Introduction
Traumatic carotid-cavernous fistula (TCCF) is an
acquired pathological arteriovenous shunt between the internal
carotid artery (ICA) and the cavernous sinus (CS) secondary to
traumatic brain injury (1). It
accounts for ~2.5-3% of all intracranial vascular malformations
(2). The diagnosis of TCCF is based
on clinical manifestations and is confirmed by radiological
modalities. Typical TCCFs drain into the ophthalmic veins, which
causes the progressive congestion of venous sinuses and subsequent
pathognomonic ocular symptoms. However, the perimedullary venous
drainage pathway and the presentation of delayed myelopathy in TCCF
are rare, and only three cases to date have been reported (3-5), at
least to the best of our knowledge. In addition, low-flow direct
CCFs are also rarely reported. Due to its rarity, there is limited
information available regarding the clinical characteristics and
the underlying mechanisms of delayed myelopathy induced by TCCF.
The present study describes the case of a patient with a history of
traumatic brain injury who presented with delayed myelopathy
consequent to spinal venous hypertension induced by TCCF.
Case report
Patient history and examination
A 45-year-old male was hospitalized at the Neural
Spinal Department of the 904th Hospital of the Chinese People's
Liberation Army Joint Logistic Support (Wuxi, China) with a 1-month
history of progressive gait disturbance in the left lower limb. A
previous history of the patient included an anterior skull base
fracture caused by a traffic accident 2 years prior. A neurological
examination revealed motor weakness (grade IV) in the left lower
limb (6), hyperreflexia, hypesthesia
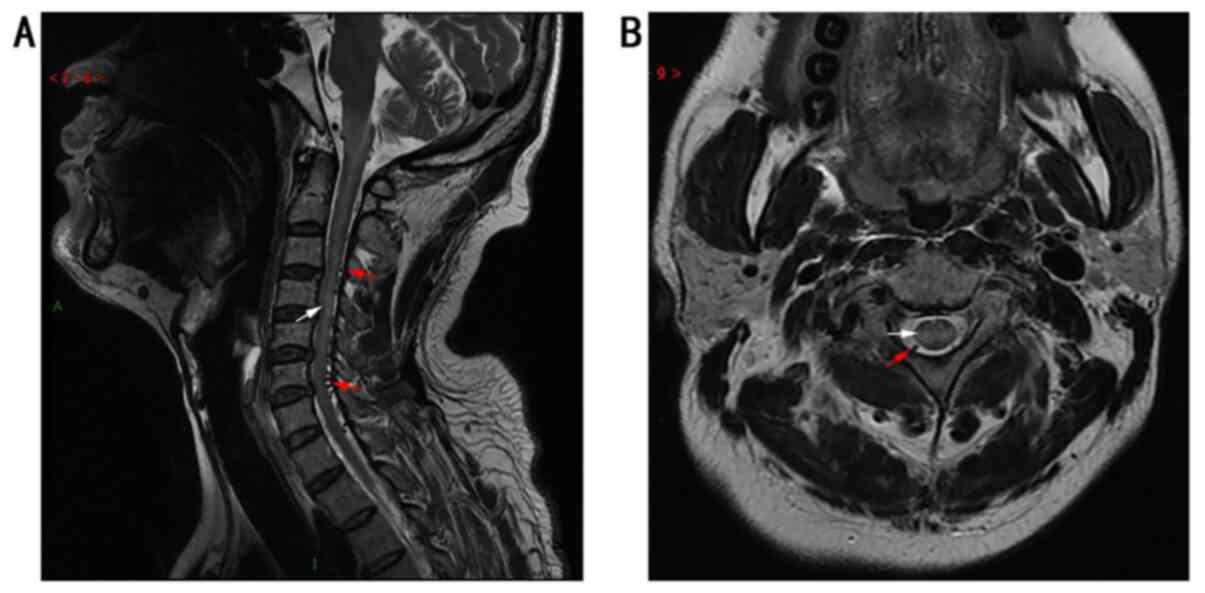
and pulsatile tinnitus. Cervical spinal magnetic resonance imaging
(MRI) revealed spinal cord edema from C1 to C6. In addition,
serpentine signal flow voids located dorsal to the spinal cord from
C1 to T1 were observed (Fig. 1).
Herniated cervical disc compression in the spinal cord was not
observed, and the cerebrospinal fluid signal surrounding the spinal
cord was normal on a T2-weighted MRI. Thus, the diagnosis of
cervical spondylotic myelopathy was excluded. The clinical symptoms
and spinal MRI result raised a high suspicion of spinal
arteriovenous malformation. The patient was transferred to the
Department of Cerebrovascular Disease of the same hospital for
further examination. A spinal digital subtraction angiography (DSA)
was performed for a clear diagnosis; however, the result was
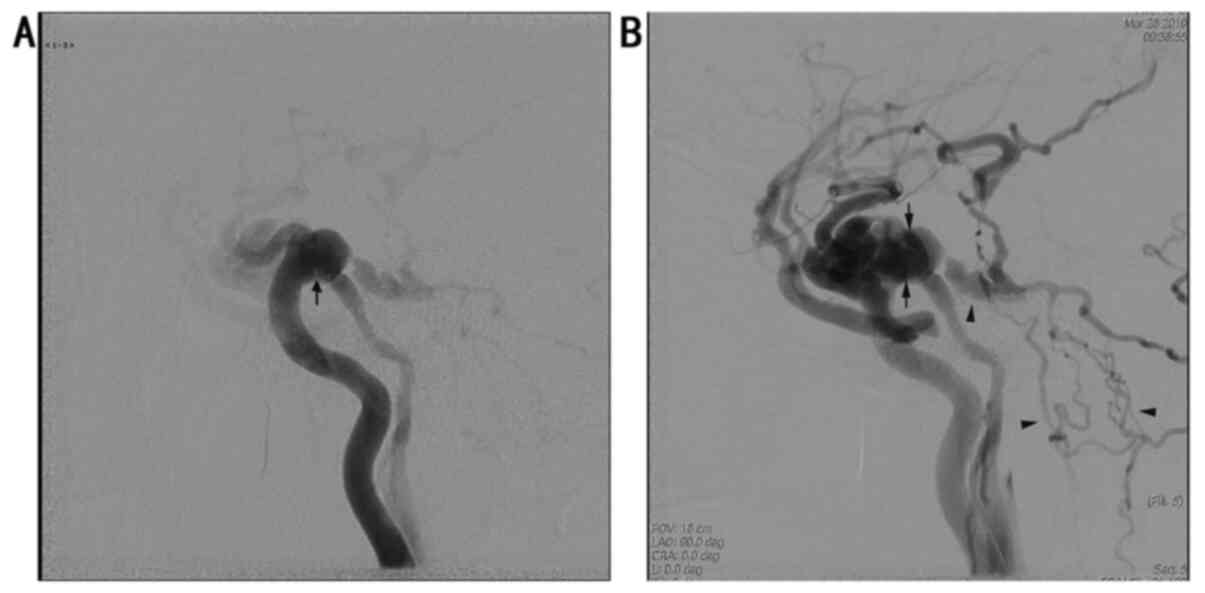
negative. A cerebral DSA was then conducted. A fistula was found
between the posterior genu of the cavernous segment of the left
internal carotid artery (ICA) and cavernous sinus (CS) wall
(Fig. 2A) with venous drainage into
the perimedullary veins via the superior petrosal sinus (SPS)
(Fig. 2B). An angiography of the
left external carotid artery (ECA) did not reveal meningeal
branches, and no filling of the ophthalmic veins was observed.
Thus, the diagnosis of a direct CCF with posterior drainage into
the perimedullary plexus, which resulted in the spinal venous
hypertension and progressive myelopathy of the patient, was
confirmed. In these cases, a possible strategy is to perform a
balloon occlusion test. However, in the case presented herein,
based on the location of the fistula, the pathological
angioarchitecture and the venous drainage pattern, the main aim was
to maintain the patency of the ICA and to occlude the fistula
orifice simultaneously. This would be achieved with the use of
coils and an Onyx liquid embolic system with the assistance of a
protecting balloon, which has a low risk of embolism of the
affected ICA. Thus, a balloon occlusion test was not performed.
Treatment strategy and course
Aspirin enteric-coated tablets (100 mg per day) and
clopidogrel (75 mg per day) were administered for 3 days prior to
treatment. The patient underwent endovascular treatment under
general anesthesia with an arterial approach. Intravenous heparin
saline was continuously used at 1,000 U/h during the treatment.
First, a guiding catheter (8-F) was placed in the petrous section
of the left ICA. A HyperForm occlusion balloon (5x30 mm; Micro
Therapeutics, Inc.) was placed in the ICA adjacent to the fistula
orifice through the guiding catheter. Subsequently, the second
microcatheter was directed into the CS through the fistula orifice
by blood flow. Three coils (one MicroPlex coil, 9x33 cm; two
HydroFrame coils, 9 mm x 31 cm and 8 mm x 33 cm; MicroVention,
Inc.) were placed in the CS to reduce the blood flow from the ICA.
The balloon was then inflated to occlude the fistula orifice to
serve as an abutment for the coils. Finally, 3 ml Onyx liquid
(Onyx™ liquid embolic system; Medtronic; cat. no. 105-7000-060) was
gradually injected into the CS. During this procedure, the Onyx
liquid was strictly prevented from flowing into the SPS and
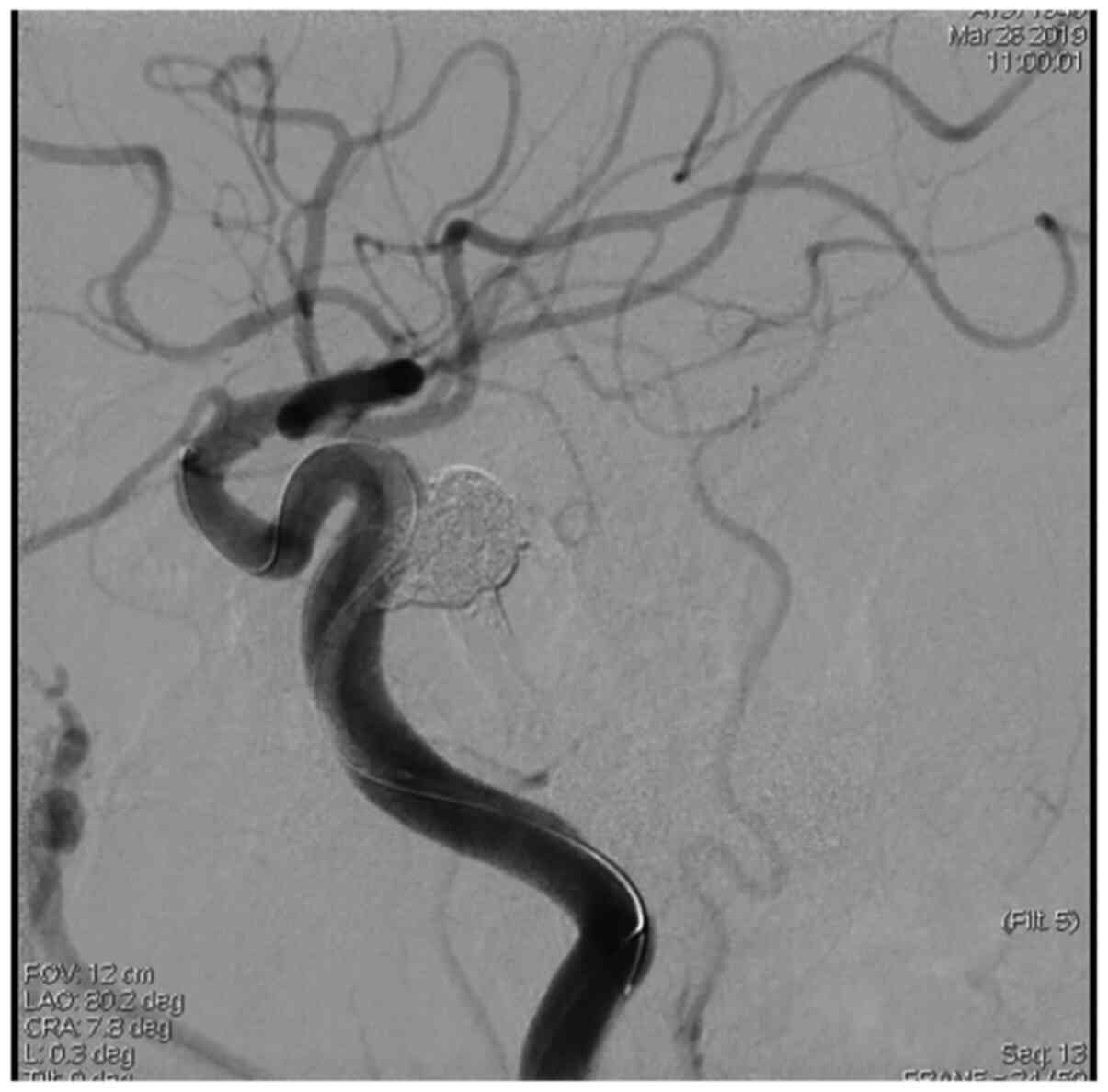
refluxing to the ICA. Subsequent angiography revealed the complete
occlusion of the fistula with no evident venous drainage into
perimedullary veins (Fig. 3). The
patient was prescribed dual antiplatelet therapy post-operatively,
including oral clopidogrel (75 mg/day) for 3 months and aspirin
(100 mg/day) for 6 months.
Post-operative course and
follow-up
The motor power of the left lower limb improved to
grade V when the patient was discharged 3 weeks later. The patient
reported a complete remission of symptoms at the 6-month follow-up.
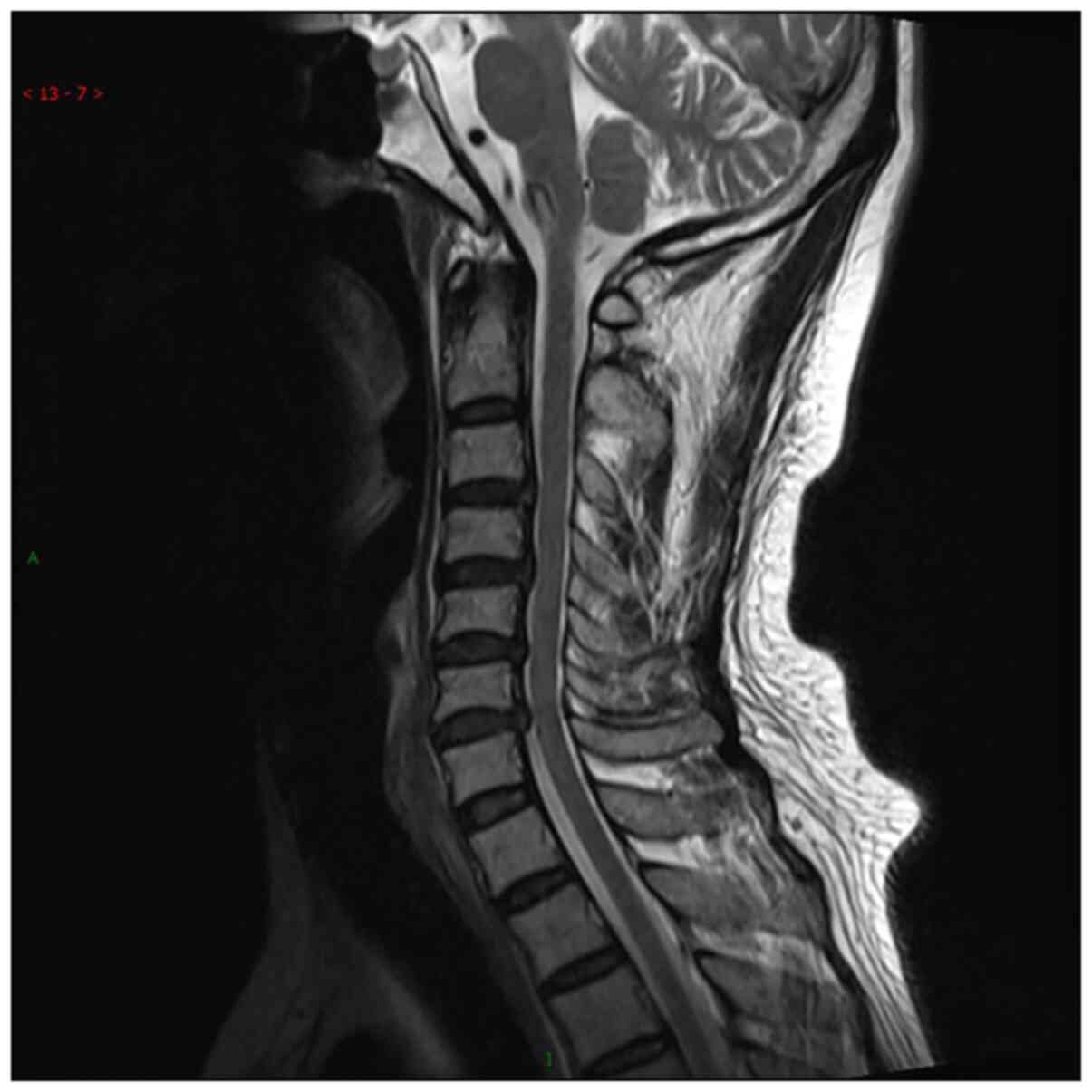
A cervical spinal MRI at the time of follow-up revealed the
disappearance of the perimedullary vein dilation and spinal cord
edema (Fig. 4).
Discussion
CCFs are abnormal communications between the ICA and
the CS and can be divided into two groups as follows: Direct CCFs
(type A, where the pathological shunt involves the cavernous
segment of the ICA and the CS directly) and indirect CCFs (types
B-D, where the pathological shunt involves the meningeal branches
of the ICA or the ECA and the CS) (7). TCCFs are direct lesions often acquired
consequent to brain injury, which account for up to 75% of all
cases (8). The drainage pathways of
CCFs flow anteriorly via the ophthalmic veins, inferiorly via the
inferior petrosal sinus, posteriorly via the deep venous system,
SPS and cerebellar veins; and superiorly via the superficial middle
cerebral vein (9). The clinical
signs and symptoms are related to the drainage pattern of the
fistula and the rapidity of progression.
Inferior and anterior drainages are the most common
routes in all CCFs. Superior and posterior drainages are noted only
in long-standing direct and high-flow fistulas. The majority of
TCCFs drain into the ophthalmic veins, which causes the progressive
congestion of venous sinuses and leads to the sudden onset of
pathognomonic ocular symptoms. The clinical presentation of
progressive myelopathy induced by CCFs is relatively rare. A
similar case presented with myelopathy was prevously reported due
to a dural arteriovenous fistula, which drains into the cervical
perimedullary veins via the petrosal sinus system (10). The patient in the present study had
the rare clinical presentation of myelopathy, and his cerebral DSA
revealed a TCCF with posterior drainage into the perimedullary
veins. To the best of our knowledge, only three cases of CCFs with
progressive myelopathy have been reported to date, among which, two
were spontaneous CCFs (3,4) and one was a TCCF (5).
Posterior drainage may be noted in high-flow and
long-standing direct fistulas. Direct fistulas are usually
considered as high-flow shunts with an acute onset of symptoms
(11). Although TCCFs manifest
symptoms at an early stage following brain injury, the longest
duration acknowledged in a previous study released was 8 weeks
(12). One proper interpretation for
the delayed clinical presentation of TCCFs is that cavernous
sinuses are composed of several small venous compartments (lateral,
medial, antero-inferior and postero-superior compartments), and
these compartments are joined by several anastomoses. Thus, the
development of a low-flow direct CCF without steal phenomena is
highly probable when the fistula orifice is inside the lateral
venous space of the CS (a narrow space with small connections with
the rest of CS) (13). In the case
presented herein, the patient suffered from a direct TCCF and
presented with delayed myelopathy at ~2 years after suffering brain
injury without any steal phenomena. Thus, the assumption was that
the fistula orifice may be located in the lateral venous
compartment, which is separated from the remaining sections of the
CS, and led to a relatively low shunt flow rate. Thus, this type of
vascular malformation may be referred to as a low-flow direct CCF
from the perspective of flow rate, based on the interval between
the initial brain injury and the onset of symptoms.
Patients with progressive myelopathy usually undergo
an MRI to confirm the presence of CCFs. In these cases, a spinal
MRI examination reveals swelling and a pathologically altered
spinal cord signal with contrast enhancement. However, an MRI has a
sensitivity of only 50% (14). Thus,
DSA is the gold standard in the diagnosis and treatment planning of
CCFs. DSA provides a thorough understanding of fistula location,
the pathological angioarchitecture and venous drainage pattern.
Endovascular embolization using embolic agents is the preferred
treatment (15). The goal of
treatment is to occlude the fistula and proximal venous drainage,
reduce venous congestion and prevent the recruitment of collateral
vessels (16). Adjacent vessels can
be recruited and CCFs may recur if residues remain in the fistula
and drainage veins. The diverse manifestations of CCFs may mimic
certain spinal cord pathologies; thus, it is suggested that CCFs as
a possible cause of cervical myelopathy need to be taken into
consideration.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and methods
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
CLD and CLZ designed the study. CLD, CLZ and FH made
substantial contributions to the acquisition and interpretation of
the patient's data. SDX, QWZ, HJW, JJC and JQ were involved in the
acquisition of the patient's data. CLD and CLZ wrote the
manuscript. FH reviewed and edited the manuscript. CLD and CLZ
confirm the authenticity of all the raw data. All authors have read
and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Medical Ethics
Committee of Xishan People's Hospital (Wuxi, China; reference no.
xs2019ky001) and the 904th Hospital of the People's Liberation Army
Joint Logistic Support (Wuxi, China; reference no. 2017-01-02).
Written informed consent was obtained from the participant.
Patient consent for publication
The patient provided consent for the publication of
his data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ringer AJ, Salud L and Tomsick TA: Carotid
cavernous fistulas: Anatomy, classification, and treatment.
Neurosurg Clin N Am. 16:279–295, viii. 2005.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Xu XQ, Liu S, Zu QQ, Zhao LB, Xia JG, Zhou
CG, Zhou WZ and Shi HB: Follow-up of 58 traumatic carotid-cavernous
fistulas after endovascular detachable-balloon embolization at a
single center. J Clin Neurol. 9:83–90. 2013.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Ko SB, Kim CK, Lee SH and Yoon BW: Carotid
cavernous fistula with cervical myelopathy. J Clin Neurosci.
16:1350–1353. 2009.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Narita Y, Watanabe Y, Hoshino T, Okada M,
Yamamoto Y and Kuzuhara S: Myelopathy due to large veins draining
recurrent spontaneous caroticocavernous fistula. Neuroradiology.
34:433–435. 1992.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Herrera DA, Vargas SA and Dublin AB:
Traumatic carotid-cavernous fistula with pontomesencephalic and
cervical cord venous drainage presenting as tetraparesis. J
Neuroimaging. 21:73–75. 2011.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Baschung Pfister P, de Bruin ED, Sterkele
I, Maurer B, de Bie RA, Knols RH and Jan YK: Manual muscle testing
and hand-held dynamometry in people with inflammatory myopathy: An
intra- and interrater reliability and validity study. PLoS One.
13(e0194531)2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Leandro L, Dolci G, Prabhu S and Corkill
R: Bilateral traumatic Caroticocavernous fistulas: A case report
and review of the literature. J Oral Maxillofac Surg. 76:826–830.
2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Docherty G, Eslami M, Jiang K and Barton
JS: Bilateral carotid cavernous sinus fistula: A case report and
review of the literature. J Neurol. 265:453–459. 2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Aralasmak A, Karaali K, Cevikol C, Senol
U, Sindel T, Toprak H, Ozdemir H and Alkan A: Venous drainage
patterns in carotid cavernous fistulas. ISRN Radiol.
2014(760267)2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Abdelsadg M, Kanodia AK, Keston P and
Galea J: Unusual case of intracranial dural AV fistula presenting
with acute myelopathy. BMJ Case Rep.
2016(bcr2016215227)2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Marín-Fernández AB, Cariati P, Román-Ramos
M, Fernandez-Solis J and Martínez-Lara I: Posttraumatic
carotid-cavernous fistula: Pathogenetic mechanisms, diagnostic
management and proper treatment. A case report. J Clin Exp Dent.
8:e226–e229. 2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Nguyen T, Cho YH, Jang YJ, Park MC and
Shin SJ: Long delayed traumatic carotid-cavernous sinus fistula. J
Craniofac Surg. 24:e237–e239. 2013.PubMed/NCBI View Article : Google Scholar
|
|
13
|
D'Angelo L, Paglia F, Caporlingua A,
Sampirisi L, Guidetti G and Santoro A: Atypical manifestation of
direct low-flow carotid-cavernous fistula: case report and review
of the literature. World Neurosurg. 125:456–460. 2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Cohen SD, Goins JL, Butler SG, Morris PP
and Browne JD: Dural arteriovenous fistula: Diagnosis, treatment,
and outcomes. Laryngoscope. 119:293–297. 2009.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Gandhi D, Chen J, Pearl M, Huang J,
Gemmete JJ and Kathuria S: Intracranial dural arteriovenous
fistulas: Classification, imaging findings, and treatment. AJNR Am
J Neuroradiol. 33:1007–1013. 2012.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kim WY, Kim JB, Nam TK, Kim YB and Park
SW: Cervical myelopathy caused by intracranial dural arteriovenous
fistula. Korean J Spine. 13:67–70. 2016.PubMed/NCBI View Article : Google Scholar
|