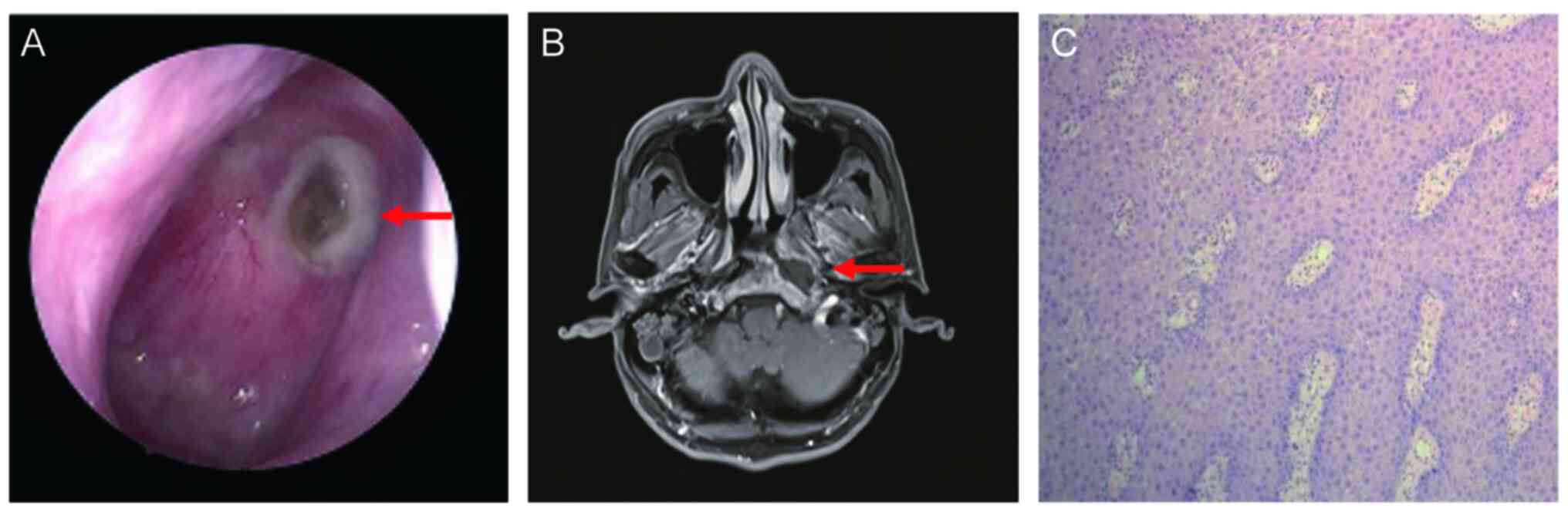
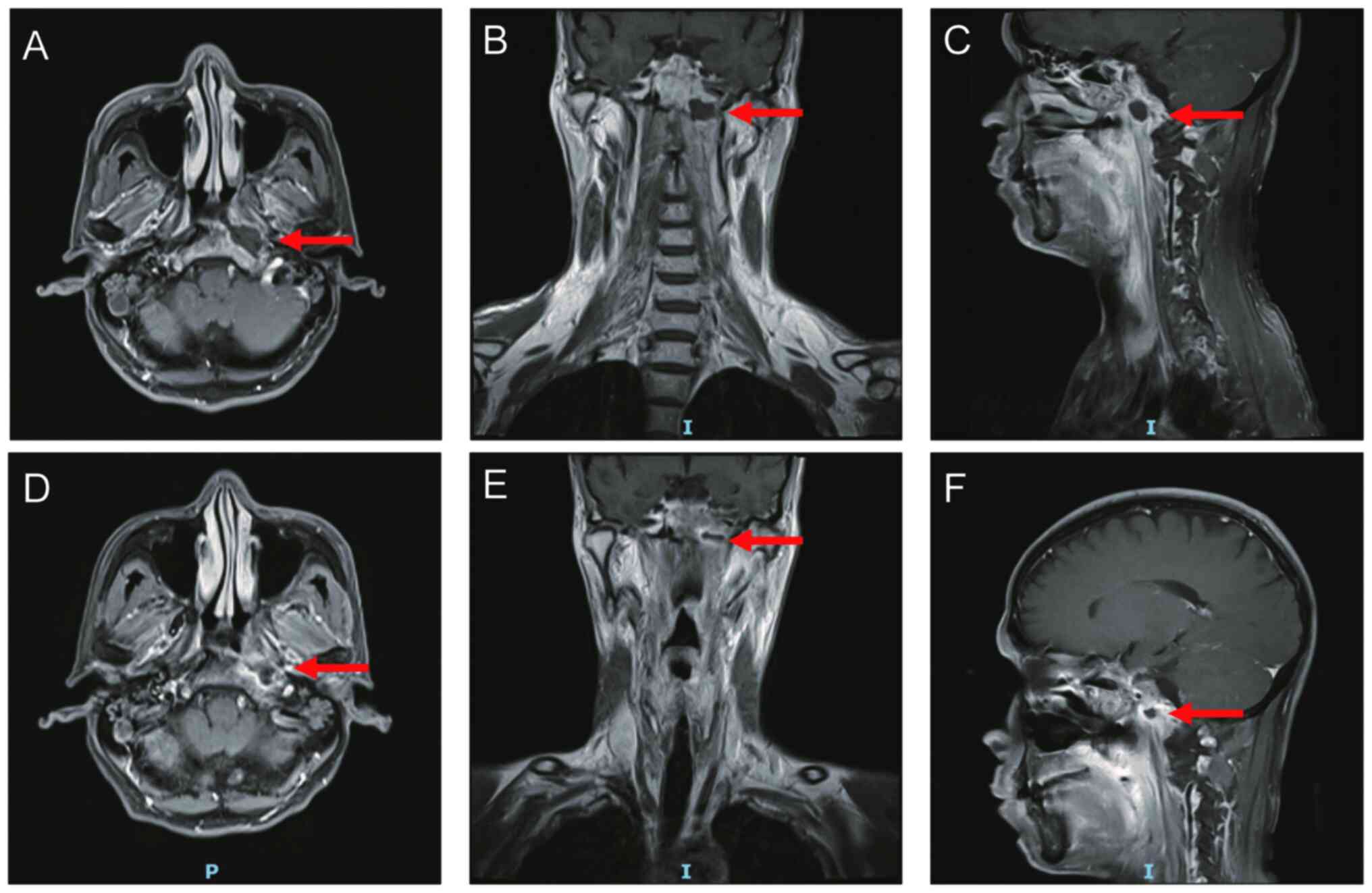
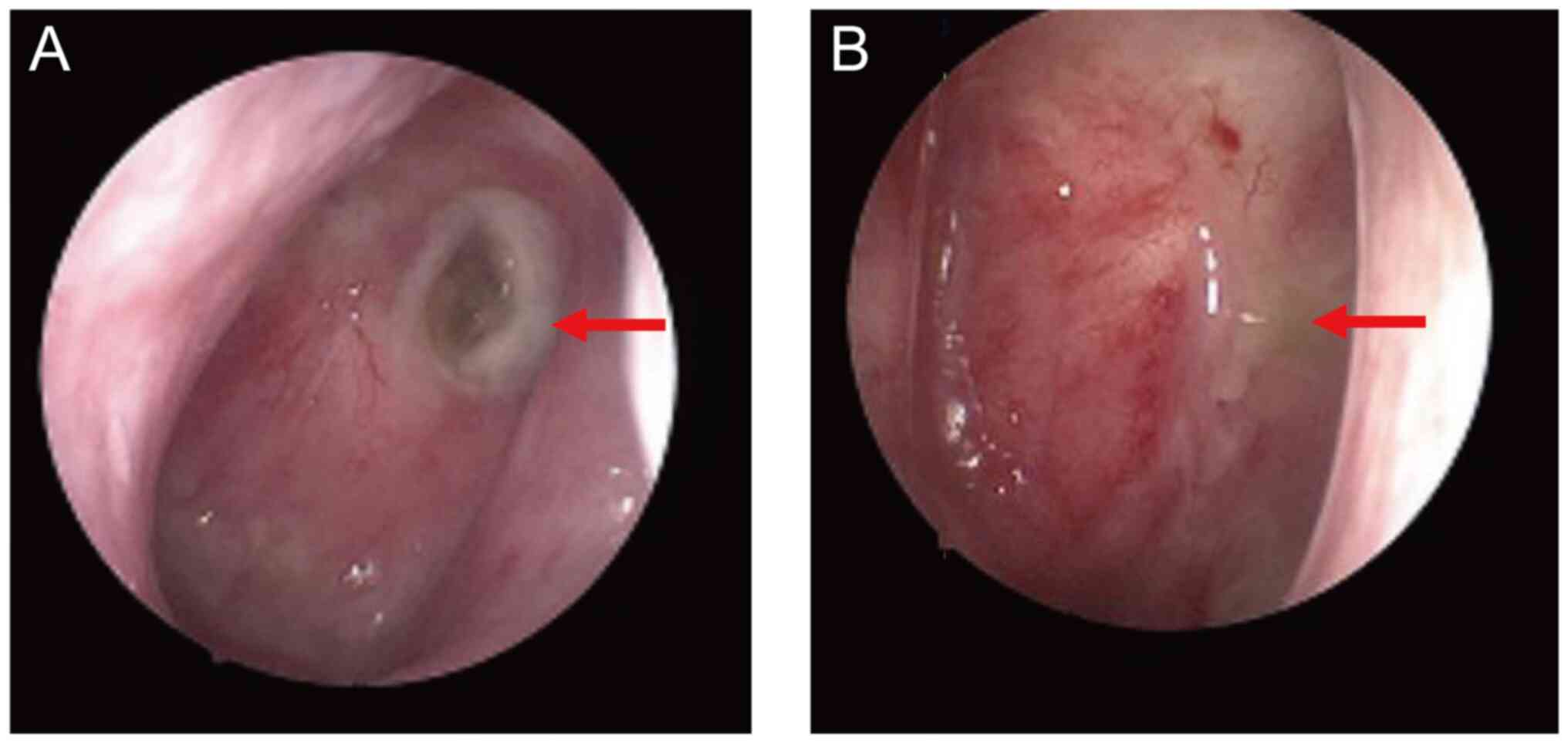
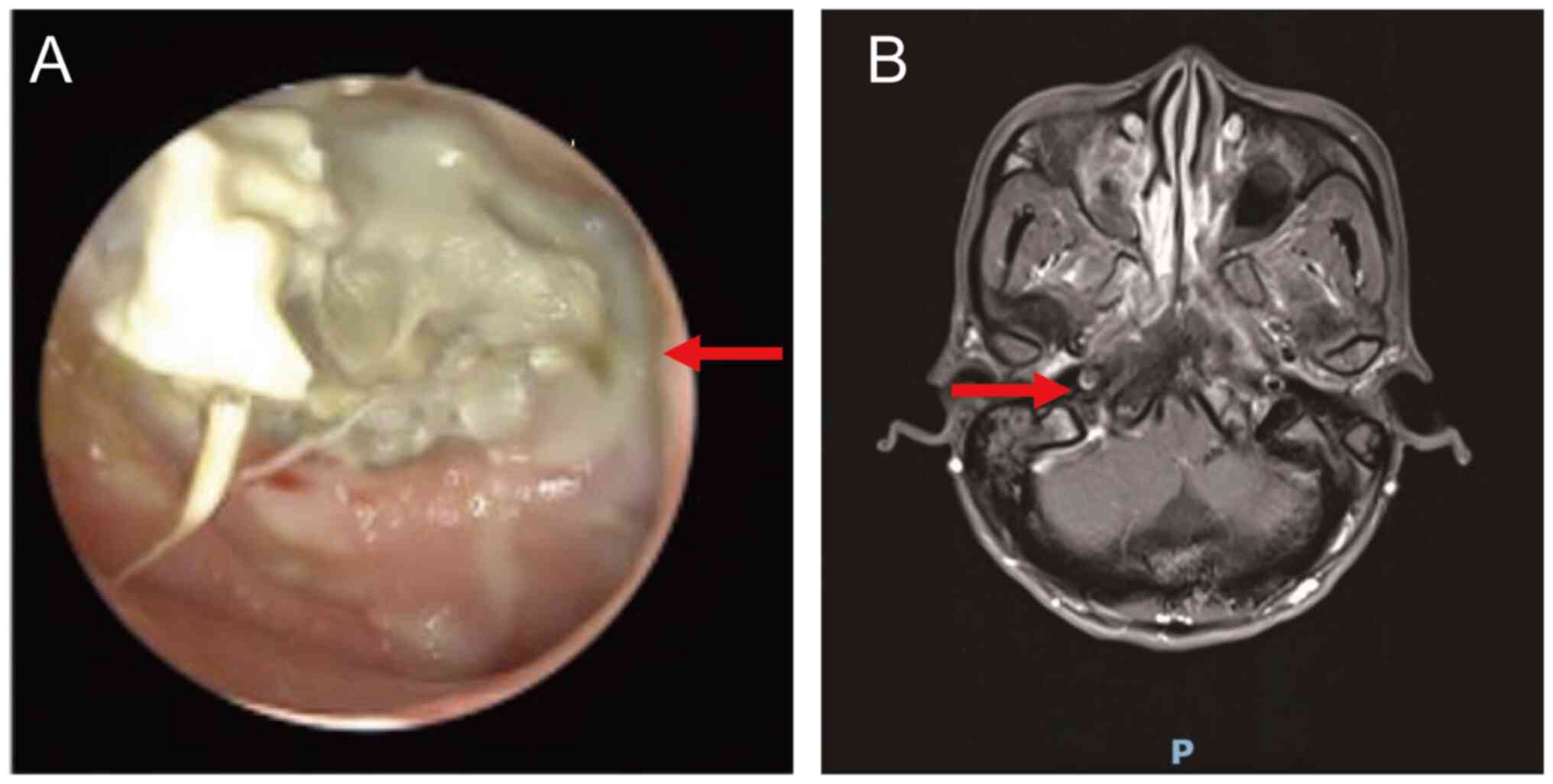
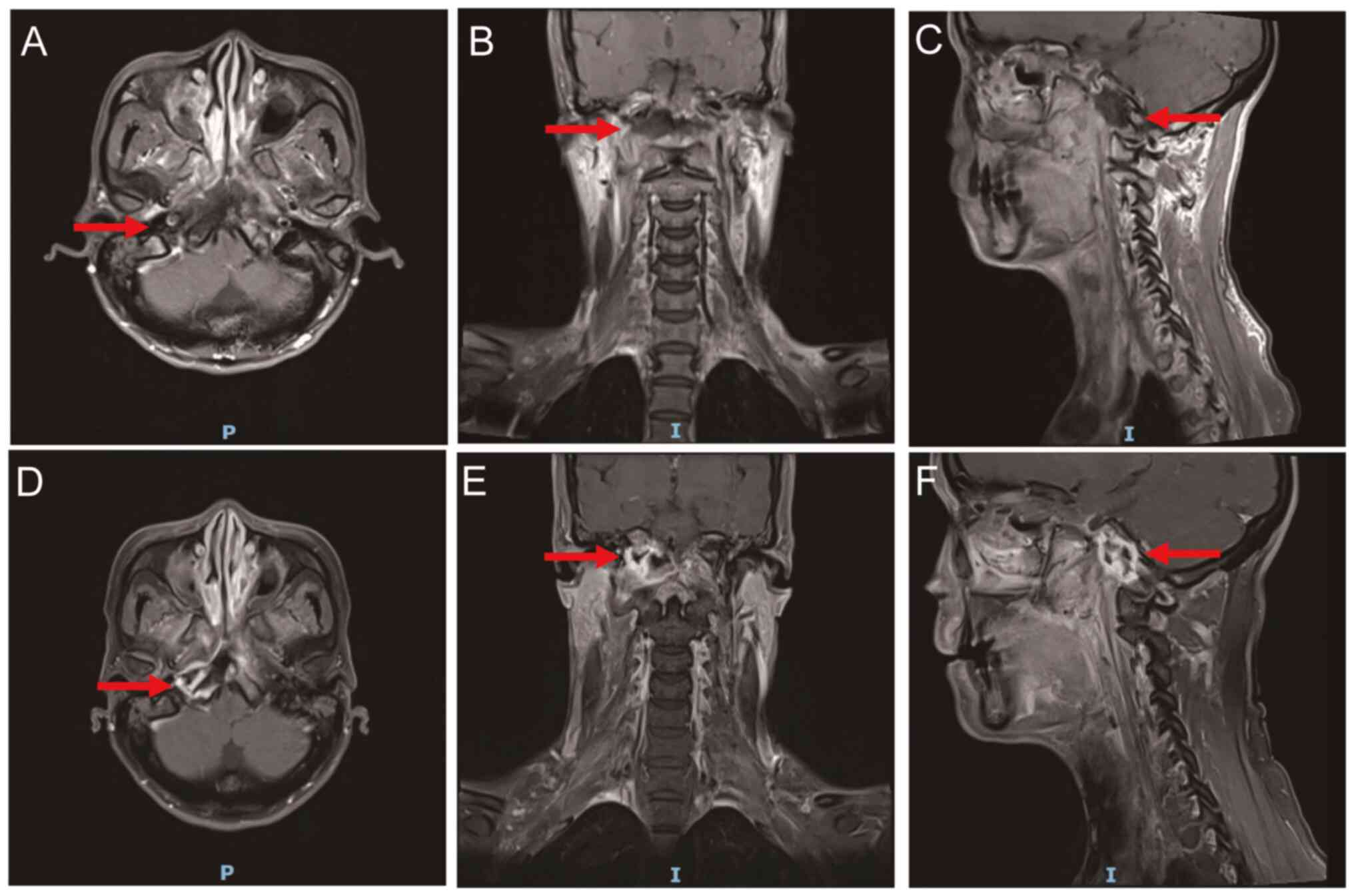
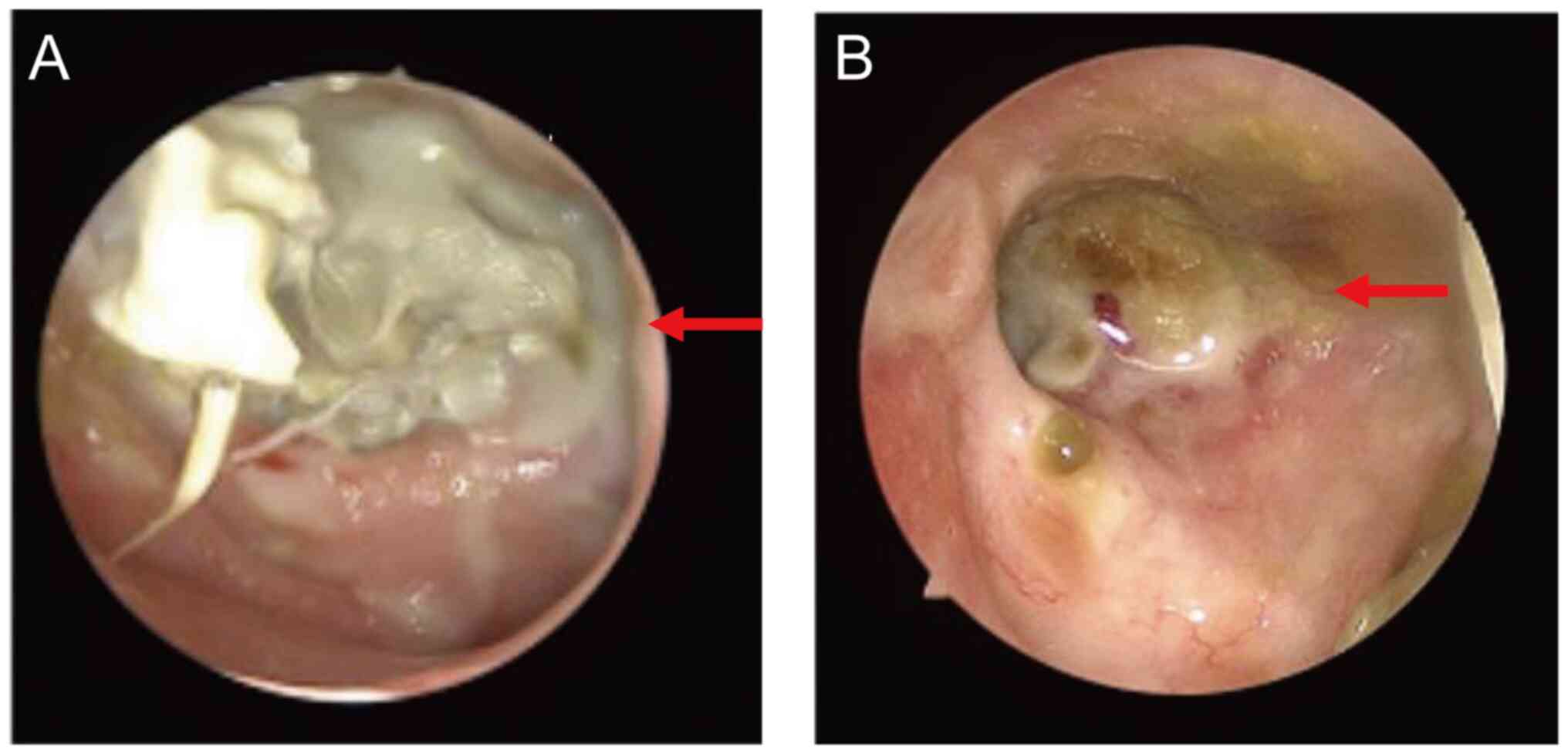
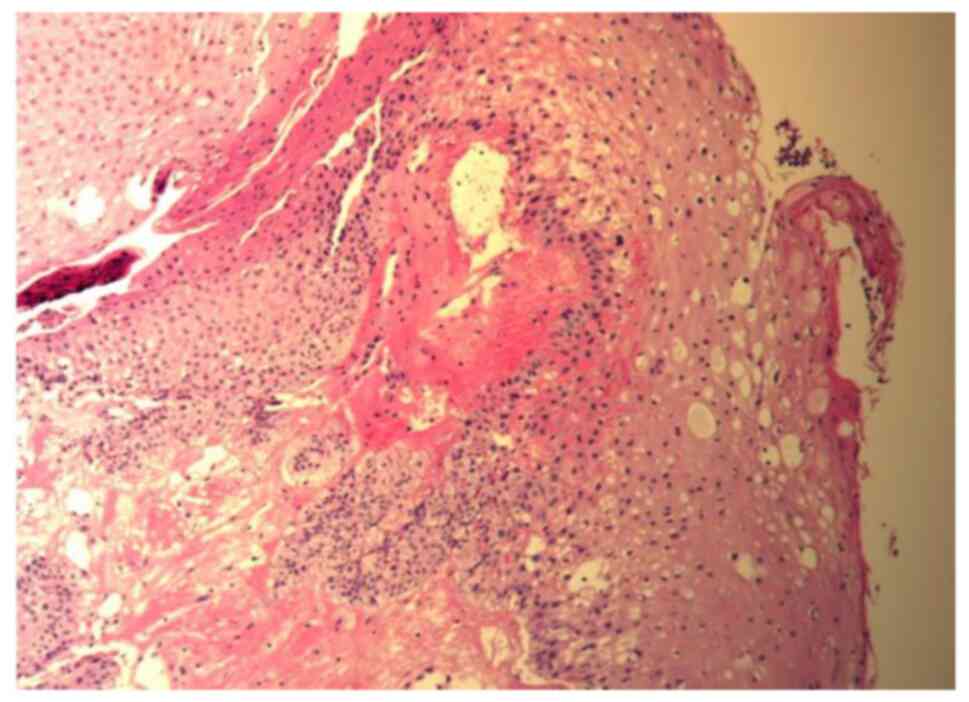
|
1
|
Luo J, Chia KS, Chia SE, Reilly M, Tan CS
and Ye W: Secular trends of nasopharyngeal carcinoma incidence in
Singapore, Hong Kong and Los Angeles Chinese populations,
1973-1997. Eur J Epidemiol. 22:513–521. 2007.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Xu M, Zang J, Luo S, Wang J and Li X:
Long-term survival outcomes and adverse effects of nasopharyngeal
carcinoma patients treated with IMRT in a non-endemic region: A
population-based retrospective study. BMJ Open.
11(e045417)2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Zhang MX, Li J, Shen GP, Zou X, Xu JJ,
Jiang R, You R, Hua YJ, Sun Y, Ma J, et al: Intensity-modulated
radiotherapy prolongs the survival of patients with nasopharyngeal
carcinoma compared with conventional two-dimensional radiotherapy:
A 10-year experience with a large cohort and long follow-up. Eur J
Cancer. 51:2587–2595. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Zeng L, Tian YM, Sun XM, Chen CY, Han F,
Xiao WW, Deng XW and Lu TX: Late toxicities after
intensity-modulated radiotherapy for nasopharyngeal carcinoma:
Patient and treatment-related risk factors. Br J Cancer. 110:49–54.
2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Li XY, Sun XS, Liu SL, Chen QY, Guo SS,
Liu LT, Yan JJ, Xie HJ, Tang QN, Liang YJ, et al: The development
of a nomogram to predict post-radiation necrosis in nasopharyngeal
carcinoma patients: A large-scale cohort study. Cancer Manag Res.
11:6253–6263. 2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Han P, Wang X, Liang F, Liu Y, Qiu X, Xu
Y, Chen R, Yu S and Huang X: Osteoradionecrosis of the Skull base
in nasopharyngeal carcinoma: Incidence and risk factors. Int J
Radiat Oncol Biol Phys. 102:552–555. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kong F, Zhou J, Du C, He X, Kong L, Hu C
and Ying H: Long-term survival and late complications of
intensity-modulated radiotherapy for recurrent nasopharyngeal
carcinoma. BMC Cancer. 18(1139)2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Han F, Zhao C, Huang SM, Lu LX, Huang Y,
Deng XW, Mai WY, The BS, Butler EB and Lu TX: Long-term outcomes
and prognostic factors of re-irradiation for locally recurrent
nasopharyngeal carcinoma using intensity-modulated radiotherapy.
Clin Oncol (R Coll Radiol). 24:569–576. 2012.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Chen MY, Mai HQ, Sun R, Guo X, Zhao C,
Hong MH and Hua YJ: Clinical findings and imaging features of 67
nasopharyngeal carcinoma patients with postradiation nasopharyngeal
necrosis. Chin J Cancer. 32:533–538. 2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Huang XM, Zheng YQ, Zhang XM, Mai HQ, Zeng
L, Liu X, Liu W, Zou H and Xu G: Diagnosis and management of skull
base osteoradionecrosis after radiotherapy for nasopharyngeal
carcinoma. Laryngoscope. 116:1626–1631. 2006.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Yang Q, Zou X, You R, Liu YP, Han Y, Zhang
YN, Guo L, Mai HQ, Xie CM, Li L, et al: Proposal for a new risk
classification system for nasopharyngeal carcinoma patients with
post-radiation nasopharyngeal necrosis. Oral Oncol. 67:83–88.
2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Huang WB, Wong STS and Chan JYW: Role of
surgery in the treatment of osteoradionecrosis and its
complications after radiotherapy for nasopharyngeal carcinoma. Head
Neck. 40:369–376. 2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Chen J, Yao Q, Huang M, Wang B, Zhang J,
Wang T, Ming Y, Zhou X, Jia Q, Huan Y, et al: A randomized Phase
III trial of neoadjuvant recombinant human endostatin, docetaxel
and epirubicin as first-line therapy for patients with breast
cancer (CBCRT01). Int J Cancer. 142:2130–2138. 2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Bao Y, Peng F, Zhou QC, Yu ZH, Li JC,
Cheng ZB, Chen L, Hu X, Chen YY, Wang J, et al: Phase II trial of
recombinant human endostatin in combination with concurrent
chemoradiotherapy in patients with stage III non-small-cell lung
cancer. Radiother Oncol. 114:161–166. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Han BH, Xiu QY, Wang HM, Shen J, Gu AQ,
Luo Y, Bai CX, Guo SL, Liu WC, Zhuang ZX, et al: A multicenter,
randomized, double-blind, placebo-controlled safety study to
evaluate the clinical effects and quality of life of
paclitaxel-carboplatin (PC) alone or combined with endostar for
advanced non-small cell lung cancer (NSCLC). Zhonghua Zhong Liu Za
Zhi. 33:854–859. 2011.PubMed/NCBI(In Chinese).
|
|
16
|
Wang J, Sun Y, Liu Y, Yu Q, Zhang Y, Li K,
Zhu Y, Zhou Q, Hou M, Guan Z, et al: (Results of randomized,
multicenter, double-blind phase III trial of rh-endostatin (YH-16)
in treatment of advanced non-small cell lung cancer patients).
Zhongguo Fei Ai Za Zhi. 8:283–290. 2005.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
17
|
Guan Y, Li A, Xiao W, Liu S, Chen B, Lu T,
Zhao C and Han F: The efficacy and safety of Endostar combined with
chemoradiotherapy for patients with advanced, locally recurrent
nasopharyngeal carcinoma. Oncotarget. 6:33926–33934.
2015.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Xing S, Fan Z, Shi L, Yang Z and Bai Y:
Successful treatment of brain radiation necrosis resulting from
triple-negative breast cancer with Endostar and short-term
hyperbaric oxygen therapy: A case report. Onco Targets Ther.
12:2729–2735. 2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zhang K, Yang S, Zhu Y, Mo A, Zhang D and
Liu L: Protection against acute radiation-induced lung injury: A
novel role for the anti-angiogenic agent Endostar. Mol Med Rep.
6:309–315. 2012.PubMed/NCBI View Article : Google Scholar
|
|
20
|
He T, Yan RN, Chen HY, Zeng YY, Xiang ZZ,
Liu F, Shao BF, Ma JC, Wang XR and Liu L: Comparing the 7th and 8th
editions of UICC/AJCC staging system for nasopharyngeal carcinoma
in the IMRT era. BMC Cancer. 21(327)2021.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Pan JJ, Ng WT, Zong JF, Chan LL,
O'Sullivan B, Lin SJ, Sze HC, Chen YB, Choi HC, Guo QJ, et al:
Proposal for the 8th edition of the AJCC/UICC staging system for
nasopharyngeal cancer in the era of intensity-modulated
radiotherapy. Cancer. 122:546–558. 2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Hua YJ, Chen MY, Qian CN, Hong MH, Zhao C,
Guo L, Guo X and Cao KJ: Postradiation nasopharyngeal necrosis in
the patients with nasopharyngeal carcinoma. Head Neck. 31:807–812.
2009.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Liu J, Ning X, Sun X, Lu H, Gu Y and Wang
D: Endoscopic sequestrectomy for skull base osteoradionecrosis in
nasopharyngeal carcinoma patients: A 10-year experience. Int J Clin
Oncol. 24:248–255. 2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Delanian S, Chatel C, Porcher R, Depondt J
and Lefaix JL: Complete restoration of refractory mandibular
osteoradionecrosis by prolonged treatment with a
pentoxifylline-tocopherol-clodronate combination (PENTOCLO): A
phase II trial. Int J Radiat Oncol Biol Phys. 80:832–839.
2011.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Robard L, Louis MY, Blanchard D, Babin E
and Delanian S: Medical treatment of osteoradionecrosis of the
mandible by PENTOCLO: Preliminary results. Eur Ann Otorhinolaryngol
Head Neck Dis. 131:333–338. 2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Lyons A and Ghazali N: Osteoradionecrosis
of the jaws: Current understanding of its pathophysiology and
treatment. Br J Oral Maxillofac Surg. 46:653–660. 2008.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Nordal RA, Nagy A, Pintilie M and Wong CS:
Hypoxia and hypoxia-inducible factor-1 target genes in central
nervous system radiation injury: A role for vascular endothelial
growth factor. Clin Cancer Res. 10:3342–3353. 2004.PubMed/NCBI View Article : Google Scholar
|
|
28
|
O'Reilly MS, Boehm T, Shing Y, Fukai N,
Vasios G, Lane WS, Flynn E, Birkhead JR, Olsen BR and Folkman J:
Endostatin: An endogenous inhibitor of angiogenesis and tumor
growth. Cell. 88:277–285. 1997.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Perletti G, Concari P, Giardini R, Marras
E, Piccinini F, Folkman J and Chen L: Antitumor activity of
endostatin against carcinogen-induced rat primary mammary tumors.
Cancer Res. 60:1793–1796. 2000.PubMed/NCBI
|
|
30
|
Bergers G, Javaherian K, Lo KM, Folkman J
and Hanahan D: Effects of angiogenesis inhibitors on multistage
carcinogenesis in mice. Science. 284:808–812. 1999.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Boehm T, Folkman J, Browder T and O'Reilly
MS: Antiangiogenic therapy of experimental cancer does not induce
acquired drug resistance. Nature. 390:404–407. 1997.PubMed/NCBI View
Article : Google Scholar
|
|
32
|
Kulke MH, Bergsland EK, Ryan DP, Enzinger
PC, Lynch TJ, Zhu AX, Meyerhardt JA, Heymach JV, Fogler WE, Sidor
C, et al: Phase II study of recombinant human endostatin in
patients with advanced neuroendocrine tumors. J Clin Oncol.
24:3555–3561. 2006.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Peng F, Xu Z, Wang J, Chen Y, Li Q, Zuo Y,
Chen J, Hu X, Zhou Q, Wang Y, et al: Recombinant human endostatin
normalizes tumor vasculature and enhances radiation response in
xenografted human nasopharyngeal carcinoma models. PLoS One.
7(e34646)2012.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Wang Y, Xuan Z, Zheng X, Han R, Zhou L,
Liu H and Liu Y: Efficacy of endostar combined with transcatheter
arterial chemoembolization and analysis of vascular endothelial
factor and C-reactive protein levels in patients with advanced
hepatocellular carcinoma under contrast enhanced ultrasound. J
BUON. 25:407–414. 2020.PubMed/NCBI
|
|
35
|
Cheng YJ, Meng CT, Ying HY, Zhou JF, Yan
XY, Gao X, Zhou N and Bai CM: Effect of Endostar combined with
chemotherapy in advanced well-differentiated pancreatic
neuroendocrine tumors. Medicine (Baltimore).
97(e12750)2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Zhou ZT, Zhou FX, Wei Q, Zou LY, Qin BF
and Peng XS: Phase II study of cisplatin/etoposide and endostar for
extensive-stage small-cell lung cancer. Cancer Chemother Pharmacol.
68:1027–1032. 2011.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Wang X, Shi Y, Jia Y, Zhao W, Zhang L, Bai
G, Ren Y, Chen YZ and Tong Z: Tolerance and pharmacokinetics of
recombinant human endostatin administered as single-dose or
multiple-dose infusions in patients with advanced solid tumors: A
phase I clinical trial. Technol Cancer Res Treat.
20(15330338211064434)2021.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Eder JP Jr, Supko JG, Clark JW, Puchalski
TA, Garcia-Carbonero R, Ryan DP, Shulman LN, Proper J, Kirvan M,
Rattner B, et al: Phase I clinical trial of recombinant human
endostatin administered as a short intravenous infusion repeated
daily. J Clin Oncol. 20:3772–3784. 2002.PubMed/NCBI View Article : Google Scholar
|