1. Introduction
LAP2-emerin-MAN1 (LEM) domain-containing protein 1
(LEMD1; also referred to as LEMP-1 and testes-specific protease 50)
is a cancer-testis antigen (CTA) that was first identified in 2004
as a novel testis-specific gene expressed in colorectal cancer
tissues (1,2). As a CTA, LEMD1 has a low expression
level in normal somatic cells and is only expressed in
immune-privileged tissues such as the testis (3). However, in tumors, abnormal
demethylation and activation of the gene promoter leads to high
expression levels of LEMD1 in various tumor tissues, including
gastric, pancreatic, colorectal and prostate cancer as well as oral
squamous cell carcinoma (OSCC) (4-6).
Consistent with the broader CTA paradigm, prototypical CTAs such as
New York esophageal squamous cell carcinoma 1 and
melanoma-associated antigen 4 exhibit testis restricted expression
yet are aberrantly activated across diverse malignancies (7). An increasing number of studies
suggest that LEMD1 serves an oncogenic role in the development and
progression of tumors in which its overexpression can promote
malignant phenotypes such as tumor cell proliferation, invasion and
metastasis (2,8). Therefore, LEMD1 has received
widespread attention in the field of tumor biology and is
considered a potential diagnostic marker and therapeutic
target.
The present review summarized the molecular
structure and biological functions of LEMD1, its expression
patterns and clinical relevance across diverse tumors, mechanistic
insights into its tumor-promoting actions, and emerging therapeutic
approaches. Unlike previous reports that investigate LEMD1 in a
single type of tumor or along a single pathway, the present review
provided two distinct advances. Firstly, it provided, to the best
of our knowledge, the first comprehensive integration of the
nuclear-inner-membrane localization of LEMD1 with five convergent
cytoplasmic signaling cascades, namely PI3K/Akt, ERK/MAPK,
Wnt/β-catenin, Ras homolog family member A (RhoA)/Rho-associated
coiled-coil-containing protein kinase (ROCK) and p53/mTORC1, each
of which were previously only examined in isolation (2,9-11).
Secondly, the present review offered a systematic synthesis of
‘expression-signaling-clinical relevance’ data for LEMD1 across
>10 tumor entities, allowing for the construction of a
cross-cancer landscape, which has not been addressed in previous
LEMD1-related studies (12,13)
and highlighted its diagnostic and therapeutic potential.
2. Molecular structure and biological
functions of LEMD1
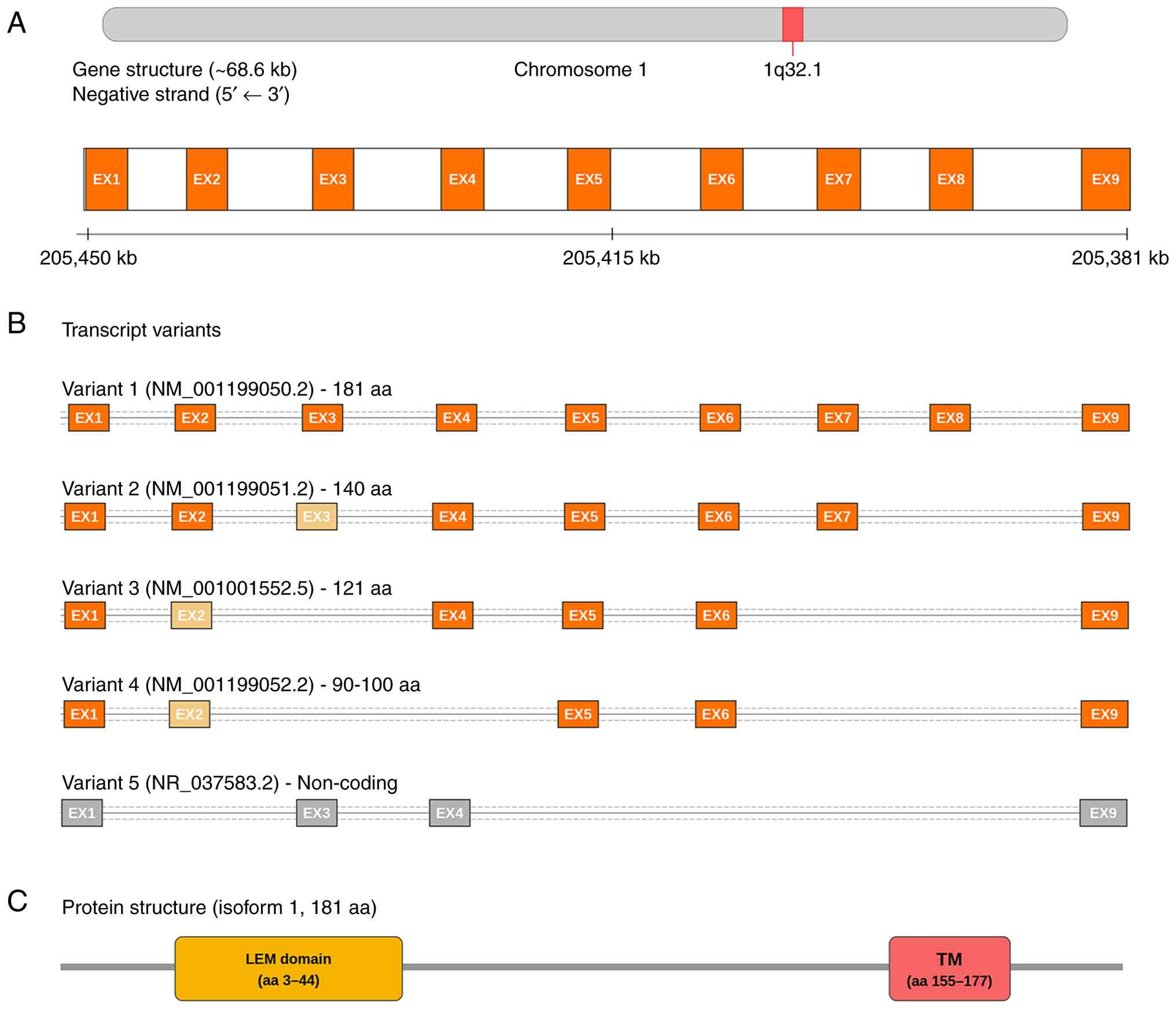
LEMD1 gene and protein structure
Located in the q32.1 region of human chromosome 1,
the LEMD1 gene contains 9 exons, encoding multiple splice variants.
At present, five transcript variants have been reported, with V1-3
mainly expressed in testis and colorectal cancer stem cells, while
V4 and 5 are expressed at low levels in normal tissues (14). The LEMD1 protein belongs to the LEM
domain protein family. The full-length protein consists of 181
amino acids with a molecular weight of ~20 kDa. It contains an
N-terminal LEM domain and a C-terminal transmembrane region, making
it a single-pass membrane protein (15,16)
localized to the inner nuclear membrane, as presented in Fig. 1. The LEM domain is a conserved
structure composed of ~40 amino acids that binds to the
barrier-to-autointegration factor (BAF) (17). In classic LEM family proteins [such
as lamina-associated polypeptide 2β (LAP2β), emerin and MAN1 (also
known as LEMD3)], the LEM domain is located on the nucleoplasmic
side and can anchor chromatin to the nuclear membrane by binding to
BAF or BAF-DNA complexes (14,18).
As a member of the aforementioned family, the LEMD1 protein is
hypothesized to have similar structural features and interaction
interfaces, potentially participating in nuclear membrane-chromatin
connections (14,19,20).
Biological functions of LEMD1 in cell
biology
Under normal physiological conditions, research on
the normal biological functions of LEMD1 is limited due to its low
expression levels in the majority of normal tissues. Previous
studies suggest that LEMD1 may serve roles in cell cycle
regulation, chromatin conformation maintenance and nuclear membrane
homeostasis in specific cell types (such as germ cells) (15,21).
Typically, chromatin anchored to the nuclear
periphery is associated with deacetylated histones, for example
H3K9 and H3K27, and heterochromatin-associated markers (such as
H3K9me2 and H3K9me3) (22), which
maintains a relatively transcriptionally silent state (16). LEMD1 may anchor specific chromatin
regions to the nuclear periphery through its LEM domain binding
with BAF, potentially influencing the epigenetic state and
transcriptional activity of these genes (16). Therefore, it is hypothesized that
LEMD1 may participate in the regulation of gene expression. Other
studies demonstrate potential associations between LEMD1 and gene
splicing and epigenetic regulation processes (14,19,20,23).
For example, a study by Li et al (11) suggests that the methylation state
of the LEMD1 gene promoter region maintains high methylation in
normal cells, resulting in silencing, while demethylation occurs in
tumor cells leading to gene activation.
Additionally, during human spermatogenesis, LEMD1
transcripts are highly enriched in post-meiotic germ cells, with
single-cell analyses showing maximal expression levels in late
spermatids (24,25). This testis-specific pattern
suggests a specialized role during spermatogenesis. Furthermore, as
developing spermatids undergo chromatin remodeling and nuclear
reshaping, a number of canonical nuclear envelope proteins (such as
emerin and LEMD3) are absent; however, LEMD1 is one of the small
number of LEM-domain factors expressed in these cells (24). Notably, LEMD1 is not detected at
the nuclear periphery in spermatids but instead localizes within
the nucleoplasm, and slowly localizes toward the posterior pole of
the nucleus as spermatids elongate (24). This dynamic localization suggests
that LEMD1 may help tether chromatin to the remaining nuclear
scaffold or otherwise contribute to nucleus remodeling during the
histone-to-protamine transition. Consistent with this hypothesis,
in human spermiogenesis, LEMD1 (along with LAP2β and lamin B1) is
suggested to substitute for the lamin B receptor (which is absent
in spermatids) as an anchoring interface between chromatin and the
nuclear envelope (26). As LEMD1
can bind to BAF, its presence in spermatids may possibly maintain
transient chromatin-nuclear envelope connections at a stage when
the lamina is being dismantled (24). These observations point to a
physiological role for LEMD1 in orchestrating spermatid nuclear
organization and stability, which may be critical for proper
spermatogenesis and subsequent sperm function. These phenomena are
consistent with the regulatory patterns of the majority of CTA
genes, suggesting that LEMD1 has limited function in normal cell
biology, while its abnormal expression primarily occurs in the
context of tumorigenesis.
3. Expression of LEMD1 and clinical
relevance in different types of tumor
LEMD1 expression levels in different
types of tumor
The present analyses using the Tumor Immune
Estimation Resource 2.0 database (https://compbio.cn/timer2/) indicated that LEMD1 mRNA
was highly expressed across ~20 types of cancer, whereas its
expression level was low or undetectable in the majority of normal
tissues, as illustrated in Fig. 2.
In 2004, a study by Yuki et al (1) first reported a high expression of
LEMD1 in colorectal cancer. Subsequently, in a study by Li et
al (4), which analyses The
Cancer Genome Atlas database and patient samples, reports that
LEMD1 is notably upregulated in tissues and cell lines of gastric
cancer compared with normal gastric mucosa and normal gastric
epithelial cells. In addition to the digestive system, upregulated
LEMD1 expression levels are reported in various solid tumors. For
example, a study by Ghafouri-Fard et al (23) examines 30 prostate cancer cases and
reports LEMD1 transcripts in 23% of them, while none are detected
in 25 cases of benign prostatic hyperplasia. In OSCC, ~35% of
patient tumors showed positive immunohistochemistry for LEMD1, with
elevated LEMD1 protein expression levels in positive tumors
(9). Both sequencing analysis and
quantitative PCR validation of LEMD1 mRNA reveal notably increased
transcription levels in papillary thyroid cancer tissues compared
with adjacent normal thyroid tissues (27). Triple-negative breast cancer
(TNBC), a type of breast cancer with a poor prognosis, reveals
higher LEMD1 expression levels in tissues compared with other
subtypes of breast cancer (11).
Pancreatic cancer tissues also exhibit abnormally high LEMD1
expression levels, with reports indicating higher mRNA and protein
levels compared with normal pancreatic tissues (2). Additionally, elevated LEMD1
expression levels are confirmed in non-small cell lung cancer
(NSCLC) and are associated with malignant tumor behaviors. For
example, higher LEMD1 levels are associated with poorer
differentiation, advanced TNM stage and lymph node metastasis, and
it predicts a shorter overall survival in patients with NSCLC
(28). Furthermore, LEMD1 is also
implicated in anaplastic large cell lymphoma (ALCL), with
nucleophosmin-anaplastic lymphoma kinase/STAT3 signaling reported
to upregulate LEMD1 expression levels (29). Overall, as a CTA, LEMD1 shows
cancer tissue-specific high expression levels in multiple types of
tumor, providing a basis for its potential use as a universal tumor
marker.
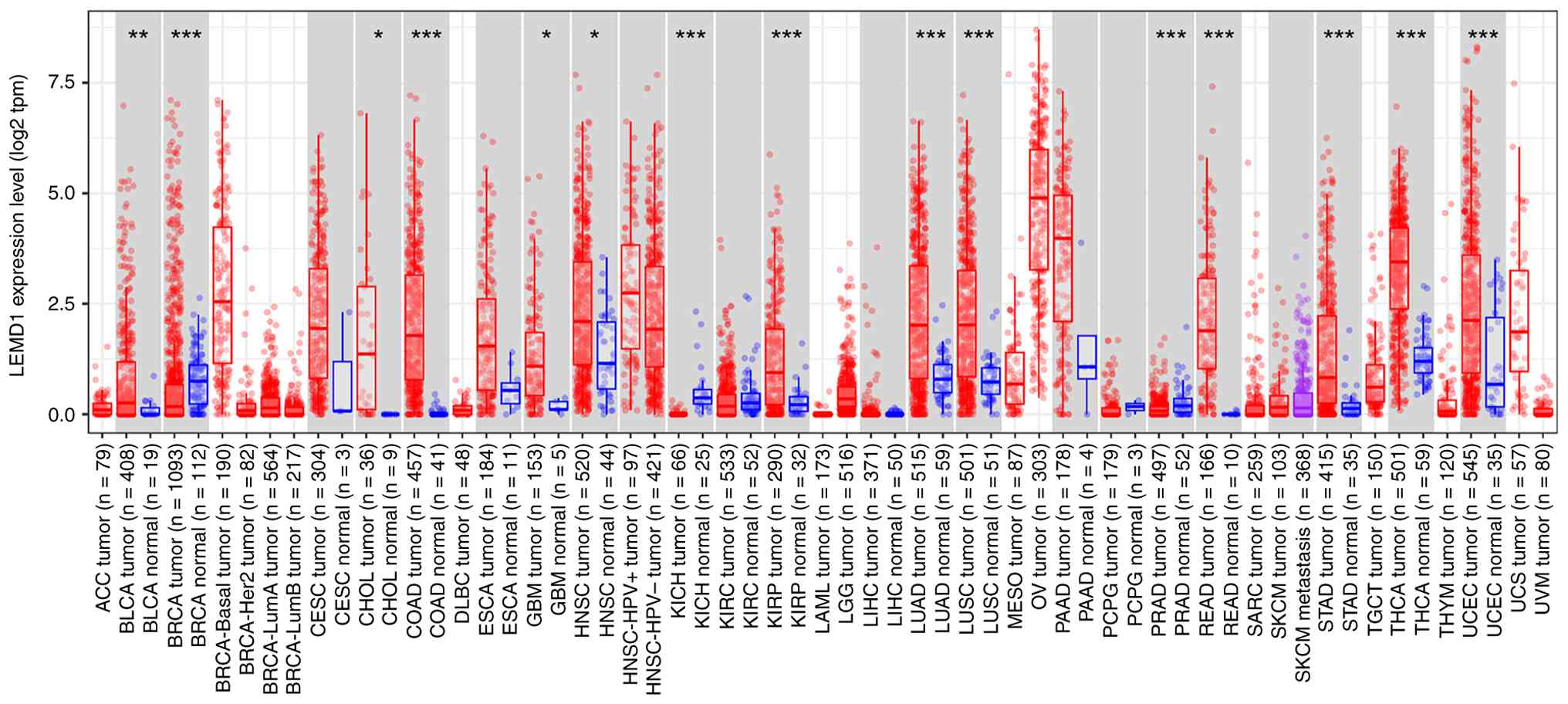 | Figure 2Expression profile of LEMD1 across
different types of tumors. Box plots showing the mRNA expression
levels of LEMD1 in tumor (red) and matched normal (blue) tissues
across multiple types of cancer, presented as log2 tpm. The sample
size for each group is indicated in parentheses on the x-axis.
Normal data are shonw only when available; therefore, a number of
types of cancer do not have corresponding normal tissue expression
levels. Differential expression analysis was carried out using
edgeR (version 4.4.2; https://bioconductor.org/packages/edgeR) based on RNA
sequencing raw counts. *P<0.05,
**P<0.01 and ***P<0.001. The figure was
generated using the Tumor Immune Estimation Resource, version 2
database (https://compbio.cn/timer2/). LEMD1,
LAP2-emerin-MAN1 domain containing 1; tpm, transcripts per million;
ACC, adrenocortical carcinoma; BLCA, bladder urothelial carcinoma;
BRCA, breast invasive carcinoma; CESC, cervical squamous cell
carcinoma; CHOL, cholangiocarcinoma; COAD, colon adenocarcinoma;
DLBC, diffuse large B-cell lymphoma; ESCA, esophageal carcinoma;
GBM, glioblastoma multiforme; HNSC, head and neck squamous cell
carcinoma; KICH, kidney chromophobe; KIRC, kidney renal clear cell
carcinoma; KIRP, kidney renal papillary cell carcinoma; LAML, acute
myeloid leukemia; LGG, lower grade glioma; LIHC, liver
hepatocellular carcinoma; LUAD, lung adenocarcinoma; LUSC, lung
squamous cell carcinoma; MESO, mesothelioma; OV, ovarian serous
cystadenocarcinoma; PAAD, pancreatic adenocarcinoma; PCPG,
pheochromocytoma and paraganglioma; PRAD, prostate adenocarcinoma;
READ, rectum adenocarcinoma; SARC, sarcoma; SKCM, skin cutaneous
melanoma; STAD, stomach adenocarcinoma; TGCT, testicular germ cell
tumor; THCA, thyroid carcinoma; THYM, thymoma; UCEC, uterine corpus
endometrial carcinoma; UCS, uterine carcinosarcoma; UVM, uveal
melanoma. |
Association between LEMD1 and
clinicopathological features
Abnormal expression of LEMD1 not only has tumor
specificity but is also notably associated with adverse
clinicopathological features in multiple types of tumor, such as
OSCC, thyroid cancer and TNBC (9,11,27),
including more advanced TNM staging, lymph node metastasis, distant
metastasis, increased tumor volume and vascular or lymphatic
invasion (9,21,30).
Previous studies suggest that in colorectal cancer, high expression
of LEMD1 is positively correlated not only with lymph node
metastasis and liver metastasis but also with tumor staging
(1,14,21,31).
A study by Sasahira et al (9) analyzed 289 patients with OSCC and
found that LEMD1 expression levels were notably associated with
tumor progression (T factor and clinical stage), nodal metastasis
and poor prognosis. Furthermore, immunostaining for cytoplasmic
LEMD1 is positive more frequently in patients with nodal metastasis
compared with those without nodal metastasis [65.7% (46/70
patients) vs. 25.1% (55/219 patients)]. Multivariate analysis
further identifies LEMD1 expression levels as an independent
predictor of disease-free survival (DFS) in patients with OSCC
(9). In patients with papillary
thyroid cancer, high LEMD1 expression levels are also notably
associated with a higher risk of lymph node metastasis (27). Additionally, in patients with TNBC,
high LEMD1 expression levels are notably associated with a higher
histological grade and shorter overall survival (OS) compared with
those with low LEMD1 expression levels (11). A study on NSCLC reports that high
LEMD1 expression levels are associated with later TNM staging,
poorer tumor differentiation and an increased likelihood of lymph
node metastasis, as well as increased cancer stem cell-like
phenotypes and invasiveness (28).
Additionally, a study by Cao et al (2) reports that LEMD1 is upregulated in
pancreatic cancer tissue, and patients with high LEMD1 expression
levels are more likely to develop distant metastasis, suggesting
that LEMD1 may promote the metastatic ability of pancreatic cancer.
These results indicate that high LEMD1 expression levels often
suggest a more aggressive tumor biological behavior and worse
clinical outcomes. By contrast, in normal tissues or benign/early
stages of tumors, LEMD1 typically remains silent or at low levels,
corresponding to an improved prognosis.
Potential diagnostic and prognostic
value of LEMD1 in tumors
Due to the absence of LEMD1 in the majority of
normal tissues and its abnormal appearance in tumors, its detection
has potential value for tumor diagnosis and prognosis assessment.
Multiple studies based on Kaplan-Meier survival analyses and
multivariate Cox regression analyses report that LEMD1 can serve as
an adverse prognostic factor for various types of tumor (9,11,21).
In cohorts with colorectal or pancreatic cancer, patients with high
LEMD1 expression levels have a notably reduced OS and DFS (2,32). A
previous study on colorectal cancer constructed a three-gene model
including LEMD1 to predict postoperative recurrence risk, with
results showing that this model can effectively identify patients
at high risk of recurrence (21).
Several studies provide performance indicators for the diagnostic
utility of LEMD1 (9,23,27).
In papillary thyroid carcinoma, LEMD1 overexpression distinguishes
tumor from normal tissue with high accuracy (receiver operating
characteristic analysis yielding ~80% sensitivity and ~90%
specificity) (27). Furthermore,
in pancreatic cancer, an autoantibody biomarker panel incorporating
LEMD1 achieved an area under the curve of 0.906 (93.3% sensitivity
and 76.7% specificity) (33),
highlighting the diagnostic potential of LEMD1 in oncology.
Furthermore, as a CTA, LEMD1 also has potential immunogenicity that
can be used for serological detection, such as the elevated LEMD1
autoantibodies detected in the blood of patients with pancreatic
cancer (33), suggesting its
research value in early diagnosis and recurrence monitoring.
Whether similar anti-LEMD1 autoantibodies are present in other
types of cancer is yet to be elucidated; however, given the status
of LEMD1 as a CTA, this is a plausible possibility that warrants
further investigation.
The survival analysis of the present review used the
Gene Expression Profiling Interactive Analysis 2 (http://GEPIA2.cancer-pku.cn/) database and revealed
that high LEMD1 expression levels were associated with a poor
prognosis in colon, rectal, pancreatic and kidney cancer, as
presented in Fig. 3. The
aforementioned evidence suggests that detecting LEMD1 expression
levels in tumor samples has the potential to provide useful
prognostic information for clinical practice and guide subsequent
treatment strategies.
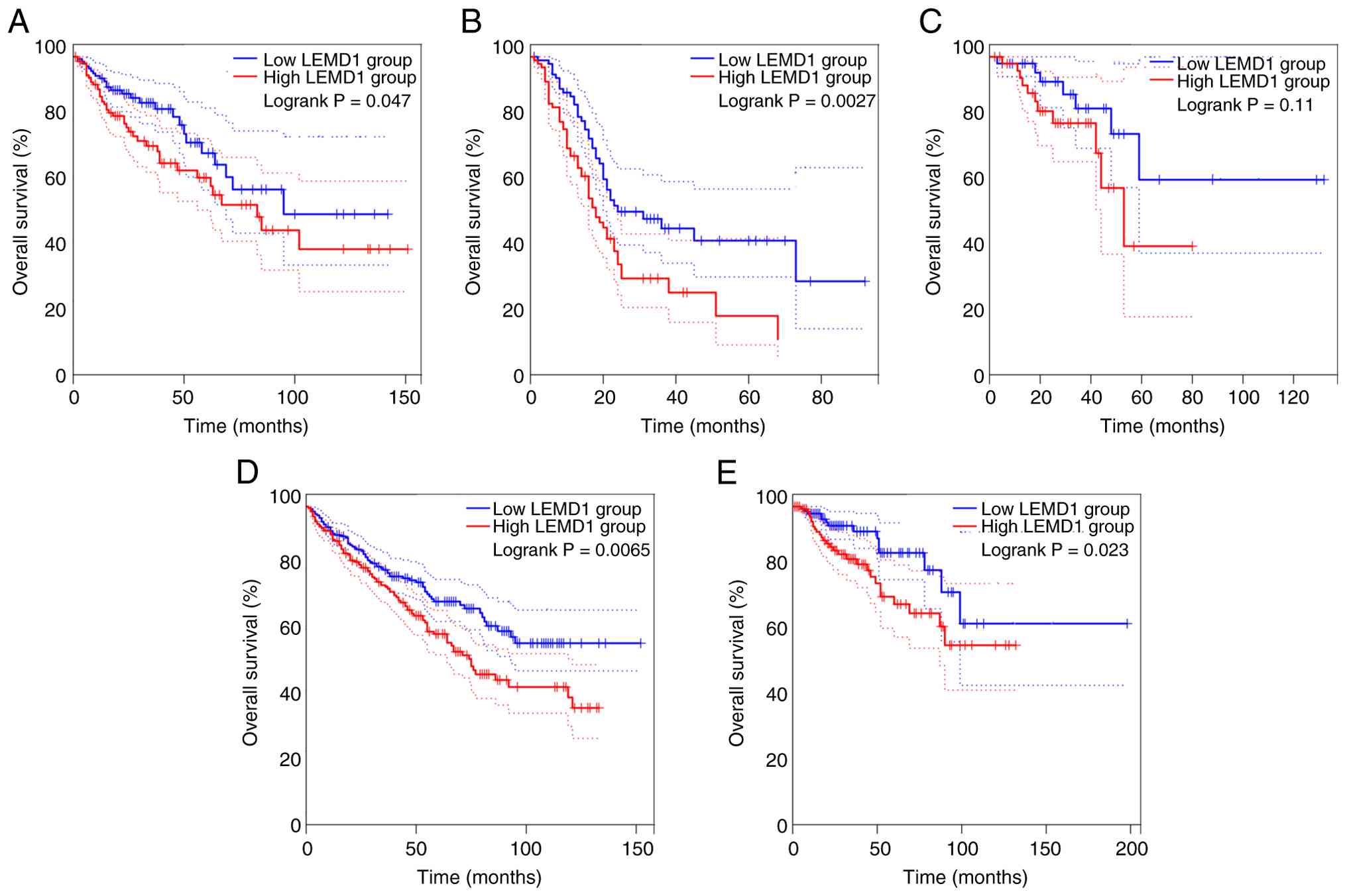 | Figure 3Kaplan-Meier overall survival curves
according to LEMD1 expression levels in five types of cancer.
Patients were divided into high and low LEMD1 expression level
groups based on the median expression level values. The blue solid
lines represent the low LEMD1 expression level groups and the red
solid lines represent the high LEMD1 expression level groups, with
dotted lines indicating the 95% confidence intervals. The log-rank
test P-values are shown in each panel. (A) Colon adenocarcinoma
(high LEMD1 group, n=135; low LEMD1 group, n=135; HR=1.6; P=0.048;
log-rank P=0.047). (B) Pancreatic adenocarcinoma (high LEMD1 group,
n=89; low LEMD1 group, n=89; HR=1.9; P=0.0031; log-rank P=0.0027).
(C) Rectum adenocarcinoma (high LEMD1 group, n=46; low LEMD1 group,
n=46; HR=2.2; P=0.11; log-rank P=0.11). (D) Kidney renal clear cell
carcinoma (high LEMD1 group, n=253; low LEMD1 group, n=250; HR=1.5;
P=0.0069; log-rank P=0.0065). (E) Kidney renal papillary cell
carcinoma (high LEMD1 group, n=140; low LEMD1 group, n=141; HR=2.1;
P=0.026; log-rank P=0.023). The figure was generated using the
GEPIA2 database (http://gepia2.cancer-pku.cn). The survival data were
derived from The Cancer Genome Atlas (database of Genotypes and
Phenotypes accession no. phs000178; https://www.ncbi.nlm.nih.gov/projects/gap/cgi-bin/study.cgi?study_id=phs000178)
using the following search parameters in the GEPIA2 database: Gene
symbol, LEMD1; type of cancer, esophageal carcinoma; analysis
method, overall survival; group cutoff, median; HR based on Cox
proportional hazards model; and 95% confidence interval. LEMD1,
LAP2-emerin-MAN1 domain-containing protein 1; HR, hazard ratio;
GEPIA2, Gene Expression Profiling Interactive Analysis 2. |
4. Molecular mechanisms of LEMD1 in tumor
development and progression
Association between LEMD1 and cell
proliferation, apoptosis and migration
Several functional experiments (2,4,9,21,27)
reveal that LEMD1 promotes tumor progression in multiple types of
cancer. For example, knocking down LEMD1 suppresses invasion and
endothelial transmigration in OSCC; in gastric cancer, it promotes
proliferation through effects on the cell cycle and apoptosis; in
colorectal cancer, it enhances proliferation, migration, invasion
and the epithelial-mesenchymal transition (EMT); and in thyroid and
pancreatic cancer, gain- and loss-of-function experiments show
effects on proliferation, migration/invasion and apoptosis
(Table I) (2,4,10,11,21,27-31,34).
A study by Li et al (4)
demonstrates in gastric cancer cells that the overexpression of
LEMD1 accelerates the G1/S phase transition and inhibits the
expression of the apoptosis-related protein Bax, which increases
tumor cell proliferation rates and promotes clone formation. By
contrast, LEMD1 knockdown inhibits cell proliferation and induces
cell cycle arrest and increases apoptosis. In vivo
experiments also demonstrate that reduced LEMD1 levels slows the
growth of transplanted tumors (4).
In colon cancer, knockdown of LEMD1 notably reduces the
proliferative activity of tumor cells (30). A study by Li and Zhang (28) on NSCLC cells reveals that
upregulation of LEMD1 confers ‘stem cell-like’ phenotypes to
tumors, including enhanced spherical clone formation and
self-renewal capacity. A study by Cao et al (2) hypothesizes that LEMD1 knockdown
notably enhances p53 pathway-mediated apoptosis. This finding
suggests that, under normal conditions, LEMD1 helps restrain the
pro-apoptotic activation of p53, likely by upregulating survival
signals or pathways that keep p53 in check. Collectively, these
studies indicate that LEMD1 promotes tumor cell proliferation and
survival across multiple types of cancer, likely by facilitating
cell-cycle progression and suppressing apoptosis, which contributes
to tumor growth.
 | Table IFunctions and potential mechanisms of
LEMD1 in different types of tumors. |
Table I
Functions and potential mechanisms of
LEMD1 in different types of tumors.
| First author,
year | Type of tumor | Expression of
LEMD1 | Functional
impact | Associated genes or
signaling pathways | (Refs.) |
|---|
| Zhang et al,
2022 | Colorectal
cancer | Highly
expressed | Promotes cell
proliferation, migration, invasion and the EMT | RhoA/ROCK1
signaling pathway | (21) |
| Luo et al,
2021 | Colorectal
cancer | Highly
expressed | Promotes cell
proliferation, migration, invasion and angiogenesis | PI3K/AKT signaling
pathway | (31) |
| Li et al,
2022 | Colorectal
cancer | Highly
expressed | Promotes cell
proliferation, migration, invasion and angiogenesis | PI3K/AKT pathway
and upstream regulation by SOX4 | (30) |
| Li et al,
2022 | Oral squamous cell
carcinoma | Highly
expressed | Promotes cell
migration and invasion | PI3K-AKT signaling
pathway, which may be activated via the LEMD1-AS1/LEMD1 axis | (34) |
| Sasahira et
al, 2020 | Oral squamous cell
carcinoma | Highly
expressed | Promotes cell
invasion, adhesion to endothelial cells and transendothelial
migration | SRPX2-uPAR/HGF
pathway | (10) |
| Li and Zhang,
2023 | Non-small cell lung
cancer | Highly
expressed | Promotes cell
proliferation, invasion, stem cell characteristics and inhibits
apoptosis | PI3K/AKT
pathway | (28) |
| Cao et al,
2022 | Pancreatic
cancer | Highly
expressed | Promotes cell
proliferation, migration, invasion and inhibits apoptosis | p53 and mTORC1
signaling pathways | (2) |
| Li et al,
2019 | Gastric cancer | Highly
expressed | Promotes cell
proliferation, cell cycle progression and inhibits apoptosis | PI3K/Akt signaling
pathway | (4) |
| Xu et al,
2021 | Papillary thyroid
cancer | Highly
expressed | Promotes cell
proliferation, migration, invasion, the EMT and inhibits
apoptosis | Wnt/β-catenin
signaling pathway | (27) |
| Li et al,
2023 | Triple-negative
breast cancer | Highly
expressed | Promotes cell
proliferation, migration, invasion, the EMT and inhibits
apoptosis | ERK signaling
pathway | (11) |
| Matsuyama et
al, 2011 | Anaplastic large
cell lymphoma | Highly
expressed | Reduces
chemotherapy sensitivity, promotes cell proliferation and inhibits
apoptosis | NPM-ALK/STAT3
pathway | (29) |
The presence of LEMD1 notably increases the invasive
and metastatic potential of cancer cells. Previous studies suggest
that LEMD1 is involved in metastatic progression in OSCC; high
LEMD1 expression levels are associated with lymph node metastasis,
and metastatic tumors have higher LEMD1 expression levels compared
with non-metastatic tumors (9,10,34).
In vitro functional experiments confirm that LEMD1 knockdown
inhibits the invasive ability of oral cancer cells and reduces the
ability of cancer cells to migrate through endothelial cell
monolayers (9). Furthermore, in
colorectal and pancreatic cancer models, LEMD1 silencing results in
decreased tumor cell invasion and migration abilities, while
overexpression made tumors more invasive (2,30). A
study by Li et al (11)
knocked down LEMD1 in TNBC cells, and the results reveal that these
tumors not only reduced their in vitro migration ability but
also reduced their tumorigenicity and metastatic ability in
vivo. LEMD1 is also associated with the regulation of the EMT.
For example, in thyroid cancer cells, LEMD1 overexpression
decreases the levels of the epithelial marker E-cadherin and
increases the expression of mesenchymal markers N-cadherin and
Vimentin, promoting cells to acquire a spindle-shaped, migratory
phenotype (27). Additionally, in
OSCC (9) and TNBC, LEMD1 promotes
transmembrane migration of vascular or lymphatic endothelial cells,
enhancing metastatic capacity (11).
Therefore, evidence suggests that LEMD1 promotes the
malignant phenotypes of tumors in multiple ways such as by
accelerating cell proliferation and inhibiting apoptosis to promote
tumor growth, while also enhancing cell migration and invasion
characteristics to facilitate tumor metastasis. In summary, LEMD1
promotes metastasis by inducing the EMT and cytoskeletal
reorganization (for example, via RhoA/ROCK signaling), which
increases cancer cell motility and invasive capability.
Interactions between LEMD1 and signal
transduction pathways
LEMD1 can regulate multiple signaling pathways
associated with tumor progression, including the PI3K/AKT,
Wnt/β-catenin and RhoA/ROCK1 pathways, which exert tumor-promoting
effects (4,21,27,30)
(Fig. 4). The PI3K/Akt signaling
pathway is important for regulating cell proliferation and
survival, and LEMD1 is associated with the abnormal activation of
this pathway in various tumors. For example, in gastric cancer
research, overexpression of LEMD1 can promote the activation of the
PI3K/Akt pathway and increase the phosphorylation levels of Akt
(4). Additionally, silencing LEMD1
decreases p-PI3K and p-AKT, whereas LEMD1 overexpression increases
p-PI3K and p-AKT (4). A study by
Li et al (30) reports that
the expression of LEMD1 is upregulated by the transcription factor
SOX4, enhancing the activity of PI3K/Akt signaling and promoting
the proliferation and invasion of colon cancer cells. Functional
tests in NSCLC also show that LEMD1-mediated malignant phenotypes
are reversed by the PI3K activator 740 Y-P, suggesting that the
action of LEMD1 depends on the activation of the PI3K/Akt pathway
(28). Taken together, these
findings indicate that aberrant activation of the PI3K/Akt pathway
is a possible primary downstream mechanism that mediates the
tumor-promoting effects of LEMD1, as this pathway is consistently
implicated in LEMD1-associated oncogenic processes across multiple
types of cancer (4,28,30).
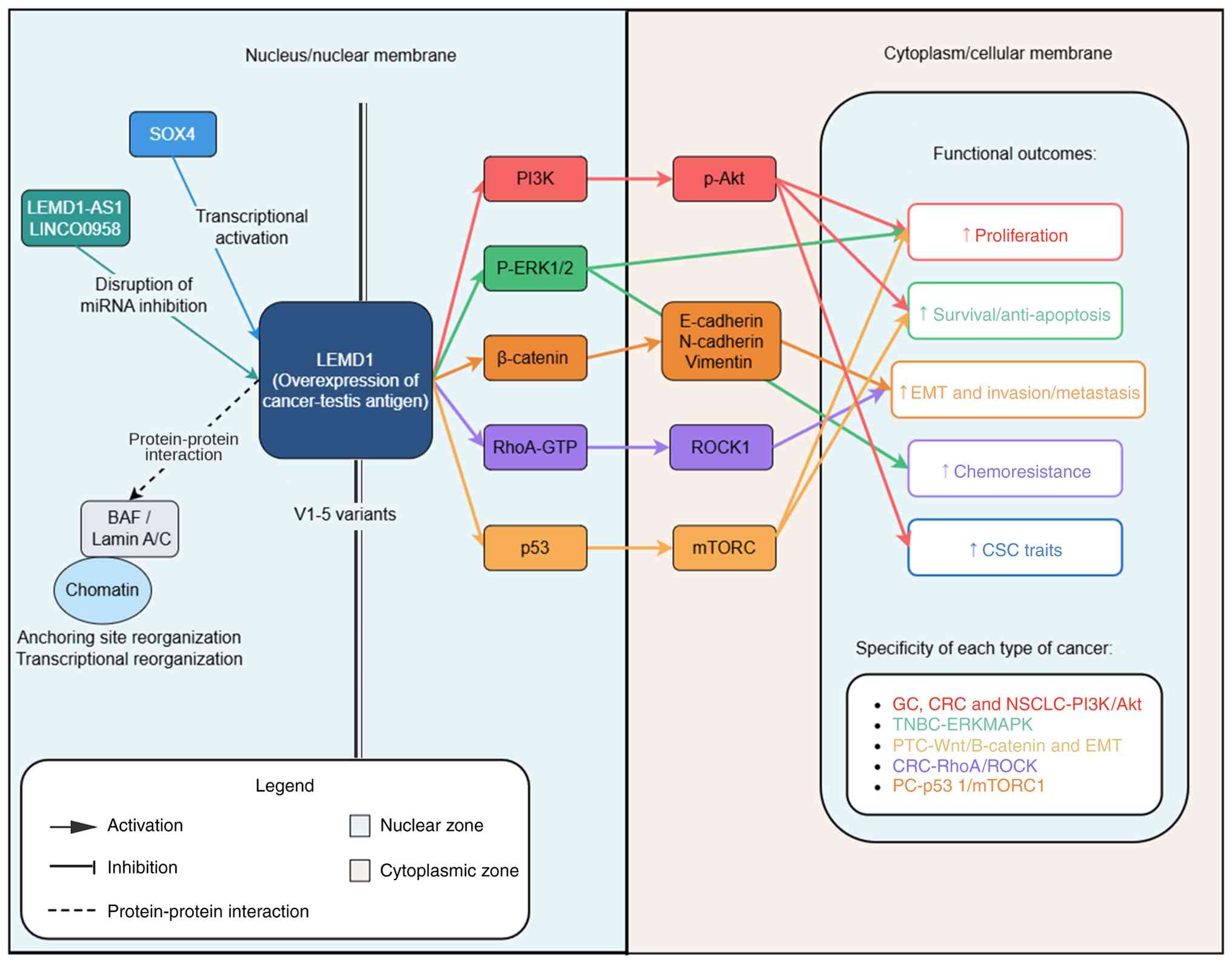 | Figure 4Schematic summary of the oncogenic
roles and signaling networks of LEMD1 in different types of tumors.
An integrated overview of the upstream regulators, downstream
signaling pathways and functional consequences associated with
LEMD1 overexpression across multiple types of cancer. In the
nucleus/nuclear membrane, SOX4 and LEMD1-AS1/LINC00958 are upstream
regulators of LEMD1, which contribute to transcriptional activation
and disruption of microRNA-mediated inhibition, respectively. LEMD1
overexpression activates multiple downstream signaling pathways,
including PI3K/p-Akt, ERK1/2, β-catenin, RhoA/ROCK and
p53/mTORC1-related pathways, and is associated with altered
expression of E-cadherin, N-cadherin and vimentin. These downstream
events are associated with increased proliferation,
survival/anti-apoptosis, the EMT and invasion/metastasis,
chemoresistance, and CSC traits (such as an enhanced sphere
formation ability, elevated expression of stemness markers and an
increased tumor-initiating potential). The tumor-type-specific
pathway associations reported in GC, CRC and NSCLC, as well as in
TNBC, PTC and PC are summarized. Arrowheads indicate activation,
T-shaped lines indicate inhibition, and dashed lines indicate
protein-protein interaction. The present figure was produced using
Adobe Illustrator 2024 (version 28; Adobe Systems, Inc.). BAF,
barrier-to-autointegration factor; CSC, cancer stem cell; CRC,
colorectal cancer; EMT, epithelial-mesenchymal transition; GC,
gastric cancer; LEMD1, LAP2-emerin-MAN1 domain-containing protein
1; LEMD1-AS1, LEMD1 antisense RNA 1; LINC00958, long intergenic
non-protein coding RNA 958; mTORC1, mechanistic target of rapamycin
complex 1; NSCLC, non-small cell lung cancer; p-Akt, phosphorylated
Akt; PC, pancreatic cancer; PTC, papillary thyroid cancer; ROCK,
Rho-associated coiled-coil containing protein kinase; RhoA, Ras
homolog family member A; SOX4, SRY-box transcription factor 4;
TNBC, triple-negative breast cancer. |
The Wnt/β-catenin pathway is also associated with
LEMD1. Using gene set enrichment analyses on thyroid cancer, a
study by Xu et al (27)
reveals that the Wnt/β-catenin pathway is notably enriched in
samples with high LEMD1 expression levels, downregulated E-cadherin
and upregulated N-cadherin and Vimentin, suggesting the occurrence
of the EMT. Further experiments confirm that knocking down LEMD1
reverses these changes, indicating that LEMD1 promotes
proliferation and migration of thyroid cancer cells by activating
the Wnt pathway to induce the EMT (27). Additionally, LEMD1 can influence
the activity of the MAPK signaling pathway. In TNBC, LEMD1
overexpression leads to the excessive activation of the ERK1/2
pathway, while silencing LEMD1 reduces downstream ERK signal
transduction, inhibiting tumor cell proliferation and migration
(11). Furthermore, the
interaction between LEMD1 and the p53/mTORC1 pathway is evident in
pancreatic cancer, in which excessive LEMD1 can inhibit the
function of the tumor suppressor protein p53 and activate mTORC1
signaling, promoting pancreatic cancer cell proliferation and
metastasis (2). Although the
precise mechanism is yet to be elucidated, LEMD1 activates
pro-survival pathways upstream of p53, including the PI3K/Akt/mTOR
pathway, which contributes to p53 inhibition and its reduced
pro-apoptotic function. LEMD1 also affects cytoskeleton
rearrangement-related pathways, such as the activation of
RhoA/ROCK1 signaling, which promotes stress fiber formation and
cell migration. A previous study investigating colorectal cancer
shows that overexpression of LEMD1 can increase RhoA activity and
enhance tumor cell migration, while interfering with LEMD1 has the
opposite effect (21). In summary,
LEMD1 may promote malignant tumor phenotypes through multi-target
signaling pathway interactions, including but not limited to
PI3K/Akt, Wnt/β-catenin, ERK/MAPK, p53/mTOR and RhoA/ROCK. Among
these pathways the PI3K/Akt axis appears to be a central downstream
node for the oncogenic effect of LEMD1, which supports its broad
involvement in several types of cancer.
LEMD1 appears to activate different downstream
signaling pathways depending on the type of tumor. For example, in
colorectal cancer, SOX4-induced transcriptional upregulation of
LEMD1 is linked to increased RhoA-GTP levels and enhanced cell
migration via activation of the RhoA/ROCK signaling cascade
(21). By contrast, in TNBC, LEMD1
overexpression downregulates ERK-inhibitory phosphatases such as
dual specificity phosphatases 5 and 6, sustaining ERK/MAPK pathway
activity, which in turn promotes tumor cell proliferation and the
EMT (11). This tissue-specific
signaling divergence may be attributed to differences in chromatin
accessibility, transcription factor repertoires or regulatory
cofactors. For example, in OSCC, particularly metastatic OSCC, the
long non-coding (lnc)RNA LEMD1-antisense RNA 1 (AS1) stabilizes
LEMD1 mRNA, which increases LEMD1 expression levels and activates
the PI3K-AKT signaling pathway (34). Additionally, the expression of
specific LEMD1 isoforms depending on the type of cancer, may
influence partner protein interactions, therefore mediating
distinct functional outcomes across tumor entities. These findings
explain the extensive biological effects of LEMD1 and indicates a
potential for further therapeutic interventions targeting
associated pathways.
Effects of LEMD1 on gene expression
levels and epigenetic regulation
As a nuclear membrane-associated protein, LEMD1 may
participate in tumorigenesis by influencing chromatin state and
gene expression levels. Firstly, expression of LEMD1 is subject to
epigenetic regulation. In normal cells, high methylation of the
LEMD1 promoter leads to transcriptional silencing, while in tumors
such as breast cancer, this promoter undergoes notable
demethylation, resulting in abnormally high LEMD1 expression
levels. This promoter demethylation positively correlates with poor
patient prognosis (11),
indicating that epigenetic activation of LEMD1 is linked to tumor
progression. Additionally, the 1q32 region, where the LEMD1 gene is
located, frequently undergoes copy number increases in certain
types of cancer (such as breast, endometrial and ovarian clear cell
carcinomas) (35-37),
which may further enhance LEMD1 expression levels (38). Besides DNA methylation and gene
amplification, transcription factors and non-coding RNAs also
participate in the regulatory network of the expression of LEMD1.
The SOX4 transcription factor can directly bind to the LEMD1
promoter and upregulate its transcription, thereby promoting the
activation of the LEMD1/PI3K/Akt axis in colon cancer cells
(30). In anaplastic lymphoma
kinase fusion-positive ALCL, the oncogene nucleophosmin-ALK induces
upregulation of microRNA (miR)-135b through the STAT3 pathway, and
miR-135b is located in the first intron of the LEMD1 gene. With the
upregulation of miR-135b, LEMD1 transcription is also co-activated
(38). This reveals that
inflammation-associated transcription factors or cytokine pathways
(such as the IL-3/STAT3 pathway) can indirectly upregulate LEMD1
through miR-135b in ALCL, suggesting a mechanistic link between
inflammatory signaling and LEMD1 expression levels.
Beyond ALCL, an associated inflammation linked
mechanism is described in epithelial tumors. In NSCLC, LEMD1
intronic miR-135b enhances NF-κB signaling by repressing
cylindromatosis tumor suppressor protein, which is a negative
regulator of the NF-κB pathway, with the expression of miR-135b
induced by IL-6/STAT3(39). As
miR-135b resides within the first intron of LEMD1, these
observations support a mechanistic connection between inflammatory
cytokine pathways and the LEMD1 locus in solid tumors, potentially
coupling IL-6/STAT3 input to NF-κB activation through an IL-6/STAT3
to miR-135b to NF-κB axis (39).
This reveals that inflammatory/cytokine signaling can promote the
expression of LEMD1 through epigenetic pathways. This connects
stimulation of the tumor microenvironment with the expression of
oncogenes.
Furthermore, lncRNAs also serve roles in regulating
LEMD1. In OSCC, LEMD1-AS1 binds to LEMD1 mRNA, stabilizing its
transcript level. This increases the LEMD1 expression levels and
activates the PI3K/Akt pathway to promote tumor metastasis
(34). Similarly, LINC00958 lncRNA
indirectly enhances the expression of LEMD1 by absorbing
miR-3064-5p (which can target and downregulate LEMD1). This
enhances the malignant behavior of tumor cells (31). LEMD1-AS1, as a cuproptosis-related
tumor suppressor, not only facilitates molecular subtyping and
prognostic evaluation of ovarian cancer, but is also a potential
biomarker for guiding personalized treatment strategies (40). These findings indicate that LEMD1
is involved in a complex gene regulatory network, with its
expression levels subject to multi-level regulation including DNA
methylation status, copy number variations, transcription factors
(such as SOX4 and STAT3) and ncRNAs. These mechanisms not only
determine the expression level of LEMD1 itself but may also
influence the expression patterns of a number of downstream genes
through LEMD1, which warrants additional in-depth research in the
future.
In addition to LEMD1 expression levels, its location
at the inner nuclear membrane suggests it may have direct effects
on gene regulation. As with other LEM-domain proteins, LEMD1 may
anchor chromatin to the nuclear periphery through interactions with
BAF or the nuclear lamina, potentially silencing or activating
specific gene regions (41).
Although direct evidence is still lacking, such a mechanism may
explain how LEMD1 influences a broad range of cellular behaviors.
If LEMD1 tethers genomic regions associated with tumor suppressor
genes or differentiation genes to the nuclear periphery, this may
repress their expression levels, contributing to the maintenance of
a proliferative, undifferentiated state in cancer cells. However,
detachment of chromatin from the repressive nuclear periphery due
to LEMD1 loss may activate tumor suppressor networks, which is
consistent with observations of increased p53 pathway activity upon
LEMD1 knockdown (2). These
hypotheses require further validation, but they highlight that the
pro-oncogenic effects of LEMD1 may stem from a combination of
signaling pathway activation and higher-order genome organization
changes.
5. Therapeutic potential of LEMD1 in
cancer
Prospects of LEMD1 as a therapeutic
target
Highly specific expression and tumor-promoting
effects of LEMD1 in tumors make it an attractive therapeutic
target. Traditional molecular targeting approaches may consider
inhibiting the function (via small molecules) or expression [via
small interfering (si)RNA and short hairpin RNA gene-silencing
strategies] of LEMD1 to inhibit tumor progression. However, as
LEMD1 is an intracellular nuclear envelope protein, designing
small-molecule drugs that act on its transmembrane region or LEM
domain is challenging. Gene-level interventions, such as via siRNA
or antisense oligonucleotides, demonstrates notable antitumor
effects in vivo (11). For
example, in mouse models of TNBC, the silencing of LEMD1 inhibits
tumor growth and metastasis (11),
suggesting that therapies targeting LEMD1 may potentially reduce
tumor malignancy. While monoclonal antibody or immunoconjugate
approaches may be theoretically considered, the predominant inner
nuclear membrane localization of LEMD1 prevents the accessibility
of extracellular epitopes, rendering such strategies impractical
for direct targeting.
A more feasible approach is the possible use of the
classification of LEMD1 as a CTA for immunotherapeutic intervention
(11). Under physiological
conditions, LEMD1 expression levels are mostly restricted to
immune-privileged germ cells, which minimizes central immune
tolerance and potentially facilitates tumor-specific immune
responses (3). Nevertheless,
developing effective LEMD1-targeted immunotherapies poses several
challenges. Firstly, intratumoral heterogeneity in LEMD1 expression
levels presents a risk of immune escape through antigen
downregulation, although this risk may be mitigated if LEMD1 is
functionally indispensable for tumor cell survival. Secondly, for
vaccine development, a critical prerequisite is the identification
of immunogenic LEMD1-derived epitopes that can be presented by
common HLA class I molecules (42). The robust activation of cytotoxic T
lymphocytes against these epitopes would be essential to overcome
any residual immune tolerances and mount effective tumor-specific
immune responses. However, previous CTA-targeted cancer vaccines,
such as recombinant melanoma-associated antigen A3 protein vaccines
and New York esophageal squamous cell carcinoma-1 (NY-ESO-1)
protein vaccines formulated with immune-stimulating complex matrix
adjuvant, only had modest clinical efficacy, with objective
response rates of ~13% in trials (43-46),
highlighting the necessity for potent adjuvants or combination
immunotherapies (such as immune checkpoint inhibitors) to enhance
the antitumor immunity. Additionally, innovative delivery systems
capable of targeting intracellular LEMD1 warrant further
investigation. In terms of immunotherapy, LEMD1, as a CTA, is a
possible target for tumor vaccines or T-cell therapies. Since
expression of LEMD1 is limited in normal tissues (occurring mainly
in the testis), immune responses against it would be tumor-specific
and spare normal cells (21). It
may be considered to use LEMD1 peptide vaccines to activate the T
cells of patients, or to expand cytotoxic T lymphocytes that
recognize LEMD1 ex vivo and reinfuse them to target
LEMD1-positive tumors. Additionally, chimeric antigen receptor
T-cell therapy may be adapted to recognize LEMD1-derived peptides
presented on tumor cell MHC molecules, provided suitable epitopes
and HLA restrictions are identified. Furthermore, advanced drug
delivery systems could possibly be used to overcome the challenge
of the intranuclear location (inner nuclear membrane) of LEMD1. For
example, nanoparticle-based carriers or ligand-directed vehicles
could potentially deliver LEMD1-targeted siRNA or protein-degrading
agents specifically into tumor cells. In summary, LEMD1 has
potential promising prospects as a therapeutic target, and these
strategies (from CTA-targeted immunotherapies to gene silencing
with innovative delivery mechanisms) warrant further
investigation.
Role of LEMD1 in anticancer drug
resistance
Tumor resistance is one of the key challenges
affecting treatment efficacy, and increasing evidence suggests
tumor-promoting genes such as LEMD1 serve a role in this process.
LEMD1 enhances tumor cell tolerance to adverse environments by
activating survival pathways such as PI3K/Akt and ERK, which may
also include tolerance to stress induced by chemotherapeutic drugs.
TNBC research reveals that knocking down LEMD1 not only inhibits
tumor proliferation but also notably increases the sensitivity of
tumor cells to paclitaxel chemotherapy (11). By contrast, TNBC cells with high
LEMD1 expression levels are relatively resistant to paclitaxel,
requiring higher drug doses to achieve the same inhibitory effect
(11). This finding suggests that
the presence of LEMD1 may confer a degree of chemotherapy
resistance to tumor cells.
The mechanism of chemotherapy resistance may be
associated with the antiapoptotic capacity that is promoted by
LEMD1(4) chemotherapeutic drugs
typically kill cancer cells by inducing DNA damage and apoptosis,
while the inhibition of apoptosis by LEMD1 would reduce the effect
of the drug. Additionally, the cancer cell stemness and the EMT
state maintained by LEMD1 are also hypothesized to be associated
with resistant phenotypes. Stem cell-like tumor cells often have an
increased tolerance to radio- and chemotherapy, which may explain
why LEMD1-enhanced stemness in NSCLC leads to more stubborn tumors
that are difficult to eliminate (28). At the same time, the EMT process is
often accompanied by increased chemotherapy resistance, and the
LEMD1-induced EMT (27) may render
tumor cells insensitive to certain drugs.
LEMD1 appears to mediate chemotherapy resistance
through multiple, reinforcing mechanisms. One major mechanism is
the activation of pro-survival signaling pathways such as PI3K/Akt.
LEMD1 overexpression activates the PI3K/Akt cascade in cancer cells
(4), which is notable because
PI3K/Akt signaling confers broad chemoresistance by upregulating
antiapoptotic effectors (such as Bcl-2 and X-linked inhibitor of
apoptosis protein) and suppressing proapoptotic factors (such as
Bax) (47). Consistently, LEMD1
knockdown sensitizes tumor cells to paclitaxel. For example, in
vitro, silencing LEMD1 in paclitaxel-treated MDA-MB-231 TNBC
cells downregulates Bcl-2 and increases Bax expression levels,
which enhances apoptosis and chemosensitivity, compared with those
with paclitaxel treatment alone (11).
Another mechanism is through inducing the EMT and
enriching cancer stem-like properties, which are well-known
inducers of drug resistance. LEMD1 silencing increases E-cadherin
and decreases N-cadherin/vimentin (mesenchymal markers) (11), indicating that LEMD1 normally
promotes a mesenchymal, migratory cell state. This shift is notable
as the EMT not only facilitates invasion but also often coincides
with the upregulation of drug efflux transporters, such as
ATP-binding cassette (ABC) sub-family B member 1/P-glycoprotein
(multidrug resistance protein 1), ABC sub-family C member
1/multidrug resistance-associated protein 1 and ABC sub-family G
member 2/breast cancer resistance protein (48-50),
as well as other adaptive mechanisms, including enhanced resistance
to apoptosis, increased DNA damage repair capacity and acquisition
of cancer stem cell-like properties (51-53),
that allow cells to evade chemotherapeutics (54). In addition, the EMT is frequently
linked to the emergence of cancer stem-like cells, which is a
subpopulation of tumor cells that are intrinsically more
chemo-resistant, partly due to their quiescent nature and ability
to survive drug-induced stress (54). By supporting a mesenchymal
phenotype and potentially maintaining a pool of stem-like cells,
LEMD1 endows tumor cells with a capacity to withstand chemotherapy,
which may explain why LEMD1-enhanced stemness in NSCLC leads to
more stubborn tumors that are difficult to eliminate (28). In summary, high LEMD1 expression
levels can protect cancer cells from chemotherapy-induced apoptosis
(via survival pathways such as PI3K/Akt) while promoting cellular
traits (such as the EMT and stemness) that undermine drug efficacy.
Clinically, this suggests that tumors with elevated LEMD1
expression levels may be predisposed to chemoresistance, and
conversely that targeting LEMD1 or its downstream signals may help
overcome resistance and improve the response to treatment (11).
Therefore, it can be hypothesized that inhibiting
LEMD1 in tumors may synergistically enhance the efficacy of
conventional treatments. Although current direct research on LEMD1
and drug resistance is limited, the aforementioned indications
suggest its potential importance. Future research is needed to
further investigate the effects of LEMD1 on the sensitivity to
various treatments (such as chemotherapy, targeted drugs and
radiotherapy) in different tumor contexts. If the resistance
spectrum corresponding to high LEMD1 expression levels can be
clarified, it may help formulate more optimized personalized
treatment plans.
6. Current research challenges and future
directions
Shortcomings and challenges in current
research
Despite notable progress in research on LEMD1 in
tumors, there are still a number of shortcomings and challenges to
overcome. Current basic research on the molecular biological
functions of LEMD1 is not in depth enough, with the majority of
studies focusing on observing the effects of LEMD1 up- or
downregulation on tumor phenotypes and signaling pathways, lacking
direct evidence for its specific role in nuclear structure and gene
regulation as a nuclear inner membrane protein. For example, it is
still unclear whether LEMD1 reshapes chromatin structure through
interactions with BAF or nuclear lamina proteins to affect gene
expression levels. There are also challenges regarding the
‘druggability’ of LEMD1 as a drug target. Since LEMD1 has no
enzymatic active site and is located on the inner nuclear membrane,
the use of traditional small molecule drugs to directly target it
is difficult. Therefore, innovative drug delivery systems or new
types of molecules are needed to achieve effective
intervention.
The heterogeneity of LEMD1 expression levels and the
functional differences across different types of tumor are yet to
be fully elucidated. Although the majority of studies consistently
suggest the cancer-promoting effects of LEMD1, there may be
exceptions in certain types of cancer. For example, in multiple
myeloma, a large-scale study involving 2,546 patients identifies
LEMD1 as a favorable prognostic marker in which patients with high
LEMD1 expression levels exhibit notably prolonged survival compared
with those with low expression (55). In prostatic adenocarcinoma, LEMD1
is frequently upregulated in tumor tissues; however, a
comprehensive study failed to demonstrate a notable prognostic
impact of LEMD1 in prostate cancer (unlike other LEM family
proteins associated with clinical outcomes) (6). By contrast, in ovarian cancer,
elevated LEMD1 expression levels are associated with improved
patient survival. An integrative genomics study reveals that
patients with ovarian tumors and high LEMD1 expression levels have
a markedly longer OS compared with the low-expression level group
(55,56). This outlier implies that the
oncogenic role of LEMD1 is not universal and might be modulated by
tissue-specific contexts. One hypothesis is that the immune
microenvironment in ovarian cancer may respond uniquely to LEMD1.
As an immunogenic CTA, high LEMD1 expression levels in ovarian
tumors might stimulate stronger anti-tumor immune responses, making
the tumor more immunologically ‘hot’ and contributing to improved
patient outcomes (57,58). In support of this, a number of CTAs
are associated with an inflamed tumor microenvironment and
favorable clinical outcomes in specific types of cancer. For
example, NY-ESO-1 expression levels in TNBC are linked to higher
tumor-infiltrating lymphocyte levels, favorable disease-free
survival and higher CD8+ T-cell counts; Hemogen
expression levels in lung adenocarcinoma are associated with
CD8+ T-cell infiltration and an improved prognosis; and
CTA-high seminomas, including preferentially expressed antigen in
melanoma-enriched tumors, demonstrate activated T-cell
infiltration, with CTA and CD8+/regulatory T-cell
signatures correlating with recurrence-free survival (57,59-62).
Alternatively, ovarian tumors might harbor unique co-factors or
signaling contexts (such as hormone-related factors or distinct
co-regulated pathways) that alter the downstream effects of LEMD1.
In gynecological cells, LEMD1 may interact with tissue-specific
partners or regulatory networks, potentially dampening its
tumor-promoting activities or even associating its high expression
levels with a more differentiated, less aggressive tumor phenotype.
In summary, LEMD1 may serve different biological roles in ovarian
cancer compared with other malignancies, highlighting the
importance of context-dependent research into its functions.
Furthermore, immunotherapy targeting LEMD1 is yet to be clinically
implemented, and its feasibility and safety are yet to be verified.
Additionally, as a member of the CTA family, the potential
immunogenicity of LEMD1 is both an advantage and a challenge as it
may promote the immune clearance of tumors but it might also be
limited by tumor immune evasion mechanisms or even trigger
autoimmune responses. Therefore, all of these issues require
further investigation.
Future research directions
To address the aforementioned shortcomings, several
directions may be worth investigating in the future. At the
fundamental level, the molecular mechanism of LEMD1 should be
analyzed in depth. Proteomics and biochemical methods could be used
to identify LEMD1-interacting proteins and clarify whether they
directly bind BAF, chromatin or nuclear lamina components.
Techniques such as chromatin immunoprecipitation sequencing could
be used to identify genomic regions affected by LEMD1 and
investigate its regulatory role in genome-wide transcription and
epigenetic modifications. This may possibly reveal the fundamental
mechanisms of the cancer-promoting effects of LEMD1. Additionally,
the function of LEMD1 should be further evaluated in cellular and
animal models. For example, LEMD1 gene knockout mouse models could
potentially be constructed to observe possible phenotypic changes
in normal development and tumor susceptibility, in order to
determine whether LEMD1 is a necessary factor for
tumorigenesis.
Furthermore, larger-scale correlation studies should
be conducted in clinical samples. This includes detecting LEMD1
expression profiles in different types of cancer and at different
stages, combined with patient treatment and survival data, to
assess the reliability and independence of LEMD1 as a prognostic or
predictive marker. Investigating LEMD1-related immunotherapy should
also be advanced. For example, by identifying the T cell epitopes
of LEMD1, the presence of natural anti-LEMD1 immune responses in
the peripheral blood of patients with cancer could be evaluated.
Once its immunogenicity is proven, attempts can be made to develop
LEMD1 vaccines or CAR-T cells, testing their anti-tumor effects in
animal models. Through these multi-faceted research efforts, a
foundation will be laid for LEMD1 to progress from laboratory to
clinical application.
7. Conclusion
As a CTA, LEMD1 demonstrates consistent
tumor-promoting effects and notable clinical relevance across
multiple tumor types. Its unique expression profile and molecular
functions make it a focal point in tumor biology research and a
possible novel opportunity for tumor therapy. Current research
preliminarily reveals various mechanisms by which LEMD1 promotes
tumor development, including activating proliferation signaling
pathways, inducing the EMT and maintaining cancer stem cell
characteristics. Furthermore, the clinicopathological importance of
LEMD1 has become clear, serving as a marker of poor prognosis in
several tumor types. Considering this, continuing in-depth research
investigating the mechanisms of action of LEMD1 and evaluating its
possible value as a diagnostic and therapeutic target has
scientific importance and potential clinical translation prospects.
Therefore, as the understanding of LEMD1 increases, this molecule
may potentially be incorporated into the landscape of precision
oncology, which may possibly provide patients with more effective
individualized diagnostic and treatment options. It may be
anticipated that multidisciplinary research investigating LEMD1
could potentially advance the understanding of the complex network
of tumor development and progression, which may potentially offer
novel treatments for refractory tumors.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
XiaoW conceived the review, conducted the literature
search, verified all source literature and the publicly available
data retrieved from the Gene Expression Omnibus for Fig. 1, and drafted the initial
manuscript. XianW revised the manuscript for important intellectual
content and provided supervisory guidance. SK critically revised
the manuscript for important intellectual content, verified all raw
data and source literature, and provided supervisory guidance. All
authors read and approved the final version of the manuscript. Data
authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, artificial
intelligence tools were used to improve the readability and
language of the manuscript or to generate images, and subsequently,
the authors revised and edited the content produced by the
artificial intelligence tools as necessary, taking full
responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Yuki D, Lin YM, Fujii Y, Nakamura Y and
Furukawa Y: Isolation of LEM domain-containing 1, a novel
testis-specific gene expressed in colorectal cancers. Oncol Rep.
12:275–280. 2004.PubMed/NCBI
|
|
2
|
Cao X, Yao N, Zhao Z, Fu Y, Hu Y, Zhu P,
Shi W and Tang L: LEM domain containing 1 promotes pancreatic
cancer growth and metastasis by p53 and mTORC1 signaling pathway.
Bioengineered. 13:7771–7784. 2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Gjerstorff MF, Andersen MH and Ditzel HJ:
Oncogenic cancer/testis antigens: Prime candidates for
immunotherapy. Oncotarget. 6:15772–15787. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Li Q, Ge Y, Chen X, Wang L, Xia Y and Xu
Z, Li Z, Wang W, Yang L, Zhang D and Xu Z: LEM domain containing 1
promotes proliferation via activating the PI3K/Akt signaling
pathway in gastric cancer. J Cell Biochem. 120:15190–15201.
2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Sasahira T and Kirita T: Hallmarks of
cancer-related newly prognostic factors of oral squamous cell
carcinoma. Int J Mol Sci. 19(2413)2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
He T, Zhang Y, Li X, Liu C, Zhu G, Yin X,
Zhang Z, Zhao K, Wang Z, Zhao P and Wang K: Collective analysis of
the expression and prognosis for LEM-domain proteins in prostate
cancer. World J Surg Oncol. 20(174)2022.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Hashimoto K, Nishimura S, Ito T, Oka N,
Kakinoki R and Akagi M: Clinicopathological assessment of
cancer/testis antigens NY-ESO-1 and MAGE-A4 in osteosarcoma. Eur J
Histochem. 66(3377)2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Wang H, Liu J, Yang J, Wang Z, Zhang Z,
Peng J, Wang Y and Hong L: A novel tumor mutational burden-based
risk model predicts prognosis and correlates with immune
infiltration in ovarian cancer. Front. Immunol.
13(943389)2022.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Sasahira T, Kurihara M, Nakashima C,
Kirita T and Kuniyasu H: LEM domain containing 1 promotes oral
squamous cell carcinoma invasion and endothelial transmigration. Br
J Cancer. 115:52–58. 2016.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Sasahira T, Kurihara-Shimomura M,
Nishiguchi Y, Shimomura H and Kirita T: Sushi repeat containing
protein X-linked 2 is a downstream signal of LEM domain containing
1 and acts as a tumor-promoting factor in oral squamous cell
carcinoma. Int J Mol Sci. 21(3655)2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Li X, Jiang S, Jiang T, Sun X, Guan Y, Fan
S and Cheng Y: LEM domain containing 1 acts as a novel oncogene and
therapeutic target for triple-negative breast cancer. Cancers
(Basel). 15(2924)2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Wang Y, Chen Z, Yang G and Yuan G:
Unveiling the roles of LEMD proteins in cellular processes. Life
Sci. 357(123116)2024.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Rose M, Burgess JT, O'Byrne K, Richard DJ
and Bolderson E: The role of inner nuclear membrane proteins in
tumourigenesis and as potential targets for cancer therapy. Cancer
Metastasis Rev. 41:953–963. 2022.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Takeda R, Hirohashi Y, Shen M, Wang L,
Ogawa T, Murai A, Yamamoto E, Kubo T, Nakatsugawa M, Kanaseki T, et
al: Identification and functional analysis of variants of a
cancer/testis antigen LEMD1 in colorectal cancer stem-like cells.
Biochem Biophys Res Commun. 485:651–657. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Barton LJ, Soshnev AA and Geyer PK:
Networking in the nucleus: a spotlight on LEM-domain proteins. Curr
Opin Cell Biol. 34:1–8. 2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Brachner A and Foisner R: Evolvement of
LEM proteins as chromatin tethers at the nuclear periphery. Biochem
Soc Trans. 39:1735–1741. 2011.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Wagner N and Krohne G: LEM-Domain
proteins: New insights into lamin-interacting proteins. Int Rev
Cytol. 261:1–26. 2007.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Herrada I, Bourgeois B, Samson C, Buendia
B, Worman HJ and Zinn-Justin S: Purification and structural
analysis of LEM-Domain proteins. Methods Enzymol. 569:43–61.
2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ai H, Yang H, Li L, Ma J, Liu K and Li Z:
Cancer/testis antigens: Promising immunotherapy targets for
digestive tract cancers. Front Immunol. 14(1190883)2023.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Li H, Chiappinelli KB, Guzzetta AA,
Easwaran H, Yen RW, Vatapalli R, Topper MJ, Luo J, Connolly RM,
Azad NS, et al: Immune regulation by low doses of the DNA
methyltransferase inhibitor 5-azacitidine in common human
epithelial cancers. Oncotarget. 5:587–598. 2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Zhang H, Xu C, Jiang F and Feng J: A
three-genes signature predicting colorectal cancer relapse reveals
LEMD1 promoting CRC cells migration by RhoA/ROCK1 signaling
pathway. Front Oncol. 12(823696)2022.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Martin CJ, Oser EA, Nagarajan P, Popova
LV, Sunkel BD, Stanton BZ and Parthun MR: Distinct classes of
lamina-associated domains are defined by differential patterns of
repressive histone methylation. Genome Res. 35:1959–1974.
2025.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Ghafouri-Fard S, Ousati Ashtiani Z, Sabah
Golian B, Hasheminasab SM and Modarressi MH: Expression of two
testis-specific genes, SPATA19 and LEMD1, in prostate cancer. Arch
Med Res. 41:195–200. 2010.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Elkhatib RA, Paci M, Boissier R, Longepied
G, Auguste Y, Achard V, Bourgeois P, Levy N, Branger N, Mitchell MJ
and Metzler-Guillemain C: LEM-domain proteins are lost during human
spermiogenesis but BAF and BAF-L persist. Reproduction.
154:387–401. 2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Karlsson M, Zhang C, Méar L, Zhong W,
Digre A, Katona B, Sjöstedt E, Butler L, Odeberg J, Dusart P, et
al: A single-cell type transcriptomics map of human tissues. Sci
Adv. 7(eabh2169)2021.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Okada Y: Sperm chromatin condensation:
Epigenetic mechanisms to compact the genome and spatiotemporal
regulation from inside and outside the nucleus. Genes Genet Syst.
97:41–53. 2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Xu M, Lin B, Zheng D, Wen J, Hu W, Li C,
Zhang X, Zhang X and Qu J: LEM domain containing 1 promotes thyroid
cancer cell proliferation and migration by activating the
Wnt/β-catenin signaling pathway and epithelial-mesenchymal
transition. Oncol. Lett. 21(442)2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Li L and Zhang P: Elevation of LEM domain
containing 1 predicts poor prognosis of NSCLC patients and triggers
malignant stemness and invasion of NSCLC cells by stimulating
PI3K/AKT pathway. Curr Mol Med. 24:366–378. 2024.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Matsuyama H, Suzuki HI, Nishimori H,
Noguchi M, Yao T, Komatsu N, Mano H, Sugimoto K and Miyazono K:
miR-135b mediates NPM-ALK-driven oncogenicity and renders
IL-17-producing immunophenotype to anaplastic large cell lymphoma.
Blood. 118:6881–6892. 2011.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Li D, Wang D, Liu H and Jiang X: LEM
domain containing 1 (LEMD1) transcriptionally activated by
SRY-related high-mobility-group box 4 (SOX4) accelerates the
progression of colon cancer by upregulating phosphatidylinositol
3-kinase (PI3K)/protein kinase B (Akt) signaling pathway.
Bioengineered. 13:8087–8100. 2022.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Luo Z, Hao S, Yuan J, Zhu K, Liu S, Zhang
J and Yao L: Long non-coding RNA LINC00958 promotes colorectal
cancer progression by enhancing the expression of LEM domain
containing 1 via microRNA miR-3064-5p. Bioengineered. 12:8100–8115.
2021.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Martinez-Romero J, Bueno-Fortes S,
Martín-Merino M, Ramirez de Molina A and De Las Rivas J: Survival
marker genes of colorectal cancer derived from consistent
transcriptomic profiling. BMC Genomics. 19 (Suppl
8)(S857)2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Maimela PWM, Smith M, Nel AJM, Bernam SDP,
Jonas EG and Blackburn JM: Humoral immunoprofiling identifies novel
biomarkers and an immune suppressive autoantibody phenotype at the
site of disease in pancreatic ductal adenocarcinoma. Front Oncol.
14(1330419)2024.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Li Z, Wang J, Wu J, Li N and Jiang C: Long
Noncoding RNA LEMD1-AS1 increases LEMD1 expression and activates
PI3K-AKT pathway to promote metastasis in oral squamous cell
carcinoma. Biomed Res Int. 2022(3543948)2022.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Mesquita B, Lopes P, Rodrigues A, Pereira
D, Afonso M, Leal C, Henrique R, Lind GE, Jerónimo C, Lothe RA and
Teixeira MR: Frequent copy number gains at 1q21 and 1q32 are
associated with overexpression of the ETS transcription factors
ETV3 and ELF3 in breast cancer irrespective of molecular subtypes.
Breast Cancer Res Treat. 138:37–45. 2013.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Depreeuw J, Stelloo E, Osse EM, Creutzberg
CL, Nout RA, Moisse M, Garcia-Dios DA, Dewaele M, Willekens K,
Marine JC, et al: Amplification of 1q32.1 refines the molecular
classification of endometrial carcinoma. Clin Cancer Res.
23:7232–7241. 2017.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Tan DS, Lambros MB, Rayter S, Natrajan R,
Vatcheva R, Gao Q, Marchiò C, Geyer FC, Savage K, Parry S, et al:
PPM1D is a potential therapeutic target in ovarian clear cell
carcinomas. Clin Cancer Res. 15:2269–2280. 2009.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Khatri R and Subramanian S: MicroRNA-135b
and its circuitry networks as potential therapeutic targets in
colon cancer. Front Oncol. 3(268)2013.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Zhao J, Wang X, Mi Z, Jiang X, Sun L,
Zheng B, Wang J, Meng M, Zhang L, Wang Z, et al:
STAT3/miR-135b/NF-κB axis confers aggressiveness and unfavorable
prognosis in non-small-cell lung cancer. Cell Death Dis.
12(493)2021.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Li N, Yu K, Huang D, Li S, Zeng D, Li J
and Fan L: Molecular characterization of cuproptosis-related
lncRNAs: Defining molecular subtypes and a prognostic signature of
ovarian cancer. Biol Trace Elem Res. 202:1428–1445. 2024.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Shevelyov YY: Interactions of chromatin
with the nuclear lamina and nuclear pore complexes. Int J Mol Sci.
24(15771)2023.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Akiyama Y, Komiyama M, Miyata H, Yagoto M,
Ashizawa T, Iizuka A, Oshita C, Kume A, Nogami M, Ito I, et al:
Novel cancer-testis antigen expression on glioma cell lines derived
from high-grade glioma patients. Oncol Rep. 31:1683–1690.
2014.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Kruit WH, Suciu S, Dreno B, Mortier L,
Robert C, Chiarion-Sileni V, Maio M, Testori A, Dorval T, Grob JJ,
et al: Selection of immunostimulant AS15 for active immunization
with MAGE-A3 protein: Results of a randomized phase II study of the
European organisation for research and treatment of cancer melanoma
group in metastatic melanoma. J Clin Oncol. 31:2413–2420.
2013.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Kruit WH, Van Ojik HH, Brichard VG,
Escudier B, Dorval T, Dréno B, Patel P, Van Baren N, Avril MF,
Piperno S, et al: Phase 1/2 study of subcutaneous and intradermal
immunization with a recombinant MAGE-3 protein in patients with
detectable metastatic melanoma. Int J Cancer. 117:596–604.
2005.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Nicholaou T, Ebert LM, Davis ID, McArthur
GA, Jackson H, Dimopoulos N, Tan B, Maraskovsky E, Miloradovic L,
Hopkins W, et al: Regulatory T-Cell-mediated attenuation of T-Cell
responses to the NY-ESO-1ISCOMATRIX vaccine in patients with
advanced malignant melanoma. Clin Cancer Res. 15:2166–2173.
2009.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Jiang J, Li L, Yu Z, Wang K, Ma J, Li D,
Guo Z, Wang L, Zhang H, Xu L and Zhang M: Clinical efficacy and
challenges of cancer-testis antigen vaccines in advanced solid
tumors: A systematic review and meta-analysis. Crit Rev Oncol
Hematol. 216(104981)2025.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Liu R, Chen Y, Liu G, Li C, Song Y, Cao Z,
Li W, Hu J, Lu C and Liu Y: PI3K/AKT pathway as a key link
modulates the multidrug resistance of cancers. Cell Death Dis.
11(797)2020.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Saxena M, Stephens MA, Pathak H and
Rangarajan A: Transcription factors that mediate
epithelial-mesenchymal transition lead to multidrug resistance by
upregulating ABC transporters. Cell Death Dis.
2(e179)2011.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Liu YR, Liang L, Zhao JM, Zhang Y, Zhang
M, Zhong WL, Zhang Q, Wei JJ, Li M, Yuan J, et al: Twist1 confers
multidrug resistance in colon cancer through upregulation of
ATP-binding cassette transporters. Oncotarget. 8:52901–52912.
2017.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Wang H, Li JM, Wei W, Yang R, Chen D, Ma
XD, Jiang GM and Wang BL: Regulation of ATP-binding cassette
subfamily B member 1 by Snail contributes to chemoresistance in
colorectal cancer. Cancer Sci. 111:84–97. 2020.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Debaugnies M, Rodríguez-Acebes S, Blondeau
J, Parent MA, Zocco M, Song Y, de Maertelaer V, Moers V, Latil M,
Dubois C, et al: RHOJ controls EMT-associated resistance to
chemotherapy. Nature. 616:168–175. 2023.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Hu J, Wang J, Guo X, Fan Q, Li X, Li K,
Wang Z, Liang S, Amin B, Zhang N, et al: MSLN induced EMT, cancer
stem cell traits and chemotherapy resistance of pancreatic cancer
cells. Heliyon. 10(e29210)2024.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Nie F, Sun X, Sun J, Zhang J and Wang Y:
Epithelial-mesenchymal transition in colorectal cancer metastasis
and progression: Molecular mechanisms and therapeutic strategies.
Cell Death Discov. 11(336)2025.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Błaszczak E, Miziak P, Odrzywolski A,
Baran M, Gumbarewicz E and Stepulak A: Triple-Negative breast
cancer progression and drug resistance in the context of
epithelial-mesenchymal transition. Cancers (Basel).
17(228)2025.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Park J, Chiu B, Delaney J, Paschal C,
Roche D, Shima J, Umesh A, Wisotzkey R, Shekar M and Kupershmidt I:
Abstract 5151: Integrative genomic analysis identifies
cancer-testis antigen LEMD1 as a prognostic biomarker. Cancer Res.
73 (Suppl 8)(5151)2013.
|
|
56
|
Taherian-Esfahani Z, Abedin-Do A, Nikpayam
E, Tasharofi B, Ghahghaei Nezamabadi A and Ghafouri-Fard S:
Cancer-Testis Antigens: A Novel Group of Tumor Biomarkers in
Ovarian Cancers. Int J Cancer Manag. 9(e4993)2016.
|
|
57
|
da Silva VL, Fonseca AF, Fonseca M, da
Silva TE, Coelho AC, Kroll JE, de Souza JES, Stransky B, de Souza
GA and de Souza SJ: Genome-wide identification of cancer/testis
genes and their association with prognosis in a pan-cancer
analysis. Oncotarget. 8:92966–92977. 2017.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Lin L, Zou X, Nong W, Ge Y, Li F, Luo B,
Zhang Q and Xie X: The potential value of cancer-testis antigens in
ovarian cancer: Prognostic markers and targets for immunotherapy.
Immun. Inflamm. Dis. 12(e1284)2024.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Ademuyiwa FO, Bshara W, Attwood K,
Morrison C, Edge SB, Karpf AR, James SA, Ambrosone CB, O'Connor TL,
Levine EG, et al: NY-ESO-1 cancer testis antigen demonstrates high
immunogenicity in triple negative breast cancer. PLoS One.
7(e38783)2012.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Lee HJ, Kim JY, Song IH, Park IA, Yu JH
and Gong G: Expression of NY-ESO-1 in triple-negative breast cancer
is associated with tumor-infiltrating lymphocytes and a good
prognosis. Oncology. 89:337–344. 2015.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Jiang Y, Yu L, Hu Q, Kang Y, You J, Huang
C, Xu X and Chen L: Cancer/testis antigen HEMGN correlated with
immune infiltration serves as a prognostic biomarker in lung
adenocarcinoma. Mol Immunol. 153:226–237. 2023.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Siska PJ, Johnpulle RAN, Zhou A, Bordeaux
J, Kim JY, Dabbas B, Dakappagari N, Rathmell JC, Rathmell WK,
Morgans AK, et al: Deep exploration of the immune infiltrate and
outcome prediction in testicular cancer by quantitative multiplexed
immunohistochemistry and gene expression profiling. Oncoimmunology.
6(e1305535)2017.PubMed/NCBI View Article : Google Scholar
|