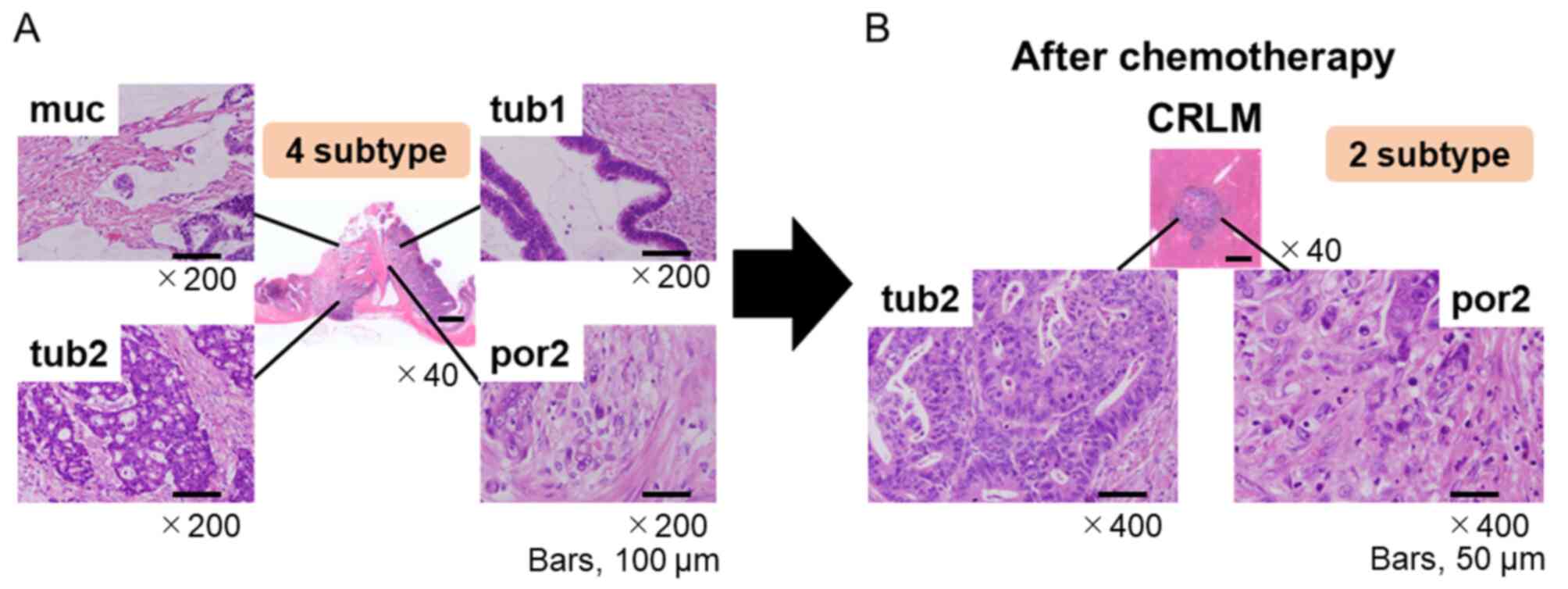
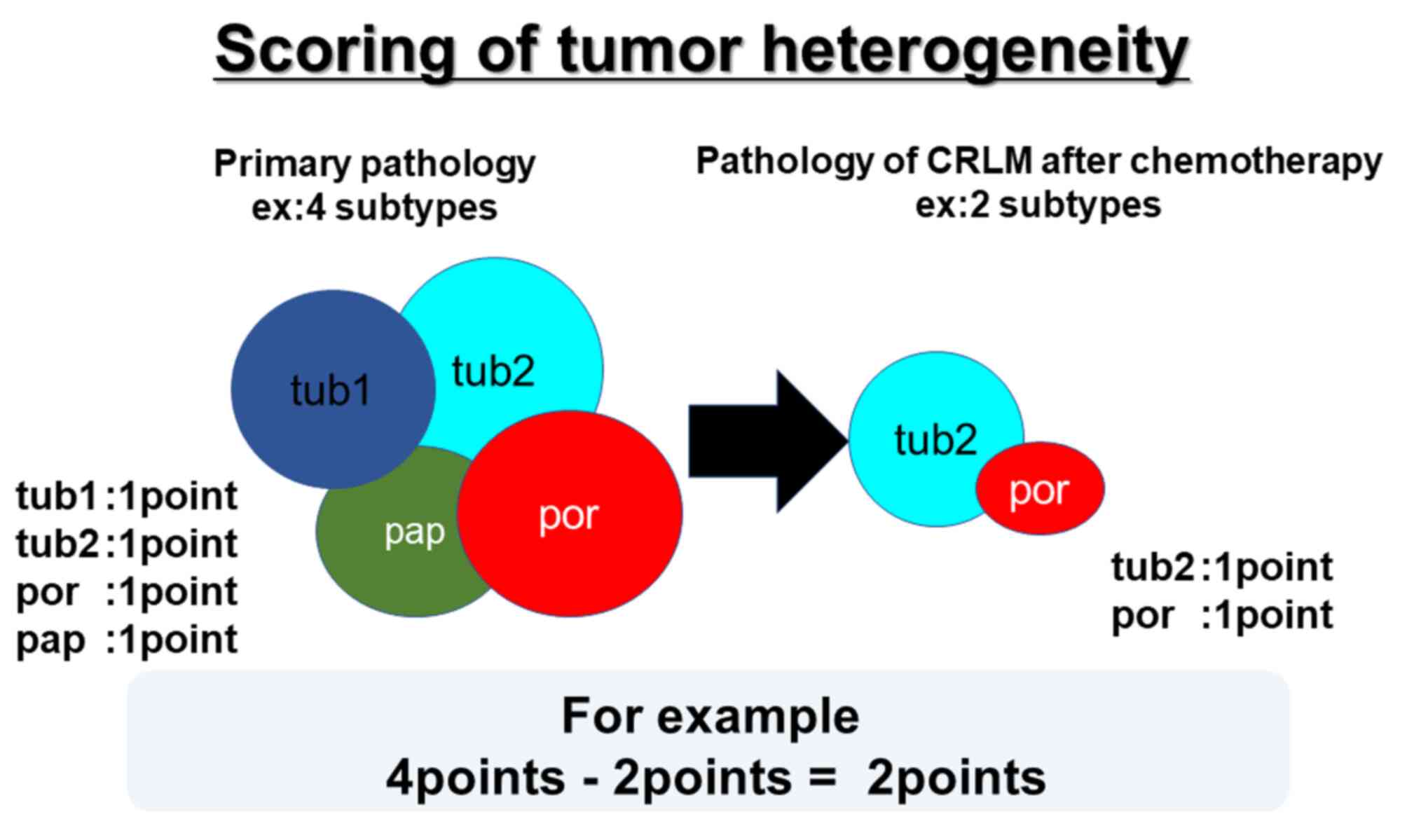
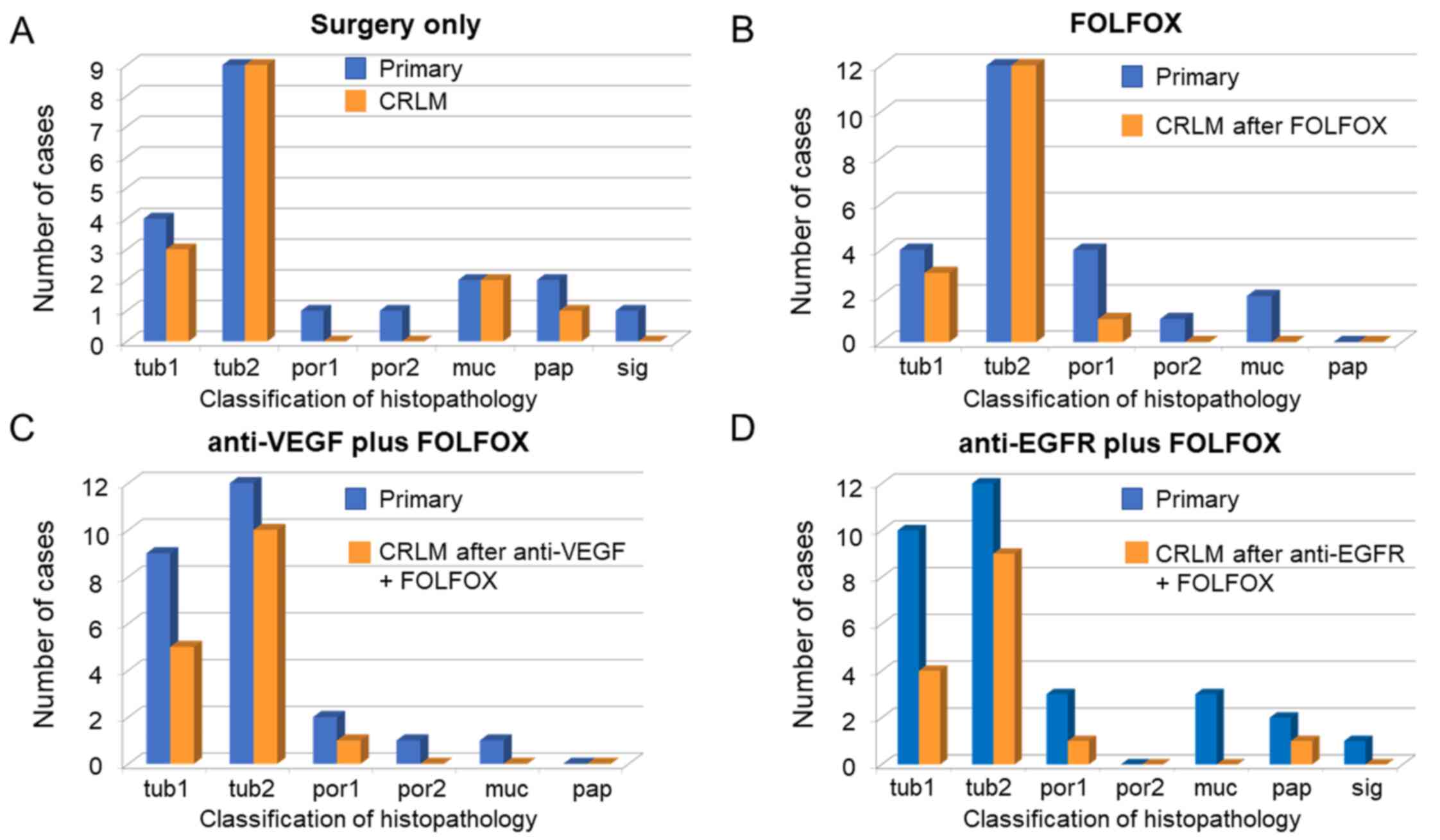
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Cancer Statistics in Japan-2018.
Foundation for Promotion of Cancer Research. Available from:
https://ganjoho.jp/reg_stat/statistics/brochure/backnumber/2018_jp.html.
|
|
3
|
NCCN GUIDELINES FOR PATIENTS 2018.
Available from: https://www.nccn.org/patients/guidelines/content/PDF/colon-patient.pdf.
|
|
4
|
Saltz LB, Clarke S, Díaz-Rubio E,
Scheithauer W, Figer A, Wong R, Koski S, Lichinitser M, Yang TS,
Rivera F, et al: Bevacizumab in combination with oxaliplatin-based
chemotherapy as first-line therapy in metastatic colorectal cancer:
A randomized phase III study. J Clin Oncol. 26:2013–2019.
2008.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumors:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Van Cutsem E, Köhne CH, Hitre E, Zaluski
J, Chang Chien CR, Makhson A, D'Haens G, Pintér T, Lim R, Bodoky G,
et al: Cetuximab and chemotherapy as initial treatment for
metastatic colorectal cancer. N Engl J Med. 360:1408–1417.
2009.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Chua TC, Saxena A, Liauw W, Kokandi A and
Morris DL: Systematic review of randomized and nonrandomized trials
of the clinical response and outcomes of neoadjuvant systemic
chemotherapy for resectable colorectal liver metastases. Ann Surg
Oncol. 17:492–501. 2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Nordlinger B, Van Cutsem E, Gruenberger T,
Glimelius B, Poston G, Rougier P, Sobrero A and Ychou M: European
Colorectal Metastases Treatment Group; Sixth International
Colorectal Liver Metastases Workshop. Combination of surgery and
chemotherapy and the role of targeted agents in the treatment of
patients with colorectal liver metastases: Recommendations from an
expert panel. Ann Oncol. 20:985–992. 2009.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Rubbia-Brandt L: Hepatic lesions induced
by systemic chemotherapy for digestive cancer. Ann Pathol.
30:421–425. 2010.PubMed/NCBI View Article : Google Scholar : (In French).
|
|
10
|
Rubbia-Brandt L, Giostra E, Brezault C,
Roth AD, Andres A, Audard V, Sartoretti P, Dousset B, Majno PE,
Soubrane O, et al: Importance of histological tumor response
assessment in predicting the outcome in patients with colorectal
liver metastases treated with neo-adjuvant chemotherapy followed by
liver surgery. Ann Oncol. 18:299–304. 2007.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Gruenberger B, Tamandl D, Schueller J,
Scheithauer W, Zielinski C, Herbst F and Gruenberger T:
Bevacizumab, capecitabine, and oxaliplatin as neoadjuvant therapy
for patients with potentially curable metastatic colorectal cancer.
J Clin Oncol. 26:1830–1835. 2008.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Gruenberger B, Schueller J, Heubrandtner
U, Wrba F, Tamandl D, Kaczirek K, Roka R, Freimann-Pircher S and
Gruenberger T: Cetuximab, gemcitabine, and oxaliplatin in patients
with unresectable advanced or metastatic biliary tract cancer: A
phase 2 study. Lancet Oncol. 11:1142–1148. 2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Poultsides GA, Bao F, Servais EL,
Hernandez-Boussard T, Dematteo RP, Allen PJ, Fong Y, Kemeny NE,
Saltz LB, Klimstra DS, et al: Pathologic response to preoperative
chemotherapy in colorectal liver metastases: Fibrosis, not
necrosis, predicts outcome. Ann Surg Oncol. 19:2797–2804.
2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Klinger M, Tamandl D, Eipeldauer S, Hacker
S, Herberger B, Kaczirek K, Dorfmeister M, Gruenberger B and
Gruenberger T: Bevacizumab improves pathological response of
colorectal cancer liver metastases treated with XELOX/FOLFOX. Ann
Surg Oncol. 17:2059–2065. 2010.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wicherts DA, de Haas RJ, Sebagh M, Saenz
Corrales E, Gorden DL, Levi F, Paule B, Azoulay D, Castaing D and
Adam R: Impact of bevacizumab on functional recovery and histology
of the liver after resection of colorectal metastases. Br J Surg.
98:399–407. 2011.PubMed/NCBI View
Article : Google Scholar
|
|
16
|
Chun YS, Vauthey JN, Boonsirikamchai P,
Maru DM, Kopetz S, Palavecino M, Curley SA, Abdalla EK, Kaur H,
Charnsangavej C and Loyer EM: Association of computed tomography
morphologic criteria with pathologic response and survival in
patients treated with bevacizumab for colorectal liver metastases.
JAMA. 302:2338–2344. 2009.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Gruenberger T, Arnold D and Rubbia-Brandt
L: Pathologic response to bevacizumab-containing chemotherapy in
patients with colorectal liver metastases and its correlation with
survival. Surg Oncol. 21:309–315. 2012.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Schwartzberg LS, Rivera F, Karthaus M,
Fasola G, Canon JL, Hecht JR, Yu H, Oliner KS and Go WY: PEAK: A
randomized, multicenter phase II study of panitumumab plus modified
fluorouracil, leucovorin, and oxaliplatin (mFOLFOX6) or bevacizumab
plus mFOLFOX6 in patients with previously untreated, unresectable,
wild-type KRAS exon 2 metastatic colorectal cancer. J Clin Oncol.
32:2240–2247. 2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Heinemann V, von Weikersthal LF, Decker T,
Kiani A, Vehling-Kaiser U, Al-Batran SE, Heintges T, Lerchenmüller
C, Kahl C, Seipelt G, et al: FOLFIRI plus cetuximab versus FOLFIRI
plus bevacizumab as first-line treatment for patients with
metastatic colorectal cancer (FIRE-3): A randomised, open-label,
phase 3 trial. Lancet Oncol. 15:1065–1075. 2014.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Venook AP, Niedzwiecki D, Lenz HJ,
Innocenti F, Fruth B, Meyerhardt JA, Schrag D, Greene C, O'Neil BH,
Atkins JN, et al: Effect of first-line chemotherapy combined with
cetuximab or bevacizumab on overall survival in patients with KRAS
wild-type advanced or metastatic colorectal cancer: A randomized
clinical trial. JAMA. 317:2392–2401. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Oki E, Emi Y, Yamanaka T, Uetake H, Muro
K, Takahashi T, Nagasaka T, Hatano E, Ojima H, Manaka D, et al:
Randomised phase II trial of mFOLFOX6 plus bevacizumab versus
mFOLFOX6 plus cetuximab as first-line treatment for colorectal
liver metastasis (ATOM trial). Br J Cancer. 121:222–229.
2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Falcão D, Alexandrino H, Caetano Oliveira
R, Martins J, Ferreira L, Martins R, Serôdio M, Martins M, Tralhão
JG, Cipriano MA, et al: Histopathologic patterns as markers of
prognosis in patients undergoing hepatectomy for colorectal cancer
liver metastases-Pushing growth as an independent risk factor for
decreased survival. Eur J Surg Oncol. 44:1212–1219. 2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Li H, Courtois ET, Sengupta D, Tan Y, Chen
KH, Goh JJL, Kong SL, Chua C, Hon LK, Tan WS, et al: Reference
component analysis of single-cell transcriptomes elucidates
cellular heterogeneity in human colorectal tumors. Nat Genet.
49:708–718. 2017.PubMed/NCBI View
Article : Google Scholar
|
|
24
|
Folprecht G, Gruenberger T, Bechstein WO,
Raab HR, Lordick F, Hartmann JT, Lang H, Frilling A, Stoehlmacher
J, Weitz J, et al: Tumor response and secondary resectability of
colorectal liver metastases following neoadjuvant chemotherapy with
cetuximab: The CELIM randomised phase 2 trial. Lancet Oncol.
11:38–47. 2010.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Rivera F, Karthaus M, Hecht JR, Sevilla I,
Forget F, Fasola G, Canon JL, Guan X, Demonty G and Schwartzberg
LS: Final analysis of the randomised PEAK trial: Overall survival
and tumor responses during first-line treatment with mFOLFOX6 plus
either panitumumab or bevacizumab in patients with metastatic
colorectal carcinoma. Int J Colorectal Dis. 32:1179–1190.
2017.PubMed/NCBI View Article : Google Scholar
|