Introduction
Reports show that in Japan, the number of cancer
patients is increasing annually (1). Similarly, in the United States, the
numbers are also increasing annually and cancer deaths are second
only to heart diseases (2). Head
and neck cancer, including oral squamous cell carcinoma (OSCC), is
the sixth leading malignancy worldwide (2,3) with
squamous cell carcinoma accounting for at least 90% of all oral
malignancies (4). Radiotherapy is
an important treatment modality for OSCC because of its relatively
high radio-sensitivity and since the maintenance of oral function
and morphology is very important for maintaining the patients'
quality of life and activities of daily living (5).
Radiotherapy should be administered carefully in
order to administer high doses to only the target area and to not
administer excess radiation to other normal tissues. In order to
avoid osteoradionecrosis (ORN), it has been found to be useful to
place spacers to increase the distance between the mandible and the
irradiation site of the tongue carcinoma during radiotherapy for
interstitial brachytherapy (6,7). In
prostate cancer, while using external beam radiation therapy, the
insertion of a hydrogel into the prostate-rectal interface to
reduce significant exposure to the rectum has been shown to be
effective in preventing radiation-related complications (8,9).
Similarly, while using external irradiation for oral cancer, it has
been considered effective to wear a spacer to maintain the distance
between the adjacent normal tissue and the irradiation site of the
lesion. X-rays used during external radiation therapy are
characterised by the maximum absorbed dose when they enter the body
within an area of about 10 mm from the body surface (10). Since oral cancer is a lesion on the
surface of the body, a bolus, which has almost the same
characteristics as the human body, can be placed over the lesion to
maximize the dose at the surface layer of the lesion by utilising
the build-up effect (11). Based on
the results of previous studies (10-13),
if bolus build-up areas are not created, there may be dose
reductions near the surface and sufficient therapeutic effects may
not be achieved.
Taking into account the extensive losses of form and
function caused by resections, radiation therapy is particularly
advantageous in cases of extensive superficial oral cancers. High
energy X-rays of 4-6 MVX used for treating oral cancer have good
linearity and can easily irradiate evenly to the edge of the
irradiation field; however, the surface dose effect is low due to
the build-up effect (14). Since
oral cancer originates from the superficial mucosa and becomes
malignant, external radiation therapy must increase the surface
dose effect and cure the primary site (12,14).
To enhance the surface dose effect of external
radiotherapy, we developed a spacer with bolus material to adhere
to the primary site and used it in patients undergoing
radiotherapy. The bolus material is a water-equivalent material
(11,12) which can be adhered to and placed
according to the contour shape of the oral mucosa to complement the
build-up area. Commercially available bolus materials are about
5-10 mm thick, and by placing them on the spacer, a large and
uniform dose distribution on the mucosal surface can be achieved.
The current National Comprehensive Cancer Network Breast guidelines
state: ‘Special consideration should be given to the use of bolus
material to ensure that the skin dose is adequate’ (15,16).
Worldwide, bolus material has been used frequently in breast cancer
treatment to increase the surface dose effect (17); however, to the best of our
knowledge, there have been only two reports of its use in oral
cancer (18,19). In these case reports, the
indications were limited to tongue cancer and cancer of the palate;
however, in our case reports, we used their method at various sites
of oral cancer and reported on the clinical courses and adverse
effects such as oral mucositis, dysgeusia, and ORN.
The main purpose of the spacer is to improve the
accuracy of positioning of the irradiation site during radiotherapy
and to prevent excessive radiation to the surrounding normal
tissues, thereby preventing ORN and severe mucositis (6,20-22).
This study described a case series in which spacers
with boluses were used at various sites in the oral cavity to
enhance the therapeutic effect of radiation therapy in oral
squamous cell carcinoma.
Patients and methods
Patients
We conducted a prospective observational study among
oral cancer patients aged >18 years who underwent adequate
medical follow-up from October 2019 and January 2021 in Yamanashi
University Hospital. This study was performed in accordance with
the Declaration of Helsinki with the approval of the Ethics
Committee of Yamanashi University Hospital (no. 2352). According to
the guidelines of the Ethics Committee, all patient data were
anonymised before use and written informed consent was obtained
from each patient and/or family before inclusion in the study.
Patients were free to withdraw from the study at any time. The
inclusion criteria for this research were as follows: Adults >20
years of age, diagnoses of oral squamous cell carcinoma, and no
recent rapid exacerbations. The exclusion criteria were as follows:
Having any acute or chronic condition that would limit the ability
of the patient to participate in the study and refusal by patient
and/or family to provide informed consent. There were no exclusions
among the enrolled patients.
Five men and two women with oral cancer with an
average age of 71.1 years (range, 47-92 years) were included in
this study and they each underwent external beam radiation using a
spacer with a bolus. Table I shows
the characteristics and treatment of the seven cases including the
underlying diseases and performance statuses of the ECOGs (23). Table
II shows the adverse events according to the Common Terminology
Criteria for Adverse Events (CTCAE) guidelines, version
5.0(24) after external
radiotherapy and the histories of tobacco use and the alcohol
intake amounts before treatment. In this study, we observed whether
spacers with boluses placed on various sites of the oral cavity
would induce to intensify local adverse events such as oral
mucositis, dysgeusia, and ORN.
 | Table IPatient characteristics. |
Table I
Patient characteristics.
| Case no. | Age, years | Sex | Site | TNM | Number of cigarettes
per day | Alcohol consumption
per day, g | Underlying
disease | ECOG: PS | Treatment | Effect of
therapy |
|---|
| 1 | 87 | F | Maxillary
gingiva | T4N2M0 | Never | Never | Dementia; HT | 2 | 7 Gy x 5
fa | PD |
| 2 | 67 | M | Floor of mouth | T3N0M0 | 30 | 70 | Hepatitis B | 1 | CCRTb: 2 Gy x 35f | CR |
| 3 | 92 | F | Buccal | T4N0M0 | Never | 10 | Hyper-lipidemia;
HT | 3 | 2 Gy x 35 f | PD |
| 4 | 81 | M | Mandibular
gingiva | T4N2M0 | 20 (former) | 60 | Prostate cancer | 1 | CCRTb: 2 Gy x 35 f | CR |
| 5 | 47 | M | Tongue | rT3N0M0 | 10 | 60 | None | 1 | CCRTb: 2 Gy x 35 f | CR |
| 6 | 74 | M | Maxillary sinus | T4N0M0 | 10 | 50 | Hyper lipidemia;
HT | 1 | CCRTb: 2 Gy x 35 f | CR |
| 7 | 50 | M | Tongue | rT3N0M0 | 10 (former) | 0 | None | 1 | CCRTb: 2 Gy x 35 f | CR |
 | Table IIInvestigation of smoking and alcohol
consumption before treatment and adverse events of CTCAE ver.
5.0a after external
radiotherapy. |
Table II
Investigation of smoking and alcohol
consumption before treatment and adverse events of CTCAE ver.
5.0a after external
radiotherapy.
| Case no. | Age, years | Sex | Mucositis | Dysgeusia | Osteonecrosis of the
jaw |
|---|
| 1 | 87 | F | 2 | 1 | 1 |
| 2 | 67 | M | 2 | 2 | None |
| 3 | 92 | F | 3 | 2 | 2 |
| 4 | 81 | M | 2 | 2 | None |
| 5 | 47 | M | 2 | 2 | None |
| 6 | 74 | M | 2 | 1 | 1 |
| 7 | 50 | M | 2 | 2 | None |
Biological effective dose
The following formula were used for the calculation
of biological effective dose (BED) (7,25-27).
BED=n x d x (1+d/[α/β])
n: Number of times of radiation therapy
d: Dose of radiation per one time (Gy)
β/α: Factor of recovery on targeted tissue (In oral
cancer, this value can be approximated as 10).
In Case 1, BED=7 (Gy) x 5x1.7=59.5 (Gy) was
calculated.
In Case 2-7, BED=2 (Gy) x 35x1.2=84.0 (Gy) was
calculated.
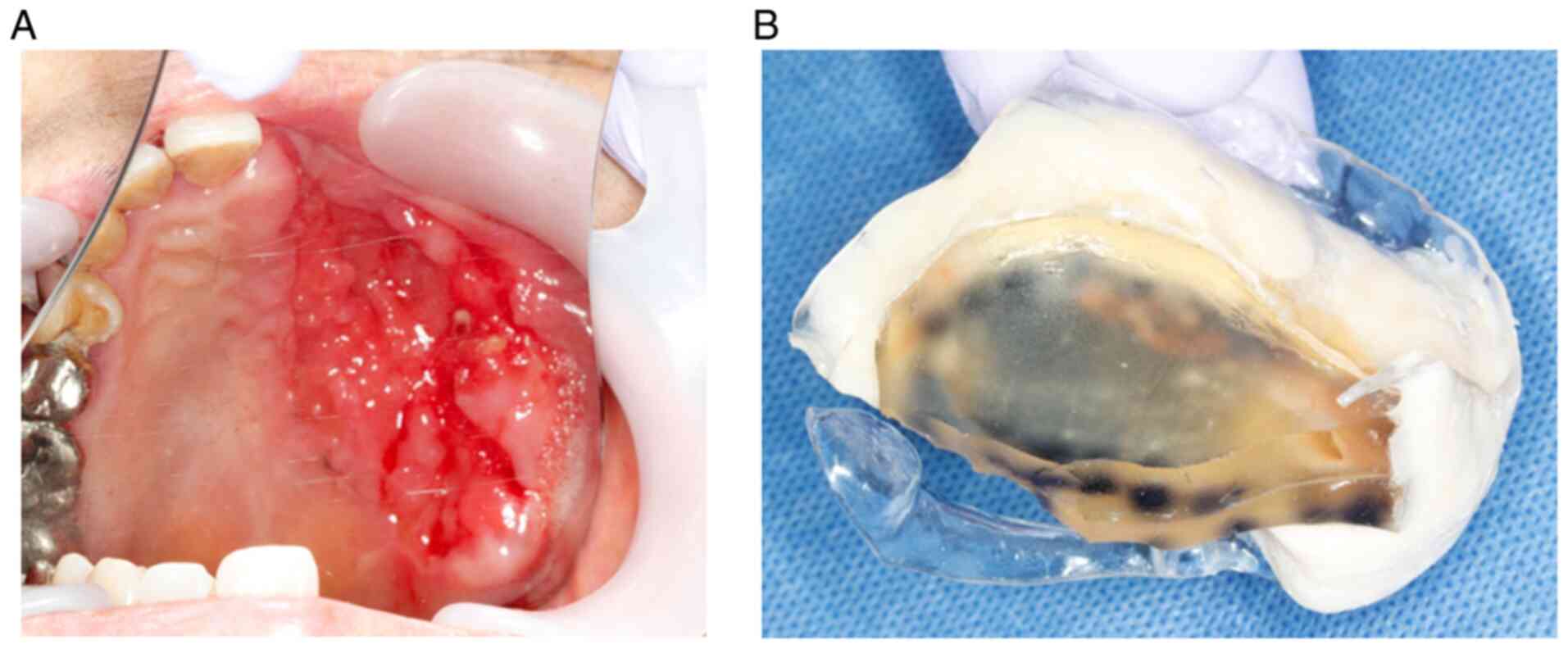
Making the spacer with a bolus
An impression of the upper and lower jaw was made
using irreversible hydrocolloid material (Algiace; Sankin Co.). A
cast of the impression was subsequently generated using hard
plaster (Newplastone; GC Co.). A 1.5-mm thick plastic disk
(Erkodur; Erkodent Co.) was pressed onto the replicated plaster
cast of the jaw using a thermoplastic former (Erkopress; Erkodent
Co.). Once the curing was completed, the plastic disk was cut-off
from the plaster cast, and the rough edges were smoothed using a
polisher. A quick self-curing resin (Ortho Crystal; Nissin Dental
Inc.) was added to the spacer to enable the bolus with a mass
density of 1.03 g/cm3 (Bolus; Toyo Medic Co.) to conform
and adhere to the irradiated area (Figs. 1 and S1). The thickness of the spacer, which
was the sum of the plastic base and bolus, was adjusted to obtain a
final thickness of ~10 mm at the equivalent radiation therapy site.
Before radiotherapy began, the spacer was set into the patient's
mouth, and we confirmed that it was painless to wear and that the
lesion could be reproducibly covered with the bolus.
Three-dimensional treatment planning was performed by a radiologist
with the spacer in the patient's mouth.
Results
Patient characteristics
Table I shows the
characteristics, treatment and its effect in each patient case. In
all cases, smoking cessation was successfully achieved, and
consuming alcohol was controlled to sobriety during/after
treatment. The overall response rate was 71.4%, with five cases
showing a complete response (CR) and two cases with progressed
disease (PD). Furthermore, as shown in Table II, osteonecrosis of the jaw as an
adverse event was ‘None’ in each of the four cases, grade 1 in two
cases, grade 2 in one case, and grade 2 or less in all the cases.
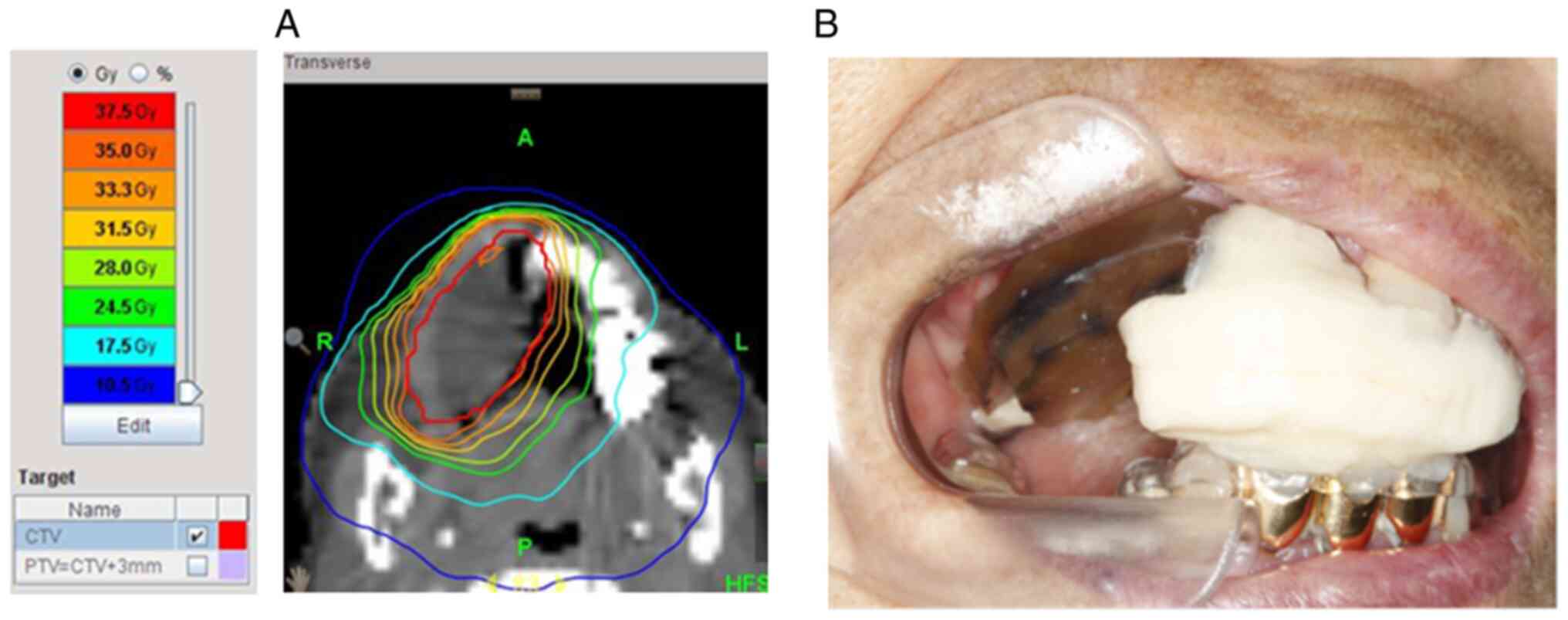
Fig. 2 shows the distribution of
radiation doses imaged during the radiation therapy planning when
the spacer was placed in the oral cavity. The bolus aligned with
the lesion, was drawn in gray and was easily identifiable because
the absorbed dose was comparable to that of water and lower than
the absorbed dose of surrounding tissue.
Clinical course of external
radiation
Except for Case 1, external radiation therapy with 4
mV X-rays consisted of 60-70 Gy/30-35 f, with a daily fraction size
of 2 Gy and five fractions per week. Case 1 was that of an elderly
patient with severe dementia, and the patient and his family
refused hospitalisation. Therefore, we decided to use a high dose
of 7 Gy per fraction with 6 mV X-rays and provide outpatient
treatment with five radiation treatments.
One year after the completion of treatment, the
patient developed recurrent symptoms, was found to have PD and died
6 months later. The other PD patient (Case 3), who was 92 years
old, was not selected for combination chemotherapy due to her poor
general condition. Six months after the completion of treatment,
she developed recurrent symptoms, her general condition
deteriorated, and she died. Each of the six patients with CR were
in a good general condition and could be treated with concurrent
cisplatin chemotherapy, which is considered to be the standard
care.
Discussion
In this study, a conventional spacer was attached to
keep the normal tissue away from the irradiation site, and bolus
material was attached to the spacer to make it about 10 mm thick to
enhance the surface dose effect at the target site.
The installation of the bolus material on the spacer
provided the following two advantages. First, since the bolus
material is a water-equivalent, gel-like material, it can be safely
fixed close to the lesion site and can assist in identifying the
area to be irradiated because it has a different absorbed dose from
other areas considered during radiotherapy planning. Second, it
increases the surface dose effect near the horizontal margins of
the oral squamous cell carcinoma, which makes it effective in
preventing recurrences in the same area. In other words, this study
was unique in two aspects: The easy identification of the target
lesion using CT with the bolus application facilitating accurate
radiotherapy planning and the enhancement of the surface dose
effect.
In contrast, since radiotherapy alone may not have
had an adequate anti-tumour effect, the combined use of cisplatin
is recommended as a curative treatment. In the two cases (Case 1
and Case 3) of recurrence in this study, the intensities of
treatment may have been too weak because radiotherapy alone was
chosen even though the patients had advanced cancer with invasions
of the jawbone. The present study suggested that spacers should be
used in combination with cisplatin for the treatment of aggressive
oral cancer. Furthermore, in Case 1, the treatment effect may have
been insufficient because the irradiation dose was 59.5 Gy (BED
equivalent), which is considered somewhat low for a curative dose
due to the choice of small fractional irradiation.
Limitations of the present study were the small
number of cases and the short follow-up periods. Since there is a
possibility that ORN may occur later than several years, it is
necessary to prevent ORN by maintaining thorough oral hygiene
during the follow-ups. It is recommended that future studies
increase the number of cases and further analyse the treatment
effect in prospective multicentre studies to confirm the
superiority of the spacer with bolus.
In conclusion, we showed that spacers with boluses
can be placed at various sites of the oral cavity and that the
osteonecrosis of the jaw as an adverse event was less than grade 2.
This treatment method requires collaboration between dental
surgeons and radiologists and may lead to a reduction in adverse
events associated with radiotherapy.
Supplementary Material
Spacer with bolus in Case 2. (A) An
image of the carcinoma of the oral floor. (B) Spacer with bolus
adjusted to adhere to the lesion site. (C) Spacer with bolus is
shown mounted in the mouth. (D) A image with complete response
obtained after radiotherapy using the applied spacer.
Acknowledgements
The authors would like to thank Professor Hiroshi
Onishi (Department of Radiology, School of Medicine, University of
Yamanashi, Chuo, Japan), for his valuable advice for radiotherapy
in clinical conference for each case.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analysed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
KY, KM, SA and MM conceived and designed the study,
and conducted data collection. KY, AM and KU researched the
literature, and performed the analysis of the data. KM, SA, MM, AM
and KU confirmed the authenticity of all the raw data. All authors
contributed to the drafting of the manuscript and critically
revised the manuscript. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Yamanashi University (approval no. 2352; Chuo,
Yamanashi, Japan). The present study was conducted in accordance
with the Declaration of Helsinki. According to the guidelines of
the Ethics Committee, all the patient data were anonymised before
use. Written informed consent was obtained from each patient and/or
the family before inclusion in the study.
Patient consent for publication
Written informed consent for publication of the oral
photographs in Figs. 1 and 2 and S1
was obtained from each patient or the family.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ministry of Health Labour and Welfare:
Ministry of Health, Labour and Welfare (In Japanese). https://www.mhlw.go.jp/bunya/kenkou/gan_toukei.html.
Accessed January 13, 2021.
|
|
2
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2020. CA Cancer J Clin. 70:7–30. 2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Massano J, Regateiro FS, Januário G and
Ferreira A: Oral squamous cell carcinoma: Review of prognostic and
predictive factors. Oral Surg Oral Med Oral Pathol Oral Radiol
Endod. 102:67–76. 2006.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Lin A: Radiation therapy for oral cavity
and oropharyngeal cancers. Dent Clin North Am. 62:99–109.
2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Yuasa K, Kawazu T, Morita M, Uehara S,
Kunitake N and Kanda S: A new, simple method of making a spacer in
interstitial brachytherapy for mobile tongue cancer. Oral Surg Oral
Med Oral Pathol Oral Radiol Endod. 89:519–521. 2000.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Miura M, Takeda M, Sasaki T, Inoue T,
Nakayama T, Fukuda H, Hoshi A, Hoshina M and Shibuya H: Factors
affecting mandibular complications in low dose rate brachytherapy
for oral tongue carcinoma with special reference to spacer. Int J
Radiat Oncol Biol Phys. 41:763–770. 1998.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Hamstra DA, Mariados N, Sylvester J, Shah
D, Karsh L, Hudes R, Beyer D, Kurtzman S, Bogart J, His RA, et al:
Continued benefit to rectal separation for prostate radiation
therapy: Final results of a phase III trial. Int J Radiat Oncol
Biol Phys. 97:976–985. 2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Susil RC, McNutt TR, DeWeese TL and Song
D: Effects of prostate-rectum separation on rectal dose from
external beam radiotherapy. Int J Radiat Oncol Biol Phys.
76:1251–1258. 2010.PubMed/NCBI View Article : Google Scholar
|
|
10
|
McKenna MG, Chen XG, Altschuler MD and
Bloch P: Calculation of the dose in the build-up region for high
energy photon beam. Treatment planning when beam spoilers are
employed. Radiother Oncol. 34:63–68. 1995.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Sroka M, Reguła J and Lobodziec W: The
influence of the bolus-surface distance on the dose distribution in
the build-up region. Rep Pract Oncol Radiother. 15:161–164.
2010.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Günhan B, Kemikler G and Koca A:
Determination of surface dose and the effect of bolus to surface
dose in electron Beams. Med Dosim. 28:193–198. 2003.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Chu JC, Coia LR, Aziz D and Stafford PM:
Dose to superficial node for patients with head and neck cancer
treated with 6 MV and 60Co photons. Radiother Oncol. 21:257–260.
1991.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Kudchadker RJ, Antolak JA, Morrison WH,
Wong PF and Hogstrom KR: Utilization of custom electron bolus in
head and neck radiotherapy. J Appl Clin Med Physics. 4:321–333.
2003.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Gradishar WJ, Anderson BO, Abraham J, Aft
R, Agnese D, Allison KH, Blair SL, Burstein HJ, Dang C, Elias AD,
et al: Breast cancer, version 3.2020, NCCN clinical practice
guidelines in oncology. J Natl Compr Canc Netw. 18:452–478.
2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Recht A, Edge SB, Solin LJ, Robinson DS,
Estabrook A, Fine RE, Fleming GF, Formenti S, Hudis C, Kirshner JJ,
et al: Postmastectomy radiotherapy: Clinical practice guidelines of
the American Society of Clinical Oncology. J Clin Oncol.
19:1539–1569. 2001.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Vu TT, Pignol JP, Rakovitch E, Spayne J
and Paszat L: Variability in radiation oncologists' opinion on the
indication of a bolus in post-mastectomy radiotherapy: An
international survey. Clin Oncol (R Coll Radiol). 19:115–119.
2007.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Baek S, Ahn S, Ju E and Jung NH:
Customized 3D bolus applied to the oral cavity and supraclavicular
area for head and neck cancer. In Vivo. 35:579–584. 2021.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Nam KY: Radiation prosthetic stents
applied to oral cancer patients during the radiation therapy: Case
reports. J Dental Rehabil Appl Sci. 36:282–288. 2020.(In
Korean).
|
|
20
|
Bedwinek JM, Shukovsky LJ, Fletcher GH and
Daley TE: Osteonecrosis in patients treated with definitive
radiotherapy for squamous cell carcinomas of the oral cavity and
Naso-and oropharynx. Radiology. 119:665–667. 1976.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Curi MM and Dib LL: Osteoradionecrosis of
the jaws: A retrospective study of the background factors and
treatment in 104 cases. J Oral Maxillofac Surg. 55:540–546.
1997.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Obinata K, Ohmori K, Tuchiya K, Nishioka
T, Shirato H and Nakamura M: Clinical study of a spacer to help
prevent osteoradionecrosis resulting from brachytherapy for tongue
cancer. Oral Surg Oral Med Oral Pathol Oral Radiol Endod.
95:246–250. 2003.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the Eastern Cooperative Oncology Group. Am J Clin
Oncol. 5:649–655. 1982.PubMed/NCBI
|
|
24
|
National Cancer Institute: Common
Terminology Criteria for Adverse Events (CTCAE) guidelines, version
5.0. 2017. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_8.5x11.pdf2017.
Accessed January 14, 2021.
|
|
25
|
Dale RG: The application of the
linear-quadratic dose-effect equation to fractionated and
protracted radiotherapy. Br J Radiol. 58:515–528. 1985.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Bentzen SM, Dörr W, Gahbauer R, Howell RW,
Joiner MC, Jones B, Jones DT, van der Kogel AJ, Wambersie A and
Whitmore G: Bioeffect modeling and equieffective dose concepts in
radiation oncology-terminology, quantities and units. Radiother
Oncol. 105:266–268. 2012.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Dutreix J: Expression of the dose rate
effect in clinical curietherapy. Radiother Oncol. 15:25–37.
1989.PubMed/NCBI View Article : Google Scholar
|