Spandidos Publications style
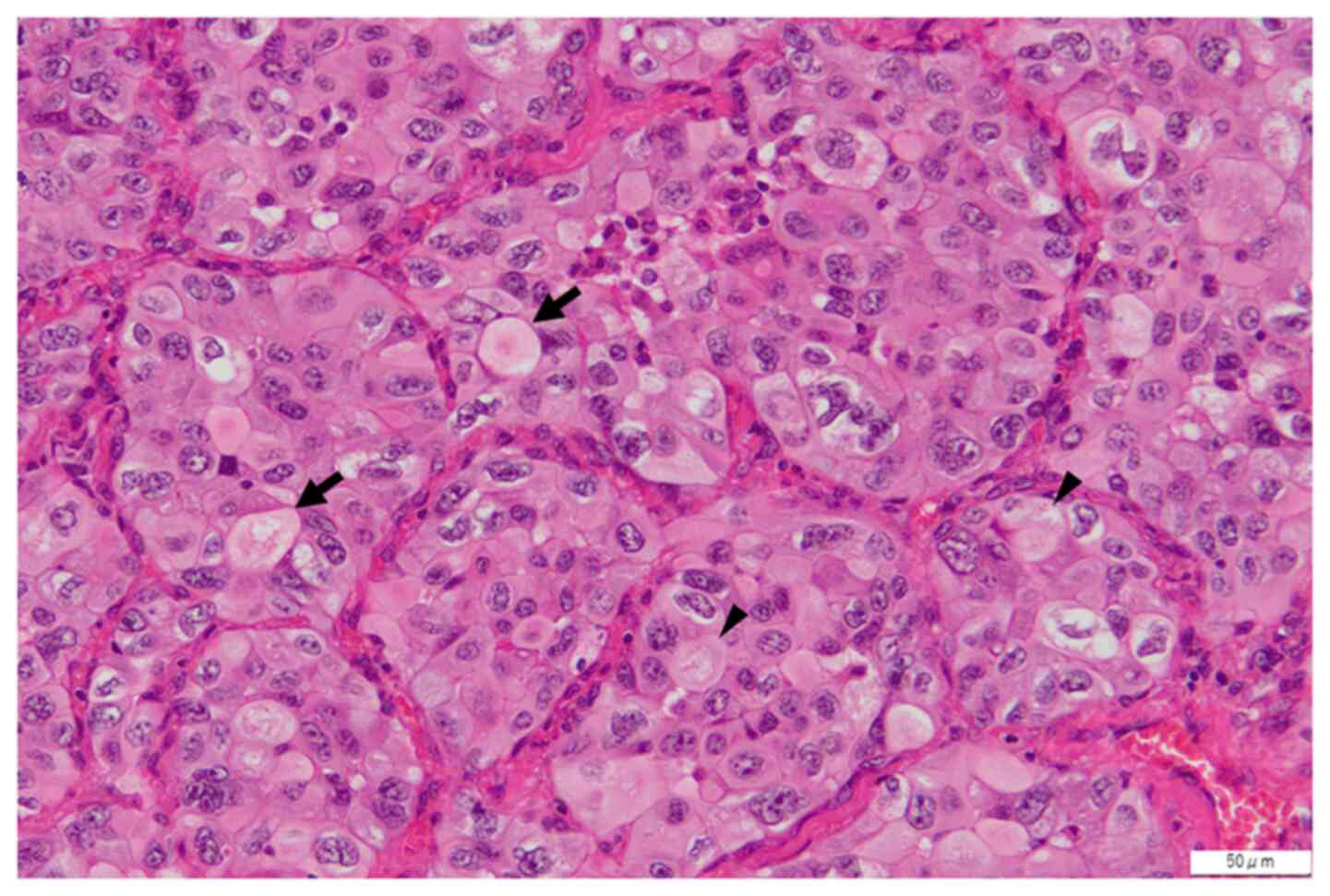
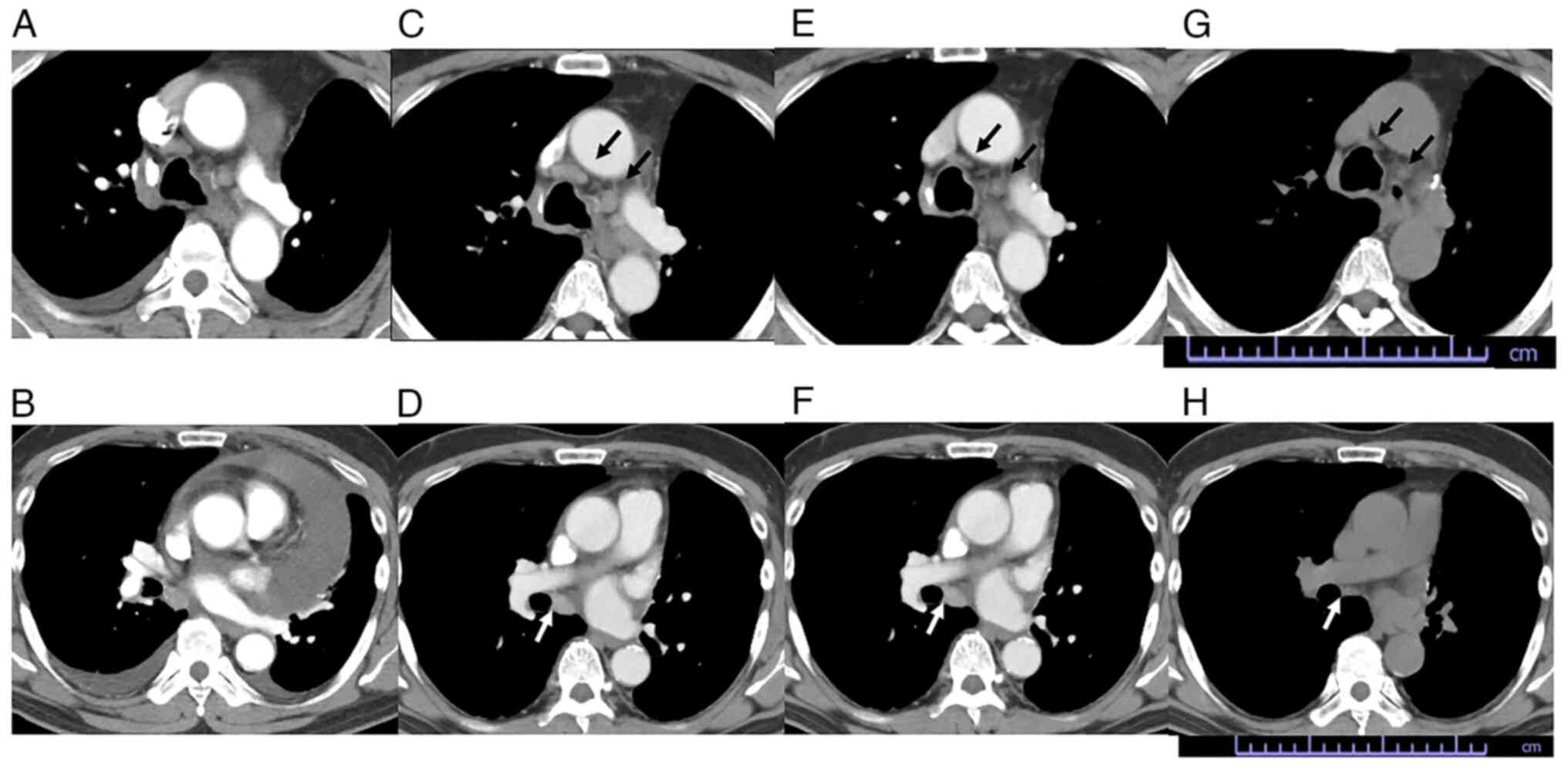
Onodera Y, Sekine A, Hagiwara E, Yamada S, Ikeda S, Tabata E, Kitamura H, Baba T, Komatsu S, Okudela K, Okudela K, et al: Successful tepotinib treatment of adenocarcinoma with MET exon 14 skipping and discordant results between Oncomine Dx target test and ArcherMET: A case report. Mol Clin Oncol 18: 49, 2023.
APA
Onodera, Y., Sekine, A., Hagiwara, E., Yamada, S., Ikeda, S., Tabata, E. ... Ogura, T. (2023). Successful tepotinib treatment of adenocarcinoma with MET exon 14 skipping and discordant results between Oncomine Dx target test and ArcherMET: A case report. Molecular and Clinical Oncology, 18, 49. https://doi.org/10.3892/mco.2023.2645
MLA
Onodera, Y., Sekine, A., Hagiwara, E., Yamada, S., Ikeda, S., Tabata, E., Kitamura, H., Baba, T., Komatsu, S., Okudela, K., Ogura, T."Successful tepotinib treatment of adenocarcinoma with MET exon 14 skipping and discordant results between Oncomine Dx target test and ArcherMET: A case report". Molecular and Clinical Oncology 18.6 (2023): 49.
Chicago
Onodera, Y., Sekine, A., Hagiwara, E., Yamada, S., Ikeda, S., Tabata, E., Kitamura, H., Baba, T., Komatsu, S., Okudela, K., Ogura, T."Successful tepotinib treatment of adenocarcinoma with MET exon 14 skipping and discordant results between Oncomine Dx target test and ArcherMET: A case report". Molecular and Clinical Oncology 18, no. 6 (2023): 49. https://doi.org/10.3892/mco.2023.2645