Introduction
Head and neck squamous cell carcinoma (HNSCC) is a
heterogeneous malignancy with a substantial global burden,
accounting for approximately 890 thousand new cases and 450
thousand deaths annually (1).
Prognosis is influenced by the clinical stage, with up to 60% of
patients being diagnosed at a locally advanced stage (2). While curative-intent treatment
remains feasible for these patients, a significant proportion are
deemed ineligible due to advanced age, comorbidities, or impaired
renal function (3). For patients
who cannot undergo definitive therapy, palliative radiotherapy (RT)
remains an important treatment option, providing rapid symptom
relief such as pain, bleeding, dysphagia, and airway compromise. In
selected cases, palliative RT can also achieve meaningful local
tumor control, which may improve performance status, facilitate
subsequent systemic therapy, and contribute to the preservation of
quality of life (4-6).
Immune checkpoint inhibitors (ICIs) targeting the
PD-1/PD-L1 axis have been used to treat unresectable, recurrent, or
metastatic HNSCC. The pivotal CheckMate-141 and KEYNOTE-048 trials
demonstrated the survival benefits of nivolumab and pembrolizumab,
respectively, establishing ICIs as standard systemic therapies in
appropriate patients (7,8). However, the optimal sequencing of
ICIs using RT in routine practice remains unclear. In locally
advanced disease, two phase III trials evaluating the concurrent
addition of ICIs to definitive chemoradiotherapy (CRT) reported
negative results: KEYNOTE-412, which tested pembrolizumab plus CRT
(9), and JAVELIN Head and Neck
100, which tested avelumab plus CRT (10). A recent phase II randomized trial
by Zandberg et al (11)
compared concurrent vs. sequential pembrolizumab with CRT, and
found that the sequential strategy was associated with superior
clinical outcomes compared to the concurrent approach. In parallel,
the development of reliable biomarkers to identify patients most
likely to benefit from ICIs remains a critical unmet need.
Present study uniquely evaluates a real-world
sequential strategy of palliative radiotherapy followed by immune
checkpoint inhibition in patients with advanced HNSCC. In addition,
we examine the prognostic impact of radiotherapy dose intensity
using the biologically effective dose. Accordingly, we conducted a
retrospective cohort study to assess overall survival,
progression-free survival, and treatment-related toxicity.
Materials and methods
Patients
This retrospective study included consecutive
patients with advanced head and neck squamous cell carcinoma who
underwent palliative radiotherapy as the initial oncologic
treatment at The University of Tokyo Hospital (Tokyo, Japan)
between January 2017 and December 2024. Eligible patients met the
following inclusion criteria: i) Pathologically confirmed head and
neck squamous cell carcinoma; ii) advanced-stage disease according
to the American Joint Committee on Cancer (AJCC) 8th edition
(stages III-IVC); iii) no prior oncologic treatment for the index
cancer; and iv) initiation of palliative radiotherapy to the head
and neck region. Patients were excluded if they had another active
malignancy at the time of radiotherapy or if clinical records were
insufficient to assess treatment exposure or survival outcomes. All
patients received palliative radiotherapy at study entry and were
subsequently classified into two groups according to
post-radiotherapy management: patients who did not receive immune
checkpoint inhibitors (RT group) and patients who received immune
checkpoint inhibitors within six months after radiotherapy
initiation (RT-ICI group).
Treatment methods
Radiotherapy techniques included intensity-modulated
radiotherapy or three-dimensional conformal radiotherapy, selected
at the discretion of the treating radiation oncologist. All
patients underwent computed tomography (CT) in the supine position
with thermoplastic mask immobilization, in accordance with our
institutional protocol (12).
Contrast-enhanced CT scans were acquired from the vertex to the
clavicle and were transferred to a treatment planning system.
Target volumes and organs at risk (OARs) were delineated according
to the institutional guidelines. The gross tumor volume (GTV) was
defined as the visible tumor on imaging. The clinical target volume
(CTV) was set to be identical to the GTV. The planning target
volume (PTV) was created by adding a 3-5 mm margin to the CTV. No
prophylactic irradiation was performed. A radiation oncologist
determined the dose and fractionation at the time of therapy. The
biologically effective dose (BED10) was calculated by
assuming an α/β ratio of 10.
Radiotherapy planning was optimized using
dose-volume histogram analyses to ensure adequate target coverage
while minimizing OAR exposure. Adjuvant ICI therapy, primarily
pembrolizumab or nivolumab, was administered intravenously in
accordance with standard institutional protocols. The ICI regimens
included pembrolizumab at a fixed dose of 200 mg per body every 3
weeks, with a 6-weekly dose of 400 mg per body permitted after more
than 1 year of treatment, or a combination chemotherapy regimen
(CF-PEM) consisting of pembrolizumab 200 mg per body on day 1,
cisplatin 80 mg/m² on day 1, and 5-fluorouracil 800 mg/m² on days
1-5.
Data collection and outcome
measures
Clinical data, including patient demographics, tumor
characteristics, radiotherapy details (total dose and fractionation
scheme), type and duration of ICI therapy, and follow-up
information, were retrospectively collected from electronic medical
records. The Karnofsky Performance Status (KPS) was documented
before the initiation of radiotherapy. The clinical outcomes
included overall survival (OS), progression-free survival (PFS),
and treatment-related adverse events, which were graded according
to the Common Terminology Criteria for Adverse Events (CTCAE),
version 5.0. OS and PFS were defined from the date of radiotherapy
initiation to death or progression, respectively, or to the date of
the last follow-up for censored cases. Survival curves for OS and
PFS were estimated using the Kaplan-Meier method, and differences
between groups were assessed using the log-rank test. Patients
without documented events were censored at the date of last
clinical follow-up. Cox proportional hazards regression models were
applied for multivariable analyses to identify factors
significantly associated with OS, with adjustment for clinically
relevant covariates. Propensity score matching (PSM) was considered
to further address potential confounding; however, given the
limited sample size, particularly in the RT-ICI group, PSM was
deemed statistically inefficient and likely to result in
substantial loss of information. Therefore, multivariable Cox
regression was adopted as the primary method for confounder
adjustment. Statistical significance was set at P<0.05. All
statistical analyses were performed using the R software (version
4.3.3; R Foundation for Statistical Computing, Vienna,
Austria).
Results
The baseline characteristics of the patients are
summarized in Table I. The cohort
comprised 74 patients with a median age of 75 years (range, 51-98
years); 61 (82.4%) were male. Performance status was generally
preserved, with a median KPS of 80, and 61% of patients had a KPS
≥80. The primary tumor sites were the oral cavity (31.1%),
hypopharynx (29.7%), oropharynx (20.3%), and other sites (18.9%).
The disease burden was largely advanced, with T4a-c in 86.5% of
patients and T3 in 13.5%. The median prescribed dose was 30 Gy
(range, 8-39 Gy), the median number of fractions was 10 (range,
1-13), and the median BED10 was 39.0 Gy (range,
11.2-50.7 Gy).
 | Table IBaseline patient characteristics
(n=74). |
Table I
Baseline patient characteristics
(n=74).
| Variable | Value |
|---|
| Median age, years
(range) | 75 (51-98) |
| Sex, n (%) | |
|
Male | 61 (82.4%) |
|
Female | 13 (17.6%) |
| Karnofsky performance
status, n (%) | |
|
90 | 13 (17.6%) |
|
80 | 32 (43.2%) |
|
70 | 16 (21.6%) |
|
60 | 8 (10.8%) |
|
50 | 3 (4.1%) |
|
40 | 2 (2.7%) |
| Primary tumor
location, n (%) | |
|
Oral
cavity | 23 (31.1%) |
|
Hypopharynx | 22 (29.7%) |
|
Oropharynx | 15 (20.3%) |
|
Larynx | 4 (5.4%) |
|
Paranasal
sinus | 4 (5.4%) |
|
Nasal
cavity | 3 (4.1%) |
|
Nasopharynx | 2 (2.7%) |
|
External
auditory canal | 1 (1.4%) |
| Clinical stage, n
(%) | |
|
III | 10 (13.5%) |
|
IVA | 25 (33.8%) |
|
IVB | 20 (27.0%) |
|
IVC | 19 (25.7%) |
| Median prescribed
dose, Gy (range) | 30 (8-39) |
| Median fractionation,
fractions (range) | 10 (1-13) |
| Median biologically
effective dosea, Gy
(range) | 39.0 (11.2-50.7) |
Seventeen patients received ICIs after palliative
radiotherapy (RT-ICI group). Of these, 15 received pembrolizumab
monotherapy and two received cisplatin/5-fluorouracil plus
pembrolizumab (CF-PEM). Among the patients in the 17 RT-ICI group,
11 (73.3%) had a Combined Positive Score (CPS) ≥20, 4 (26.7%) had
CPS 1-19, and none had CPS 0; CPS was not assessed in two patients
(11.8%). The median number of ICI administration cycles was nine
(range, 2-27). The remaining 57 patients comprised the RT group
without ICI; of these, six subsequently received chemotherapy after
radiotherapy (oral S-1 in three patients and
cisplatin/5-fluorouracil plus cetuximab in three).
The median follow-up was 6.2 months (range,
0.3-51.8). A significant difference in OS was observed between the
RT-ICI and RT groups (P<0.001). The 6-month, 1-year, and 2-year
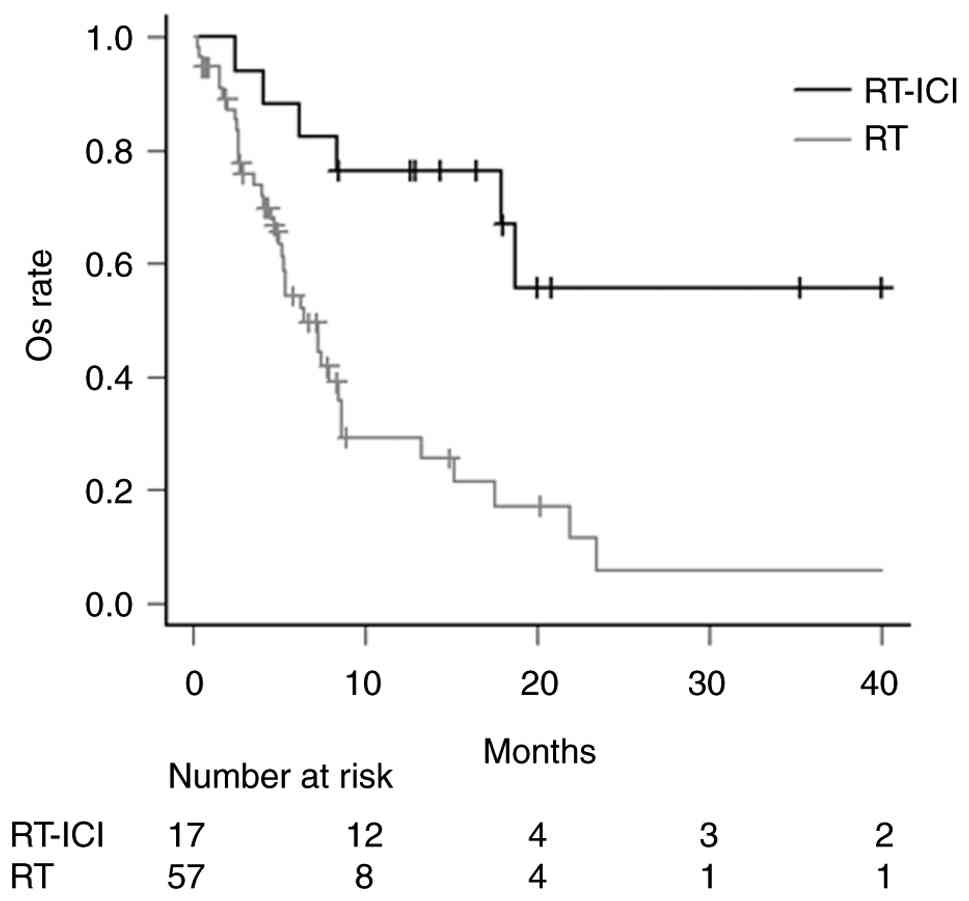
OS rates were 88.2% 76.5, and 55.8% in the RT-ICI group, compared
with 54.3, 29.2, and 5.7% in the RT group, respectively (Fig. 1). The PFS rate in the RT-ICI group
were 76.5% at 6 months, 52.3% at 1 year, and 20.9% at 2 years.
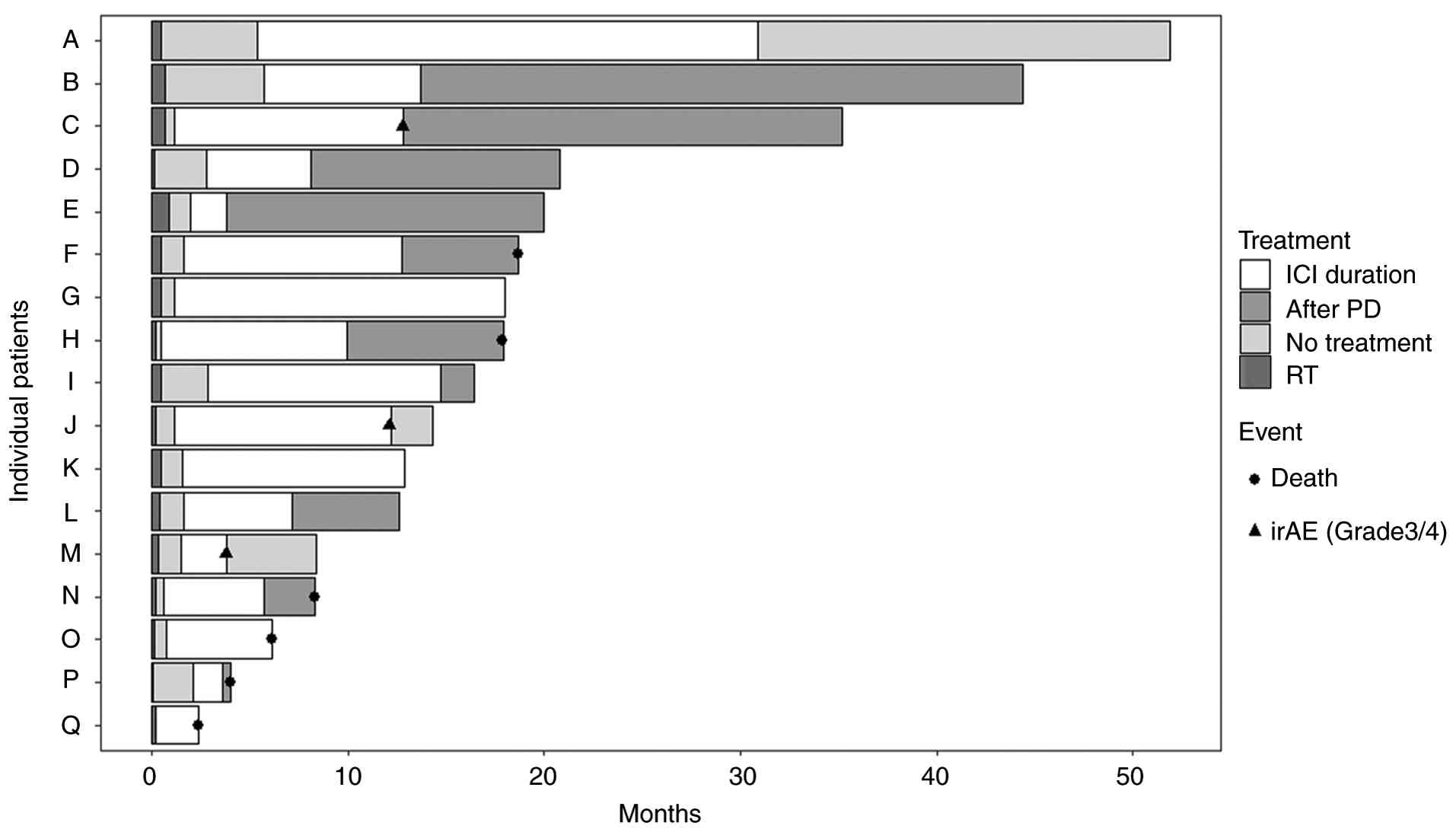
Among the 17 patients in the RT-ICI group, 10 discontinued
treatments due to disease progression during the study period
(Fig. 2). Post-progression
management included systemic therapy in five patients (paclitaxel
plus cetuximab in four and pemigatinib in one), local therapy in
two patients (BNCT or TPL), and best supportive care in three
patients. Three patients discontinued ICI due to grade ≥3
immune-related adverse events (irAEs), specifically grade 4
hepatitis, grade 3 cytokine-release syndrome, and grade 3 adrenal
insufficiency. Two patients experienced grade 3 mucositis as
adverse events related to palliative radiotherapy.
In univariate analysis, RT-ICI treatment [hazard
ratio (HR), 0.23; 95% confidence interval (CI), 0.10-0.56;
P=0.001], a BED10 ≥39 Gy (HR, 0.44; 95% CI, 0.23-0.85;
P=0.015), and better performance status (HR, 2.02; 95% CI,
1.09-3.73; P=0.025) were significantly associated with OS (Table II). In multivariate analysis,
performance status (HR, 1.99; 95% CI, 1.02-3.88; P=0.043), RT-ICI
treatment (HR, 0.22; 95% CI, 0.09-0.55; P=0.001), and a
BED10 ≥39 Gy (HR, 0.46; 95% CI, 0.24-0.91; P=0.025)
remained independent prognostic factors.
 | Table IIUnivariate and multivariate Cox
regression analyses for overall survival. |
Table II
Univariate and multivariate Cox
regression analyses for overall survival.
| Covariable | Univariate hazard
ratio (95% CI) | Univariate
P-value | Multivariate hazard
ratio (95% CI) | Multivariate
P-value |
|---|
| Age | | | | |
|
≤75
(reference) vs. >75 years | 1.121
(0.614-2.047) | 0.710 | 1.754
(0.832-3.698) | 0.140 |
| Sex | | | | |
|
Male
(reference) vs. Female | 1.166
(0.513-2.649) | 0.713 | 0.939
(0.378-2.333) | 0.892 |
| Karnofsky performance
status | | | | |
|
80-100
(reference) vs. ≤70 | 2.018
(1.092-3.731) | 0.025 | 1.991
(1.022-3.879) | 0.043 |
| Distant
metastasis | | | | |
|
No
(reference) vs. Yes | 1.189
(0.598-2.365) | 0.621 | 1.543
(0.712-3.345) | 0.272 |
| Treatment
modality | | | | |
|
RT
(reference) vs. RT-ICI | 0.233
(0.097-0.560) | 0.001 | 0.221
(0.090-0.546) | 0.001 |
| Biologically
effective dose | | | | |
|
<39
(reference) vs. ≥ 39 Gy | 0.441
(0.229-0.853) | 0.015 | 0.464
(0.237-0.907) | 0.025 |
Discussion
Our study demonstrated that the addition of ICI
therapy following palliative radiotherapy resulted in favorable and
durable outcomes, with a 2-year OS of 55.8% and 2-year PFS of 20.9%
in patients with advanced HNSCC. These results compare favorably
with previously reported outcomes for palliative radiotherapy
alone, in which median survival ranged from 7 to 11 months in
similar cohorts (13,14).
According to the updated results of the Phase III
KEYNOTE-048 study, with a median follow-up of 45 months,
pembrolizumab monotherapy achieved a median OS of 11.6 months,
while pembrolizumab combined with chemotherapy achieved a median OS
of 13.6 months in the overall cohort (15). Two-year OS rates remained at
approximately 20-30% with pembrolizumab-based regimens, although
differences in patient selection, disease stage, and treatment
intent should be considered. Collectively, these findings support
the idea that radiotherapy may potentiate the efficacy of ICIs
through immunogenic modulation, thereby leading to durable survival
benefits. In line with this, Cheng et al (16) reported in a retrospective study of
113 patients with recurrent or metastatic HNSCC that the addition
of radiotherapy to immunotherapy significantly improved outcomes,
with higher response rates (ORR 67.6% vs. 39.5%) and prolonged
survival (median PFS 20.0 vs. 4.0 months; median OS not reached vs.
26.0 months) compared with immunotherapy alone.
The synergistic potential of radiotherapy and ICIs
is supported by preclinical evidence. Ionizing radiation induces
tumor cell death and enhances tumor antigen release and
presentation by antigen-presenting cells, thereby promoting
peripheral T cell priming with cell lines of breast cancer in
vitro and in vivo (17). This process augments the efficacy
of ICIs such as PD-1/PD-L1 blockade by facilitating T cell
reactivation (18). In addition,
radiotherapy can modulate the tumor microenvironment by increasing
vascular permeability and chemokine production and promoting immune
cell infiltration into the tumor (19-21).
Consequently, a hot tumor microenvironment enhances responsiveness
to ICIs and fosters therapeutic synergy (22-24).
The clinical efficacy of ICI administration after
radiotherapy has been clearly demonstrated in other malignancies,
particularly lung cancer. The PACIFIC trial demonstrated that
consolidation therapy with durvalumab, an anti-PD-L1 monoclonal
antibody, following chemoradiotherapy significantly improved
outcomes in stage III non-small-cell lung cancer. The 5-year OS
rate was 42.9% with durvalumab vs. 33.4% with a placebo, confirming
that a durable survival benefit was maintained beyond 5 years
(25). The ADRIATIC trial in
limited-stage small-cell lung cancer showed that durvalumab
consolidation after chemoradiotherapy prolonged both PFS and OS
compared to placebo (26). Our
findings are consistent with these observations. Further studies
are warranted to clarify the optimal sequencing of radiotherapy and
immune checkpoint inhibition. Given the retrospective,
single-institution nature of the present study, prospective
randomized trials are needed to formally evaluate different RT-ICI
sequencing strategies. In addition, optimization of treatment
timing should be explored, as the efficacy of ICIs may vary
according to their class and the temporal relationship with
radiotherapy. Finally, mechanistic investigations incorporating
translational biomarkers and advanced imaging approaches are
required to elucidate how radiotherapy modulates the tumor
microenvironment and immune responsiveness, thereby informing
rational integration of RT and ICIs in future clinical
practice.
In the present study, a higher radiation dose
(BED10 ≥39 Gy) was independently associated with
improved survival outcomes, suggesting a possible dose-response
relationship even in the palliative setting. However, the potential
survival benefit of dose escalation must be carefully weighed
against the risk of treatment-related toxicity, particularly in
palliative patients for whom symptom control and quality of life
are primary treatment goals. Higher-dose radiotherapy has been
proposed to enhance antitumor immune responses through increased
tumor antigen release and immune activation, which may augment the
efficacy of immune checkpoint inhibitors (27). Supporting this concept, a
meta-analysis by Viani et al (28), including 28 studies and 1,986
patients, reported that higher dose was associated with superior
overall response rates and an improved therapeutic ratio.
Stereotactic body radiation therapy (SBRT), a novel technique that
enables highly concentrated dose delivery to the target while
sparing surrounding normal tissues, represents a promising strategy
(29,30). Its clinical value is currently
being evaluated in a phase III randomized trial comparing SBRT with
conventional palliative radiotherapy for painful bone metastases
(NCT06065449).
However, the optimal dose and fractionation schedule
for palliative RT in the context of sequential RT-ICI therapy
remains uncertain. Dose escalation must be carefully balanced
against treatment-related toxicity, particularly mucositis,
xerostomia, and swallowing dysfunction, which can substantially
affect quality of life in patients with advanced head and neck
cancer. Notably, a phase III randomized trial in patients
unsuitable for curative treatment demonstrated no significant
differences in OS or PFS between 20 Gy in five fractions and a
dose-escalated 30 Gy in five fractions regimen, with comparable
toxicity profiles, suggesting that moderate dose escalation may be
feasible but not necessarily associated with clear survival benefit
(31).
Despite its retrospective nature, this analysis was
conducted at a single institution with relatively consistent
clinical practices regarding radiotherapy planning and immune
checkpoint inhibitor administration. Nevertheless, several
important limitations should be acknowledged. First, the
retrospective design is inherently subject to selection bias, which
may have influenced treatment allocation and could have resulted in
an overestimation of survival outcomes, thereby limiting the
generalizability of the findings. In particular, the comparison
between the RT-ICI and RT-alone groups is susceptible to immortal
time bias, as patients in the RT-ICI group must, by definition,
have survived long enough to receive subsequent immunotherapy. In
the absence of formal time-dependent statistical adjustments, such
as a landmark analysis, the observed survival differences should
therefore be interpreted with caution and cannot be considered
fully causal. Second, the median follow-up period was relatively
short, and long-term survival estimates were based on a limited
number of patients at risk, as reflected by the wide confidence
intervals; accordingly, greater emphasis should be placed on
earlier endpoints. Third, the small sample size, particularly in
the RT-ICI cohort, limits statistical power and may affect the
robustness of the observed associations. In addition, key
biological variables relevant to immunotherapy response, such as
PD-L1 combined positive score (CPS), were not available for all
patients. Given the incomplete availability of CPS data and the
limited number of evaluable cases, we were unable to reliably
assess the interaction between PD-L1 status, radiotherapy dose
intensity, and survival outcomes, either through subgroup analyses
or inclusion as covariates in multivariable models. This limitation
may have constrained our ability to elucidate potential effect
modification by tumor immunophenotype and should be considered when
interpreting the observed survival benefits. Furthermore, the study
population was predominantly male, elderly, and had relatively
preserved performance status, which limits extrapolation of the
findings to younger patients, female patients, or more frail
subgroups, including those with poor performance status or very
advanced age. Finally, although radiotherapy planning and delivery
were performed according to standardized institutional protocols,
some degree of heterogeneity in radiotherapy techniques and
dose-fractionation schedules remained across patients; this
residual variability may have influenced outcome interpretation and
limits reproducibility. Prospective studies with larger cohorts,
comprehensive biomarker profiling, and appropriate time-dependent
analyses are warranted to better define the optimal radiotherapy
dose intensity, including relevant thresholds, as well as the
timing and sequencing of ICI administration.
In conclusion, in patients with advanced HNSCC, ICI
given within 6 months after palliative RT was associated with
better OS and PFS. Higher radiation dose was also associated with
better outcomes; however, these findings should be interpreted as
associations rather than evidence of a causal effect. Prospective
studies with appropriate time-dependent analyses are needed to
confirm these results and to guide the integration of RT and ICI in
advanced head and neck cancer.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
AK was a major contributor to writing this
manuscript, and collected and assembled the data. AK and HY
analyzed and interpreted the clinical data. YS conceived of this
study, and participated in its design and coordination. AO and MM
made substantial contributions to data curation and interpretation
of data. YI, KY, KK and YS contributed to the acquisition of
clinical data. AO, MM, YI, KY, KK, HY and YS revised the manuscript
critically for important intellectual content. AK and HY confirm
the authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study was conducted in accordance with
the ethical principles outlined in The Declaration of Helsinki. The
study was approved by the Research Ethics Committee of the Faculty
of Medicine of the University of Tokyo (IRB no. 3372-7; Tokyo,
Japan). The requirement for informed consent was waived due to the
retrospective design of the study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Nenclares P, Rullan A, Tam K, Dunn LA, St
John M and Harrington KJ: Introducing checkpoint inhibitors into
the curative setting of head and neck cancers: Lessons learned,
future considerations. Am Soc Clin Oncol Educ Book. 42:1–16.
2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Kim SS, Liu HC and Mell LK: Treatment
considerations for patients with locoregionally advanced head and
neck cancer with a contraindication to cisplatin. Curr Treat
Options Oncol. 24:147–161. 2023.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Grewal AS, Jones J and Lin A: Palliative
radiation therapy for head and neck cancers. Int J Radiat Oncol
Biol Phys. 105:254–266. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Weiss ML, Domschikowski J, Krug D,
Sonnhoff M, Nitsche M, Hoffmann W, Becker-Schiebe M, Bock F,
Hoffmann M, Schmalz C, et al: The impact of palliative radiotherapy
on health-related quality of life in patients with head and neck
cancer-results of a multicenter prospective cohort study. Clin
Transl Radiat Oncol. 41(100633)2023.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Fabian A, Domschikowski J, Hoffmann M,
Weiner O, Schmalz C, Dunst J and Krug D: Patient-reported outcomes
assessing the impact of palliative radiotherapy on quality of life
and symptom burden in head and neck cancer patients: A systematic
review. Front Oncol. 11(683042)2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Ferris RL, Blumenschein G Jr, Fayette J,
Guigay J, Colevas AD, Licitra L, Harrington K, Kasper S, Vokes EE,
Even C, et al: Nivolumab for recurrent squamous-cell carcinoma of
the head and neck. N Engl J Med. 375:1856–1867. 2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Burtness B, Harrington KJ, Greil R,
Soulières D, Tahara M, de Castro G Jr, Psyrri A, Basté N, Neupane
P, Bratland Å, et al: Pembrolizumab alone or with chemotherapy
versus cetuximab with chemotherapy for recurrent or metastatic
squamous cell carcinoma of the head and neck (KEYNOTE-048): A
randomised, open-label, phase 3 study. Lancet. 394:1915–1928.
2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Machiels JP, Tao Y, Licitra L, Burtness B,
Tahara M, Rischin D, Alves G, Lima IPF, Hughes BGM, Pointreau Y, et
al: Pembrolizumab plus concurrent chemoradiotherapy versus placebo
plus concurrent chemoradiotherapy in patients with locally advanced
squamous cell carcinoma of the head and neck (KEYNOTE-412): A
randomised, double-blind, phase 3 trial. Lancet Oncol. 25:572–587.
2024.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Lee NY, Ferris RL, Psyrri A, Haddad RI,
Tahara M, Bourhis J, Harrington K, Chang PM, Lin JC, Razaq MA, et
al: Avelumab plus standard-of-care chemoradiotherapy versus
chemoradiotherapy alone in patients with locally advanced squamous
cell carcinoma of the head and neck: A randomised, double-blind,
placebo-controlled, multicentre, phase 3 trial. Lancet Oncol.
22:450–462. 2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Zandberg DP, Vujanovic L, Clump DA, Isett
BP, Wang H, Sica G, Bao R, Li H, Ohr J, Skinner HD, et al:
Randomized phase II study of concurrent versus sequential
pembrolizumab in combination with chemoradiation in locally
advanced head and neck cancer. J Clin Oncol. 43:2572–2582.
2025.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Katano A and Yamashita H: Early-stage
hypopharyngeal squamous cell carcinoma treated with radical
radiotherapy at a uniform dose of 70 Gy in 35 fractions: A
single-center study. Eur Arch Otorhinolaryngol. 281:4401–4407.
2024.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Pandey KC, Revannasiddaiah S, Pant NK,
Nautiyal V, Rastogi M and Gupta MK: Palliative radiotherapy in
locally advanced head and neck cancer after failure of induction
chemotherapy: Comparison of two fractionation schemes. Indian J
Palliat Care. 19:139–145. 2013.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Katano A, Minamitani M, Tongyu G, Ohira S
and Yamashita H: Survival following palliative radiotherapy for
head and neck squamous cell carcinoma: Examining treatment
indications in elderly patients. Cancer Diagn Progn. 4:46–50.
2024.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Harrington KJ, Burtness B, Greil R,
Soulières D, Tahara M, de Castro G Jr, Psyrri A, Brana I, Basté N,
Neupane P, et al: Pembrolizumab with or without chemotherapy in
recurrent or metastatic head and neck squamous cell carcinoma:
Updated results of the phase III KEYNOTE-048 study. J Clin Oncol.
41:790–802. 2023.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Cheng Y, Wang J, Sun J, Zhong Y and Wu Q:
Radiotherapy improves the clinical outcomes of recurrent or
metastatic head and neck cancers treated with immunotherapy. Discov
Oncol. 16(988)2025.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Krombach J, Hennel R, Brix N, Orth M,
Schoetz U, Ernst A, Schuster J, Zuchtriegel G, Reichel CA,
Bierschenk S, et al: Priming anti-tumor immunity by radiotherapy:
Dying tumor cell-derived DAMPs trigger endothelial cell activation
and recruitment of myeloid cells. Oncoimmunology.
8(e1523097)2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Liao Y, Deng J, Yang X, Wang D and Du X:
Advances in radiotherapy enhancing the efficacy of immune
checkpoint inhibitors in malignant. Front Oncol.
15(1611036)2025.
|
|
19
|
Liu S, Wang W, Hu S, Jia B, Tuo B, Sun H,
Wang Q, Liu Y and Sun Z: Radiotherapy remodels the tumor
microenvironment for enhancing immunotherapeutic sensitivity. Cell
Death Dis. 14(679)2023.
|
|
20
|
Wang Y, Berg T, Verona AW and Morris ZS:
NImmuno-radiobiology: Effects of radiation therapy on immune cells,
tumor microenvironment, susceptibility of tumor cells to
immune-mediated destruction, and anti-tumor immunity. Int J Radiat
Oncol Biol Phys: S0360-3016(26)00088-X, 2026 (Epub ahead of
print).
|
|
21
|
Lin A, Xiong M, Jiang A, Chen L, Huang L,
Li K, Wong HZH, Zhang J, Liu Z, Cheng Q, et al: Tumor immunotherapy
and microbiome: From bench-to-bedside applications. MedComm (2020).
7(e70454)2026.
|
|
22
|
Chen C, Liu Y and Cui B: Effect of
radiotherapy on T cell and PD-1/PD-L1 blocking therapy in tumor
microenvironment. Hum Vaccin Immunother. 17:1555–1567. 2021.
|
|
23
|
Khosravi GR, Mostafavi S, Bastan S,
Ebrahimi N, Gharibvand RS and Eskandari N: Immunologic tumor
microenvironment modulators for turning cold tumors hot. Cancer
Commun (Lond). 44:521–553. 2024.
|
|
24
|
Benoit A, Vogin G, Duhem C, Berchem G and
Janji B: Lighting up the fire in the microenvironment of cold
tumors: A major challenge to improve cancer immunotherapy. Cells.
12(1787)2023.
|
|
25
|
Spigel DR, Faivre-Finn C, Gray JE, Vicente
D, Planchard D, Paz-Ares L, Vansteenkiste JF, Garassino MC, Hui R,
Quantin X, et al: Five-year survival outcomes from the PACIFIC
trial: Durvalumab after chemoradiotherapy in stage III
non-small-cell lung cancer. J Clin Oncol. 40:1301–1311. 2022.
|
|
26
|
Cheng Y, Spigel DR, Cho BC, Laktionov KK,
Fang J, Chen Y, Zenke Y, Lee KH, Wang Q, Navarro A, et al:
Durvalumab after chemoradiotherapy in limited-stage small-cell lung
cancer. N Engl J Med. 391:1313–1327. 2024.
|
|
27
|
Pontoriero A, Critelli P, Chillari F,
Ferrantelli G, Sciacca M, Brogna A, Parisi S and Pergolizzi S:
Modulation of radiation doses and chimeric antigen receptor T
cells: A promising new weapon in solid tumors-a narrative review. J
Pers Med. 13(1261)2023.
|
|
28
|
Viani GA, Gouveia AG, Matsuura FK, Neves
LVF, Marta GN, Chua MLK and Moraes YF: Assessing the efficacy of
palliative radiation treatment schemes for locally advanced
squamous cell carcinoma of the head and neck: A meta-analysis. Rep
Pract Oncol Radiother. 28:137–146. 2023.
|
|
29
|
Katano A, Minamitani M, Ohira S and
Yamashita H: Recent advances and challenges in stereotactic body
radiotherapy. Technol Cancer Res Treat.
23(15330338241229363)2024.
|
|
30
|
Katano A: Exploring the current challenges
and pioneering clinical applications of stereotactic radiotherapy
in cancer treatment. Technol Cancer Res Treat.
24(15330338251333658)2025.
|
|
31
|
Mallick S, Dagar A, Ghosh A, Aashita Raj
J, Hazarika S, Meena JK, Kumar A, Sharma J, Panda S, et al: Optimum
radiation dose for palliation in head and neck squamous cell
carcinoma (OpRAH)-A phase 3 randomized controlled trial. Radiother
Oncol. 202(110611)2025.
|