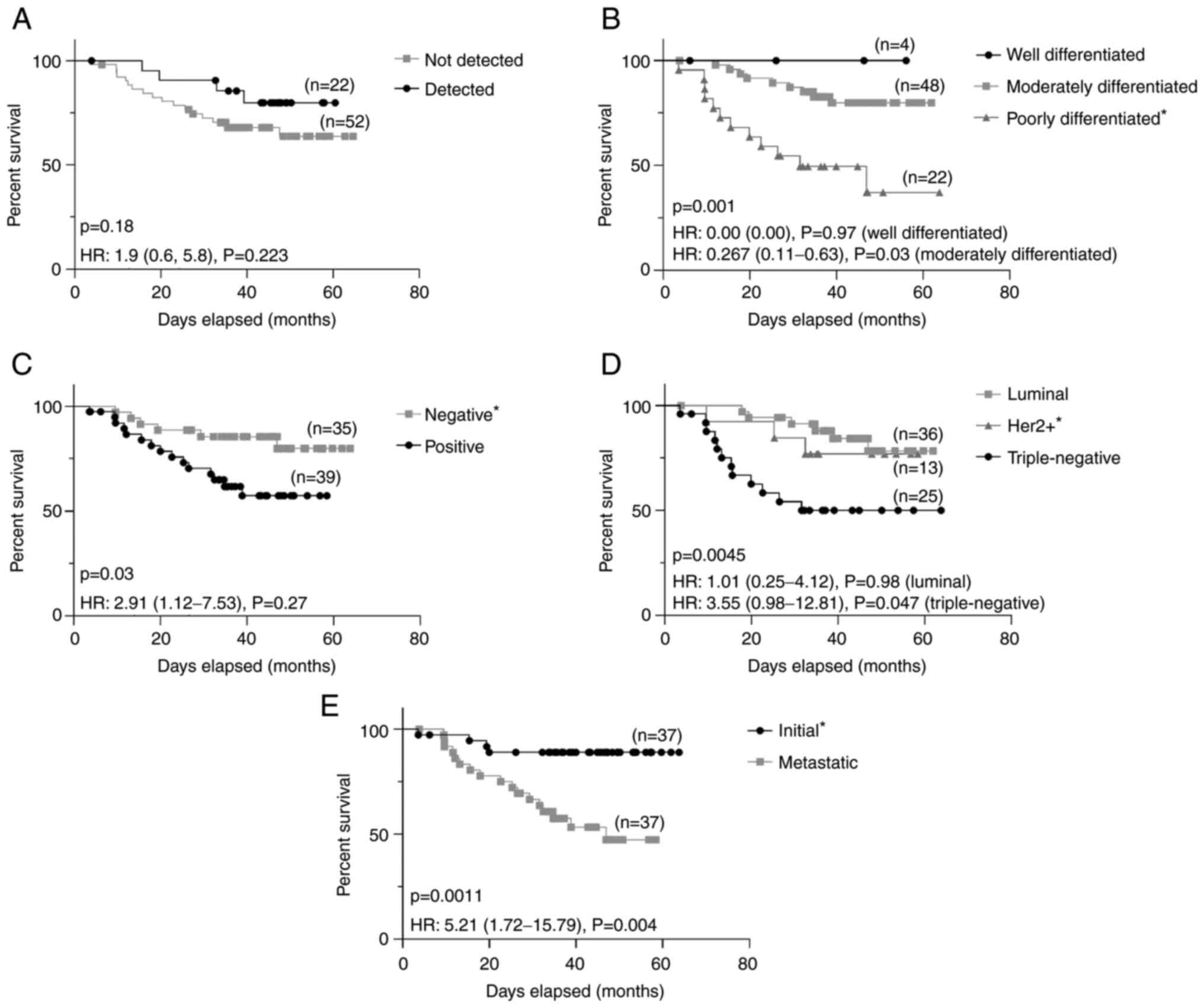
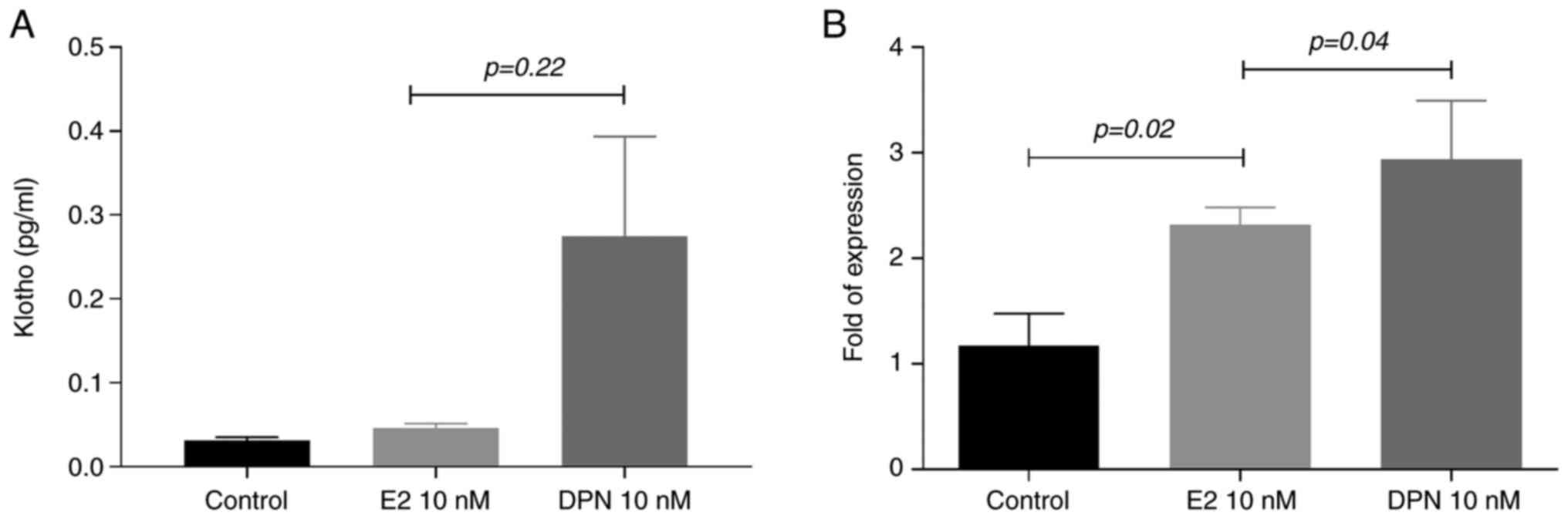
|
1
|
Kurosu H, Yamamoto M, Clark JD, Pastor JV,
Nandi A, Gurnani P, McGuinness OP, Chikuda H, Yamaguchi M,
Kawaguchi H, et al: Suppression of aging in mice by the hormone
Klotho. Science. 309:1829–1833. 2005.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kuro-o M, Matsumara Y, Aizawa H, Kawaguchi
H, Suga T, Utsugi T, Ohyama Y, Kurabayashi M, Kaname T, Kume E, et
al: Mutation of the mouse Klotho gene leads to a syndrome
resembling ageing. Nature. 390:45–51. 1997.PubMed/NCBI View
Article : Google Scholar
|
|
3
|
Nabeshima YI: Klotho: A fundamental
regulator of aging. Ageing Res Rev. 1:627–638. 2002.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Sopjani M and Dërmaku-Sopjani M:
Klotho-dependent cellular transport regulation. Vitam Horm.
101:59–84. 2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kharitonenkov A: FGFs and metabolism. Curr
Opin Pharmacol. 9:805–810. 2009.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Urakawa I, Yamazaki Y, Shimada T, Iijima
K, Hasegawa H, Okawa K, Fujita T, Fukumoto S and Yamashita T:
Klotho converts canonical FGF receptor into a specific receptor for
FGF23. Nature. 444:770–774. 2006.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kurosu H, Ogawa Y, Miyoshi M, Yamamoto M,
Nandi A, Rosenblatt KP, Baum MG, Schiavi S, Hu MC, Moe OW and
Kuro-o M: Regulation of fibroblast growth factor-23 signaling by
Klotho. J Biol Chem. 281:6120–6123. 2006.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Ito S, Kinoshita S, Shiraishi N, Nakagawa
S, Sekine S, Fujimori T and Nabeshima YI: Molecular cloning and
expression analyses of mouse betaklotho, which encodes a novel
Klotho family protein. Mech Dev. 98:115–119. 2000.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Ito S, Fujimori T, Hayashizaki Y and
Nabeshima Y: Identification of a novel mouse membrane-bound family
1 glycosidase-like protein, which carries an atypical active site
structure. Biochim Biophys Acta. 1576:341–345. 2002.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Kim JH, Hwang KH, Park KS, Kong ID and Cha
SK: Biological role of anti-aging protein Klotho. J Lifestyle Med.
5:1–6. 2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Chen CD, Tung TV, Liang J, Zeldich E,
Tucker Zhou TB, Turk BE and Abraham CR: Identification of cleavage
sites leading to the shed form of the anti-aging protein Klotho.
Biochemistry. 53:5579–5587. 2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Chen CD, Podvin S, Gillespie E, Leeman SE
and Abraham CR: Insulin stimulates the cleavage and release of the
extracellular domain of Klotho by ADAM10 and ADAM17. Proc Natl Acad
Sci USA. 104:19796–19801. 2007.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Bloch L, Sineshchekova O, Reichenbach D,
Reiss K, Saftig P, Kuro-o M and Kaether C: Klotho is a substrate
for alpha-, beta- and gamma-secretase. FEBS Lett. 583:3221–3224.
2009.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Akimoto T, Yoshizawa H, Watanabe Y, Numata
A, Yamazaki T, Takeshima E, Iwazu K, Komada T, Otani N, Morishita
Y, et al: Characteristics of urinary and serum soluble Klotho
protein in patients with different degrees of chronic kidney
disease. BMC Nephrol. 13(155)2012.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Matsumura Y, Aizawa H, Shiraki-lida T,
Nagai R, Kuro-o M and Nabeshima Y: Identification of the human
Klotho gene and its two transcripts encoding membrane and secreted
Klotho protein. Biochem Biophys Res Commun. 242:626–630.
1998.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Imura A, Tsuji Y, Murata M, Maeda R,
Kubota K, Iwano A, Obuse C, Togashi K, Tominaga M, Kita N, et al:
Alpha-klotho as a regulator of calcium homeostasis. Science.
316:1615–1618. 2007.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Xuan NT and Hai N: Changes in expression
of Klotho affect physiological processes, diseases, and cancer.
Iran J Basic Med Sci. 21:3–8. 2018.PubMed/NCBI
|
|
18
|
Zhou X and Wang X: Klotho: A novel
biomarker for cancer. J Cancer Res Clin Oncol. 141:961–969.
2015.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Perri F, Longo F, Giuliano M, Sabbatino F,
Favia G, Ionna F, Addeo R, Della Vittoria Scarpati G, Di Lorenzo G
and Pisconti S: Epigenetic control of gene expression: Potential
implications for cancer treatment. Crit Rev Oncol Hematol.
111:166–172. 2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Chen B, Ma X, Liu S, Zhao W and Wu J:
Inhibition of lung cancer cells growth, motility and induction of
apoptosis by Klotho, a novel secreted Wnt antagonist, in a
dose-dependent manner. Cancer Biol Ther. 13:1221–1228.
2012.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Chen TJ, Ren H, Thakur A, Yang T, Li Y,
Zhang S, Wang T and Chen M: Decreased level of Klotho contributes
to drug resistance in lung cancer cells: Involving in
Klotho-mediated cell autophagy. DNA Cell Biol. 35:751–757.
2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Wang Y, Chen L, Huang G, He D, He J, Xu W,
Zou C, Zong F, Li Y, Chen B, et al: Klotho sensitizes human lung
cancer cell line to cisplatin via PI3k/Akt pathway. PLoS One.
8(e57391)2013.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Abramovitz L, Rubinek T, Ligumsky H, Bose
S, Barshack I, Avivi C, Kaufman B and Wolf I: KL1 internal repeat
mediates Klotho tumor suppressor activities and inhibits bFGF and
IGF-I signaling in pancreatic cancer. Clin Cancer Res.
17:4254–4266. 2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Rubinek T, Shulman M, Israeli S, Bose S,
Avraham A, Zundelevich A, Evron E, Gal-Yam EN, Kaufman B and Wolf
I: Epigenetic silencing of the tumor suppressor Klotho in human
breast cancer. Breast Cancer Res Treat. 133:649–657.
2012.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Wolf I, Levanon-Cohen S, Bose S, Ligumsky
H, Sredni B, Kanety H, Kuro-o M, Karlan B, Kaufman B, Koeffler HP
and Rubinek T: Klotho: A tumor suppressor and a modulator of the
IGF-1 and FGF pathways in human breast cancer. Oncogene.
27:7094–7105. 2008.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Doi S, Zou Y, Togao O, Pastor JV, John GB,
Wang L, Shiizaki K, Gotschall R, Schiavi S, Yorioka N, et al:
Klotho inhibits transforming growth factor-beta1 (TGF-beta1)
signaling and suppresses renal fibrosis and cancer metastasis in
mice. J Biol Chem. 286:8655–8665. 2011.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Tang X, Fan Z, Wang Y, Ji G, Wang M, Lin J
and Huang S: Expression of Klotho and β-catenin in esophageal
squamous cell carcinoma, and their clinicopathological and
prognostic significance. Dis Esophagus. 29:207–214. 2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Tang X, Wang Y, Fan Z, Ji G, Wang M, Lin
J, Huang S and Meltzer SJ: Klotho: A tumor suppressor and modulator
of the Wnt/β-catenin pathway in human hepatocellular carcinoma. Lab
Invest. 96:197–205. 2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Lee J, Jeong DJ, Kim J, Lee S, Park JH,
Chang B, Jung SI, Yi L, Han Y, Yang Y, et al: The anti-aging gene
Klotho is a novel target for epigenetic silencing in human cervical
carcinoma. Mol Cancer. 9(109)2010.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Usuda J, Ichinose S, Ishizumi T, Ohtani K,
Inoue T, Saju H, Kakihana M, Kajiwara N, Uchida O, Nomura M, et al:
Klotho is a novel biomarker for good survival in resected large
cell neuroendocrine carcinoma of the lung. Lung Cancer. 72:355–359.
2011.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Yan Y, Wang Y, Xiong Y, Lin X, Zhou P and
Chen Z: Reduced Klotho expression contributes to poor survival
rates in human patients with ovarian cancer, and overexpression of
Klotho inhibits the progression of ovarian cancer partly via the
inhibition of systemic inflammation in nude mice. Mol Med Rep.
15:1777–1785. 2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kim JH, Hwang KH, Lkhagvadorj S, Jung JH,
Chung HC, Park HS, Kong ID, Eom M and Cha SK: Klotho plays a
critical role in clear cell renal cell carcinoma progression and
clinical outcome. Korean J Physiol Pharmacol. 20:297–304.
2016.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Li XX, Huang LY, Peng JJ, Liang L, Shi DB,
Zheng HT and Cai SJ: Klotho suppresses growth and invasion of colon
cancer cells through inhibition of IGF1R-mediated PI3K/AKT pathway.
Int J Oncol. 45:611–618. 2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Seo M, Kim MS, Jang A, Chung HJ, Noh Y,
Kim DH, Lee J, Ko K and Myung SC: Epigenetic suppression of the
anti-aging gene Klotho in human prostate cancer cell lines. Anim
Cells Syst (Seoul). 21:223–232. 2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Kusaba T, Okigaki M, Matui A, Murakami M,
Ishikawa K, Kimura T, Sonomura K, Adachi Y, Shibuya M, Shirayama T,
et al: Klotho is associated with VEGF receptor-2 and the transient
receptor potential canonical-1 Ca2+ channel to maintain endothelial
integrity. Proc Natl Acad Sci USA. 107:19308–19313. 2010.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Galkina E and Ley K: Vascular adhesion
molecules in atherosclerosis. Arterioscler Thromb Vasc Biol.
27:2292–2301. 2007.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Maekawa Y, Ishikawa K, Yasuda O, Oguro R,
Hanasaki H, Kida I, Takemura Y, Ohishi M, Katsuya T and Rakugi H:
Klotho suppresses TNF-alpha-induced expression of adhesion
molecules in the endothelium and attenuates NF-kappaB activation.
Endocrine. 35:341–346. 2009.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Xie B, Zhou J, Shu G, Liu DC, Zhou J, Chen
J and Yuan L: Restoration of Klotho gene expression induces
apoptosis and autophagy in gastric cancer cells: Tumor suppressive
role of Klotho in gastric cancer. Cancer Cell Int.
13(18)2013.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Shu G, Xie B, Ren F, Liu DC and Zhou J, Li
Q, Chen J, Yuan L and Zhou J: Restoration of Klotho expression
induces apoptosis and autophagy in hepatocellular carcinoma cells.
Cell Oncol (Dordr). 36:121–129. 2013.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Dalton GD, Xie J, An SW and Huang CL: New
insights into the mechanism of action of soluble Klotho. Front
Endocrinol (Lausanne). 8(323)2017.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Mencke R, Olauson H and Hillebrands JL:
Effects of Klotho on fibrosis and cancer: A renal focus on
mechanisms and therapeutic strategies. Adv Drug Deliv Rev.
121:85–100. 2017.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2018. CA Cancer J Clin. 68:7–30. 2018.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386.
2015.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2020. CA Cancer J Clin. 70:7–30. 2020.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Malhotra GK, Zhao X, Band H and Band V:
Histological, molecular and functional subtypes of breast cancers.
Cancer Biol Ther. 10:955–960. 2010.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Harbeck N, Thomssen C and Gnant M: St.
Gallen 2013: Brief preliminary summary of the consensus discussion.
Breast Care (Basel). 8:102–109. 2013.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Akaza H: TNM classification. Nihon
Hinyokika Gakkai Zasshi. 85:229–241. 1994.PubMed/NCBI View Article : Google Scholar : (In Japanese).
|
|
48
|
Brown RB and Razzaque MS: Phosphate
toxicity and tumorigenesis. Biochim Biophys Acta Rev Cancer.
1869:303–309. 2018.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Ligumsky H, Rubinek T, Merenbakh-Lamin K,
Yeheskel A, Sertchook R, Shahmoon S, Aviel-Ronen S and Wolf I:
Tumor suppressor activity of Klotho in breast cancer is revealed by
structure-function analysis. Mol Cancer Res. 13:1398–1407.
2015.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Trošt N, Peña-Llopis S, Koirala S, Stojan
J, Potts PR, Fon Tacer K and Martinez ED: γKlotho is a novel marker
and cell survival factor in a subset of triple negative breast
cancers. Oncotarget. 7:2611–2628. 2016.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Untch M, Gerber B, Harbeck N, Jackisch C,
Marschner N, Möbus V, von Minckwitz G, Loibl S, Beckmann MW,
Blohmer JU, et al: 13th st. Gallen international breast cancer
conference 2013: primary therapy of early breast cancer evidence,
controversies, consensus-opinion of a german team of experts
(Zurich 2013). Breast Care (Basel). 8:221–229. 2013.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Dembinski TC, Leung CK and Shiu RP:
Evidence for a novel pituitary factor that potentiates the
mitogenic effect of estrogen in human breast cancer cells. Cancer
Res. 45:3083–3089. 1985.PubMed/NCBI
|
|
53
|
U.S National Library of Medicine (NIH):
BLAST®: Basic local alignment search tool. National
Center for Biotechnology Information, Bethesda, MD, 2021.
|
|
54
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Ragab HM, Samy N, Afify M, El Maksoud NA
and Shaaban HM: Assessment of Ki-67 as a potential biomarker in
patients with breast cancer. J Genet Eng Biotechnol. 16:479–484.
2018.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Park S, Park S, Kim J, Ahn S, Park KH and
Lee H: Assessment of Ki-67 for predicting effective prognosis in
breast cancer subtypes. Biomed Sci Lett. 24:9–14. 2018.
|
|
57
|
Colomer R, Aranda-López I, Albanell J,
García-Caballero T, Ciruelos E, López-García MÁ, Cortés J, Rojo F,
Martín M and Palacios-Calvo J: Biomarkers in breast cancer: A
consensus statement by the Spanish society of medical oncology and
the Spanish society of pathology. Clin Transl Oncol. 20:815–826.
2018.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Kuro-o M: Klotho and aging. Biochim
Biophys Acta. 1790:1049–1058. 2009.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Kuro-o M: Klotho in health and disease.
Curr Opin Nephrol Hypertens. 21:362–368. 2012.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Arbel Rubinstein T, Shahmoon S, Zigmond E,
Etan T, Merenbakh-Lamin K, Pasmanik-Chor M, Har-Zahav G, Barshack
I, Vainer GW, Skalka N, et al: Klotho suppresses colorectal cancer
through modulation of the unfolded protein response. Oncogene.
38:794–807. 2019.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Chang B, Kim J, Jeong D, Jeong Y, Jeon S,
Jung SI, Yang Y, Kim KI, Lim JS, Kim C and Lee MS: Klotho inhibits
the capacity of cell migration and invasion in cervical cancer.
Oncol Rep. 28:1022–1028. 2012.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Sopjani M, Rinnerthaler M, Kruja J and
Dermaku-Sopjani M: Intracellular Signaling of the aging suppressor
protein Klotho. Curr Mol Med. 15:27–37. 2015.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Pako J, Bikov A, Barta I, Matsueda H,
Puskas R, Galffy G, Kerpel-Fronius A, Antus B and Horvath I:
Assessment of the circulating Klotho protein in lung cancer
patients. Pathol Oncol Res. 26:233–238. 2020.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Oh HJ, Oh H, Nam BY, You JS, Ryu DR, Kang
SW and Chung YE: The protective effect of Klotho against
contrast-associated acute kidney injury via the antioxidative
effect. Am J Physiol Renal Physiol. 317:F881–F889. 2019.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Brominska B, Gabryel P,
Jarmolowska-Jurczyszyn D, Janicka-Jedyńska M, Kluk A, Trojanowski
T, Brajer-Luftmann B, Woliński K, Czepczyński R, Gut P, et al:
Klotho expression and nodal involvement as predictive factors for
large cell lung carcinoma. Arch Med Sci. 15:1010–1016.
2019.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Soliman NA and Yussif SM: Ki-67 as a
prognostic marker according to breast cancer molecular subtype.
Cancer Biol Med. 13:496–504. 2016.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Inwald EC, Klinkhammer-Schalke M,
Hofstädter F, Zeman F, Koller M, Gerstenhauer M and Ortmann O:
Ki-67 is a prognostic parameter in breast cancer patients: Results
of a large population-based cohort of a cancer registry. Breast
Cancer Res Treat. 139:539–552. 2013.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Wu Q, Ma Q, Deng Y, Luo W, Zhao Y, Li W
and Zhou Q: Prognostic value of ki-67 in patients with resected
triple-negative breast cancer: A meta-analysis. Front Oncol.
9(1068)2019.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Bruey JM, Kantarjian H, Estrov Z, Zhang Z,
Ma W, Albitar F, Abdool A, Thomas D, Yeh C, O'Brien S and Albitar
M: Circulating Ki-67 protein in plasma as a biomarker and
prognostic indicator of acute lymphoblastic leukemia. Leuk Res.
34:173–176. 2010.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Goldhirsch A, Wood WC, Coates AS, Gelber
RD, Thürlimann B and Senn HJ: Panel members. Strategies for
subtypes-dealing with the diversity of breast cancer: Highlights of
the St. Gallen international expert consensus on the primary
therapy of early breast cancer 2011. Ann Oncol. 22:1736–1747.
2011.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Varga Z, Diebold J, Dommann-Scherrer C,
Frick H, Kaup D, Noske A, Obermann E, Ohlschlegel C, Padberg B,
Rakozy C, et al: How reliable is Ki-67 immunohistochemistry in
grade 2 breast carcinomas? A QA study of the Swiss working group of
breast- and gynecopathologists. PLoS One. 7(e37379)2012.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Polley MY, Leung SC, McShane LM, Gao D,
Hugh JC, Mastropasqua MG, Viale G, Zabaglo LA, Penault-Llorca F,
Bartlett JM, et al: An international ki67 reproducibility study. J
Natl Cancer Inst. 105:1897–1906. 2013.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Jonat W and Arnold N: Is the Ki-67
labelling index ready for clinical use? Ann Oncol. 22:500–502.
2011.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Gudlaugsson E, Skaland I, Janssen EA,
Smaaland R, Shao Z, Malpica A, Voorhorst F and Baak JP: Comparison
of the effect of different techniques for measurement of Ki67
proliferation on reproducibility and prognosis prediction accuracy
in breast cancer. Histopathology. 61:1134–1144. 2012.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Mikami Y, Ueno T, Yoshimura K, Tsuda H,
Kurosumi M, Masuda S, Horii R, Toi M and Sasano H: Interobserver
concordance of Ki67 labeling index in breast cancer: Japan breast
cancer research group Ki67 ring study. Cancer Sci. 104:1539–1543.
2013.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Christopoulos PF, Msaouel P and
Koutsilieris M: The role of the insulin-like growth factor-1 system
in breast cancer. Mol Cancer. 14(43)2015.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Oloomi M, Moazzezy N and Bouzari S:
Comparing blood versus tissue-based biomarkers expression in breast
cancer patients. Heliyon. 6(e03728)2020.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Diniz RW, Guerra MR, Cintra JR, Fayer VA
and Teixeira MT: Disease-free survival in patients with
non-metastatic breast cancer. Rev Assoc Med Bras (1992).
62:407–413. 2016.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Li X, Yang J, Peng L, Sahin AA, Huo L,
Ward KC, O'Regan R, Torres MA and Meisel JL: Triple-negative breast
cancer has worse overall survival and cause-specific survival than
non-triple-negative breast cancer. Breast Cancer Res Treat.
161:279–287. 2017.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Carter CL, Allen C and Henson DE: Relation
of tumor size, lymph node status, and survival in 24,740 breast
cancer cases. Cancer. 63:181–187. 1989.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Dai X, Li Y, Bai Z and Tang XQ: Molecular
portraits revealing the heterogeneity of breast tumor subtypes
defined using immunohistochemistry markers. Sci Rep.
5(14499)2015.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Usuda J, Ichinose S, Ishizumi T, Ohtani K,
Inoue T, Saji H, Kakihana M, Kajiwara N, Uchida O, Nomura M, et al:
Klotho predicts good clinical outcome in patients with
limited-disease small cell lung cancer who received surgery. Lung
Cancer. 74:332–337. 2011.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Zhang J, Chen X, Shen L, Wang X, Wang L,
Sun X and Qu S: The association between lymph node stage and
clinical prognosis in thyroid cancer. Front Endocrinol (Lausanne).
11(90)2020.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Nottegar A, Veronese N, Senthil M, Roumen
RM, Stubbs B, Choi AH, Verheuvel NC, Solmi M, Pea A, Capelli P, et
al: Extra-nodal extension of sentinel lymph node metastasis is a
marker of poor prognosis in breast cancer patients: A systematic
review and an exploratory meta-analysis. Eur J Surg Oncol.
42:919–925. 2016.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Li Y, Xiao H and Xue F: Overexpression of
Klotho suppresses growth and pulmonary metastasis of osteosarcoma
in vivo. Genet Mol Biol. 43(e20190229)2020.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Zhou X, Fang X, Jiang Y, Geng L, Li X, Li
Y, Lu K, Li P, Lv X and Wang X: Klotho, an anti-aging gene, acts as
a tumor suppressor and inhibitor of IGF-1R signaling in diffuse
large B cell lymphoma. J Hematol Oncol. 10(37)2017.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Lambert AW, Pattabiraman DR and Weinberg
RA: Emerging biological principles of metastasis. Cell.
168:670–691. 2017.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Ruijtenberg S and van den Heuvel S:
Coordinating cell proliferation and differentiation: Antagonism
between cell cycle regulators and cell type-specific gene
expression. Cell Cycle. 15:196–212. 2016.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Chen CD, Li H, Liang J, Hixson K, Zeldich
E and Abraham CR: The anti-aging and tumor suppressor protein
Klotho enhances differentiation of a human oligodendrocytic hybrid
cell line. J Mol Neurosci. 55:76–90. 2015.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Fan J and Sun Z: The antiaging gene Klotho
regulates proliferation and differentiation of adipose-derived stem
cells. Stem Cells. 34:1615–1625. 2016.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Ahrens HE, Huettemeister J, Schmidt M,
Kaether C and von Maltzahn J: Klotho expression is a prerequisite
for proper muscle stem cell function and regeneration of skeletal
muscle. Skelet Muscle. 8(20)2018.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Toan NK, Tai NC, Kim SA and Ahn SG:
Soluble Klotho regulates bone differentiation by upregulating
expression of the transcription factor EGR-1. FEBS Lett.
594:290–300. 2020.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Sárvári M, Kalló I, Hrabovszky E, Solymosi
N, Rodolosse A, Vastagh C, Auer H and Liposits Z: Hippocampal gene
expression is highly responsive to estradiol replacement in
middle-aged female rats. Endocrinology. 156:2632–2645.
2015.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Lojkin I, Rubinek T, Orsulic S,
Schwarzmann O, Karlan BY, Bose S and Wolf I: Reduced expression and
growth inhibitory activity of the aging suppressor Klotho in
epithelial ovarian cancer. Cancer Lett. 362:149–157.
2015.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Wolf I, Laitman Y, Rubinek T, Abramovitz
L, Novikok I, Beeri R, Kuro-O M, Koeffler HP, Catane R, Freedman
LS, et al: Functional variant of Klotho: A breast cancer risk
modifier among BRCA1 mutation carriers of Ashkenazi origin.
Oncogene. 29:26–33. 2010.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Mahmoud AM, Al-Alem U, Ali MM and Bosland
MC: Genistein increases estrogen receptor beta expression in
prostate cancer via reducing its promoter methylation. J Steroid
Biochem Mol Biol. 152:62–75. 2011.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Li Y, Chen F, Wei A, Bi F, Zhu X, Yin S,
Lin W and Cao W: Klotho recovery by genistein via promoter histone
acetylation and DNA demethylation mitigates renal fibrosis in mice.
J Mol Med (Berl). 97:541–552. 2019.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Oz OK, Hajibeigi A, Howard K, Cummins CL,
van Abel M, Bindels RJ, Word RA, Kuro-o M, Pak CY and Zerwekh JE:
Aromatase deficiency causes altered expression of molecules
critical for calcium reabsorption in the kidneys of female mice *.
J Bone Miner Res. 22:1893–1902. 2007.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Carrillo-López N, Román-García P,
Rodríguez-Rebollar A, Fernández-Martín JL, Naves-Díaz M and
Cannata-Andía JB: Indirect regulation of PTH by estrogens may
require FGF23. J Am Soc Nephrol. 20:2009–2017. 2009.PubMed/NCBI View Article : Google Scholar
|