Introduction
Colorectal cancer (CRC) is a major global health
concern; it is the third most frequently diagnosed malignancy and a
leading cause of cancer-related deaths worldwide (1). The 10-year survival rate for CRC is
58%, but this varies depending on race and ethnicity (2). In Mexico, the mortality rate due to
CRC is increasing by 1.3% in women and 2.7% in men annually.
However, public health insurance policies do not cover the cost of
CRC screening despite its importance in diagnosis and treatment.
This lack of programs focused on early detection is a cause of
concern in Mexico (3). Certain
lifestyle factors, such as smoking, unhealthy diet, obesity and
high alcohol consumption, are associated with the incidence of
sporadic CRC; these factors are essentially linked to chronic
inflammation. The Tumor-Node-Metastasis (TNM) classification uses
clinical parameters to illustrate the development of CRC, its
clinical outcomes and how it responds to treatment (4).
Inflammation is extensively involved in several
cancer hallmarks, including evasion of apoptosis, tumor growth,
proliferation and metastasis, thereby playing a crucial role in
promoting tumorigenesis (5).
Nuclear factor-κB (NF-κB) is a family of inducible transcription
factors that regulates the inflammatory response in various human
malignant diseases by controlling the expression of
pro-inflammatory genes (6,7). The NF-κB family comprises five hetero
or homodimers: RelA/p65, RelB, NF-κB1 (p50/p105), NF-κB2 (p52/p100)
and c-Rel. Multiple post-translational modifications, particularly
phosphorylation, regulate NF-κB signaling (8,9).
Several studies have shown an association between increased
activity of NF-κB and CRC development (8,10).
The principal regulators of NF-κB activation are cytokines such as
tumor necrosis factor-α (TNF-α), which trigger the IKK complex,
leading to the phosphorylation of the p65 subunit and translocation
of NF-κB dimers to the nucleus, regulating gene expression
associated with tumorigenesis (8,11).
Previous studies indicate that TNF-α promotes
phosphorylation of NF-κB/p65 on serine 536 and 529 in various
inflammation-related pathologies, including cancer (12-15).
TNF-α is a pro-inflammatory cytokine produced by macrophages and
commonly found in the tumor microenvironment (12). In previous studies, and one by the
present authors it was found that TNF-α gene expression was higher
in advanced stages of patients with sporadic CRC compared with
early stages. TNF-α activity has been proposed as a potential
marker for CRC development (14-17).
However, it remains uncertain what clinical impact TNF-α/NF-κB
signaling has on patients with sporadic CRC.
The aim of the present study was to investigate the
TNF-α/NF-κB signaling activity and its correlation with the
development and progression of sporadic CRC. To assess the
activation of NF-κB, the phosphorylation of p65 Ser536 and Ser529
was assessed through phospho-flow cytometry. Additionally, TNF-α
gene expression was determined by reverse
transcription-quantitative PCR (RT-qPCR) and the protein level was
detected using flow cytometry and ELISA.
Materials and methods
Participants
In total, 122 patients >18 years old (adults)
diagnosed with clinical and histological sporadic CRC who agreed to
participate and signed the informed consent form were enrolled from
December 2018 to June 2023 as experimental group in the present
study. A calibration control group consisting of 30 tissue samples
of individuals without a history of inflammatory bowel disease was
also included. The collection of calibration control (non-CRC)
tissues was conducted during the same time period. These
individuals were included after undergoing routine preventive
colonoscopy due to non-specific symptoms that ultimately resulted
negative for neoplasia (such as chronic constipation, unexplained
anemia or mild abdominal pain). The tissues were obtained as
biopsies of non-cancerous colonic mucosa during these procedures.
Therefore, no additional hospitalization was required specifically
for sample collection. Additionally, blood samples were collected
from 100 healthy individuals between December 2018 and May 2025 to
establish a control group for evaluating serum TNF-α levels. All
experimental and control samples were collected at the Civil
Hospital of Guadalajara ‘Dr. Juan I. Menchaca’ (Guadalajara,
Mexico). Patients who had taken drugs that could modify the natural
activity of TNF-α, and NF-kB or those who voluntarily decided that
the analysis of their samples should not be used in the project
were excluded from this study. Further exclusions included those
with unsuccessful laboratory procedures or with the impossibility
of access to the new biological samples. The pathology service
determined the CRC stage using the TNM classification according to
the American Joint Committee on Cancer (AJCC) 8th edition (18). Only non-treated patients were
included in the study. The present study was performed following
the Declaration of Helsinki and was approved by the ‘Hospital Civil
de Guadalajara Ethics Committee’ (approval no. 21711; Guadalajara,
Mexico). The patients provided written and verbal informed consent
for scientific purposes to publish any associated data from the
present study.
Tissue samples
In each case, tumor tissue and adjacent normal
mucosa from distant regions of the same patient's resected tumor
were obtained. The sampling was performed according to the ‘Cancer
Care Quality Measures: Diagnosis and Treatment of Colorectal
Cancer’, issued by the ‘Agency for Health Care Research and
Quality’ (19,20). Samples were collected using
phosphate-buffered saline and transported to the laboratory for
immediate flow cytometric experiments. For RNA isolation,
RNAlater® Stabilization Solution (cat. no. AM7020;
Thermo Fisher Scientific, Inc.) was used and further processed for
qPCR analysis. Tissue samples from non-patients with CRC were
collected during preventive colonoscopy using the same procedure.
The remaining tissues were stored at -80˚C for any additional
experiments.
Serum samples
Blood samples were collected preoperatively from 122
patients diagnosed with CRC constituting the experimental group,
along with 100 healthy participants who served as the control
group. The samples were centrifuged at 1,811 x g for 5 min at room
temperature in the Eppendorf centrifuge 5810. Then, the serum was
separated and stored at -80˚C until further analysis.
Flow cytometric assays
Tissues (5-10 mg) suspended in PBS were minced with
a sharp blade. The procedure to obtain single-cell suspensions
before flow cytometric experiments was carried out as proposed by
Ali et al (21) and adapted
to the samples as follows: The minced tissue was subjected to
enzymatic digestion in 1 ml of 0.1% trypsin solution, followed by
dissociation with 0.1% collagenase. The phospho-flow cytometric
method was used to measure the phosphorylated NF-κB/p65 residues
using the Perfix Phospho-Epitopes Exposure kit (cat. no. B26976;
Beckman Coulter, Inc.), anti-NF-κB p65pS529-PE (cat. no.
130-120-252; Miltenyi Biotec GmbH) and anti-NF-κB p65pS536-Alexa
Fluor (cat. no. A88939; Beckman Coulter, Inc.) at a working
dilution of 1:50 (2 µl/test) incubated for 30 min at room
temperature while protected from light, following the
manufacturer's recommended protocol. To assess TNF-α activity, cell
surface protein was detected. The cell suspensions were processed
and stained with anti-human TNFα Alexa Fluor 488 antibody (cat. no.
53-7349-41 Affymetrix; Thermo Fisher Scientific, Inc.) using the
‘Staining Cell Surface Antigens for Flow Cytometry Reagents’ from
Affymetrix eBioscience, which includes the following reagents and
steps on its protocol: Pre-incubation of the cell suspension with
20 µl Human Fc Receptor Binding Inhibitor (cat. no. 14-9161) for
10-20 min at 2-25˚C. The TNFα Alexa Fluor 488 antibody at a working
dilution of 1:200 (2 µl/test) was combined with an appropriate
volume of Flow Cytometry Staining Buffer (cat. no. 00-4222) so that
the final staining volume was 100 µl. Next, the cell suspensions
were incubated for 40 min at 4˚C and protected from light. After
incubation, the cells were washed by adding 2 ml/tube of Flow
Cytometry Staining Buffer and centrifuged at 400-600 x g for 5 min
at room temperature. Data were acquired using a Gallios 10 Flow
Cytometer (Beckman Coulter, Inc.). The Gallios software v.10
(Beckman Coulter, Inc.) was utilized to analyze 20,000 events.
Gating was applied to exclude cell debris and autofluorescence.
ELISA
The TNF-α level was measured in serum samples of
patients with CRC and control participants using the human TNF-α
ELISA Kit (cat. no. KHC3011; Thermo Fisher Scientific, Inc.).
Experiment validation was performed using standards provided by the
kit manufacturers and analyzed with the MultiSkan FC automated
microplate reader (Thermo Fisher Scientific, Inc.). The optical
density at 450/550 nm was relied upon to detect the concentrations
of each sample. The mean concentration and the standard deviation
of the optical density of control patients were utilized to
establish the ELISA test cut-off.
Gene expression
A total of 10-15 mg of tissue was cut into small
pieces and then collected in 0.5 ml of TRI® reagent
(cat. no. T3934; Sigma-Aldrich; Merck KGaA). Each sample was
homogenized in Tissue Lyser LT (Qiagen Inc.) for 3 min at 25 Hz.
The following steps of RNA isolation were performed according to
the manufacturer's instructions (Qiagen Inc.). RNA was quantified
using a NanoDrop 1000 spectrophotometer (Thermo Fisher Scientific,
Inc.). Reverse transcription was performed using 1 µg total RNA
treated with DNase1 amplification grade and the High-Capacity cDNA
Reverse Transcription kit (cat. no. 4387406; Applied Biosystems;
Thermo Fisher Scientific, Inc.) in a GeneAmp PCR System 9700
thermal cycler. The reaction was carried out by following the given
conditions: 25˚C for 25 min, 37˚C for 120 min, 85˚C for 5 min, and
infinite hold at 4˚C.
qPCR was performed using 1 µl of cDNA per reaction,
TaqMan® Gene Expression Master Mix (cat. no. 4369016;
Applied Biosystems; Thermo Fisher Scientific, Inc.) and
TaqMan®MGB Probes Gene Expression Assays (cat. no.
4331182; Applied Biosystems; Thermo Fisher Scientific, Inc.) with
FAM-NFQ detector for NFKB1 (ID no: Hs00765730_m1),
RELA (ID no: Hs00153294_m1) and TNF (ID no:
Hs00174128_m1) primers. The reaction was performed in a 7900HT Fast
Real-Time PCR System linked to SDS 2.4 software (Applied
Biosystems; Thermo Fisher Scientific, Inc.). Cycling conditions
were as follows: 50˚C for 2 min, 95˚C for 10 min, 95˚C for 15 sec,
and 60˚C for 1 min (40 cycles). Relative quantification (RQ)
analysis was performed using the Livak method
(2-ΔΔCq) with amplification
efficiencies >95% (22). The
GUSB gene (Hs99999908_m1) was used as a housekeeping
reference and normal adjacent mucosa as a calibrator. The
experimental samples were evaluated in triplicate.
Statistical analysis
The data were analyzed using the SPSS 20.0 software
(IBM Corp.). The NF-κB/p65 activation was evaluated using
Kruskal-Wallis test and Dunn's test for a post hoc analysis, and
then differences in active cell percentage pairwise comparison
among groups were assessed by the Mann-Whitney U test. The Wilcoxon
matched-pairs test was used to examine differences in gene
expression and anti-human TNF-α levels between tumor tissues and
adjacent normal mucosa. TNF-α serum levels data were first
evaluated for normality and homogeneity of variances, using the
Shapiro-Wilk and Levene's tests, respectively. Therefore, a one-way
analysis of variance (ANOVA) was conducted to compare the group
means with Tukey's post hoc test. The correlation between molecular
parameters and TNM groups was analyzed using the Pearson
correlation coefficient test. P<0.05 was considered to indicate
a statistically significant difference.
Results
Participants
A total of 122 patients with CRC were included, of
which 83 were males and 39 were females, all unrelated to
hereditary colorectal syndromes. The tissues were collected during
surgery and in total, 92 tissues corresponded to colon and 30 to
rectal cancer. The mean age was 56 years, ranging from 34 to 62
years; none of the participants had been previously treated with
radiotherapy or chemotherapy. The TNM classification to establish
tumor staging resulted in I=18, II=34, III=48 and IV=22 patients
per stage. Participants were classified into early stages (E) and
advanced stages (A) to analyze the results. The E group included 52
participants from stages I and II, while the A group included 70
participants from stages III and IV. This categorization was
implemented to enable an in-depth analysis of the collected data
and identify significant differences between the groups. A total of
30 individuals, of whom 8 were male and 22 were female, who were
established with non-cancerous condition after colonoscopy and
histopathology examination, were included as control calibrators
for tissue experiments comparison. The mean age was 39 years,
ranging from 26 to 48 years; none of the participants in the
control group had an inflammatory bowel disease history (Table I). Additionally, blood samples were
collected from 100 healthy individuals to establish a control group
for evaluating serum TNF-α levels (Table II). This approach aimed to
establish an appropriate cut-off threshold for the experimental
groups, thus ensuring the validation of the assay and enabling the
attainment of statistically significant results across all
groups.
 | Table IParameters of sporadic CRC and
non-cancerous tissue control group. |
Table I
Parameters of sporadic CRC and
non-cancerous tissue control group.
| A, Clinical
parameters |
|---|
| Parameter | CRC group
(n=122) | Non-cancerous
tissue control group (n=30) |
|---|
| Age, years | | |
|
Mean ±
SD) | 56±7.31 | 39±6.57 |
|
≤40, N
(%) | 9 (7.4) | 19 (63.3) |
|
41-60, N
(%) | 54 (44.3) | 11 (36.7) |
|
>60, N
(%) | 59 (48.4) | 0 (0.0) |
| Sex, N (%) | | |
|
Male | 83 (68.0) | 8 (26.7) |
|
Female | 39 (32.0) | 22 (73.3) |
| B, Pathological
parameters |
| Parameter | CRC group
(n=122) | Control group
(n=30) |
| TNM stage, N
(%) | | |
|
I | 18 (14.8) | N/A |
|
II | 34 (27.9) | N/A |
|
III | 48 (39.3) | N/A |
|
IV | 22 (18.0) | N/A |
| Tumor location, N
(%) | | |
|
Colon | 92 (75.4) | N/A |
|
Rectum | 30 (24.6) | N/A |
| Histopathology
result, N (%) | | |
|
CRC | 122 (100.0) | 0 (0.0) |
|
Non-cancerous
condition | 0 (0.0) | 30 (100.0) |
| Disease history, N
(%) | | |
|
IBD | 97 (79.5) | 0 (0.0) |
|
No IBD | 25 (20.5) | 30 (100.0) |
 | Table IIParameters of healthy control
group. |
Table II
Parameters of healthy control
group.
| Clinical
parameters |
|---|
| Parameter | Healthy control
group (n=100) |
|---|
| Age, years | |
|
Mean ±
SD | 46±6.39 |
|
≤40, N
(%) | 37(37) |
|
41-60, N
(%) | 63(63) |
|
>60, N
(%) | 0 (0) |
| Sex, N (%) | |
|
Male | 59 (68.0) |
|
Female | 41 (32.0) |
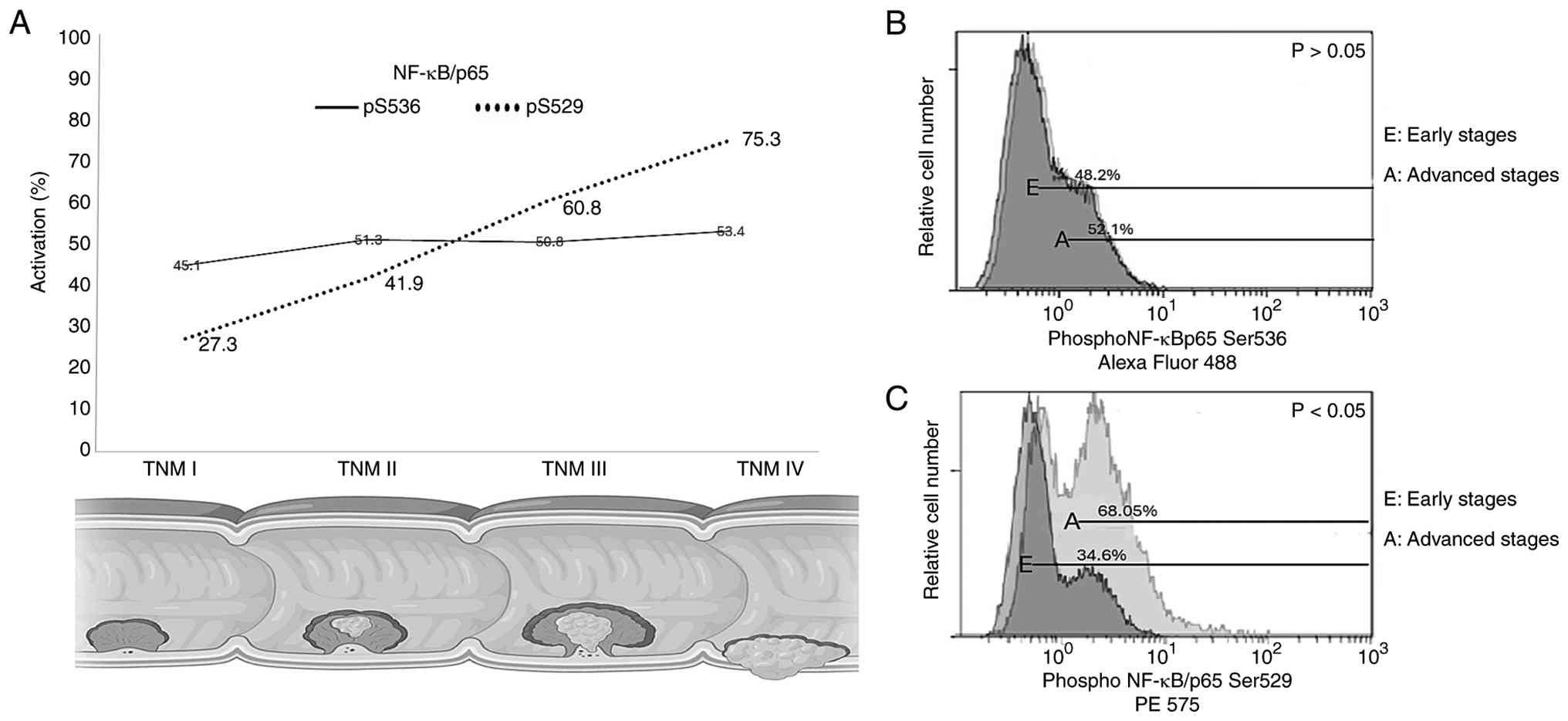
NF-ĸB/p65 activation in CRC
development
Phospho-flow cytometric analysis of NF-κB revealed
that both p65 (pS536) and (pS529) were active in patients with CRC.
The activation levels were compared with those found in adjacent
normal mucosa. Differences between early and advanced TNM groups
were analyzed using non-neoplastic tissues as a control group;
results are described below and reported in Table III. NF-ĸB/p65 (pS536) activation
evaluated by TNM stages was reported as follows: I=45.1%, II=51.3%,
III=50.8% and IV=53.4% (Fig. 1A).
Compared with the control group, the activation was significantly
higher in all stages (P<0.05). However, there was no significant
increase between the early stages (48.2%) and advanced stages
(52.1%; Fig. 1B). Furthermore, the
analysis of NF-ĸB/p65 (pS529) activation by TNM stages revealed:
I=27.3%, II=41.9%, III=60.8% and IV=75.3% (Fig. 1A). There was a significant
increment between the early (34.6%) and advanced stages (68.05%;
Fig. 1C). Therefore, NF-ĸB/p65
(pS529) activation exhibited a positive association with CRC
progression (P<0.05).
 | Table IIIEvaluation of NF-κB/p65 activation in
Tumor-Node-Metastasis stages. |
Table III
Evaluation of NF-κB/p65 activation in
Tumor-Node-Metastasis stages.
| | NF-κB/p65
(pS536) | NF-κB/p65
(pS529) |
|---|
| Stage | activation, % | activation, % |
| Early (I + II) | 48.2 | 34.6 |
|
I | 45.1 | 27.3 |
|
II | 51.3 | 41.9 |
| Advanced (III +
IV) | 52.1 | 68.05 |
|
III | 50.8 | 60.8 |
|
IV | 53.4 | 75.3 |
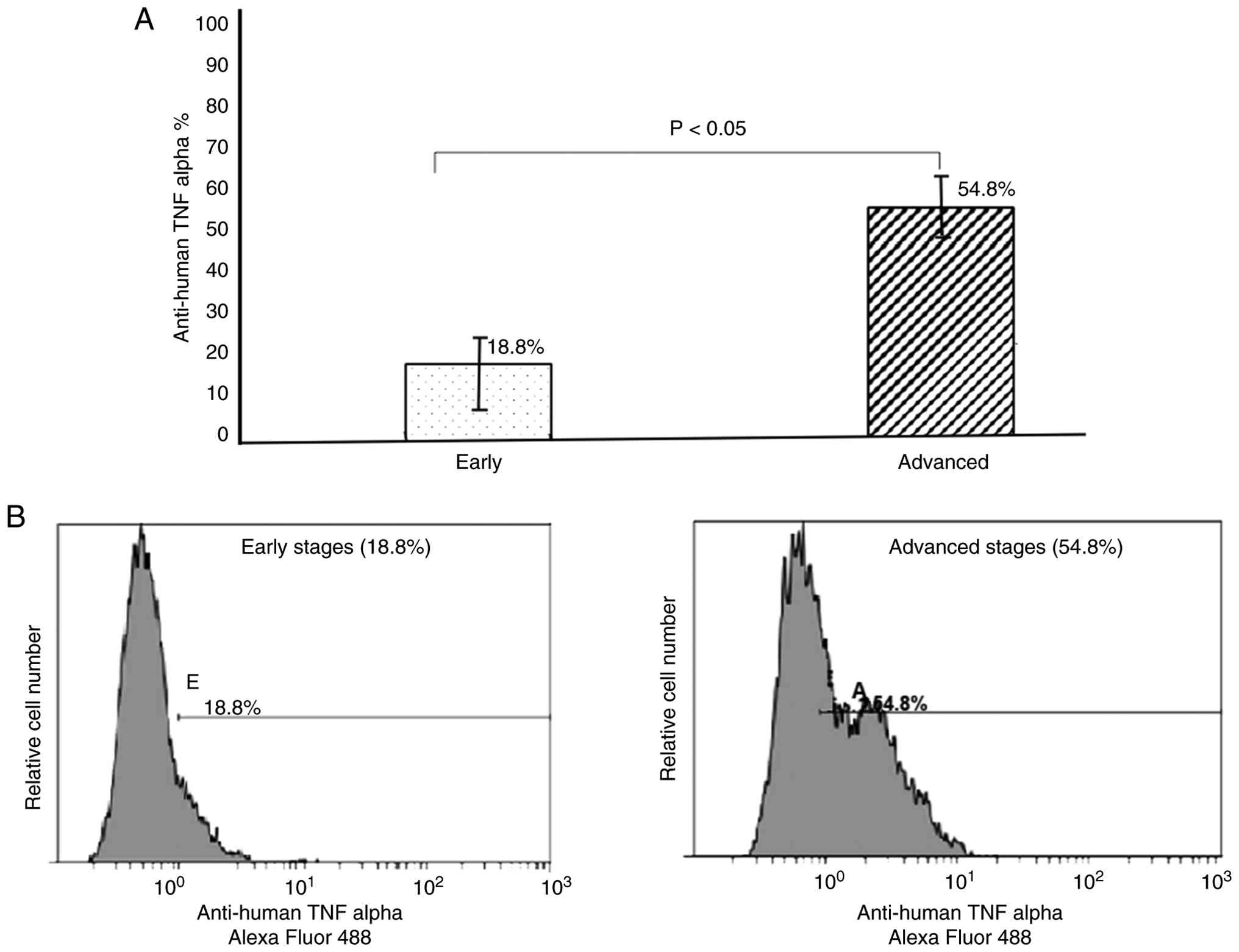
TNF-α cell surface protein is
upregulated in CRC
It was found that both early and advanced CRC-stage
groups exhibited significant upregulation of TNF-α in CRC cells
compared with non-tumoral adjacent mucosa cells, as assessed
through cell surface antigen evaluation. The comparison between
tumor groups revealed a significant increase in TNF-α levels
(E=18.8% and A=54.8%; P<0.05) as the disease progresses, with a
difference of 36.0% (Fig. 2). The
correlation between TNF-α cell surface levels and NF-κB activation
of TNM groups showed a weak positive correlation with p65 (pS536;
ρ=0.216; P<0.05), while the analysis with p65 (pS529) exhibited
a strong positive correlation (ρ=0.801; P<0.05) as revealed in
Table IV. The results closely
matched the comparison between TNF-α serum levels and the
phosphorylated NF-κB/p65 residues, suggesting that the behavior
between TNF-α serum and cell surface antigen levels may be due to
an effect associated with that producing NF-κB activation.
 | Table IVNF-κB/p65 correlation analysis with
the evaluated parameters. |
Table IV
NF-κB/p65 correlation analysis with
the evaluated parameters.
| | Correlation with
NF-ĸB/p65 (pS536) activation | Correlation with
NF-ĸB/p65 (pS529) activation |
|---|
| Parameter | ρ | Correlation | ρ | Correlation |
|---|
| TNF-α cell
surface | 0.216 | Weak positive | 0.801 | Strong
positive |
| TNF-α serum | 0.469 | Moderate
positive | 0.862 | Strong
positive |
| TNFA gene
expression | 0.616 | Moderate
positive | 0.921 | Strong
positive |
| RELA gene
expression | 0.731 | Moderate
positive | 0.891 | Strong
positive |
| NFKB1 gene
expression | 0.104 | Weak positive | 0.294 | Weak positive |
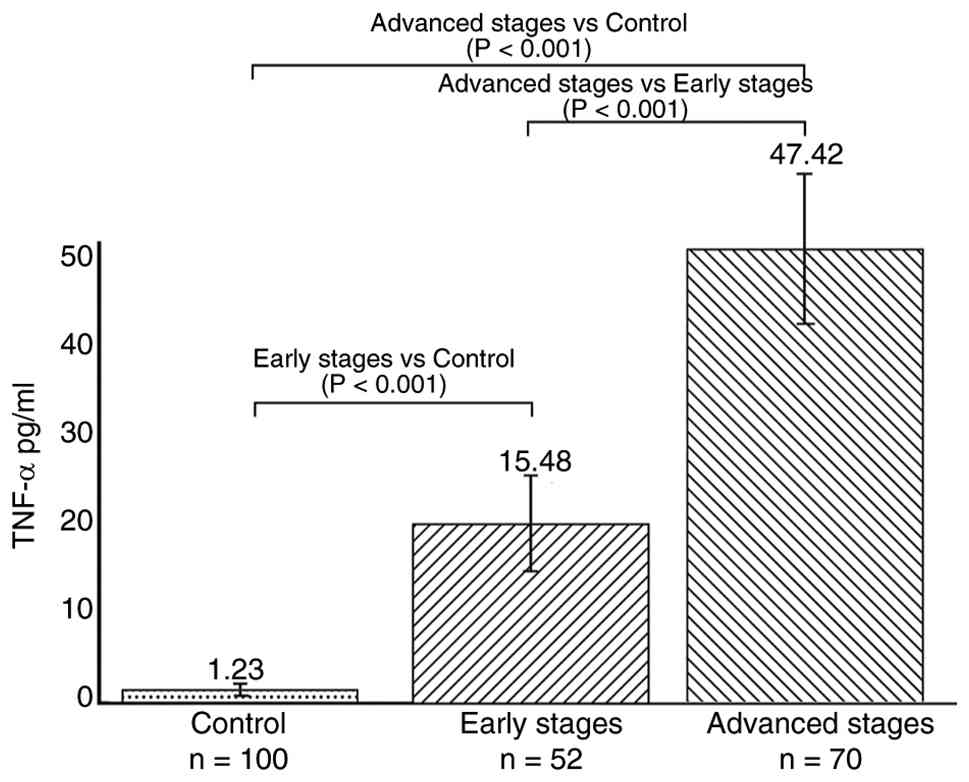
TNF-α serum levels provide early CRC
diagnosis
The cut-off value was calculated using the formula:
Cut-off=Mean concentration of controls + 3 times the standard
deviation to validate the ELISA test. Additionally, the 95th
percentile of the mean was considered, resulting in
Cut-off=1.23+3x0.43)=2.52 pg/ml. This approach is a widely accepted
and effective method for determining the presence of analytes in a
given sample. By employing this method, the reliability and
accuracy of the ELISA test is enhanced, enabling us to identify
valid inferences from the results.
After measuring serum TNF-α levels, the mean values
obtained for each group were as follows: Control group
(C)=1.23±0.43 pg/ml; Early-stage group (E)=15.48±4.2 pg/ml; and
Advanced-stage group (A)=47.42±7.1 pg/ml. The data met the
assumptions of normality and homogeneity of variances, assessed
using the Shapiro-Wilk and Levene's tests, respectively. Therefore,
a one-way analysis of variance (ANOVA) was conducted to compare the
group means. The ANOVA revealed statistically significant
differences in TNF-α levels among the (P<0.0001). Tukey's post
hoc analysis showed that the E group exhibited significantly higher
TNF-α levels compared with that in the C group (P<0.001),
whereas the A group showed significantly higher levels compared
with those in both the E and C groups (P<0.001 in both cases;
Fig. 3).
The correlation between the serum level of TNF-α and
NF-κB activation was assessed based on TNM groups; this analysis
indicated a moderate positive correlation (ρ=0.469; P<0.05) with
p65 (pS536), while a strong positive correlation (ρ=0.862;
P<0.05) was observed with p65 (pS529; Table IV).
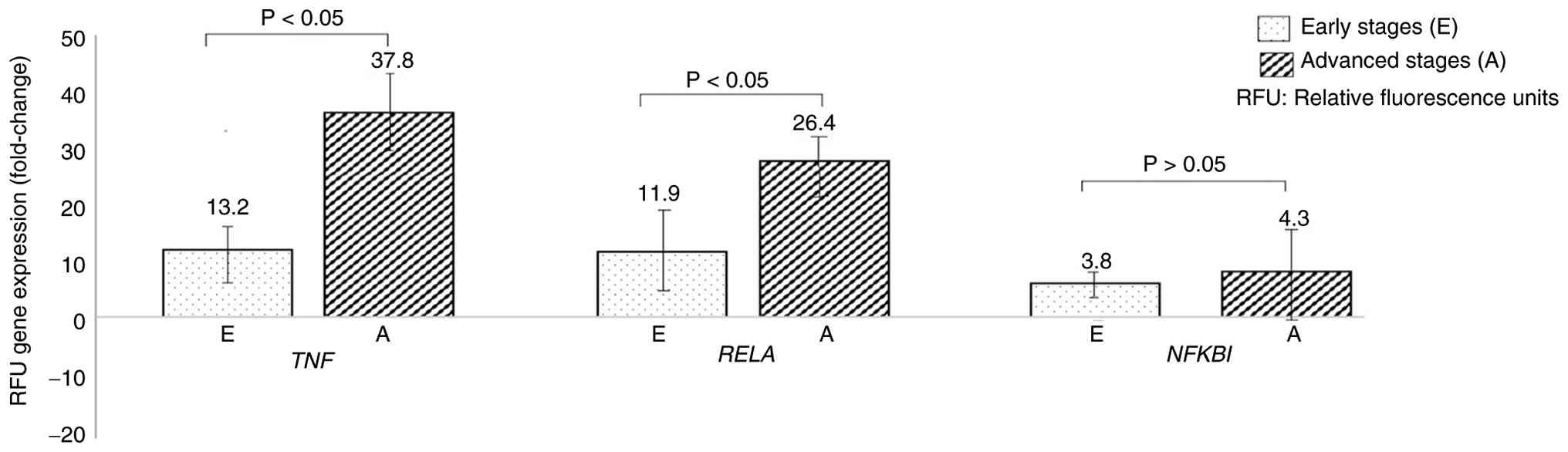
TNF gene expression interacts with the
NF-κB heterodimer genes
Additionally, the impact on gene expression and the
probable interaction of the TNF-α/NF-κB signaling pathway was
determined. To identify the most stable housekeeping gene suitable
for use as an internal control for the qPCR assays, three candidate
genes were compared, namely GUSB, ACTB and
ABL, in both experimental and calibrator samples.
GUSB showed a mean Cq of 23.375±0.61 in experimental samples
compared with 23.638±0.47 in calibrator samples (P=0.429).
ACTB showed a median Cq of 29.785±1.02 in experimental
samples compared with 28.914±1.24 in calibrator samples (P=0.686).
ABL exhibited a median Cq of 28.726±1.57 in experimental
samples and 28.278±1.34 in calibrator samples (P=0.739). These
findings indicate that there were no significant differences in any
case. However, GUSB exhibited minimal standard deviation
among the three genes, making it the most suitable for the present
experiment (data not shown). Therefore, it was selected as the
internal control for qPCR assays.
The Livak method
(2-ΔΔCq) was utilized to quantify
the relative expression of TNFA, RELA and
NFKB1 genes. All genes were upregulated in tumor tissue
compared with that in the adjacent normal mucosa. A significant
increase in TNFA and RELA expression in advanced
stages compared with the early stages was found, with a fold change
of A=37.8, E=13.2 and A=26.4 and E=11.9, respectively (P<0.05).
However, no significant differences were observed in the expression
of NFKB1, with a fold change of A=4.3 and E=3.8 (Fig. 4). These findings suggest that
TNFA and RELA might play a role in the progression of
the disease, whereas NFKB1 might not be associated. The
correlation test between gene expression profiles and NF-κB
activation indicated that TNFA has a moderate positive
correlation with p65 (pS536; ρ=0.616; P<0.05) and a strong
positive correlation with p65 (pS529; ρ=0.921; P<0.05).
Similarly, RELA presented a moderate positive correlation
with p65 (pS536; ρ=0.731; P<0.05) and a strong positive
correlation with p65 (pS529; ρ=0.891; P<0.05). Conversely,
NFKB1 revealed a weak positive correlation with both p65
(pS536) (ρ=0.104; P<0.05) and p65 (pS529; ρ=0.294; P<0.05).
These findings are summarized in Table IV.
Discussion
CRC is characterized by dysregulated inflammation
stimulus. An essential pro-inflammatory molecule involved is NF-κB,
which promotes gene transcription in tumorigenesis (23). The NF-κB/p65 subunit
phosphorylation is a critical post-translational activation
mechanism triggered by TNF-α stimulation in cancer development
(23-25).
Recent studies demonstrated that dysregulated NF-κB activity
contributes to tumor progression and confers drug resistance by
inhibiting apoptosis (26). The
present study was designed to evaluate NF-κB activity in patients
with sporadic CRC by measuring the phosphorylation of its subunit
p65 residues (Ser529) and (Ser536) together with TNF-α cell surface
protein, serum and gene expression levels. The goal was to
determine whether there is a correlation between the NF-κB
activation status and TNF-α levels in normal adjacent mucosa
compared with tumor tissue. Patients were grouped according to TNM
classification in early stages (I + II=E) and advanced stages (III
+ IV=A) to statistically analyze the disease progression. The
validation of each experiment performed in this project was a
fundamental part of reporting the obtained results.
NF-κB/p65 has been frequently reported to be
upregulated in CRC. However, most studies have not compared normal
adjacent mucosa with tumor tissue from the same patient or analyzed
the results by comparison with the TNM classification (27-29).
Berkovich et al (30)
utilized immunohistochemistry to compare the expression of NF-κB in
colonic adenocarcinoma specimens, colonic adenomas and inflammatory
colonic tissues. The study observed a similar expression level
between polypoid and inflammatory cases, but it was significantly
higher in CRC than in both. The findings support the role of NF-κB
activity early in the adenoma-to-carcinoma sequence, consistent
with the results of the present study. Although the researchers did
not evaluate the phosphorylated residues associated with the
analysis, valuable findings were provided (30). Lewander et al (25,31)
conducted two studies to assess the activation of NF-κB p65 (pS536)
in Swedish patients with CRC using immunohistochemistry. NF-κB p65
(pS536) activation was higher in patients with CRC than in normal
mucosa. Both studies concluded that NF-κB p65 (pS536) is an
independent prognostic factor in patients with CRC, but it is not
directly associated with the response of radiotherapy based on
recurrence and survival (25). In
accordance with these studies, the authors of the present study had
previously published a study (15), in which upregulation of
NF-κB/p65(pS536) in the tumor tissue was observed compared with the
normal adjacent mucosa using immunohistochemistry in sporadic CRC.
However, p65 (pS529) could not be statistically analyzed due to the
limited quantity and quality of samples available during evaluation
(15). According to previous
studies, the nuclear immunopositivity of NF-κB/p65 in CRC cells has
been widely reported and linked to their high transcriptional
activity (25,31-33).
However, conflicting findings have suggested that NF-κB is
consistently present in the cytoplasm (15,31),
which is contradictory to the predicted outcomes. The
immunohistochemistry technique commonly used for diagnosis implies
that the number of cells analyzed is a limiting factor in research
experiments. Due to unexpected and controversial findings of some
recent studies (15,16,25,31-33),
an experimental design with larger sample groups to compare and use
of phospho-flow cytometry to analyze a more significant and
specific number of cells (20,000 events), are proposed.
Phospho-flow cytometry is a reliable method that
consistently and reproducibly measures levels of phosphorylated
proteins in single cells. This technique has recently been used for
screening intracellular signaling events in different diseases
(34-37).
In 2023, Toney et al (34)
applied the method in a CRC model, measuring the phosphorylation of
STAT1, STAT3 and STAT5, induced by IL-6, IL-10 and IL-2,
respectively. The experimental design of the present study involved
significant challenges, as the authors were not able to find
previous studies that used the technique to analyze NF-κB
phosphorylated residues in CRC tissue. The fluorescence-labeled
antibodies used in the present study recognized NF-κB only when
phosphorylated on the specific amino acid residues (pS536) and
(pS529), which strongly indicated the activation of the
transcription factor. Previously, the authors of the present study
have reported such evaluation in cell cultures of different
pediatric leukemias with replicable results (38,39).
Based on these precedents, a protocol for efficiently separating
CRC cells was standardized for analysis by phospho-flow cytometry,
obtaining the results reported in the present study. Furthermore,
the present study revealed that both NF-κB/p65 (pS536) and (pS529)
are significantly active in patients with sporadic CRC. The TNM
analysis conducted on the results indicated that p65 (pS536) is
strongly correlated to the onset of CRC, while p65 (pS529) is
associated with CRC progression; these findings underscore the
importance of how phospho-flow cytometry can integrate clinical and
histopathological analysis with phospho-protein detection, which
provides an advantage over conventional techniques in monitoring
the activity of these biomarkers for early diagnosis and as
indicators of progression. To the best of the authors' knowledge,
this is the first study that standardizes and proposes a protocol
for evaluating NF-κB/p65 (pS536) and (pS529) in CRC tissue and
adjacent normal mucosa cells.
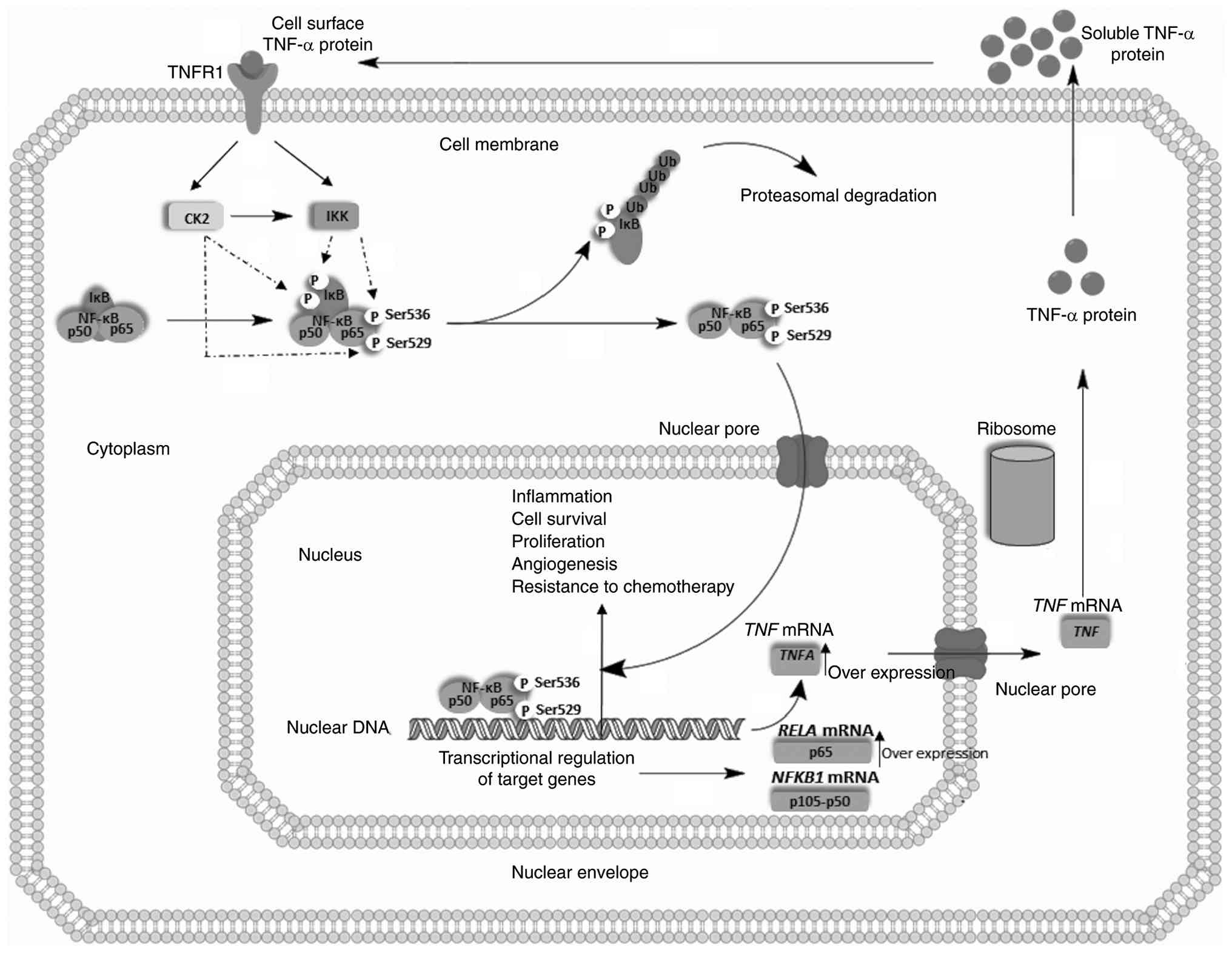
A summary of the findings of the present study is
described below along with a proposed model presented in Fig. 5. The role of TNF-α in the
inflammatory stimulus leading to solid tumor development is well
established (40-42).
The present study provided substantial evidence of a gradual
increase in cell surface TNF-α expression as CRC progresses. The
results of the TNF-α cell surface analysis revealed a weak positive
correlation with p65 (pS536), while a strong positive correlation
was observed with p65 (pS529). These findings confirm an
association between TNF-α and p65 (pS536), which is produced by IKK
activity and is involved in the early stages of CRC. On the other
hand, the signaling of TNF-α and p65 (pS529) mediated by the
dysregulated activity of CK2 after TNF-α stimulus is critical to
the progression of CRC; concurrently, CK2 potentiates IKK activity.
The phosphorylation of IκB promotes its degradation, and
consequently, NF-κB is released from the IκB inhibitory complex.
NF-κB is translocated into the nucleus, resulting in the regulation
of biological processes associated with tumorigenesis. NF-κB
transcriptionally regulates the genes that codify to p65
(RELA) and p50 subunit (NFKB1). The TNFA gene
is also regulated by NF-κB, and it is found to be overexpressedin
the sporadic CRC model. TNF mRNA is exported to the cytoplasm and
translated into the functional TNF-α protein in the ribosome. The
tumoral cell identifies the increase in the TNF-α protein level and
exports it out of the cell. The soluble TNF-α level is now
increased, causing the proposed exponentially dysregulated
TNF-α/NF-κB signaling in sporadic CRC. Cell surface proteins play
an essential role in biomedical research due to their ability to
serve as cellular markers and their extracellular accessibility for
pharmacological intervention (Fig.
5). Despite their importance, information regarding individual
cell surface protein repertoires, known as ‘the surfaceome’ remains
limited (43). By evaluating the
level of TNF-α on cell surfaces, the present study has revealed
crucial insight into the molecular signaling of sporadic CRC.
Therefore, the authors strongly recommend to include this
assessment in any comprehensive analysis of sporadic CRC to better
understand its molecular mechanisms.
Previous studies indicated that TNF-α gene
expression levels are significantly higher in patients with CRC and
associated with advanced stages (44,45).
Additionally, an increase of soluble TNF-α was observed in the
serum of these patients. It has been reported that patients with
low TNF-α serum levels have a significantly higher survival rate
than those with high levels (44,46).
The present study found that individuals with sporadic CRC had
considerably higher levels of TNF-α in serum than healthy controls.
Furthermore, within the tumoral group, there was a significant
difference in TNF-α levels between patients in advanced stages and
those in early stages. The study examined the association between
TNF-α serum levels and NF-κB activation of TNM groups. The analysis
showed a moderate positive correlation between TNF-α serum levels
and p65 (pS536) activation and a strong positive correlation with
p65 (pS529) activation. These findings suggest that TNF-α serum
levels are associated with NF-κB activation, which is known to play
a key role in tumor development and progression (Fig. 5). Overall, the results indicated
that TNF-α serum levels could potentially serve as a predictive
biomarker for CRC development, and the correlation with NF-κB
activation further supports the involvement of TNF-α in CRC
pathogenesis.
Similarly, increased levels of the TNF-α gene
in CRC tissue and significant upregulation in advanced stages
compared with early stages were observed. The correlation test
between gene expression profiles and NF-κB activation indicated
that TNF-α was strongly correlated with CRC progression.
Upregulation of the RELA and NFKB1 genes indicated a
molecular feedback mechanism during the inflammatory response.
These findings reinforced that the NF-κB pathway dysregulation
impacts the transcriptional, translational and replicational levels
(Fig. 5).
Despite its established relationship with various
pathologies, there is a lack of clinically useful research on the
TNF-α/NF-κB level profile. The results of the present study
highlighted the importance of TNF-α/NF-κB interaction in CRC;
therefore, its evaluation has potential as a biomarker in
predicting CRC prognosis and development. Such predictive ability
can allow for proactive measures to be taken in detecting and
treating the disease earlier, which can positively impact patient
outcomes. Identifying biomarkers in this context can aid in the
early detection and timely intervention of CRC, a disease that
globally poses significant health threats. With these
considerations in mind, it is crucial to continue pursuing research
into the potential of TNF-α assessment as a predictive biomarker
for CRC. The implications of these findings are significant, as
they could lead to the development of cost-effective and
non-invasive screening methods for CRC in Mexico and in other
developing nations.
In conclusion, the present study provides important
insights into the relationship between TNF-α/NF-κB signaling and
CRC development. It is demonstrated that assessing TNF-α/NF-κB
activity is of utmost importance for patients diagnosed with
sporadic CRC. The findings revealed that TNF-α and NF-κB/p65 have a
positive correlation in the development of CRC. Moreover, the
analysis of TNM groups suggests that the correlation between TNF-α
and p65 (pS536) plays a crucial role in the early stages,
contributing to the initiation and establishment of CRC. On the
other hand, TNF-α and p65 (pS529) signaling are relevant to the
advanced stages as well, which implies that they may play a role in
the growth and spread of CRC. Overall, the present study
highlighted the significance of TNF-α/NF-κB activity as a potential
biomarker for monitoring and predicting CRC progression. It is
essential to consider the potential limitations of this study. The
methodological limitations are related to the relative
quantification design of the data. It is important to note that
while the control group patients did not have a diagnosis of
adenocarcinoma of the colon or rectum, they might still have
irregular levels of inflammatory markers. Despite these
limitations, the invasiveness of the procedure for collecting the
colorectal sample makes these individuals the ideal participants to
be controls for the study. Additionally, the measurement of IKK and
CK2 kinases, which are proposed as critical regulators of NF-kB
activation, should be considered, as well as other residues
phosphorylated by these kinases.
It was proposed that monitoring TNF-α/NF-κB activity
could be a valuable parameter in managing CRC, thus increasing the
percentage of success and improving the quality of life of the
patient. Specifically, determining TNF-α serum levels could
potentially be used as a diagnostic factor for early stages instead
of relying on other, more invasive tests. This approach is
particularly interesting due to the growing incidence of CRC
worldwide. Indeed, detecting CRC in its early stages is critical
for successful treatment outcomes. In light of these findings, the
potential benefits of monitoring TNF-α/NF-κB activity and their
molecular interactions in managing CRC must be further
analyzed.
Acknowledgements
The authors would like to thank Dr F.
Bustos-Rodríguez and Dr J.A.Valenzuela-Pérez (Department of
Oncological Pathology, National Cancer Institute, Guadalajara,
Mexico) for technical support in the present study.
Funding
Funding: The present study was partially supported by the State
Council of Science and Technology of Jalisco (grant no.
5-2010-1-1083; COECYTJAL; Guadalajara, México) and the National
Council of Science and Technology (grant no. 590826/305844 CONACYT;
México).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
UFSB participated in the study design, collection of
the tissues, analysis and interpretation of the experimental data,
as well as performed immunohistochemical experiments and RNA
isolation, and was a significant contributor to the writing of the
manuscript. DFS participated in the study design and collection of
the tissues, analyzed and interpreted the experimental data,
performed gene expression analysis and was a significant
contributor to the writing of the manuscript. LBM participated in
the study design, analysis and interpretation of the experimental
data and was involved in drafting the manuscript and revising it
critically for important intellectual content. ABJ participated in
the collection of the tissues, analyzed and interpreted the patient
data and validated the gene expression assays. JAVP coordinated the
clinicopathological diagnosis of the patients, participated in the
surgeries of the patients, classified the samples according to the
criteria of the study and revised the manuscript. JRCR made a
substantial contribution to the conception and design of the study
and was involved in drafting the manuscript and revising it
critically for important intellectual content. ACR coordinated the
present study, contributed substantially to its conception and
design, as well as the analysis and interpretation of the
experimental and clinicopathological data. ACR was also involved in
drafting the manuscript and revising it critically for important
intellectual content, and providing the final approval of the
version to be published. UFSB, LBM and ACR confirm the authenticity
of all the raw data. All authors read and approved the final
version of the manuscript.
Ethics approval and consent to
participate
The present study was performed following the
Declaration of Helsinki and was approved by the ‘Hospital Civil de
Guadalajara Ethics Committee’ (approval no. 21711; Guadalajara,
Mexico). The hospital Ethics Committee is affiliated to the ‘Ethics
Committee from the State of Jalisco’ with investigation no.
77/UG-JAL/2011. The present study was registered at the National
Bioethics Commission with the no. CONBIOÉTICA
14-CEI-008-20161212. The patients provided written and verbal
informed consent for scientific purposes to publish any associated
data from the present study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Xi Y and Xu P: Global colorectal cancer
burden in 2020 and projections to 2040. Transl Oncol.
14(101174)2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Siegel RL, Miller KD, Fedewa SA, Ahnen DJ,
Meester RGS, Barzi A and Jemal A: Colorectal cancer statistics,
2017. CA Cancer J Clin. 67:177–193. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Espinosa-Tamez P, Suazo-Zepeda E,
Sánchez-Blas H, Meneses-Medina M, Huitzil-Meléndez FD, Van Loon K,
Potter M and Lajous M: National and state-level colorectal cancer
mortality trends in Mexico, 1998-2018. Salud Publica Mex. 64:5–13.
2021.PubMed/NCBI View
Article : Google Scholar
|
|
4
|
Li J, Ma X, Chakravarti D, Shalapour S and
DePinho RA: Genetic and biological hallmarks of colorectal cancer.
Genes Dev. 35:787–820. 2021.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Bhat AA, Nisar S, Singh M, Ashraf B,
Masoodi T, Prasad CP, Sharma A, Maacha S, Karedath T, Hashem S, et
al: Cytokine- and chemokine-induced inflammatory colorectal tumor
microenvironment: Emerging avenue for targeted therapy. Cancer
Commun (Lond). 42:689–715. 2022.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Zhang T, Ma C, Zhang Z, Zhang H and Hu H:
NF-κB signaling in inflammation and cancer. MedComm (2020).
2:618–653. 2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Yu H, Lin L, Zhang Z, Zhang H and Hu H:
Targeting NF-κB pathway for the therapy of diseases: Mechanism and
clinical study. Signal Transduct Target Ther. 5(209)2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Mitchell S, Tsui R, Tan ZC, Pack A and
Hoffmann A: The NF-κB multidimer system model: A knowledge base to
explore diverse biological contexts. Sci Signal.
16(eabo2838)2023.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Prescott JA, Mitchell JP and Cook SJ:
Inhibitory feedback control of NF-κB signalling in health and
disease. Biochem J. 478:2619–2664. 2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Bacher S, Meier-Soelch J, Kracht M and
Schmitz ML: Regulation of transcription factor NF-κB in its natural
habitat: The nucleus. Cells. 10(753)2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Hayden MS and Ghosh S: Regulation of NF-κB
by TNF family cytokines. Semin Immunol. 26:253–266. 2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Moreno-Lorenzana D, Torres-Barrera P,
Flores-Lopez G, Chávez-González MA, Isordia-Salas I, Yoder MC,
Majluf-Cruz A and Alvarado-Moreno JA: Self-regulation of TNF-α
induces dysfunction of endothelial colony-forming cells from
patients with venous thromboembolic disease. Arch Med Res.
53:680–687. 2022.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Mattioli I, Sebald A, Bucher C, Charles
RP, Nakano H, Doi T, Kracht M and Schmitz ML: Transient and
selective NF-kappa B p65 serine 536 phosphorylation induced by T
cell costimulation is mediated by I kappa B kinase beta and
controls the kinetics of p65 nuclear import. J Immunol.
172:6336–6344. 2004.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Tsuchiya R, Tanaka T, Hozumi Y, Nakano T,
Okada M, Topham MK, Iino M and Goto K: Downregulation of
diacylglycerol kinase ζ enhances activation of cytokine-induced
NF-κB signaling pathway. Biochim Biophys Acta. 1853:361–369.
2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
González-Quezada BA, Santana-Bejarano UF,
Corona-Rivera A, Pimentel-Gutiérrez HJ, Silva-Cruz R,
Ortega-De-la-Torre C, Franco-Topete R, Franco-Topete K,
Centeno-Flores MW, Maciel-Gutiérrez VM, et al: Expression profile
of NF-κB regulated genes in sporadic colorectal cancer patients.
Oncol Lett. 15:7344–7354. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Xu H, Liu T, Li J, Chen F, Xu J, Hu L,
Jiang L, Xiang Z, Wang X and Sheng J: Roburic acid targets TNF to
inhibit the NF-κB signaling pathway and suppress human colorectal
cancer cell growth. Front Immunol. 13(853165)2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Alotaibi AG, Li JV and Gooderham NJ:
Tumour necrosis factor-alpha (TNF-α)-induced metastatic phenotype
in colorectal cancer epithelial cells: Mechanistic support for the
role of MicroRNA-21. Cancers (Basel). 15(627)2023.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Amin MB, Greene FL, Edge SB, Compton CC,
Gershenwald JE, Brookland RK, Meyer L, Gress DM, Byrd DR and
Winchester DP: The eighth edition AJCC cancer staging manual:
Continuing to build a bridge from a population-based to a more
‘personalized’ approach to cancer staging. CA Cancer J Clin.
67:93–99. 2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Patwardhan MB, Samsa GP, McCrory DC,
Fisher DA, Mantyh CR, Morse MA, Prosnitz RG, Cline KE and Gray RN:
Cancer care quality measures: Diagnosis and treatment of colorectal
cancer. Evid Rep Technol Assess. (Full Rep):1–116. 2006.PubMed/NCBI
|
|
20
|
Hawkins AT, Rothman RL, Geiger TM, Canedo
JR, Edwards-Hollingsworth K, LaNeve DC and Penson DF:
Patient-reported outcome measures in colon and rectal surgery: A
systematic review and quality assessment. Dis Colon Rectum.
63:1156–1167. 2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Ali MY, Anand SV, Tangella K, Ramkumar D
and Saif TA: Isolation of primary human colon tumor cells from
surgical tissues and culturing them directly on soft elastic
substrates for traction cytometry. J Vis Exp.
(e52532)2015.PubMed/NCBI View
Article : Google Scholar
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 4:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Wu D, Wu P, Zhao L, Huang L, Zhang Z, Zhao
S and Huang J: NF-κB expression and outcomes in solid tumors: A
systematic review and meta-analysis. Medicine (Baltimore).
94(e1687)2015.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Andersen V, Christensen J, Overvad K,
Tjønneland A and Vogel U: Polymorphisms in NFkB, PXR, LXR and risk
of colorectal cancer in a prospective study of Danes. BMC Cancer.
10(484)2010.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Lewander A, Gao J, Adell G, Zhang H and
Sun XF: Expression of NF-κB p65 phosphorylated at serine-536 in
rectal cancer with or without preoperative radiotherapy. Radiol
Oncol. 45:279–284. 2011.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Kumar A, Singh AK, Singh H, Thareja S and
Kumar P: Regulation of thymidylate synthase: An approach to
overcome 5-FU resistance in colorectal cancer. Med Oncol.
40(3)2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Braumüller H, Mauerer B, Andris J, Berlin
C, Wieder T and Kesselring R: The cytokine network in colorectal
cancer: Implications for new treatment strategies. Cells.
12(138)2022.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Borowczak J, Szczerbowski K, Maniewski M,
Kowalewski A, Janiczek-Polewska M, Szylberg A, Marszałek A and
Szylberg Ł: The role of inflammatory cytokines in the pathogenesis
of colorectal carcinoma-recent findings and review. Biomedicines.
10(1670)2022.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Jana A, Krett NL, Guzman G, Khalid A,
Ozden O, Staudacher JJ, Bauer J, Baik SH, Carroll T, Yazici C and
Jung B: NFkB is essential for activin-induced colorectal cancer
migration via upregulation of PI3K-MDM2 pathway. Oncotarget.
8:37377–37393. 2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Berkovich L, Gerber M, Katzav A, Kidron D
and Avital S: NF-kappa B expression in resected specimen of colonic
cancer is higher compared to its expression in inflammatory bowel
diseases and polyps. Sci Rep. 12(16645)2022.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Lewander A, Gao J, Carstensen J, Arbman G,
Zhang H and Sun XF: NF-κB p65 phosphorylated at serine-536 is an
independent prognostic factor in Swedish colorectal cancer
patients. Int J Colorectal Dis. 27:447–452. 2012.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kojima M, Morisaki T, Sasaki N, Nakano K,
Mibu R, Tanaka M and Katano M: Increased nuclear factor-kB
activation in human colorectal carcinoma and its correlation with
tumor progression. Anticancer Res. 24:675–681. 2004.PubMed/NCBI
|
|
33
|
Berardi R, Maccaroni E, Mandolesi A,
Mantello G, Onofri A, Biscotti T, Pierantoni C, Siquini W,
Marmorale C, Guerrieri M, et al: Nuclear factor-κB predicts outcome
in locally advanced rectal cancer patients receiving neoadjuvant
radio-chemotherapy. Dig Liver Dis. 44:617–622. 2012.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Toney NJ, Schlom J and Donahue RN:
Phosphoflow cytometry to assess cytokine signaling pathways in
peripheral immune cells: Potential for inferring immune cell
function and treatment response in patients with solid tumors. J
Exp Clin Cancer Res. 42(247)2023.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Bitar M, Boldt A, Freitag MT, Gruhn B,
Köhl U and Sack U: Evaluating STAT5 phosphorylation as a mean to
assess T cell proliferation. Front Immunol. 10(722)2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Davies R, Vogelsang P, Jonsson R and Appel
S: An optimized multiplex flow cytometry protocol for the analysis
of intracellular signaling in peripheral blood mononuclear cells. J
Immunol Methods. 436:58–63. 2016.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Canto E, Isobe N and Didonna A: MS-EPIC
Study Group. Hauser SL and Oksenberg JR: Aberrant STAT
phosphorylation signaling in peripheral blood mononuclear cells
from multiple sclerosis patients. J Neuroinflammation.
15(72)2018.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Santana-Bejarano UF, Bobadilla-Morales L,
Mendoza-Maldonado L, Torres-Anguiano E, Brukman-Jiménez SA,
Barba-Barba CC, Corona-Rivera JR and Corona-Rivera A: In
vitro effect of curcumin in combination with chemotherapy drugs
in Ph+ acute lymphoblastic leukemia cells. Oncol Lett.
17:5224–5240. 2019.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Pimentel-Gutiérrez HJ, Bobadilla-Morales
L, Barba-Barba CC, Ortega-De-La-Torre C, Sánchez-Zubieta FA,
Corona-Rivera JR, González-Quezada BA, Armendáriz-Borunda JS,
Silva-Cruz R and Corona-Rivera A: Curcumin potentiates the effect
of chemotherapy against acute lymphoblastic leukemia cells via
downregulation of NF-κB. Oncol Lett. 12:4117–4124. 2016.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Bates RC and Mercurio AM: Tumor necrosis
factor-alpha stimulates the epithelial-to-mesenchymal transition of
human colonic organoids. Mol Biol Cell. 14:1790–1800.
2003.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Ou B, Zhao J, Guan S, Feng H, Wangpu X,
Zhu C, Zong Y, Ma J, Sun J, Shen X, et al: CCR4 promotes metastasis
via ERK/NF-κB/MMP13 pathway and acts downstream of TNF-α in
colorectal cancer. Oncotarget. 30:47637–47649. 2016.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Jovanovic DV, Di Battista JA, Martel J,
Jolicoeur FC, He Y, Zhang M, Mineau F and Pelletier JP: IL-17
stimulates the production and expression of proinflammatory
cytokines, IL-beta and TNF-alpha, by human macrophages. J Immunol.
160:3513–3521. 1998.PubMed/NCBI
|
|
43
|
Bausch-Fluck D, Hofmann A, Bock T, Frei
AP, Cerciello F, Jacobs A, Moest H, Omasits U, Gundry RL, Yoon C,
et al: A mass spectrometric-derived cell surface protein atlas.
PLoS One. 10(e0121314)2015.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Al Obeed OA, Alkhayal KA, Al Sheikh A,
Zubaidi AM, Vaali-Mohammed MA, Boushey R, Mckerrow JH and Abdulla
MH: Increased expression of tumor necrosis factor-α is associated
with advanced colorectal cancer stages. World J Gastroenterol.
20:18390–18396. 2014.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Grimm M, Kim M, Rosenwald A, von Raden BH,
Tsaur I, Meier E, Heemann U, Germer CT, Gasser M and Waaga-Gasser
AM: Tumour-mediated TRAIL-receptor expression indicates effective
apoptotic depletion of infiltrating CD8+ immune cells in clinical
colorectal cancer. Eur J Cancer. 46:2314–2323. 2010.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Stanilov N, Miteva L, Dobreva Z and
Stanilova S: Colorectal cancer severity and survival in correlation
with tumour necrosis factor-alpha. Biotechnol Biotechnol Equip.
28:911–917. 2014.PubMed/NCBI View Article : Google Scholar
|