Introduction
Recently, the use of biosimilars has been promoted
from the perspective of medical cost optimization. Biosimilar
trastuzumab, indicated for human epidermal growth factor receptor
type 2 (HER2)-positive breast cancer, was launched in 2018.
Clinically, biosimilar trastuzumab is used less frequently than
HERCEPTIN® (reference trastuzumab), but new research
(1-4)
now recognizes it as a viable treatment option along with reference
trastuzumab. However, although its equivalence/identity has been
confirmed, a biosimilar is not exactly the same compound as its
biotech predecessor, and thus monitoring for adverse events is
important regardless of the history of use of the reference
biopharmaceutical. Trastuzumab, for example, requires a
particularly careful response to infusion reactions (IR) (5) and monitoring for cardiac dysfunction
(6-8).
Clinically, it is assumed that there will be cases of i) treatment
with reference trastuzumab, ii) treatment with biosimilar
trastuzumab, and iii) switching from reference trastuzumab to
trastuzumab biosimilar. However, there is an extreme lack of
information on IR and cardiac dysfunction, especially in cases of
treatment switching.
Among the many regimens, since we assessed IR and
cardiac dysfunction, we focused on patients without concomitant use
of potentially IR-inducing biopharmaceutical pertuzumab (PER) and
without prior treatment with a potentially cardiac-inducing
anthracycline regimen. Considering this background, we decided to
focus on the patients who selected adjuvant therapy with paclitaxel
(PTX) plus trastuzumab as initial therapy.
In this study, we aimed to gain further insight into
the safety of biosimilar trastuzumab by evaluating the incidence of
IR and changes in cardiac function after switching from reference
to biosimilar trastuzumab in the first treatment PTX + trastuzumab
regimen.
Materials and methods
Patients and study design
The medical records of patients who were given PTX +
trastuzumab regimen receiving their first treatment as adjuvant
therapy at the Saitama Cancer Center (Saitama, Japan) between April
2018 and March 2022 were retrospectively reviewed. The reference
trastuzumab was HERCEPTIN® (Chugai Pharmaceutical Co.,
Ltd.), while the biosimilar trastuzumab was Trastuzumab BS for I.V
Infusion 60/150 mg ‘NK’: Trastuzumab-NK (Nippon Kayaku Co., Ltd.)
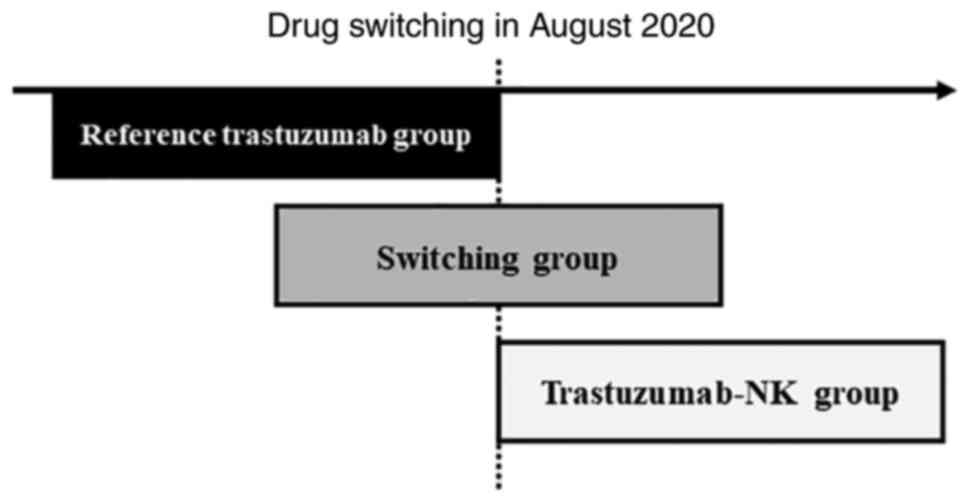
was used. We switched from reference trastuzumab to trastuzumab-NK
on August 19, 2020 (Fig. 1).
Patients were divided into three groups: a reference
trastuzumab-only group (reference trastuzumab group), a group that
switched from reference trastuzumab to trastuzumab-NK and continued
treatment (switching group), and a trastuzumab-NK-only group
(trastuzumab-NK group). Eligible patients were excluded if they had
previously received chemotherapy or were receiving PTX and
anticancer drugs other than trastuzumab. Patients were not excluded
based on the presence or absence of ER/PR, stage classification, or
radiation therapy.
Treatment
The PTX + trastuzumab regimen consisted of
trastuzumab at 8 mg/kg in 90 min for the first dose, 6 mg/kg in 30
min for the second and subsequent doses, PTX at 80 mg/m2
for 1 h, trastuzumab administered triweekly, and PTX administered
weekly. Dexamethasone 9.9 mg, d-Chlorpheniramine Maleate 5 mg, and
famotidine 20 mg were administered as premedication. This sequence
of administration of drugs is standard in our hospital.
Survey items
Baseline patient characteristics were recorded, such
as age, weight, BMI, HER2 score, left ventricular ejection fraction
(LVEF), trastuzumab dose per body weight, dosing time, and IR
expression status. For IR, the presence of Grade 1 or higher was
evaluated based on the Common Terminology Criteria for Adverse
Events (CTCAE) version 5.0. Cardiac function was assessed based on
the Guidelines for the Diagnosis and Treatment of Acute and Chronic
Heart Failure (JCS 2017/JHFS 2017), and changes were evaluated
based on LVEF test results before and at any point after treatment
initiation.
Statistical analysis
Statistical analysis was performed using JMP 14 (SAS
Institute, Cary, NC, USA). IR expression in the reference
trastuzumab group and the trastuzumab-NK group were compared using
the Fisher's exact test. P<0.05 was considered to indicate a
statistically significant difference.
Results
Baseline clinical characteristics
There were 37 patients, including 21 in the
reference trastuzumab group, 9 in the switching group, and 7 in the
trastuzumab-NK group (Table I).
There were no changes in premedication.
 | Table IBaseline patient characteristics. |
Table I
Baseline patient characteristics.
| Characteristic | Reference trastuzumab
group (n=21) | Switching group
(n=9) | Trastuzumab-NK group
(n=7) |
|---|
| Age, years | | | |
|
Mean
(SD) | 62.1 (10.6) | 63.7 (10.9) | 58.7 (13.4) |
|
Median
(range) | 65.0 (43-76) | 66.0 (47-79) | 62.0 (38-73) |
| Weight, kg | | | |
|
Mean
(SD) | 56.2 (9.2) | 52.5 (8.7) | 56.9 (7.9) |
|
Median
(range) | 54.6 (45.5-79.1) | 48.9 (41.8-63.0) | 54.0 (48.8-69.0) |
| BMI,
kg/m2 | | | |
|
Mean
(SD) | 23.0 (3.78) | 21.5 (2.88) | 23.6 (3.24) |
|
Median
(range) | 22.1 (17.5-30.6) | 22.5 (17.6-25.5) | 22.6 (20.0-28.2) |
| HER2 score | | | |
|
+2 | 4 | 0 | 0 |
|
+3 | 17 | 9 | 7 |
| Stage | | | |
|
IA | 13 | 8 | 7 |
|
IIA | 8 | 0 | 0 |
|
IIIB | 0 | 1 | 0 |
| +Radiation
therapy | 6 | 2 | 0 |
| LVEF result, % | | | |
|
Mean
(SD) | 68.3 (3.6) | 67.7 (3.9) | 66.0 (3.3) |
|
Median
(range) | 67.5 (63.0-79.0) | 69.0 (60.0-72.0) | 66.0 (62.0-71.0) |
IR expression
IR of Grade 1 or higher developed in 5 patients
(24%) in the reference trastuzumab group, 3 patients (33%) in the
switching group, and 2 patients (29%) in the trastuzumab-NK group
(Table II). Notably 3 patients in
the switching group developed IR at the first dose of reference
trastuzumab. There was no significant difference in IR expression
between the reference trastuzumab group and trastuzumab-NK group
(P>0.999). IR was characterized by the presence of fever,
chills, erythema, skin rash, pruritus, and flushing. The time of
onset was the day of or the day after the start of treatment in 7
out of 9 patients (Table
III).
 | Table IIIncidence of infusion reactions. |
Table II
Incidence of infusion reactions.
| Expression of
infusion reaction | Reference trastuzumab
group (n=21)a | Switching group
(n=9) | Trastuzumab-NK group
(n=7)a | P-valuea |
|---|
| Yes | 5 (24%) | 3 (33%) | 2 (29%) | >0.999 |
| No | 16 (76%) | 6 (67%) | 5 (71%) | |
 | Table IIIDetails of infusion reaction. |
Table III
Details of infusion reaction.
| Group | Patient number | Adverse events | Drugs | Days from start of
treatment until adverse even |
|---|
| Reference trastuzumab
Group | 1 | Pruritic | Reference
trastuzumab | 2 |
| | 2 | Chill | Reference
trastuzumab | 1 |
| | 3 | Fever | Reference
trastuzumab | 2 |
| | 4 | Pruritic | Reference
trastuzumab | 1 |
| | 5 | Eczema | Reference
trastuzumab | 141 |
| Switching group | 6 | Rash | Reference
trastuzumab | 294 |
| | 7 | Eczema | Reference
trastuzumab | 1 |
| | 8 | Stomachache | Reference
trastuzumab | 1 |
| Trastuzumab-NK
group | 9 | Flushed face | Trastuzumab-NK | 2 |
| | 10 | Fever | Trastuzumab-NK | 1 |
Cardiac function assessment
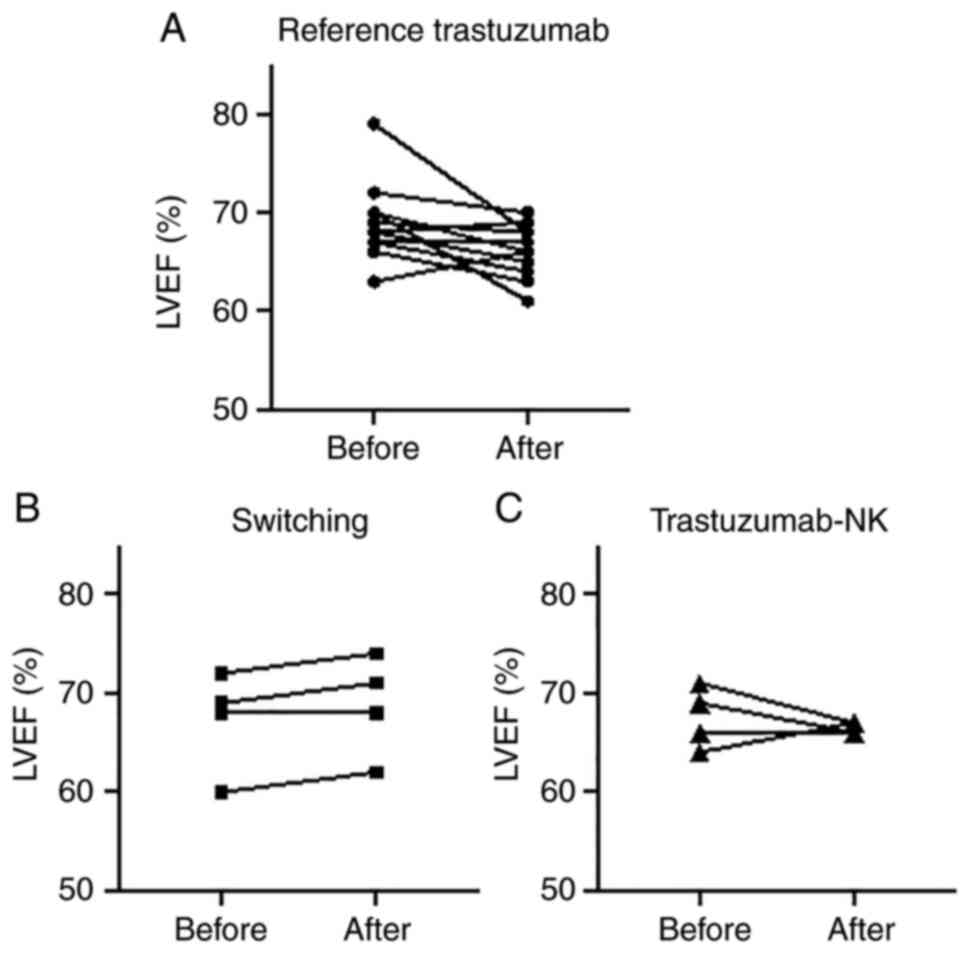
All patients had an LVEF of at least 60% before the
start of treatment and no patients had heart failure (Table I). LVEF was measured after the
start of treatment in 20 patients (reference trastuzumab group: 12
patients, switching group: 4 patients, trastuzumab-NK group: 4
patients), and no patient discontinued treatment due to a decrease
in LVEF caused by treatment (Fig.
2).
Discussion
Trastuzumab is an anti-HER2 humanized monoclonal
antibody biopharmaceutical indicated for the treatment of
HER2-positive breast cancer and advanced or recurrent unresectable
gastric cancer (9). Since breast
cancers with confirmed HER2 overexpression account for
approximately 20% of all breast cancer patients (10), it is extremely important to
accumulate safety information on trastuzumab biosimilars, a key
drug in the treatment of breast cancer. Although this is a
different trastuzumab biosimilar from the product used in the
present study, the safety information associated with this
biosimilar is reported in a study (11).
The result of this study showed no significant
difference in the frequency of IR expression between reference
trastuzumab and trastuzumab-NK (Table
II), which is similar to a previous study (12,13).
Based on these results, we confirmed IR in the switching group and
found that IR did not occur with switching, but rather only with
reference trastuzumab administration. The authors initially
expected IR to occur when switching from reference trastuzumab to
trastuzumab-NK and continuing treatment (i.e., at the first dose of
trastuzumab-NK), although IR did not occur during the switch in all
9 patients (Table II). We believe
there are two reasons for this. First is the difference between the
initial reference trastuzumab dose and the trastuzumab-NK dose at
the time of switching. One of the risk factors for IR with
trastuzumab is a high dose per body weight (14). Especially for the loading dose, an
8 mg/kg triweekly dose is reported to have a higher incidence of IR
than a 4 mg/kg weekly dose and 6 mg/kg biweekly dose (14). The initial dose of trastuzumab in
the reference trastuzumab group and the biosimilar group was 8
mg/kg, whereas the dose of trastuzumab-NK in the switching group
was 6 mg/kg. This is a lower dose than in the other two groups,
which may have been one reason why IR was not induced. Second, the
dose of trastuzumab-NK in the switching group corresponds to a
maintenance dose. In general, it is known that the frequency of IR
expression is not increased with maintenance dosing of trastuzumab
compared to the initial dose, even at higher maintenance doses
(e.g., 6 mg/kg) during triweekly and shorter durations such as 30
min (15,16). Due to these considerations, IR did
not occur even after 30 min of administration. On the other hand,
since the frequency of IR expression in trastuzumab-NK is about
11%, IR may still occur during the switch (12). However, the results of this study
suggest that IR may be less likely to occur at the time of
switching, even at the maintenance dose (e.g., 6 mg/kg). In
addition, this study found that IR was less likely to occur even if
the maintenance dosing time (30 min) was used, providing meaningful
information on dosing conditions when switching. Although dosage
and administration at the time of switching should be carefully
considered, the results of this study suggest that switching to
trastuzumab-NK in patients who have previously received reference
trastuzumab may be less likely to cause IR.
Cardiac function was then assessed using LVEF based
on the JCS 2017/JHFS 2017 guidelines. One patient in the reference
trastuzumab group exhibited a decrease in LVEF of more than 10
points before and after treatment, but LVEF was still within normal
limits, and continued treatment after the decrease in LVEF. The
normal range of LVEF in Japanese women is 64±5% (17), and heart failure below 50% is often
considered a criterion for discontinuation of treatment. A decrease
in LVEF can be influenced by multiple factors other than
trastuzumab, including a history of cardiac disease, age, known
risk factors for trastuzumab-related cardiac dysfunction (e.g.,
history of anthracycline use), and low baseline LVEF (6,18).
We believe that the inclusion of patients on the first-line PTX +
trastuzumab regimen and the assessment of cardiac function before
the initiation of therapy in this study allowed us to exclude the
influence of these risk factors for cardiac dysfunction and to
evaluate the impact of trastuzumab. Considering that none of the
patients in any group had a decrease in LVEF below 50%, the results
suggest that switching from reference trastuzumab to trastuzumab-NK
and continuing treatment is not significantly different than in the
other two groups. Currently, periodic post-treatment LVEF testing
is not performed on all the patients at our hospital because it is
not a required part of the regimen. However, a certain percentage
of patients may experience a decline in cardiac function during
trastuzumab therapy, and thus monitoring is still necessary.
This study has some limitations. First, this is a
retrospective observational study with a small sample size in a
single center. Furthermore, the effect of PTX on cardiotoxicity
cannot be eliminated. PTX has a small number of cardiotoxic
effects; however, compared to the cardiotoxicity of trastuzumab,
these effects are negligible and unlikely to be of concern in the
clinical settings (19).
Therefore, in this study, which aims to evaluate the safety of
switching from reference trastuzumab to biosimilars, the results
will not be affected by PTX because the effect of PTX on cardiac
function is comparable in all the three groups of patients without
prior treatment. However, we believe that one of the strengths of
this study is that we were able to secure patients without
concomitant use of the biopharmaceutical PER, as well as without
prior treatment with an anthracycline regimen, which can induce IR
and cardiac dysfunction, respectively. In particular, PER is often
used in combination with trastuzumab, and in the case of
combination regimens, it is difficult to identify which IR is
caused by which drug. In addition, prior treatment with an
anthracycline is associated with an increased risk of cardiac
dysfunction (6-8),
and the effect of the prior treatment cannot be excluded, since the
effect of trastuzumab on cardiac function cannot be adequately
assessed. The fact that we were able to evaluate the impact of
switching to trastuzumab under these conditions is significant,
although further studies including a larger sample size is needed
to validate our findings.
In summary, this study suggests that switching from
reference trastuzumab to trastuzumab biosimilar may not have a
significant effect on the frequency of IR expression or the
occurrence of cardiac dysfunction.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
All data generated or analyzed during the present
study are included in this published article.
Authors' contributions
TA conceived the study. TA, AS and DO designed the
experiments. TA, AS, DO and KM analyzed the data. TA and AS wrote
the paper. All authors confirm the authenticity of all the raw
data, and provided intellectual input, and are responsible for the
contents of the paper, including the data, analysis, and
interpretation. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Saitama Cancer Center (approval no. 1296). As this was
a retrospective observational study, consent was not obtained from
individual patients. An information disclosure document about this
study was created and published for the study patient, guaranteeing
the opportunity for the study patient to refuse.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Esteva FJ, Baranau YV, Baryash V, Manikhas
A, Moiseyenko V, Dzagnidze G, Zhavrid E, Boliukh D, Stroyakovskiy
D, Pikiel J, et al: Efficacy and safety of CT-P6 vs. reference
trastuzumab in HER2-positive early breast cancer: Updated results
of a randomised phase 3 trial. Cancer Chemother Pharmacol.
84:839–847. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Rugo HS, Barve A, Waller CF,
Hernandez-Bronchud M, Herson J, Yuan J, Sharma R, Baczkowski M,
Kothekar M, Loganathan S, et al: Effect of a proposed trastuzumab
biosimilar compared with trastuzumab on overall response rate in
patients with ERBB2 (HER2)-positive metastatic breast cancer: A
randomized clinical trial. JAMA. 317:37–47. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Pegram MD, Bondarenko I, Zorzetto MMC,
Hingmire S, Iwase H, Krivorotko PV, Lee KS, Li RK, Pikiel J,
Aggarwal R, et al: PF-05280014 (a trastuzumab biosimilar) plus
paclitaxel compared with reference trastuzumab plus paclitaxel for
HER2-positive metastatic breast cancer: A randomised, double-blind
study. Br J Cancer. 120:172–182. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Esteva FJ, Stebbing J, Wood-Horrall RN,
Winkle PJ, Lee SY and Lee SJ: A randomised trial comparing the
pharmacokinetics and safety of the biosimilar CT-P6 with reference
trastuzumab. Cancer Chemother Pharmacol. 81:505–514.
2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Sato K and Kohgo Y: Infusion reactions.
Gan To Kagaku Ryoho. 35:1671–1674. 2008.PubMed/NCBI(In Japanese).
|
|
6
|
Suter TM, Procter M, van Veldhuisen DJ,
Muscholl M, Bergh J, Carlomagno C, Perren T, Passalacqua R, Bighin
C, Klijn JG, et al: Trastuzumab-associated cardiac adverse effects
in the herceptin adjuvant trial. J Clin Oncol. 25:3859–3865.
2007.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Sengupta PP, Northfelt DW, Gentile F,
Zamorano JL and Khandheria BK: Trastuzumab-induced cardiotoxicity:
Heart failure at the crossroads. Mayo Clin Proc. 83:197–203.
2008.PubMed/NCBI View
Article : Google Scholar
|
|
8
|
Kolberg HC, Colleoni M, Demetriou GS,
Santi P, Tesch H, Fujiwara Y, Tomasevic Z and Hanes V: Cardiac
safety of the trastuzumab biosimilar ABP 980 in Women with
HER2-positive early breast cancer in the randomized, double-blind,
active-controlled LILAC Study. Drug Saf. 43:233–242.
2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Smith I, Procter M, Gelber RD, Guillaume
S, Feyereislova A, Dowsett M, Goldhirsch A, Untch M, Mariani G,
Baselga J, et al: 2-year follow-up of trastuzumab after adjuvant
chemotherapy in HER2-positive breast cancer: A randomised
controlled trial. Lancet. 369:29–36. 2007.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Slamon DJ, Clark GM, Wong SG, Levin WJ,
Ullrich A and McGuire WL: Human breast cancer: Correlation of
relapse and survival with amplification of the HER-2/neu oncogene.
Science. 235:177–182. 1987.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Li RK, Tokunaga E, Adamchuk H, Vladimirov
V, Yanez E, Lee KS, Bondarenko I, Vana A, Hilton F, Ishikawa T, et
al: Long-Term Safety and effectiveness of PF-05280014 (a
Trastuzumab Biosimilar) Treatment in patients with HER2-Positive
metastatic breast cancer: Updated results of a randomized,
double-blind study. BioDrugs. 36:55–69. 2022.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Stebbing J, Baranau Y, Baryash V, Manikhas
A, Moiseyenko V, Dzagnidze G, Zhavrid E, Boliukh D, Stroyakovskii
D, Pikiel J, et al: CT-P6 compared with reference trastuzumab for
HER2-positive breast cancer: A randomised, double-blind,
active-controlled, phase 3 equivalence trial. Lancet Oncol.
18:917–928. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Tatsuta R, Sumimoto T, Nakahara R, Tanaka
R and Itoh H: Comparison of treatment safety between brand-name
product and biosimilar of trastuzumab. Gan To Kagaku Ryoho.
48:945–949. 2021.PubMed/NCBI(In Japanese).
|
|
14
|
Thompson LM, Eckmann K, Boster BL, Hess
KR, Michaud LB, Esteva FJ, Hortobágyi GN and Barnett CM: Incidence,
risk factors, and management of infusion-related reactions in
breast cancer patients receiving trastuzumab. Oncologist.
19:228–234. 2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Chan AS, Freeswick S and Sklarin NT:
Trastuzumab 6 mg/kg and 4 mg/kg can be infused safely over 30
minutes. Cancer Res. 69 (2 suppl)(S3150)2009.
|
|
16
|
Ring A, Simcock R, Mitra S, Bloomfield D,
King J, Simpson S, Sadler G and Webb A: Infusion of trastuzumab
maintenance doses over 30 minutes. Ann Oncol. 19:1509–1510.
2008.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Tsutsui H, Isobe M, Ito H, Ito H, Okumura
K, Ono M, Kitakaze M, Kinugawa K, Kihara Y, Goto Y, et al: JCS
2017/JHFS 2017 guideline on diagnosis and treatment of acute and
chronic heart failure-Digest Version. Circ J. 83:2084–2184.
2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Guarneri V, Lenihan DJ, Valero V, Durand
JB, Broglio K, Hess KR, Michaud LB, Gonzalez-Angulo AM, Hortobagyi
GN and Esteva FJ: Long-term cardiac tolerability of trastuzumab in
metastatic breast cancer: The M.D. Anderson Cancer Center
experience. J Clin Oncol. 24:4107–4115. 2006.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Čelutkienė J, Pudil R, López-Fernández T,
Grapsa J, Nihoyannopoulos P, Bergler-Klein J, Cohen-Solal A,
Farmakis D, Tocchetti CG, von Haehling S, et al: Role of
cardiovascular imaging in cancer patients receiving cardiotoxic
therapies: A position statement on behalf of the Heart Failure
Association (HFA), the European Association of Cardiovascular
Imaging (EACVI) and the Cardio-Oncology Council of the European
Society of Cardiology (ESC). Eur J Heart Fail. 22:1504–1524.
2020.PubMed/NCBI View Article : Google Scholar
|