Introduction
The number of patients receiving cancer chemotherapy
has increased in parallel with the rise in morbidity and mortality
from cancer (1). With the
increased use of oral anticancer drugs, advances in supportive care
for cancer, and changes in the healthcare environment, chemotherapy
has shifted from inpatient to outpatient settings (2,3).
However, cancer drug therapy is associated with a wide variety of
adverse events that can decrease patients' quality of life and
reduce dose intensity. Early intervention by healthcare providers
for adverse events is effective in prolonging survival and
continuing treatment (4).
Collaboration of pharmacists with physicians in providing
pharmacological intervention is effective for maintaining quality
of life, laboratory testing rates and treatment retention (5-7).
Care by the healthcare team is important, and pharmacists provide
safe cancer drug therapy within the healthcare team.
Capecitabine plus oxaliplatin (CAPOX) is a
frequently used treatment regimen for colorectal and gastric cancer
as postoperative adjuvant chemotherapy (8-10).
Treatment consists of capecitabine (1,000 mg/m2 twice
daily on days 1-14 of each cycle) and oxaliplatin (130
mg/m2 intravenously on day 1 of each cycle). Treatment
continues for eight 3-week cycles. CAPOX therapy in postoperative
adjuvant chemotherapy for gastric cancer has been reported to cause
nausea (87% all grades, 10% grade 3/4), peripheral neuropathy (76%
all grades, 33% grade 3/4) and hand-foot syndrome (48% all grades)
in Japanese patients (10).
Maintaining dose intensity is important in postoperative adjuvant
chemotherapy, and adherence to capecitabine is important for this
purpose. Nausea, vomiting and diarrhea are reported to be the main
causes of decreased adherence to CAPOX therapy (11). Since CAPOX therapy is started after
gastrectomy in patients with gastric cancer, special attention
should be paid to decreased adherence and dose intensity due to
gastrointestinal toxicity (10).
Pharmacists are experts in medications, and in
recent years, pharmacists have met with patients to assess the
severity of side effects (11-13).
The Cancer Institute Hospital, Japanese Foundation for Cancer
Research, provides a pharmacy outpatient clinic for patients
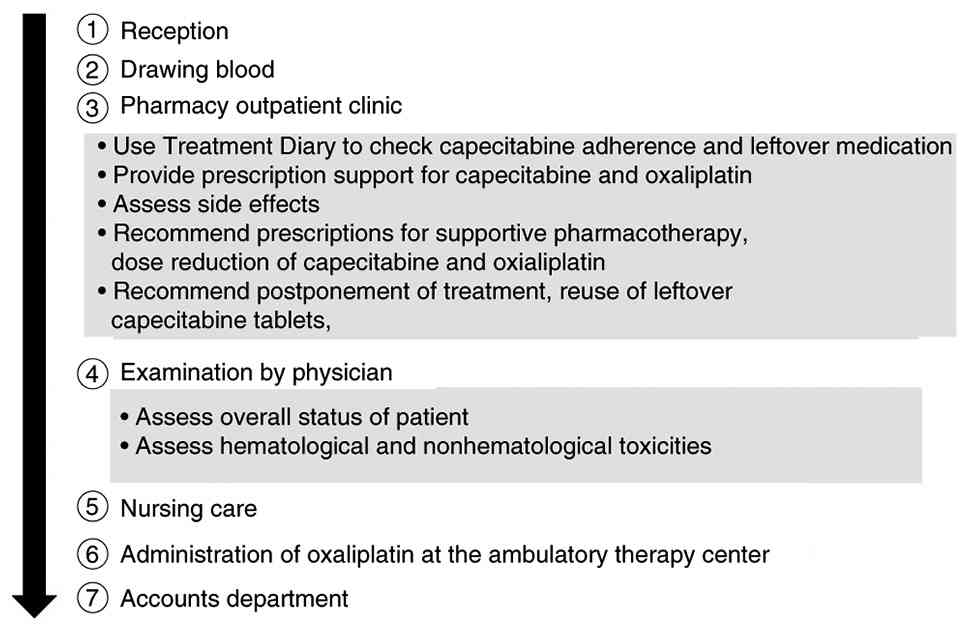
undergoing outpatient chemotherapy (Fig. 1). In The Cancer Institute Hospital,
proactive intervention by pharmacists to check for adherence and
side effects (which are one cause of nonadherence) is considered
necessary, and a pharmacy outpatient clinic is held for patients
receiving oral chemotherapy treatment. The main tasks related to
CAPOX performed in the pharmacy outpatient clinic are as follows:
i) Use of the capecitabine treatment diary to check for
capecitabine adherence and leftover medication; ii) provision of
prescription support for CAPOX treatment; iii) assessment of side
effects; and iv) suggestion of prescriptions for medication as
supportive therapy (13). In
practice, prescription support for CAPOX treatment consists of a
pharmacist recording the CAPOX doses and the administration period
required for the next cycle in the electronic medical record (EMR).
This is then checked and authorized by a physician to enable the
prescription to be issued. If capecitabine is left over from the
previous cycle, the amount to be prescribed is adjusted
accordingly. There have been reports of a decrease in the rate of
inquiries related to capecitabine as a result of pharmacy
outpatient clinics, in addition to decreases in emergency visits
and emergency hospitalizations (14,15).
However, there are no studies of pharmacists designing
prescriptions of anticancer drugs for physicians, which contributed
to patient care. Therefore, in the present study, dose intensity
and severity of side effects of CAPOX therapy as adjuvant
chemotherapy after surgery for gastric cancer before and after
outpatient pharmacy clinic follow-up were evaluated.
Study design and treatment
This was a retrospective, observational study.
Patients with pathological Stage II/III gastric cancer who
underwent D2 gastrectomy were included. CAPOX was selected based on
the evidence from the CLASSIC trial and in accordance with the
institutional treatment protocols approved by the Regimen Review
Committee of The Cancer Institute Hospital, which designated CAPOX
as a preferred combination therapy for patients at high risk of
recurrence during the study period (16). Data were obtained from consecutive
patients who received CAPOX treatment as adjuvant therapy for
gastric cancer. The CAPOX regimen comprised a 2-h intravenous
infusion of oxaliplatin 130 mg/m2 on day 1, and oral
capecitabine at 1,000 mg/m2 twice daily was given for 14
days on a 3-week cycle. Interviews were held at the pharmacy
outpatient clinic between November 1, 2015 and April 30, 2021 at
the Cancer Institute Hospital of the Japanese Foundation for Cancer
Research using EMRs. The exclusion criterion was limited to
patients for whom the RDI could not be calculated accurately due to
unknown leftover capecitabine amounts; consequently, two patients
were excluded. The Clinical Research Ethics Review Committee of the
Japanese Foundation for Cancer Research Cancer Institute Hospital
approved the present study (approval no. 2021-GB-017). Following
this approval, data for this retrospective analysis were extracted
from EMRs between November 2021 and March 2023.
Statistical analysis
All analyses were prespecified based on the study
objectives. Given the retrospective, observational design and small
sample size, the results are presented primarily using descriptive
statistics (mean, percentage, and event counts). Results are
presented as the mean ± SD [95% confidence interval (CI)]. The 95%
CIs for the mean relative dose intensities (RDIs) were calculated
using the standard method for a single sample mean.
No formal hypothesis testing was performed to
compare the RDIs or adverse event counts before and after the
pharmacist intervention. The comparison of pre- and
post-intervention data serves only to show descriptive changes and
potential associations, not to establish statistical significance
or causality. Statistical analyses were performed using JMP Pro
version 14 (SAS Institute Japan).
Pharmacy outpatient clinic and
prescription proposals by pharmacists to physicians
In the pharmacy outpatient clinic of the Cancer
Institute Hospital of the Japanese Foundation for Cancer Research,
pharmacists checked adherence to oral anticancer drugs and assessed
side effects, which are one cause of non-adherence, before the
patients were examined by their physicians (12,13).
In the pharmacy outpatient clinic, pharmacists conducted direct
patient interviews and assessments. In particular, the pharmacists
played two important roles, confirmation and suggestion.
Confirmation involved checking the patients' adherence to
capecitabine and evaluating the side effects. Suggestion involved
including suggestions in patients' EMRs regarding the most
effective prescription for supportive pharmacotherapy, the timing
of the next anticancer drug dose, and the administration regimen.
Recommendations for prescriptions at the pharmacy outpatient clinic
from the second cycle to the end of treatment were assessed. The
number of prescription recommendations of medications for
supportive care, reuse of leftover capecitabine, postponement of
treatment, dose reduction of each drug, and suspension of
oxaliplatin were evaluated. The EMR was utilized as a tool for
checking patient history, documenting adherence and side effects,
and communicating prescription suggestions to physicians. No
artificial intelligence was involved in the patient interview or
assessment process during the study period.
Number of occurrences of Grade ≥3
non-hematological toxicity before and after pharmacist-suggested
prescription of supportive care medications and
pharmacist-suggested dose reduction of anticancer drugs
The number of Grade ≥3 non-hematological toxicity
events over the treatment period was examined. Adverse events were
evaluated based on pharmacist assessments. Of these, Grade ≥3
non-hematological adverse events for which supportive care
medications were proposed and Grade ≥3 non-hematological toxicity
in the next course after the proposal were evaluated. Grade ≥3
non-hematological toxicity for which dose reductions of each drug
were proposed were also investigated. Adverse events were graded
according to the Common Terminology Criteria for Adverse Events
version 4.0 (CTCAE v4.0; https://ctep.cancer.gov/protocoldevelopment/electronic_applications/ctc.htm#ctc_40).
RDI before and after
pharmacist-proposed CAPOX dose reduction
The number of recommendations for dose reduction of
CAPOX from the second course to the end of treatment was studied.
The RDI was calculated before and after a pharmacist-recommended
dose reduction of each drug and subsequent physician-prescribed
dose reduction of each drug. The RDI was calculated based on
previous studies (10,17,18).
In the present study, the RDI was calculated with consideration of
treatment delays and dose reductions. RDI (%) was defined as: RDI
(%)=(Actual dose intensity/Planned dose intensity) x100.
The planned dose intensity was calculated by
dividing the cumulative dose (mg/m²) that would have been
administered if the treatment had been completed without any dose
reductions or delays by the planned treatment duration (days). The
actual dose intensity was calculated by dividing the actual
cumulative dose (mg/m²) administered by the actual treatment
duration (days), including any delays or interruptions. This method
reflects the impact of both dose modifications and treatment
schedule changes on the overall intensity of chemotherapy. The
overall RDI was calculated based on the total planned and actual
doses over the entire treatment course, whereas the RDI before and
after the dose-reduction recommendation was calculated using the
data from only one course prior to and one course following the
intervention.
Results
Patients and characteristics
Data were obtained from 61 consecutive patients with
gastric cancer. Two patients were excluded from the final analysis
due to an unclear number of capecitabine tablets taken, leading to
a final total of 59 patients included. Patient characteristic data
for the 59 patients analyzed are reported in Table I. The median age was 61 years
(range: 23-77 years), and the study included 40 male and 19 female
patients. In terms of surgical procedure, 23 (39.0%) underwent
total gastrectomy, 33 (55.9%) underwent distal gastrectomy, 1
(1.7%) underwent cardiac gastrectomy, and 2 (3.4%) underwent other
procedures. The mean duration of CAPOX treatment was 7.39 cycles
(range: 1-8 cycles).
 | Table IClinicopathological characteristics of
patients (total n=59). |
Table I
Clinicopathological characteristics of
patients (total n=59).
| Clinicopathological
characteristics | Number of patients, n
(%) |
|---|
| Median age, years
(range) | 61.0 (23-77) |
| Sex | |
|
Male | 40 (67.8) |
|
Female | 19 (32.2) |
| Eastern Cooperative
Oncology Group performance status | |
|
0 | 30 (50.8) |
|
1 | 29 (49.2) |
| Pathological
stage | |
|
IIA | 4 (6.8) |
|
IIB | 29 (49.1) |
|
IIIC | 23 (39.0) |
|
Others | 3 (5.1) |
| Surgical
procedure | |
|
Total
gastrectomy | 23 (39.0) |
|
Distal
gastrectomy | 33 (55.9) |
|
Cardiac
gastrectomy | 1 (1.7) |
|
Others | 2 (3.4) |
| Duration of use
(cycle) | |
|
Mean
(SD) | 7.39 (1.59) |
|
Range | 1-8 |
| Creatinine clearance
(ml/min) | |
|
Median
(Range) | 88.3
(38.4-168.2) |
Prescription recommendations at the
pharmacy outpatient clinic to physicians
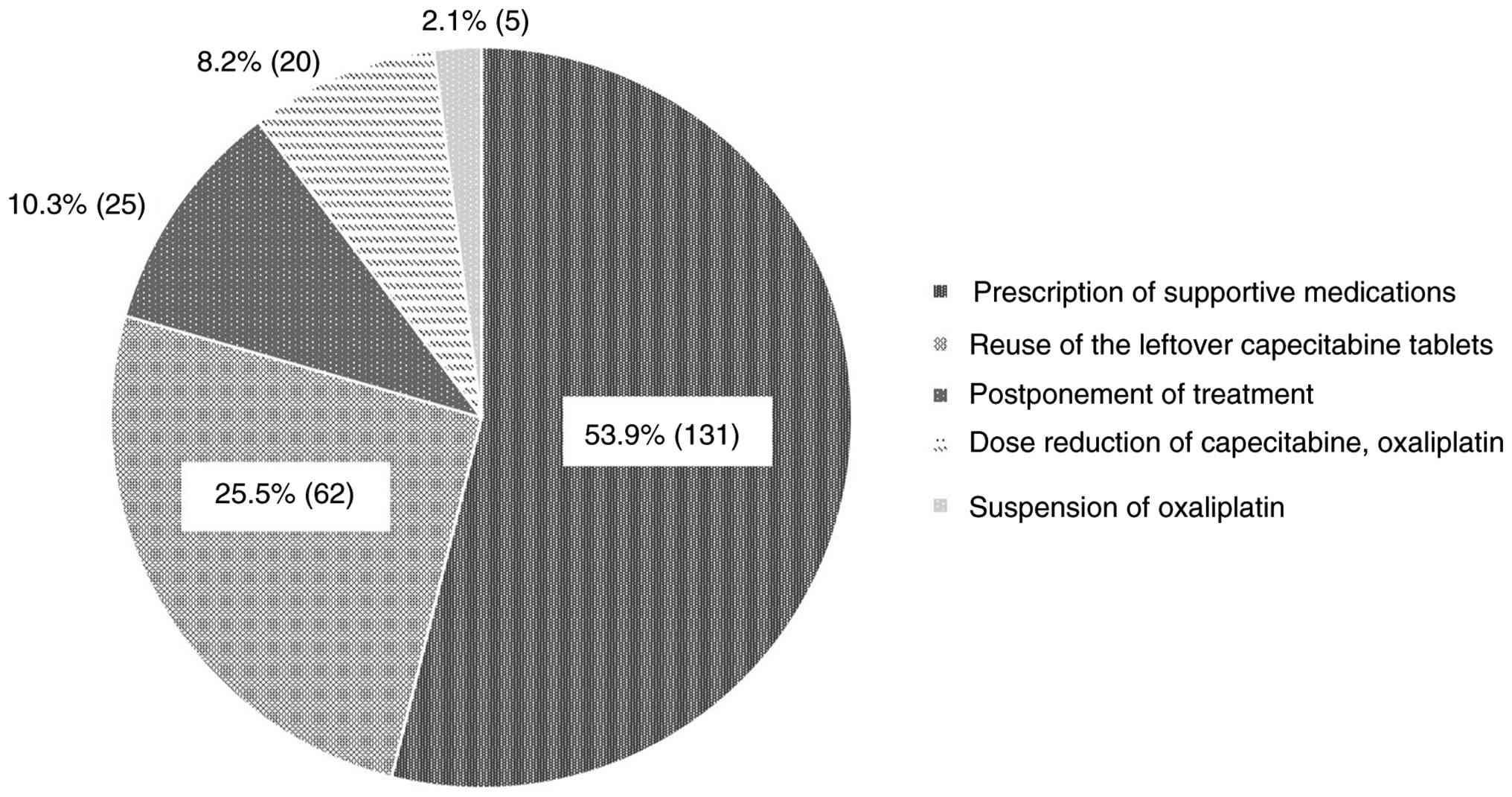
In total, prescription recommendations at the
pharmacy outpatient clinic were observed in 243 instances. (Note:
‘Instances’ refers to the total number of intervention events,
including multiple recommendations made for a single patient,
reflecting the frequency and workload of pharmacist intervention).
The most common prescription recommendation was prescription of
supportive medication (53.9%, 131 instances), followed by reuse of
the leftover capecitabine tablets (25.5%, 62 instances),
postponement of treatment (10.3%, 25 instances), dose reduction of
each drug (8.2%, 20 instances) and suspension of oxaliplatin (2.1%,
5 instances) (Fig. 2). Supportive
care medications for adverse events were recommended for nausea and
vomiting (37.4%, 49 instances), hand-foot syndrome (24.4%, 32
instances), diarrhea (7.6%, 10 instances), peripheral neuropathy
(4.6%,6 instances), anorexia (2.3%, 3 instances) and others (23.7%,
31 instances).
Grade ≥3 non-hematological adverse
events before and after recommended prescriptions for supportive
care medication and recommended dose reduction of each drug
In total, Grade ≥3 non-hematological toxicities were
observed in 25 instances. The most common Grade ≥3
non-hematological toxicities were hand-foot syndrome (32.0%, 8
instances), peripheral neuropathy (32.0%, 8 instances), fatigue
(12.0%, 3 instances), diarrhea (8.0%, 2 instances), nausea (4.0%, 1
instance), constipation (4.0%,1 instance), and others (8.0%, 2
instances) (Table II). Others
included oxaliplatin hypersensitivity (4.0%, 1 instance) and
gastrointestinal toxicity (4.0%, 1 instance). The number of Grade
≥3 non-hematological toxicities was counted for one treatment
course before and one course after the pharmacists'
recommendations. This comparison reflects short-term changes in
toxicity severity following dose adjustment proposals. However, it
does not account for long-term toxicity trends or cumulative
effects over the entire treatment duration. Prescription of
supportive care medications was recommended for 5 instances of
Grade ≥3 hand-foot syndrome and 1 instance of Grade ≥3 peripheral
neuropathy. Dose reduction of capecitabine was recommended for 4
instances of Grade ≥3 hand-foot syndrome and 1 instance of Grade ≥3
fatigue. In the next course, after the recommended prescription of
supportive care medications, Grade ≥3 hand-foot syndrome decreased
to 1 instance. Similarly, after the recommended dose reduction of
capecitabine, Grade ≥3 hand-foot syndrome and fatigue were not
observed (Table III).
 | Table IIPrescription recommendations for Grade
3≥ non-hematological adverse events by phamacists. |
Table II
Prescription recommendations for Grade
3≥ non-hematological adverse events by phamacists.
| Grade 3≥
non-hematologic toxicities | Number of instances
(n=25) | Number of
intervention instances | Prescription of
supportive medication | Dose reduction of
capecitabine | Both
recommendations | Others |
|---|
| Hand-foot
syndrome | 8 | 8 | 3 | 2 | 2 | 1 |
| Peripheral
neuropathy | 8 | 7 | 1 | 0 | 0 | 6 |
| Fatigue | 3 | 2 | 0 | 1 | 0 | 1 |
| Diarrhea | 2 | 0 | 0 | 0 | 0 | 0 |
| Nausea | 1 | 1 | 0 | 0 | 0 | 1 |
| Constipation | 1 | 1 | 0 | 0 | 0 | 1 |
| Others | 2 | 0 | 0 | 0 | 0 | 0 |
 | Table IIIThe number of Grade ≥3
non-hematological adverse events before and after pharmacist's
recommendations. |
Table III
The number of Grade ≥3
non-hematological adverse events before and after pharmacist's
recommendations.
| | Number of Grade ≥3
non-hematological toxicities |
|---|
| Intervention |
Pre-recommended |
Post-recommended |
|---|
| Recommended
prescription of supportive medication | | |
|
Hand-foot
syndrome | 5 | 1 |
|
Peripheral
neuropathy | 1 | 1 |
|
Fatigue | 0 | 0 |
|
Subtotal | 6 | 2 |
| Recommended dose
reduction of capecitabine | | |
|
Hand-foot
syndrome | 4 | 0 |
|
Peripheral
neuropathy | 0 | 0 |
|
Fatigue | 1 | 0 |
|
Subtotal | 5 | 0 |
RDI before and after dose reduction of
each drug
All 14 recommendations for capecitabine dose
reduction by pharmacists were reflected in the physicians'
prescriptions for dose reduction. The mean RDI was 72.5±18.8% [95%
confidence interval (CI), 61.2-83.7%] before capecitabine dose
reduction and 90.4±14.8% (95% CI, 81.4-99.3%) after dose reduction.
Among the 14 cases with proposed capecitabine dose reduction,
treatment delays occurred in 9 cases before the proposal and 3
after, and the number of patients unable to receive the medication
during the treatment period was 5 before and 2 after. Of 6
recommendations for oxaliplatin dose reduction by pharmacists, 5
recommendations for oxaliplatin dose reduction were reflected in
the physicians' prescriptions for dose reduction. The mean RDI was
83.8% (95% CI, 56.7-111.0%) before oxaliplatin dose reduction and
73.8% (95% CI, 62.5-85.1%) after dose reduction. The relative dose
intensities of CAPOX were counted one course each before and after
the pharmacists' recommendations. This analysis is limited to a
short-term comparison and does not reflect maintenance of the
overall RDI over the entire treatment duration. For the RDI
comparison before and after the dose reduction recommendation, the
RDI was calculated for one treatment course each. The mean overall
RDI was 67.8±20.2% (95% CI, 62.5-73.1%) for capecitabine and
62.2±20.7% (95% CI, 56.9-67.6%) for oxaliplatin.
Discussion
In the present study, the RDI and severity of side
effects of CAPOX therapy as adjuvant chemotherapy after surgery for
gastric cancer before and after pharmacy outpatient clinic
follow-up of patients were investigated. Examining the number of
adverse events before and after the pharmacist suggested
prescriptions, pharmacological intervention at the pharmacy
outpatient clinic had an impact on decreasing the severity of
adverse events. In particular, the suggestion of supportive care
medications and capecitabine reduction for Grade 3 or higher
hand-foot syndrome was effective in reducing the severity of severe
symptoms. Therefore, pharmacists working with physicians to provide
pharmacological interventions can manage adverse events
appropriately in patients. It was also evident that the pharmacy
outpatient clinic led to maintenance of RDI.
In the present study, there were various types of
prescribing suggestions in the pharmacy outpatient clinic for the
physicians. The highest percentage of suggestions made by
pharmacists were for supportive care medications (53.9%, 131
instances). This result was consistent with previous studies
(7,19). Supportive care medications for
nausea and vomiting were the most frequently suggested, at 37.4%.
Chemotherapy-induced nausea and vomiting are side effects that
significantly affect patients undergoing outpatient cancer
chemotherapy (20). Significant
improvements in quality of life have been demonstrated after
pharmacological intervention for nausea and vomiting (5). The pharmacists may have contributed
to the maintenance of RDI by assessing the patients' conditions and
suggesting appropriate supportive care medications for nausea and
vomiting. Dose reduction suggestions for anticancer drugs accounted
for 8.2% (20 instances). This may have provided favorable support
for peripheral neuropathy, for which there is no supportive care
medication of known benefit.
Outpatient pharmacological interventions focused on
supportive care medication suggestions for Grade 3 or higher
non-hematological toxicities and suggested reductions in anticancer
drugs. Before and after the proposed supportive care, the number of
Grade ≥3 hand-foot syndrome instances decreased from 5 to 1. This
assessment was limited to a comparison between one treatment course
prior to and one course following the intervention, and the
reduction in Grade severity cannot exclude the possibility of
improvement due to the natural course of the condition. However,
peripheral neuropathy remained at one instance and did not improve.
The pharmacists' suggestions for supportive care medications
decreased the severity of hand-foot syndrome, but not the severity
of peripheral neuropathy. Hand-foot syndrome improves with drug
withdrawal, but peripheral neuropathy is a cumulative condition and
does not improve rapidly, and no drug has clearly demonstrated
benefit for peripheral neuropathy. It has been reported that the
implementation of a pharmacy outpatient clinic for peripheral
neuropathy significantly improved patient quality of life, but
there was no significant difference in the occurrence of Grade ≥2
peripheral neuropathy (5). It was
also reported that supportive care medications were primarily
suggested for peripheral neuropathy, and that anticancer drug dose
reductions were suggested for 11.1% of prescriptions proposed
(5). In the present study, the
lack of improvement in the severity of peripheral neuropathy may be
due to the fact that the authors did not propose a reduction in the
dose of anticancer drugs for Grade 3 or higher peripheral
neuropathy. The proposed capecitabine dose reduction for 5
instances of Grade 3 or higher hand-foot syndrome was reduced to 1
instance of Grade 3 or higher hand-foot syndrome. Given this, when
pharmacists collaborate with physicians in cancer drug therapy, the
severity of side effects is reduced.
Pharmacists have an important role in the management
of adverse effects of chemotherapy. Pharmacists are not only
responsible for dispensing medications and providing medication
guidance, but also for interviewing patients and collaborating with
physicians to manage adverse drug reactions. Pharmacist care has
also been reported to improve treatment of patients with HIV, and
the present study also showed results demonstrating direct benefit
for patients through pharmacist care (21).
The RDI for patients for whom pharmacy outpatient
clinic follow-up was provided was studied. CAPOX had mean RDIs of
67.8 and 62.2%, respectively. The reported RDI for capecitabine was
67.2% in a previous study, which is almost the same as in the
present study (67.8 vs. 67.2%) (10). The results may indicate that the
dose intensity for capecitabine was maintained through the
implementation of the pharmaceutical outpatient clinic. However,
the reported RDI for oxaliplatin was lower in the present study
than that reported in the aforementioned previous study (62.2 vs.
73.4%) (10). This may be due to
the fact that the RDI in the present study was calculated taking
into consideration the duration of treatment. The RDI for
oxaliplatin may also be due to its susceptibility to dose reduction
or treatment deferral due to adverse events. The contrasting trends
in RDI following dose reduction for the two drugs warrant further
discussion. The observed increase in capecitabine RDI from 72.5 to
90.4% after dose-reduction suggestions may be attributed to
improved treatment continuity. Specifically, dose reduction likely
alleviated severe adverse events, thereby reducing the frequency of
subsequent treatment delays or interruptions. Since the RDI is
calculated based on the dose per unit time, the reduction in delays
resulted in an overall higher dose intensity per day. Conversely,
the decrease in the oxaliplatin RDI from 83.8 to 73.8% after dose
reduction suggests a different mechanism. This drop is likely due
to the nature of its major dose-limiting toxicity, peripheral
neuropathy. Since peripheral neuropathy is a cumulative condition,
dose reduction often fails to prevent subsequent treatment
deferrals or extensions of treatment duration. Since the RDI
calculation strictly accounts for the actual treatment duration
(days), the extended treatment time due to the persistent
cumulative toxicity resulted in a relatively lower overall dose
intensity per unit time. This distinction highlights that, though
capecitabine dose optimization aids acute toxicity management and
schedule maintenance, oxaliplatin's cumulative toxicity can negate
the positive RDI impact of dose reduction. In the CLASSIC trial, a
minimum of 6 cycles of either capecitabine or oxaliplatin was found
to be important for the efficacy of adjuvant chemotherapy (16). Therefore, it is important to
continue administration with appropriate dose modification. In the
present study, pharmacist prescribing suggestions reduced the
severity of adverse events and maintained the RDI for capecitabine.
Therefore, pharmacological intervention by a pharmacist is
necessary to maintain RDI.
The impact of the pharmacists' suggestions for
capecitabine dose reduction on the RDI was examined. In the present
study, proposals for dose reduction of anticancer drugs by
pharmacists included 14 instances for capecitabine and 5 instances
for oxaliplatin. After the pharmacist suggested a reduced dose of
the anticancer drug and the physician implemented the dose
adjustment, the mean RDI for capecitabine increased from 72.5 to
90.4%. Specifically, the reduction in treatment delays (from 9 to 3
cases) and interruptions (from 5 to 2 cases) following dose
optimization directly contributed to the increased RDI. These
findings suggest that appropriate dose reduction to alleviate
adverse events can improve treatment continuity, which is more
effective for sustaining dose intensity than attempting to maintain
a higher dose that leads to frequent treatment cessations. These
results indicate that pharmacists' suggestions for reducing the
dose of anticancer drugs are useful for maintaining the RDI.
Regarding the choice of postoperative adjuvant chemotherapy, the
JACCRO GC-07 trial recently established the efficacy of S-1 plus
docetaxel (DS) for Stage III gastric cancer, demonstrating superior
outcomes compared with S-1 monotherapy. While the present study
focused on the CAPOX regimen, the clinical significance and
pharmacist-led management of DS therapy are also considered
important areas for future investigation and comparative analysis
(17). It has been reported that
interprofessional collaboration between clinical pharmacists and
physicians in the outpatient setting is necessary, and without
collaboration in the form of adverse event interventions,
prescription suggestions, and patient education, as in the present
study, no results will accrue directly to patients (22).
The present study has several limitations. Because
it focused on postoperative CAPOX therapy for gastric cancer and
evaluated RDI retrospectively, it was not possible to compare the
pharmacy outpatient clinic follow-up group with a non-pharmacy
outpatient clinic follow-up group. Therefore, it is not clear
whether maintenance of the RDI is due to the implementation of
pharmacy outpatient clinic follow-up. The absence of a control
group makes it difficult to determine whether pharmacist
intervention truly affects the RDI and survival indicators such as
disease-free survival (DFS) and overall survival (OS), which limits
the clinical translatability of the findings. Efficacy and survival
indicators were not assessed primarily because this was a
retrospective, observational study with the main objective of
evaluating the impact of pharmacist intervention on RDI maintenance
and adverse event management. The study's design, characterized by
the absence of a control group and a small sample size, made it
statistically inappropriate to draw robust conclusions regarding
long-term clinical outcomes such as DFS or OS. Future studies
should consider incorporating a control group design, ideally
consisting of patients who do not receive follow-up in the
dedicated pharmacist outpatient clinic. In addition, survival time
analyses such as disease-free survival and overall survival were
not performed. Although the pharmacists' prescribing suggestions
reduced the severity of Grade 3 or higher non-hematological
toxicities, improvement in quality of life could not be assessed.
Furthermore, the comparison of RDI values with historical data was
exploratory and cannot exclude the influence of confounding factors
such as differences in patient background characteristics or
advances in medical care (10).
Finally, the small sample size (n=59) from a single institution and
the lack of stratification by surgical method (total/partial
gastrectomy) or age significantly limit the generalizability of the
findings, particularly to high-risk groups such as patients
following total gastrectomy. Furthermore, performing a robust
sensitivity analysis (for example, excluding patients with early
treatment completion) was not feasible due to the small,
single-center, retrospective nature of the study, since such
exclusions would have rendered the remaining sample size
insufficient for meaningful analysis. This represents another
constraint on the generalizability of the present findings. In
addition, the relationship between the RDI and long-term outcomes
such as DFS or OS was not assessed. Given that the average RDI in
the present cohort was below the 80% threshold associated with
survival benefits in previous studies, the clinical relevance of
RDI maintenance in this context remains unclear.
In conclusion, the usefulness of pharmacy outpatient
clinic follow-up was evaluated based on the RDI. Although this
study did not include a control group, pharmacist outpatient
follow-up may have contributed to maintenance of the RDI and the
continuation of treatment by suggesting supportive care medications
and dosage reductions of anticancer drugs to the physician.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the Japanese Society
of Pharmaceutical Oncology (grant no. 2022-07).
Availability of data and materials
The data generated in the present study are included
in the figures and/or tables of this article.
Authors' contributions
MN, TY, KKa, HS, DT and KS conceived the study,
participated in its design and coordination, and performed
statistical analyses and interpretation. MN, TY, KKa, YK, KKo, TA,
WS and MH performed data curation. DT, MO, KC, TW, KY and MY
contributed to the study design, provided critical intellectual
input, and supervised the research. MN, TY, KKa and YK drafted the
manuscript. All authors read and approved the final version of the
manuscript. MN, TY, KKa and YK confirm the authenticity of all the
raw data.
Ethics approval and consent to
participate
The present study was approved by Clinical Research
Ethics Review Committee of the Japanese Foundation for Cancer
Research Cancer Institute Hospital (approval no. 2021-GB-017;
Tokyo, Japan). The use of the opt-out method in lieu of written,
informed consent was approved by the Clinical Research Ethics
Review Committee of the hospital.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Banna GL, Collovà E, Gebbia V, Lipari H,
Giuffrida P, Cavallaro S, Condorelli R, Buscarino C, Tralongo P and
Ferraù F: Anticancer oral therapy: Emerging related issues. Cancer
Treat Rev. 36:595–605. 2010.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Denois VR, Poirson J, Nourissat A, Jacquin
JP, Guastalla JP and Chauvin F: Adherence with oral chemotherapy:
Results from a qualitative study of the behaviour and
representations of patients and oncologists. Eur J Cancer Care
(Engl). 20:520–527. 2011.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Basch E, Deal AM, Dueck AC, Scher HI, Kris
MG, Hudis C and Schrag D: Overall survival results of a trial
assessing patient-reported outcomes for symptom monitoring during
routine cancer treatment. JAMA. 318:197–198. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Fujii H, Ueda Y, Hirose C, Ohata K, Sekiya
K, Kitahora M, Sadaka S, Yamamoto S, Watanabe D, Kato-Hayashi H, et
al: Pharmaceutical intervention for adverse events improves quality
of life in patients with cancer undergoing outpatient chemotherapy.
J Pharm Health Care Sci. 8(8)2022.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ikesue H, Kusuda K, Satsuma Y, Nishiwaki
F, Miura R, Masuda Y, Hirabatake M, Muroi N, Fujimoto D, Morimoto
T, et al: Evaluation of the usefulness of protocol-based
pharmacist-facilitated laboratory monitoring to ensure the safety
of immune checkpoint inhibitors in patients with lung cancer. J
Clin Pharm Ther. 45:1288–1294. 2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Hirabatake M, Ikesue H, Iwama Y, Irie K,
Yoshino S, Yamasaki T, Hashida T, Kawakita M and Muroi N:
Pharmacist-urologist collaborative management improves clinical
outcomes in patients with castration-resistant prostate cancer
receiving enzalutamide. Front Pharmacol. 13(901099)2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Haller DG, Tabernero J, Maroun J, de Braud
F, Price T, Van Cutsem E, Hill M, Gilberg F, Rittweger K and
Schmoll HJ: Capecitabine plus oxaliplatin compared with
fluorouracil and folinic acid as adjuvant therapy for stage III
colon cancer. J Clin Oncol. 29:1465–1471. 2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Bang YJ, Kim YW, Yang HK, Chung HC, Park
YK, Lee KH, Lee KW, Kim YH, Noh SI, Cho JY, et al: Adjuvant
capecitabine and oxaliplatin for gastric cancer after D2
gastrectomy (CLASSIC): A phase 3 open-label, randomised controlled
trial. Lancet. 379:315–321. 2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Fuse N, Bando H, Chin K, Ito S, Yoshikawa
T, Tsuburaya A, Terashima M, Kawashima Y, Fukunaga T, Gotoh M, et
al: Adjuvant capecitabine plus oxaliplatin after D2 gastrectomy in
Japanese patients with gastric cancer: A phase II study. Gastric
Cancer. 20:332–340. 2017.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kawakami K, Yokokawa T, Kobayashi K,
Sugisaki T, Suzuki K, Suenaga M, Yamaguchi K, Inoue A, Machida Y,
Yamaguchi T and Hama T: Self-reported adherence to capecitabine on
XELOX treatment as adjuvant therapy for colorectal cancer. Oncol
Res. 25:1625–1631. 2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Sugita K, Kawakami K, Yokokawa T, Sugisaki
T, Takiguchi T, Aoyama T, Suzuki K, Suenaga M, Yamaguchi K, Inoue
A, et al: Self-Reported adherence to trifluridine and tipiracil
hydrochloride for metastatic colorectal cancer: A retrospective
cohort study. Oncology. 91:224–230. 2016.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Kawakami K, Aoyama T, Yokokawa T,
Kobayashi K, Takahari D, Chin K, Ide T, Machida Y, Yamaguchi K and
Hama T: The combined use of 5 or more drugs is a factor related to
lower adherence to S-1 in S-1 and oxaliplatin treatment for
advanced gastric cancer. Biol Pharm Bull. 44:1075–1080.
2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Mae T, Yokokawa T, Kawakami K, Yagi N,
Suenaga M, Shinozaki E, Matsusaka S, Mizunuma N and Hama T:
Usefulness of pharmaceutical outpatient clinic in XELOX therapy.
Jpn J Pharm Health Care Sci. 37:611–615. 2011.
|
|
15
|
Horasawa S, Matsui R, Kawasumi K and Saito
S: The usefulness of outpatient pharmacy services in adjuvant
chemotherapy for gastric cancer. Gan To Kagaku Ryoho. 43:1091–1095.
2016.PubMed/NCBI(In Chinese).
|
|
16
|
Noh SH, Park SR, Yang HK, Chung HC, Chung
IJ, Kim SW, Kim HH, Choi JH, Kim HK, Yu W, et al: Adjuvant
capecitabine plus oxaliplatin for gastric cancer after D2
gastrectomy (CLASSIC): 5-year follow-up of an open-label,
randomised phase 3 trial. Lancet Oncol. 15:1389–1396.
2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Yoshida K, Kodera Y, Kochi M, Ichikawa W,
Kakeji Y, Sano T, Nagao N, Takahashi M, Takagane A, Watanabe T, et
al: Addition of docetaxel to oral fluoropyrimidine improves
efficacy in patients with stage III gastric cancer: Interim
analysis of JACCRO GC-07, a randomized controlled trial. J Clin
Oncol. 37:1296–1304. 2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Hryniuk WM and Goodyear M: The calculation
of received dose intensity. J Clin Oncol. 8:1935–1937.
1990.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Aogi K, Takeuchi H, Saeki T, Aiba K,
Tamura K, Iino K, Imamura CK, Okita K, Kagami Y, Tanaka R, et al:
Optimizing antiemetic treatment for chemotherapy-induced nausea and
vomiting in Japan: Update summary of the 2015 Japan society of
clinical oncology clinical practice guidelines for antiemesis. Int
J Clin Oncol. 26:1–17. 2021.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Ihbe-Heffinger A, Ehlken B, Bernard R,
Berger K, Peschel C, Eichler HG, Deuson R, Thödtmann J and Lordick
F: The impact of delayed chemotherapy-induced nausea and vomiting
on patients, health resource utilization and costs in German cancer
centers. Ann Oncol. 15:526–536. 2004.PubMed/NCBI View Article : Google Scholar
|
|
21
|
de Maat MM, de Boer A, Koks CH, Mulder JW,
Meenhorst PL, van Gorp EC, Mairuhu AT, Huitema AD and Beijnen JH:
Evaluation of clinical pharmacist interventions on drug
interactions in outpatient pharmaceutical HIV-care. J Clin Pharm
Ther. 29:121–130. 2004.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Shahrami B, Forough AS, Najmeddin F,
Hadidi E, Toomaj S, Javadi MR, Gholami K and Sadeghi K:
Identification of drug-related problems followed by clinical
pharmacist interventions in an outpatient pharmacotherapy clinic. J
Clin Pharm Ther. 47:964–972. 2022.PubMed/NCBI View Article : Google Scholar
|