The technical success rate was 90.3% with a
clinical success rate of 87.7%. However, despite the high clinical
success rate, 26.3% (15/57) of the patients ultimately required
surgery in the long term (23). The
rate of surgery in their ECM group was lower than that in our ECM
group [44.4% (4/9)], but it was higher than that in our CRC group
[7.02% (4/57)]. In addition, since Ahn et al did not compare
their data with the CRC group; it is unclear whether SEMS is
equally effective for CRC as well as ECM patients. However, we
agree with the opinion of Ahn et al that SEMS placement with
colorectal obstruction caused by ECM is effective in selected
patients considering their prognosis because SEMS is less
burdensome compared to surgery (23). Keswani et al demonstrated
that patients with ECM had a significantly lower clinical success
rate than those with CRC (94.1 vs. 20%; P<0.0001), but they have
a higher rate of complications (P=0.046). Moreover, the
multivariable analysis revealed that ECM was a predictor of
complications (20). In our study,
one patient in the CRC group developed perforation during SEMS
placement at the ascending colon when using a guidewire to traverse
the obstruction. However, both groups showed high technical success
rates. With regard to the clinical success, all patients in the CRC
group showed clinical improvement, but some patients in the ECM
group did not show clinical improvement even after successful stent
placement. In our study, re-obstruction occurred in the CRC group
at 6 months after stent placement, with a longer placement
resulting in in-growth and stool impaction. In the ECM group,
re-obstruction occurred relatively earlier after stent placement
(median patency duration: 79 days).
One patient in the CRC group developed perforation
when receiving ramucirumab treatment. Ramucirumab and bevacizumab
are anti-VEGF antibody drugs that have been reported to be
associated with the risk of gastrointestinal perforation during
stent placement (36).
Faraz et al reported that the technical and
clinical success rates are decreased in patients with peritoneal
carcinomatosis and multifocal disease. However, these factors
should not discourage attempts for stent placement, especially if
the benefits outweigh the risks (37). Furthermore, the use of SEMS should
be carefully considered for patients who respond to chemotherapy,
which results in longer survival duration, as the ECM group is
likely to have a shorter stent patency duration. In some cases,
colorectal obstruction associated with ECM did not improve after
SEMS placement. These patients presented with multiple stenoses,
intestinal stenosis, impaired bowel movement, or impaired digestive
tract motility owing to the presence of an omental cake (14,20,21).
Colorectal stents may be an excellent palliative treatment option
compared with surgery or an ileus tube. However, when complications
occur, emergency surgery is often required in the ECM group.
Therefore, careful consideration of the patient's presentation and
prognosis is required when placing a stent in a patient with ECM.
In addition, it is important that an adequate explanation is
provided to patients and families regarding potential complications
that may develop shortly after stent placement. In this study, as
in previous reports, colorectal stent placement was inferior in the
ECM group as compared to that in the CRC group in terms of safety
and efficacy. Furthermore, patients in the ECM group had a
significantly higher frequency of palliative stent placement and
chemotherapy before stent placement than those in the CRC group.
These factors were considered to be associated with a higher number
of complications. However, the results of the multivariate analysis
showed that the ECM group was a risk factor for re-obstruction.
Keswani et al reported a similar conclusion (20). This retrospective, single-centre
study, with its small number of patients, requires further
expansion.
In conclusion, in patients with ECM, SEMS placement
was associated with a lower clinical success rate and increased
risk for complications, especially re-obstruction. The time elapsed
before the onset of complications was short, and the ECM itself was
a risk factor for re-obstruction, suggesting that placement of SEMS
for malignant colorectal obstruction in ECM is not optimal.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analysed during this study are
included in this published article.
Authors' contributions
TS designed the current study and established
patient selection criteria. TS, YN, AI, MA, MI, TI, TH and TY
formed the study groups, analysed and interpreted patient data, and
wrote the manuscript. TS and AI confirm the authenticity of all the
raw data. All authors read and approved the final manuscript and
agree to be accountable for all aspects of the research in ensuring
that the accuracy or integrity of any part of the work are
appropriately investigated and resolved.
Ethics approval and consent to
participate
The current study was approved by the Standards of
the Official Conduct Committee at Saiseikai Niigata Hospital
(approval no. E17-28). All patients provided written informed
consent and the current study adhered to the principles of the
Declaration of Helsinki of 1964.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Frago R, Ramirez E, Millan M, Kriesler E,
del Valle E and Biondo S: Current management of acute malignant
large bowel obstruction: A systematic review. Am J Surg.
207:127–138. 2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Pisano M, Zorcolo L, Merli C, Cimbanassi
S, Poiasina E, Ceresoli M, Agresta F, Allievi N, Bellanova G,
Coccolini F, et al: 2017 WSES guidelines on colon and rectal cancer
emergencies: Obstruction and perforation. World J Emerg Surg.
13(36)2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Cheynel N, Cortet M, Lepage C, Benoit L,
Faivre J and Bouvier AM: Trends in frequency and management of
obstructing colorectal cancers in a well-defined population. Dis
Colon Rectum. 50:1568–1575. 2007.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Deans GT, Krukowski ZH and Irwin ST:
Malignant obstruction of the left colon. Br J Surg. 81:1270–1276.
1994.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Faraz S, Salem SB, Schattner M, Mendelsohn
R, Markowitz A, Ludwig E, Zheng J, Gerdes H and Shah PM: Predictors
of clinical outcome of colonic stent in patents with malignant
large-bowel obstruction because of extracolonic malignancy.
Gastrointest Endosc. 87:1310–1317. 2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Sebastian S, Johnston S, Geoghegan T,
Torreggiani W and Buckley M: Pooled analysis of the efficacy and
safety of self-expanding metal stenting in malignant colorectal
obstruction. Am J Gastroenterol. 99:2051–2057. 2004.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Carne PW, Frye JN, Robertson GM and
Frizelle FA: Stents or open operation for palliation of colorectal
cancer: A retrospective, cohort study of perioperative outcome and
long-term survival. Dis Colon Rectum. 47:1455–1461. 2004.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Faragher IG, Chaitowitz IM and Stupart DA:
Long-term results of palliative stenting or surgery for incurable
obstructing colon cancer. Colorectal Dis. 10:668–672.
2008.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Angenete E, Asplund D, Bergström M and
Park PO: Stenting for colorectal cancer obstruction compared to
surgery-a study of consecutive patients in a single institution.
Int J Colorectal Dis. 27:665–670. 2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Dohmoto M, Rupp KD and Hohlbach G:
Endoscopically-implanted prosthesis in rectal carcinoma. Dtsch Med
Wochenschr. 115(915)1990.PubMed/NCBI(In German).
|
|
11
|
van Hooft JE, van Halsema EE, Vanbiervliet
G, Beets-Tan RG, DeWitt JM, Donellan F, Dumonceau JM, Glynne-Jones
RG, Hassan C, Jiménez-Perez J, et al: Self-expandable metal stents
for obstructing colonic and extracolonic cancer: European society
of gastrointestinal endoscopy (ESGE) clinical guideline. Endoscopy.
46:990–1053. 2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Davila RE, Rajan E, Adler D, Hirota WK,
Jacobson BC, Leighton JA, Qureshi W, Zuckerman MJ, Fanelli R,
Hambrick D, et al: ASGE guideline: The role of endoscopy in the
diagnosis, staging, and management of colorectal cancer.
Gastrointest Endosc. 61:1–7. 2005.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang Y, Hu H, Wang M, Han X, Zhang Q, Yu
L, Chen Y and Wang G: Self-expanding metallic stent as a bridge to
surgery versus emergency surgery for acute obstructive colorectal
cancer: A retrospective study. Cancer Manag Res. 11:2709–2718.
2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Kim BK, Hong SP, Heo HM, Kim JY, Hur H,
Lee KY, Cheon JH, Kim TI and Kim WH: Endoscopic stenting is not as
effective for palliation of colorectal obstruction in patients with
advanced gastric cancer as emergency surgery. Gastrointest Endosc.
75:294–301. 2012.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kim JY, Kim SG, Im JP, Kim JS and Jung HC:
Comparison of treatment outcomes of endoscopic stenting for colonic
and extracolonic malignant obstruction. Surg Endosc. 27:272–277.
2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Moon SJ, Kim SW, Lee BI, Lim CH, Kim JS,
Soo J, Park JM, Lee IS, Choi MG and Choi KY: Palliative stent for
malignant colonic obstruction by extracolonic malignancy: A
comparison with colorectal cancer. Dig Dis Sci. 59:1891–1897.
2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kim JH, Song HY, Park JH, Ye BD, Yoon YS
and Kim JC: Metallic stent placement in the palliative treatment of
malignant colonic obstructions: Primary colonic versus extracolonic
malignancies. J Vasc Interv Radiol. 22:1727–1732. 2011.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Trompetas V, Saunders M, Gossage J and
Anderson H: Shortcomings in colonic stenting to palliate large
bowel obstruction from extracolonic malignancies. Int J Colorectal
Dis. 25:851–854. 2010.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Shin SJ, Kim TI, Kim BC, Lee YV, Song SY
and Kim WH: Clinical application of self-expandable metallic stent
for treatment of colorectal obstruction caused by extrinsic
invasive tumors. Dis Colon Rectum. 51:578–583. 2008.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Keswani RN, Azar RR, Edmundowicz SA, Zhang
Q, Ammar T, Banerjee B, Early DS and Jonnalagadda SS: Stenting for
malignant colonic obstruction: A comparison of efficacy and
complications in colonic versus extracolonic malignancy.
Gastrointest Endosc. 69:675–680. 2009.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Yoshida S, Isayama H and Koike K:
Palliative self-expandable metallic stent placement for colorectal
obstruction caused by an extracolonic malignancy. Gastrointest
Interv. 3:75–79. 2014.
|
|
22
|
Kim JH, Ku YS, Jeon TJ, Park JY, Chung
J-W, Kwon KA, Park DK and Kim YJ: The efficacy of self-expanding
metal stents for malignant colorectal obstruction by noncolonic
malignancy with peritoneal carcinomatosis. Dis Colon Rectum.
56:1228–1232. 2013.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Ahn JS, Hong SN, Chang DK, Kim YH and Kim
ER: Efficacy of uncovered self-expandable metallic stent for
colorectal obstruction by extracolonic malignancy. World J
Gastrointest Oncol. 12:1005–1013. 2020.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Keymling M: Colorectal stenting.
Endoscopy. 35:234–238. 2003.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Keränen I, Lepistö A, Udd M, Halttunen J
and Kylänpää L: Stenting for malignant colorectal obstruction: A
single-center experience with 101 patients. Surg Endosc.
26:423–430. 2012.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Kanda Y: Investigation of the freely
available easy-to-use software ‘EZR’ for medical statistics. Bone
Marrow Transplant. 48:452–458. 2013.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Matsuzawa T, Ishida H, Yoshida S, Isayama
S, Kuwai T, Maetani I, Shimada M, Yamada T, Saito S, Tomita M, et
al: A Japanese prospective multicenter study of self-expandable
metal stent placement for malignant colorectal obstruction:
Short-term safety and efficacy within 7 days of stent procedure in
513 cases. Gastrointest Endosc. 82:697–707.e1. 2015.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Otsuji A, Saida Y, Enomoto T, Takabayashi
K, Nakamura Y, Katagi M, Nagao S, Watanabe R, Dotai K, Nagaoki Y,
et al: Feasibility of colorectal obstruction scoring system (CROSS)
in colonic stent cases. Prog Dig Endosc. 82:68–71. 2013.
|
|
29
|
Ribeiro IB, Bernardo WM, Martins BDC, de
Moura DTH, Baba ER, Josino IR, Miyahima NT, Coronel Cordero MA,
Visconti TAC, Ide E, et al: Colonic stent versus emergency surgery
as treatment of malignant colonic obstruction in the palliative
setting: A systematic review and meta-analysis. Endosc Int Open.
6:E558–E567. 2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Ribeiro IB, de Moura DTH, Thompson CC and
de Moura EGH: Acute abdominal obstruction: Colon stent or emergency
surgery? An evidence-based review. World J Gastrointest Endosc.
11:193–208. 2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Watt AM, Faragher IG, Griffin TT, Rieger
NA and Maddern GJ: Self-expanding metallic stents for relieving
malignant colorectal obstruction: A systematic review. Ann Surg.
246:24–30. 2007.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Pothuri B, Guirguis A, Gerdes H, Barakat
RR and Chi DS: The use of colorectal stents for palliation of
large-bowel obstruction due to recurrent gynecologic cancer.
Gynecol Oncol. 95:513–517. 2004.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Carter J, Valmader S, Dalrymple C,
Atkinson K and Young C: Management of large bowel obstruction in
advanced ovarian cancer with intraluminal stents. Gynecol Oncol.
84:176–179. 2002.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Caceres A, Zhou Q, Iasonos A, Gerdes H,
Chi DS and Barakat RR: Colorectal stents for palliation of
large-bowel obstructions in recurrent gynecologic cancer: An
updated series. Gynecol Oncol. 108:482–485. 2008.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Baraza W, Lee F, Brown S and Hurlstone DP:
Combination endo-radiological colorectal stenting: A prospective
5-year clinical evaluation. Colorectal Dis. 10:901–906.
2008.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Manes G, de Bellis M, Fuccio L, Repici A,
Masci E, Ardizzone S, Mangiavillano B, Carlino A, Rossi GB,
Occhipinti P and Cennamo V: Endoscopic palliation in patients with
incurable malignant colorectal obstruction by means of
self-expanding metal stent: Analysis of results and predictors of
outcomes in a large multicenter series. Arch Surg. 146:1157–1162.
2011.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Faraz S, Salem SB, Schattner M, Mendelsohn
R, Markowitz A, Ludwig E, Zheng J, Gerdes H and Shah PM: Predictors
of clinical outcome of colonic stents in patients with malignant
large-bowel obstruction because of extracolonic malignancy.
Gastrointest Endosc. 87:1310–1317. 2018.PubMed/NCBI View Article : Google Scholar
|